Study of the Structure and Antimicrobial Activity of Ca-Deficient Ceramics on Chlorhexidine Nanoclay Substrate
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Modifications and Preparation of Samples
2.3. Analytical Methods
2.4. Antibacterial Test
3. Results and Discussions
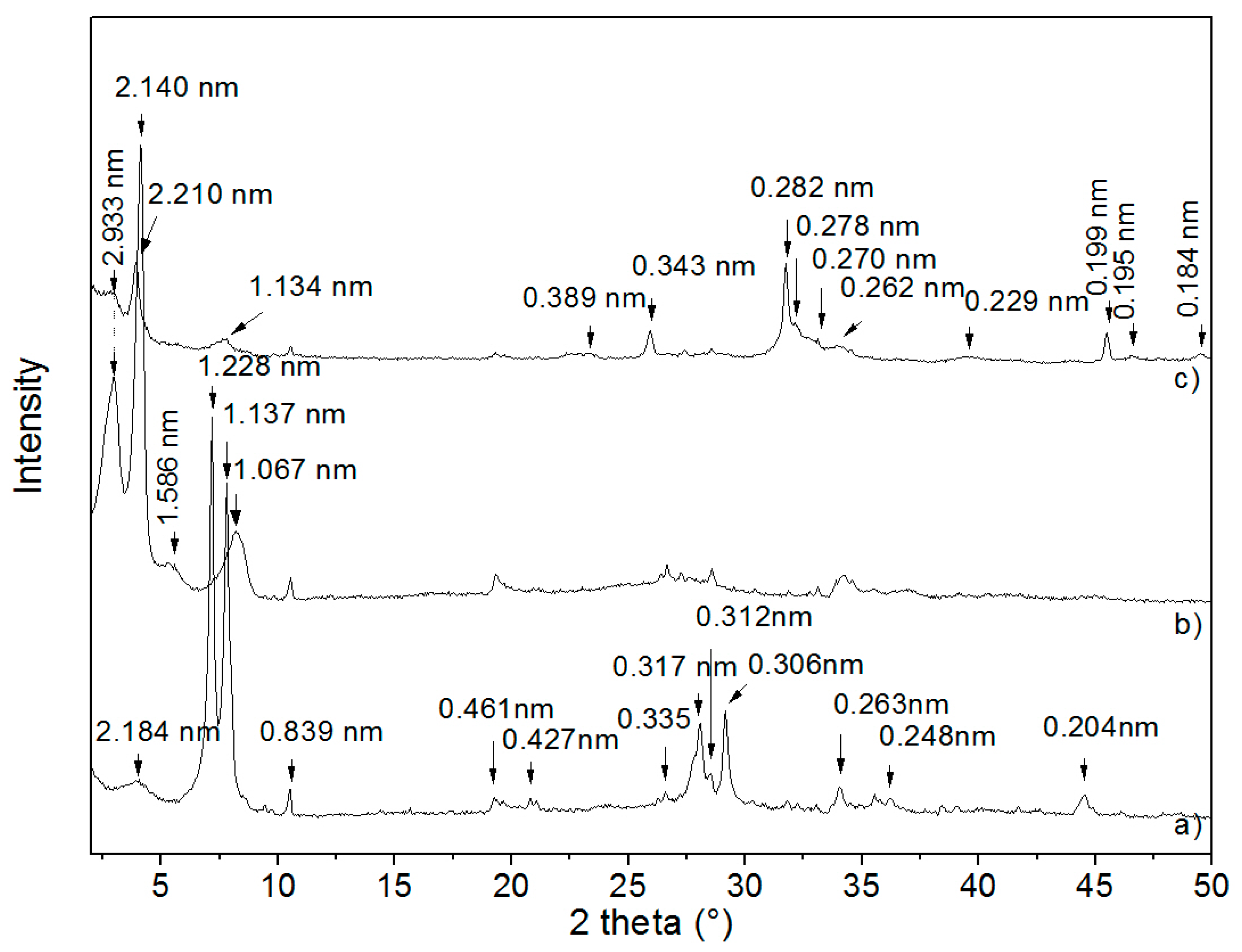
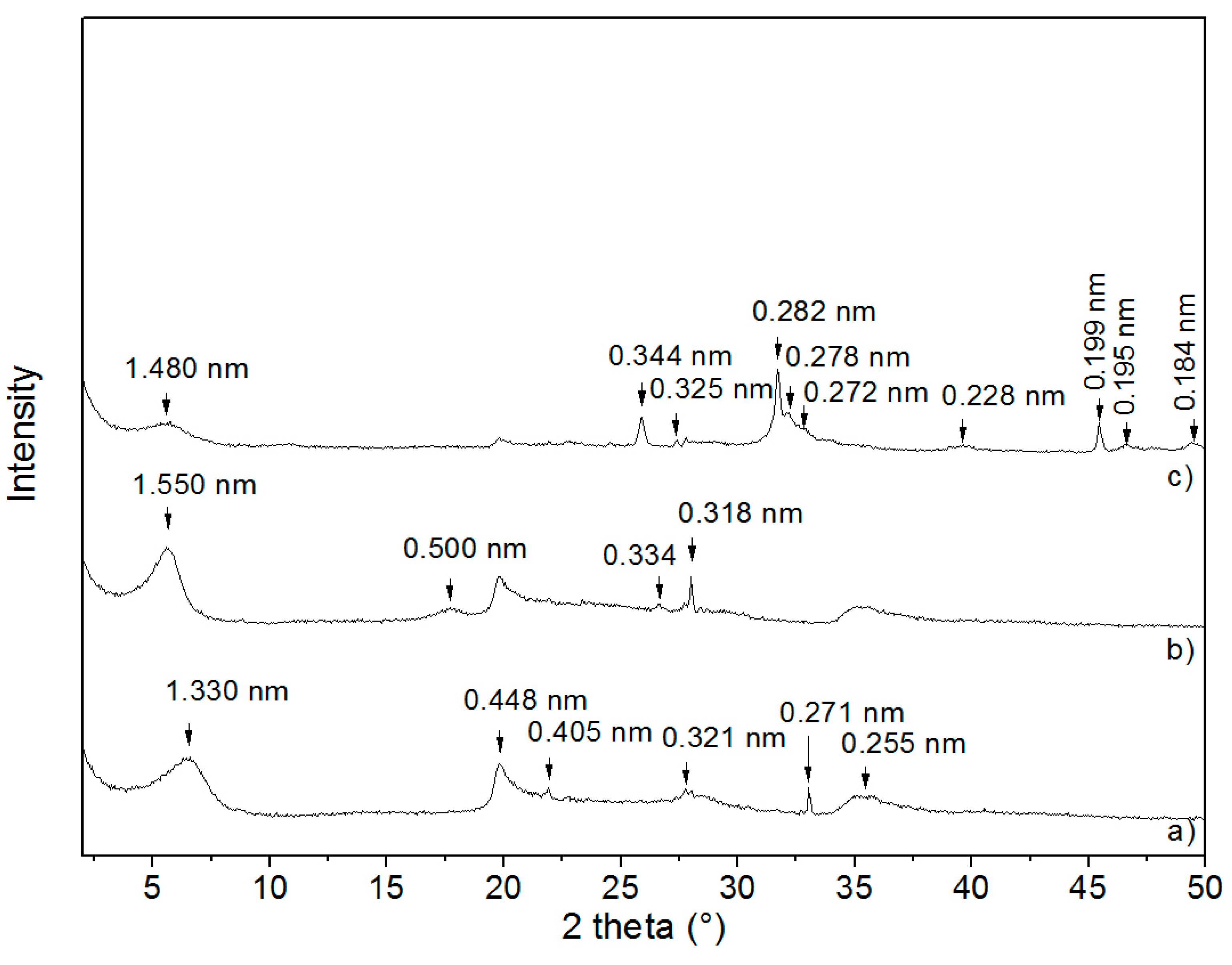
3.1. X-ray Diffraction Analysis
3.2. FTIR Spectroscopy
3.3. Antimicrobial Test
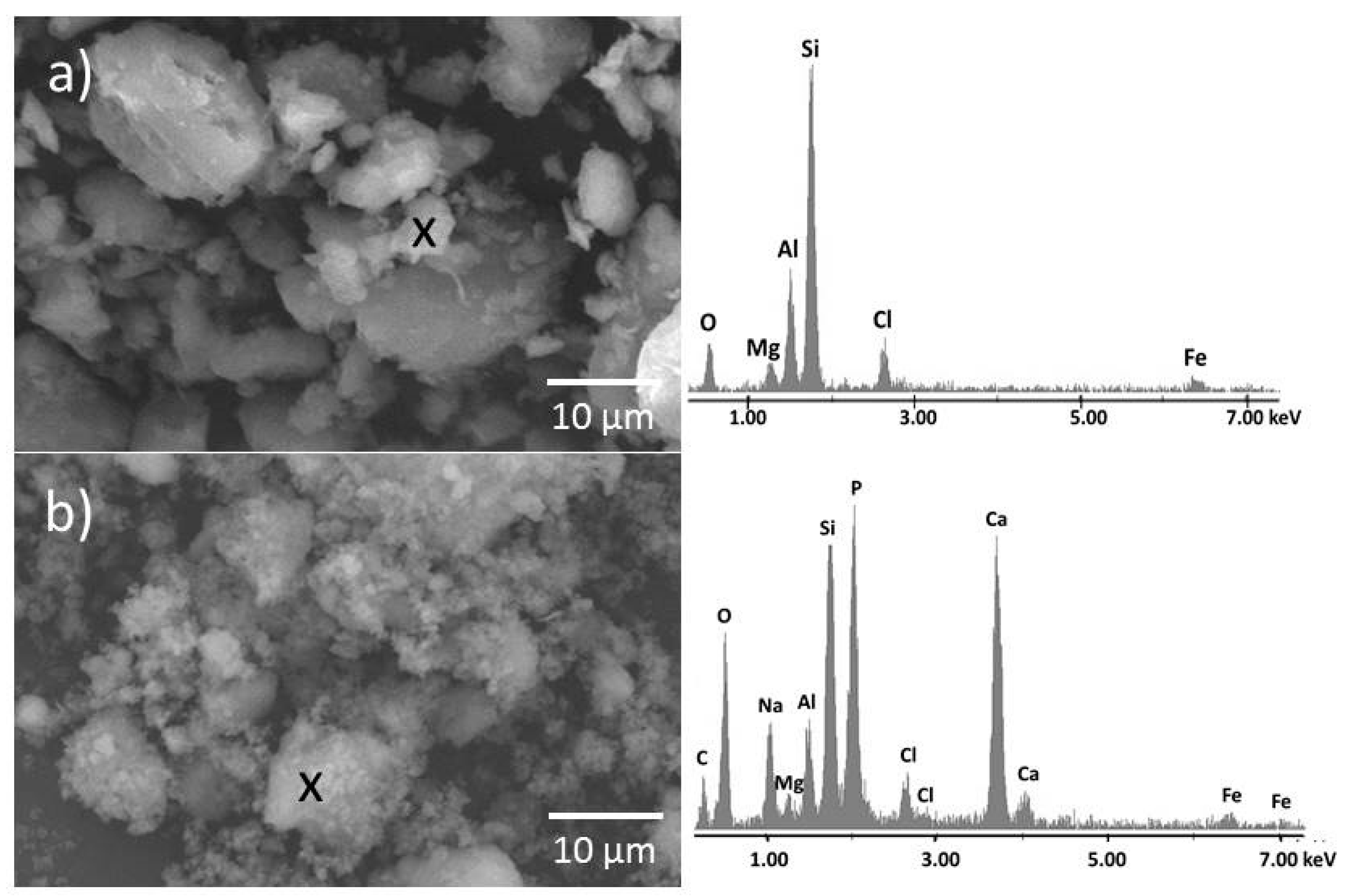
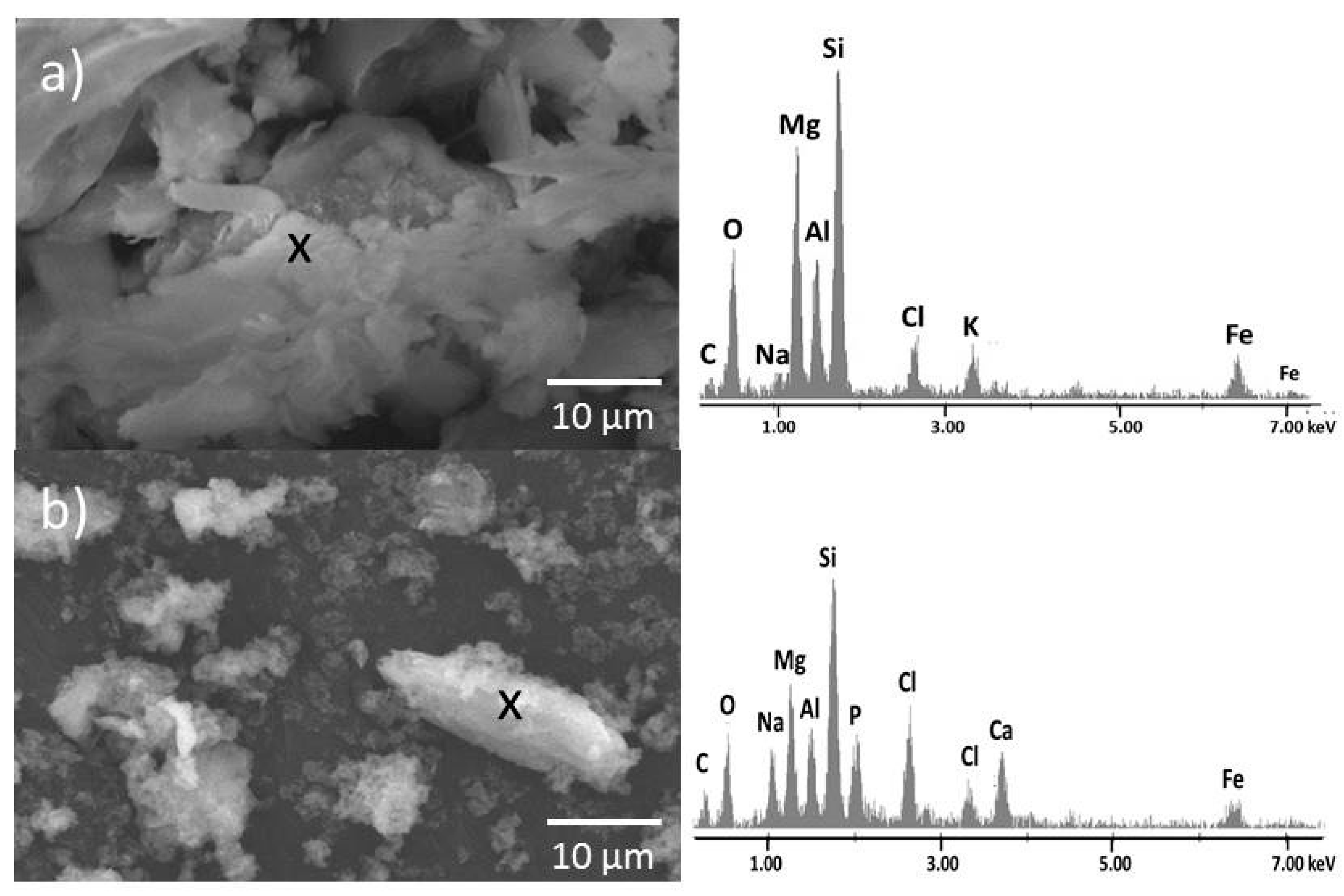
3.4. Scanning Electron Microscopy
3.5. Specific Surface Area Measurement
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, L.; Pan, H.; Tao, J.; Xu, X.; Mao, C.; Gu, X.; Tang, R. Repair of enamel by using hydroxyapatite nanoparticles as the building block. J. Mater. Chem. 2008, 18, 4079–4084. [Google Scholar] [CrossRef]
- Jarcho, M. Calcium phosphate ceramics as hard tissue prosthetics. Clin. Orthop. Rel. Res. 1981, 157, 259–278. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphates in nature, biology and medicine. Materials 2009, 2, 399–498. [Google Scholar] [CrossRef]
- Šupová, M. Isolation and preparation of nanoscale bioapatites from natural sources: A review. J. Nanosci. Nanotechnol. 2014, 14, 546–563. [Google Scholar] [CrossRef]
- Carretero, M.I.; Pozo, M. Clay and non–clay minerals in the pharmaceutical industry Part I. Excipients and medical applications. Appl. Clay. Sci. 2009, 46, 73–80. [Google Scholar] [CrossRef]
- Ambre, A.; Katti, K.S.; Katti, D.R. In situ mineralized hydroxyapatite on amino acid modified nanoclays as novel bone biomaterials. Mater. Sci. Eng. C 2011, 31, 1017–1029. [Google Scholar] [CrossRef]
