Hydrogen Embrittlement Behavior of 18Ni 300 Maraging Steel Produced by Selective Laser Melting
Abstract
1. Introduction
2. Materials and Methods
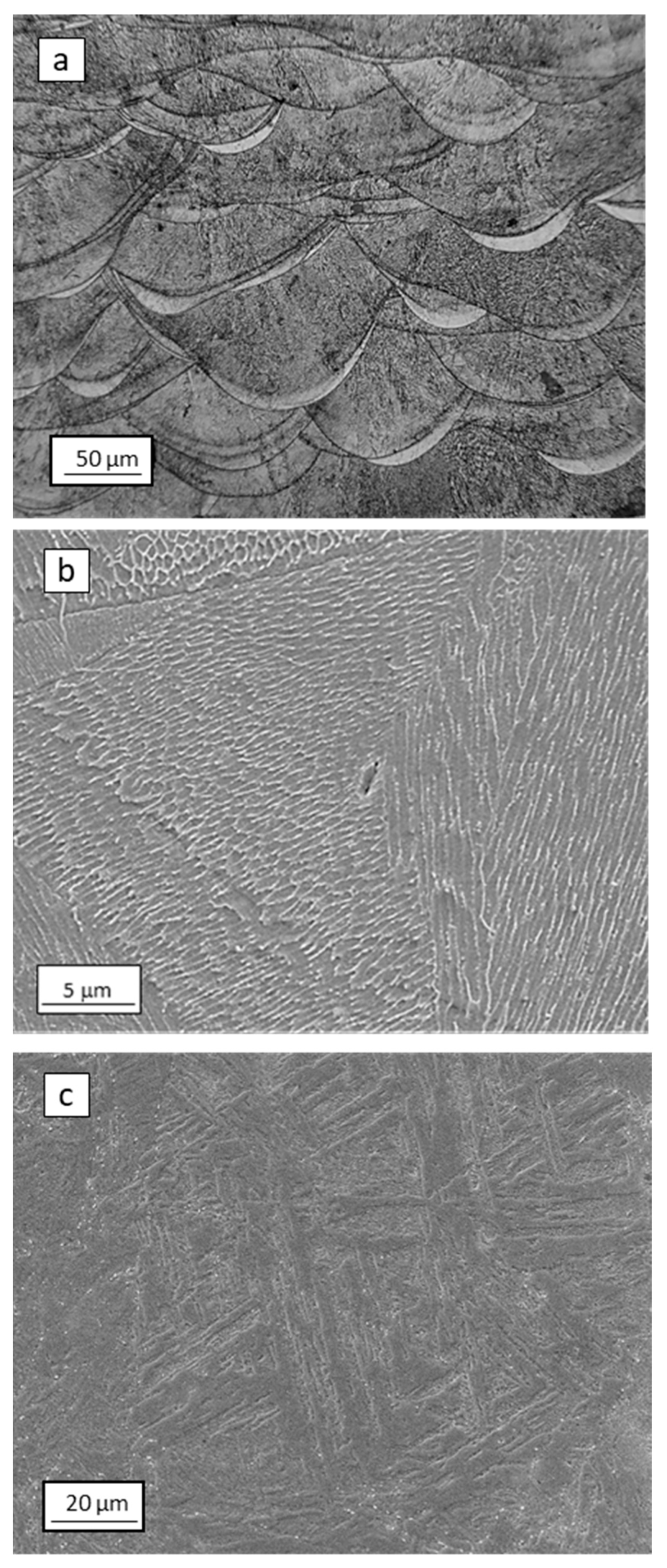
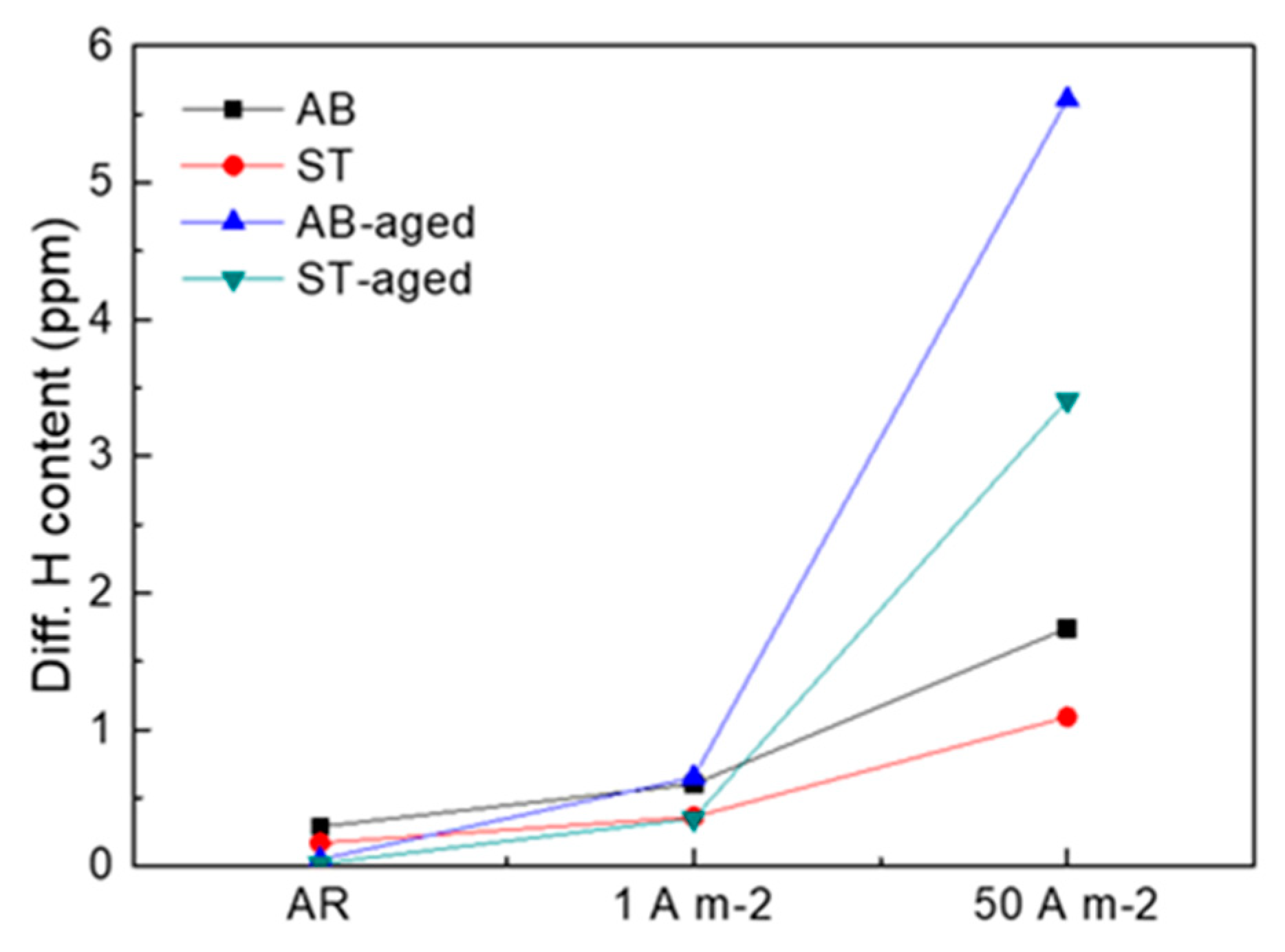
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pardal, J.M.; Tavares, S.S.M.; Terra, V.F.; Da Silva, M.R.; Dos Santos, D.R. Modeling of precipitation hardening during the aging and overaging of 18Ni-Co-Mo-Ti Maraging 300 steel. J. Alloys Compd. 2015, 393, 109–113. [Google Scholar] [CrossRef]
- Sha, W.; Cerezo, A.; Smith, G.D.W. Phase Chemistry and Precipitation Reactions in Maraging Steels: Part I. Introduction and Study of Co-Containing C-300 Steel. Metall. Mater. Trans. A 1993, 24, 1221–1232. [Google Scholar] [CrossRef]
- Pardal, J.M.; Tavares, S.S.M.; Fonseca, M.P.C.; Abreu, H.F.G.; Silva, J.J.M. Study of the austenite quantification by X-ray diffraction in the 18Ni-Co-Mo-Ti Maraging 300 steel. J. Mater. Sci. 2006, 41, 2301–2307. [Google Scholar] [CrossRef]
- Guo, Z.; Sha, W.; Li, D. Quantification of phase transformation kinetics of 18 wt.% Ni C250 Maraging steel. Mater. Sci. Eng. A 2004, 373, 10–20. [Google Scholar] [CrossRef]
- Tewari, R.; Mazumder, S.; Batra, I.S.; Dey, G.K.; Banerjee, S. Precipitation in 18 wt.% Maraging steel of grade 350. Acta Mater. 2000, 48, 1187–1200. [Google Scholar] [CrossRef]
- Jägle, E.A.; Choi, P.P.; van Humbeeck, J.; Raabe, D. Precipitation and austenite reversion behavior of a Maraging steel produced by selective laser melting. J. Mater. Res. 2014, 29, 2072–2079. [Google Scholar] [CrossRef]
- Li, X.; Yin, Z. Reverted austenite during aging in 18Ni (350) Maraging steel. Mater. Lett. 1995, 24, 239–242. [Google Scholar] [CrossRef]
- Viswanathan, U.K.; Dey, G.K.; Sethumadhavan, V. Effects of austenite reversion during overageing on the mechanical properties of 18 Ni (350) Maraging steel. Mater. Sci. Eng. A 2005, 398, 367–372. [Google Scholar] [CrossRef]
- Tavares, S.S.M.; Abreu, H.F.G.; Neto, J.M.; Da Silva, M.R.; Popa, I. A thermomagnetic study of the martensite-austenite phase transition in the Maraging 350 steel. J. Alloys Compd. 2003, 358, 152–156. [Google Scholar] [CrossRef]
