Highly Transparent, Flexible and Conductive CNF/AgNW Paper for Paper Electronics
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of TEMPO-oxidized CNFs
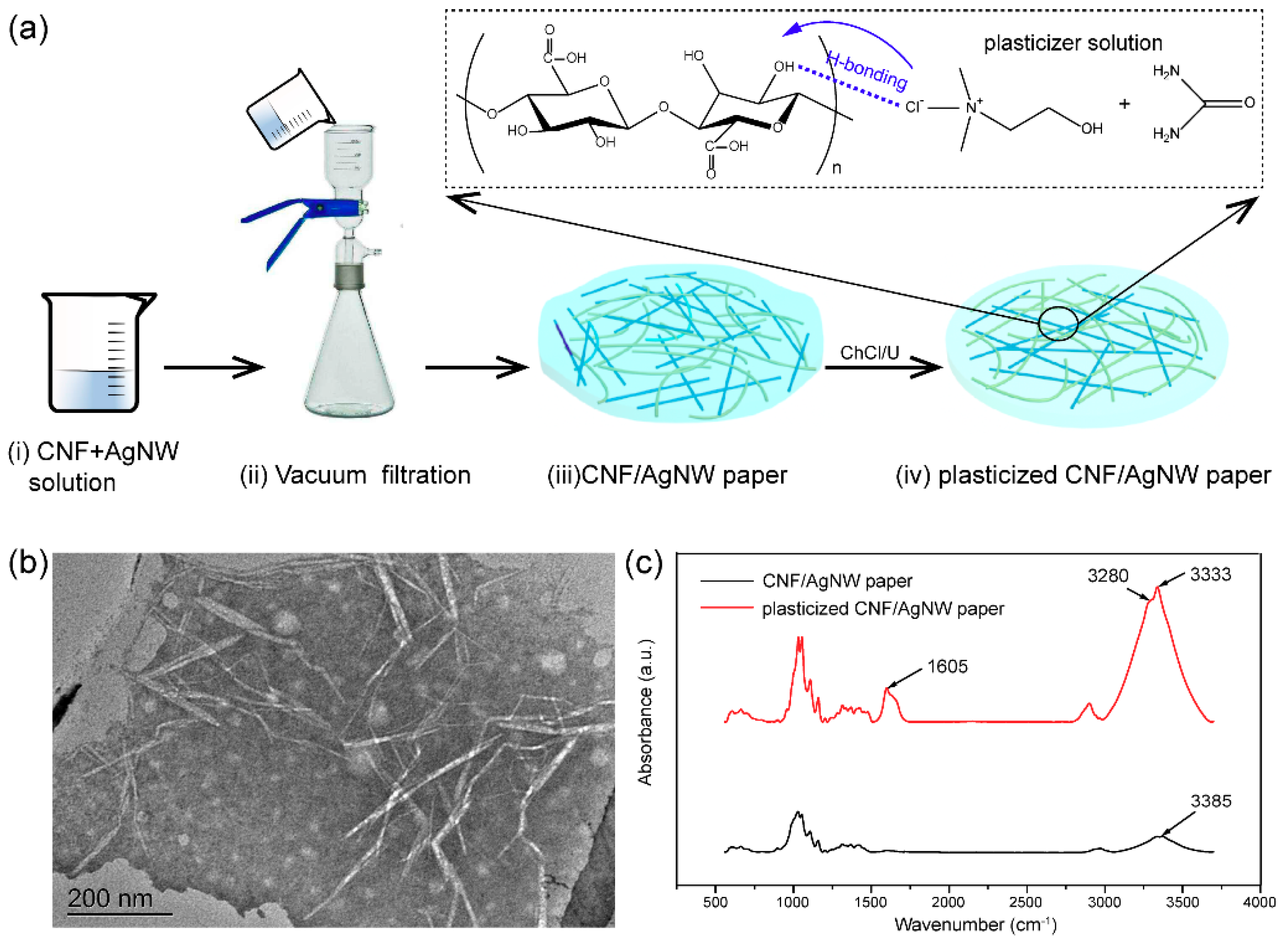
2.3. Fabrication of Conductive Paper
3. Characterizations
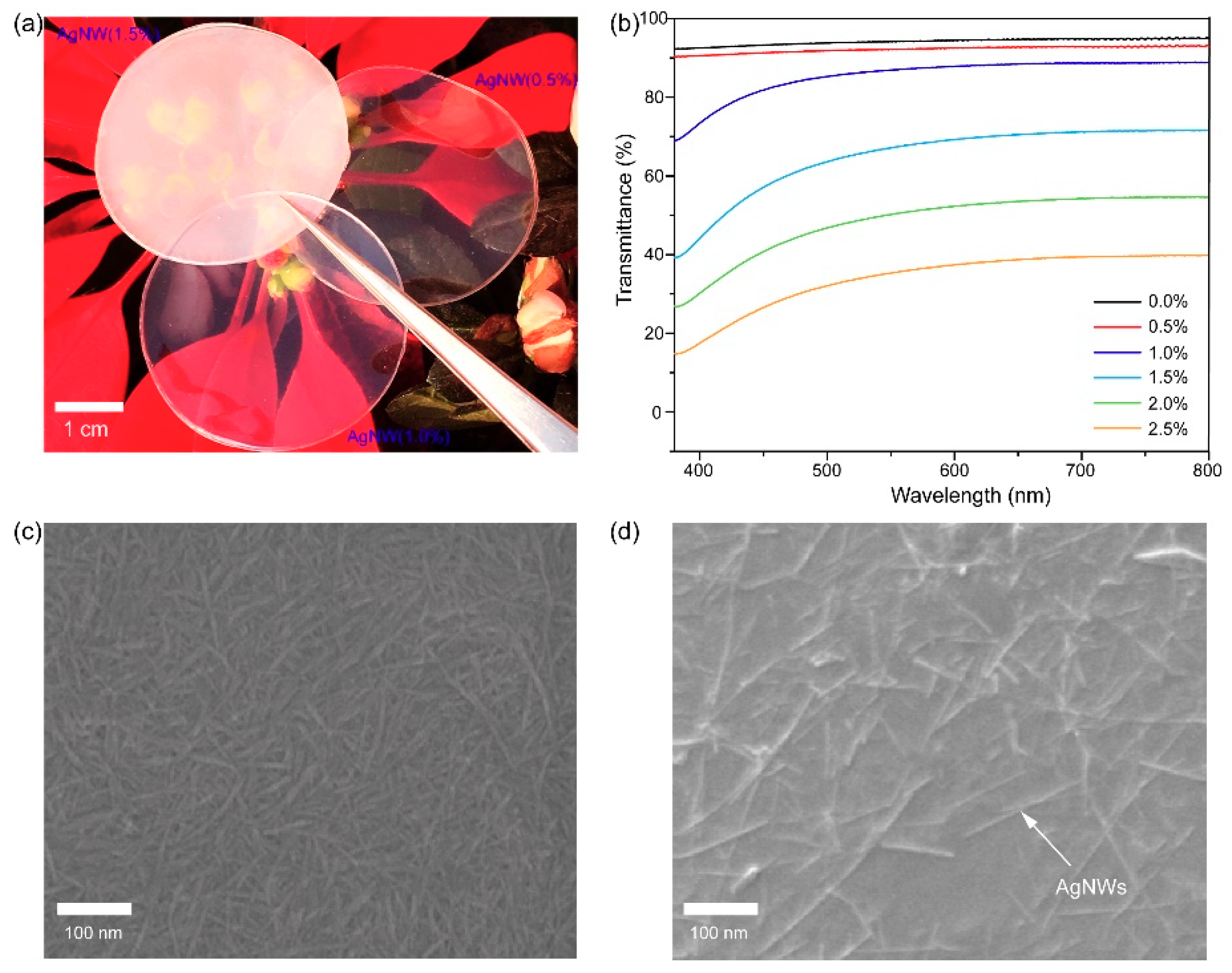
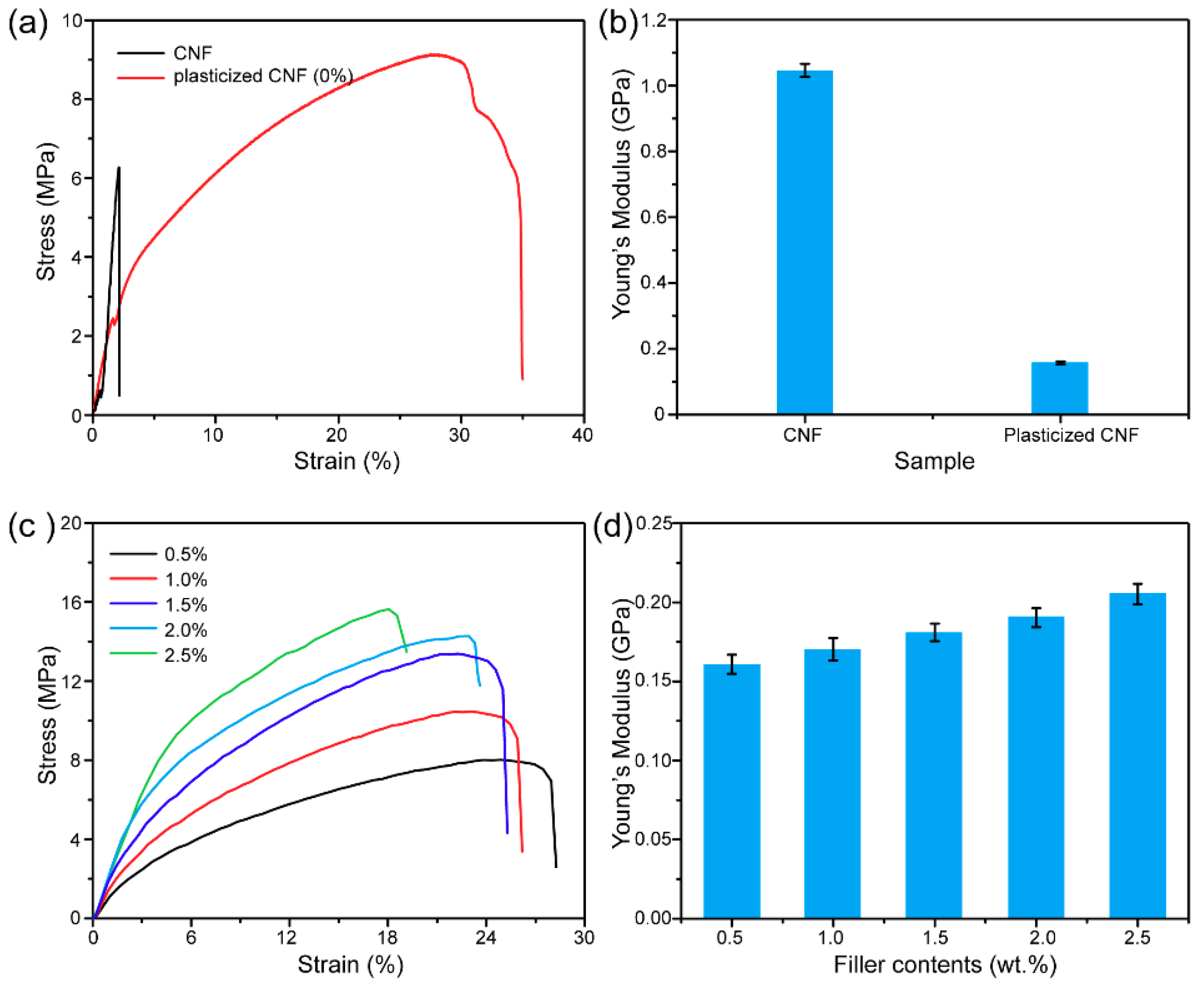
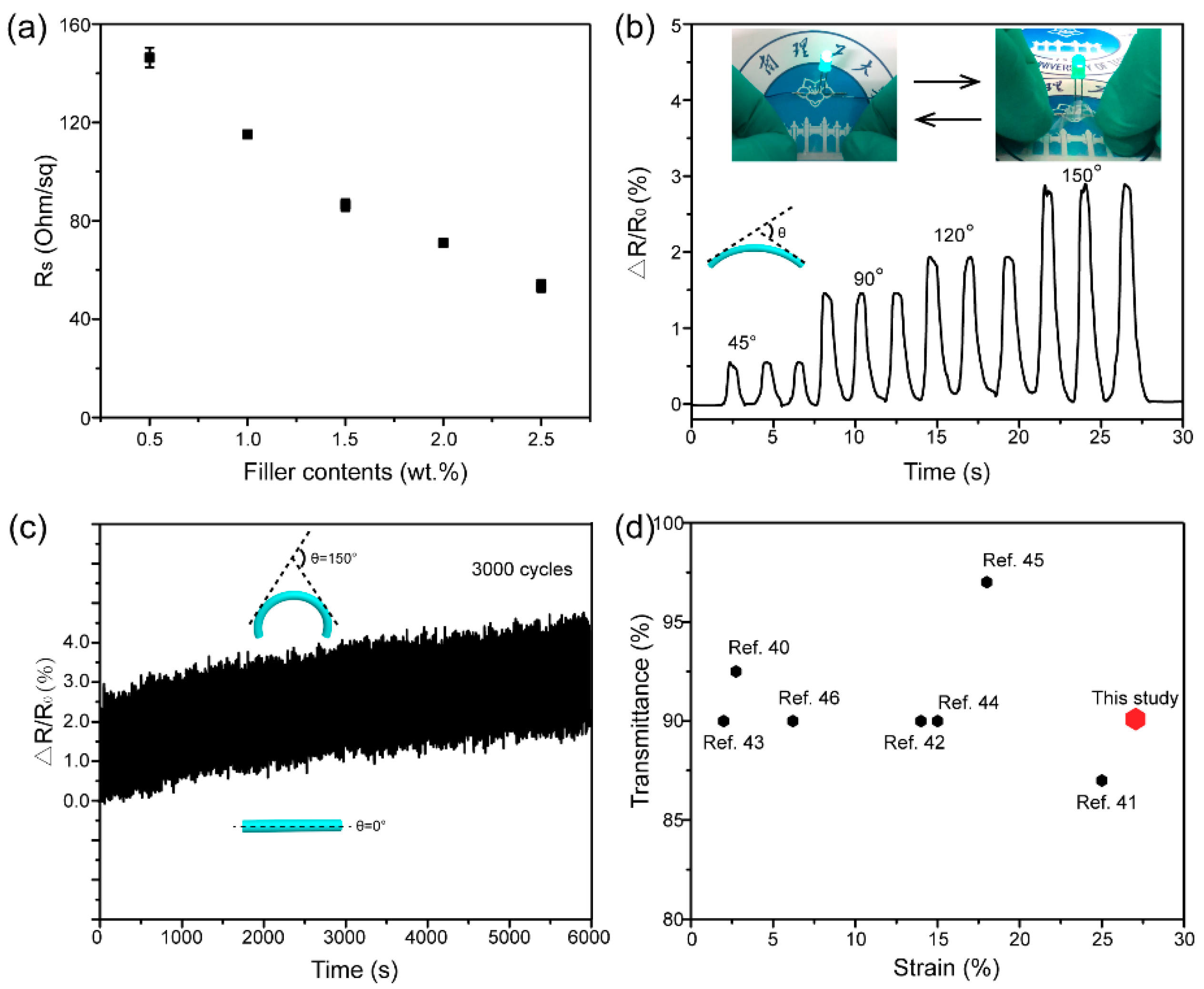
4. Results
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tobjörk, D.; Österbacka, R. Paper electronics. Adv. Mater. 2011, 23, 1935–1961. [Google Scholar] [CrossRef] [PubMed]
- Yao, B.; Jing, Z.; Kou, T.; Yu, S.; Liu, T.; Li, Y. Paper-Based Electrodes for Flexible Energy Storage Devices. Adv. Sci. 2017, 4, 1700107. [Google Scholar] [CrossRef] [PubMed]
- Compton, O.C.; Dikin, D.A.; Putz, K.W.; Brinson, L.C.; Nguyen, S.T. Electrically conductive “alkylated” graphene paper via chemical reduction of amine-functionalized graphene oxide paper. Adv. Mater. 2010, 22, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Shao, Z.; Wu, X.; Wang, X.; Li, J.; Zhang, Y.; Wang, W.; Wang, F. Cellulose nanofibers/reduced graphene oxide flexible transparent conductive paper. Carbohydr. Polym. 2013, 97, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Hyun, W.J.; Park, O.O.; Chin, B.D. Foldable graphene electronic circuits based on paper substrates. Adv. Mater. 2013, 25, 4729–4734. [Google Scholar] [CrossRef]
- Jabbour, L.; Gerbaldi, C.; Chaussy, D.; Zeno, E.; Bodoardo, S.; Beneventi, D. Microfibrillated cellulose–graphite nanocomposites for highly flexible paper-like Li-ion battery electrodes. J. Mater. Chem. 2010, 20, 7344–7347. [Google Scholar] [CrossRef]