- Kneiflová, J. Evaluation of the bactericidal effectiveness of disinfection agents using a suspension micromethod. Ceskoslovenska Epidemiol. Mikrobiol. Imunol. 1988, 37, 97–104. (In Czech) [Google Scholar]
- Marcos, C.; Argüelles, A.; Ruíz–Conde, A.; Sánchez–Soto, P.J.; Blanco, J.A. Study of the dehydration process of vermiculites by applying a vacuum pressure: Formation of interstratified phases. Mineral. Mag. 2003, 67, 1253–1268. [Google Scholar] [CrossRef]
- Marcos, C.; Arango, Y.C.; Rodriguez, I. X–ray diffraction studies of the thermal behaviour of commercial vermiculites. Appl. Clay Sci. 2009, 42, 368–378. [Google Scholar] [CrossRef]
- Valášková, M.; Kupková, J.; Simha Martynková, G.; Seidlerová, J.; Tomášek, V.; Ritz, M.; Kočí, K.; Klemm, V.; Rafaja, D. Comparable study of vermiculites from four commercial deposits prepared with fixed ceria nanocomposites. Appl. Clay. Sci. 2018, 151, 164–174. [Google Scholar] [CrossRef]
- Samlíková, M.; Holešová, S.; Hundáková, M.; Pazdziora, E.; Jankovič, Ľ.; Valášková, M. Preparation of antibacterial chlorhexidine/vermiculite and release study. Int. J. Miner. Process. 2017, 159, 1–6. [Google Scholar] [CrossRef]
- Simha Martynková, G.; Valášková, M.; Čapková, P.; Matějka, V. Structural ordering of organovermiculite: Experiments and modelling. J. Colloid. Interface. Sci. 2007, 313, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Alves, J.L.; Rosa, P.D.T.V.E.; Morales, A.R. Evaluation of organic modification of montmorillonite with ionic and nonionic surfactants. App. Clay. Sci. 2017, 150, 23–33. [Google Scholar] [CrossRef]
