Comprehensive Review of Polymer Architecture for All-Solid-State Lithium Rechargeable Batteries
Abstract
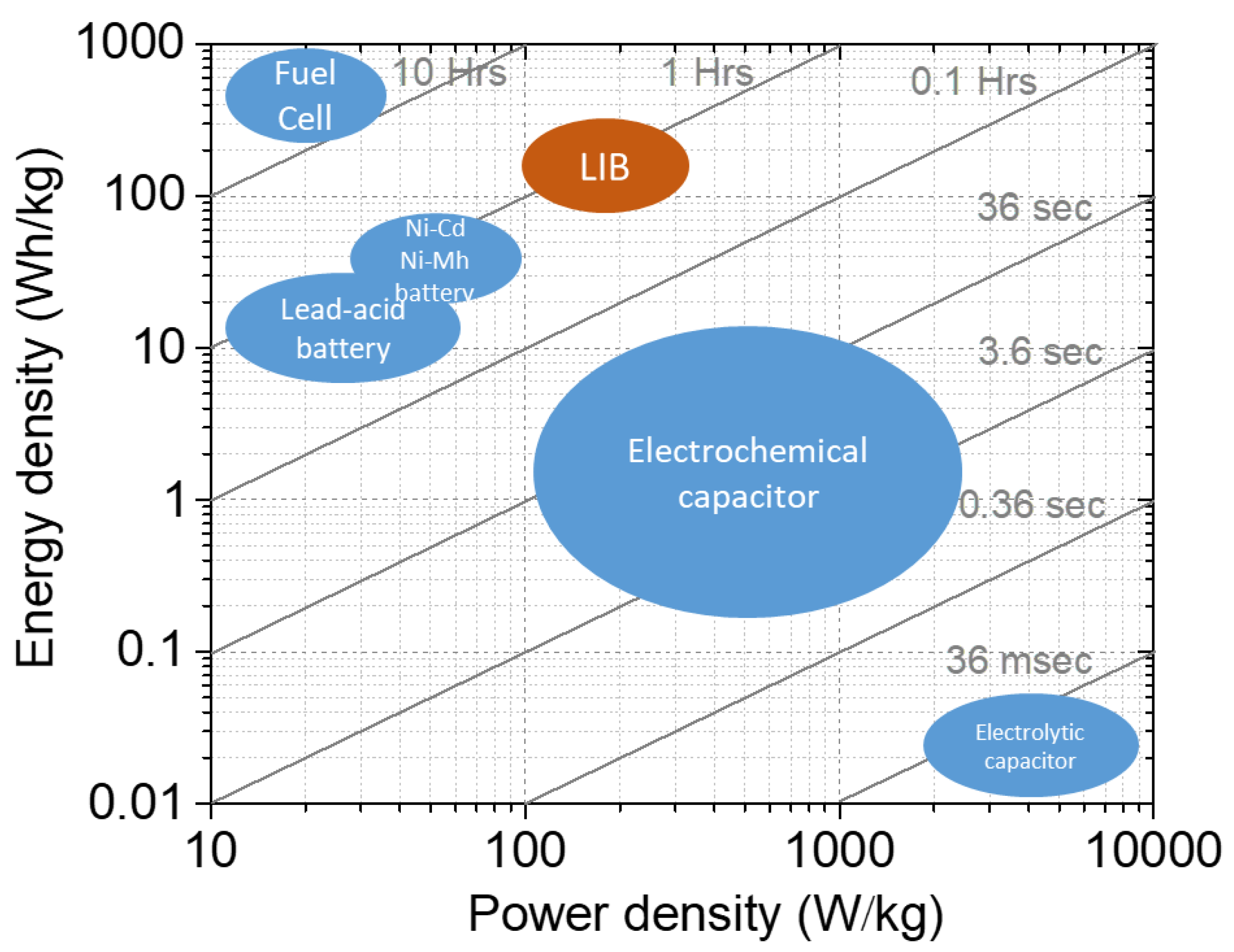
1. Introduction
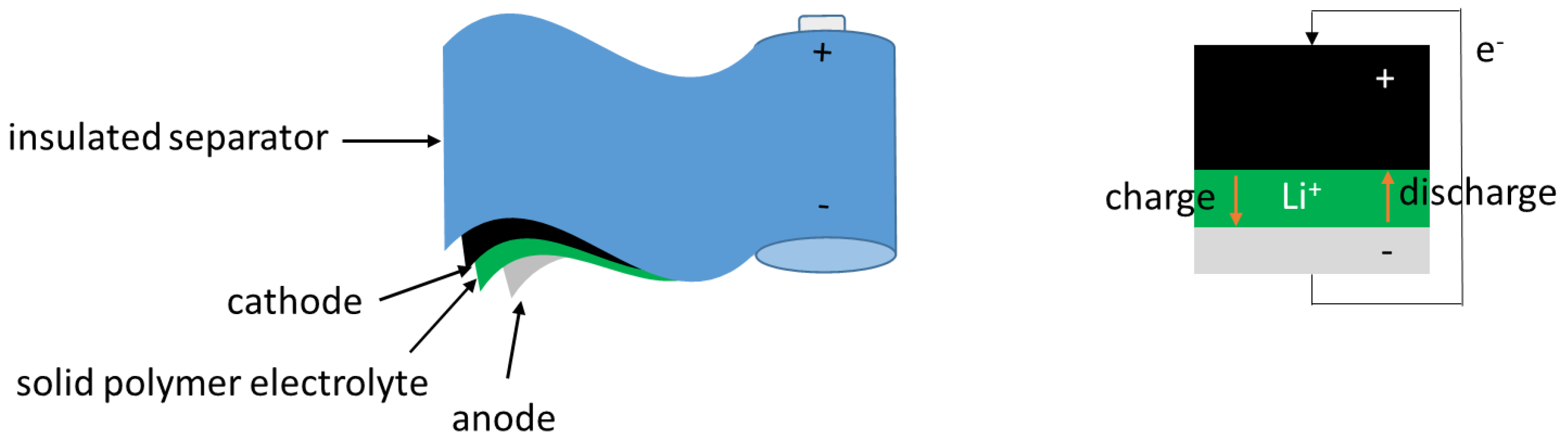
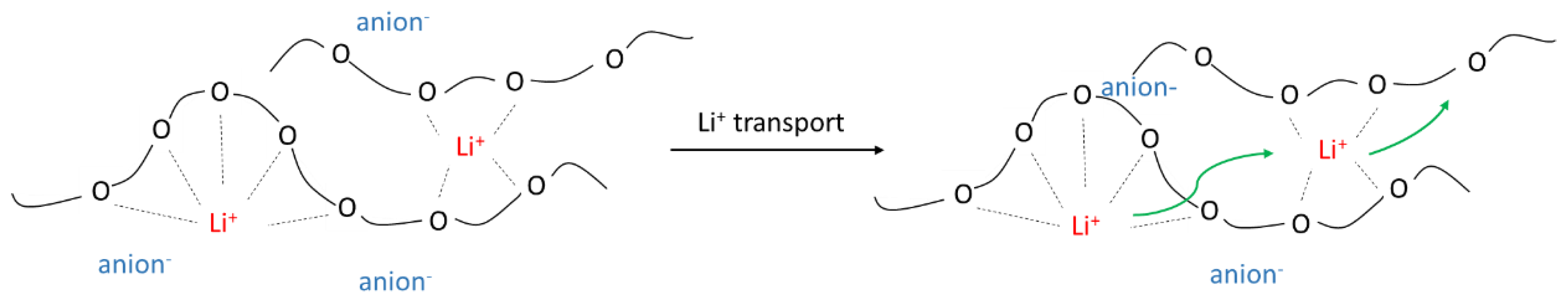
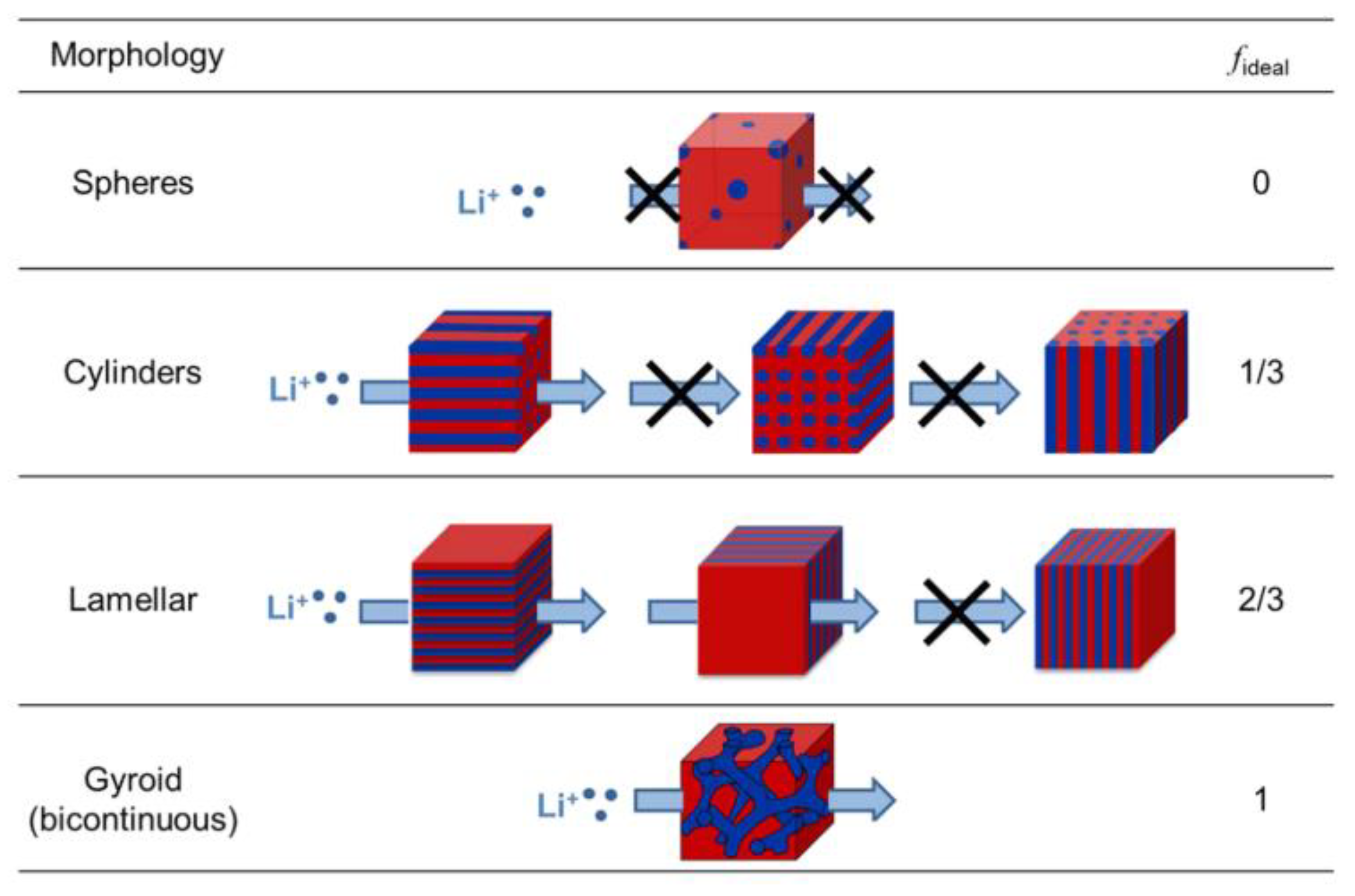
2. Theoretical Background
3. PEO-Based SPEs
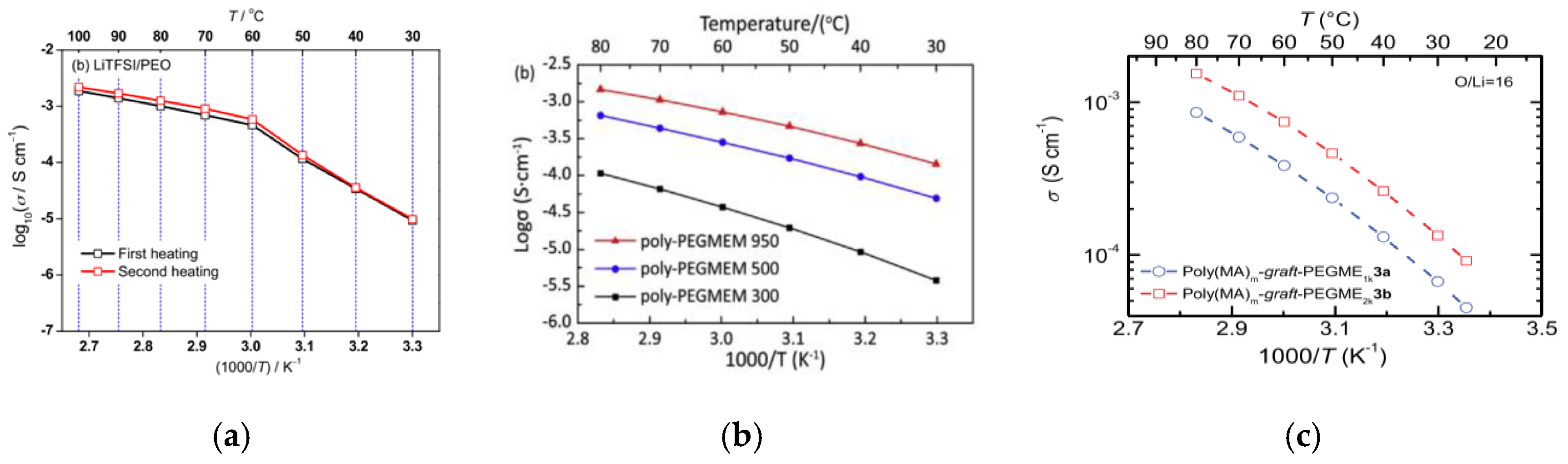
3.1. Linear Polymer
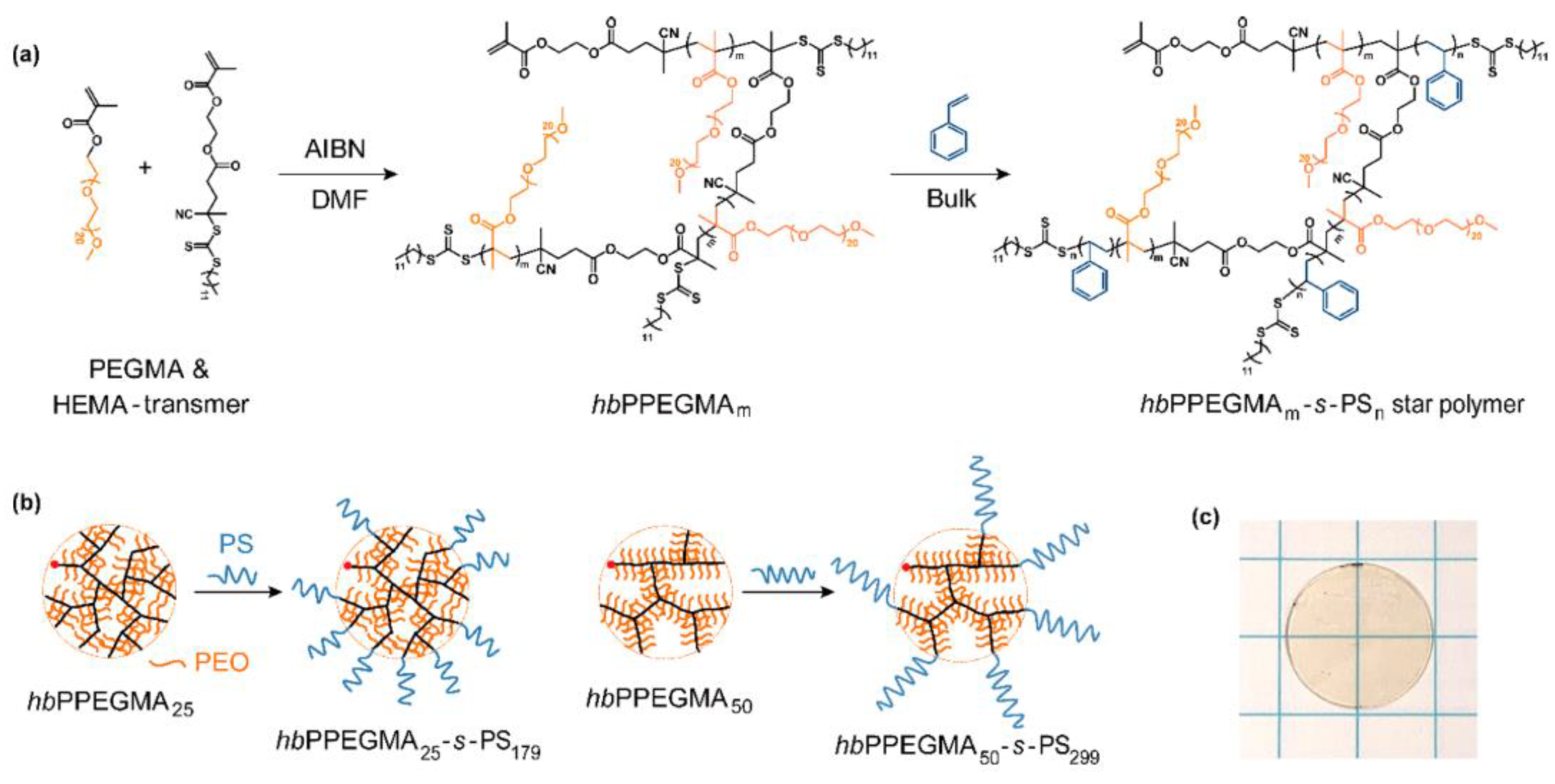
3.2. Comb-Like Polymer
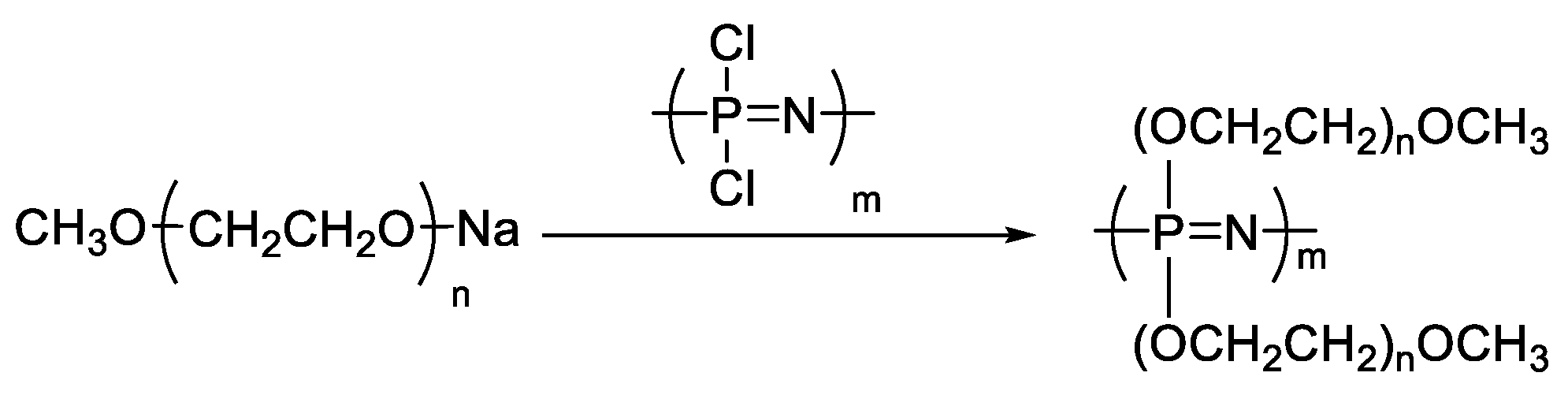
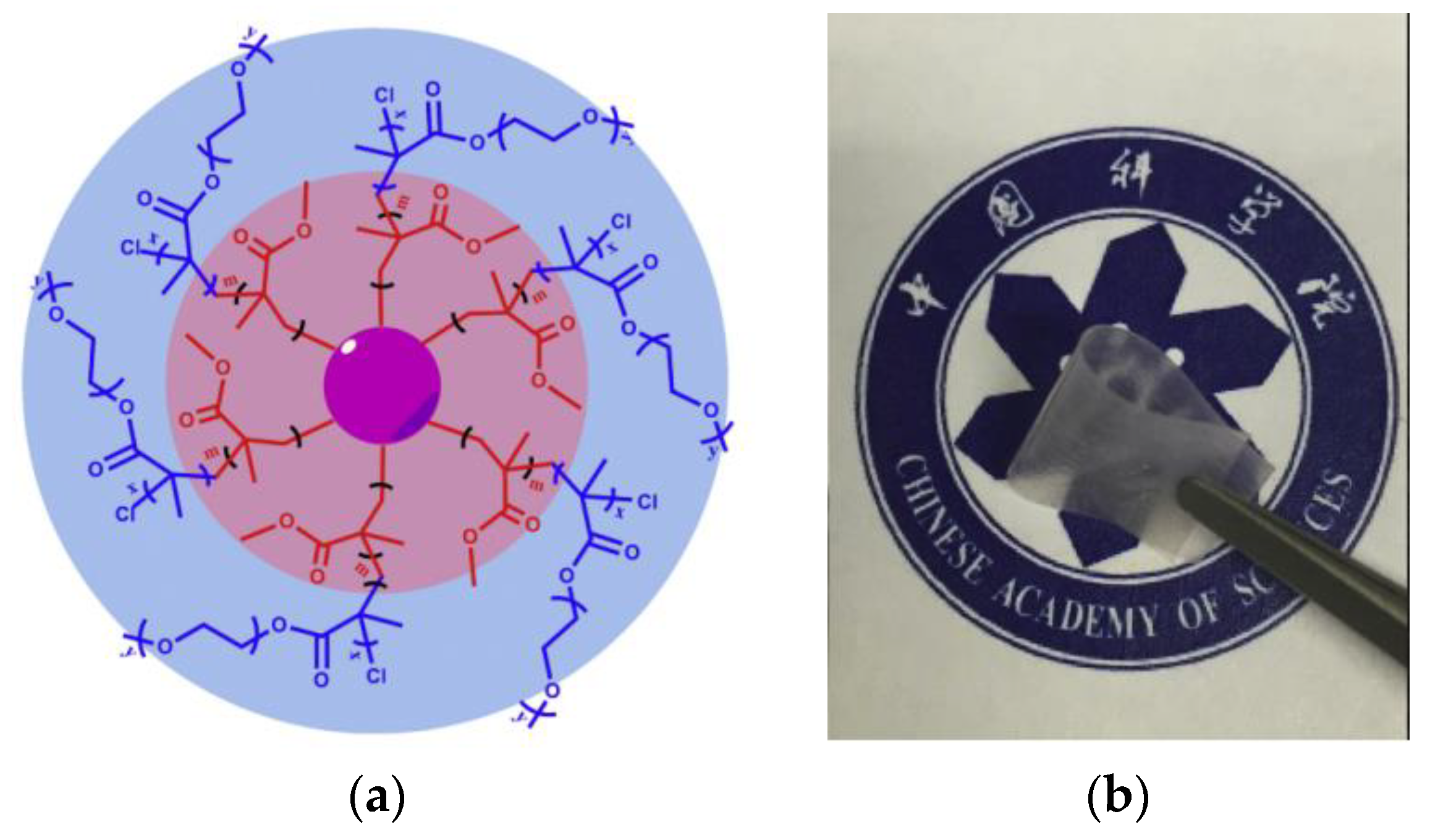
3.3. Hyper-Branched Polymer
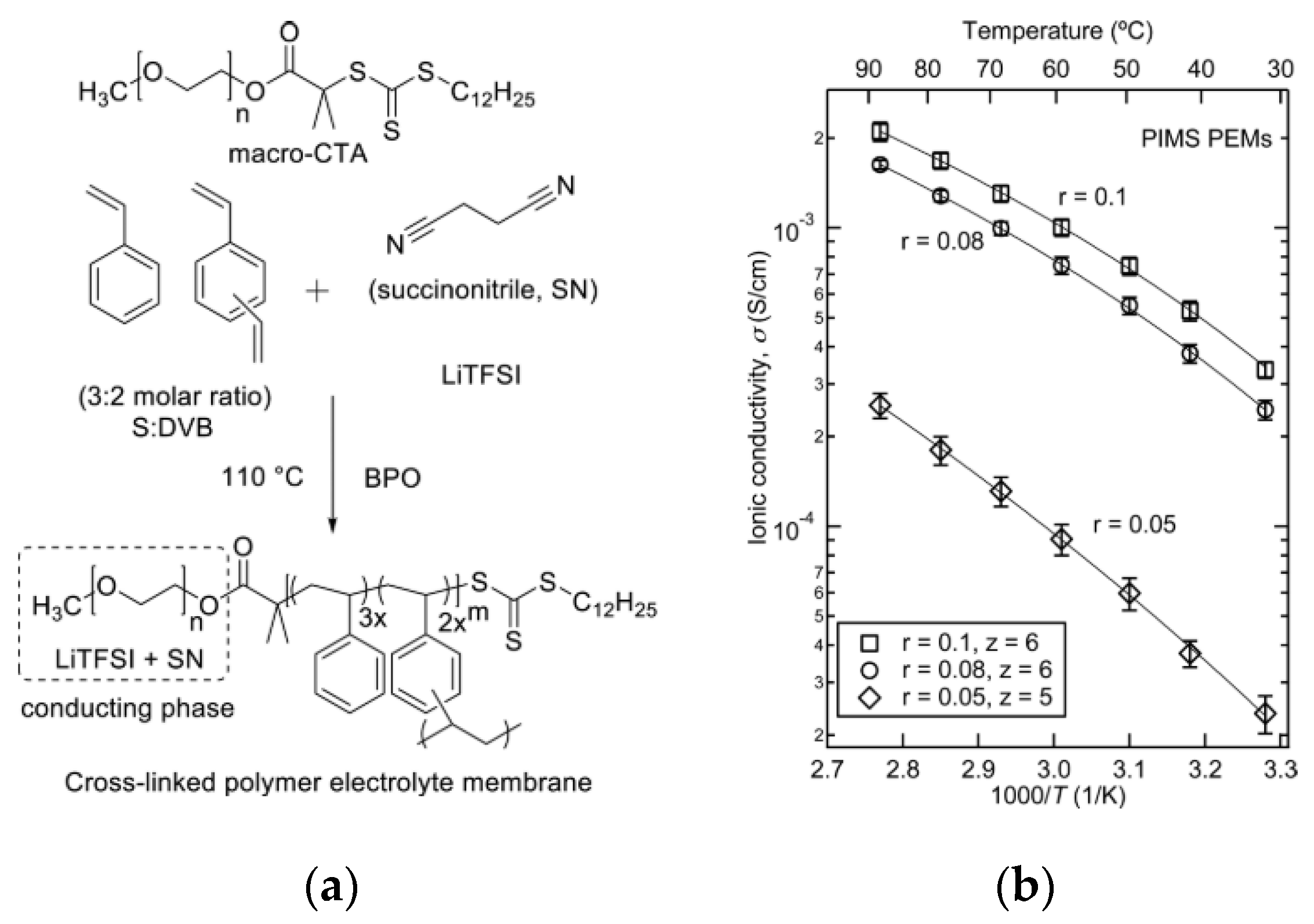
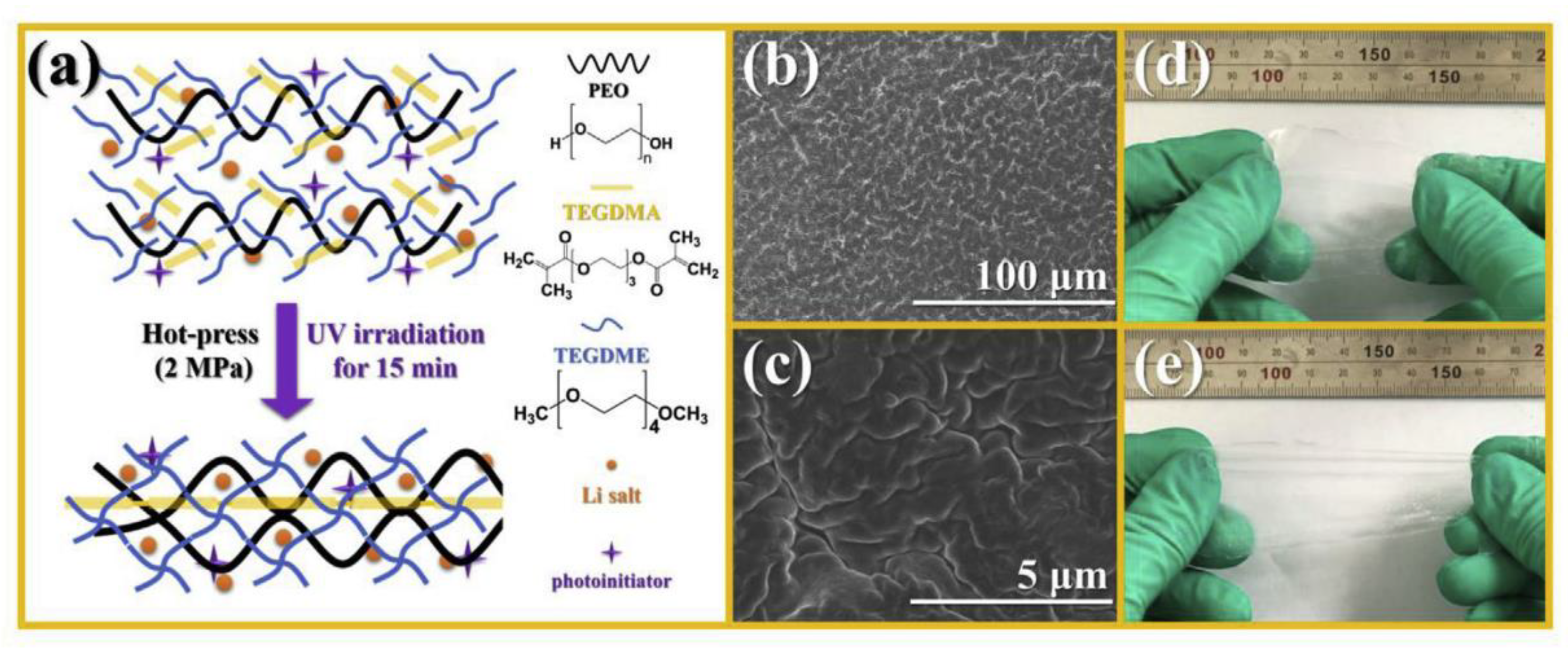
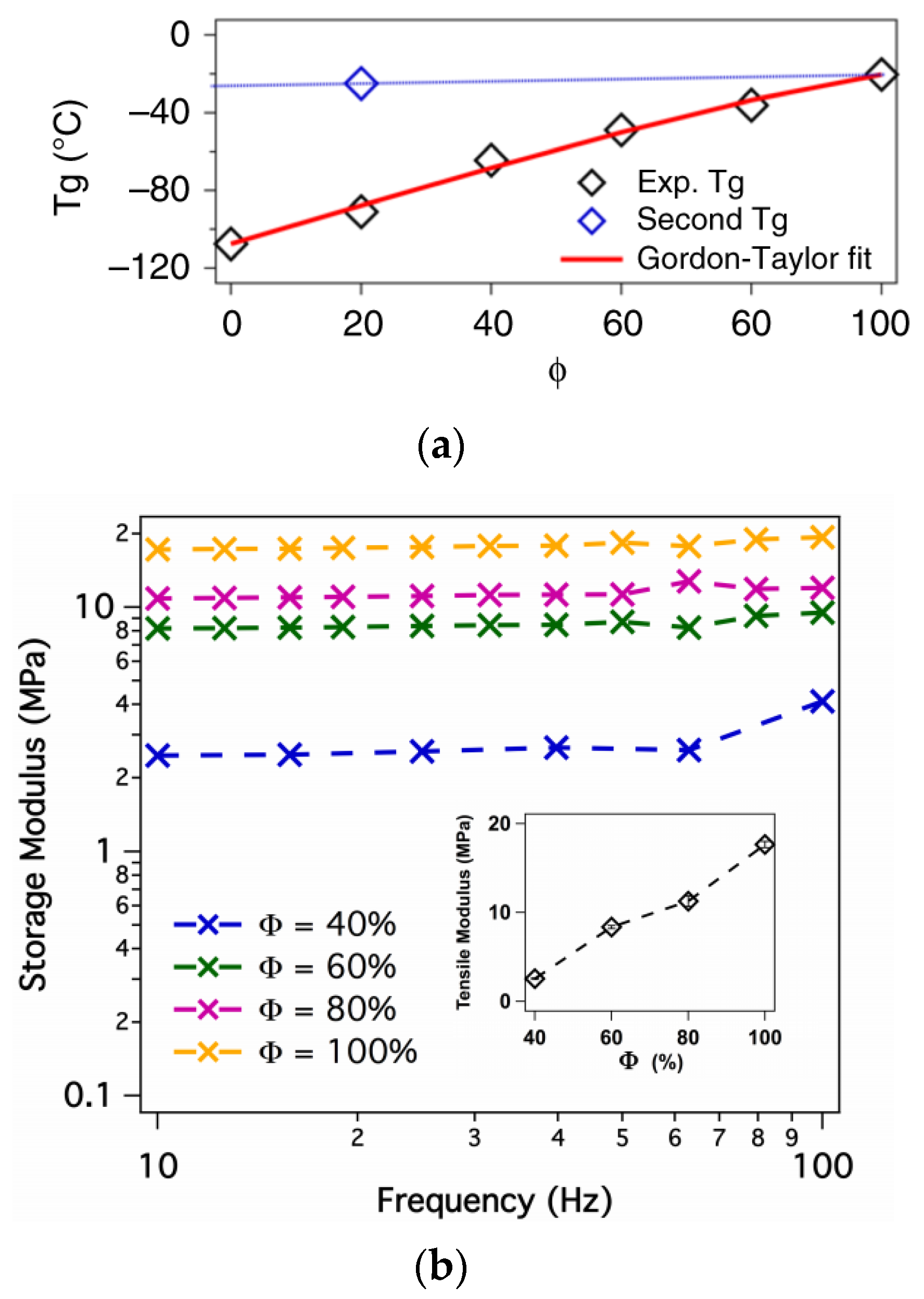
3.4. Crosslinked Polymer
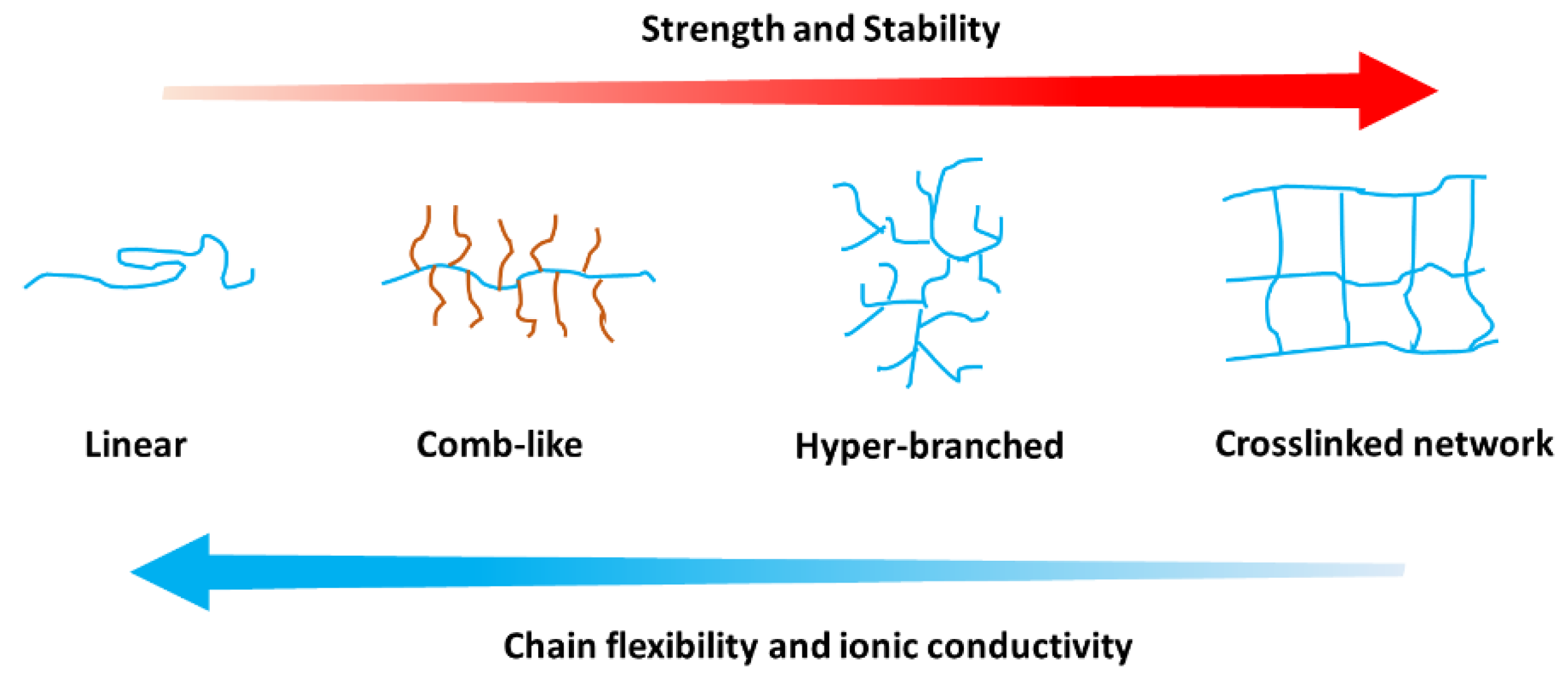
3.5. Discussion
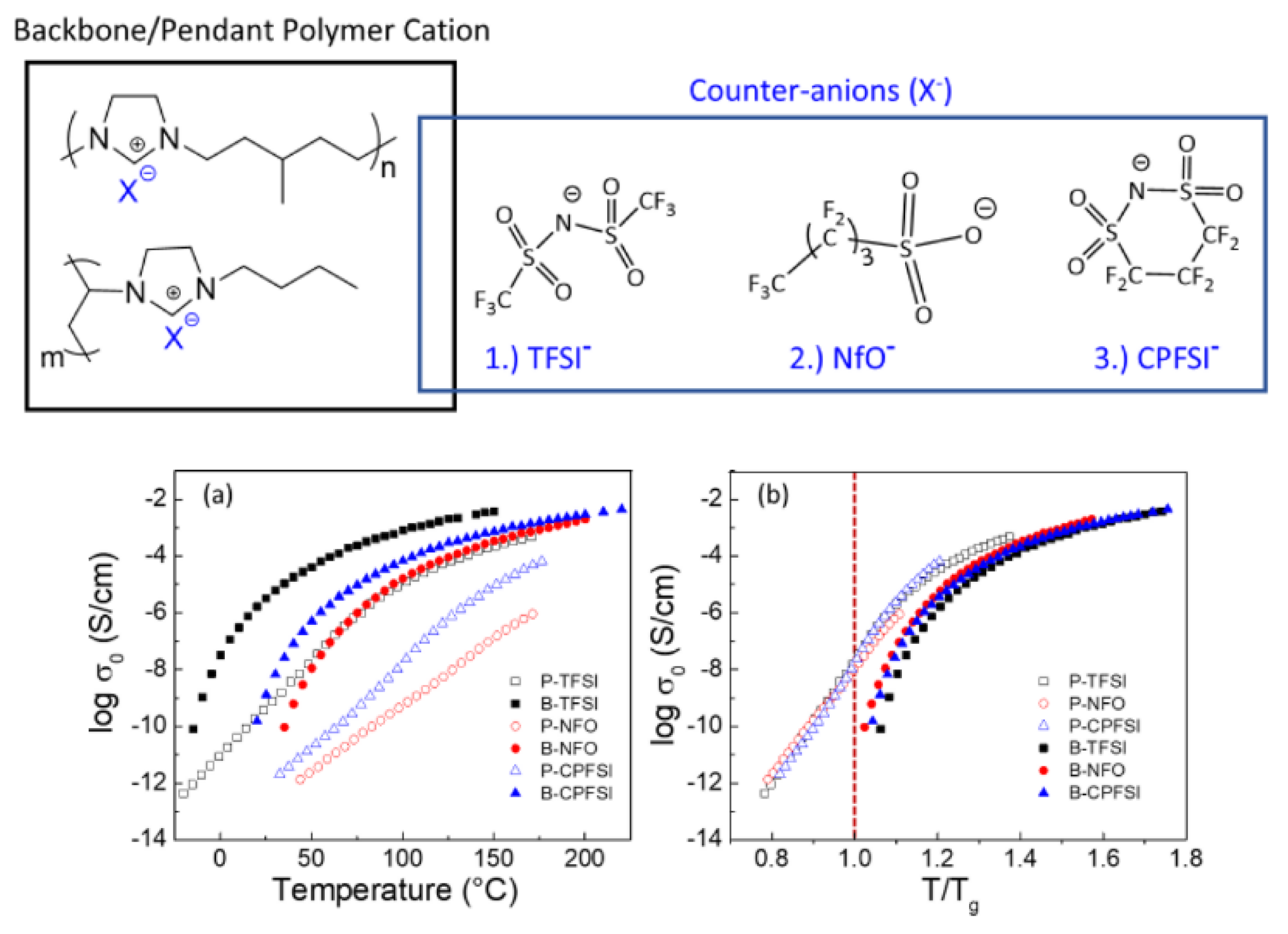
4. PIL-Based SPEs
5. Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Burke, A.; Miller, M. The power capability of ultracapacitors and lithium batteries for electric and hybrid vehicle applications. J. Power Sources 2011, 196, 514–522. [Google Scholar] [CrossRef]
- Cairns, E.J.; Albertus, P. Batteries for electric and hybrid-electric vehicles. Annu. Rev. Chem. Biomol. Eng. 2010, 1, 299–320. [Google Scholar] [CrossRef]
- Frackowiak, E. Carbon materials for supercapacitor application. Phys. Chem. Chem. Phys. 2007, 9, 1774–1785. [Google Scholar] [CrossRef] [PubMed]
- Jeżowski, P.; Crosnier, O.; Deunf, E.; Poizot, P.; Béguin, F.; Brousse, T. Safe and recyclable lithium-ion capacitors using sacrificial organic lithium salt. Nat. Mater. 2018, 17, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Padbury, R.; Zhang, X.W. Lithium–oxygen batteries—Limiting factors that affect performance. J. Power Sources 2011, 196, 4436–4444. [Google Scholar] [CrossRef]
- Marcinek, M.; Syzdek, J.; Marczewski, M.; Piszcz, M.; Niedzicki, L.; Kalita, M.; Plewa-Marczewska, A.; Bitner, A.; Wieczorek, P.; Trzeciak, T.; et al. Electrolytes for Li-ion transport—Review. Solid State Ion. 2015, 276, 107–126. [Google Scholar] [CrossRef]
- Murata, K.; Izuchi, S.; Yoshihisa, Y. An overview of the research and development of solid polymer electrolyte batteries. Electrochim. Acta 2000, 45, 1501–1508. [Google Scholar] [CrossRef]
- Bruce, P.G.; Vincent, C.A. Polymer electrolytes. J. Chem. Soc. Faraday Trans. 1993, 89, 3187–3203. [Google Scholar] [CrossRef]
- Sequeira, C.; Santos, D. Polymer Electrolytes-Fundamentals and Applications; Woodhead Publishing: Sawston, UK, 2010; ISBN 978-1-84569-772-3. [Google Scholar]
- Amine, K. Batteries: Polymers switch for safety. Nat. Energy 2016, 1, 15018. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, L.; Yu, Y.; Sun, J. Progress of enhancing the safety of lithium ion battery from the electrolyte aspect. Nano Energy 2019, 55, 93–114. [Google Scholar] [CrossRef]
- Armand, M.B.; Chabagno, J.M.; Duclot, M. Polymeric solid electrolytes. In Proceedings of the Second International Meeting on Solid Electrolytes, St Andrews, Scotland, 20–22 September 1978. [Google Scholar]
- Gauthier, M.; Fauteux, D.; Vassort, G.; Bélanger, A.; Duval, M.; Ricoux, P.; Chabagno, J.-M.; Muller, D.; Rigaud, P.; Armand, M.B.; et al. Assessment of polymer-electrolyte batteries for EV and ambient temperature applications. J. Electrochem. Soc. 1985, 132, 1333–1340. [Google Scholar] [CrossRef]
- Chatani, Y.; Okamura, S. Crystal structure of poly(ethylene oxide)—Sodium iodide complex. Polymer 1987, 28, 1815. [Google Scholar] [CrossRef]
- Zhang, H.; Li, C.M.; Eshetu, G.G.; Laruelle, S.; Grugeon, S.; Zaghib, K.; Julien, C.; Mauger, A.; Guyomard, D.; Rojo, T.; et al. From solid-solution electrodes and the Rocking-Chair concept to today’s batteries. Angew. Chem. Int. Ed. 2020, 59, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Letourneau, C.; Gauthier, M.; Belanger, A.; Kuller, D.; Hoffman, J. Lithium polymer battery pack design. In Proceedings of the 14th International Electric Vehicle Symposium and Exposition (EVS—14), Orlando, FL, USA, 15–17 December 1997. [Google Scholar]