- Weng, Z.; Su, Y.; Wang, D.W.; Li, F.; Du, J.; Cheng, H.M. Graphene–cellulose paper flexible supercapacitors. Adv. Energy Mater. 2011, 1, 917–922. [Google Scholar] [CrossRef]
- Huang, L.; Chen, D.; Ding, Y.; Feng, S.; Wang, Z.L.; Liu, M. Nickel–cobalt hydroxide nanosheets coated on NiCo2O4 nanowires grown on carbon fiber paper for high-performance pseudocapacitors. Nano Lett. 2013, 13, 3135–3139. [Google Scholar] [CrossRef]
- Huang, L.; Chen, D.; Ding, Y.; Wang, Z.L.; Zeng, Z.; Liu, M. Hybrid composite Ni (OH)2@ NiCo2O4 grown on carbon fiber paper for high-performance supercapacitors. ACS Appl. Mater. Interfaces 2013, 5, 11159–11162. [Google Scholar] [CrossRef]
- Xiao, J.; Wan, L.; Yang, S.; Xiao, F.; Wang, S. Design hierarchical electrodes with highly conductive NiCo2S4 nanotube arrays grown on carbon fiber paper for high-performance pseudocapacitors. Nano Lett. 2014, 14, 831–838. [Google Scholar] [CrossRef]
- Yang, L.; Cheng, S.; Ding, Y.; Zhu, X.; Wang, Z.L.; Liu, M. Hierarchical Network Architectures of Carbon Fiber Paper Supported Cobalt Oxide Nanonet for High-Capacity Pseudocapacitors. Nano Lett. 2012, 12, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Huang, Y.; Zhang, Z.; Duan, D.; Hao, X.; Liu, S. Preparation and structural evolution of well aligned-carbon nanotube arrays onto conductive carbon-black layer/carbon paper substrate with enhanced discharge capacity for Li–air batteries. Chem. Eng. J. 2016, 283, 911–921. [Google Scholar] [CrossRef]