- Scherrer, P. Bestimmung der Grösse und der inneren Struktur von Kolloidteilchen mittels Röntgensrahlen [Determination of the size and internal structure of colloidal particles using X–rays], Nachr Ges Wiss Goettingen. Math. Phys. Kl. 1918, 98–100. [Google Scholar]
- Farmer, V.C. The infrared Spectra of Minerals; Farmer, V.C., Ed.; The Mineralogical Society: London, UK, 1974. [Google Scholar]
- Madejová, J.; Komadel, P. Baseline studies of the clay minerals society source clays: Infrared methods. Clays Clay Miner. 2001, 49, 410–432. [Google Scholar]
- Moenke, H.H.W. Silica, the three–dimensional silicates, borosilicates and beryllium silicates. In Infrared Spectra of Minerals; Farmer, V.C., Ed.; Mineralogical Society: London, UK, 1974; pp. 365–382. [Google Scholar]
- Silverstein, R.M.; Basser, G.C.; Morrill, T.C. Spectrometric Identification of Organic Compounds, 2nd ed.; John Wiley & Sons Inc.: New York, NY, USA, 1991. [Google Scholar]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies, Tables and Charts, 3rd ed.; John Wiley & Sons Inc.: Chichester, UK, 2001. [Google Scholar]
- Holešová, S.; Valášková, M.; Hlaváč, D.; Madejová, J.; Samlíková, M.; Tokarský, J.; Pazdziora, E. Antibacterial kaolinite/urea/chlorhexidine nanocomposites: Experiment and molecular modelling. Appl. Surf. Sci. 2014, 305, 783–791. [Google Scholar] [CrossRef]
- Panda, R.N.; Hsieh, M.F.; Chung, R.J.; Chin, T.S. FTIR, XRD, SEM and solid state NMR investigations of carbonate–containing hydroxyapatite nanoparticles synthesized by hydroxide–gel technique. J. Phys. Chem. Solids 2003, 64, 193–199. [Google Scholar] [CrossRef]