- Zeng, X.-Q.; Li, M.; Abd El-Hady, D.; Alshitari, W.; Al-Bogami, A.S.; Lu, J.; Amine, K. Commercialization of lithium battery technologies for electric vehicles. Adv. Energy Mater. 2019, 9, 1900161. [Google Scholar] [CrossRef]
- Randau, S.; Weber, D.A.; Kötz, O.; Koerver, R.; Braun, P.; Weber, A.; Ivers-Tiffée, E.; Adermann, T.; Kulisch, J.; Zeier, W.G.; et al. Benchmarking the performance of all-solid-state lithium batteries. Nat. Energy 2020, 5, 259–270. [Google Scholar] [CrossRef]
- History: Over 20 Years of Research and Development to Produce a Unique Technology: LMP®. Available online: https://www.blue-solutions.com/en/blue-solutions/company/presentation/histoire/ (accessed on 28 May 2020).
- Springer, T.E.; Zawodzinski, T.A.; Gottesfeld, S. Polymer electrolyte fuel cell model. J. Electrochem. Soc. 1991, 138, 2334–2342. [Google Scholar] [CrossRef]
- Su’ait, M.S.; Rahman, M.Y.A.; Ahmad, A. Review on polymer electrolyte in dye-sensitized solar cells. Sol. Energy 2015, 115, 452–470. [Google Scholar] [CrossRef]
- Alexandre, S.A.; Silva, G.G.; Santamaría, R.; Trigueiro, J.P.C.; Lavall, R.L. A highly adhesive PIL/IL gel polymer electrolyte for use in flexible solid state supercapacitors. Electrochim. Acta 2019, 299, 789–799. [Google Scholar] [CrossRef]
- Xue, Z.; He, D.; Xie, X. Poly(ethylene oxide)-based electrolytes for lithium-ion batteries. J. Mater. Chem. A 2015, 3, 19218–19253. [Google Scholar] [CrossRef]
- Arya, A.; Sharma, A.L. Insights into the use of polyethylene oxide in energy storage/conversion devices: A critical review. J. Phys. D Appl. Phys. 2017, 50, 443002. [Google Scholar] [CrossRef]
- Cheng, S.; Smith, D.M.; Pan, Q.; Wang, S.; Li, C.Y. Anisotropic ion transport in nanostructured solid polymer electrolytes. RSC Adv. 2015, 5, 48793–48810. [Google Scholar] [CrossRef]
- Watanabe, M.; Thomas, M.L.; Zhang, S.; Ueno, K.; Yasuda, T.; Dokko, K. Application of ionic liquids to energy storage and conversion materials and devices. Chem. Rev. 2017, 117, 7190–7239. [Google Scholar] [PubMed]
- Eshetu, G.G.; Mecerreyes, D.; Forsyth, M.; Zhang, H.; Armand, M. Polymeric ionic liquids for lithium-based rechargeable batteries. Mol. Syst. Des. Eng. 2019, 4, 294–309. [Google Scholar] [CrossRef]
- Keller, M.; Varzi, A.; Passerini, S. Hybrid electrolytes for lithium metal batteries. J. Power Sources 2018, 392, 206–225. [Google Scholar] [CrossRef]
- Dirican, M.; Yan, C.; Zhu, P.; Zhang, X. Composite solid electrolytes for all-solid-state lithium batteries. Mater. Sci. Eng. R Rep. 2019, 136, 27–46. [Google Scholar]
- Zhang, H.; Li, C.; Piszcz, M.; Coya, E.; Rojo, T.; Rodriguez-Martinez, L.M.; Armand, M.; Zhou, Z. Single lithium-ion conducting solid polymer electrolytes: Advances and perspectives. Chem. Soc. Rev. 2017, 46, 797–815. [Google Scholar]
- Zhang, H.; Eshetu, G.G.; Judez, X.; Li, C.; Rodriguez-Martínez, L.M.; Armand, M. Electrolyte additives for lithium metal anodes and rechargeable lithium metal batteries: Progress and perspectives. Angew. Chem. Int. Ed. 2018, 57, 15002–15027. [Google Scholar]
- Mauger, A.; Julien, C.M.; Paolella, A.; Armand, M.; Zaghib, K. A comprehensive review of lithium salts and beyond for rechargeable batteries: Progress and perspectives. Mater. Sci. Eng. R Rep. 2018, 134, 1–21. [Google Scholar] [CrossRef]
- Olsher, U.; Izatt, R.M.; Bradshaw, J.S.; Dalley, N.K. Coordination chemistry of lithium ion: A crystal and molecular structure review. Chem. Rev. 1991, 91, 137–164. [Google Scholar] [CrossRef]
- Jeong, K.; Park, S.; Lee, S.-Y. Revisiting polymeric single lithium-ion conductors as an organic route for all-solid-state lithium ion and metal batteries. J. Mater. Chem. A 2019, 7, 1917–1935. [Google Scholar] [CrossRef]
- Budd, P.M. Chapter 11—Polyelectrolytes. In Comprehensive Polymer Science and Supplements, 1st ed.; Pergamon Press: Oxford, UK, 1989; Volume 1, pp. 215–230. [Google Scholar]