- Tang, Y.; Mosseler, J.A.; He, Z.; Ni, Y. Imparting Cellulosic Paper of High Conductivity by Surface Coating of Dispersed Graphite. Ind. Eng. Chem. Res. 2014, 53, 10119–10124. [Google Scholar] [CrossRef]
- Johnston, J.H.; Kelly, F.M.; Moraes, J.; Borrmann, T.; Flynn, D. Conducting polymer composites with cellulose and protein fibres. Curr. Appl. Phys. 2006, 6, 587–590. [Google Scholar] [CrossRef]
- Kelly, F.M.; Johnston, J.H.; Borrmann, T.; Richardson, M.J. Functionalised hybrid materials of conducting polymers with individual fibres of cellulose. Eur. J. Inorg. Chem. 2007, 2007, 5571–5577. [Google Scholar] [CrossRef]
- Lou, B.; Chen, C.; Zhou, Z.; Zhang, L.; Wang, E.; Dong, S. A novel electrochemical sensing platform for anions based on conducting polymer film modified electrodes integrated on paper-based chips. Talanta 2013, 105, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Youssef, A.M.; El-Samahy, M.A.; Rehim, M.H.A. Preparation of conductive paper composites based on natural cellulosic fibers for packaging applications. Carbohydr. Polym. 2012, 89, 1027–1032. [Google Scholar] [CrossRef]
- Russo, A.; Ahn, B.Y.; Adams, J.J.; Duoss, E.B.; Bernhard, J.T.; Lewis, J.A. Pen-on-paper flexible electronics. Adv. Mater. 2011, 23, 3426–3430. [Google Scholar] [CrossRef]
- Cho, S.J.; Choi, K.H.; Yoo, J.T.; Kim, J.H.; Lee, Y.H.; Chun, S.J.; Park, S.B.; Choi, D.H.; Wu, Q.; Lee, S.Y. Hetero-Nanonet Rechargeable Paper Batteries: Toward Ultrahigh Energy Density and Origami Foldability. Adv. Funct. Mater. 2015, 25, 6029–6040. [Google Scholar] [CrossRef]
- Hu, L.; Wu, H.; La Mantia, F.; Yang, Y.; Cui, Y. Thin, flexible secondary Li-ion paper batteries. ACS Nano 2010, 4, 5843–5848. [Google Scholar] [CrossRef]