- Mistra, D.N. Interaction of chlorhexidine digluconate with and adsorption of chlorhexidine on hydroxyapatite. J. Biomed. Mater Res. 1994, 28, 1375–1381. [Google Scholar] [CrossRef]
- Gen–Tao, Z.; Qi–Zhi, Y.; Jie, N.; Gu, J. Formation of aragonite mesocrystals and implication for biomineralization. Am. Mineral 2009, 94, 293–302. [Google Scholar]
- Pazourková, L.; Hundáková, M.; Peikertová, P.; Simha Martynková, G. Preparation of calcium–deficient hydroxyapatite particles on vermiculite by precipitation and sonication. J. Aust. Ceram Soc. 2017, 53, 775–785. [Google Scholar] [CrossRef]
- Narayan, R. Advances in Bioceramics and Porous Ceramics VIII: Ceramic Engineering and Science Proceedings; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Holešová, S.; Samlíková, M.; Pazdziora, E.; Valášková, M. Antibacterial activity of organomontmorillonites and organovermiculites prepared using chlorhexidine diacetátem. Appl. Clay Sci. 2013, 83, 17–23. [Google Scholar]
- Tarte, P. Infra–red spectra of inorganic aluminates and characteristic vibrational frequencies of AlO4 tetrahedra and AlO6 octahedra. Spectrochim Acta A Mol. Spectrosc. 1967, 23, 2127–2143. [Google Scholar] [CrossRef]
- Yahiaoui, A.; Belbachir, M.; Hachemaoui, A. An acid exchanged montmorillonite clay–catalyzed synthesis of polyepichlorhydrin. Int. J. Mol. Sci. 2003, 4, 548–561. [Google Scholar] [CrossRef]
- Masindi, V.; Gitari, M.; Tutu, H.; Debeer, M. Synthesis of cryptocrystalline magnesite–bentonite clay composite and its application for neutralization and attenuation of inorganic contaminants in acidic and metalliferous mine drainage. J. Water Proc. Eng. 2017, 15, 2–17. [Google Scholar] [CrossRef]
