Powder Pressed Cuprous Iodide (CuI) as A Hole Transporting Material for Perovskite Solar Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Solar Cell Fabrication
2.2.1. CuI Powder Pressing
2.2.2. Deposition of Spiro-OMeTAD
2.3. Electrical Characterization
2.4. Structural Characterization
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Juarez-Perez, E.J.; Wang, S.; Ono, L.K.; Qi, Y.; Leyden, M.R.; Hawash, Z. Role of the Dopants on the Morphological and Transport Properties of Spiro-MeOTAD Hole Transport Layer. Chem. Mater. 2016, 28, 5702–5709. [Google Scholar] [CrossRef]
- Jung, H.S.; Park, N.G. Perovskite solar cells: From materials to devices. Small 2015, 11, 10–25. [Google Scholar] [CrossRef]
- Velauthapillai, D.; Rangasamy, B.; Natarajan, M.; Santhanam, A.; Selvaraj, Y.; Asokan, V.; Sundaram, S.; Yuvapragasam, A.; Madurai Ramakrishnan, V.; Pitchaiya, S. The Performance of CH3NH3PbI3 - Nanoparticles based – Perovskite Solar Cells Fabricated by Facile Powder press Technique. Mater. Res. Bull. 2018, 108, 61–72. [Google Scholar]
- Zhou, H.; Shi, Y.; Dong, Q.; Zhang, H.; Xing, Y.; Wang, K.; Du, Y.; Ma, T. Hole-Conductor-Free, Metal-Electrode-Free TiO2/CH3NH3PbI3 Heterojunction Solar Cells Based on a Low-Temperature Carbon Electrode. J. Phys. Chem. Lett. 2014, 3241–3246. [Google Scholar] [CrossRef]
- Cheng, Y.; So, F.; Tsang, S.-W. Progress in air-processed perovskite solar cells: from crystallization to photovoltaic performance. Mater. Horizons 2019. [Google Scholar] [CrossRef]
- Hua, Y.; Liu, P.; Li, Y.; Sun, L.; Kloo, L. Composite Hole-Transport Materials Based on a Metal-Organic Copper Complex and Spiro-OMeTAD for Efficient Perovskite Solar Cells. Sol. RRL 2018, 2, 1700073. [Google Scholar] [CrossRef]
- Kumara, G.R.A.; Konno, A.; Shiratsuchi, K.; Tsukahara, J.; Tennakone, K. Dye-sensitized solid-state solar cells: Use of crystal growth inhibitors for deposition of the hole collector. Chem. Mater. 2002, 14, 954–955. [Google Scholar] [CrossRef]
