The Investigation of High-Temperature SAW Oxygen Sensor Based on ZnO Films
Abstract
:1. Introduction
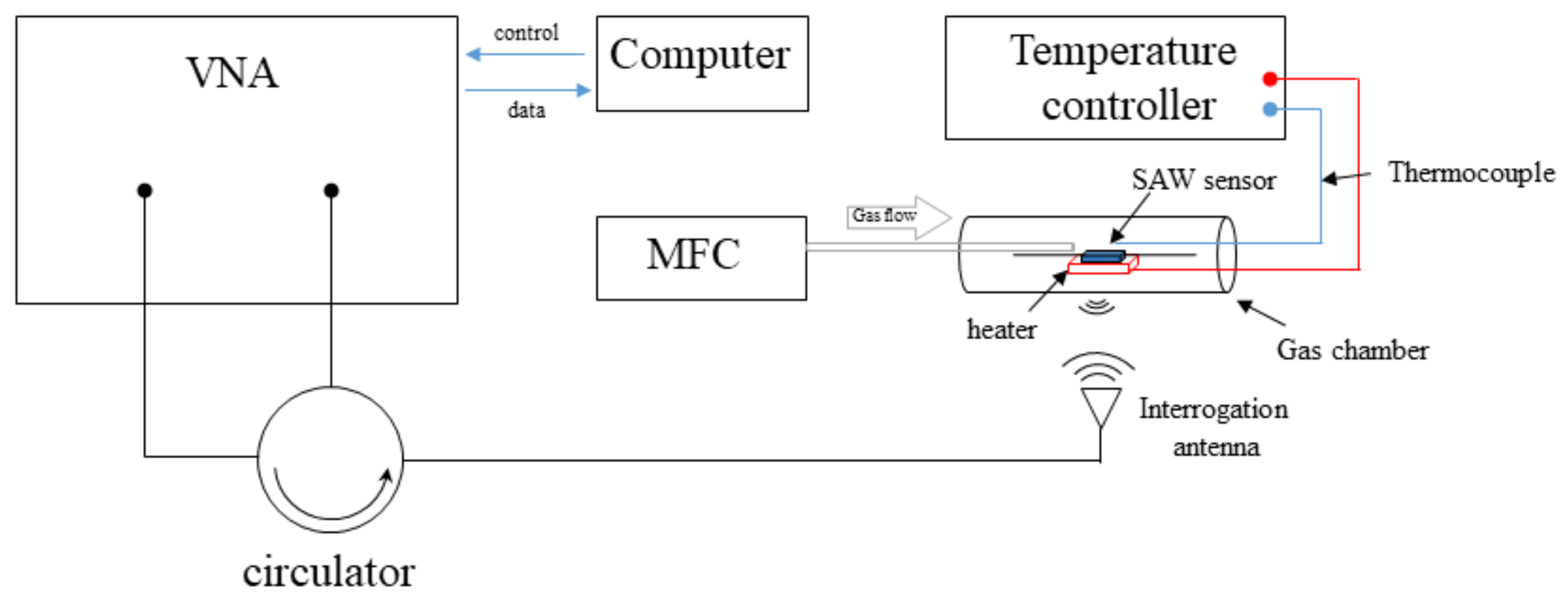
2. Experimental Setup
3. Results and Discussions
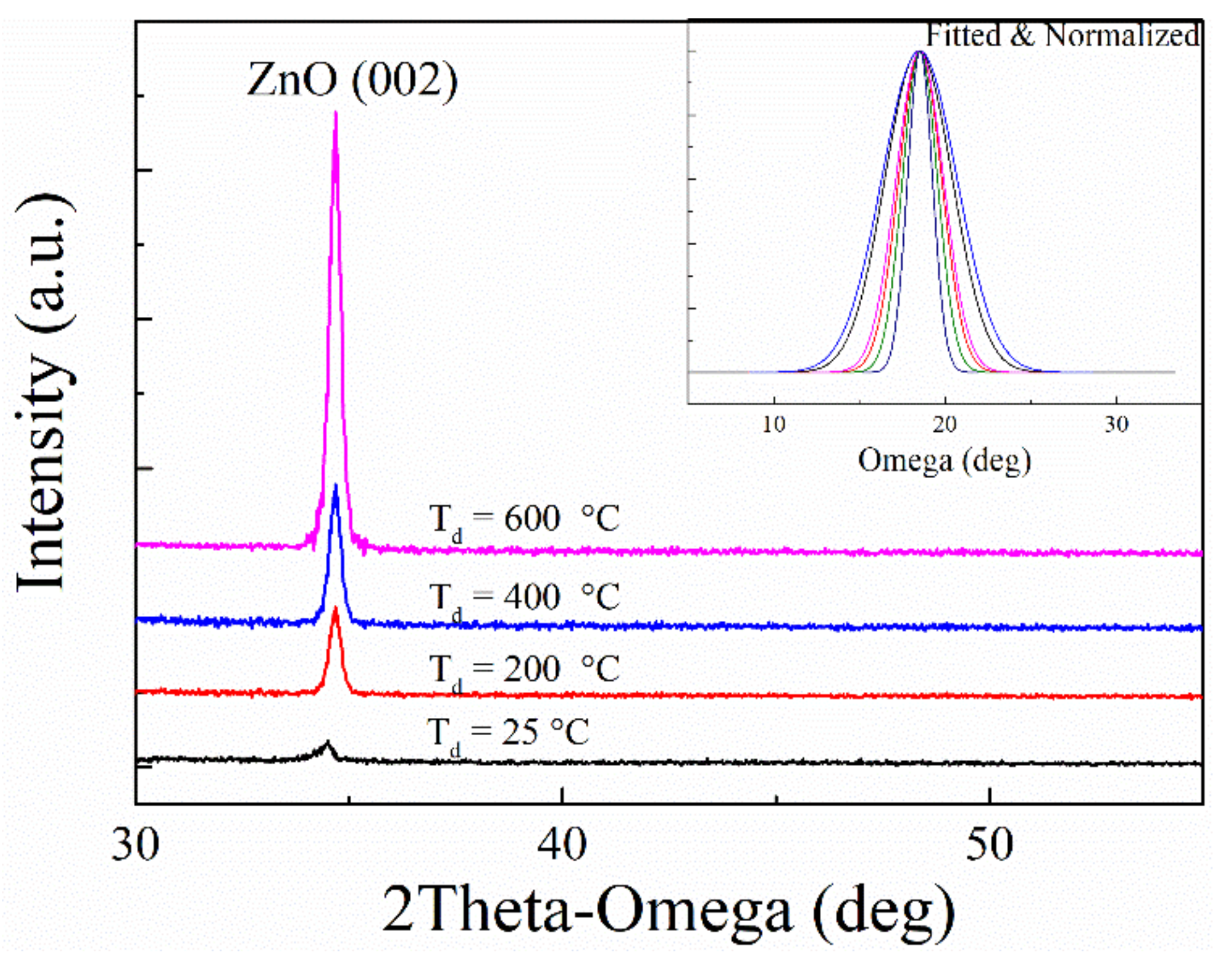
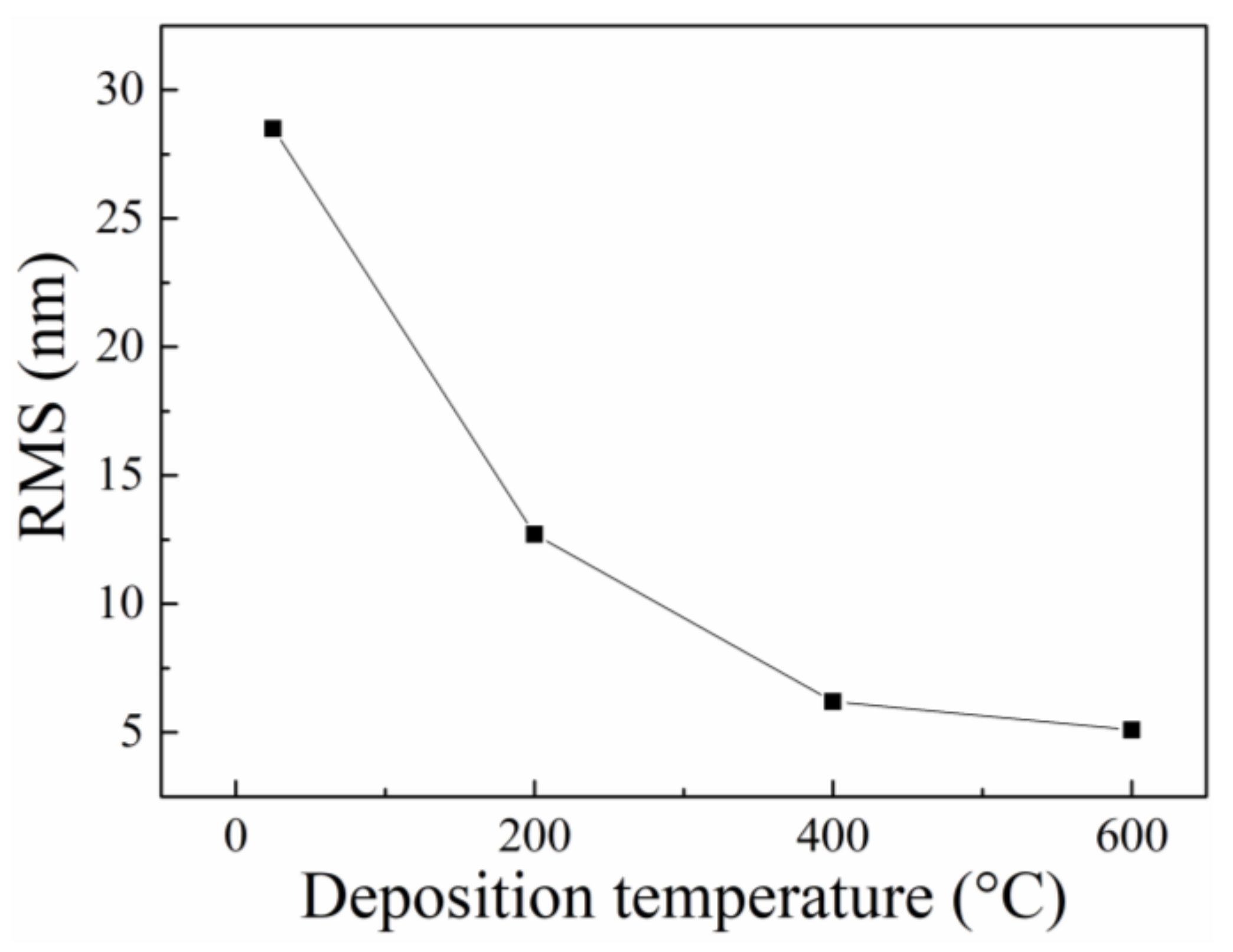
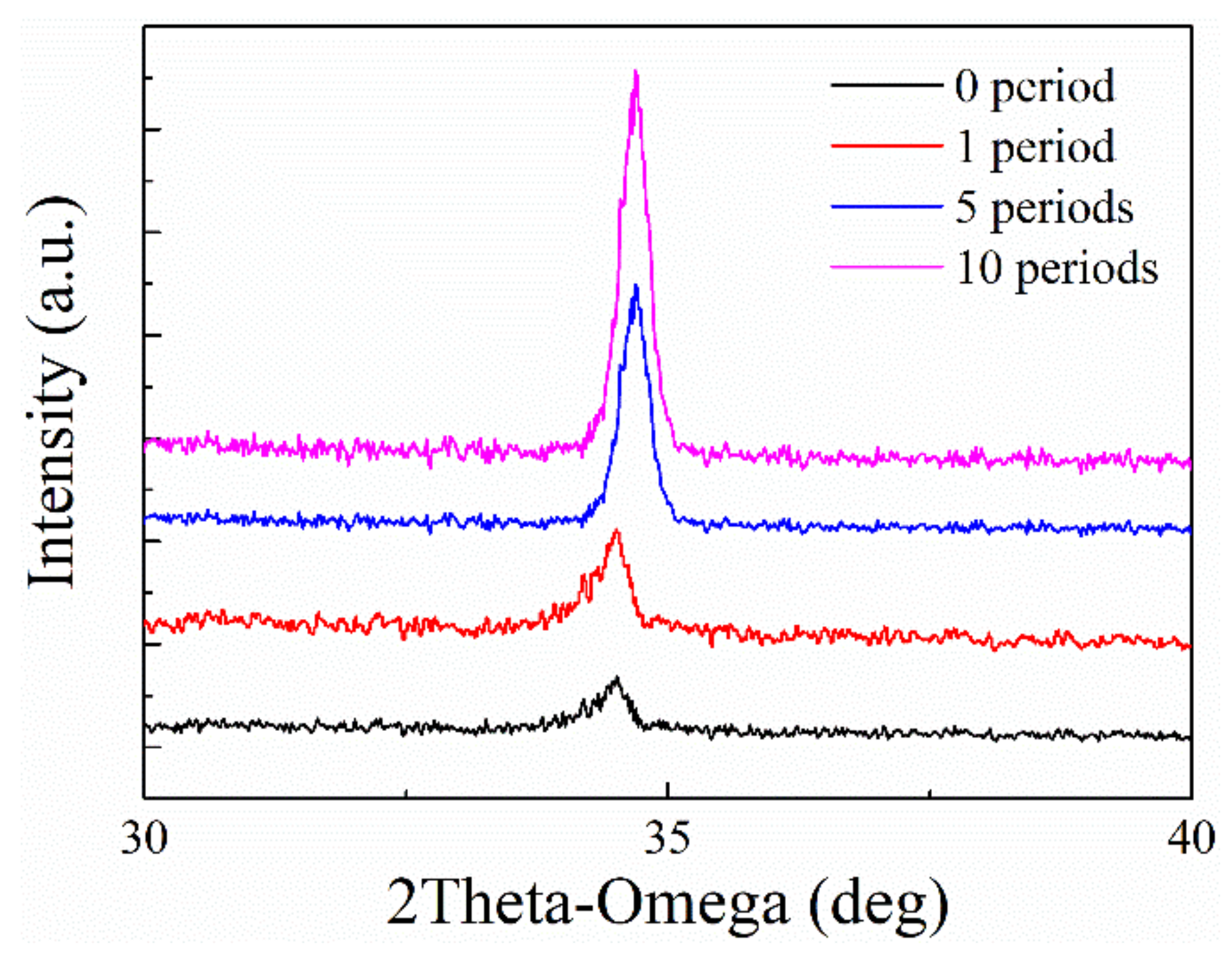
3.1. Structural Characterization
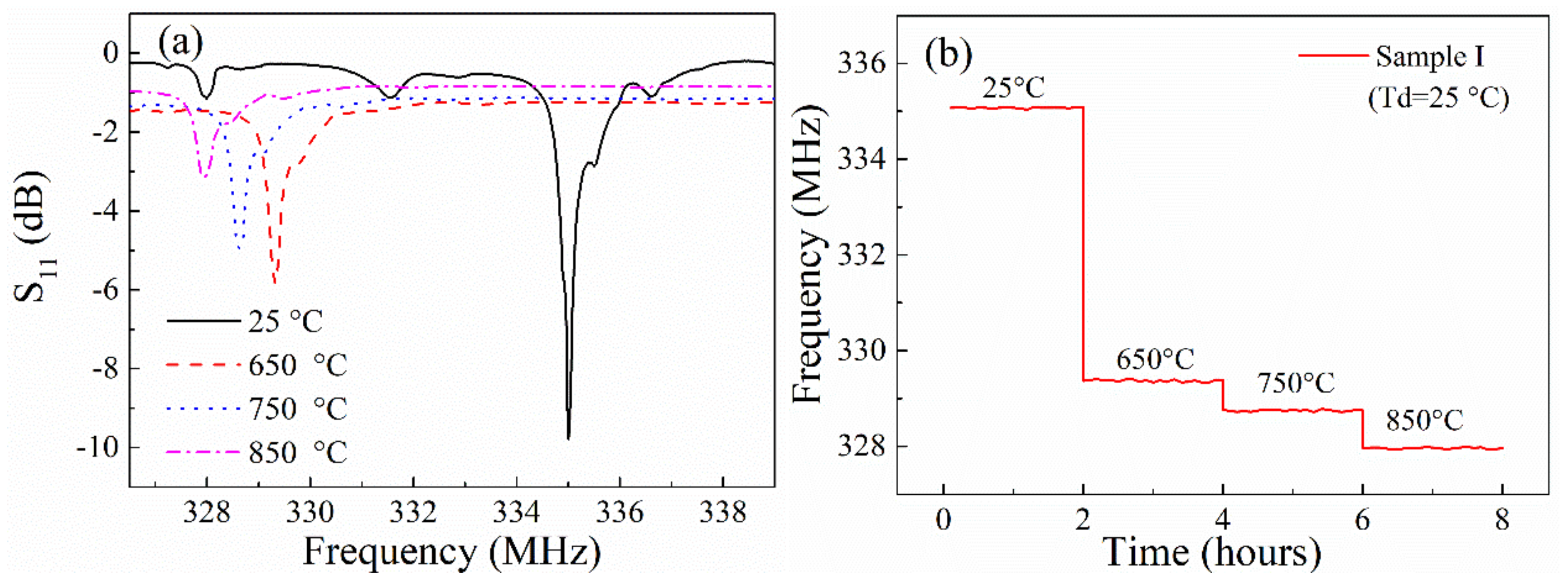
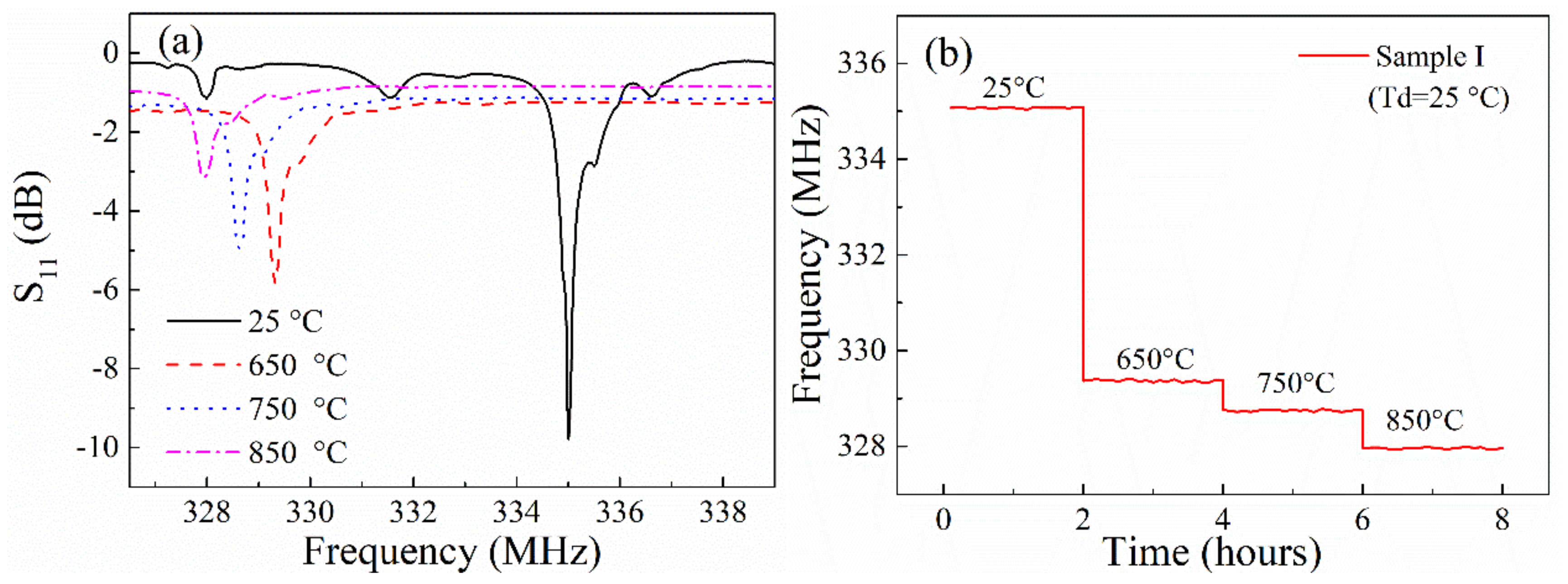
3.2. High Temperature Operation Property and Discussion
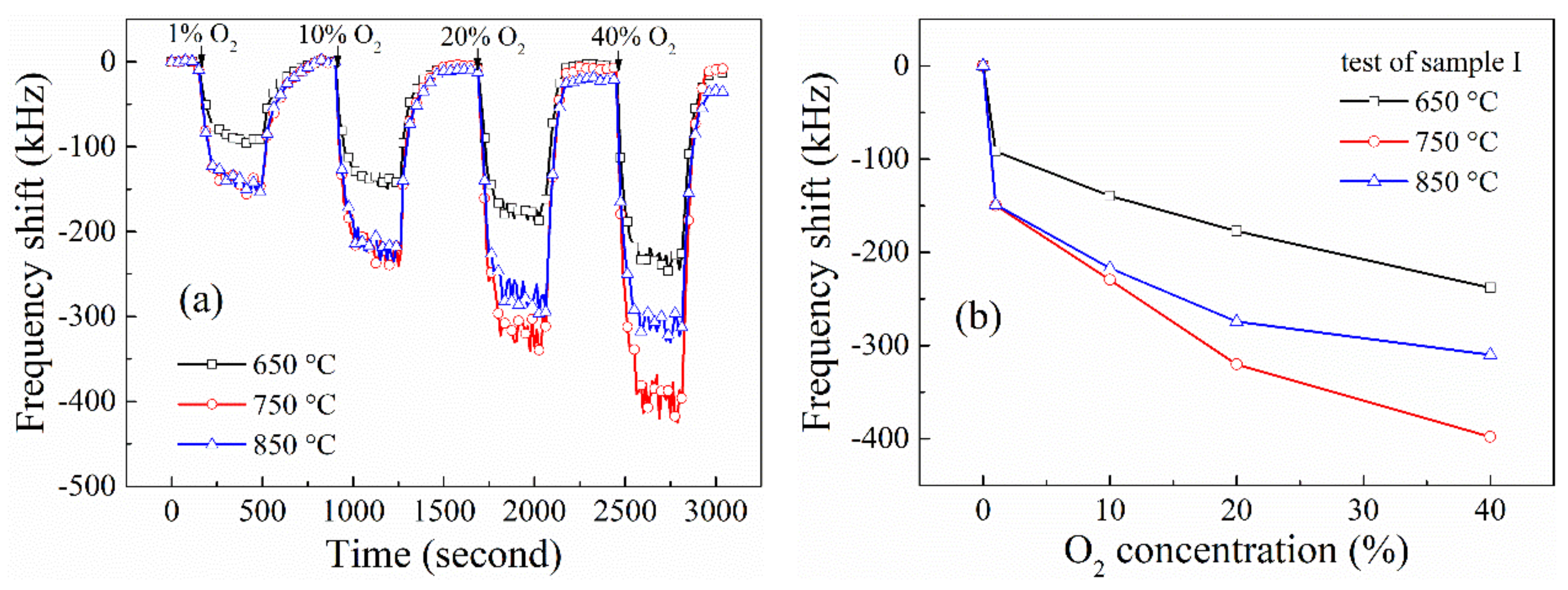
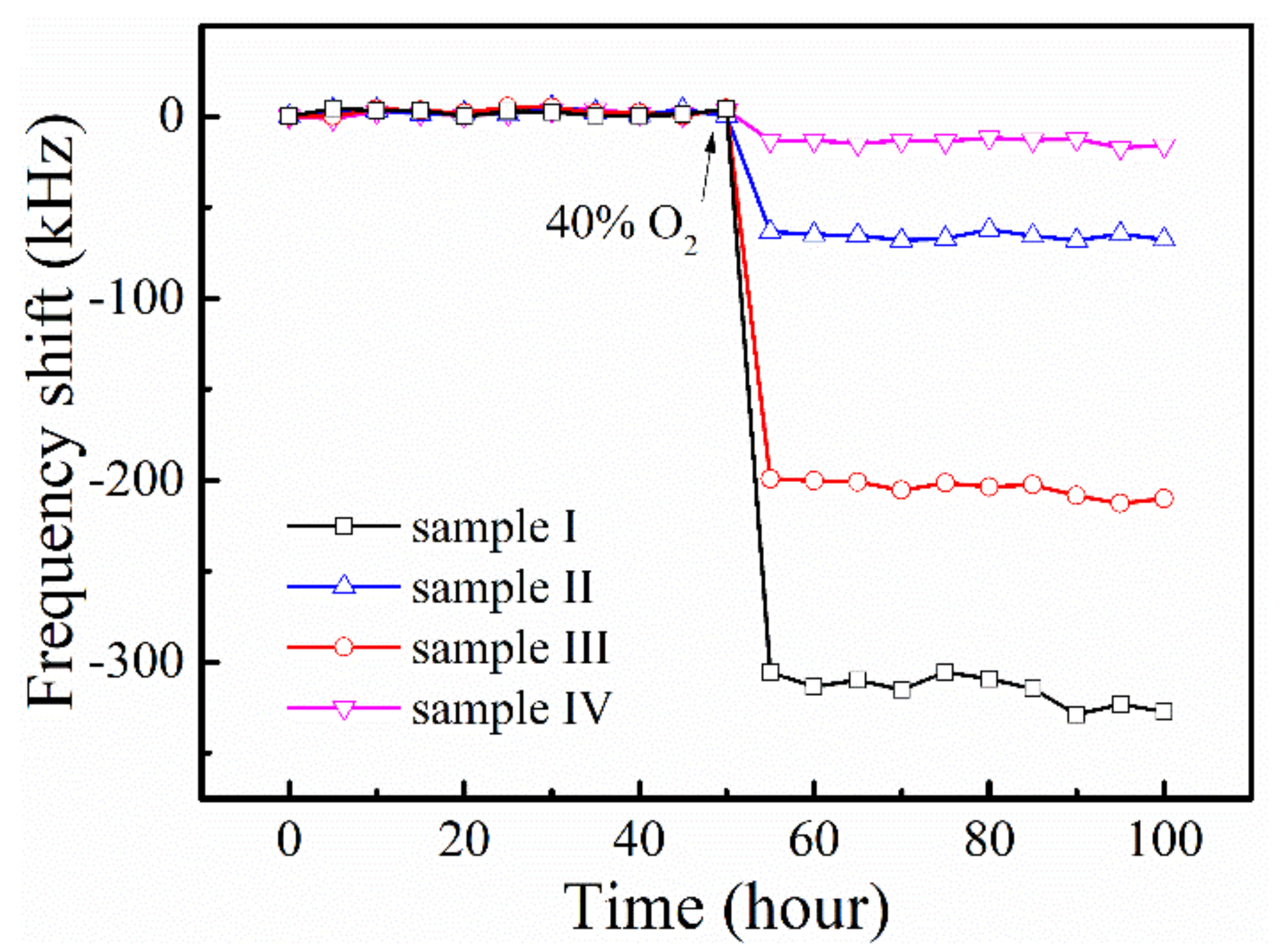
3.3. O2 Gas-Sensing Performance
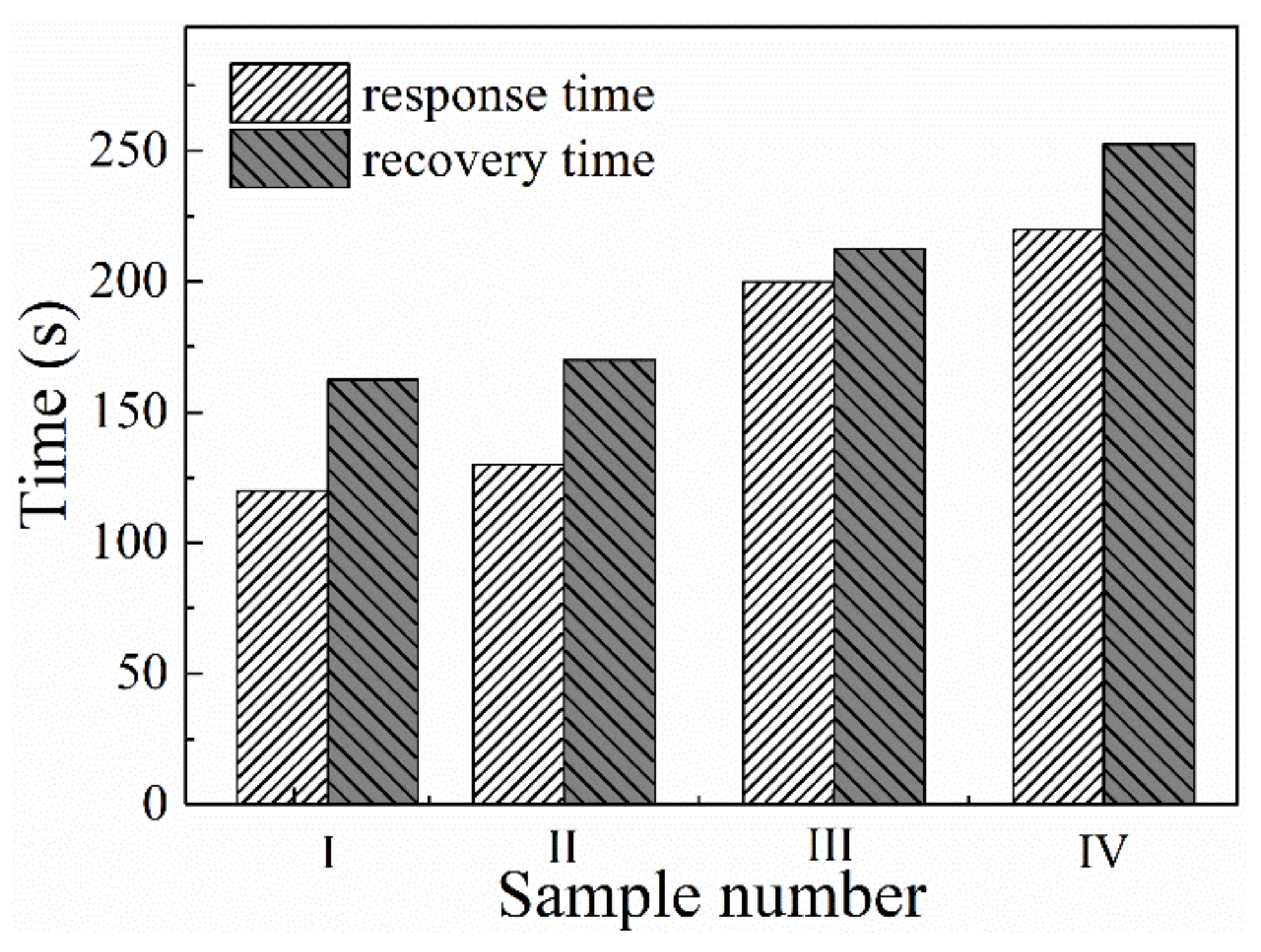
3.4. Stability
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhu, R.; Desroches, M.; Yoon, B.; Swager, T.M. Wireless oxygen sensors enabled by Fe (II)-polymer wrapped carbon nanotubes. ACS Sens. 2017, 2, 1044–1050. [Google Scholar]