- Berthier, C.; Gorecki, W.; Minier, M.; Armand, M.B.; Chabagno, J.M.; Rigaud, P. Microscopic investigation of ionic conductivity in alkali metal salts-poly(ethylene oxide) adducts. Solid State Ion. 1983, 11, 91–95. [Google Scholar] [CrossRef]
- Ward, I.M.; Boden, N.; Cruickshank, J.; Leng, S.A. NMR studies of ionic mobility and molecular mobility in polymer electrolytes. Electrochim. Acta 1995, 40, 2071–2076. [Google Scholar]
- Borodin, O.; Smith, G.D.; Henderson, W. Li+ cation environment, transport, and mechanical properties of the LiTFSI doped N-Methyl-N-alkylpyrrolidinium+TFSI- ionic liquids. J. Phys. Chem. B 2006, 110, 16879–16886. [Google Scholar] [CrossRef] [PubMed]
- Kreuer, K.-D.; Rabenau, A.; Weppner, W. Vehicle mechanism, a new model for the interpretation of the conductivity of fast proton conductors. Angew. Chem. Int. Ed. 1982, 21, 208–209. [Google Scholar] [CrossRef]
- Ganesan, V. Ion transport in polymeric ionic liquids: Recent developments and open questions. Mol. Syst. Des. Eng. 2019, 4, 280–293. [Google Scholar]
- Frenck, L.; Sethi, G.K.; Maslyn, J.A.; Balsara, N.P. Factors that control the formation of dendrites and other morphologies on lithium metal anodes. Front. Energy Res. 2019, 7, 115. [Google Scholar] [CrossRef]
- Barai, P.; Higa, K.; Srinivasan, V. Effect of initial state of lithium on the propensity for dendrite formation: A theoretical study. J. Electrochem. Soc. 2016, 164, A180–A189. [Google Scholar] [CrossRef]
- Fenton, D.E.; Parker, J.M.; Wright, P.V. Complexes of alkali metal ions with poly(ethylene oxide). Polymer 1973, 14, 589. [Google Scholar]
- Sun, B.; Xu, C.; Mindemark, J.; Gustafsson, T.; Edström, K.; Brandell, D. At the polymer electrolyte interfaces: The role of the polymer host in interphase layer formation in Li-batteries. J. Mater. Chem. A 2015, 3, 13994–14000. [Google Scholar]
- Yoon, H.-K.; Chung, W.-S.; Jo, N.-J. Study on ionic transport mechanism and interactions between salt and polymer chain in PAN based solid polymer electrolytes containing LiCF3SO3. Electrochim. Acta 2004, 50, 289–293. [Google Scholar] [CrossRef]
- Ferry, A. Ionic interactions and transport properties in methyl terminated poly(propylene glycol)(4000) complexed with LiCF3SO3. J. Phys. Chem. B 1997, 101, 150–157. [Google Scholar] [CrossRef]
- Ferry, A. Effects of dynamic spatial disorder on ionic transport properties in polymer electrolytes based on poly(propylene glycol)(4000). J. Chem. Phys. 1997, 107, 9168–9175. [Google Scholar] [CrossRef]
- Alamgir, M.; Moulton, R.D.; Abraham, K.M. Li+-conductive polymer electrolytes derived from poly(1,3-dioxolane) and polytetrahydrofuran. Electrochim. Acta 1991, 36, 773–782. [Google Scholar] [CrossRef]
- Sahadeo, E.; Wang, Y.; Lin, C.-F.; Li, Y.; Rubloff, G.; Lee, S.B. Mg2+ ion-catalyzed polymerization of 1,3-dioxolane in battery electrolytes. Chem. Comm. 2020, 56, 4583–4586. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Liu, X.; Stalin, S.; Khan, K.; Archer, L. Solid-state polymer electrolytes with in-built fast interfacial transport for secondary lithium batteries. Nat. Energy 2019, 4, 365–373. [Google Scholar] [CrossRef]
- Wu, X.-L.; Xin, S.; Seo, H.-H.; Kim, J.; Guo, Y.-G.; Lee, J.-S. Enhanced Li+ conductivity in PEO–LiBOB polymer electrolytes by using succinonitrile as a plasticizer. Solid State Ion. 2011, 186, 1–6. [Google Scholar] [CrossRef]
- Johan, M.R.; Shy, O.H.; Ibrahim, S.; Mohd, Y.; Siti, M.; Hui, T.Y. Effects of Al2O3 nanofiller and EC plasticizer on the ionic conductivity enhancement of solid PEO–LiCF3SO3 solid polymer electrolyte. Solid State Ion. 2011, 196, 41–47. [Google Scholar] [CrossRef]