- Grau, G.; Kitsomboonloha, R.; Swisher, S.L.; Kang, H.; Subramanian, V. Printed transistors on paper: Towards smart consumer product packaging. Adv. Funct. Mater. 2014, 24, 5067–5074. [Google Scholar] [CrossRef]
- Hu, L.; Zheng, G.; Yao, J.; Liu, N.; Weil, B.; Eskilsson, M.; Karabulut, E.; Ruan, Z.; Fan, S.; Bloking, J.T. Transparent and conductive paper from nanocellulose fibers. Energy Environ. Sci. 2013, 6, 513–518. [Google Scholar] [CrossRef]
- Mirica, K.A.; Weis, J.G.; Schnorr, J.M.; Esser, B.; Swager, T.M. Mechanical drawing of gas sensors on paper. Angew. Chem. Int. Ed. 2012, 51, 10740–10745. [Google Scholar] [CrossRef] [PubMed]
- Breer, H. Olfactory receptors: Molecular basis for recognition and discrimination of odors. Anal. Bioanal. Chem. 2003, 377, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Di Pietrantonio, F.; Benetti, M.; Cannatà, D.; Verona, E.; Palla-Papavlu, A.; Fernández-Pradas, J.M.; Serra, P.; Staiano, M.; Varriale, A.; D’Auria, S. A surface acoustic wave bio-electronic nose for detection of volatile odorant molecules. Biosens. Bioelectron. 2015, 67, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Park, T.H. Recent advances in the development of bioelectronic nose. Biotechnol. Bioprocess Eng. 2010, 15, 22–29. [Google Scholar] [CrossRef]
- Wasilewski, T.; Gębicki, J.; Kamysz, W. Advances in olfaction-inspired biomaterials applied to bioelectronic noses. Sens. Actuators B Chem. 2018, 257, 511–537. [Google Scholar] [CrossRef]