- Wang, H.; Wang, J.; Chen, L.; Yao, Y.; Sun, Q.; Qunming, Z. Integrated microoxygen sensor based on nanostructured TiO2 thin films. Micro Nano Lett. 2015, 10, 597–602. [Google Scholar]
- Chaabouni, F.; Abaab, M.; Rezig, B. Metrological characteristics of ZnO oxygen sensor at room temperature. Sens. Actuators B Chem. 2004, 100, 200–204. [Google Scholar] [CrossRef]
- Gębicki, J.; Kloskowski, A.; Chrzanowski, W.; Stepnowski, P.; Namiesnik, J. Application of ionic liquids in amperometric gas sensors. Crit. Rev. Anal. Chem. 2016, 46, 122–138. [Google Scholar]
- Boeker, P. On ‘electronic nose’ methodology. Sens. Actuators B Chem. 2014, 204, 2–17. [Google Scholar]
- Wales, D.J.; Grand, J.; Ting, V.P.; Burke, R.D.; Edler, K.J.; Bowen, C.R.; Mintova, S.; Burrows, A.D. Gas sensing using porous materials for automotive applications. Chem. Soc. Rev. 2015, 44, 4290–4321. [Google Scholar] [CrossRef] [Green Version]
- Jiang, X.; Kim, K.; Zhang, S.; Johnson, J.; Salazar, G. High-temperature piezoelectric sensing. Sensors 2014, 14, 144–169. [Google Scholar]
- Fu, Q.; Wang, J.; Zhou, D.; Luo, W. Passive wireless SAWR sensor system model including the effects of antenna distances. Sens. Actuators A Phys. 2009, 150, 151–155. [Google Scholar] [CrossRef]
- Canabal, A.; Davulis, P.M.; Harris, G.M.; Da Cunha, M.P. High-temperature battery-free wireless microwave acoustic resonator sensor system. Electron. Lett. 2010, 46, 471–472. [Google Scholar]
- Li, C.; Liu, X.; Shu, L.; Li, Y. AlN-based surface acoustic wave resonators for temperature sensing applications. Mater. Express 2015, 5, 367–370. [Google Scholar] [CrossRef]
