Enamel Anti-Demineralization Effect of Orthodontic Adhesive Containing Bioactive Glass and Graphene Oxide: An In-Vitro Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Bioactive Glass/Graphene Oxide
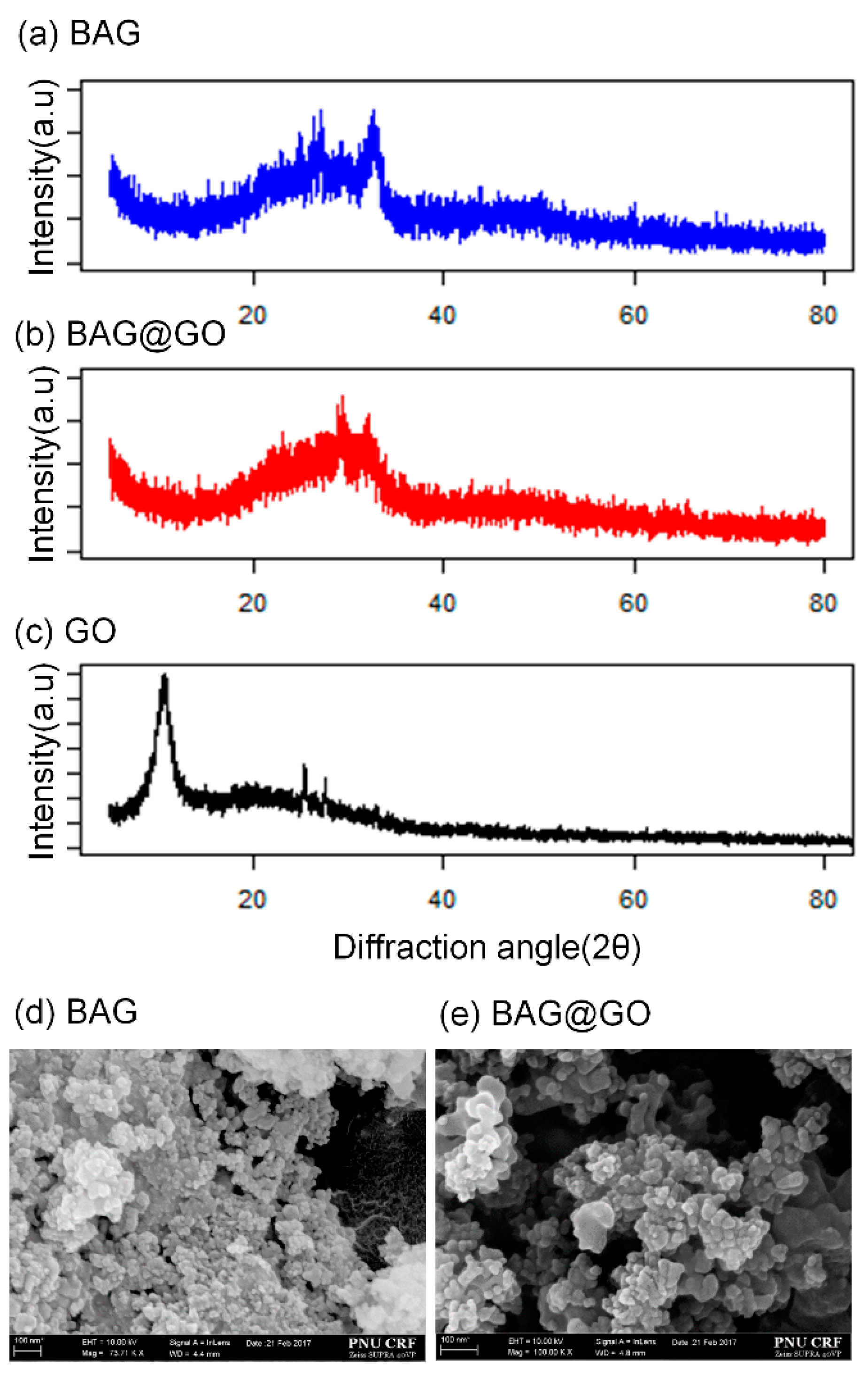
2.2. Characterization of Bioactive Glass/Graphene Oxide
2.3. Mechanical Properties
2.3.1. Disk Preparation for Mechanical Properties
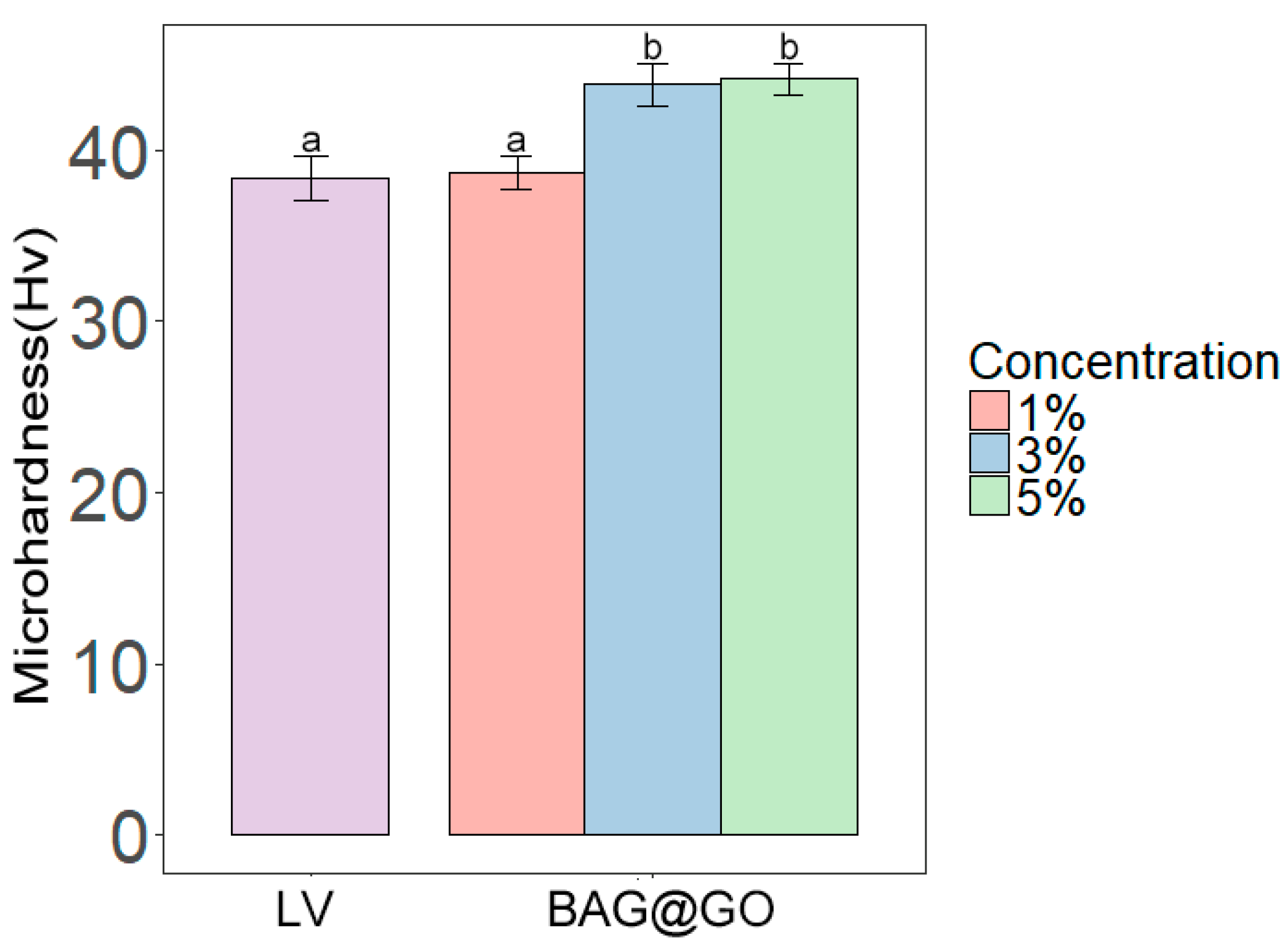
2.3.2. Microhardness
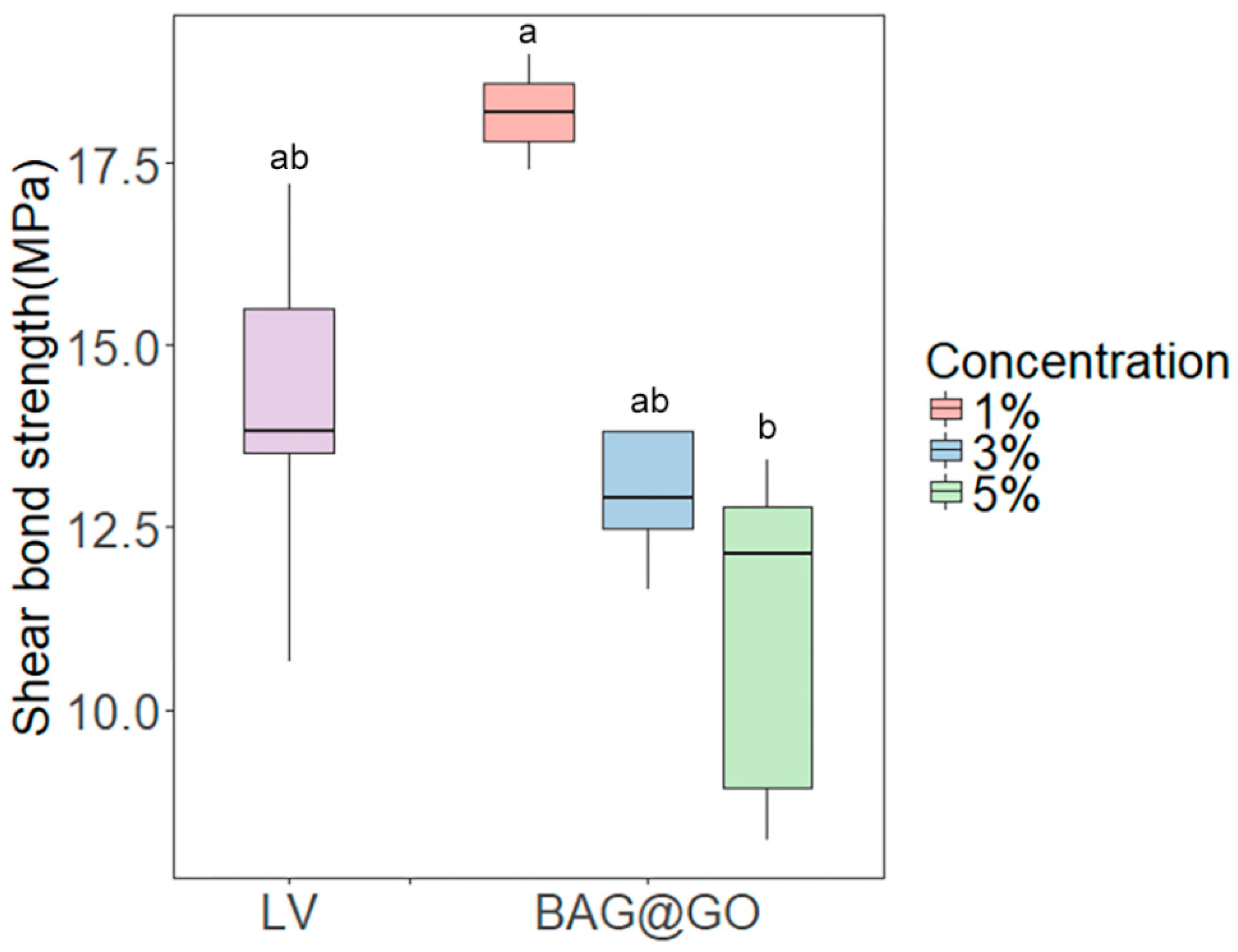
