3D Printing to Support the Shortage in Personal Protective Equipment Caused by COVID-19 Pandemic
Abstract
1. Introduction and Scope
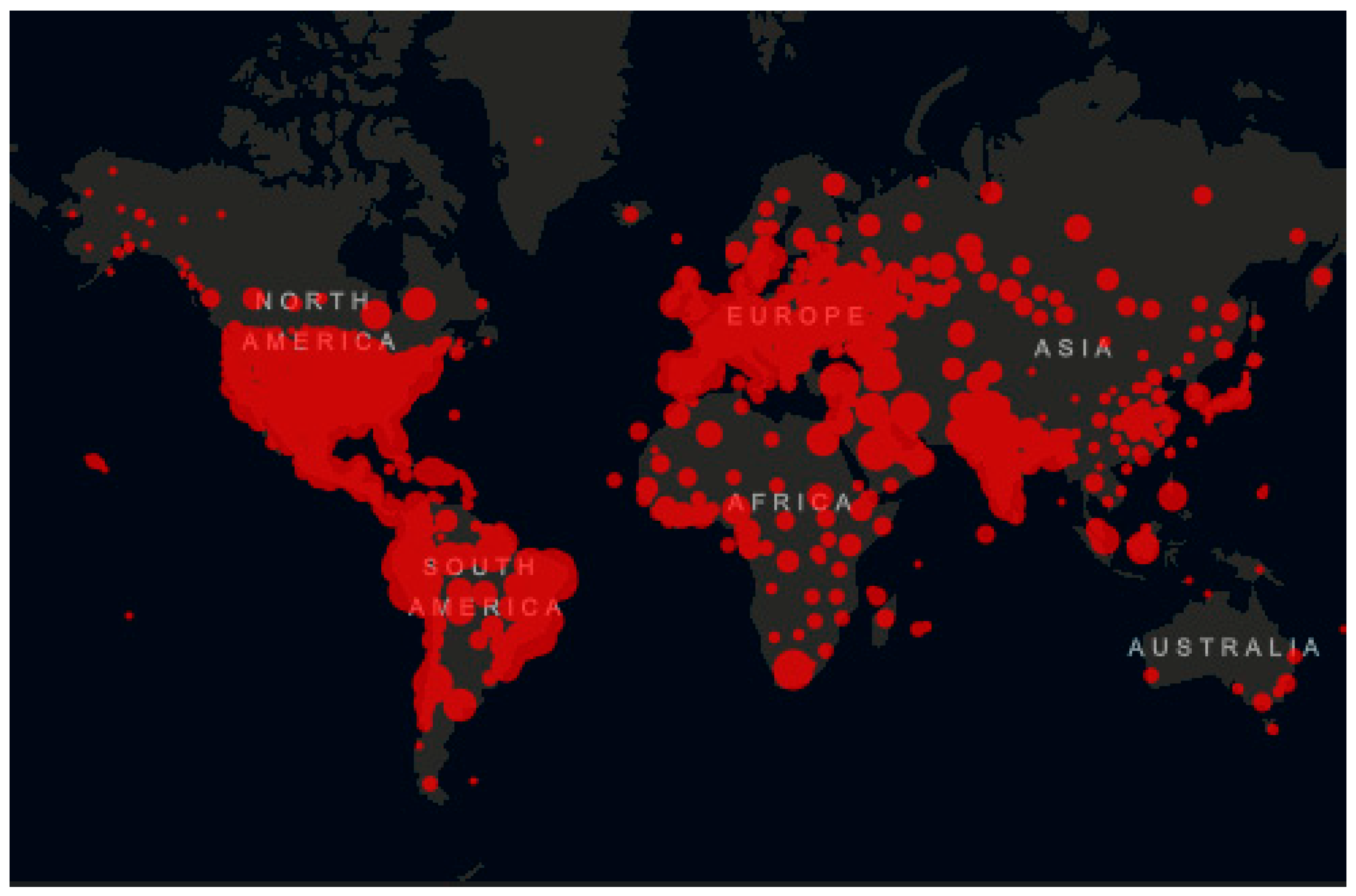
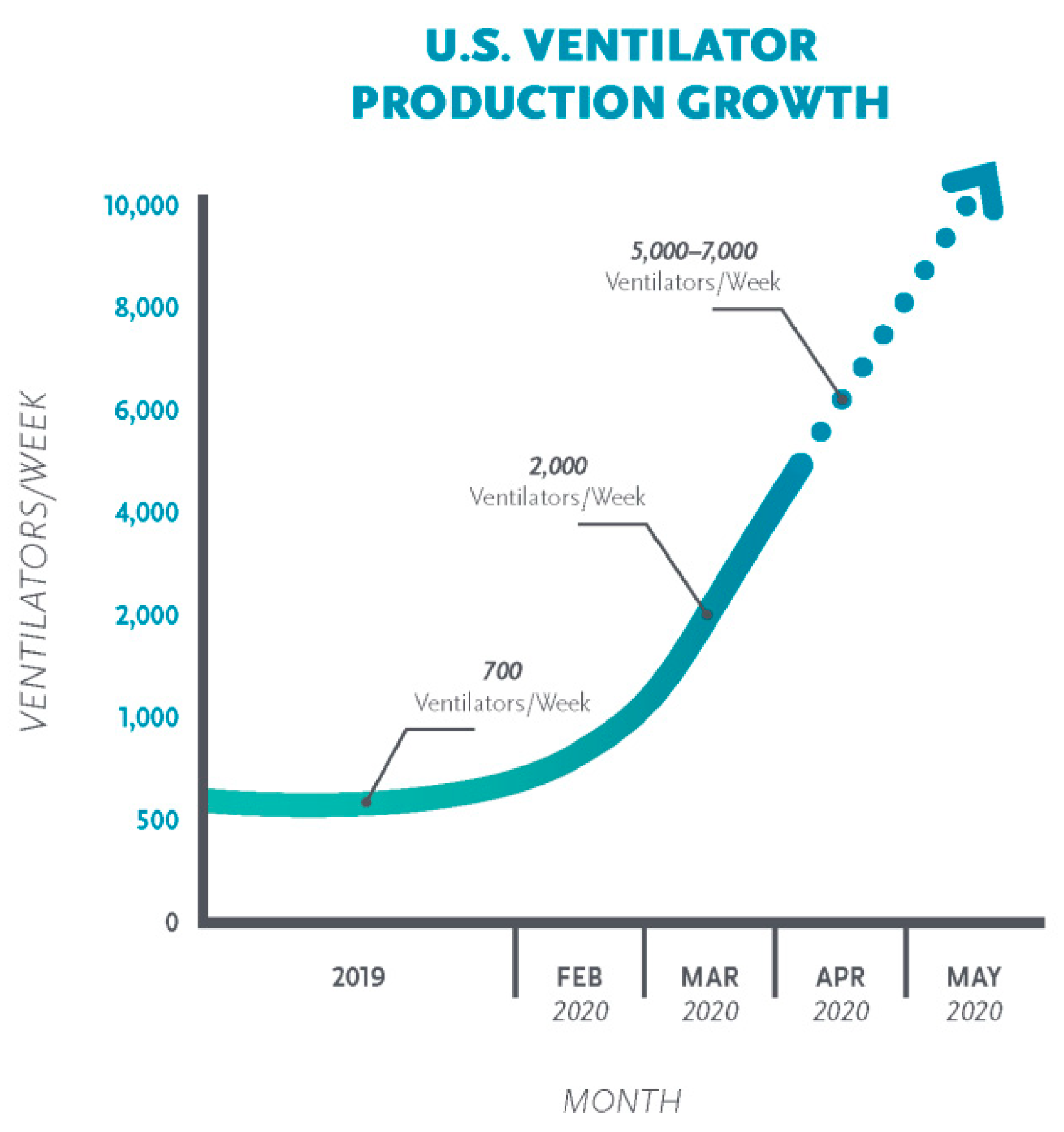
2. Shortage in Medical Supplies Against COVID-19
3. AM Technology Against COVID-19
3.1. An Up-To-Date Review of the AM Technology in Medical Engineering
- The design of new products with the realization of functional prototypes: the bio-compatibility of certain materials enables patient testing and the fabrication of pathological organ prototypes to facilitate preoperative planning and analysis of surgical treatments.