- Asghar, A.; Abdul Samad, S.; Singh Lalia, B.; Hashaikeh, R. PEG based quasi-solid polymer electrolyte: Mechanically supported by networked cellulose. J. Membr. Sci. 2012, 421, 85–90. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, N.; Zhang, M.; Li, Y.; Chu, P.K.; Guo, X.; Di, Z.; Wang, X.; Li, H. Flexible and ion-conducting membrane electrolytes for solid-state lithium batteries: Dispersion of garnet nanoparticles in insulating polyethylene oxide. Nano Energy 2016, 28, 447–454. [Google Scholar] [CrossRef]
- Phan, T.N.T.; Issa, S.; Gigmes, D. Poly(ethylene oxide)-based block copolymer electrolytes for lithium metal batteries. Polym. Int. 2019, 68, 7–13. [Google Scholar] [CrossRef]
- Rosso, M.; Brissot, C.; Teyssot, A.; Dollé, M.; Sannier, L.; Tarascon, J.-M.; Bouchet, R.; Lascaud, S. Dendrite short-circuit and fuse effect on Li/polymer/Li cells. Electrochim. Acta 2006, 51, 5334–5340. [Google Scholar] [CrossRef]
- Blonsky, P.M.; Shriver, D.F.; Austin, P.; Allcock, H.R. Polyphosphazene solid electrolytes. J. Am. Chem. Soc. 1984, 106, 6854–6855. [Google Scholar] [CrossRef]
- Allcock, H.R.; Austin, P.E.; Neenan, T.X.; Sisko, J.T.; Blonsky, P.M.; Shriver, D.F. Polyphosphazenes with etheric side groups: Prospective biomedical and solid electrolyte polymers. Macromolecules 1986, 19, 1508–1512. [Google Scholar] [CrossRef]
- Tonge, J.S.; Blonsky, P.M.; Shriver, D.F.; Allcock, H.R.; Austin, P.E.; Neenan, T.X.; Sisko, J.T. Effect of side-chain length and crosslinking on ionic conductivity in polyphosphazene solid electrolytes. Proc. Electrochem. Soc. 1987, 87, 533–544. [Google Scholar]
- Allcock, H.R.; Sunderland, N.J.; Ravikiran, R.; Nelson, J.M. Polyphosphazenes with novel architectures: influence on physical properties and behavior as solid polymer electrolytes. Macromolecules 1998, 31, 8026–8035. [Google Scholar] [CrossRef]
- Allcock, H.R.; Kuharcik, S.E.; Reed, C.S.; Napierala, M.E. Synthesis of polyphosphazenes with ethyleneoxy-containing side groups: New solid electrolyte materials. Macromolecules 1996, 29, 3384–3389. [Google Scholar] [CrossRef]
- Allcock, R.; O’Connor, S.J.M.; Olmeijer, D.L.; Napierala, M.E.; Cameron, C.G. Polyphosphazenes bearing branched and linear oligoethyleneoxy side groups as solid solvents for ionic conduction. Macromolecules 1996, 29, 7544–7552. [Google Scholar] [CrossRef]
- Oh, B.; Vissers, D.; Zhang, Z.; West, R.; Tsukamoto, H.; Amine, K. New interpenetrating network type poly(siloxane-g-ethylene oxide) polymer electrolyte for lithium battery. J. Power Sources 2003, 119, 442–447. [Google Scholar] [CrossRef]
- Borodin, O.; Smith, G.D. Molecular Dynamics simulations of comb-branched poly(epoxide ether)-based polymer electrolytes. Macromolecules 2007, 40, 1252–1258. [Google Scholar] [CrossRef]
- Wang, Z.H.; Chen, S.J.; Huang, Z.; Wei, Z.Y.; Shen, L.; Gu, H.; Xu, X.X.; Yao, X.Y. High conductivity polymer electrolyte with comb-like structure via a solvent-free UV-cured method for large-area ambient all-solid-sate lithium batteries. J. Mater. 2019, 5, 195–203. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, C.Y.; Zheng, L.P.; Xu, F.; Feng, W.F.; Li, H.; Huang, X.J.; Armand, M.; Nie, J.; Zhou, Z.B. Lithium bis(fluorosulfonyl)imide/poly(ethylene oxide) polymer electrolyte. Electrochim. Acta 2014, 133, 529–538. [Google Scholar] [CrossRef]
- Rosenbach, D.; Mödl, N.; Hahn, M.; Petry, J.; Danzer, M.A.; Thelakkat, M. Synthesis and comparative studies of solvent-free brush polymer electrolytes for lithium batteries. ACS Appl. Energy Mater. 2019, 2, 3373–3388. [Google Scholar] [CrossRef]
- Kim, D.-W.; Park, J.-K.; Gong, M.-S.; Song, H.-Y. Effect of grafting degree and side PEO chain length on the ionic conductivities of NBR-g-PEO based polymer electrolytes. Polym. Eng. Sci. 1994, 34, 1305–1313. [Google Scholar] [CrossRef]
- Sun, J.; Stone, G.M.; Balsara, N.P.; Zuckermann, R.N. Structure–conductivity relationship for peptoid-based PEO–mimetic polymer electrolytes. Macromolecules 2012, 45, 5151–5156. [Google Scholar] [CrossRef]
- Guo, M.; Zhang, M.; He, D.; Hu, J.; Wang, X.; Gong, C.; Xie, X.; Xue, Z. Comb-like solid polymer electrolyte based on polyethylene glycol-grafted sulfonated polyether ether ketone. Electrochim. Acta 2017, 255, 396–404. [Google Scholar] [CrossRef]