2.3.3. Shear Bond Strength
2.4. Biological Properties
2.4.1. Cell Viability Assay
2.4.2. Antibacterial Test
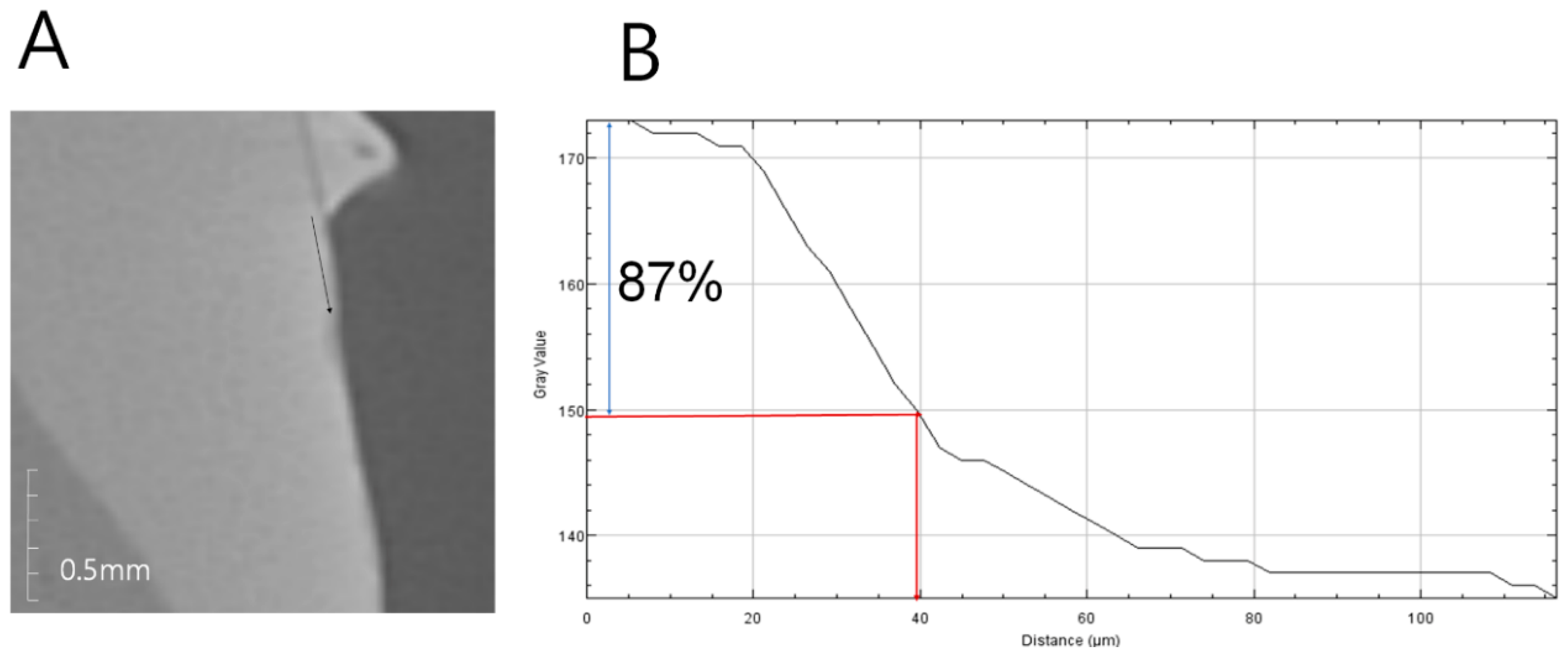
2.5. Anti-Demineralizationtest
2.6. Statistical Analysis
3. Results
3.1. Characterisation of BAG@GO
3.2. Mechanical Properties
3.2.1. Microhardness
3.2.2. Shear Bond Strength
3.2.3. Adhesive Remnant Index (ARI) Score
3.3. Biological Properties
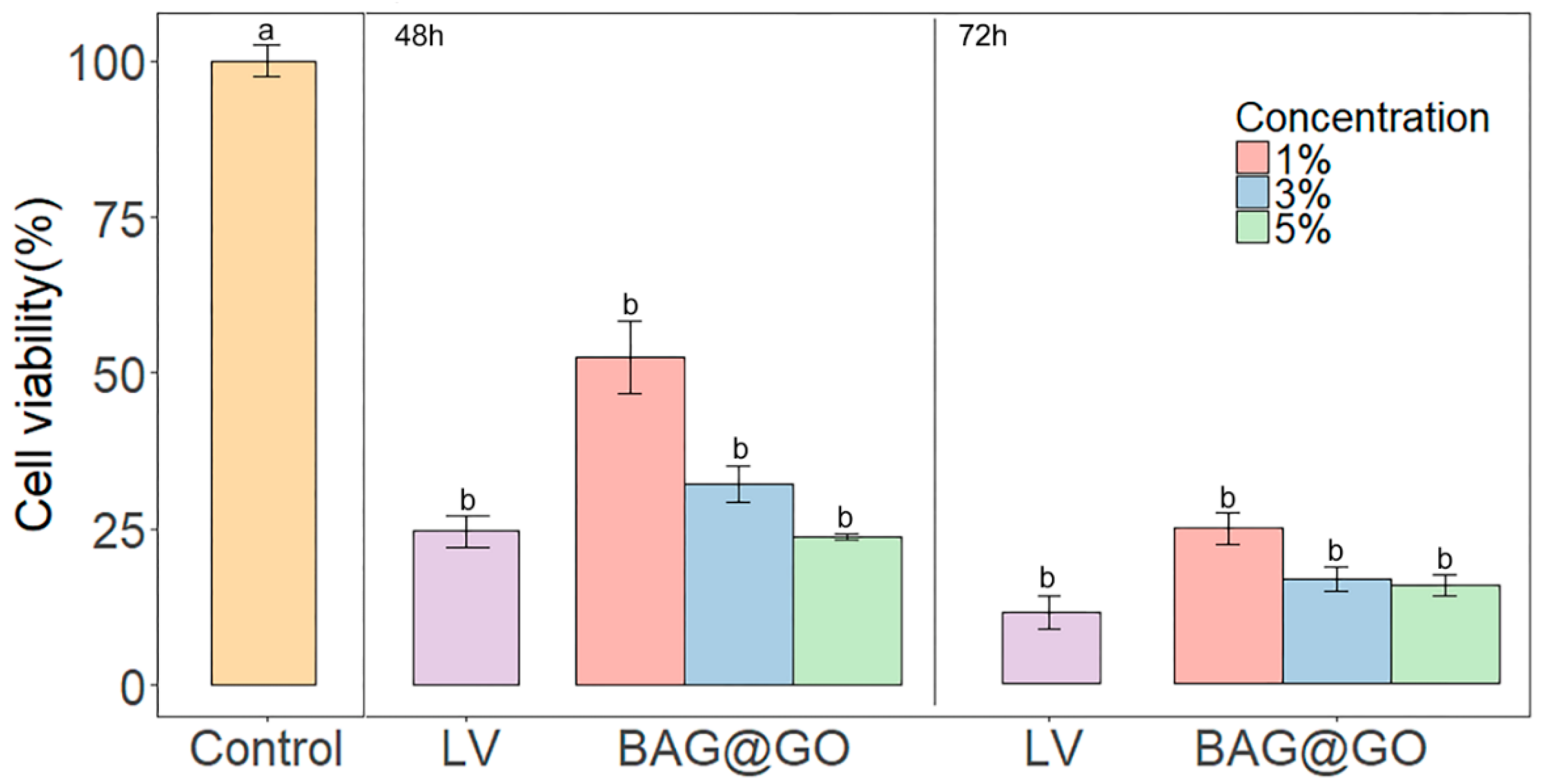
3.3.1. Cell Viability Test