- Kumar, R.; Al-Dossary, O.; Kumar, G.; Umar, A. Zinc oxide nanostructures for NO 2 gas–sensor applications: A review. Nano-Micro Lett. 2015, 7, 97–120. [Google Scholar] [CrossRef]
- Devkota, J.; Ohodnicki, P.; Greve, D. SAW sensors for chemical vapors and gases. Sensors 2017, 17, 801. [Google Scholar] [CrossRef]
- Afzal, A.; Iqbal, N.; Mujahid, A.; Schirhagl, R. Advanced vapor recognition materials for selective and fast responsive surface acoustic wave sensors: A review. Anal. Chim. Acta 2013, 787, 36–49. [Google Scholar] [CrossRef]
- Wang, H.; Chen, L.; Wang, J.; Sun, Q.; Zhao, Y. A micro oxygen sensor based on a nano sol-gel TiO2 thin film. Sensors 2014, 14, 16423–16433. [Google Scholar] [CrossRef]
- Mhlongo, G.H.; Shingange, K.; Tshabalala, Z.P.; Dhonge, B.P.; Mahmoud, F.A.; Mwakikunga, B.W.; Motaung, D.E. Room temperature ferromagnetism and gas sensing in ZnO nanostructures: Influence of intrinsic defects and Mn, Co, Cu doping. Appl. Surf. Sci. 2016, 390, 804–815. [Google Scholar] [CrossRef]
- Arunraja, L.; Thirumoorthy, P.; Karthik, A.; Rajendran, V.; Edwinpaul, L. EDTA-decorated nanostructured ZnO/CdS thin films for oxygen gas sensing applications. J. Electron. Mater. 2016, 45, 4100–4107. [Google Scholar] [CrossRef]
- Fan, Z.; Wang, D.; Chang, P.C.; Tseng, W.Y.; Lu, J.G. ZnO nanowire field-effect transistor and oxygen sensing property. Appl. Phys. Lett. 2004, 85, 5923–5925. [Google Scholar] [CrossRef] [Green Version]
- Thiele, J.A.; Da Cunha, M.P. High temperature LGS SAW gas sensor. Sens. Actuators B Chem. 2006, 113, 816–822. [Google Scholar] [CrossRef]