- Hamedi, M.M.; Campbell, V.E.; Rothemund, P.; Güder, F.; Christodouleas, D.C.; Bloch, J.F.; Whitesides, G.M. Electrically Activated Paper Actuators. Adv. Funct. Mater. 2016, 26, 2446–2453. [Google Scholar] [CrossRef]
- Nyholm, L.; Nyström, G.; Mihranyan, A.; Strømme, M. Toward flexible polymer and paper-based energy storage devices. Adv. Mater. 2011, 23, 3751–3769. [Google Scholar] [CrossRef]
- He, M.; Zhang, K.; Chen, G.; Tian, J.; Su, B. Ionic gel paper with long-term bendable electrical robustness for use in flexible electroluminescent devices. ACS Appl. Mater. Interfaces 2017, 9, 16466–16473. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2002, 9, 70–71. [Google Scholar]
- Abbott, A.P.; Ballantyne, A.D.; Conde, J.P.; Ryder, K.S.; Wise, W.R. Salt modified starch: Sustainable, recyclable plastics. Green Chem. 2012, 14, 1302–1307. [Google Scholar] [CrossRef]
- Ramesh, S.; Shanti, R.; Morris, E. Studies on the plasticization efficiency of deep eutectic solvent in suppressing the crystallinity of corn starch based polymer electrolytes. Carbohydr. Polym. 2012, 87, 701–706. [Google Scholar] [CrossRef]
- Okita, Y.; Saito, T.; Isogai, A. Entire Surface Oxidation of Various Cellulose Microfibrils by TEMPO-Mediated Oxidation. Biomacromolecules 2010, 11, 1696–1700. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Kimura, S.; Nishiyama, Y.; Isogai, A. Cellulose Nanofibers Prepared by TEMPO-Mediated Oxidation of Native Cellulose. Biomacromolecules 2007, 8, 2485–2491. [Google Scholar] [CrossRef]
- Saito, T.; Nishiyama, Y.; Putaux, J.-L.; Vignon, M.; Isogai, A. Homogeneous Suspensions of Individualized Microfibrils from TEMPO-Catalyzed Oxidation of Native Cellulose. Biomacromolecules 2006, 7, 1687–1691. [Google Scholar] [CrossRef]