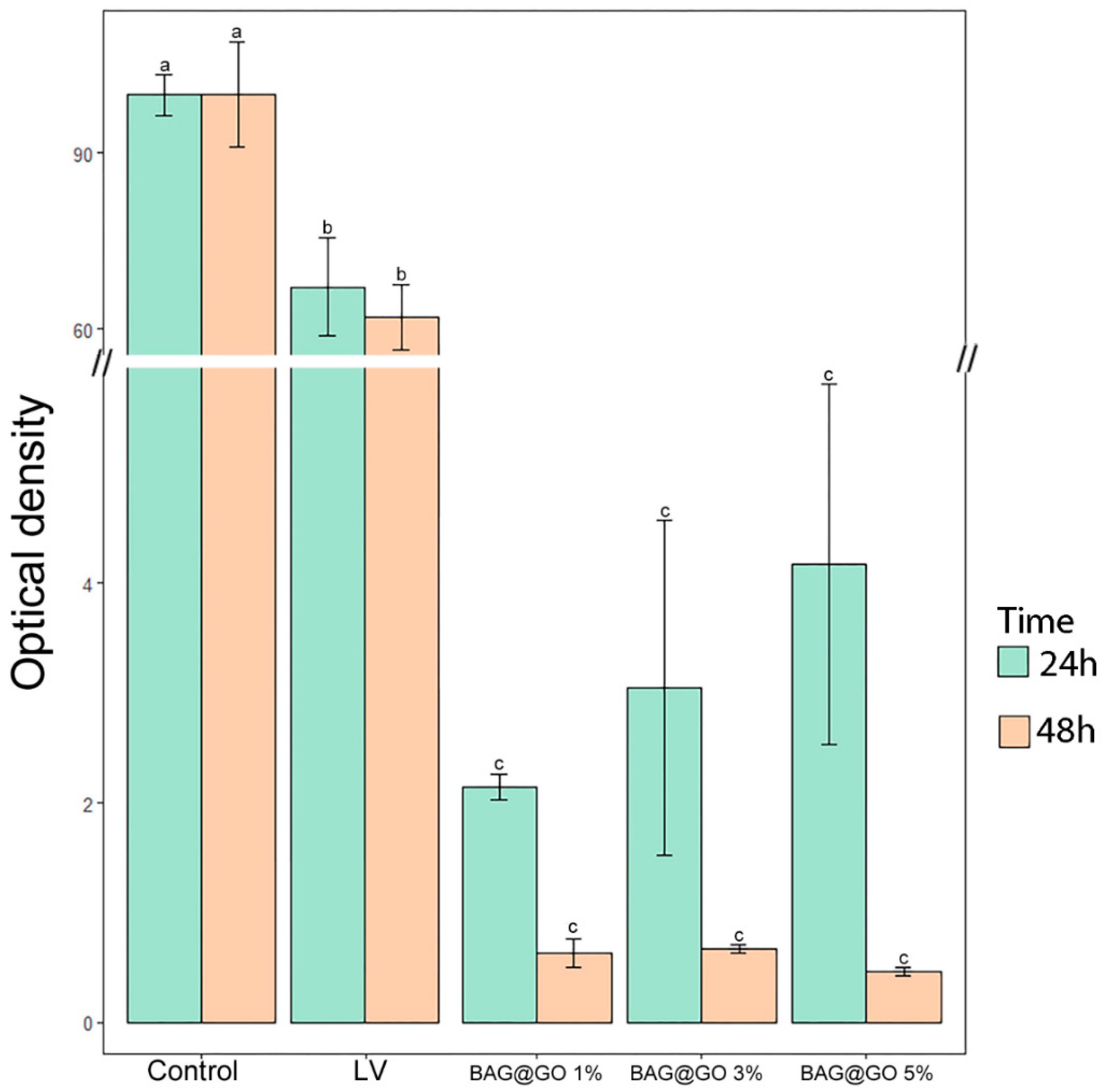
3.3.2. Antibacterial Properties
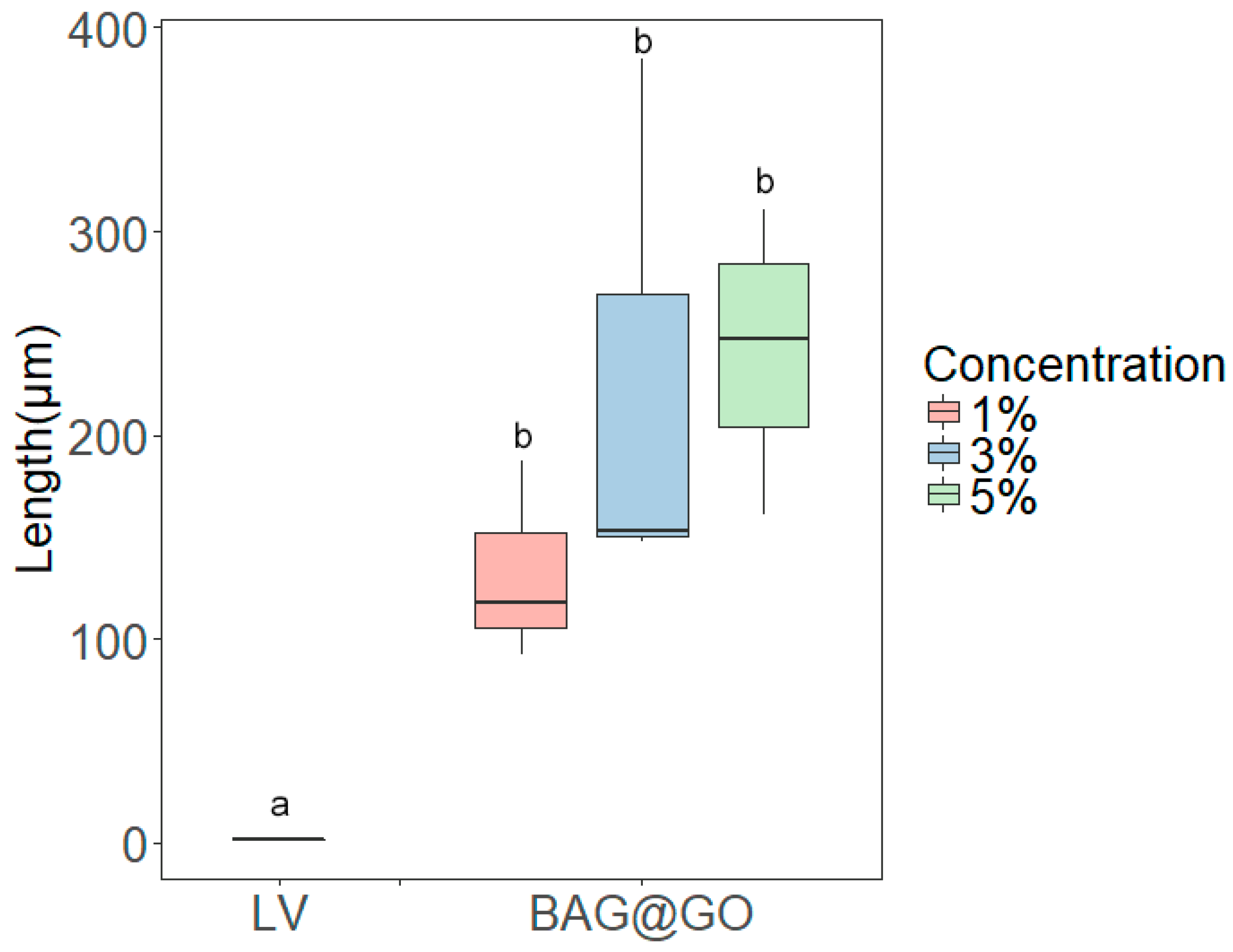
3.4. Anti-Demineralization Test
4. Discussion
5. Conclusions
- Mechanical properties of orthodontic bonding adhesive enriched in graphene oxide and bioactive glass were appropriate for clinical application.
- Biological properties of orthodontic bonding adhesive enriched in graphene oxide and bioactive glass were safe for application to patients.