- The production of prostheses and implants: 3D printing offers the possibility to create even the most complex shapes of compatible polymer materials or titanium from scans or MRI scans. AM is ideal for the unit manufacture of complex parts.
- Surgery: The 3D impressions produced are used as templates to prepare for surgery, identify areas of intervention, form any titanium parts, and finally prepare cutting or drilling templates. In addition to the time saved in the operating room (and the savings for the hospital), one can count on a faster recovery of the patient.
- Medical research and education: 3D printing is also used in medical research to visualize and enable organ or concept manipulation.
- Research on direct manufacturing of tissues with complete vital tasks [12,13]: although these applications are still far from widespread clinical implementation due to a number of fundamental method and scientific problems that remain to be solved, major scientific advancement and applications have been conducted in these areas.
- surgical departments and hospitals
- medical and pharmaceutical laboratories
- prosthetists
- universities
- teaching and education
- medical research
3.2. SWOT Analysis of AM in Medical Application
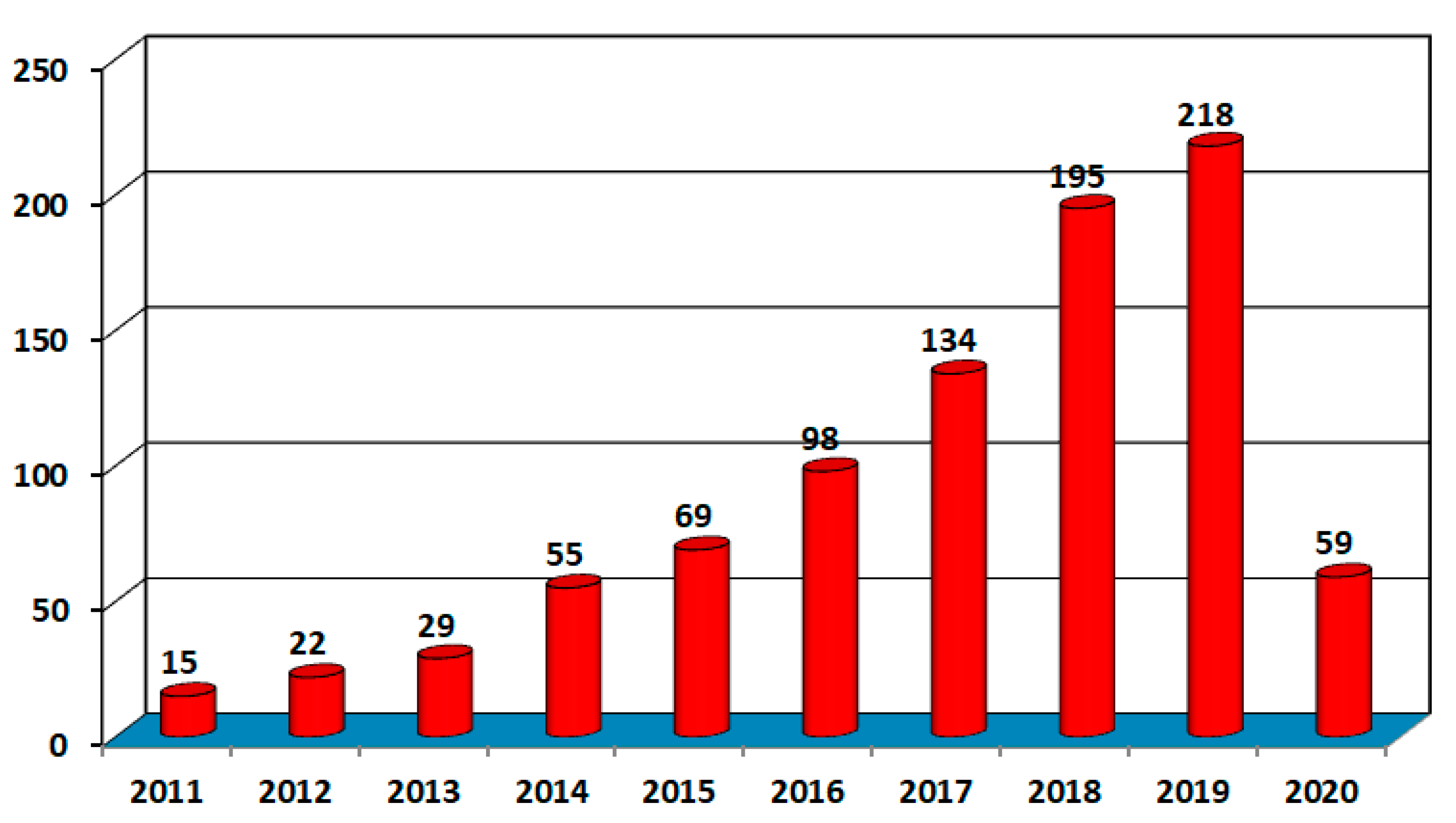
3.3. Implications of the COVID-19 on the Global AM Industry
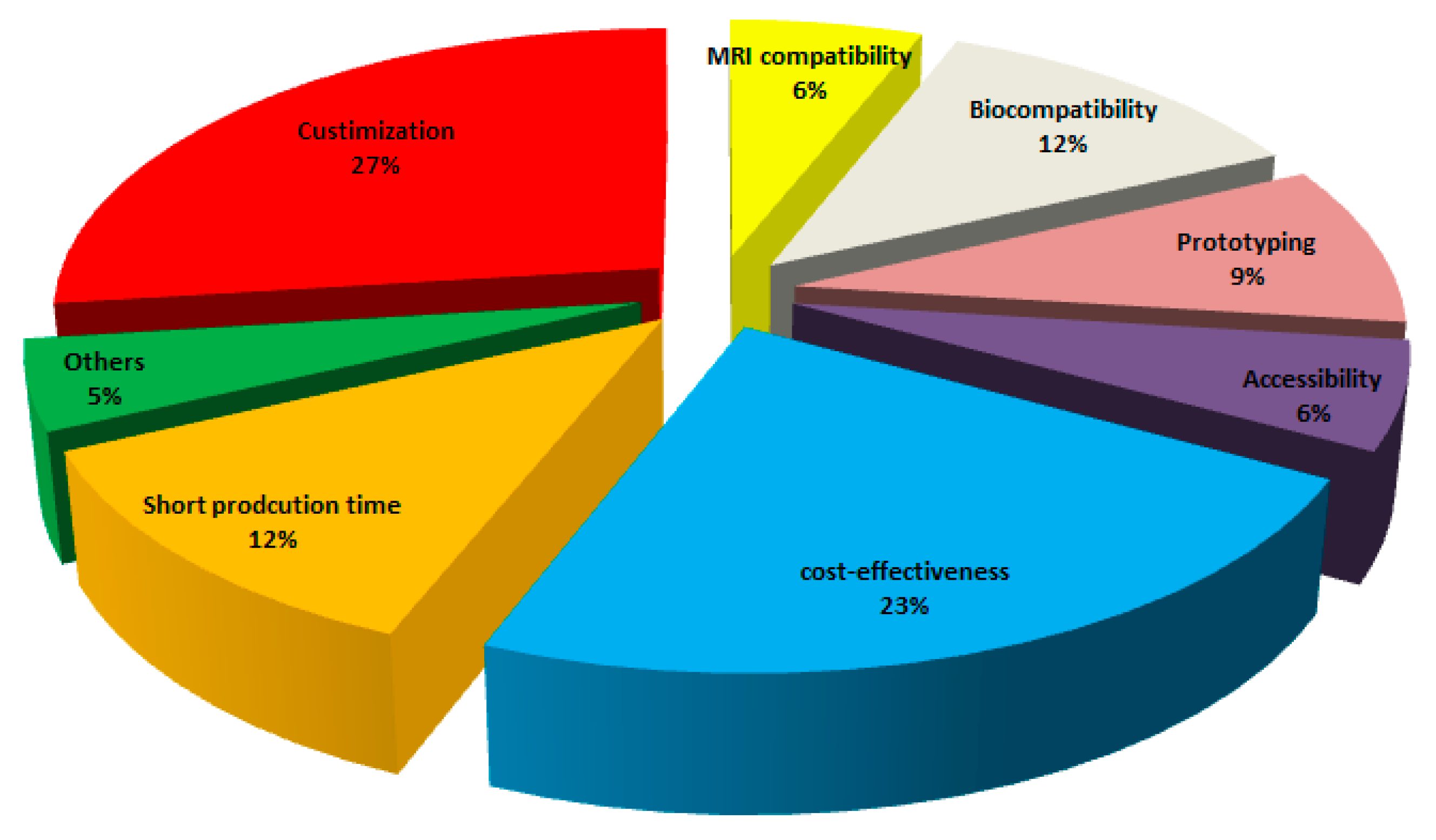
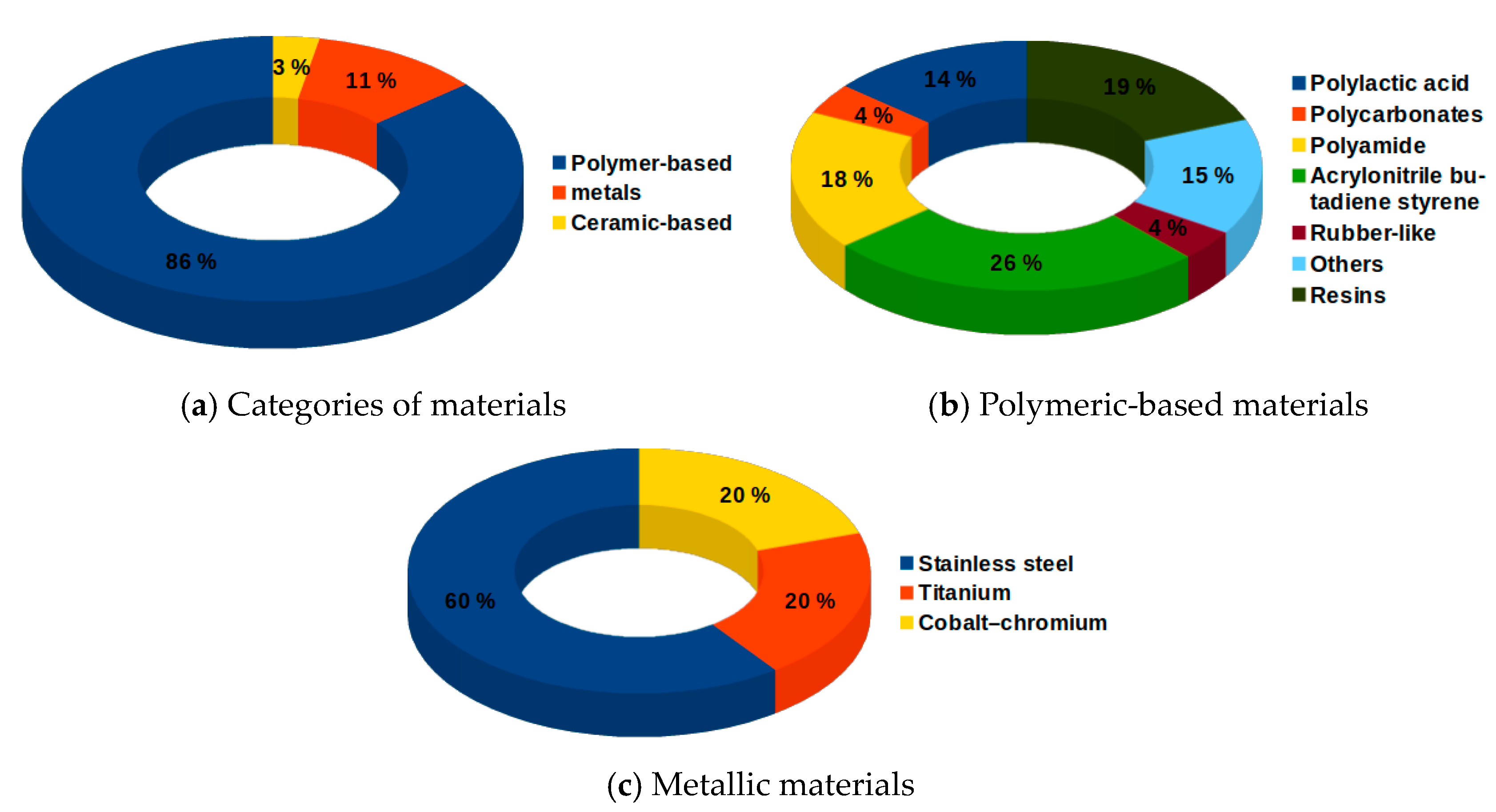
3.4. Research Status on Materials Used for the 3D-Printed Medical Parts
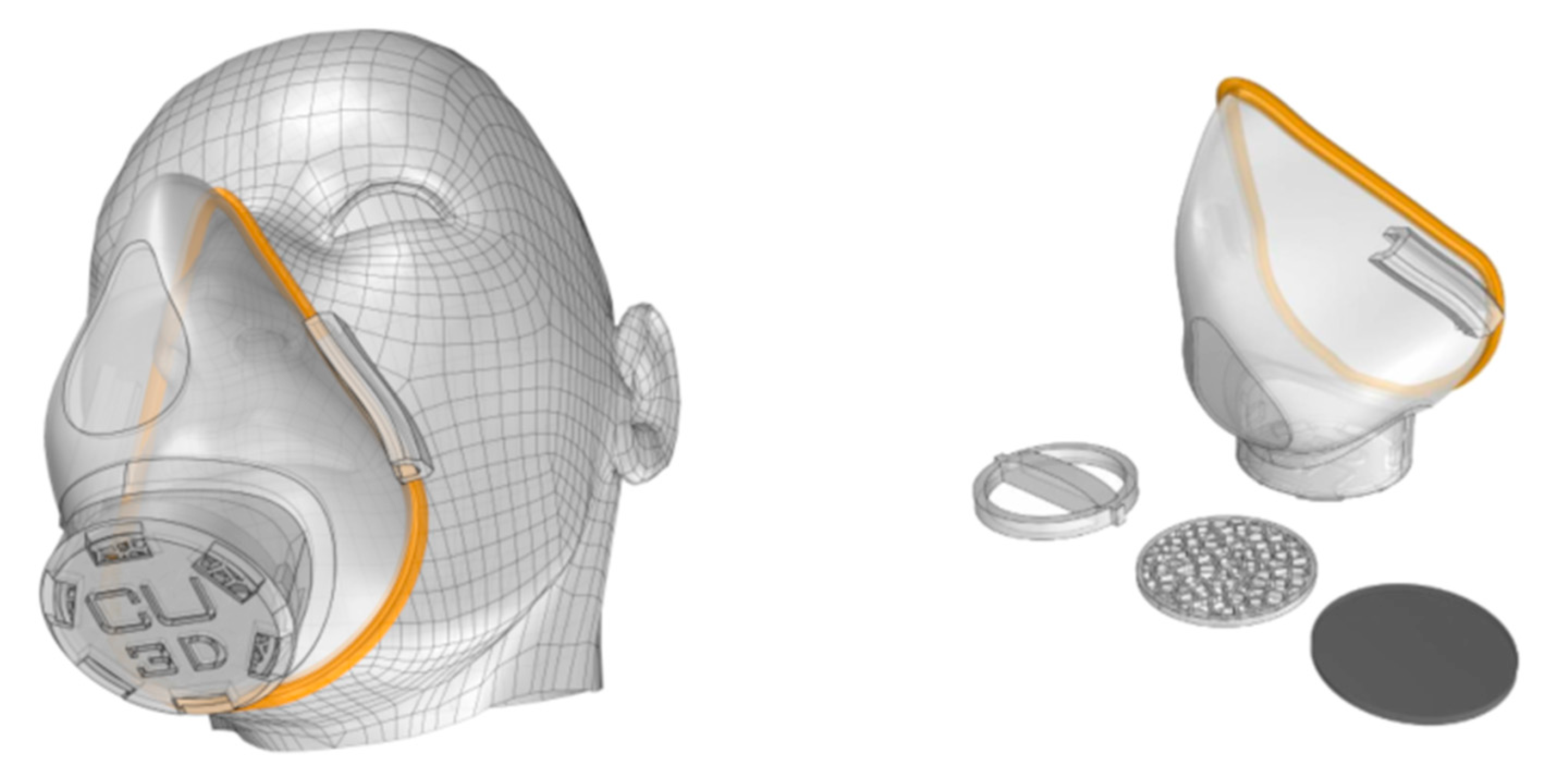
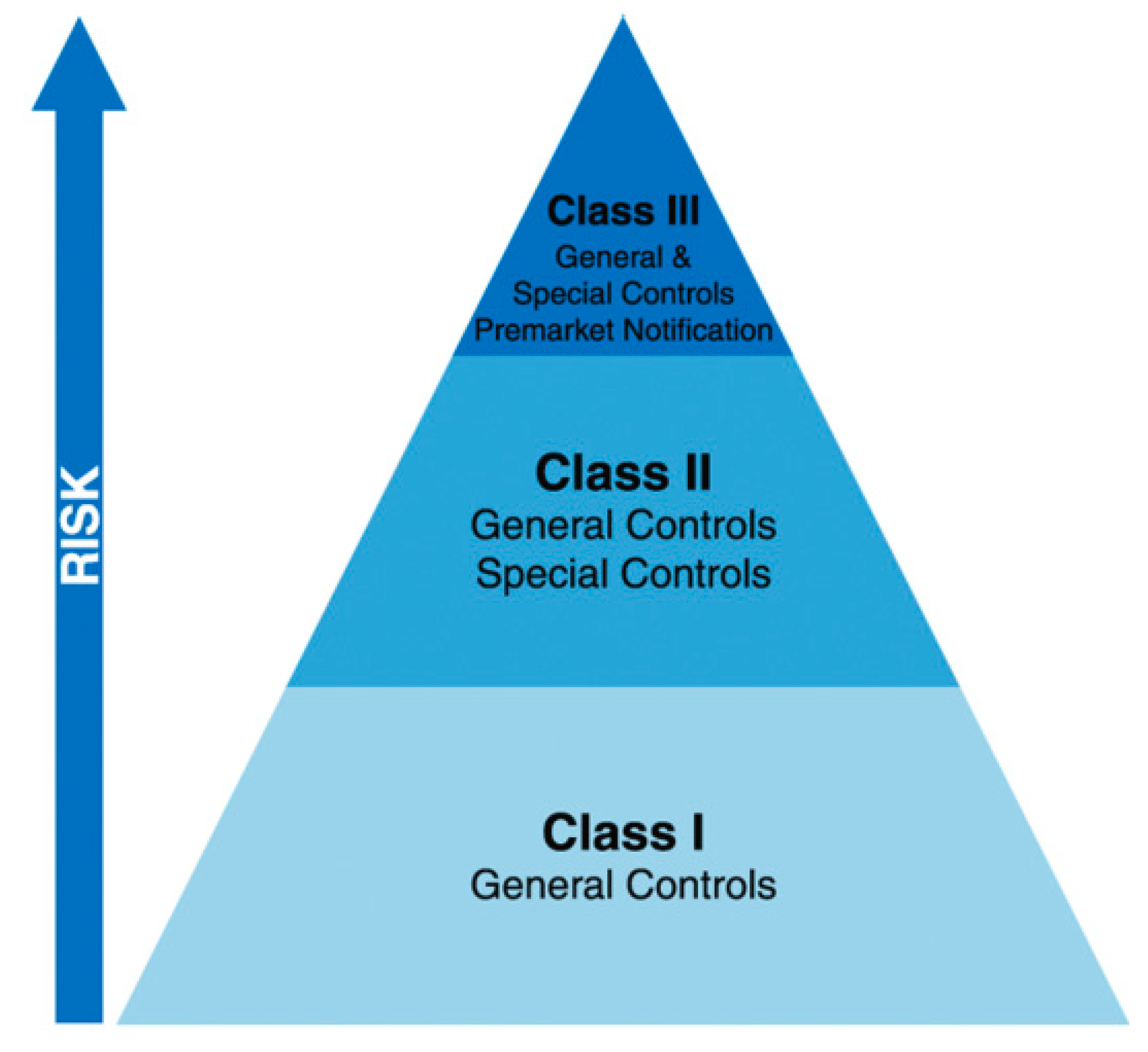
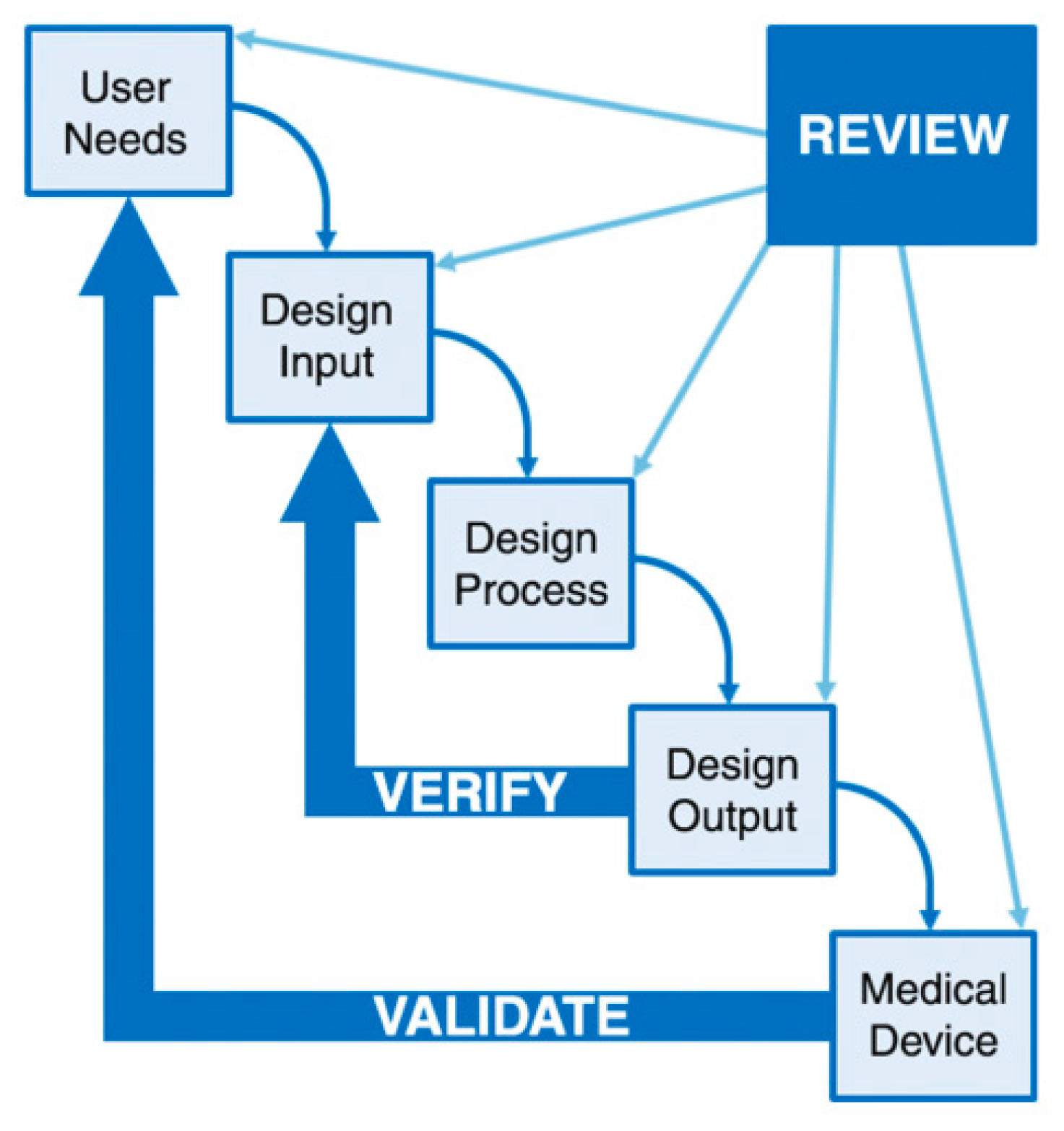
3.5. Technical Considerations for the 3D-Printed Medical Devices
- The device should be washed with soap and water.
- Once washed, it should be rinsed well with clean water.
- Equipment must be disinfected to inactivate any remaining pathogens. For this, chemical disinfection must be used, not using an autoclave, since PLActive (material from which the mask is printed) does not tolerate 80 °C or more.
4. Conclusions and Final Remarks
Funding
Conflicts of Interest
References
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. The Lancet infectious diseases. Lancet Infect Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- World Health Organization. Disease Outbreak News. 2020. Available online: https://www.who.int/csr/don/en/ (accessed on 15 April 2020).