- Zardalidis, G.; Pipertzis, A.; Mountrichas, G.; Pispas, S.; Mezger, M.; Floudas, G. Effect of polymer architecture on the ionic conductivity. Densely grafted poly(ethylene oxide) brushes doped with LiTf. Macromolecules 2016, 49, 2679–2687. [Google Scholar] [CrossRef]
- Glynos, E.; Pantazidis, C.; Sakellariou, G. Designing all-polymer nanostructured solid electrolytes: Advances and prospects. ACS Omega 2020, 5, 2531–2540. [Google Scholar] [CrossRef]
- Aldalur, I.; Zhang, H.; Piszcz, M.; Oteo, U.; Rodriguez-Martinez, L.M.; Shanmukaraj, D.; Rojo, T.; Armand, M. Jeffamine® based polymers as highly conductive polymer electrolytes and cathode binder materials for battery application. J. Power Sources 2017, 347, 37–46. [Google Scholar] [CrossRef]
- Aldalur, I.; Martinez-Ibañez, M.; Piszcz, M.; Rodriguez-Martinez, L.M.; Zhang, H.; Armand, M. Lowering the operational temperature of all-solid-state lithium polymer cell with highly conductive and interfacially robust solid polymer electrolytes. J. Power Sources 2018, 383, 144–149. [Google Scholar] [CrossRef]
- Kerr, J.B.; Sloop, S.E.; Liu, G.; Han, Y.B.; Hou, J.; Wang, S.E. From molecular models to system analysis for lithium battery electrolytes. J. Power Sources 2002, 110, 389–400. [Google Scholar] [CrossRef]
- Kuan, W.-F.; Remy, R.; Mackay, M.E.; Epps, T.H. Controlled ionic conductivity via tapered block polymer electrolytes. RSC Adv. 2015, 5, 12597–12604. [Google Scholar] [CrossRef]
- Harris, D.J.; Bonagamba, T.J.; Schmidt-Rohr, K.; Soo, P.P.; Sadoway, D.R.; Mayes, A.M. Solid-state NMR investigation of block copolymer electrolyte dynamics. Macromolecules 2002, 35, 3772–3774. [Google Scholar] [CrossRef]
- Huang, B.; Cook, C.C.; Mui, S.; Soo, P.P.; Staelin, D.H.; Mayes, A.; Sadoway, D.R. High energy density, thin-film, rechargeable lithium batteries for marine field operations. J. Power Sources 2001, 97, 674–676. [Google Scholar] [CrossRef]
- Feng, S.; Shi, D.; Liu, F.; Zheng, L.; Nie, J.; Feng, W.; Huang, X.; Armand, M.; Zhou, Z. Single lithium-ion conducting polymer electrolytes based on poly[(4-styrenesulfonyl)(trifluoromethanesulfonyl)imide] anions. Electrochim. Acta 2013, 93, 254–263. [Google Scholar]
- Porcarelli, L.; Shaplov, A.S.; Salsamendi, M.; Nair, J.R.; Vygodskii, Y.S.; Mecerreyes, D.; Gerbaldi, C. Single-ion block copoly(ionic liquid)s as electrolytes for all-solid state lithium batteries. ACS Appl. Mater. Interfaces 2016, 8, 10350–10359. [Google Scholar] [CrossRef]
- Niitani, T.; Amaike, M.; Nakano, H.; Dokko, K.; Kanamura, K. Star-shaped polymer electrolyte with microphase separation structure for all-solid-state lithium batteries. J. Electrochem. Soc. 2009, 156, A577–A583. [Google Scholar] [CrossRef]
- Ren, S.; Chang, H.; He, L.; Dang, X.; Fang, Y.; Zhang, L.; Li, H.; Hu, Y.; Lin, Y. Preparation and ionic conductive properties of all-solid polymer electrolytes based on multiarm star block polymers. J. Appl. Polym. Sci. 2013, 129, 1131–1142. [Google Scholar]
- Wang, A.; Xu, H.; Zhou, Q.; Liu, X.; Li, Z.; Gao, R.; Wu, N.; Guo, Y.; Li, H.; Zhang, L. A new all-solid-state hyperbranched star polymer electrolyte for lithium ion batteries: Synthesis and electrochemical properties. Electrochim. Acta 2016, 212, 372–379. [Google Scholar] [CrossRef]
- Wang, A.; Xu, H.; Zhou, Q.; Liu, X.; Li, Z.; Gao, R.; Liu, X.; Zhang, L. Electrochemical performances of a new solid composite polymer electrolyte based on hyperbranched star polymer and ionic liquid for lithium-ion batteries. J. Solid State Electrochem. 2017, 21, 2355–2364. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, Y.; Liang, Y.; Dong, H.; Hao, F.; Wang, A.; Zhu, Y.; Cui, X.; Yao, Y. Hyperbranched PEO-based hyperstar solid polymer electrolytes with simultaneous improvement of ion transport and mechanical strength. ACS Appl. Energy Mater. 2019, 2, 1608–1615. [Google Scholar]