- Tennakone, K.; Kumara, G.R.R.A.; Kottegoda, I.R.M.; Wijayantha, K.G.U.; Perera, V.P.S. A solid-state photovoltaic cell sensitized with a ruthenium bipyridyl complex. J. Phys. D. Appl. Phys. 1998, 31, 1492–1496. [Google Scholar] [CrossRef]
- Tennakone, K.; Kumara, G.R.R.A.; Kottegoda, I.R.M.; Wijayantha, K.G.U. The photostability of dye-sensitized solid state photovoltaic cells: Factors determining the stability of the pigment in a nanoporous n-TiO2/cyanidin/p-Cul cell. Semicond. Sci. Technol. 1997, 12, 128–132. [Google Scholar] [CrossRef]
- Tennakone, K.; Kumara, G.R.R.A.; Wijayantha, K.G.U. The suppression of the recombination of photogenerated carriers in a dye-sensitized nano-porous solid-state photovoltaic cell. Semicond. Sci. Technol. 1996, 11, 1737–1739. [Google Scholar] [CrossRef]
- Search, H.; Journals, C.; Contact, A.; Iopscience, M.; Address, I.P. A dye-sensitized nano-porous solid-state photovoltaic cell. Semicond. Sci. Technol. 1995, 10, 1689. [Google Scholar]
- Yamada, N.; Ino, R.; Ninomiya, Y. Truly Transparent p-Type γ-CuI Thin Films with High Hole Mobility. Chem. Mater. 2016, 28, 4971–4981. [Google Scholar] [CrossRef]
- Kumara, G.R.A.; Okuya, M.; Murakami, K.; Kaneko, S.; Jayaweera, V.V.; Tennakone, K. Dye-sensitized solid-state solar cells made from magnesiumoxide-coated nanocrystalline titanium dioxide films: Enhancement of the efficiency. J. Photochem. Photobiol. A Chem. 2004, 164, 183–185. [Google Scholar] [CrossRef]
- Kumara, G.R.A.; Kaneko, S.; Okuya, M.; Tennakone, K. Fabrication of Dye-Sensitized Solar Cells Using Triethylamine Hydrothiocyanate as a CuI Crystal Growth Inhibitor. Langmuir 2002, 18, 10493–10495. [Google Scholar] [CrossRef]