- Wang, S.; Peng, X.; Zhong, L.; Jing, S.; Cao, X.; Lu, F.; Sun, R. Choline chloride/urea as an effective plasticizer for production of cellulose films. Carbohydr. Polym. 2015, 117, 133–139. [Google Scholar] [CrossRef]
- Xiao, F.; Song, J.; Gao, H.; Zan, X.; Xu, R.; Duan, H. Coating graphene paper with 2D-assembly of electrocatalytic nanoparticles: A modular approach toward high-performance flexible electrodes. ACS Nano 2012, 6, 100–110. [Google Scholar] [CrossRef]
- Hu, L.; Choi, J.W.; Yang, Y.; Jeong, S.; La Mantia, F.; Cui, L.; Cui, Y. Highly conductive paper for energy-storage devices. Proc. Natl. Acad. Sci. USA 2009, 106, 21490–21494. [Google Scholar] [CrossRef]
- Fang, Z.; Zhu, H.; Yuan, Y.; Ha, D.; Zhu, S.; Preston, C.; Chen, Q.; Li, Y.; Han, X.; Lee, S.; et al. Novel Nanostructured Paper with Ultrahigh Transparency and Ultrahigh Haze for Solar Cells. Nano Lett. 2014, 14, 765–773. [Google Scholar] [CrossRef]
- Gao, X.; Huang, L.; Wang, B.; Xu, D.; Zhong, J.; Hu, Z.; Zhang, L.; Zhou, J. Natural Materials Assembled, Biodegradable, and Transparent Paper-Based Electret Nanogenerator. ACS Appl. Mater. Interfaces 2016, 8, 35587–35592. [Google Scholar] [CrossRef]
- Ji, S.; Hyun, B.G.; Kim, K.; Lee, S.Y.; Kim, S.-H.; Kim, J.-Y.; Song, M.H.; Park, J.-U. Photo-patternable and transparent films using cellulose nanofibers for stretchable origami electronics. NPG Asia Mater. 2016, 8, e299. [Google Scholar] [CrossRef]