- Grewal, M.S.; Tanaka, M.; Kawakami, H. Bifunctional poly(ethylene glycol) based crosslinked network polymers as electrolytes for all-solid-state lithium ion batteries. Polym. Int. 2019, 68, 684–693. [Google Scholar] [CrossRef]
- Hasan, N.; Pulst, M.; Samiullah, M.H.; Kressler, J. Comparison of Li+-ion conductivity in linear and crosslinked poly(ethylene oxide). J. Polym. Sci. B Polym. Phys. 2019, 57, 21–28. [Google Scholar] [CrossRef]
- Carvalho, L.M.; Guégan, P.; Cheradame, H.; Gomes, A.S. Variation of the mesh size of PEO-based networks filled with TFSILi: From an Arrhenius to WLF type conductivity behavior. Eur. Polym. J. 2000, 36, 401–409. [Google Scholar]
- Niitani, T.; Shimada, M.; Kawamura, K.; Dokko, K.; Rho, Y.-H.; Kanamura, K. Synthesis of Li+ Ion Conductive PEO-PSt Block Copolymer Electrolyte with Microphase Separation Structure. Electrochem. Solid State Lett. 2005, 8, A385–A388. [Google Scholar]
- Phan, T.T.N.; Robinet, M.; Bouchet, R.; Bertin, D. PS-b-PEO-b-PS based solid polymer electrolytes for lithium rechargeable batteries. PMSE Prepr. 2008, 99, 329–330. [Google Scholar]
- Zhang, B.; Zhang, Y.; Zhang, N.; Liu, J.; Cong, L.; Liu, J.; Sun, L.; Mauger, A.; Julien, C.M.; Xie, H.; et al. Synthesis and interface stability of polystyrene-poly(ethylene glycol)-polystyrene triblock copolymer as solid-state electrolyte for lithium-metal batteries. J. Power Sources 2019, 428, 93–104. [Google Scholar]
- Young, W.-S.; Kuan, W.-F.; Epps, T.H. Block copolymer electrolytes for rechargeable lithium batteries. J. Polym. Sci. Part B Polym. Phys. 2014, 52, 1–16. [Google Scholar]
- Villaluenga, I.; Chen, X.C.; Devaux, D.; Hallinan, D.; Balsara, N.P. Nanoparticle-driven assembly of highly conducting hybrid block copolymer electrolytes. Macromolecules 2015, 48, 358–364. [Google Scholar] [CrossRef]
- Irwin, M.T.; Hickey, R.J.; Xie, S.; So, S.; Bates, F.; Lodge, T.P. Structure-conductivity relationships in ordered and disordered salt-doped diblock copolymer/homopolymer blends. Macromolecules 2016, 49, 6928–6939. [Google Scholar]
- Chopade, S.; Au, J.G.; Li, Z.; Schmidt, P.W.; Hillmyer, M.A.; Lodge, T.P. Robust polymer electrolyte membranes with high ambient-temperature lithium-ion conductivity via polymerization-induced microphase separation. ACS Appl. Mater. Interfaces 2017, 9, 14561–14565. [Google Scholar] [CrossRef]
- Wu, N.; Shi, Y.-R.; Lang, S.-Y.; Zhou, J.M.; Liang, J.-Y.; Wang, W.; Tan, S.-J.; Yin, Y.-X.; Wen, R.; Guo, Y.-G. Self-healable solid polymeric electrolytes for stable and flexible lithium metal batteries. Angew. Chem. Int. Ed. 2019, 58, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lu, W.; Cong, L.; Liu, J.; Sun, L.; Mauger, A.; Julien, C.M.; Xie, H.; Liu, J. Cross-linking network based on Poly(ethylene oxide): Solid polymer electrolyte for room temperature lithium battery. J. Power Sources 2019, 420, 63–72. [Google Scholar] [CrossRef]
- Choudhury, S.; Stalin, S.; Vu, D.; Warren, A.; Deng, Y.; Biswal, P.; Archer, L.A. Solid-state polymer electrolytes for high-performance lithium metal batteries. Nat. Commun. 2019, 10, 4398. [Google Scholar] [CrossRef] [PubMed]
- Porcarelli, L.; Gerbaldi, C.; Bella, F.; Nair, J.R. Super soft all-ethylene oxide polymer electrolyte for safe all-solid lithium batteries. Sci. Rep. 2016, 6, 19892. [Google Scholar] [CrossRef] [PubMed]
- Khurana, R.; Schaefer, J.L.; Archer, L.A.; Coates, G.W. Suppression of lithium dendrite growth using cross-linked polyethylene/poly(ethylene oxide) electrolytes: A new approach for practical lithium-metal polymer batteries. J. Am. Chem. Soc. 2014, 136, 7395–7402. [Google Scholar] [PubMed]
- Porcarelli, L.; Shaplov, A.S.; Bella, F.; Nair, J.R.; Mecerreyes, D.; Gerbaldi, C. Single-ion conducting polymer electrolytes for lithium metal polymer batteries that operate at ambient temperature. ACS Energy Lett. 2016, 1, 678–682. [Google Scholar]