- Christians, J.A.; Fung, R.C.M.; Kamat, P.V. An inorganic hole conductor for Organo-lead halide perovskite solar cells. improved hole conductivity with copper iodide. J. Am. Chem. Soc. 2014, 136, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Tetreault, N.; Nazeeruddin, M.K.; Tanaka, S.; Grätzel, M.; Qin, P.; Ito, S.; Manabe, K.; Nishino, H. Inorganic hole conductor-based lead halide perovskite solar cells with 12.4% conversion efficiency. Nat. Commun. 2014, 5, 1–6. [Google Scholar]
- Huangfu, M.; Shen, Y.; Zhu, G.; Xu, K.; Cao, M.; Gu, F.; Wang, L. Copper iodide as inorganic hole conductor for perovskite solar cells with different thickness of mesoporous layer and hole transport layer. Appl. Surf. Sci. 2015, 357, 2234–2240. [Google Scholar] [CrossRef]
- Qin, T.; Lv, S.; Yu, Y.; Zhang, J.; Liu, Z.; Wei, F.; Xu, H.; Cui, G.; Pang, S.; Hu, H. NH2CH═NH2PbI3: An Alternative Organolead Iodide Perovskite Sensitizer for Mesoscopic Solar Cells. Chem. Mater. 2014, 26, 1485–1491. [Google Scholar]
- Sepalage, G.A.; Meyer, S.; Pascoe, A.; Scully, A.D.; Huang, F.; Bach, U.; Cheng, Y.B.; Spiccia, L. Copper(I) Iodide as Hole-Conductor in Planar Perovskite Solar Cells: Probing the Origin of J-V Hysteresis. Adv. Funct. Mater. 2015, 25, 5650–5661. [Google Scholar] [CrossRef]
- Kokubun, Y.; Watanabe, H.; Wada, M. Electrical Properties of CuI Thin Films. Jpn. J. Appl. Phys. 1971, 10, 864. [Google Scholar] [CrossRef]
- Wang, H.; Yu, Z.; Jiang, X.; Li, J.; Cai, B.; Yang, X.; Sun, L. Efficient and Stable Inverted Planar Perovskite Solar Cells Employing CuI as Hole-Transporting Layer Prepared by Solid–Gas Transformation. Energy Technol. 2017, 5, 1836–1843. [Google Scholar] [CrossRef]
- Gharibzadeh, S.; Nejand, B.A.; Moshaii, A.; Mohammadian, N.; Alizadeh, A.H.; Mohammadpour, R.; Ahmadi, V.; Alizadeh, A. Two-step physical deposition of a compact cui hole-transport layer and the formation of an interfacial species in perovskite solar cells. ChemSusChem 2016, 9, 1929–1937. [Google Scholar] [CrossRef] [PubMed]
