New Perspectives in the Use of Biomaterials for Periodontal Regeneration
Abstract
1. Introduction
2. Periodontal Regeneration and Biomaterials
2.1. Barrier Membranes
- Nonresorbable
- a.
- Cellulose acetate
- b.
- Expanded polytetrafluoroethylene (e-PTFE) with or without titanium reinforcement
- c.
- Dense polytetrafluoroethylene (d-PTFE)
- d.
- Titanium-reinforced high-density polytetrafluoroethylene (Ti-d-PTFE)
- Resorbable
- a.
- Natural
- b.
- Synthetic
2.2. Grafting Biomaterials
2.3. Biological Agents
2.4. 3D Scaffolds
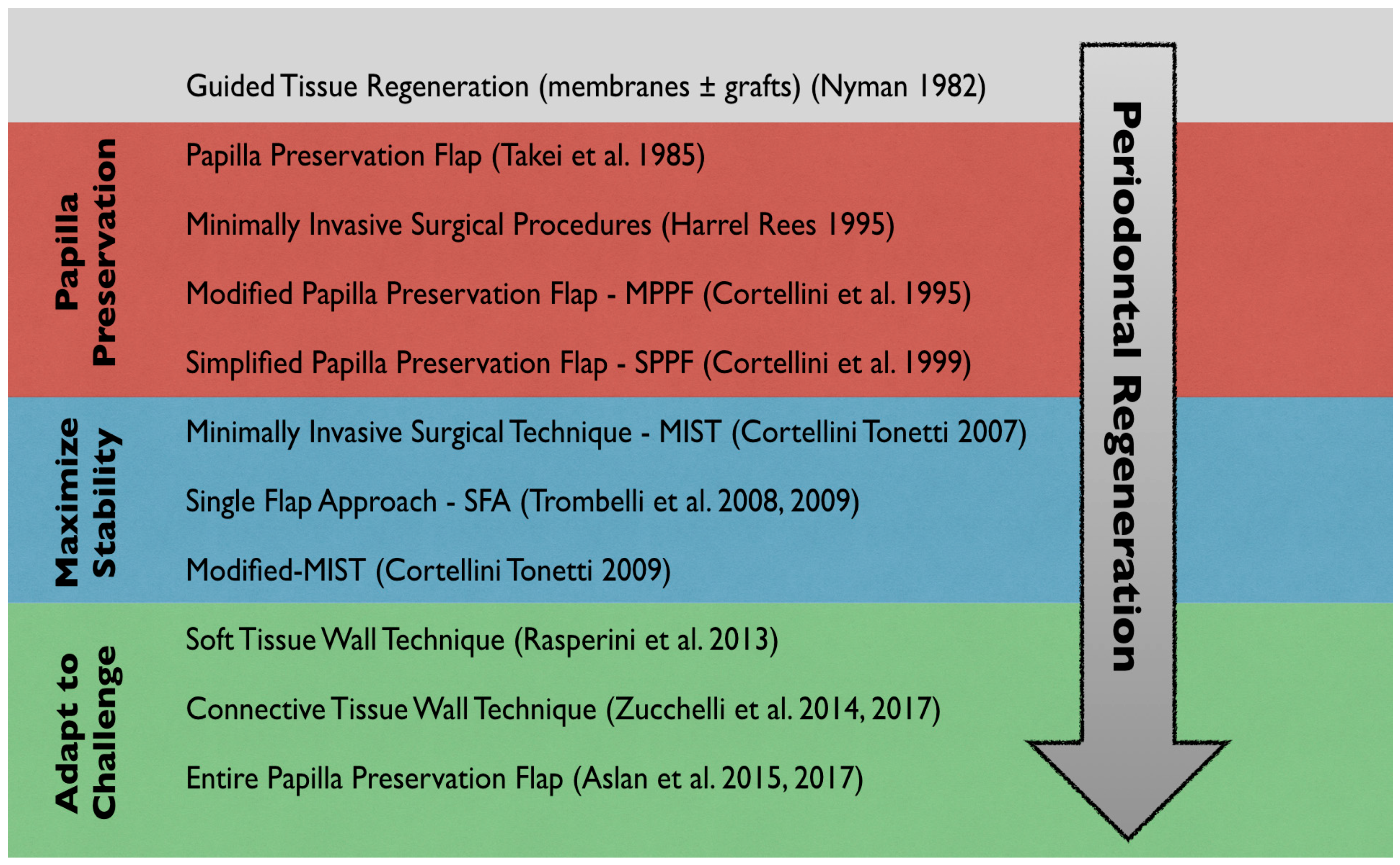
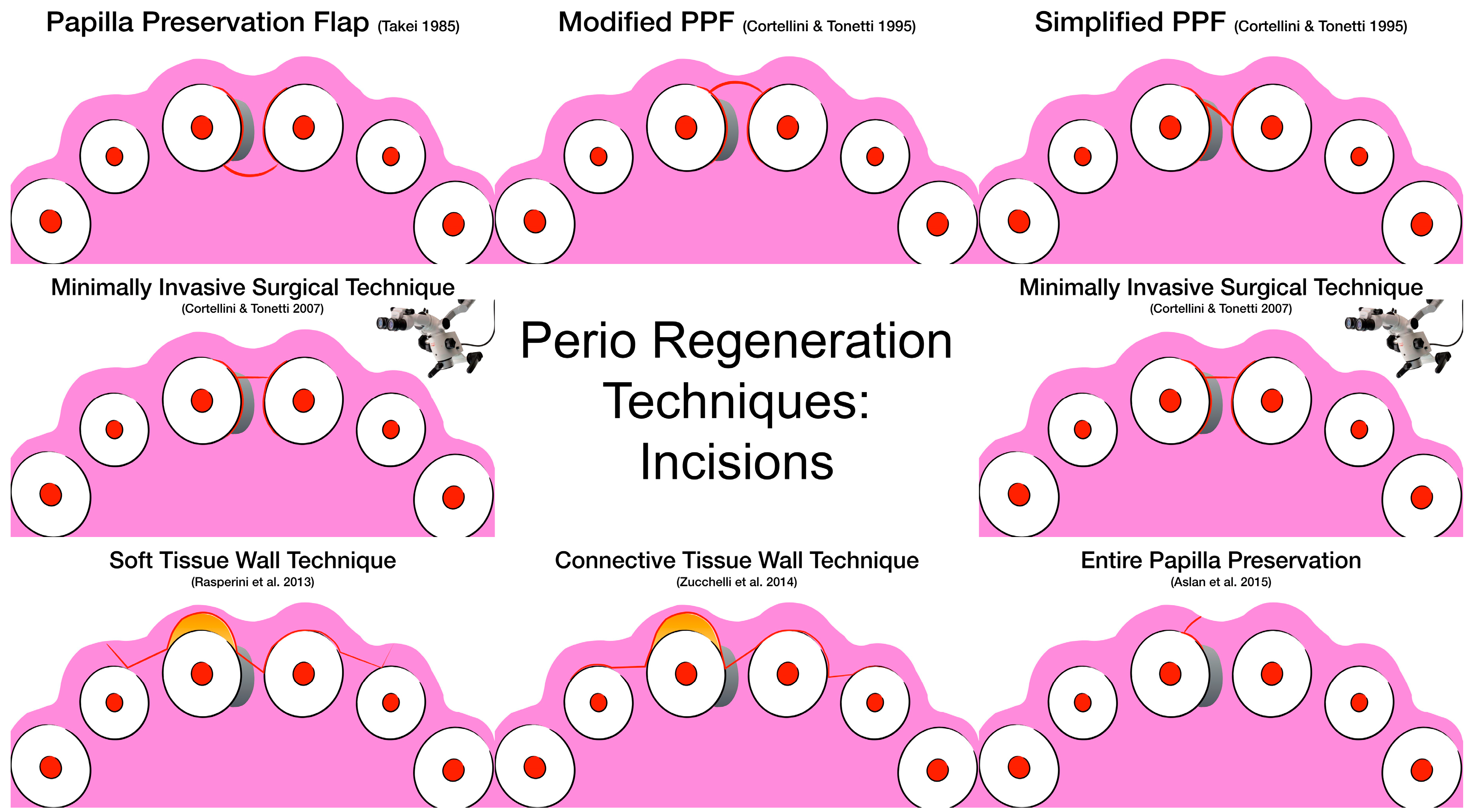
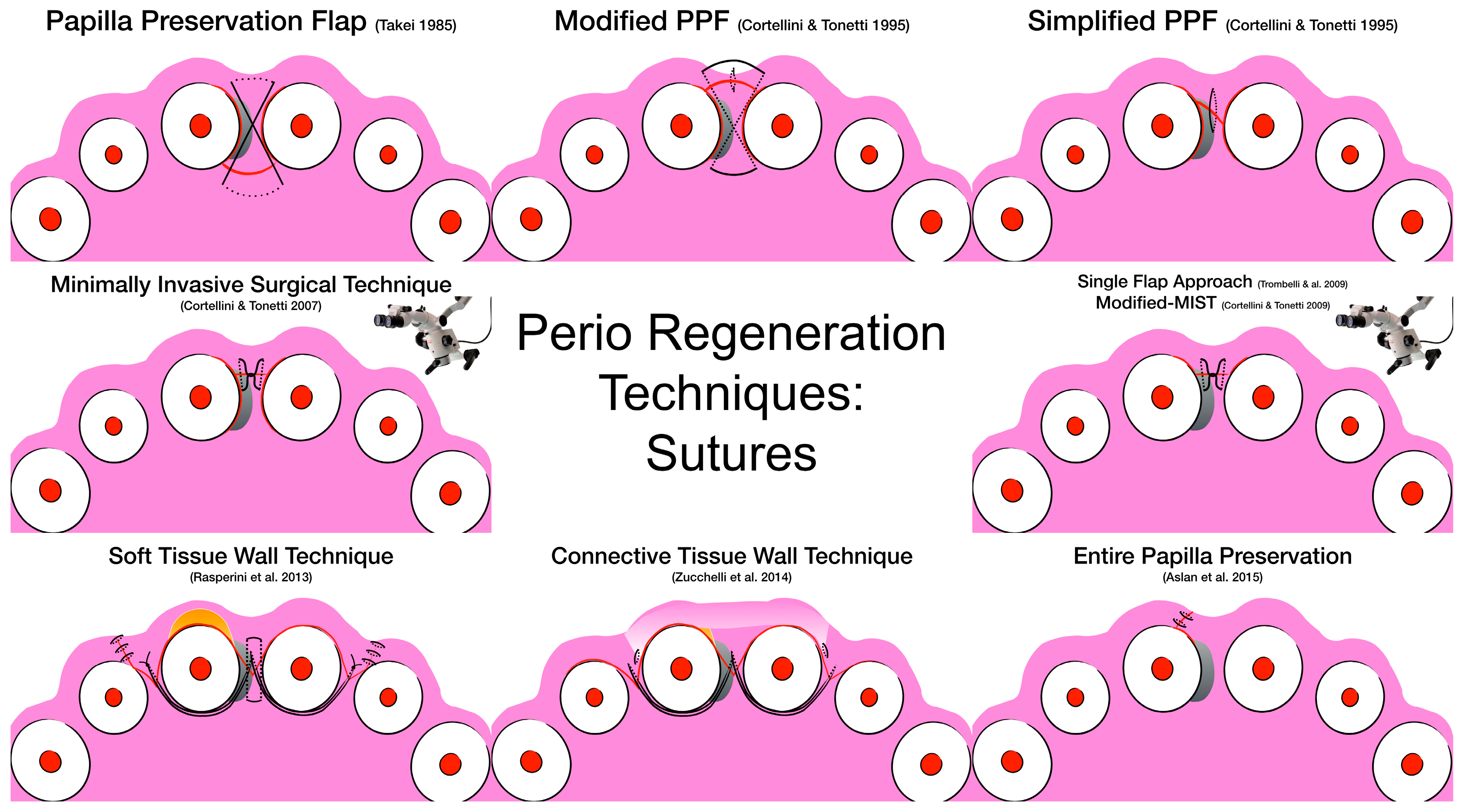
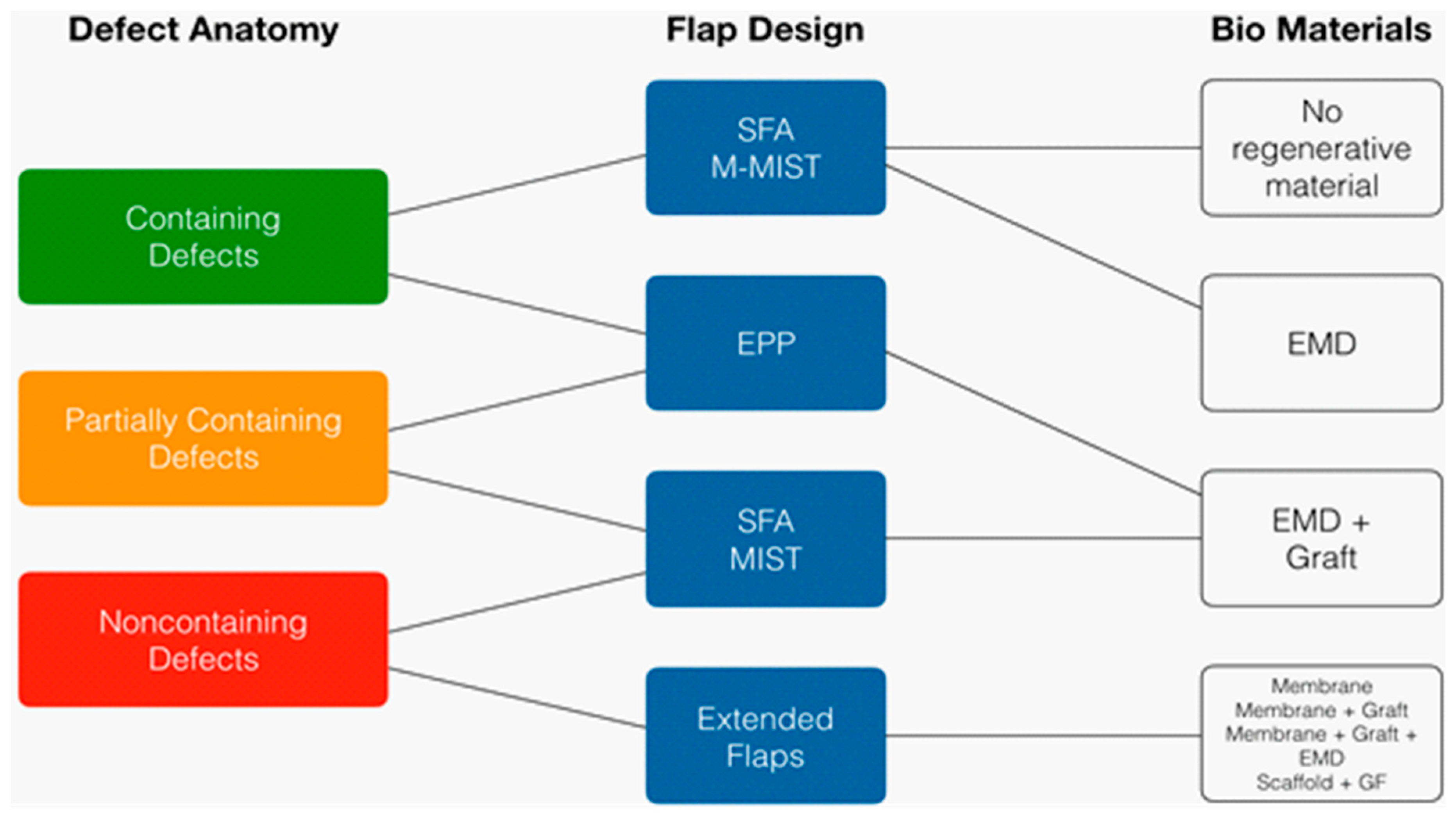
3. Surgical Techniques and Biomaterials
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bartold, P.M.; Van Dyke, T.E. A host-mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontology 2000 2013, 62, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Caton, J.; Armitage, G.; Berglundh Chapple, I.; Jepsen, S.; Kornman, K.; Mealey, B.; Papapanou, P.; Sanz, M.; Tonetti, M. A new classification scheme for periodontal and peri-implant diseases and conditions-introduction and key changes from the 1999 classification. J. Periodontol. 2018, 89 (Suppl. 1), S1–S8. [Google Scholar] [CrossRef] [PubMed]
- Löe, H.; Anerud, A.; Boysen, H.; Morrison, E. Natural history of periodontal disease in man. Rapid, moderate and no loss of attachment in Sri Lankan labourers 14 to 46 years of age. J. Clin. Periodontol. 1986, 13, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Eke, P.I.; Dye, B.A.; Wei, L.; Slade, G.D.; Thornton-Evans, G.O.; Borgnakke, W.S.; Taylor, G.W.; Page, R.C.; Beck, J.D.; Genco, R.J. Update on Prevalence of Periodontitis in Adults in the United States: NHANES 2009 to 2012. J. Periodontol. 2015, 86, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Aimetti, M.; Perotto, S.; Castiglione, A.; Mariani, G.M.; Ferrarotti, F.; Romano, F. Prevalence of periodontitis in an adult population from an urban area in North Italy: Findings from a cross-sectional population-based epidemiological survey. J. Clin. Periodontol. 2015, 42, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Kassebaum, N.J.; Bernabé, E.; Dahiya, M.; Bhandari, B.; Murray, C.J.; Marcenes, W. Global burden of severe periodontitis in 1990–2010: A systematic review meta-regression. J. Dent. Res. 2014, 93, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Matuliene, G.; Pjetursson, B.E.; Salvi, G.E.; Schmidlin, K.; Bragger, U.; Zwahlen, M.; Lang, N.P. Influence of residual pockets on progression of periodontitis and tooth loss: Results after 11 years of maintenance. J. Clin. Periodontol. 2008, 35, 685–695. [Google Scholar] [CrossRef]
- American Academy of Periodontology. Glossary of Periodontal Terms; American Academy of Periodontology: Chicago, IL, USA, 2001. [Google Scholar]
- Cortellini, P.; Tonetti, M. Clinical concepts for regenerative therapy in intrabony defects. Periodontology 2000 2015, 68, 282–307. [Google Scholar] [CrossRef]
- Waerhaug, J. Healing of the dento-dents-epithelial junction following plaque control. I: As observed in human biopsy material. J. Periodontol. 1978, 49, 1–8. [Google Scholar] [CrossRef]
- Waerhaug, J. Healing of the dento-dents-epithelial junction following plaque control. II: As observed on extracted teeth. J. Periodontol. 1978, 49, 119–134. [Google Scholar] [CrossRef]
- Caton, J.G.; Zander, H.A. The attachment between tooth and gingival tissues after periodic root planing and soft tissue curettage. J. Periodontol. 1979, 50, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.G.; Gunsolley, J.C. Guided tissue regeneration for the treatment of periodontal intrabony and furcation defects. A systematic review. Ann. Periodontol. 2003, 8, 266–302. [Google Scholar] [CrossRef] [PubMed]
- Needleman, I.G.; Worthington, H.V.; Giedrys-Leeper, E.; Tucker, R.J. Guided tissue regeneration for periodontal infra-bony defects. Chocrane Database Syst. Rev. 2006, 19, CD001724. [Google Scholar] [CrossRef] [PubMed]
