Optimization of the SiC Powder Source Material for Improved Process Conditions During PVT Growth of SiC Boules
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of the SiC Source Material
2.2. Crystal Growth of SiC Boules
2.3. In Situ Visulaization of the Growth Process
2.4. Characterization Methods
3. Results and Discussion
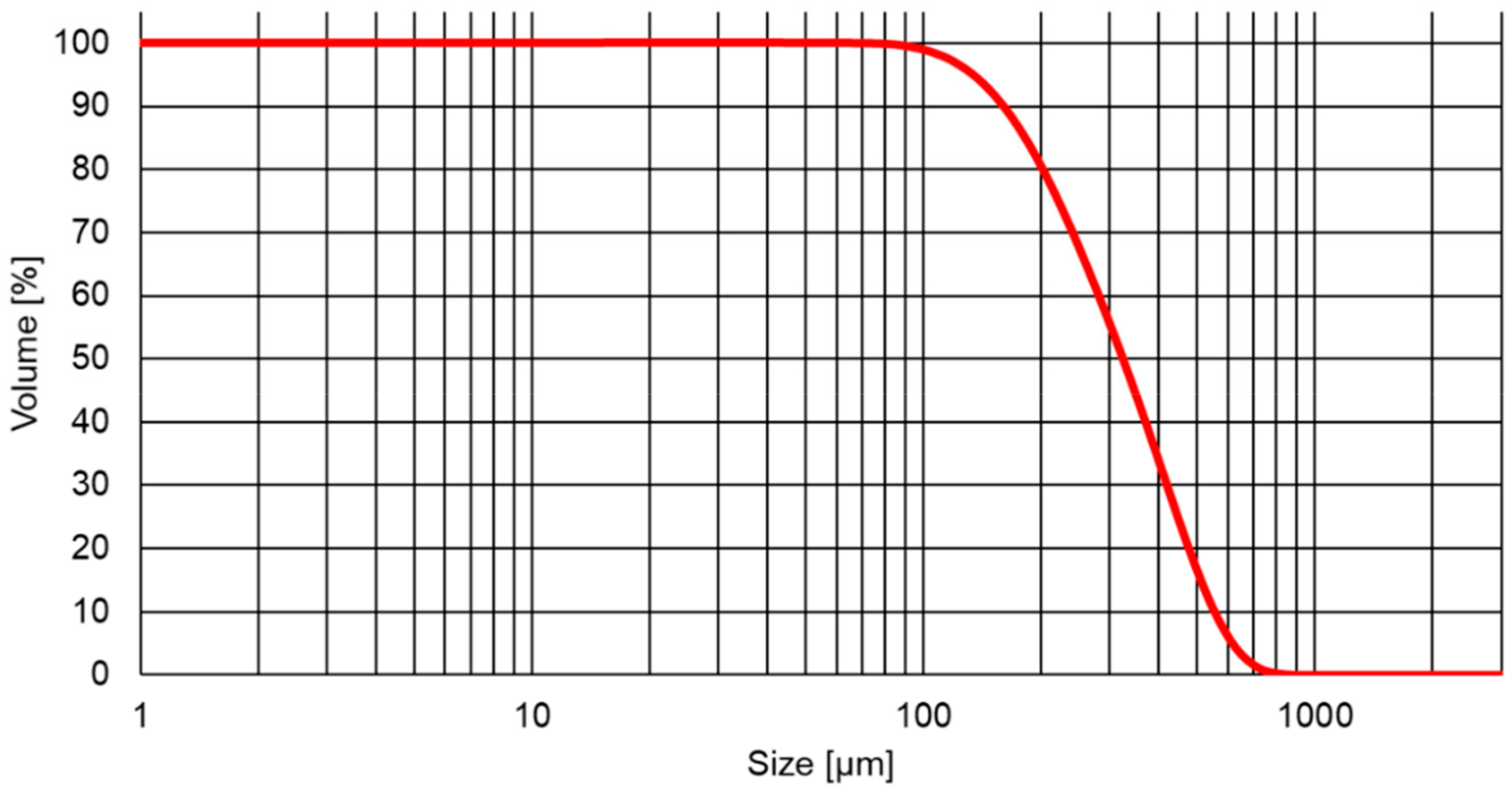
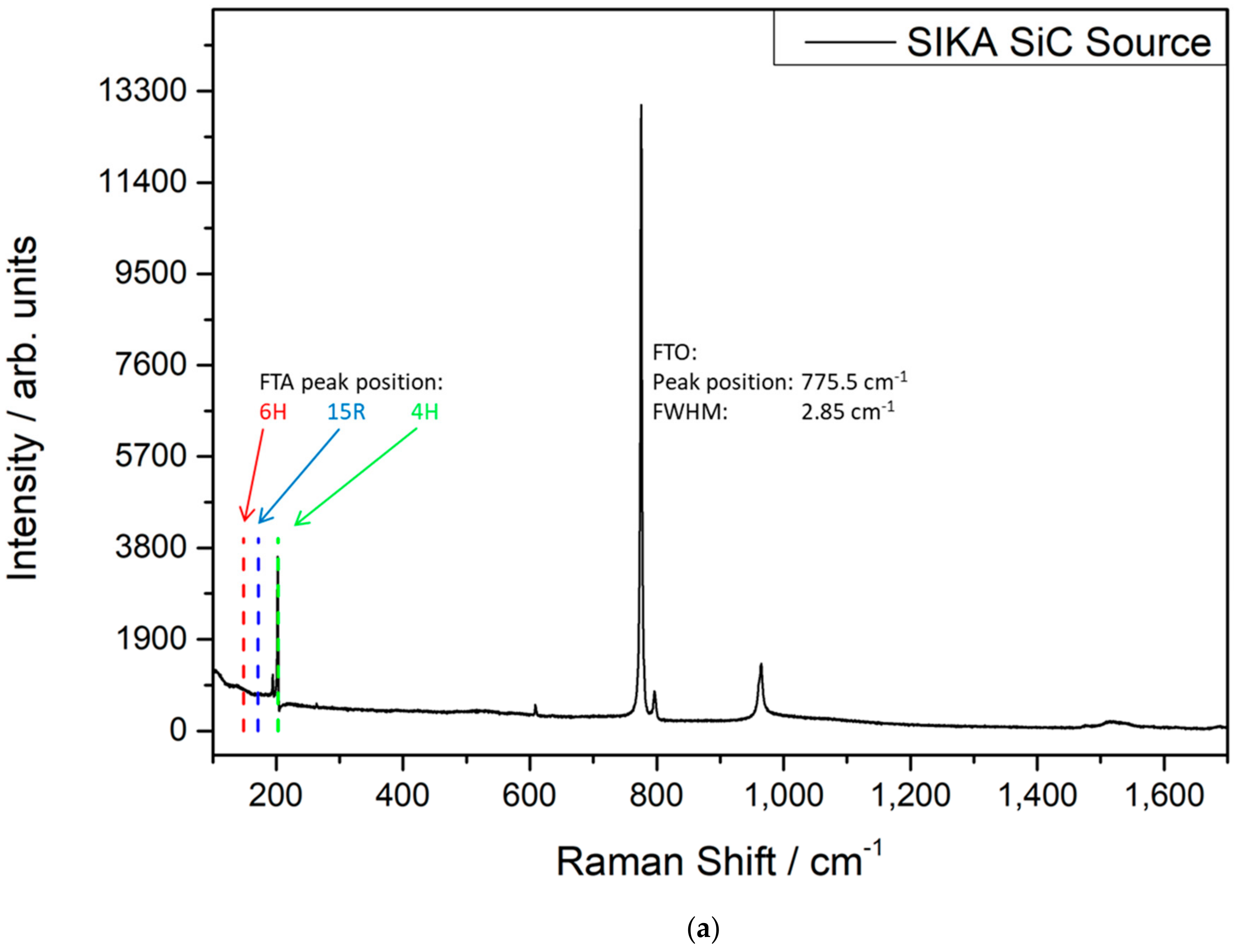
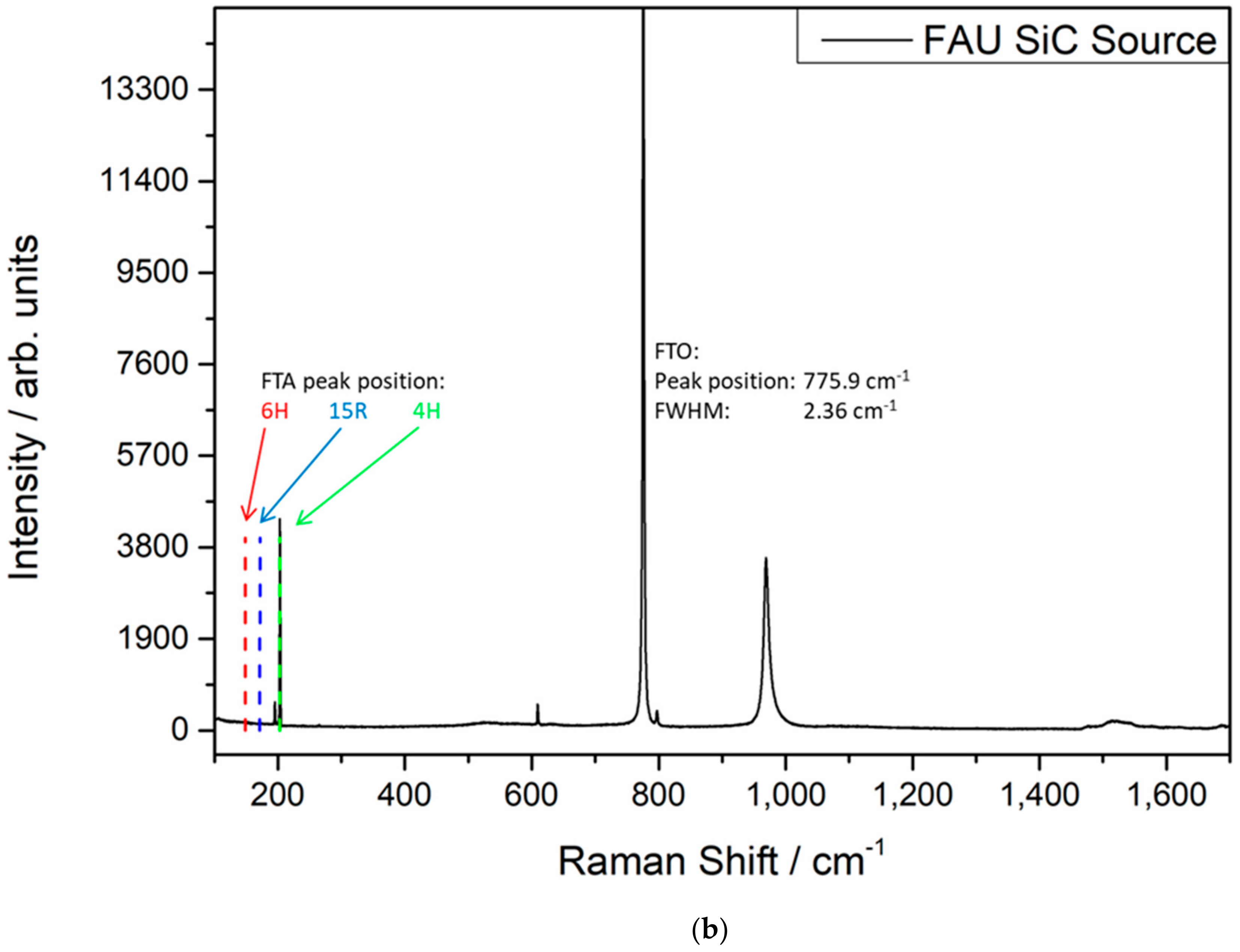
3.1. Properties of the SiC Powder Sources
3.2. Sublimation Behavior of the SiC Powder
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Wellmann, P.J. Review of SiC materials technology. Semicond. Sci. Technol. 2018, 33, 103001. [Google Scholar] [CrossRef]
- Ziegler, G.; Lanig, P.; Theis, D.; Weyerich, C. Single crystal growth of SiC substrate material for blue light emitting diodes. IEEE Trans. Electron. Dev. 1983, 30, 277–281. [Google Scholar] [CrossRef]
- Tairov, Y.M.; Tsvetkov, V.F. Progress in controlling the growth of polytypic crystals. Prog. Cryst. Growth Charact. 1983, 7, 111–163. [Google Scholar] [CrossRef]
- Tairov, Y.M. Growth of bulk SiC. Mater. Sci. Eng. B 1995, 29, 83–89. [Google Scholar]