- Marshall, K.P.; Walton, R.I.; Hatton, R.A. Tin perovskite/fullerene planar layer photovoltaics: Improving the efficiency and stability of lead-free devices. J. Mater. Chem. A 2015, 3, 11631–11640. [Google Scholar] [CrossRef]
- Chen, D.; Wang, Y.; Lin, Z.; Huang, J.; Chen, X.; Pan, D.; Huang, F. Growth strategy and physical properties of the high mobility p-type cui crystal. Cryst. Growth Des. 2010, 10, 2057–2060. [Google Scholar] [CrossRef]
- Li, M.H.; Hsu, C.W.; Shen, P.S.; Cheng, H.M.; Chi, Y.; Chen, P.; Guo, T.F. Novel spiro-based hole transporting materials for efficient perovskite solar cells. Chem. Commun. 2015, 51, 15518–15521. [Google Scholar] [CrossRef] [PubMed]
- Poplavskyy, D.; Nelson, J. Nondispersive hole transport in amorphous films of methoxy-spirofluorene-arylamine organic compound. J. Appl. Phys. 2003, 93, 341–346. [Google Scholar] [CrossRef]
- Schmidt-Mende, L.; Grätzel, M. TiO2 pore-filling and its effect on the efficiency of solid-state dye-sensitized solar cells. Thin Solid Films 2006, 500, 296–301. [Google Scholar] [CrossRef]
- Kaushik, D.K.; Selvaraj, M.; Ramu, S.; Subrahmanyam, A. Thermal evaporated Copper Iodide (CuI) thin films: A note on the disorder evaluated through the temperature dependent electrical properties. Sol. Energy Mater. Sol. Cells 2017, 165, 52–58. [Google Scholar] [CrossRef]
- Cao, G.; Huang, F.; Tian, J.; Siffalovic, P.; Li, M. From scalable solution fabrication of perovskite films towards commercialization of solar cells. Energy Environ. Sci. 2018. [Google Scholar]
- Nazari, P.; Ansari, F.; Abdollahi Nejand, B.; Ahmadi, V.; Payandeh, M.; Salavati-Niasari, M. Physicochemical Interface Engineering of CuI/Cu as Advanced Potential Hole-Transporting Materials/Metal Contact Couples in Hysteresis-Free Ultralow-Cost and Large-Area Perovskite Solar Cells. J. Phys. Chem. C 2017, 121, 21935–21944. [Google Scholar] [CrossRef]