- Trombelli, L.; Heitz-Mayfield, L.; Needleman, I.; Moles, D.; Scabbia, A. A systematic review of graft materials and biological agents for periodontal intraosseous defects. J. Clin. Periodontol. 2002, 29 (Suppl. 3), 117–135. [Google Scholar] [CrossRef]
- Scantelbury, T.; Ambruster, J. The development of guided regeneration: Making the impossible possible and the unpredictable predictable. J. Evid. Based Dent. Pract. 2012, SI, 101–117. [Google Scholar] [CrossRef]
- Melcher, A.H. On the repair potential of periodontal tissues. J. Periodontol. 1976, 47, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Nyman, S.; Lindhe, J.; Karring, T.; Rylander, H. New attachment following surgical treatment of human periodontal disease. J. Clin. Periodontol. 1982, 9, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Wikesjo, U.M.; Nilveus, R. Periodontal repair in dogs: Effect of wound stabilization on healing. J. Periodontol. 1990, 61, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Wikesjo, U.M.; Claffey, N.; Egelberg, J. Periodontal repair in dogs. Effect of heparin treatment of the root surface. J. Clin. Periodontol. 1991, 18, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Cardaropoli, G.; Araújo, M.; Lindhe, J. Dynamics of bone tissue formation in tooth extraction sites. An experimental study in dogs. J. Clin. Periodontol. 2003, 30, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Takei, H.H.; Han, T.; Carranza, F.A., Jr.; Kenney, E.B.; Lekovic, V. Flap technique for periodontal bone implants. Papilla preservation technique. J. Periodontol. 1985, 56, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Cortellini, P.; Prato, G.; Tonetti, M.S. The modified papilla preservation technique. A new surgical approach for interproximal regenerative procedures. J. Periodontol. 1995, 66, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Cortellini, P.; Prato, G.; Tonetti, M.S. The simplified papilla preservation flap. A novel surgical approach for the management of soft tissues in regenerative procedures. Int. J. Periodontics Restor. Dent. 1999, 19, 589–599. [Google Scholar]
- Cortellini, P.; Tonetti, M.S. Microsurgical approach to periodontal regeneration. Initial evaluation in a case cohort. J. Periodontol. 2001, 72, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Trombelli, L.; Farina, R.; Franceschetti, G. Single flap approach in periodontal reconstructive surgery. Dent. Cadmos 2007, 8, 15–25. (In Italian) [Google Scholar]
- Trombelli, L.; Farina, R.; Franceschetti, G.; Calura, G. Single-flap approach with buccal access in periodontal reconstructive procedures. J. Periodontol. 2009, 80, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Cortellini, P.; Tonetti, M.S. Improved wound stability with a modified minimally invasive surgical technique in the regenerative treatment of isolated interdental intrabony defects. J. Clin. Periodontol. 2009, 36, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Rasperini, G.; Acunzo, R.; Barnett, A.; Pagni, G. The soft tissue wall technique for the regenerative treatment of non-contained infrabony defects: A case series. Int. J. Periodontics Restor. Dent. 2013, 33, e79–e87. [Google Scholar] [CrossRef] [PubMed]
- Susin, C.; Wikesjo, U. Regenerative periodontal therapy: 30 years of lessons learned and unlearned. Periodontology 2000, 2013, 232–242. [Google Scholar] [CrossRef]
- Bunyaratavej, P.; Wang, H. Collagen Membranes: A Review. J. Periodontol. 2001, 72, 215–229. [Google Scholar] [CrossRef]
- Chipasco, M.; Abati, S.; Romeo, E.; Vogel, G. Clinical outcome of autogenous bone blocks or guided bone regeneration with e-PTFE membranes for the reconstruction of narrow edentulous ridges. Clin. Oral. Implants Res. 1999, 10, 278–288. [Google Scholar] [CrossRef]
- Jovanovic, S.A.; Nevins, M. Bone formation utilizing titanium-reinforced barrier membranes. Int. J. Periodontics Rsotor. Dent. 1995, 15, 56–69. [Google Scholar] [CrossRef]