- Luo, G.; Yuan, B.; Guan, T.; Cheng, F.; Zhang, W.; Chen, J. Synthesis of single lithium-ion conducting polymer electrolyte membrane for solid-state lithium metal batteries. ACS Appl. Energy Mater. 2019, 2, 3028–3034. [Google Scholar] [CrossRef]
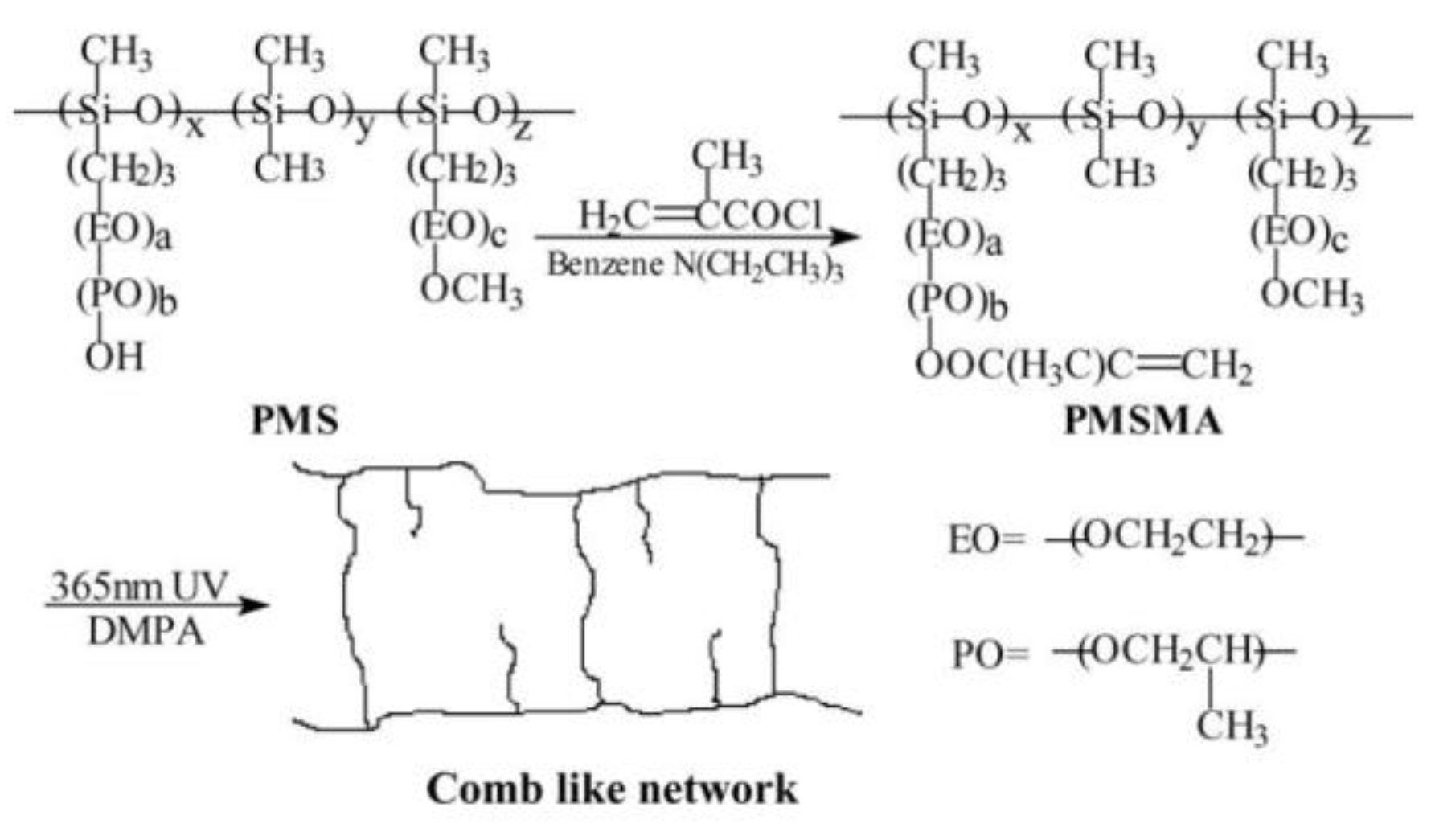
- Jiang, H.; Fang, S. All solid-state comb-like network polymer electrolytes based on poly(methylsiloxane). J. Power Sources 2006, 159, 673–678. [Google Scholar] [CrossRef]
- Zhang, Z.; Sherlock, D.; West, R.; West, R.; Amine, K.; Lyons, L.J. Cross-linked network polymer electrolytes based on a polysiloxane backbone with oligo(oxyethylene) side chains: synthesis and conductivity. Macromolecules 2003, 36, 9176–9180. [Google Scholar] [CrossRef]
- Daigle, J.-C.; Asakawa, Y.; Vijh, A.; Hovington, P.; Armand, M.; Zaghib, K. Exceptionally stable polymer electrolyte for a lithium battery based on cross-linking by a residue-free process. J. Power Sources 2016, 332, 213–221. [Google Scholar]
- Grewal, M.S.; Tanaka, M.; Kawakami, H. Free-standing polydimethylsiloxane-based cross-linked network solid polymer electrolytes for future lithium ion battery applications. Electrochim. Acta 2019, 307, 148–156. [Google Scholar] [CrossRef]
- Xia, Y.; Fujieda, T.; Tatsumi, K.; Prosini, P.P.; Sakai, T. Thermal and electrochemical stability of cathode materials in solid polymer electrolyte. J. Power Sources 2001, 92, 234–243. [Google Scholar] [CrossRef]
- Xia, Y.; Tatsumi, K.; Fujieda, T.; Prosini, P.P.; Sakai, T. Solid-State Lithium-Polymer Batteries Using Lithiated MnO2 Cathodes. J. Electrochem. Soc. 2000, 147, 2050–2056. [Google Scholar] [CrossRef]
- Ebadi, M.; Marchiori, C.; Mindemark, J.; Brandell, D.; Araujo, C.M. Assessing structure and stability of polymer/lithium-metal interfaces from first-principles calculations. J. Mater. Chem. A 2019, 7, 8394–8404. [Google Scholar] [CrossRef]
- Zaghib, K.; Armand, M.; Gauthier, M. Electrochemistry of anodes in solid-state Li-ion polymer batteries. J. Electrochem. Soc. 1998, 145, 3135. [Google Scholar]
- Yang, Q.; Zhang, Z.; Sun, X.-G.; Hu, Y.-S.; Xing, H.; Dai, S. Ionic liquids and derived materials for lithium and sodium batteries. Chem. Soc. Rev. 2018, 47, 2020–2064. [Google Scholar] [CrossRef]
- Osada, I.; de Vries, H.; Scrosati, B.; Passerini, S. Ionic-liquid-based polymer electrolytes for battery applications. Angew. Chem. Int. Ed. 2016, 55, 500–513. [Google Scholar] [CrossRef]
- MacFarlane, D.R.; Forsyth, M.; Howlett, P.C.; Kar, M.; Passerini, S.; Pringle, J.M.; Ohno, H.; Watanabe, M.; Yan, F.; Zheng, W.; et al. Ionic liquids and their solid-state analogues as materials for energy generation and storage. Nat. Rev. Mater. 2016, 1, 15005. [Google Scholar]
- Appetecchi, G.B.; Montanino, M.; Passerini, S. Ionic liquid-based electrolytes for high energy, safer lithium batteries. In Ionic Liquids: Science and Applications, ACS Symposium Series; ACS Publications: Washington, DC, WA, USA, 2012; Chapter 4; Volume 1117, pp. 67–128. [Google Scholar]
- Armand, M.; Endres, F.; MacFarlane, D.R.; Ohno, H.; Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 2009, 8, 621. [Google Scholar]
- Ogihara, W.; Washiro, S.; Nakajima, H.; Ohno, H. Effect of cation structure on the electrochemical and thermal properties of ion conductive polymers obtained from polymerizable ionic liquids. Electrochim. Acta 2006, 51, 2614–2619. [Google Scholar] [CrossRef]
- Keith, J.R.; Rebello, N.J.; Cowen, B.J.; Ganesan, V. Influence of counterion structure on conductivity of polymerized ionic liquids. ACS Macro Lett. 2019, 8, 387–392. [Google Scholar] [CrossRef]
- Ohno, H.; Yoshizawa, M.; Ogihara, W. Development of new class of ion conductive polymers based on ionic liquids. Electrochim. Acta 2004, 50, 255–261. [Google Scholar] [CrossRef]
- Fan, F.; Wang, W.; Holt, A.P.; Feng, H.; Uhrig, D.; Lu, X.; Hong, T.; Wang, Y.; Kang, N.-G.; Mays, J.; et al. Effect of molecular weight on the ion transport mechanism in polymerized ionic liquids. Macromolecules 2016, 49, 4557–4570. [Google Scholar] [CrossRef]