- Thanihaichelvan, M.; Sockiah, K.; Balashangar, K.; Ravirajan, P. Cadmium sulfide interface layer for improving the performance of titanium dioxide/poly (3-hexylthiophene) solar cells by extending the spectral response. J. Mater. Sci. Mater. Electron. 2015, 26, 3558–3563. [Google Scholar] [CrossRef]
- Loheeswaran, S.; Balashangar, K.; Jevirshan, J.; Ravirajan, P. Controlling Recombination Kinetics of Hybrid Nanocrystalline Titanium Dioxide/Polymer Solar Cells by Inserting an Alumina Layer at the Interface. J. Nanoelectron. Optoelectron. 2013, 8, 484–488. [Google Scholar] [CrossRef]
- Loheeswaran, S.; Thanihaichelvan, M.; Ravirajan, P.; Nelson, J. Controlling recombination kinetics of hybrid poly-3-hexylthiophene (P3HT)/titanium dioxide solar cells by self-assembled monolayers. J. Mater. Sci. Mater. Electron. 2017, 28, 4732–4737. [Google Scholar] [CrossRef]
- Balashangar, K.; Paranthaman, S.; Thanihaichelvan, M.; Amalraj, P.A.; Velauthapillai, D.; Ravirajan, P. Multi-walled carbon nanotube incorporated nanoporous titanium dioxide electrodes for hybrid polymer solar cells. Mater. Lett. 2018, 219, 265–268. [Google Scholar] [CrossRef]
- Cojocaru, L.; Uchida, S.; Jayaweera, P.V.V.; Kaneko, S.; Wang, H.; Nakazaki, J.; Kubo, T.; Segawa, H. Effect of TiO2 Surface Treatment on the Current–Voltage Hysteresis of Planar-Structure Perovskite Solar Cells Prepared on Rough and Flat Fluorine-Doped Tin Oxide Substrates. Energy Technol. 2017, 5, 1762–1766. [Google Scholar] [CrossRef]
- Zhu, B.L.; Zhao, X.Z. Transparent conductive CuI thin films prepared by pulsed laser deposition. Phys. Status Solidi Appl. Mater. Sci. 2011, 208, 91–96. [Google Scholar] [CrossRef]
- Yue, S.; Gu, M.; Liu, X.; Zhang, J.; Huang, S.; Liu, B.; Ni, C. Effect of ZnI2 cosolute on quality and performance of γ-CuI ultrafast scintillation crystal grown via evaporation method in acetonitrile solvent. Opt. Mater. (Amst). 2017, 66, 308–313. [Google Scholar] [CrossRef]