- Blumenthal, M.N. A clinical comparison of collagen membranes with e-PTFE membranes in the treatment of human mandibular buccal class II furcation defects. J. Periodontol. 1993, 64, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Guillemin, M.; Mellonig, J.; Brunswold, M. Healing in periodontal defects reated by decalcified freeze-dried bone allografts in combination with e-PTFE membranes. (I) Clinical and scanning electron microscope analysis. J. Clin. Periodontol. 1993, 20, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Selvig, K.; Kersten, B.; Chamberlain, A.; Wikesjo, U.M.E.; Nilveus, R. Regenerative surgery of intrabony periodontal defects using e-PTFE barrier membranes. Scanninf electron microscopic evaluation of retrieved membranes vs. clinical healing. J. Periodontol. 1992, 63, 974–978. [Google Scholar] [CrossRef] [PubMed]
- Sigurdsson, T.J.; Hardwick, R.; Bogle, G.C.; Wikesjo, U.M.E. Periodontal repair in dogs: Space provision by reinforced ePTFE membranes enhances bone and cementum regeneration in large supraalveolar defects. J. Periodontol. 1994, 65, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Benic, G.; Hammerle, C. Horizontal bone augmentation by means of guided bone regeneration. Periodontology 2000 2014, 66, 13–40. [Google Scholar] [CrossRef] [PubMed]
- Johns, L.P.; Merritt, K.; Agarwal, S.; Ceravolo, F.J. Immunogenicity of a bovine collagen membrane in guided tissue regeneration. J. Dent. Res. 1992, 71, 298–301. [Google Scholar]
- Wang, H.-L.; O’Neal, R.B.; Thomas, C.L.; Shyr, Y.; MacNeil, R.L. Evaluation of an absorbable collagen membrane in treating class II furcation defects. J. Periodontol. 1994, 65, 1029–1036. [Google Scholar] [CrossRef]
- Schlegel, A.K.; Möhler, H.; Busch, F.; Mehl, A. Preclinical and clinical studies of a collagen membrane (Bio-Gide). Biomaterials 1997, 18, 535–538. [Google Scholar] [CrossRef]
- Simion, M.; Trisi, P.; Maglione, M.; Piattelli, A. A preliminary report on a method for studying the permeability of expanded polytetrafluoroethylene membrane to bacteria in vitro: A scanning electron microscopic and histological study. J. Periodontol. 1994, 65, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, L.; Zhou, Z.; Lai, H.; Xu, P.; Liao, L.; Wei, J. Biodegradale Polymer Membranes Applied in Guided Bone/Tissue Regeneration: A Review. Polymers 2016, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.M.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent. Mater. 2012, 28, 703–721. [Google Scholar] [CrossRef] [PubMed]
- Simion, M.; Nevins, M.; Rocchietta, I.; Fontana, F.; Maschera, E.; Schupbach, P.; Kim, D. Vertical ridge augmentation using an equine block infused with recombinant human platelet-derived growth factor-BB: A histologic study in a canine model. Int. J. Periodontics Restor. Dent. 2009, 29, 245–255. [Google Scholar]
- Avila-Ortiz, G.; Elengovan, S.; Karimbux, N. Bone grafting substitutes for periodontal use available in the Unites States. Clin. Adv. Periodontics 2013, 3, 187–190. [Google Scholar] [CrossRef]
- Urist, M.R. Bone formation by autoinduction. Science 1965, 150, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Burchardt, H. The biology of bone graft repair. Clin. Orthop. Relat. Res. 1983, 174, 28–42. [Google Scholar] [CrossRef]
- Tsu, Y.T.; Wang, H.L. How to Select Replacement Grafts for Various Periodontal and Implant Indications. Clin. Adv. Periodontics 2013, 167–179. [Google Scholar] [CrossRef]
- Sheikh, Z.; Hamdan, N.; Ikeda, Y.; Grynpas, M. Natural graft tissues and synthetic biomaterials for periodontal and alveolar bone reconstructive applications: A review. Biomater. Res. 2017, 21–29. [Google Scholar] [CrossRef]
- Marx, R.E.; Carlson, E.R. Creutzfeldt-Jakob disease from allogeneic dura: A review of risks and safety. J. Oral Maxillofac. Surg. 1991, 49, 272–274. [Google Scholar] [CrossRef]
- Carlson, E.R.; Marx, R.E.; Buck, B.E. The potential for HIV transmission through allogeneic bone. A review of risks and safety. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1995, 80, 17–23. [Google Scholar] [CrossRef]
