Macro-Encapsulation of Inorganic Phase-Change Materials (PCM) in Metal Capsules
Abstract
1. Introduction
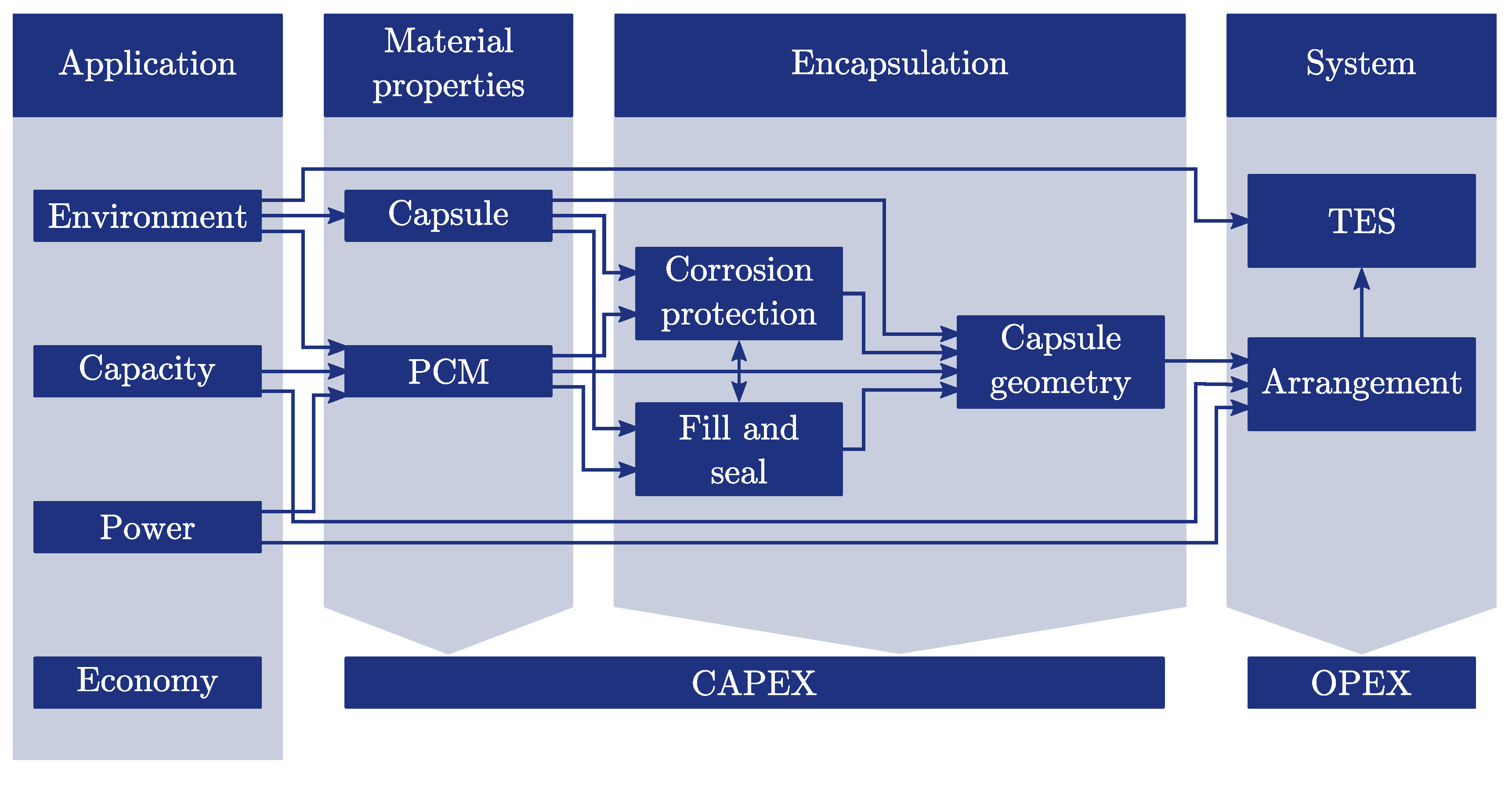
2. Analysis of the Development Process of Macro-Encapsulated TES
2.1. Application
2.2. Material Properties
2.3. Encapsulation
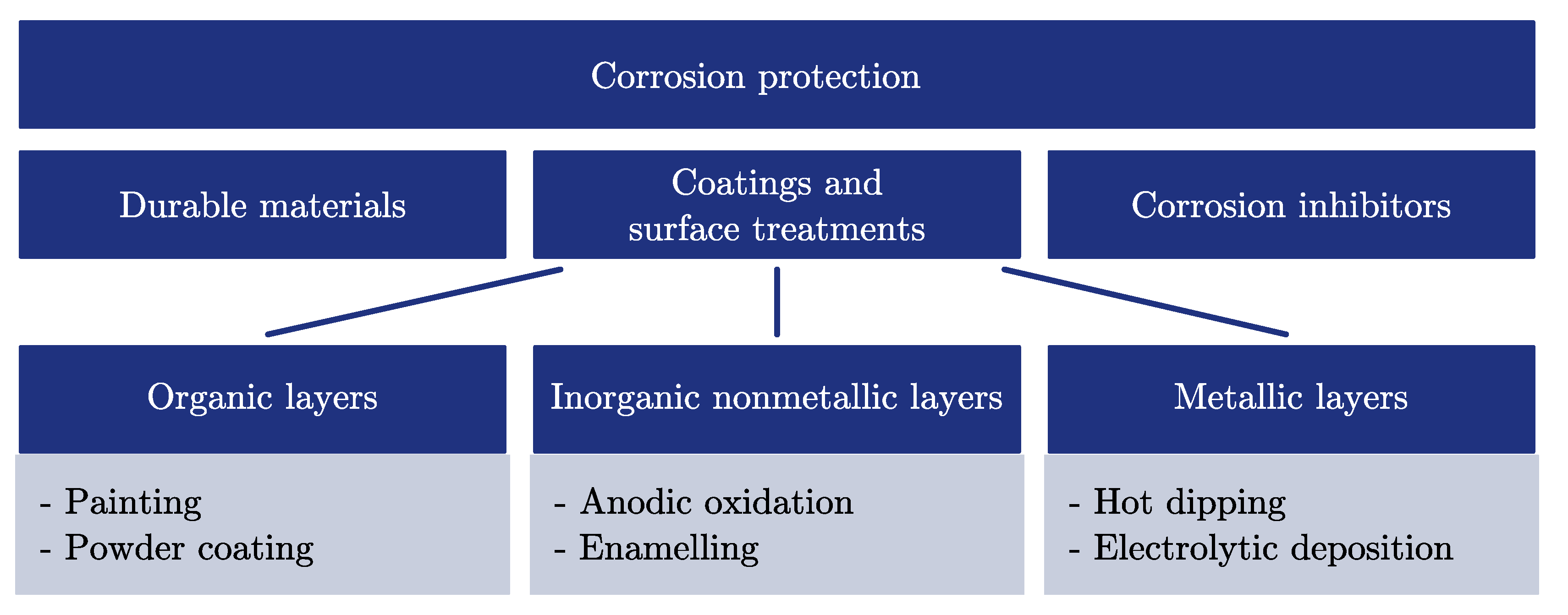
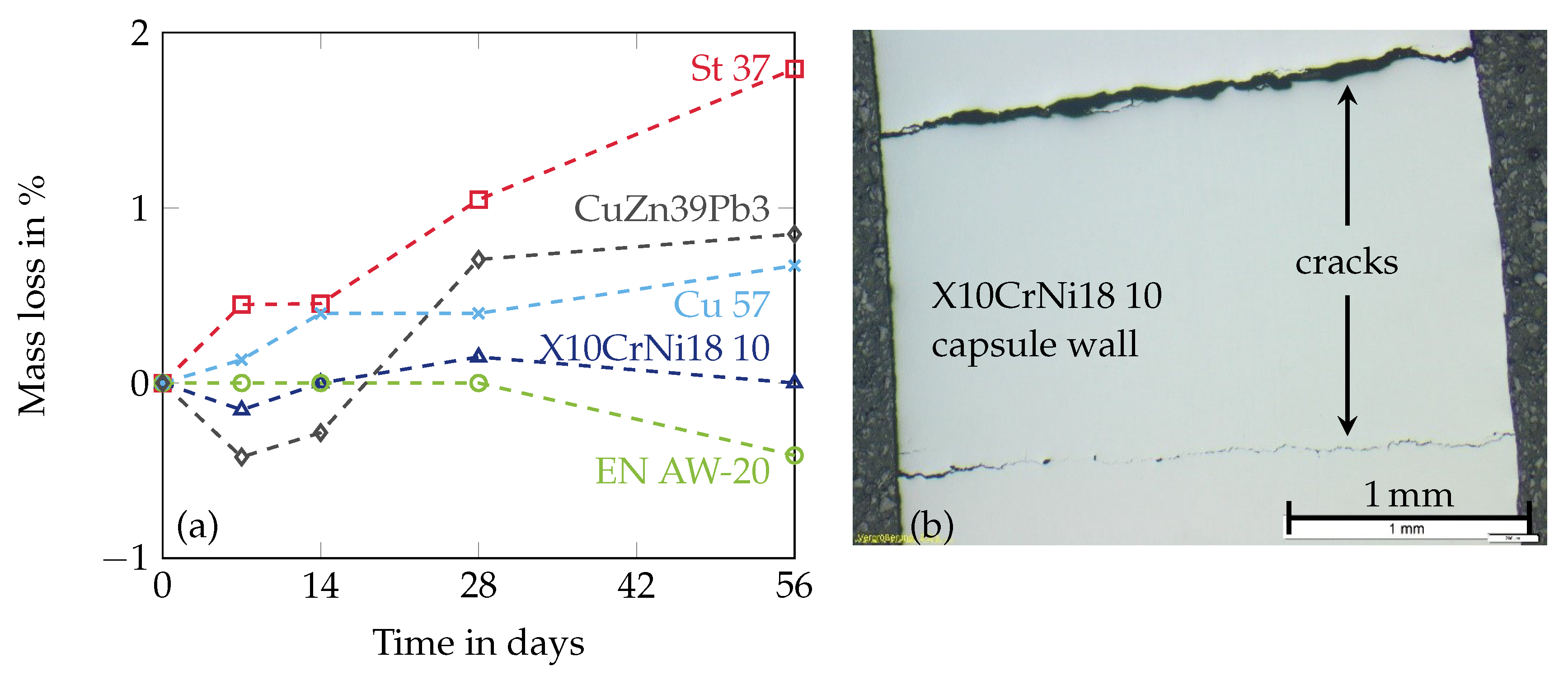
2.3.1. Corrosion Protection
Durable Materials
Coatings and Surface Treatments
Corrosion Inhibitors
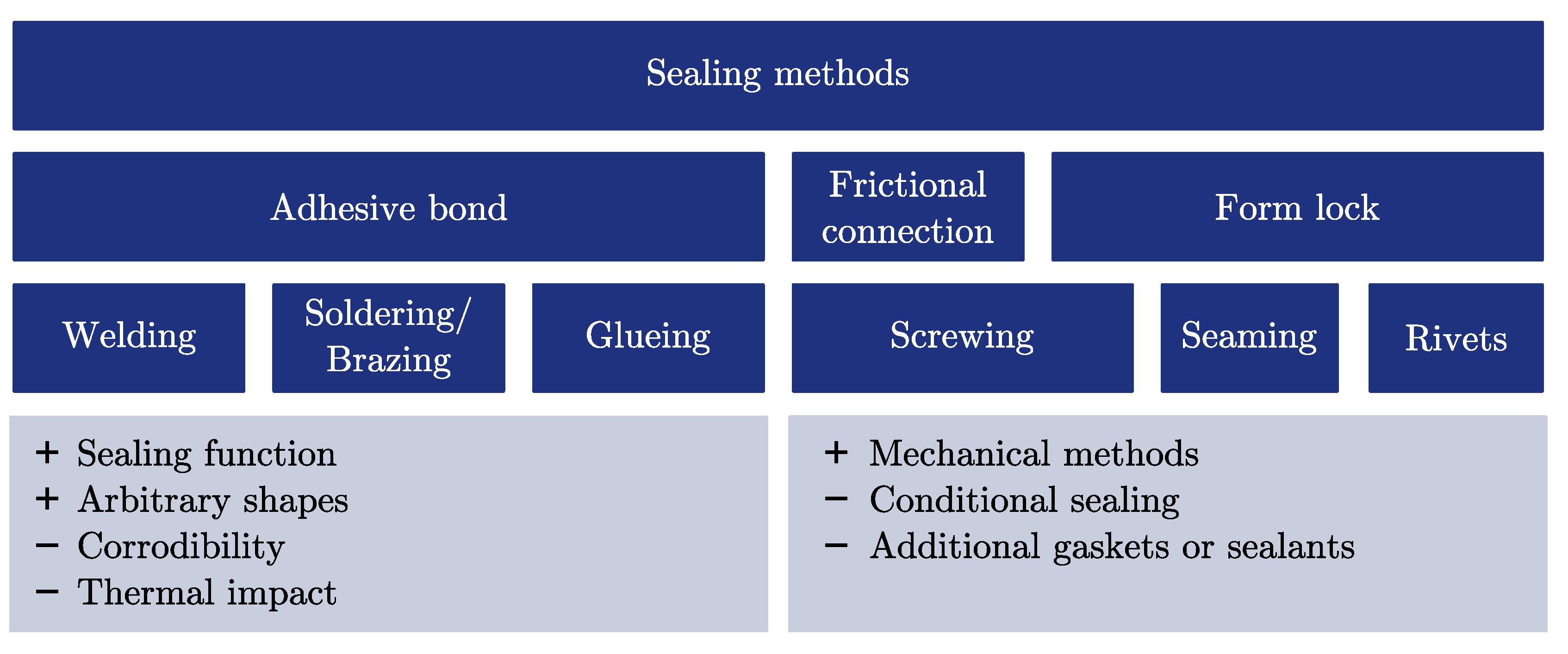
2.3.2. Fill and Seal
2.3.3. Capsule Geometry
2.4. System
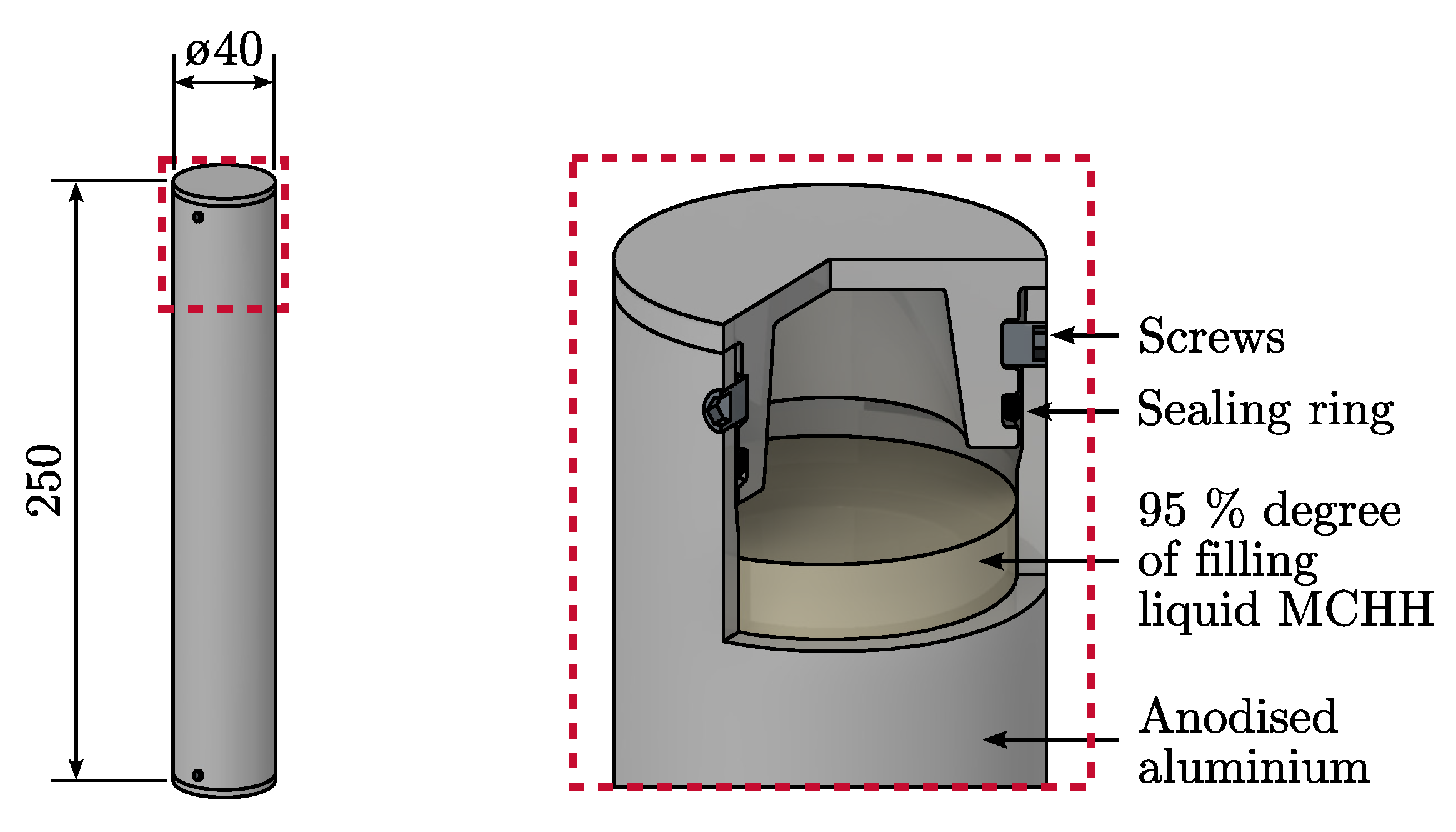
3. Exemplary Implementation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CAPEX | Capital expenditures |
| MCHH | Magnesiumchloride hexahydrate |
| OPEX | Operational expenditures |
| PCM | phase-change material |
| TES | Thermal energy storage |
References
- Tamme, R.; Bauer, T.; Hahne, E. Heat Storage Media. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009; pp. 285–313. [Google Scholar]