- Ranney, M.L.; Griffeth, V.; Jha, A.K. Critical Supply Shortages—The Need for Ventilators and PPEduring the Covid-19 Pandemic. N. Engl. J. Med. 2020, 382. [Google Scholar] [CrossRef] [PubMed]
- A Better Answer to the Ventilator Shortage as the Pandemic Rages on. Available online: https://www.weforum.org/agenda/2020/04/covid-19-ventilator-shortage-manufacturing-solution/ (accessed on 15 April 2020).
- El Moumen, A.; Tarfaoui, M.; Lafdi, K. AM of polymer composites: Processing and modeling approaches. Compos. Part B Eng. 2019, 171, 166–182. [Google Scholar] [CrossRef]
- El Moumen, A.; Tarfaoui, M.; Lafdi, K. Modelling of the temperature and residual stress fields during 3D printing of polymer composites. Int. J. Adv. Manuf. Technol. 2019, 104, 1661–1676. [Google Scholar] [CrossRef]
- Henhammer, A.; Pammer, S. AM for Medical Applications in TechMatch; TexasInstruments: Dallas, TX, USA, 2017. [Google Scholar]
- Szymczyk, P.; Łabowska, M.B.; Detyna, J.; Michalak, I.; Gruber, P. A review of fabrication polymer scaffolds for biomedical applications using AM techniques. Biocybernetics and Biomedical Engineering. Biocybern. Biomed. Eng. 2020, 40, 624–638. [Google Scholar] [CrossRef]
- 3D Printing: The New Tool Available to Doctors. Available online: https://www.3dnatives.com/en/medical-3d-printing170720184/ (accessed on 18 April 2020).
- Allied Market Research. Available online: https://www.alliedmarketresearch.com/ (accessed on 18 April 2020).
- Velu, R.; Calais, T.; Jayakumar, A.; Raspall, F. A Comprehensive Review on Bio-Nanomaterials for Medical Implants and Feasibility Studies on Fabrication of Such Implants by AMTechnique. Materials 2020, 13, 92. [Google Scholar] [CrossRef] [PubMed]
- Beneke, F.; Metzen, M.; Bergers, D. Study of a Process Chain for the Use of Medical Graphic Data in Technical Applications; Materialise Archives: Leuven, Belgium, 2003. [Google Scholar]
- Bibb, R.; Eggbeer, D.; Evans, P.; Bocca, A.; Sugar, A. Rapid manufacture of custom-fitting surgical guides. Rapid Prototyp. J. 2009, 15, 346–354. [Google Scholar] [CrossRef]
- Mohammed, A.; Elshaer, A.; Sareh, P.; Elsayed, M.; Hassanin, H. AM technologies for drug delivery applications. Int. J. Pharm. 2020, 580, 119245. [Google Scholar] [CrossRef]
- Tzavellas, A.N.; Kenanidis, E.; Potoupnis, M.; Tsiridis, E. 3D printing in orthopedic surgery. In 3D Printing: Applications in Medicine and Surgery; Elsevier: Amsterdam, The Netherlands, 2020; pp. 133–141. [Google Scholar]
- Javaid, M.; Haleem, A. AM applications in medical cases: A literature based review. Alexandria J. Med. 2018, 54, 411–422. [Google Scholar] [CrossRef]
- Zuniga, J.M.; Cortes, A. The role of AM and antimicrobial polymers in the COVID-19 pandemic. Expert Rev. Med. Devices 2020, 17, 477–481. [Google Scholar] [CrossRef]
- Culmone, C.; Smit, G.; Breedveld, P. AM of medical instruments: A state-of-the-art review. Addit. Manuf. 2019, 27, 461–473. [Google Scholar]
- Touri, M.; Kabirian, F.; Saadati, M.; Ramakrishna, S.; Mozafari, M. AM of biomaterials—The evolution of rapid prototyping. Adv. Eng. Mater. 2019, 21, 1800511. [Google Scholar] [CrossRef]
- Cawley, T.; Ross, T. Bone Screw Incorporating a Porous Surface Formed by an Additive Manufacturing Process. U.S. Patent 10,561,456, 18 February 2020. [Google Scholar]
- Backes, E.H.; Pires, L.D.N.; Beatrice, C.A.G.; Costa, L.C.; Passador, F.R.; Pessan, L.A. Fabrication of biocompatible composites of poly (lactic acid)/hydroxyapatite envisioning medical applications. Polym. Eng. Sci. 2020, 60, 636–644. [Google Scholar] [CrossRef]
- Schwarzer, E.; Holtzhausen, S.; Scheithauer, U.; Ortmann, C.; Oberbach, T.; Moritz, T.; Michaelis, A. Process development for additive manufacturing of functionally graded alumina toughened zirconia components intended for medical implant application. J. Eur. Ceram. Soc. 2019, 39, 522–530. [Google Scholar] [CrossRef]
- Sharma, A.; Oh, M.C.; Kim, J.T.; Srivastava, A.K.; Ahn, B. Investigation of electrochemical corrosion behavior of additive manufactured Ti–6Al–4V alloy for medical implants in different electrolytes. J. Alloys Compd. 2020, 830, 154620. [Google Scholar] [CrossRef]
- Yan, Q.; Dong, H.; Su, J.; Han, J.; Song, B.; Wei, Q.; Shi, Y. A review of 3D printing technology for medical applications. Engineering 2018, 4, 729–742. [Google Scholar] [CrossRef]
- Salmi, M.; Paloheimo, K.S.; Tuomi, J.; Wolff, J.; Mäkitie, A. Accuracy of medical models made by AM (rapid manufacturing). J. Cranio-Maxillofac. Surg. 2013, 41, 603–609. [Google Scholar] [CrossRef]
- Mihalko, W.M. AM of arthroplasty implants. In 3D Printing in Orthopaedic Surgery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 49–53. [Google Scholar]
- Giannatsis, J.; Dedoussis, V. Additive fabrication technologies applied to medicine and health care: A review. Int. J. Adv. Manuf. Technol. 2009, 40, 116–127. [Google Scholar] [CrossRef]
- Examplatory Use of 3D Printing to Provide Medical Supplies during Coronavirus (COVID-19) Pandemic in 2020. Available online: https://www.statista.com/statistics/1107198/covid-19-3d-printing-medical-supplies (accessed on 8 May 2020).