- Mellonig, J.T.; Bowers, G.M.; Bright, R.W.; Lawrence, J.J. Clinical evaluation of freeze-dried bone allografts in periodontal osseous defects. J. Periodontol. 1976, 47, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, J.M.; James, R.A.; Lozada, J.; Cordova, C.; GaRey, D.J. Histological response and clinical evaluation of heterograft and allograft materials in the elevation of the maxillary sinus for the preparation of endosteal dental implant sites. Simultaneous sinus elevation and root form implantation: An eight-month autopsy report. J. Oral Implantol. 1989, 15, 141–144. [Google Scholar] [PubMed]
- Kubler, N.; Reuther, J.; Kirchner, T.; Priessnitz, B.; Sebald, W. Osteoinductive, morphologic, and biomechanical properties of autolyzed, antigen-extracted, allogeneic human bone. J. Oral Maxillofac. Surg. 1993, 51, 1346–1357. [Google Scholar] [CrossRef]
- Frost, D.E.; Fonseca, R.J.; Burkes, E.J., Jr. Healing of interpositional allogeneic lyophilized bone grafts following total maxillary osteotomy. J. Oral Maxillofac. Surg. 1982, 40, 776–786. [Google Scholar] [CrossRef]
- Becker, W.; Schenk, R.; Higuchi, K.; Lekholm, U.; Becker, B.E. Variations in bone regeneration adjacent to implants augmented with barrier membranes alone or with demineralized freeze-dried bone or autologous grafts: A study in dogs. Int. J. Oral Maxillofac. Implants 1995, 10, 143–154. [Google Scholar] [PubMed]
- Wetzel, A.C.; Stich, H.; Caffesse, R.G. Bone apposition onto oral implants in the sinus area filled with different grafting materials. A histological study in beagle dogs. Clin. Oral. Implants Res. 1995, 6, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Honig, J.F.; Merten, H.A.; Heinemann, D.E. Risk of transmission of agents associated with Creutzfeldt-Jakob disease and bovine spongiform encephalopathy. Plast. Reconstr. Surg. 1999, 103, 1324–1325. [Google Scholar] [CrossRef]
- Lang, N.P.; Hammerle, C.; Oesch, B.; Schenk, R.K. Risk of transmission of agents associated with Creutzfeldt-Jakob disease and bovine spongiform encephalopathy. Plast. Reconstr. Surg. 2000, 105, 2273–2275. [Google Scholar] [CrossRef]
- Hurzeler, M.B.; Kohal, R.J.; Naghshbandi, J.; Mota, L.F.; Conradt, J.; Hutmacher, D.; Caffesse, R.G. Evaluation of a new bioresorbable barrier to facilitate guided bone regeneration around exposed implant threads. An experimental study in the monkey. Int. J. Oral Maxillofac. Surg. 1998, 27, 315–320. [Google Scholar] [CrossRef]
- Hammerle, C.H.; Lang, N.P. Single stage surgery combining transmucosal implant placement with guided bone regeneration and bioresorbable materials. Clin. Oral. Implants Res. 2001, 12, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Hallman, S.; Lundgren, S.; Sennerby, L. Histologic analysis of clinical biopsies taken 6 months and 3 years after maxillary sinus floor augmentation with 80% bovine hydroxyapatite and 20% autogenous bone mixed with fibrin glue. Clin. Implant Dent. Relat. Res. 2001, 3, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Mordenfeld, A.; Hallman, M.; Johansson, C.B.; Albrektsson, T. Histological and histomorphometrical analyses of biopsies harvested 11 years after maxillary sinus floor augmentation with deproteinized bovine and autogenous bone. Clin. Oral. Implants Res. 2010, 21, 961–970. [Google Scholar] [CrossRef] [PubMed]
- Bauer, T.W.; Muschler, G.F. Bone graft materials. An overview of the basic science. Clin. Orthop. Relat. Res. 2000, 371, 10–27. [Google Scholar] [CrossRef]
- Sasaoka, K.; Seto, K.; Tsugita, M.; Tsuru, S. An immunological study of tricalcium phosphate supplied by three different manufacturers. J. Clin. Lab. Immunol. 1989, 30, 197–202. [Google Scholar] [PubMed]
- Zinner, I.D.; Small, S.A. Sinus-lift graft: Using the maxillary sinuses to support implants. J. Am. Dent. Assoc. 1996, 127, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Coetzee, A.S. Regeneration of bone in the presence of calcium sulfate. Arch. Otolaryngol. 1980, 106, 405–409. [Google Scholar] [CrossRef]
- Chiroff, R.; White, E.W.; Weber, K.N.; Roy, D.M. Tissue ingrowth of Replaminefrom implants. J. Biomed. Mater. Res. 1975, 9, 29–45. [Google Scholar] [CrossRef]
- Guillemin, G.J.; Patat, L.; Fournie, J.; Chetail, M. The use of coral as a bone graft substitute. J. Biomed. Mater. Res. 1987, 21, 557–567. [Google Scholar] [CrossRef]