- Madhavan, V.; Ali, A.; Baloch, B.; Manekkathodi, A.; Zimmermann, I.; Roldán-carmona, C.; Grancini, G.; Buffiere, M.; Nazeeruddin, M.K.; Belaidi, A.; et al. CuI and CuSCN as Hole Transport Materials for Perovskite Solar Cells. In Qatar Foundation Annual Research Conference Proceedings Volume 2018 Issue 1; Hamad bin Khalifa University Press (HBKU Press): Doha, Qatar, 2018. [Google Scholar]
- Liu, M.; Endo, M.; Shimazaki, A.; Wakamiya, A.; Tachibana, Y. Identifying an Optimum Perovskite Solar Cell Structure by Kinetic Analysis: Planar, Mesoporous Based, or Extremely Thin Absorber Structure. ACS Appl. Energy Mater. 2018, 1, 3722–3732. [Google Scholar] [CrossRef]
- Jeon, N.J.; Lee, J.; Noh, J.H.; Nazeeruddin, M.K.; Grätzel, M.; Seok, S.I. Efficient inorganic-organic hybrid perovskite solar cells based on pyrene arylamine derivatives as hole-transporting materials. J. Am. Chem. Soc. 2013, 135, 19087–19090. [Google Scholar] [CrossRef]
- Zhu, X.D.; Ma, X.J.; Wang, Y.K.; Li, Y.; Gao, C.H.; Wang, Z.K.; Jiang, Z.Q.; Liao, L.S. Hole-Transporting Materials Incorporating Carbazole into Spiro-Core for Highly Efficient Perovskite Solar Cells. Adv. Funct. Mater. 2019, 29, 1–8. [Google Scholar] [CrossRef]
- Ke, W.; Fang, G.; Wang, J.; Qin, P.; Tao, H.; Lei, H.; Liu, Q.; Dai, X.; Zhao, X. Perovskite solar cell with an efficient TiO2 compact film. ACS Appl. Mater. Interfaces 2014, 6, 15959–15965. [Google Scholar] [CrossRef] [PubMed]
- Nouri, E.; Mohammadi, M.R.; Lianos, P. Inverted perovskite solar cells based on lithium-functionalized graphene oxide as an electron-transporting layer. Chem. Commun. 2017, 53, 1630–1633. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Y.; Deng, L.L.; Dai, S.M.; Wang, X.; Tian, C.B.; Zhan, X.X.; Xie, S.Y.; Huang, R.B.; Zheng, L.S. Low-cost solution-processed copper iodide as an alternative to PEDOT:PSS hole transport layer for efficient and stable inverted planar heterojunction perovskite solar cells. J. Mater. Chem. A 2015, 3, 19353–19359. [Google Scholar] [CrossRef]