- Dorozhkin, S.I.; Avrov, D.D.; Rastegaev, V.P.; Tairov, Y.M. Growth of SiC ingots with high rate. Mater. Sci. Eng. B 1997, 46, 296–299. [Google Scholar]
- Augustine, G.; Balakrishna, V.; Brandt, C.D. Growth and characterization of high-purity SiC single crystals. J. Cryst. Growth 2000, 211, 339–342. [Google Scholar] [CrossRef]
- Hobgood, H.M.; Barrett, D.L.; McHugh, J.P.; Clarke, R.C.; Sriram, S.; Burk, A.A.; Greggi, J.; Brandt, C.D.; Hopkins, R.H.; Choyke, W.J. Large diameter 6H-SiC for microwave device applications. J. Cryst. Growth 1994, 137, 181–186. [Google Scholar] [CrossRef]
- Arzig, M.; Hsiao, T.C.; Wellmann, P.J. Optimization of the SiC powder source size distribution for the sublimation growth of long crystal boules. Adv. Mater. Proc. 2018, 3, 540–543. [Google Scholar]
- Wellmann, P.J.; Bickermann, M.; Hofmann, D.; Kadinski, L.; Selder, M.; Straubinger, T.L.; Winnacker, A. In situ visualization and analysis of silicon carbide physical vapor transport growth using digital X-ray imaging. J. Cryst. Growth 2000, 216, 263–272. [Google Scholar] [CrossRef]
- Wellmann, P.J.; Hofmann, D.; Kadinski, L.; Selder, M.; Straubinger, T.L.; Winnacker, A. Impact of source material on silicon carbide vapor transport growth process. J. Cryst. Growth 2001, 225, 312–316. [Google Scholar] [CrossRef]