- Radev, L.; Hristov, V.; Michailova, I.; Fernandes, H.; Salvado, M. In vitro bioactivity of biphasic calcium phosphate silicate glass–ceramic in CaO–SiO2–P2O5 system. Proces. Appl. Cer. 2010, 4, 15–24. [Google Scholar] [CrossRef]
- Musić, S.; Filipović–Vinceković, N.; Sekovanić, L. Precipitation of amorphous SiO2 particles and their properties. Braz. J. Chem. Eng. 2011, 28, 89–94. [Google Scholar] [CrossRef]
- Ciobanu, C.; Iconaru, S.; Pasuk, I.; Vasile, B.; Lupu, A.; Hermenean, A.; Dinischiotu, A.; Predoi, D. Structural properties of silver doped hydroxyapatite and their biocompatibility. Mater. Sci. Eng. C 2013, 33, 1395–1402. [Google Scholar] [CrossRef]
- Bai, X.; More, K.; Rouleau, C.; Rabiel, A. Functionally graded hydroxyapatite coatings doped with antibacterial components. Acta Biomater. 2010, 6, 2264–2273. [Google Scholar] [CrossRef]
- Katti, K.; Katti, D. Effect of clay–water interactions on swelling in montmorillonite clay. 2018. Available online: https://www.researchgate.net/publication/242072574_Effect_Of_Clay–Water_Interactions_On_Swelling_In_Montmorillonite_Clay (accessed on 5 September 2018).
- Daud, N.; Bahri, I.; Malek, N.; Hermawan, H.; Saidin, S. Immobilization of antibacterial chlorhexidine on stainless steel using crosslinking polydopamine film: Towards infection resistant medical devices. Colloids Surf B 2016, 145, 130–139. [Google Scholar] [CrossRef]
- Šontevska, V.; Jovanovski, G.; Makreski, P.; Raškovska, A.; Šoptrajanova, B. Minerals from Macedonia. XXI. Vibrational Spectroscopy as Identificational Tool for Some Phyllosilicate Minerals. Acta Chim. Slov. 2008, 55, 757–766. [Google Scholar]