- Martin, R.B.; Chapman, M.W.; Holmes, R.E.; Sartoris, D.J.; Shors, E.C.; Gordon, J.E.; Heitter, D.O.; Sharkey, N.A.; Zissimos, A.G. Effects of bone ingrowth on the strength and non-invasive assessment of a coralline hydroxyapatite material. Biomaterials 1989, 10, 481–488. [Google Scholar] [CrossRef]
- Bang, G.; Urist, M.R. Bone induction in excavation chambers in matrix of decalcified dentin. Arch. Surg. 1967, 94, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.B.; Urist, M.R. Dentin matrix transformation: Rapid induction of alkaline phosphatase and cartilage. Science 1970, 167, 896–898. [Google Scholar] [CrossRef] [PubMed]
- Bowers, G.; Felton, F.; Middleton, C.; Glynn, D.; Sharp, S.; Mellonig, J.; Corio, R.; Emerson, J.; Park, S.; Suzuki, J.; et al. Histologic comparison of regeneration in human intrabony defects when osteogenin is combined with demineralized freeze-dried bone allograft and with purified bovine collagen. J. Periodontol. 1991, 62, 690–702. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.E.; Williams, R.C.; Polson, A.M.; Howell, T.H.; Reddy, M.S.; Zappa, U.E.; Antoniades, H.N. A combination of platelet-derived and insulin-like growth factors enhances periodontal regeneration. J. Clin. Periodontol. 1989, 16, 545–548. [Google Scholar] [CrossRef] [PubMed]
- Hammarström, L. Enamel matrix, cementum development and regeneration. J. Clin. Periodontol. 1997, 24, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Rasperini, G.; Silvestri, M.; Ricci, G. Long-term clinical observation of treatment of infrabony defects with enamel matrix derivative (Emdogain): Surgical reentry. Int. J. Periodontics Restor. Dent. 2005, 25, 121–127. [Google Scholar]
- Rösing, C.K.; Aass, A.M.; Mavropoulos, A.; Gjermo, P. Clinical and radiographic effect of enamel matrix derivative in the treatment of intrabony periodontal defects: A 12-month longitudinal placebo-controlled trial in adult periodontitis patients. J. Periodontol. 2005, 76, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Koop, R.; Merheb, J.; Quirynen, M. Periodontal regeneratio with enamel matrix derivative in reconstructive periodontal therapy: A systematic review. J. Periodontol. 2012, 83, 707–720. [Google Scholar] [CrossRef]
- Esposito, M.; Grusovin, M.G.; Papanikolaou, N.; Coulthard, P.; Worthington, H.V. Enamel matrix derivative (Emdogain(R)) for periodontal tissue regeneration in intrabony defects. Cochrane Database Syst. Rev. 2009, 4, CD003875. [Google Scholar] [CrossRef]
- Nevins, M.; Camelo, M.; Nevins, M.L.; Schenk, R.K.; Lynch, S.E. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J. Periodontol. 2003, 74, 1282–1292. [Google Scholar] [CrossRef]
- Nevins, M.; Giannobile, W.V.; McGuire, M.K.; Kao, R.T.; Mellonig, J.T.; Hinrichs, J.E.; McAllister, B.S.; Murphy, K.S.; McClain, P.K.; Nevins, M.; et al. Platelet-derived growth factor stimulates bone fill and rate of attachment level gain: Results of a large multicenter randomized controlled trial. J. Periodontol. 2005, 76, 2205–2215. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, A.; Rajababu, P.; Rohini, S.; Butchibabu, K.; Naveen, A.; Reddy, P.K.; Vidyasagar, S.; Satyanarayana, D.; Pavan Kumar, S. Multi-centre, randomized clinical trial on the efficacy and safety of recombinant human platelet-derived growth factor with β-tricalcium phosphate in human intra-osseous periodontal defects. J. Clin. Periodontol. 2011, 38, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Darby, I.B.; Morris, K.H. A systematic review of the use of growth factors in human periodontal regeneration. J. Periodontol. 2013, 84, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Takiguchi, T.; Suzuki, R.; Yamaguchi, A.; Deguchi, K.; Shionome, M.; Miyazawa, Y.; Nishihara, T.; Nagumo, M.; Hasegawa, K. Recombinant human bone morphogenetic protein-2 stimulates osteoblastic differentiation in cells isolated from human periodontal ligament. J. Dent. Res. 1999, 78, 1624–1633. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, U.; Renton, L. Bone morphogenetic proteins and the induction of periodontal tissue regeneration. Periodontology 2000 2006, 41, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Howell, T.H.; Fiorellini, J.; Jones, A.; Alder, M.; Nummikoski, P.; Lazaro, M.; Lilly, L.; Cochran, D. Afeasibility study evaluating rhBMP-2/absorbable collagen sponge device for local alveolar ridge preservation or augmentation. Int. J. Periodontics Restor. Dent. 1997, 17, 125–139. [Google Scholar]