- Chae, M.P.; Rozen, W.M.; McMenamin, P.G.; Findlay, M.W.; Spychal, R.T.; Hunter-Smith, D.J. Emerging applications of bedside 3D printing in plastic surgery. Front. Surg. 2015, 2, 1–14. [Google Scholar] [CrossRef]
- Ulmeanu, M.E.; Doicin, C.V.; Baila, D.I.; Rennie, A.E.W.; Neagu, C.; Laha, S. Comparative evaluation of optimum AM technology to fabricate bespoke medical prototypes of composite materials. Mater. Plast. 2015, 52, 416–422. [Google Scholar]
- O’Neill, K.J.; Laina, V.; Wilson, S. Referrals between consultants. The wait to be seen in a specialist plastic surgery hand clinic. Clin. Gov. Int. J. 2008, 13, 261–268. [Google Scholar] [CrossRef]
- Singh, R. Process capability analysis of fused deposition modelling for plastic components. Rapid Prototyp. J. 2014, 20, 69–76. [Google Scholar] [CrossRef]
- Bajpai, P.; Bajpai, M. Development of a high-performance hybrid epoxy silicone resin for coatings. Pigment Resin Technol. 2010, 39, 96–100. [Google Scholar] [CrossRef]
- Haleem, A.; Javaid, M. 3D Scanning Applications in Medical Field: A Literature-Based Review. Clin. Epidemiol. Glob. Health 2018, 7. [Google Scholar] [CrossRef]
- Chohan, J.S.; Singh, R.; Boparai, K.S. Thermal and surface characterization of ABS replicas made by FDM for rapid tooling applications. Rapid Prototyp. J. 2018, 24, 28–36. [Google Scholar] [CrossRef]
- Haleem, A.; Javaid, M.; Vaishya, R. 5D printing and its expected applications in Orthopaedics. J. Clin. Orthop. Trauma 2018. [Google Scholar] [CrossRef]
- Drummer, D.; Cifuentes-Cuéllar, S.; Rietzel, D. Suitability of PLA/TCP for fused deposition modelling. Rapid Prototyp. J. 2012, 18, 500–507. [Google Scholar] [CrossRef]
- Hamad, K.; Kaseem, M.; Yang, H.W.; Deri, F.; Ko, Y.G. Properties and medical applications of polylactic acid: A review. Express Polym. Lett. 2015, 9, 435–455. [Google Scholar] [CrossRef]
- Nachtane, M.; Tarfaoui, M.; Ledoux, Y.; Khammassi, S.; Leneveu, E.; Pelleter, J. Experimental Investigation on the dynamic behavior of 3D printed CF-PEKK composite under cyclic uniaxial Compression. Compos. Struct. 2020, 247, 112474. [Google Scholar] [CrossRef]
- Wang, R.; Shang, J.; Li, X.; Wang, Z.; Luo, Z. Novel topological design of 3D Kagome structure for additive manufacturing. Rapid Prototyp. J. 2018, 24, 261–269. [Google Scholar] [CrossRef]
- Paul, S.C.; van Zijl, G.P.A.G.; Tan, M.J.; Gibson, I. A review of 3D concrete printing systems and materials properties: Current status and future research prospects. Rapid Prototyp. J. 2018, 24, 784–798. [Google Scholar] [CrossRef]
- Jin, Y.; Li, X.; Campbell, R.I.; Ji, S. Visualizing the hotspots and emerging trends of 3D printing through scientometrics. Rapid Prototyp. J. 2018, 24, 801–812. [Google Scholar] [CrossRef]
- Jelínek, F.; Breedveld, P. Design for additive manufacture of fine medical instrumentation dragonflex case study. J. Mech. Des. 2015, 137, 111416. [Google Scholar] [CrossRef]
- Jelínek, F.; Pessers, R.; Breedveld, P. DragonFlex smart steerable laparoscopic instrument. J. Med. Devices 2014, 8, 015001. [Google Scholar] [CrossRef]
- Polonsky, A.T.; Lenthe, W.C.; Echlin, M.P.; Livescu, V.; Gray, G.T., III; Pollock, T.M. Solidification-driven orientation gradients in additively manufactured stainless steel. Acta Mater. 2020, 183, 249–260. [Google Scholar] [CrossRef]
- Zhao, Y.; Koizumi, Y.; Aoyagi, K.; Yamanaka, K.; Chiba, A. Isothermal γ → ε phase transformation behavior in a Co-Cr-Mo alloy depending on thermal history during electron beam powder-bed additive manufacturing. J. Mater. Sci. Technol. 2020, 50, 162–170. [Google Scholar] [CrossRef]
- Zhang, D.; Qiu, D.; Gibson, M.A.; Zheng, Y.; Fraser, H.L.; StJohn, D.H.; Easton, M.A. Additive manufacturing of ultrafine-grained high-strength titanium alloys. Nature 2019, 576, 91–95. [Google Scholar] [CrossRef]
- Karathanasopoulos, N.; Dos Reis, F.; Diamantopoulou, M.; Ganghoffer, J.F. Mechanics of beams made from chiral metamaterials: Tuning deflections through normal-shear strain couplings. Mater. Des. 2020, 189, 108520. [Google Scholar] [CrossRef]
- Tancogne-Dejean, T.; Karathanasopoulos, N.; Mohr, D. Stiffness and strength of hexachiral honeycomb-like metamaterials. J. Appl. Mech. 2019, 86, 111010. [Google Scholar] [CrossRef]
- HackThePandemic. Available online: https://copper3d.com/hackthepandemic/ (accessed on 8 May 2020).
- Swennen, G.R.J.; Pottel, L.; Haers, P.E. Custom-made 3D-printed face masks in case of pandemic crisis situations with a lack of commercially available FFP2/3 masks. Int. J. Oral Maxillofac. Surg. 2019, 49, 673–677. [Google Scholar] [CrossRef]
- FDA Approves First 3D-Printed Mask for COVID-19 Support. Available online: https://www.governmentciomedia.com/fda-approves-first-3d-printed-mask-COVID-19-support (accessed on 10 May 2020).
- 3D-Printed Masks. Available online: https://lowellmakes.com/3d-printed-masks/# (accessed on 8 May 2020).
- Di Prima, M.; Coburn, J.; Hwang, D.; Kelly, J.; Khairuzzaman, A.; Ricles, L. Additively manufactured medical products—The FDA perspective. 3D Print. Med. 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- FDA. Public Workshop—AM of Medical Devices: An Interactive Discussion on the Technical Considerations of 3D Printing; FDA: Rockville, MD, USA, 2014.
- FDA. Technical Considerations for Additive Manufactured Medical Devices-Guidance for Industry and Food and Drug Administration Staff; FDA: Rockville, MD, USA, 2017.
- Fed Regist. Investigational Device Exemptions: Supplemental Applications; 63: 64625. Codified at 21 CFR Sec. 812.35; OFR: Washington, DC, USA, 1998.
- FDA. “Design Control Guidance for Medical Device Manufacturers”, Guidance for Industry and Food and Drug Administration Staff; FDA: Rockville, MD, USA, 1997.