- Lerot, L.; Low, P. Effect of swelling on the infrared absorption spectrum of montmorillonite. Clays Clay Miner 1976, 14, 191–199. [Google Scholar] [CrossRef]
- Li, X.; Su, H.; Ren, G.; Wang, S. The role of MgO in the performance of Pd/SiO2/cordierite monolith catalyst for the hydrogenation of 2–ethyl–anthraquinone. Appl. Catal. A 2016, 517, 168–175. [Google Scholar] [CrossRef]
- Che, C.; Glotch, T.; Bish, D.; Michalski, J.; Xu, W. Spectroscopic study of the dehydration and/or dehydroxylation of phyllosilicate and zeolite minerals. J. Geophys Res. Plan. 2011, 116. [Google Scholar] [CrossRef]
- Senthil Kumar, R.; Rajkumar, P. Characterization of minerals in air dust particles in the state of Tamilnadu, India through ftir spectroscopy. Atmos. Chem. Phys. Discuss. 2013, 13, 22221–22248. [Google Scholar] [CrossRef]
- Ovadyahu, D.; Yariv, S.; Lapides, I. Mechanochemical Adsorption of Phenol by Tot Swelling Clay Minerals. J. Therm. Anal. Calorim. 1998, 51, 415–430. [Google Scholar] [CrossRef]
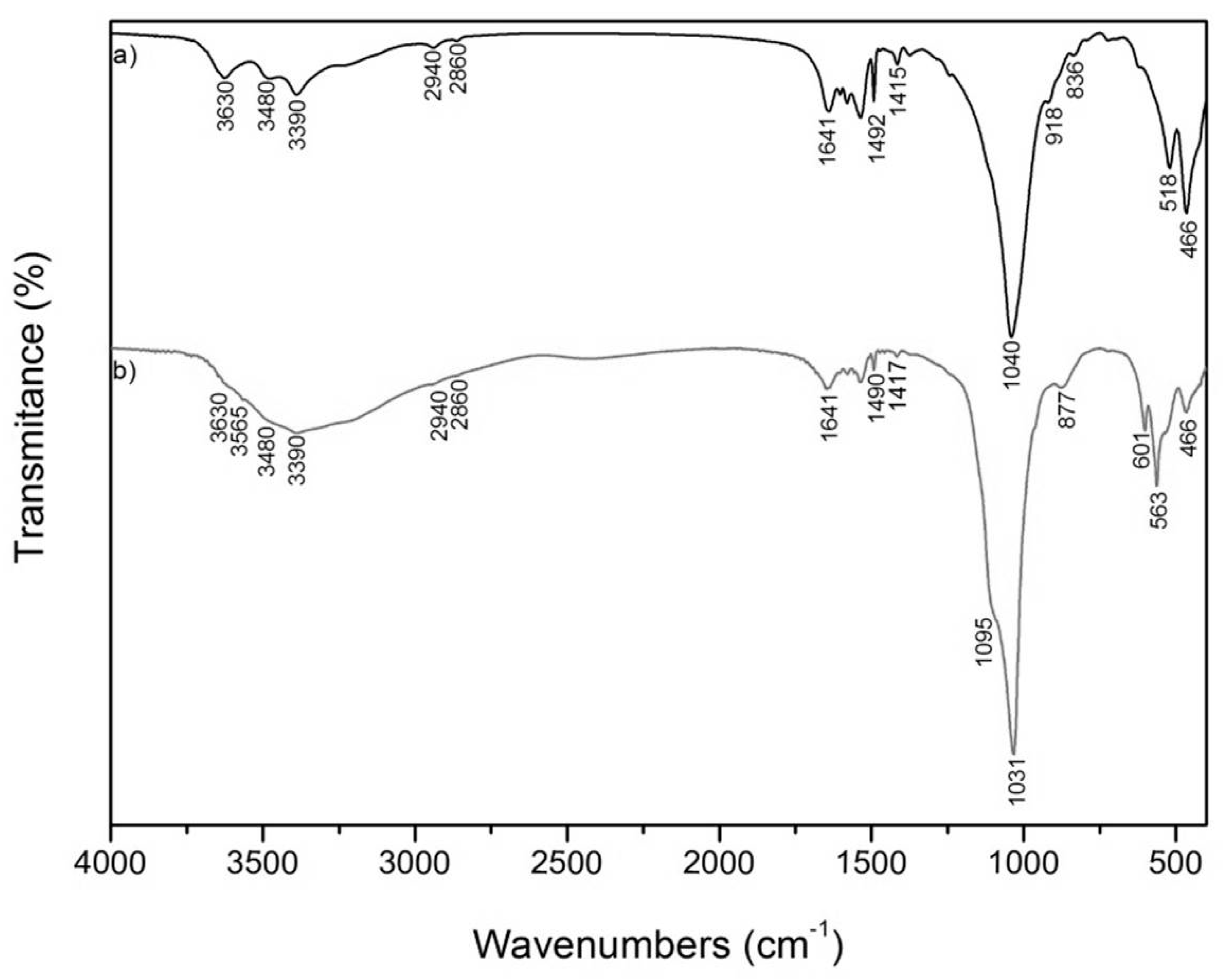


| MCA (cm−1) | MCAH (cm−1) | VCA (cm−1) | VCAH (cm−1) | Assignments |
|---|---|---|---|---|
| - | - | 823 | - | C–H vibration deformation of methylene group [25] |
| 832 | - | - | - | C–H out-of-plane vibration of 1,4-substitued aromatic ring [26] |
| - | 867 | 877 | 869 | AlO4, Al–O stretching vibration [27] |
| 883 | - | - | - | AlFe3+OH [28] |
| 915 | - | 917 | - | Al–OH–Al [29] |
| 941 | - | - | 944 | Si–O symmetric [30] |
| - | - | 956 | - | Si–O stretching [31] |
| - | 959 | - | 960 | ν1 PO43− [32] |
| - | 991 | 1001 | 990 | Si-O-Si [30] |
| - | 1028 | - | 1030 | ν3 PO43− [33] |
| 1034 | - | - | - | Si–O stretching vibration [34] |