- Cochran, D.L.; Jones, A.A.; Lilly, L.C.; Fiorellini, J.P.; Howell, H. Evaluation of recombinant human bone morphogeneticprotein-2 in oral applications including the use of endosseous implants: 3-year results of a pilot study in humans. J. Periodontol. 2000, 71, 1241–1243. [Google Scholar] [CrossRef]
- Fiorellini, J.P.; Howell, T.H.; Cochran, D.; Malmquist, J.; Lilly, L.C.; Spagnoli, D.; Toljanic, J.; Jones, A.; Nevins, M. Randomized study evaluating recombinant human bonemorphogenetic protein-2 for extraction socket augmentation. J. Periodontol. 2005, 76, 605–613. [Google Scholar] [CrossRef]
- Giannobile, W.V.; Ryan, S.; Shih, M.-S.; Su, D.L.; Kaplan, P.L.; Chan, T.C.K. Recombinant human osteogenic protein-1 (OP-1) stimulates periodontal wound healing in class III furcation defects. J. Periodontol. 1998, 69, 129–137. [Google Scholar] [CrossRef]
- Wikesjö, U.M.E.; Guglielmoni, P.; Promsudthi, A.; Cho, K.S.; Trombelli, L.; Selvig, K.A.; Jin, L.; Wozney, J.M. Periodontal repair in dogs: Effect of rhBMP-2 concentration on regeneration of alveolar bone and periodontal attachment. J. Clin. Periodontol. 1999, 26, 392–393. [Google Scholar] [CrossRef]
- Urist, M.R. Bone: Formation by autoinduction. 1965. Clin. Orthop. Relat. Res. 2002, 395, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Boyne, P.J.; Marx, R.E.; Nevins, M.; Triplett, G.; Lazaro, E.; Lilly, L.C.; Alder, M.; Nummikoski, P. A feasibility study evaluating rhBMP-2/absorbable collagen sponge for maxillary sinus floor augmentation. Int. J. Periodontics Restor. Dent. 1997, 17, 11–13. [Google Scholar]
- Triplett, R.G.; Nevins, M.; Marx, R.E.; Spagnoli, D.B.; Oates, T.W.; Moy, P.K.; Boyne, P.J. Pivotal, randomized, parallel evaluation of recombinant human bone morphogenetic protein-2/absorbable collagen sponge and autogenous bone graft for maxillary sinus floor augmentation. J. Oral Maxillofac. Surg. 2009, 67, 1947–1960. [Google Scholar] [CrossRef] [PubMed]
- Corinaldesi, G.; Piersanti, L.; Piattelli, A.; Iezzi, G.; Pieri, F.; Marchetti, C. Augmentation of the floor of the maxillary sinus with recombinant human bone morphogenetic protein-7: A pilot radiological and histological study in humans. Br. J. Oral Maxillofac. Surg. 2013, 51, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Wikesjö, U.M.; Sorensen, R.G.; Kinoshita, A.; Jian Li, X.; Wozney, J.M. Periodontal repair in dogs: Effect of recombinant human bone morphogenetic protein-12 (rhBMP-12) on regeneration of alveolar bone and periodontal attachment. J. Clin. Periodontol. 2004, 31, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Mayahara, H.; Ito, T.; Nagai, H.; Miyajima, H.; Tsukuda, R.; Taketomi, S.; Mizoguchi, J.; Kato, K. In vivo stimulation of endosteal bone formation by basic fibroblast growth factor in rats. Growth Factors 1993, 9, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Hanada, K.; Tamura, M.; Shibanushi, T.; Nigi, H.; Tagawa, M.; Fukumoto, S.; Matsumoto, T. Stimulation of endosteal bone formation by systemic injections of recombinant basic fibroblast growth factor in rats. Endocrinology 1995, 136, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Takayama, S.; Kitamura, M.; Shimabukuro, Y.; Yanagi, K.; Ikezawa, K.; Saho, T.; Nozaki, T.; Okada, H. Recombinant human basic fibroblast growth factor (bFGF) stimulates periodontal regeneration in class II furcation defects created in beagle dogs. J. Periodontal Res. 2003, 38, 97–103. [Google Scholar] [CrossRef]
- Takayama, S.; Murakami, S.; Shimabukuro, Y.; Kitamura, M.; Okada, H. Periodontal regeneration by FGF-2 (bFGF) in primate models. J. Dent. Res. 2001, 80, 2075–2079. [Google Scholar] [CrossRef]
- Kitamura, M.; Akamatsu, M.; Machigashira, M.; Hara, Y.; Sakagami, R.; Hirofuji, T.; Hamachi, T.; Maeda, K.; Yokota, M.; Kido, J.; et al. FGF-2 stimulates periodontal regeneration: Results of a multi-center randomized clinical trial. J. Dent. Res. 2011, 90, 35–40. [Google Scholar] [CrossRef]