- Liu, X.; Peng, B.; Zhang, W.; Zhu, J.; Liu, X.; Wei, M. Improvement of High-Temperature Stability of Al2O3/Pt/ZnO/Al2O3 Film Electrode for SAW Devices by Using Al2O3 Barrier Layer. Materials 2017, 10, 1377. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Peng, B.; Yang, Z.; Wang, R.; Deng, S.; Liu, X. High-temperature SAW wireless strain sensor with langasite. Sensors 2015, 15, 28531–28542. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Kumar, G.; Al-Dossary, O.; Umar, A. ZnO nanostructured thin films: Depositions, properties and applications—A review. Mater. Express 2015, 5, 3–23. [Google Scholar] [CrossRef]
- Bartelt, M.C.; Evans, J.W. Scaling analysis of diffusion-mediated island growth in surface adsorption processes. Phys. Rev. B 1992, 46, 12675. [Google Scholar] [CrossRef]
- Xia, Y.; Xiong, J.; Zhang, F.; Xue, Y.; Wang, L.; Guo, P.; Xu, P.; Zhao, X.; Tao, B. Morphology evolvement of CeO2 cap layer for coated conductors. Appl. Surf. Sci. 2012, 263, 508–512. [Google Scholar] [CrossRef]
- Wang, Z.L. Zinc oxide nanostructures: Growth, properties and applications. J. Phys. Condens. Matter 2004, 16, R829. [Google Scholar] [CrossRef]
- Gu, F.; You, D.; Wang, Z.; Han, D.; Guo, G. Improvement of gas-sensing property by defect engineering in microwave-assisted synthesized 3D ZnO nanostructures. Sens. Actuators B Chem. 2014, 204, 342–350. [Google Scholar] [CrossRef]
- Vakulov, Z.E.; Zamburg, E.G.; Khakhulin, D.A.; Ageev, O.A. Thermal stability of ZnO thin films fabricated by pulsed laser deposition. Mater. Sci. Semicond. Process. 2017, 66, 21–25. [Google Scholar] [CrossRef]


| Sample Number | Td (°C) | tZnO (nm) | *FWHM of ZnO Film (°) |
|---|---|---|---|
| I | 25 | 204.5 | 10.2 |
| II | 200 | 202.6 | 6.4 |
| III | 400 | 195.8 | 3.7 |
| IV | 600 | 200.2 | 2.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shu, L.; Wang, X.; Yan, D.; Fan, L.; Wu, W. The Investigation of High-Temperature SAW Oxygen Sensor Based on ZnO Films. Materials 2019, 12, 1235. https://doi.org/10.3390/ma12081235
Shu L, Wang X, Yan D, Fan L, Wu W. The Investigation of High-Temperature SAW Oxygen Sensor Based on ZnO Films. Materials. 2019; 12(8):1235. https://doi.org/10.3390/ma12081235
Chicago/Turabian StyleShu, Lin, Xuemin Wang, Dawei Yan, Long Fan, and Weidong Wu. 2019. "The Investigation of High-Temperature SAW Oxygen Sensor Based on ZnO Films" Materials 12, no. 8: 1235. https://doi.org/10.3390/ma12081235
APA StyleShu, L., Wang, X., Yan, D., Fan, L., & Wu, W. (2019). The Investigation of High-Temperature SAW Oxygen Sensor Based on ZnO Films. Materials, 12(8), 1235. https://doi.org/10.3390/ma12081235