- Farrell, A.J.; Norton, B.; Kennedy, D.M. Corrosive effects of salt hydrate phase change materials used with aluminium and copper. J. Mater. Process. Technol. 2006, 175, 198–205. [Google Scholar] [CrossRef]
- Ushak, S.; Marín, P.; Galazutdinova, Y.; Cabeza, L.F.; Farid, M.M.; Grágeda, M. Compatibility of materials for macroencapsulation of inorganic phase change materials: Experimental corrosion study. Appl. Therm. Eng. 2016, 107, 410–419. [Google Scholar] [CrossRef]
- Porisini, F.C. Salt hydrates used for latent heat storage: Corrosion of metals and reliability of thermal performance. Sol. Energy 1988, 41, 193–197. [Google Scholar] [CrossRef]
- El-Sebaii, A.; Al-Amir, S.; Al-Marzouki, F.; Faidah, A.S.; Al-Ghamdi, A.; Al-Heniti, S. Fast thermal cycling of acetanilide and magnesium chloride hexahydrate for indoor solar cooking. Energy Convers. Manag. 2009, 50, 3104–3111. [Google Scholar] [CrossRef]
- Milián, Y.E.; Gutiérrez, A.; Grágeda, M.; Ushak, S. A review on encapsulation techniques for inorganic phase change materials and the influence on their thermophysical properties. Renew. Sustain. Energy Rev. 2017, 73, 983–999. [Google Scholar] [CrossRef]
- Su, W.; Darkwa, J.; Kokogiannakis, G. Review of solid-liquid phase change materials and their encapsulation technologies. Renew. Sustain. Energy Rev. 2015, 48, 373–391. [Google Scholar] [CrossRef]
- Jacob, R.; Bruno, F. Review on shell materials used in the encapsulation of phase change materials for high temperature thermal energy storage. Renew. Sustain. Energy Rev. 2015, 48, 79–87. [Google Scholar] [CrossRef]
- Cristopia Energy Systems. Thermal Hermal Energy Storage. Available online: http://pdf.medicalexpo.com/pdf/ciat/cristopia/78902-79307.html#open (accessed on 13 September 2018).
- Phase Change Material Products Limited. Thermal Storage. Available online: http://www.pcmproducts.net/files/thermal_storage_catalogue.pdf (accessed on 13 September 2018).