- Kraus, D.; Jäger, A.; Abuduwali, N.; Deschner, J.; Lossdörfer, S. Intermittent PTH(1-34) signals through protein kinase A to regulate osteoprotegerin production in human periodontal ligament cells in vitro. Clin. Oral Investig. 2012, 16, 611–613. [Google Scholar] [CrossRef] [PubMed]
- Lossdörfer, S.; Götz, W.; Jäger, A. PTH(1-34) affects osteoprotegerin production in human PDL cells in vitro. J. Dent. Res. 2005, 84, 634–638. [Google Scholar] [CrossRef] [PubMed]
- Lossdörfer, S.; Götz, W.; Jäger, A. Parathyroid hormone modifies human periodontal ligament cell proliferation and survival in vitro. J. Periodontal Res. 2006, 41, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Bashutski, J.D.; Eber, R.M.; Kinney, J.S.; Benavides, E.; Maitra, S.; Braun, T.; Giannobile, W.V.; McCauley, L.K. Teriparatide and osseous regeneration in the oral cavity. N. Engl. J. Med. 2010, 363, 2396–2405. [Google Scholar] [CrossRef] [PubMed]
- Kuchler, U.; Luvizuto, E.R.; Tangl, S.; Watzek, G.; Gruber, R. Short-termteriparatide delivery and osseointegration: A clinical feasibility study. J. Dent. Res. 2011, 90, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Boyapati, L.; Wang, H.-L. The role of platelet-rich plasma in sinus augmentation: A critical review. Implant Dent. 2006, 15, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, N.; Greenwell, H.; Hill, M.; Vida, R.L.; Scheetz, J.P. Root coverage using acellular dermal matrix and comparing a coronally positioned tunnel with and without platelet-rich plasma: A pilot study in humans. J. Periodontol. 2009, 80, 397–404. [Google Scholar] [CrossRef]
- Khairy, N.M.; Shendy, E.E.; Askar, N.A.; El-Rouby, D.H. Effect of platelet rich plasma on bone regeneration in maxillary sinus augmentation (randomized clinical trial). Int. J. Oral Maxillofac. Surg. 2013, 42, 249–255. [Google Scholar] [CrossRef]
- Kawase, T.; Kamiya, M.; Kobayashi, M.; Tanaka, T.; Okuda, K.; Wolff, L.F.; Yoshie, H. The heat compression technique for the conversion of platelet-rich fibrin preparation to a barrier membrane with a reduced rate of biodegradation. J. Biomed. Mater. Res. B Appl. Biomater. 2015, 103, 825–831. [Google Scholar] [CrossRef]
- Döri, F.; Kov´acs, V.; Arweiler, N.B.; Huszár, T.; Gera, I.; Nikolidakis, D.; Sculean, A. Effect of platelet rich plasma on the healing of intrabony defects treated with an anorganic bovine bone mineral: A pilot study. J. Periodontol. 2009, 80, 1599–1605. [Google Scholar] [CrossRef]
- Chambrone, L.; Tatakis, D.N. Periodontal soft tissue root coverage procedures: A systematic review from the AAP regeneration workshop. J. Periodontol. 2015, 86, S8–S51. [Google Scholar] [CrossRef] [PubMed]
- Steffensen, B.; Weber, H.P. Relationship between the radiographic periodontal defect angle and healing after treatment. J. Periodontol. 1989, 60, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Choi, S.H.; Chai, J.K.; Cho, K.S.; Moon, I.S.; Wikesjö, U.M.; Kim, C.K. Periodontal repair in surgically created intrabony defects in dogs: Influence of the number of bone walls in healing response. J. Periodontol. 2004, 75, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Seunarine, K.; Gadegaard, N.; Tormen, M.; Meredith, D.O.; Riehle, M.O.; Wilkinson, C.D. 3D polymer scaffolds for tissue engineering. Nanomedicine (Lond.) 2006, 1, 281–296. [Google Scholar] [CrossRef] [PubMed]
- Dalby, M.J.; Gadegaard, N.; Tare, R.; Andar, A.; Riehle, M.O.; Herzyk, P.; Wilkinson, C.D.; Oreffo, R.O. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat. Mater. 2007, 6, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Brammer, K.S.; Oh, S.; Gallagher, J.O.; Jin, S. Enhanced cellular mobility guided by TiO2 nanotube surfaces. Nano Lett. 2008, 8, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Angwarawong, T.; Dubas, S.T.; Arksornnukit, M.; Pavasant, P. Differentiation of MC3T3-E1 on poly(4-styrenesulfonic acid-co-maleic acid)sodium salt-coated films. Dent. Mater. J. 2011, 30, 158–169. [Google Scholar] [CrossRef]