| - | 1039 | - | - | ν3 PO43− [33] |
| 1043 | - | - | - | Aromatic amine CA [35] |
| - | - | - | 1049 | Aromatic amine [35] |
| - | - | 1064 | - | νasymSi–O [36] |
| 1093 | 1092 | 1094 | - | Si–O, C–Cl stretching vibration of halogen compounds [37] |
| - | - | - | 1109 | Asymmetric Si–O–Si [38] |
| - | 1114 | - | - | Si–O stretching [38] |
| 1122 | - | - | - | Si–O–Si [39] |
| - | 1144 | - | 1151 | Symmetric Si–O [40] |
| 1168 | - | 1164 | - | C–OH stretching [41] |
| Sample | Enterococcus Faecalis MIC (%, w/v) | Staphylococcus Aureus MIC(%, w/v) | ||||||||
| 0.5 h | 2 h | 4 h | 24 h | 120 h | 0.5 h | 2 h | 4 h | 24 h | 120 h | |
| NaMMT | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 |
| MCA | 3.33 | 3.33 | 3.33 | 0.014 | 0.014 | 0.37 | 0.041 | 0.014 | 0.014 | 0.014 |
| MCAH | 3.33 | 3.33 | 3.33 | 1.11 | 0.12 | 1.11 | 1.11 | 1.11 | 0.014 | 0.014 |
| NaVER | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | 10 |
| VCA | 10 | 3.33 | 10 | 0.014 | 0.014 | 0.37 | 0.12 | 0.014 | 0.014 | 0.014 |
| VCAH | 10 | 10 | 10 | 10 | 0.37 | 3.33 | 3.33 | 3.33 | 0.014 | 0.014 |
| Escherichia Coli MIC (%, w/v) | Pseudomonas Aeruginosa MIC(%, w/v) | |||||||||
| 0.5 h | 2 h | 4 h | 24 h | 120 h | 0.5 h | 2 h | 4 h | 24 h | 120 h | |
| NaMMT | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 |
| MCA | 1.11 | 1.11 | 1.11 | 0.014 | 0.014 | 3.33 | 3.33 | 3.33 | 1.11 | 1.11 |
| MCAH | 3.33 | 1.11 | 1.11 | 0.12 | 0.014 | 10 | 1.11 | 3.33 | 1.11 | 1.11 |
| NaVER | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 | >10 |
| VCA | 3.33 | 1.11 | 1.11 | 0.014 | 0.014 | 10 | 10 | 10 | 10 | 10 |