- Höhlein, S.; König-Haagen, A.; Brüggemann, D. Thermophysical Characterization of MgCl2·6H2O, Xylitol and Erythritol as Phase Change Materials (PCM) for Latent Heat Thermal Energy Storage (LHTES). Materials 2017, 10, 444. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Zhao, W.; Sabol, J.C.; Tuzla, K.; Neti, S.; Oztekin, A.; Chen, J.C. Encapsulated phase change materials for energy storage - Characterization by calorimetry. Sol. Energy 2013, 87, 117–126. [Google Scholar] [CrossRef]
- Zhang, H.; Baeyens, J.; Degrève, J.; Cáceres, G.; Segal, R.; Pitié, F. Latent heat storage with tubular-encapsulated phase change materials (PCMs). Energy 2014, 76, 66–72. [Google Scholar] [CrossRef]
- COMSOL Multiphysics. Available online: https://www.comsol.com (accessed on 13 September 2018).
- Blaney, J.J.; Neti, S.; Misiolek, W.Z.; Oztekin, A. Containment capsule stresses for encapsulated phase change materials. Appl. Thermal Eng. 2013, 50, 555–561. [Google Scholar] [CrossRef]
- Lane, G.A. Low temperature heat storage with phase change materials. Int. J. Ambient Energy 1980, 1, 155–168. [Google Scholar] [CrossRef]
- Gonçalves, L.; Probert, S. Thermal-energy storage: Dynamic performance characteristics of cans each containing a phase-change material, assembled as a packed-bed. Appl. Energy 1993, 45, 117–155. [Google Scholar] [CrossRef]
- Ibáñez, M.; Cabeza, L.F.; Solé, C.; Roca, J.; Nogués, M. Modelization of a water tank including a PCM module. Appl. Thermal Eng. 2006, 26, 1328–1333. [Google Scholar] [CrossRef]
- Hauer, A.; Hiebler, S.; Reuß, M. Wärmespeicher, 5th ed.; Fraunhofer IRB Verlag: Stuttgart, Germany, 2013. [Google Scholar]
- Miró, L.; Gasia, J.; Cabeza, L.F. Thermal energy storage (TES) for industrial waste heat (IWH) recovery: A review. Appl. Energy 2016, 179, 284–301. [Google Scholar] [CrossRef]
- Lane, G.A. Phase change materials for energy storage nucleation to prevent supercooling. Sol. Energy Mater. Sol. Cells 1992, 27, 135–160. [Google Scholar] [CrossRef]