- Wellmann, P.; Herro, Z.; Winnacker, A.; Püsche, R.; Hundhausen, M.; Kulik, A.; Bogdanov, M.; Karpov, S.; Ramm, M.; Makarov, Y.; et al. In situ visualization of SiC physical vapor transport crystal growth. J. Cryst. Growth 2005, 275, e1807–e1812. [Google Scholar] [CrossRef]
- Wellmann, P.; Neubauer, G.; Fahlbusch, L.; Salamon, M.; Uhlmann, N. Growth of SiC bulk crystals for application in power electronic devices—Process design, 2D and 3D X-ray in situ visualization and advanced doping. Cryst. Res. Technol. 2015, 50, 2–9. [Google Scholar] [CrossRef]
- Sakwe, S.A.; Herro, Z.G.; Wellmann, P.J. Development of a KOH Defect Etching Furnace with Absolute In-Situ Temperature Measurement Capability. Mater. Sci. Forum 2005, 483–485, 283. [Google Scholar] [CrossRef]
- Sakwe, S.A.; Müller, R.; Wellmann, P.J. Optimization of KOH etching parameters for quantitative defect recognition in n- and p-type doped SiC. J. Cryst. Growth 2006, 289, 520–526. [Google Scholar] [CrossRef]




| Property | Unit | Value |
|---|---|---|
| LPD | g/cm3 | 1.60 |
| Free C | % wt | 0.01 |
| Total oxygen | % wt | 0.02 |
| Element | Concentration (ppm wt) |
|---|---|
| Al | 5.9 |
| B | 0.23 |
| Ba | 0.05 |
| C | Matrix |
| Ca | 0.67 |
| Cl | 1.5 |
| Cr | 0.41 |