- Kuray, P.; Noda, T.; Matsumoto, A.; Iacob, C.; Inoue, T.; Hickner, M.A.; Runt, J. Ion transport in pendant and backbone polymerized ionic liquids. Macromolecules 2019, 52, 6438–6448. [Google Scholar] [CrossRef]
- Delhorbe, V.; Bresser, D.; Mendil-Jakani, H.; Rannou, P.; Bernard, L.; Gutel, T.; Lyonnard, S.; Picard, L. Unveiling the ion conduction mechanism in imidazolium-based poly(ionic liquids): A comprehensive investigation of the structure-to-transport interplay. Macromolecules 2017, 50, 4309–4321. [Google Scholar] [CrossRef]
- Yin, K.; Zhang, Z.; Yang, L.; Hirano, S.-I. An imidazolium-based polymerized ionic liquid via novel synthetic strategy as polymer electrolytes for lithium ion batteries. J. Power Sources 2014, 258, 150–154. [Google Scholar] [CrossRef]
- Zhang, P.; Li, M.; Yang, B.; Fang, Y.; Jiang, X.; Veith, G.M.; Sun, X.-G.; Dai, S. Polymerized ionic networks with high charge density: Quasi-solid electrolytes in lithium-metal batteries. Adv. Mater. 2015, 27, 8088–8094. [Google Scholar] [CrossRef]
- Tian, X.; Yi, Y.; Yang, P.; Liu, P.; Qu, L.; Li, M.; Hu, Y.-S.; Yang, B. High-charge density polymerized ionic networks boosting high ionic conductivity as quasi-solid electrolytes for high-voltage batteries. ACS Appl. Mater. Interfaces 2019, 11, 4001–4010. [Google Scholar] [CrossRef]
- Zhou, D.; Liu, R.; Zhang, J.; Qi, X.; He, Y.-B.; Li, B.; Yang, Q.-H.; Hu, Y.-S.; Kang, F. In situ synthesis of hierarchical poly(ionic liquid)-based solid electrolytes for high-safety lithium-ion and sodium-ion batteries. Nano Energy 2017, 33, 45–54. [Google Scholar] [CrossRef]
| Backbone | Chemical Structure | Salt | Conductivity (S∙cm−1) | Potential Window (V) | Ref. |
|---|---|---|---|---|---|
| Poly(epoxide ether) | 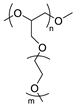 | LiTFSI | 3.2 × 10−4 (60 °C) | ND | [75] |
| Polysiloxane | 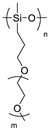 | LiTFSI | >10−4 (25 °C) | 4.5 | [63] |
| Polyphosphazene | 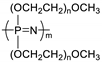 | lithium triflate (LiSO3CF3) | ~ 3 × 10−5 (25 °C) | ND | [61] |
| Polymethacrylate | 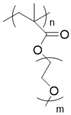 | LiTFSI | 1.44 × 10−4 (30 °C) 7.26 × 10−4 (60 °C) | > 5 | [65] |
| Poly(acrylonitrile-butadiene) | 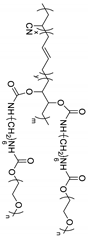 | LiSO3CF3 | ~3 × 10−5 (25 °C) | ND | [68] |
| Polynorbornene | 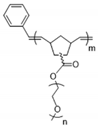 | LiTFSI | >3 × 10−5 (25 °C) >7 × 10−4 (80 °C) | ~ 3.5 | [67] |
| Poly(hydroxyl styrene) | 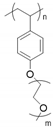 | LiSO3CF3 | ∼6 × 10−5 (60 °C) | ND | [71] |
| Polyether ether ketone | 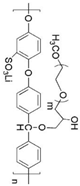 | LiClO4 | > 10−5 (room temperature) | ND | [70] |
| Polypeptoid | 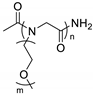 | LiTFSI | ~10−5 (50 °C) 2.6 × 10−4 (100 °C) | ND | [69] |
| Poly(ethylene-co-maleic anhydride) | 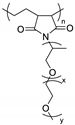 | LiTFSI | 4.5 × 10−5 (room temperature) 5.3 × 10−4 (70 °C) | >4 (stainless steel electrode) >3 (aluminum electrode) | [72] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Daigle, J.-C.; Zaghib, K. Comprehensive Review of Polymer Architecture for All-Solid-State Lithium Rechargeable Batteries. Materials 2020, 13, 2488. https://doi.org/10.3390/ma13112488
Zhang X, Daigle J-C, Zaghib K. Comprehensive Review of Polymer Architecture for All-Solid-State Lithium Rechargeable Batteries. Materials. 2020; 13(11):2488. https://doi.org/10.3390/ma13112488
Chicago/Turabian StyleZhang, Xuewei, Jean-Christophe Daigle, and Karim Zaghib. 2020. "Comprehensive Review of Polymer Architecture for All-Solid-State Lithium Rechargeable Batteries" Materials 13, no. 11: 2488. https://doi.org/10.3390/ma13112488
APA StyleZhang, X., Daigle, J.-C., & Zaghib, K. (2020). Comprehensive Review of Polymer Architecture for All-Solid-State Lithium Rechargeable Batteries. Materials, 13(11), 2488. https://doi.org/10.3390/ma13112488