- Xie, N.; Huang, Z.; Luo, Z.; Gao, X.; Fang, Y.; Zhang, Z. Inorganic Salt Hydrate for Thermal Energy Storage. Appl. Sci. 2017, 7, 1317. [Google Scholar] [CrossRef]
- Sandnes, B.; Rekstad, J. Supercooling salt hydrates: Stored enthalpy as a function of temperature. Sol. Energy 2006, 80, 616–625. [Google Scholar] [CrossRef]
- Yoshii, Y.; Kuraoka, M.; Sengoku, K.; Ohachi, T. Induction time and three-electrode current vs. voltage characteristics for electrical nucleation of concentrated solutions of sodium acetate trihydrate. J. Cryst. Growth 2002, 237, 414–418. [Google Scholar] [CrossRef]
- Seo, K.; Suzuki, S.; Kinoshita, T.; Hirasawa, I. Effect of Ultrasonic Irradiation on the Crystallization of Sodium Acetate Trihydrate Utilized as Heat Storage Material. Chem. Eng. Technol. 2012, 35, 1013–1016. [Google Scholar] [CrossRef]
- Lane, G.A. Solar Heat Storage: Latent Heat Material Volume 1: Background and Scientific Principles; CRC Press: Boca Raton, FL, USA, 1983. [Google Scholar]
- Rathod, M.K.; Banerjee, J. Thermal stability of phase change materials used in latent heat energy storage systems: A review. Renew. Sustain. Energy Rev. 2013, 18, 246–258. [Google Scholar] [CrossRef]
- Werner, R. Compatibility of organic latent heat storage materials and plastic container materials. Heat Recovery Syst. CHP 1987, 7, 383–388. [Google Scholar] [CrossRef]
- Castellón, C.; Martorell, I.; Cabeza, L.F.; Fernández, A.I.; Manich, A.M. Compatibility of plastic with phase change materials (PCM). Int. J. Energy Res. 2011, 35, 765–771. [Google Scholar] [CrossRef]
- Brüggemann, D.; König-Haagen, A.; Kasibhatla, R.R.; Höhlein, S.; Glatzel, U.; Völkl, R.; Agarkov, N. Entwicklung makroverkapselter Latentwärmespeicher für den straßengebundenen Transport von Abwärme (MALATrans). Final report, Universität Bayreuth, Bayreuth, 2017. Available online: https://doi.org/10.2314/GBV:897196937 (accessed on 13 September 2018).