- Li, Z.; Chen, J.; Li, H.; Zhang, Q.; Chen, Z.; Zheng, X.; Fang, G.; Wang, H.; Hao, Y. A facilely synthesized “spiro” hole-transporting material based on spiro[3.3]heptane-2,6-dispirofluorene for efficient planar perovskite solar cells. RSC Adv. 2017, 7, 41903–41908. [Google Scholar] [CrossRef]
- Gheno, A.; Vedraine, S.; Ratier, B.; Bouclé, J. π-Conjugated Materials as the Hole-Transporting Layer in Perovskite Solar Cells. Metals 2016, 6, 21. [Google Scholar] [CrossRef]
- Yang, L.; Barrows, A.T.; Lidzey, D.G. Perovskite solar cells with CuI inorganic hole conductor of Ti thin film solvent annealing treatment method. Jpn. J. Appl. Phys. 2017, 56, 08MC04. [Google Scholar]
- Karuppuchamy, S.; Murugadoss, G.; Ramachandran, K.; Saxena, V.; Thangamuthu, R. Inorganic based hole transport materials for perovskite solar cells. J. Mater. Sci. Mater. Electron. 2018, 29, 8847–8853. [Google Scholar] [CrossRef]

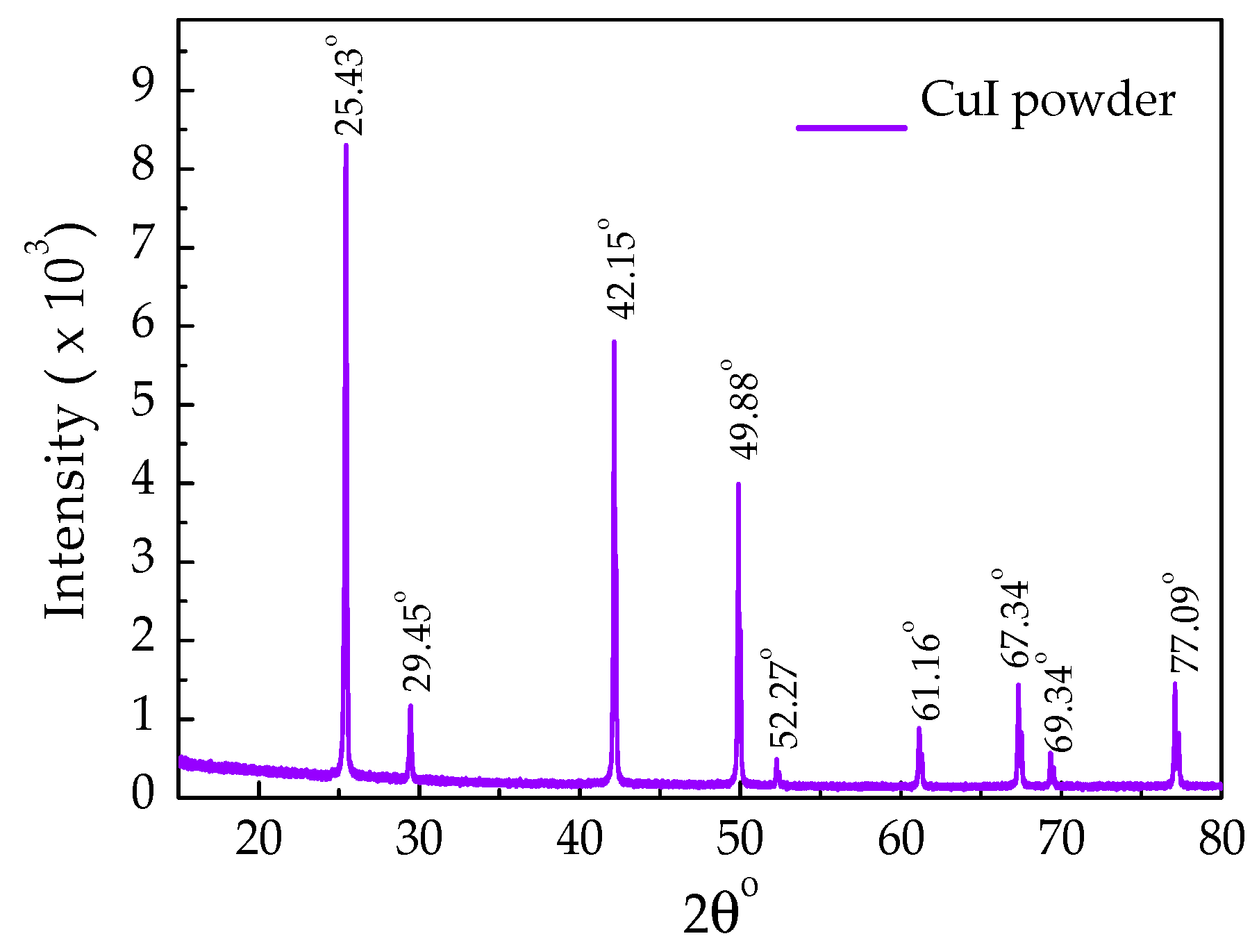
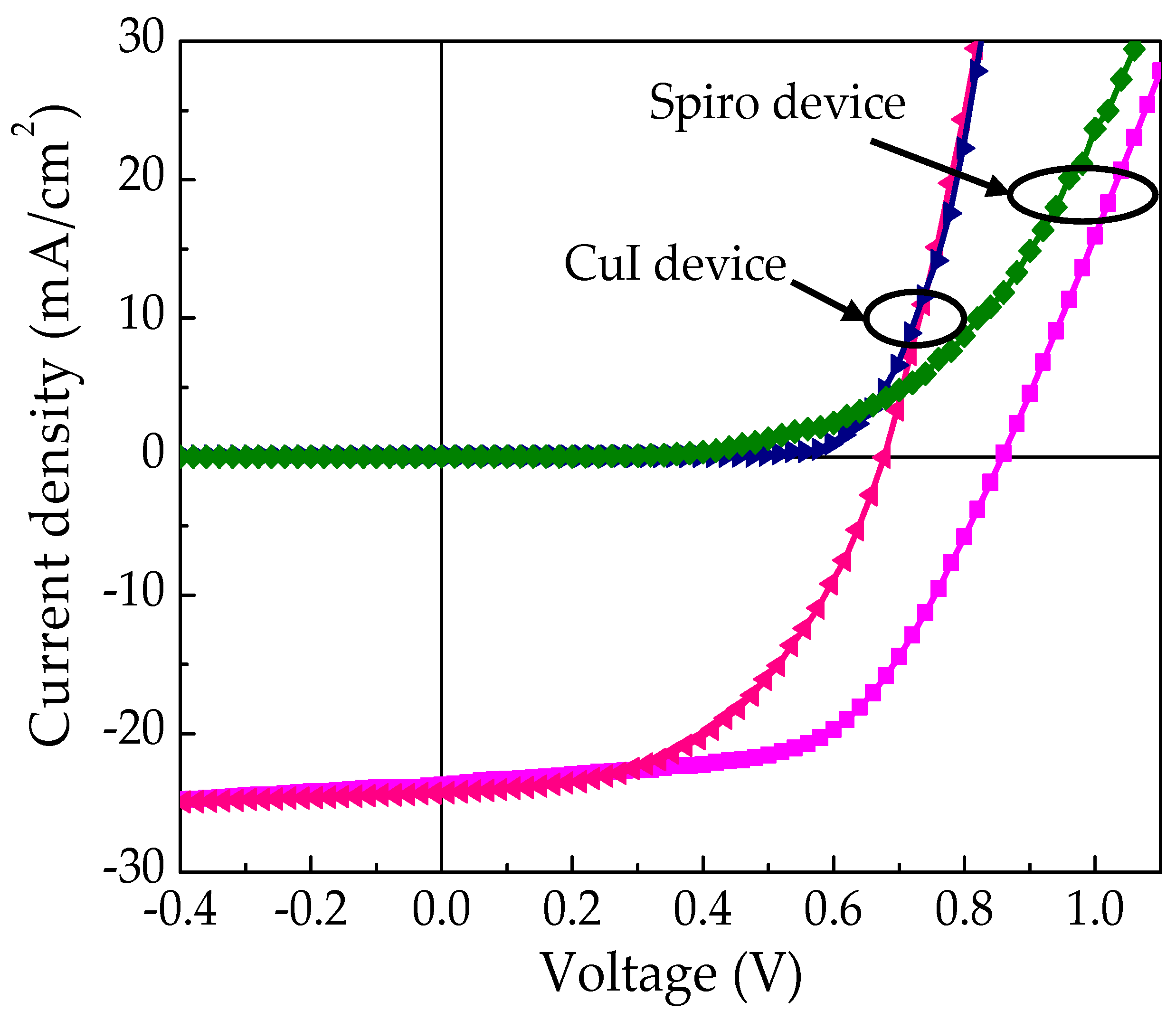
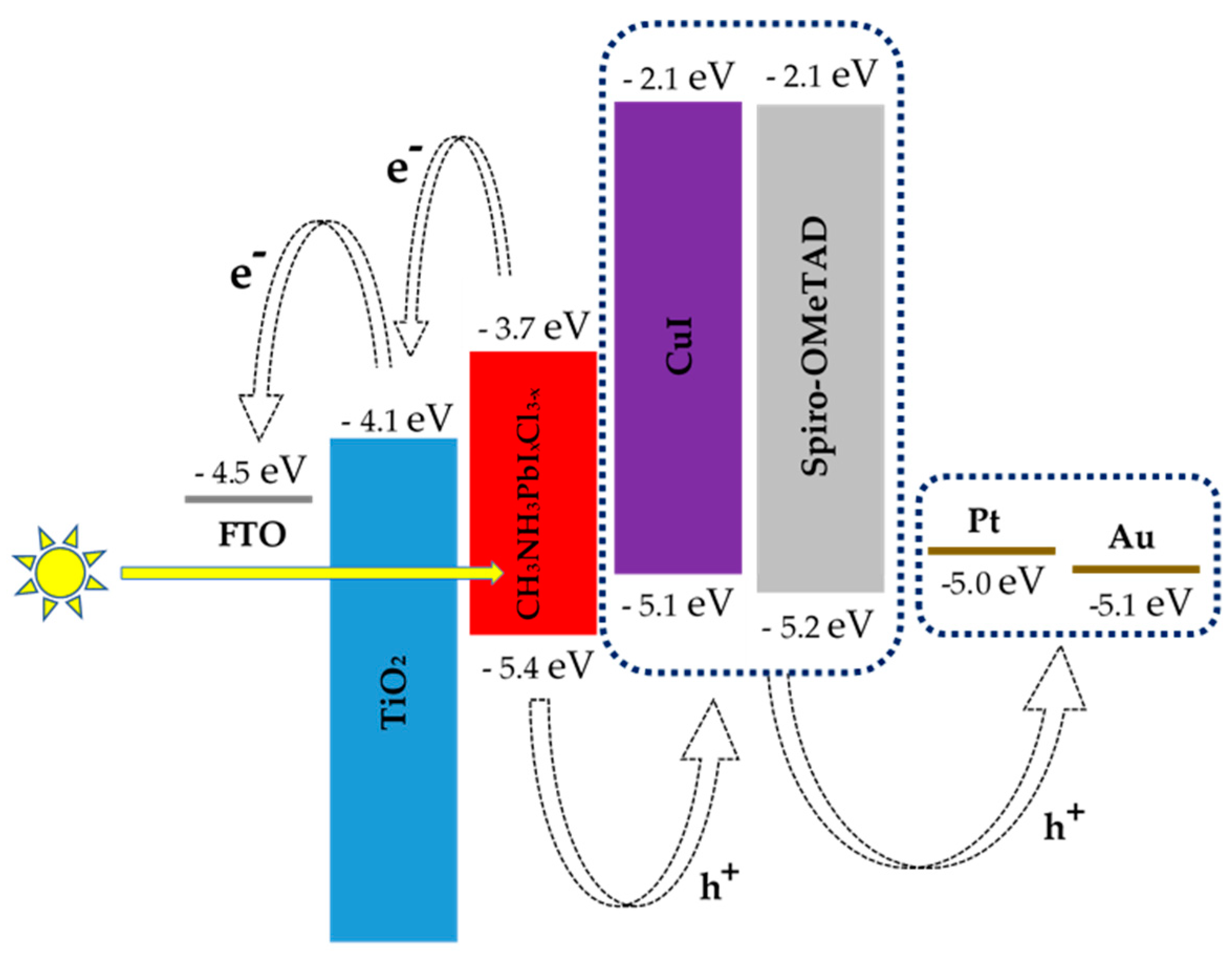
| Hole Transporting Material (HTM) | JSC (mA/cm2) | VOC (V) | FF | PCE (ŋ%) |
|---|---|---|---|---|
| Spiro-OMeTAD | 22.4 ± 1.7 23.7 | 0.79 ± 0.03 0.85 | 0.56 ± 0.07 0.59 | 9.9 ± 1.3 11.9 |
| CuI | 24.09 ± 1.4 24.23 | 0.66 ± 0.02 0.67 | 0.49 ± 0.03 0.50 | 7.8 ± 0.3 8.1 |
| Device Structure | CuI Deposition Method | Jsc (mA/cm2) | VOC (V) | FF | PCE (ŋ%) | Reference |
|---|---|---|---|---|---|---|
| TiO2/CH3NH3PbI3/CuI | Solution pumping process | 17.8 | 0.55 | 0.62 | 6.0 | [15] |
| TiO2/CH3NH3PbIxCl3−x/CuI | Spray coating method | 22.3 | 0.61 | 0.42 | 5.8 | [17] |