- Kempen, K.; Yasa, E.; Thijs, L.; Kruth, J.-P.; Van Humbeeck, J. Microstructure and mechanical properties of Selective Laser Melted 18Ni-300 steel. Phys. Proc. 2011, 12, 255–263. [Google Scholar] [CrossRef]
- Casalino, G.; Campanelli, S.L.; Contuzzi, N.; Ludovico, A.D. Experimental investigation and statistical optimization of the selective laser melting process of a Maraging steel. Opt. Laser Technol. 2015, 65, 151–158. [Google Scholar] [CrossRef]
- Casati, R.; Lemke, J.N.; Tuissi, A.; Vedani, M. Aging Behaviour and Mechanical Performance of 18-Ni 300 Steel Processed by Selective Laser Melting. Metals 2016, 6, 218. [Google Scholar] [CrossRef]
- DebRoy, T.; Wei, H.L.; Zuback, J.S.; Mukherjee, T.; Elmerb, J.W.; Milewskic, J.O.; Beese, A.M.; Wilson-Heida, A.; Ded, A.; Zhange, W. Additive manufacturing of metallic components—Process, structure and properties. Prog. Mater. Sci. 2018, 92, 112–224. [Google Scholar] [CrossRef]
- Casati, R.; Coduri, M.; Lecis, N.; Andrianopoli, C.; Vedani, M. Microstructure and mechanical behavior of hot-work tool steels processed by Selective Laser Melting. Mater. Charact. 2018, 137, 50–57. [Google Scholar] [CrossRef]
- Lemke, J.N.; Casati, R.; Lecis, N.; Andrianopoli, C.; Varone, S.; Montanari, R.; Vedani, M. Design of Wear-Resistant Austenitic Steels for Selective Laser Melting. Metall. Mater. Trans. A 2018, 49, 962–971. [Google Scholar] [CrossRef]
- Mutua, J.; Nakata, S.; Onda, T.; Chen, Z.C. Optimization of selective laser melting parameters and influence of post heat treatment on microstructure and mechanical properties of Maraging steel. Mater. Des. 2018, 139, 486–497. [Google Scholar] [CrossRef]
- Takata, N.; Nishida, R.; Suzuki, A.; Kobashi, M.; Kato, M. Crystallographic Features of Microstructure in Maraging Steel Fabricated by Selective Laser Melting. Metals 2018, 8, 440. [Google Scholar] [CrossRef]
- Casati, R.; Vedani, M. Aging Response of an A357 Al Alloy Processed by Selective Laser Melting. Adv. Eng. Mater. 2019, 21, 1800406. [Google Scholar] [CrossRef]
- Casati, R.; Nasab, N.H.; Coduri, M.; Tirelli, V.; Vedani, M. Effects of Platform Pre-Heating and Thermal-Treatment Strategies on Properties of AlSi10Mg Alloy Processed by Selective Laser Melting. Metals 2018, 8, 954. [Google Scholar] [CrossRef]