- Ayala, R.; Zhang, C.; Yang, D.; Hwang, Y.; Aung, A.; Shroff, S.S.; Arce, F.T.; Lal, R.; Arya, G.; Varghese, S. Engineering the cell-material interface for controlling stem cell adhesion, migration, and differentiation. Biomaterials 2011, 32, 3700–3711. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, Y.; Maeda, H. Recent developments of functional scaffolds for craniomaxillofacial bone tissue engineering applications. Sci. World J. 2013, 2013, 863157. [Google Scholar] [CrossRef]
- Asa’ad, F.; Pagni, G.; Pilipchuk, S.; Giannì, A.; Giannobile, W.; Rasperini, G. 3-D Printed Scaffolds & Biomaterials: Review Alveolar Bone Augmentation & Periodontal Regeneration Applications. Int. J. Dent 2016, 2016, 1239842. [Google Scholar] [CrossRef]
- Park, C.H.; Rios, H.F.; Jin, Q.; Sugai, J.V.; Padial-Molina, M.; Taut, A.D.; Flanagan, C.L.; Hollister, S.J.; Giannobile, W.V. Tissue engineering bone-ligament complexes using fiber-guiding scaffolds. Biomaterials 2012, 33, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Rios, H.F.; Taut, A.D.; Padial-Molina, M.; Flanagan, C.L.; Pilipchuk, S.P.; Hollister, S.J.; Giannobile, W.V. Image-based, fiber guiding scaffolds: A platform for regenerating tissue interfaces. Tissue Eng. Part C Methods 2014, 20, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Rasperini, G.; Pilipchuk, S.P.; Flanagan, C.L.; Park, C.H.; Pagni, G.; Hollister, S.J.; Giannobile, W.V. 3D-printed Bioresorbable Scaffold for Periodontal Repair. J. Dent. Res. 2015, 94 (Suppl. 9), 153S–157S. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.; Buduneli, N.; Cortellini, P. Entire papilla preservation technique: A novel approach for regenerative treatment of deep and wide intrabony defects. Int. J. Periodontics Restor. Dent. 2017, 37, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.; Buduneli, N.; Cortellini, P. Entire papilla preservation technique in the regenerative treatment of deep intrabony defects: 1-year results. J. Clin. Periodontol. 2017, 44, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Harrel, S.K.; Rees, T.D. Granulation tissue removal in routine and minimally invasive surgical procedures. Compend. Contin. Educ. Dent. 1995, 16, 960–967. [Google Scholar] [PubMed]
- Cortellini, P.; Tonetti, M. A minimally invasive surgical technique with an enamel matrix derivative in the regenerative treatment of intra-bony defects: A novel approach to limit morbidity. J. Clin. Periodontol. 2007, 34, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Stavropoulos, A.; Chiantells, G.; Costa, D.; Steigmann, M.; Windisch, P.; Scluean, A. Clinical and histologic evaluation of a granular bovine bone biomaterial used as an adjunct to GTR with a bioresorbable bovine pericardium membrane in the treatment of intrabony defects. J. Periodontol. 2011, 82, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Tal, H. The prevalence and distribution of intrabony defects in dry mandibles. J. Periodontol. 1984, 55, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Vrotsos, J.A.; Parashis, A.O.; Theofanatos, G.D.; Smulow, J.B. Prevalence and distribution of bone defects in moderate and advanced adult periodontitis. J. Clin. Periodontol. 1999, 26, 44–48. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ausenda, F.; Rasperini, G.; Acunzo, R.; Gorbunkova, A.; Pagni, G. New Perspectives in the Use of Biomaterials for Periodontal Regeneration. Materials 2019, 12, 2197. https://doi.org/10.3390/ma12132197
Ausenda F, Rasperini G, Acunzo R, Gorbunkova A, Pagni G. New Perspectives in the Use of Biomaterials for Periodontal Regeneration. Materials. 2019; 12(13):2197. https://doi.org/10.3390/ma12132197
Chicago/Turabian StyleAusenda, Federico, Giulio Rasperini, Raffaele Acunzo, Angelina Gorbunkova, and Giorgio Pagni. 2019. "New Perspectives in the Use of Biomaterials for Periodontal Regeneration" Materials 12, no. 13: 2197. https://doi.org/10.3390/ma12132197
APA StyleAusenda, F., Rasperini, G., Acunzo, R., Gorbunkova, A., & Pagni, G. (2019). New Perspectives in the Use of Biomaterials for Periodontal Regeneration. Materials, 12(13), 2197. https://doi.org/10.3390/ma12132197