| VCAH | 10 | 3.33 | 3.33 | 0.12 | 0.37 | 10 | 10 | 3.33 | 3.33 | 10 |
| Candida Albicans MIC (%, w/v) | - | |||||||||
| 0.5 h | 2 h | 4 h | 24 h | 120 h | ||||||
| NaMMT | >10 | >10 | >10 | >10 | >10 | |||||
| MCA | 0.12 | 0.041 | 0.014 | 0.014 | 0.014 | |||||
| MCAH | 10 | 1.11 | 0.12 | 1.11 | 0.37 | |||||
| NaVER | >10 | >10 | >10 | >10 | >10 | |||||
| VCA | >10 | 0.12 | 0.041 | 0.014 | 0.041 | |||||
| VCAH | >10 | 0.37 | 1.11 | 1.11 | 0.37 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pazourková, L.; Reli, M.; Hundáková, M.; Pazdziora, E.; Predoi, D.; Simha Martynková, G.; Lafdi, K. Study of the Structure and Antimicrobial Activity of Ca-Deficient Ceramics on Chlorhexidine Nanoclay Substrate. Materials 2019, 12, 2996. https://doi.org/10.3390/ma12182996
Pazourková L, Reli M, Hundáková M, Pazdziora E, Predoi D, Simha Martynková G, Lafdi K. Study of the Structure and Antimicrobial Activity of Ca-Deficient Ceramics on Chlorhexidine Nanoclay Substrate. Materials. 2019; 12(18):2996. https://doi.org/10.3390/ma12182996
Chicago/Turabian StylePazourková, Lenka, Magda Reli, Marianna Hundáková, Erich Pazdziora, Daniela Predoi, Gražyna Simha Martynková, and Khalid Lafdi. 2019. "Study of the Structure and Antimicrobial Activity of Ca-Deficient Ceramics on Chlorhexidine Nanoclay Substrate" Materials 12, no. 18: 2996. https://doi.org/10.3390/ma12182996
APA StylePazourková, L., Reli, M., Hundáková, M., Pazdziora, E., Predoi, D., Simha Martynková, G., & Lafdi, K. (2019). Study of the Structure and Antimicrobial Activity of Ca-Deficient Ceramics on Chlorhexidine Nanoclay Substrate. Materials, 12(18), 2996. https://doi.org/10.3390/ma12182996