| TiO2/CH3NH3PbI3/CuI | Spin coating method | 14.7 | 0.42 | 0.40 | 2.2 | [47] |
| TiO2/CH3NH3PbI3/CuI | Doctor blading method | 16.7 | 0.78 | 0.57 | 7.5 | [19] |
| TiO2/CH3NH3PbI3/CuI | Gas-solid treatment | 32.7 | 0.73 | 0.31 | 7.4 | [22] |
| CuI/CH3NH3PbI3/PCBM | Doctor blading method | 12.3 | 0.57 | 0.47 | 3.4 | [48] |
| CuI/CsSnI3/C60/BCP | Thermal evaporation | 8.94 | 0.36 | 0.54 | 2.1 | [23] |
| TiO2/CH3NH3PbI3/CuI/Cu | Thermal evaporation | 23.0 | 0.85 | 0.47 | 9.2 | [30] |
| TiO2/CH3NH3PbIxCl3−x/CuI | Powder pressing method | 24.23 | 0.67 | 0.50 | 8.1 | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uthayaraj, S.; Karunarathne, D.G.B.C.; Kumara, G.R.A.; Murugathas, T.; Rasalingam, S.; Rajapakse, R.M.G.; Ravirajan, P.; Velauthapillai, D. Powder Pressed Cuprous Iodide (CuI) as A Hole Transporting Material for Perovskite Solar Cells. Materials 2019, 12, 2037. https://doi.org/10.3390/ma12132037
Uthayaraj S, Karunarathne DGBC, Kumara GRA, Murugathas T, Rasalingam S, Rajapakse RMG, Ravirajan P, Velauthapillai D. Powder Pressed Cuprous Iodide (CuI) as A Hole Transporting Material for Perovskite Solar Cells. Materials. 2019; 12(13):2037. https://doi.org/10.3390/ma12132037
Chicago/Turabian StyleUthayaraj, Siva, D. G. B. C. Karunarathne, G. R. A. Kumara, Thanihaichelvan Murugathas, Shivatharsiny Rasalingam, R. M. G. Rajapakse, Punniamoorthy Ravirajan, and Dhayalan Velauthapillai. 2019. "Powder Pressed Cuprous Iodide (CuI) as A Hole Transporting Material for Perovskite Solar Cells" Materials 12, no. 13: 2037. https://doi.org/10.3390/ma12132037
APA StyleUthayaraj, S., Karunarathne, D. G. B. C., Kumara, G. R. A., Murugathas, T., Rasalingam, S., Rajapakse, R. M. G., Ravirajan, P., & Velauthapillai, D. (2019). Powder Pressed Cuprous Iodide (CuI) as A Hole Transporting Material for Perovskite Solar Cells. Materials, 12(13), 2037. https://doi.org/10.3390/ma12132037