- Hirth, J.P. Effects of hydrogen on the properties of iron and steel. Metall. Mater. Trans. A 1980, 11, 861–890. [Google Scholar] [CrossRef]
- Kim, J.S.; Lee, Y.H.; Lee, D.L.; Park, K.T.; Lee, C.S. Microstructural influences on hydrogen delayed fracture of high strength steels. Mater. Sci. Eng. A 2009, 505, 105–110. [Google Scholar] [CrossRef]
- Kameda, J.; McMahon, C.J. Solute segregation and hydrogen-induced intergranular fracture in an alloy steel. Metall. Mater. Trans. A 1983, 14, 903–911. [Google Scholar] [CrossRef]
- Chun, Y.S.; Park, K.-T.; Lee, C.S. Delayed static failure of twinning-induced plasticity steels. Scr. Mater. 2012, 66, 960–965. [Google Scholar] [CrossRef]
- Park, I.-J.; Jeong, K.-H.; Jung, J.-G.; Lee, C.S.; Lee, Y.-K. The mechanism of enhanced resistance to the hydrogen delayed fracture in Al-added Fe–18Mn–0.6C twinning-induced plasticity steels. Int. J. Hydrogen Energy 2012, 37, 9925–9932. [Google Scholar] [CrossRef]
- Kürnsteiner, P.; Wilms, M.B.; Weisheit, A.; Barriobero-Vila, P.; Jagle, E.A.; Raabe, D. Massive nanoprecipitation in an Fe-19Ni-xAl Maraging steel triggered by the intrinsic heat treatment during laser metal deposition. Acta Mater. 2017, 129, 52–60. [Google Scholar] [CrossRef]
- Zhu, X.; Li, W.; Zhao, H.; Wang, L.; Jin, X. Hydrogen trapping sites and hydrogen-induced cracking in high strength quenching & partitioning (Q & P) treated steel, International. Int. J. Hydrogen Energy 2014, 39, 13031–13040. [Google Scholar]
- Kirchheim, R.; Pundt, A. Segregation of hydrogen at dislocations. In Proceedings of the ICF11, Turin, Italy, 20–25 March 2005. [Google Scholar]
- Santos, L.P.M.; Béreš, M.; Bastos, I.N.; Tavares, S.S.M.M.; Abreu, H.F.G.G.; Gomes da Silva, M.J.; Da Silva, M.J.G. Hydrogen embrittlement of ultra high strength 300 grade maraging steel. Corros. Sci. 2015, 101, 12–18. [Google Scholar] [CrossRef]
- Chun, Y.S.; Kim, J.S.; Park, K.-T.; Lee, Y.K.; Lee, C.S. Role of ε martensite in tensile properties and hydrogen degradation of high-Mn steels. Mater. Sci. Eng. A 2012, 533, 87–95. [Google Scholar] [CrossRef]
- Solheim, K.G.; Solberg, J.K.; Walmsley, J.; Rosenqvist, F.; Bjørnå, T.H. The role of retained austenite in hydrogen embrittlement of supermartensitic stainless steel. Eng. Fail. Anal. 2013, 34, 140–149. [Google Scholar] [CrossRef]