- International Organization for Standardization. Biological Evaluation of Medical Devices; ISO/IEC JTC 1. ISO/IEC 10993:2013; ISO/IEC: Geneva, Switzerland, 2013. [Google Scholar]
- FDA. Use of International Standard ISO-10993, Biological Evaluation of Medical Devices Part 1: Evaluation and Testing. Draft Guidance for Industry and Food and Drug Administration Staff; FDA: Rockville, MD, USA, 2013.
- Stratasys Helps: Responding to the COVID-19 Crisis. Available online: https://www.stratasys.com/COVID-19 (accessed on 10 May 2020).
- Shereen, M.A.; Khan, S.; Kazmi, A.; Bashir, N.; Siddique, R. COVID-19 infection: Origin, transmission, and characteristics of human coronaviruses. J. Adv. Res. 2020, 24, 91–98. [Google Scholar] [CrossRef] [PubMed]
- FDA. Enforcement Policy for Sterilizers, Disinfectant Devices, and Air Purifiers During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency Guidance for Industry and Food and Drug Administration Staff; FDA: Rockville, MD, USA, 2020.






| Authors | Description |
|---|---|
| T Cawley [20] | Use of 3D printing to manufacture a bone screw incorporating a porous surface to improve bony fixation |
| EH Backes [21] | Development of bioactive composites with biodegradable polymers using AM for medical applications |
| Eric Schwarzer [22] | Process development for AM of functionally graded alumina toughened zirconia components intended for medical implant application |
| Ashutosh Sharma [23] | Investigation of electrochemical corrosion behavior of additive manufactured Ti–6Al–4V alloy for medical implants in different electrolytes |
| Method | Drug Formulation |
|---|---|
| Fused Deposition Modeling (FDM) |
|
| Stereolithography (SLA) |
|
| Selective Laser Sintering (SLS) |
|
| Strengths | Weaknesses |
| Time reduction Sustainability Chain efficiency Better control of the construction process Minimizes transportation cost Part consolidation Light weighting parts Complex geometry Inner specifics | Expertise needed Production speed Less awareness Dimensional accuracy Post-processing needed Material limitation Cost of the AM technology printer Low production volume |
| Opportunities | Threats |
| Distributed Manufacturing Multi products batches Manufacturing on demand Potential growth market Design flexibility Allowing customization New product development | Less research collaboration with the industrial sector Reverse engineering Copyright problems/IP issues Regional and country regulations Dangerous weapons and security challenge |
| 3D Printing Companies | Number of Parts Produced |
|---|---|
| Consortium—Formlabs, Carbon, EnvisionTec, and Origin: nasophryngeal swabs (potential weekly capacity) | 4,000,000 |
| Nexa3D (3D printing manufacturer, United States): Test swabs (potential weekly production capacity) | 500,000 |
| Stratasys & Origin (United States): Nasopharyngeal swabs (Planned production capacity per day) | 190,000 |
| Nissan (car manufacturer, Japan): Face shields (potential weekly production capacity) | 100,000 |
| Voodoo Manufacturing (3D printing, United States): Face shields and swabs (weekly capacity for 2500 face shields and 50,000 swabs) | 52,500 |
| Ricoh 3D (Printing, UK): Face shields (weekly capacity) | 40,000 |
| 3D Hubs (3D manufacturing, The Netherlands): Face shields (coordinated effort through the COVID-19 Manufacturing Fund) | 20,000 |
| Forecast 3D (Industrial 3D printing, United States): Face shields, stopgap masks, nasopharyngeal swabs, and other PPE products (daily part production capacity) | 10,000 |
| Nexa3D (3D printing manufacturer, United States): Face shields (potential weekly production capacity) | 10,000 |
| Prusa Research (3D printing company, Czech Republic): Face shields | 10,000 |
| Mobility/Medical goes Additive consortium (around 50 enterprises, Germany): Face shields | 5000 |
| PERA CD- N95 mask lining bracket—Farsoon Technologies (China) -Safety goggle & Mask adjuster. (Large- Safety googles- scale PPE manufacturer, China): (2000 daily) | - |
| Protolabs (3D printing company): Ventilator components | 3000 |
| Fast Radius (Additive manufacturing solutions, United States): Face shield kits (inital shipment, potential daily production capacity of 10,000) | 1500 |
| Azul3D (3D printing manufacturer): Face shields (Current daily capacity; Goal of 20,000 face shields per week) | 1000 |
| SmileDirectClub (Digital dentistry enterprise): Face shields (initial shipment; potential capacity of 7500 per day) | 1000 |
| Photocentric (3D printing company, UK): Valves for respirators (trial run; potential capacity of 40,000 per week) | - |
| Y Soft 3D (Enterprise solutions, Czech Republic): Face shields (daily production capacity) | 500 |
| Weerg & PressUP (Italy): Protective visors | 500 |
| BCN3D (3D printing manufacturer): Face shields (initial shipment with 2000 more planned to ship) | 400 |
| Formlabs (3D printing company, United States): Test swabs (300 in one batch; potential capacity of 75–150 k per day) | 300 |
| Photocentric (Photopolymer manufacturer, UK): Face shield parts (first batch of prints; potential daily capacity for 4860 parts) | 200 |
| Omni3D (Industrial 3D Printing, Poland): Face shields (daily capacity) | 120 |
| Consortium led by Leitat technology center (Zona Franca Consortium, Spain): Pieces for respirators (planned daily production) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tarfaoui, M.; Nachtane, M.; Goda, I.; Qureshi, Y.; Benyahia, H. 3D Printing to Support the Shortage in Personal Protective Equipment Caused by COVID-19 Pandemic. Materials 2020, 13, 3339. https://doi.org/10.3390/ma13153339
Tarfaoui M, Nachtane M, Goda I, Qureshi Y, Benyahia H. 3D Printing to Support the Shortage in Personal Protective Equipment Caused by COVID-19 Pandemic. Materials. 2020; 13(15):3339. https://doi.org/10.3390/ma13153339
Chicago/Turabian StyleTarfaoui, Mostapha, Mourad Nachtane, Ibrahim Goda, Yumna Qureshi, and Hamza Benyahia. 2020. "3D Printing to Support the Shortage in Personal Protective Equipment Caused by COVID-19 Pandemic" Materials 13, no. 15: 3339. https://doi.org/10.3390/ma13153339
APA StyleTarfaoui, M., Nachtane, M., Goda, I., Qureshi, Y., & Benyahia, H. (2020). 3D Printing to Support the Shortage in Personal Protective Equipment Caused by COVID-19 Pandemic. Materials, 13(15), 3339. https://doi.org/10.3390/ma13153339