| Cu | 0.18 |
| F | <0.1 |
| Fe | 1.3 |
| In | Binder |
| Na | 0.62 |
| Ni | 0.33 |
| P | 0.13 |
| Si | Matrix |
| S | 1.6 |
| Ti | 2.8 |
| V | 0.19 |
| Zn | 0.45 |
| Zr | 0.22 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ellefsen, O.M.; Arzig, M.; Steiner, J.; Wellmann, P.; Runde, P. Optimization of the SiC Powder Source Material for Improved Process Conditions During PVT Growth of SiC Boules. Materials 2019, 12, 3272. https://doi.org/10.3390/ma12193272
Ellefsen OM, Arzig M, Steiner J, Wellmann P, Runde P. Optimization of the SiC Powder Source Material for Improved Process Conditions During PVT Growth of SiC Boules. Materials. 2019; 12(19):3272. https://doi.org/10.3390/ma12193272
Chicago/Turabian StyleEllefsen, Oda Marie, Matthias Arzig, Johannes Steiner, Peter Wellmann, and Pål Runde. 2019. "Optimization of the SiC Powder Source Material for Improved Process Conditions During PVT Growth of SiC Boules" Materials 12, no. 19: 3272. https://doi.org/10.3390/ma12193272
APA StyleEllefsen, O. M., Arzig, M., Steiner, J., Wellmann, P., & Runde, P. (2019). Optimization of the SiC Powder Source Material for Improved Process Conditions During PVT Growth of SiC Boules. Materials, 12(19), 3272. https://doi.org/10.3390/ma12193272