- Lee, J.; Lee, T.; Kwon, Y.J.; MunJang, D.-J.; Yoo, Y.; Lee, C.S. Effects of vanadium carbides on hydrogen embrittlement of tempered martensitic steel. Met. Mater. Int. 2016, 22, 364–372. [Google Scholar] [CrossRef]
- Venezuela, J.; Zhou, Q.; Liu, Q.; Li, H.; Zhang, M.; Dargusch, M.; Atrens, A. The influence of microstructure on the hydrogen embrittlement susceptibility of martensitic advanced high strength steels. Mater. Today Commun. 2018, 17, 1–14. [Google Scholar] [CrossRef]
- Tsay, L.W.; Lu, H.L.; Chen, C. The effect of grain size and aging on hydrogen embrittlement of a maraging steel. Corros. Sci. 2008, 50, 2506–2511. [Google Scholar] [CrossRef]
- Wang, G.; Yan, Y.; Li, J.; Huang, J.; Qiao, L.; Volinsky, A.A. Microstructure effect on hydrogen-induced cracking in TM210 maraging steel. Mater. Sci. Eng. A 2013, 586, 142–148. [Google Scholar] [CrossRef]
- Barsanti, M.; Beghini, M.; Frasconi, F.; Ishak, R.; Monelli, B.D.; Valentini, R. Experimental study of hydrogen embrittlement in Maraging steels. Procedia Struct. Integr. 2018, 8, 501–508. [Google Scholar] [CrossRef]






| Ni | Mo | Co | Ti | Al | Si | Fe |
|---|---|---|---|---|---|---|
| 17.6 | 5.3 | 9.6 | 0.7 | 0.09 | 0.2 | Bal. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, Y.J.; Casati, R.; Coduri, M.; Vedani, M.; Lee, C.S. Hydrogen Embrittlement Behavior of 18Ni 300 Maraging Steel Produced by Selective Laser Melting. Materials 2019, 12, 2360. https://doi.org/10.3390/ma12152360
Kwon YJ, Casati R, Coduri M, Vedani M, Lee CS. Hydrogen Embrittlement Behavior of 18Ni 300 Maraging Steel Produced by Selective Laser Melting. Materials. 2019; 12(15):2360. https://doi.org/10.3390/ma12152360
Chicago/Turabian StyleKwon, Young Jin, Riccardo Casati, Mauro Coduri, Maurizio Vedani, and Chong Soo Lee. 2019. "Hydrogen Embrittlement Behavior of 18Ni 300 Maraging Steel Produced by Selective Laser Melting" Materials 12, no. 15: 2360. https://doi.org/10.3390/ma12152360
APA StyleKwon, Y. J., Casati, R., Coduri, M., Vedani, M., & Lee, C. S. (2019). Hydrogen Embrittlement Behavior of 18Ni 300 Maraging Steel Produced by Selective Laser Melting. Materials, 12(15), 2360. https://doi.org/10.3390/ma12152360