- Kang, W.; Yan, C.; Foo, C.Y.; Lee, P.S. Foldable Electrochromics Enabled by Nanopaper Transfer Method. Adv. Funct. Mater. 2015, 25, 4203–4210. [Google Scholar] [CrossRef]
- Nogi, M.; Yano, H. Transparent Nanocomposites Based on Cellulose Produced by Bacteria Offer Potential Innovation in the Electronics Device Industry. Adv. Mater. 2008, 20, 1849–1852. [Google Scholar] [CrossRef]
- Xiong, J.; Li, S.; Ye, Y.; Wang, J.; Qian, K.; Cui, P.; Gao, D.; Lin, M.-F.; Chen, T.; Lee, P.S. A Deformable and Highly Robust Ethyl Cellulose Transparent Conductor with a Scalable Silver Nanowires Bundle Micromesh. Adv. Mater. 2018, 30, 1802803. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Fang, Z.; Wang, Z.; Dai, J.; Yao, Y.; Shen, F.; Preston, C.; Wu, W.; Peng, P.; Jang, N.; et al. Extreme Light Management in Mesoporous Wood Cellulose Paper for Optoelectronics. ACS Nano 2016, 10, 1369–1377. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, R.; Zhang, K.; Chen, G. Highly Transparent, Flexible and Conductive CNF/AgNW Paper for Paper Electronics. Materials 2019, 12, 322. https://doi.org/10.3390/ma12020322
Li R, Zhang K, Chen G. Highly Transparent, Flexible and Conductive CNF/AgNW Paper for Paper Electronics. Materials. 2019; 12(2):322. https://doi.org/10.3390/ma12020322
Chicago/Turabian StyleLi, Ren’ai, Kaili Zhang, and Guangxue Chen. 2019. "Highly Transparent, Flexible and Conductive CNF/AgNW Paper for Paper Electronics" Materials 12, no. 2: 322. https://doi.org/10.3390/ma12020322
APA StyleLi, R., Zhang, K., & Chen, G. (2019). Highly Transparent, Flexible and Conductive CNF/AgNW Paper for Paper Electronics. Materials, 12(2), 322. https://doi.org/10.3390/ma12020322