- Zoch, H.W.; Spur, G. Handbuch Wärmebehandeln und Beschichten; Carl Hanser Verlag: München, Germany, 2015. [Google Scholar]
- Hale, D.V.; Hoover, M.J.; O’Neill, M.J. Phase change materials handbook. Technical report. Lockheed Missiles and Space Company: Huntsville, AL, USA, 1971. [Google Scholar]
- Ostermann, F. Anwendungstechnologie Aluminium; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Jelinek, T.W. Oberflächenbehandlung von Aluminium; Eugen G. Leuze Verlag: Saulgau, Germany, 1997. [Google Scholar]
- Drossel, G.; Friedrich, S.; Huppatz, W.; Kammer, C.; Lehnert, W.; Liesenberg, O.; Mader, W.; Paul, M.; Rudolf, A.; Thate, W.; Zeltner, S.; Wenglorz, W. Aluminium-Taschenbuch / 2. Umformung von Aluminium-Werkstoffen, Giessen von Aluminium-Teilen, Oberflächenbehandlung von Aluminium, Recycling und Ökologie, 16th ed.; Aluminium-Verlag Marketing & Kommunikation GmbH: Düsseldorf, Germany, 2009. [Google Scholar]
- Ashby, M. Materials Selection in Mechanical Design, 4th ed.; Butterworth-Heinemann: Burlington, MA, USA, 2011. [Google Scholar]
- Zhang, Z.; Tian, N.; Zhang, W.; Huang, X.; Ruan, L.; Wu, L. Inhibition of carbon steel corrosion in phase-change-materials solution by methionine and proline. Corros. Sci. 2016, 111, 675–689. [Google Scholar] [CrossRef]
- Fleischer, A.S. Thermal Energy Storage Using Phase Change Materials; SpringerBriefs in Applied Sciences and Technology, Springer International Publishing: Berlin, Germany, 2015. [Google Scholar]
- Dong, Z.; Cui, H.; Tang, W.; Chen, D.; Wen, H. Development of Hollow Steel Ball Macro-Encapsulated PCM for Thermal Energy Storage Concrete. Materials 2016, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- BIKAR METALLE. EN AW-6060. Available online: http://www.bikar.com/fileadmin/download/6060-komplett.pdf (accessed on 13 September 2018).
- LOCTITE. Loctite® EA 9514. Available online: http://www.loctite.de/produktsuche-29727.htm?nodeid=8802629386241 (accessed on 13 September 2018).
- Chemours. Selection Guide. Available online: https://www.chemours.com/Viton/en_US/assets/downloads/viton-selection-guide.pdf (accessed on 13 September 2018).
- Hartmut Bauer Großhandel für Flaschen, Gläser und Konservendosen e.K. Available online: https://www.flaschenbauer.de/konservendosen/?p=1 (accessed on 13 September 2018).

| Property | Value |
|---|---|
| Melting Temperature | 115.1 ± 0.1 |
| Supercooling * | 2.8 |
| Melting enthalpy between 114–118 | 166.9 ± 1.2 |
| Solid state heat capacity at 100 | 1.83 ± 0.06 |
| Liquid state heat capacity at 120 | 2.57 ± 0.06 |
| Solid state density at 20 | 1.5955 ± 0.0002 |
| Liquid state density at 120 | 1.4557 ± 0.0004 |
| Solid state thermal conductivity at 110 | 0.70 ± 0.05 |
| Liquid state thermal conductivity at 120 | 0.63 ± 0.04 |
| Description | Costs Per Capsule in € | Costs Per PCM in € | Share in % |
|---|---|---|---|
| Manufacturing | 8.68 | 44.64 | 48 |
| Capsule material | 4.55 | 23.38 | 25 |
| Corrosion protection | 4.68 | 24.05 | 26 |
| PCM (MCHH) | 0.22 | 1.11 | 1 |
| Total | 18.11 | 93.19 | 100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Höhlein, S.; König-Haagen, A.; Brüggemann, D. Macro-Encapsulation of Inorganic Phase-Change Materials (PCM) in Metal Capsules. Materials 2018, 11, 1752. https://doi.org/10.3390/ma11091752
Höhlein S, König-Haagen A, Brüggemann D. Macro-Encapsulation of Inorganic Phase-Change Materials (PCM) in Metal Capsules. Materials. 2018; 11(9):1752. https://doi.org/10.3390/ma11091752
Chicago/Turabian StyleHöhlein, Stephan, Andreas König-Haagen, and Dieter Brüggemann. 2018. "Macro-Encapsulation of Inorganic Phase-Change Materials (PCM) in Metal Capsules" Materials 11, no. 9: 1752. https://doi.org/10.3390/ma11091752
APA StyleHöhlein, S., König-Haagen, A., & Brüggemann, D. (2018). Macro-Encapsulation of Inorganic Phase-Change Materials (PCM) in Metal Capsules. Materials, 11(9), 1752. https://doi.org/10.3390/ma11091752