- More studies, especially in vivo studies, are needed to extensively test orthodontic bonding adhesive containing BAG@GO.
Author Contributions
Funding
Conflicts of Interest
References
- Nyvad, B.; Fejerskov, O.; Baelum, V. Dental Caries: The Disease and its Clinical Management, 3rd ed.; Blackwell Munksgaard: Oxford, UK, 2008. [Google Scholar]
- Ristic, M.; Vlahovic, S.M.; Sasic, M.; Zelic, O. Clinical and microbiological effects of fixed orthodontic appliances on periodontal tissues in adolescents. Orthod. Craniofac. Res. 2007, 10, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Toi, C.S.; Mogodiri, R.; Cleaton-Jones, P.E. Cleaton-Jones Mutans streptococci and lactobacilli on healthy and carious teeth in the same mouth of children with and without dental caries. Microb. Ecol. Health Dis. 2009, 12, 35–41. [Google Scholar]
- Lee, S.M.; Kim, I.R.; Park, B.S.; Lee, D.J.; Ko, C.C.; Son, W.S.; Kim, Y.I. Remineralization Property of an Orthodontic Primer Containing a Bioactive Glass with Silver and Zinc. Materials 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Fernando, D.; Attik, N.; Pradelle-Plasse, N.; Jackson, P.; Grosgogeat, B.; Colon, P. Bioactive glass for dentin remineralization: A. systematic review. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 76, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Chang, J.; Liu, M.; Ning, C. Study on antibacterial effect of 45S5 Bioglass. J. Mater. Sci. Mater. Med. 2009, 20, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Mehrali, M.; Moghaddam, E.; Seyed Shirazi, S.F.; Baradaran, S.; Mehrali, M.; Latibari, S.T.; Metselaar, H.S.; Kadri, N.A.; Zandi, K.; Osman, N.A. Mechanical and in vitro biological performance of graphene nanoplatelets reinforced calcium silicate composite. PLoS ONE 2014, 9, e106802. [Google Scholar] [CrossRef] [PubMed]
- Mohammadrezaei, D.; Golzar, H.; Rezai Rad, M.; Omidi, M.; Rashedi, H.; Yazdian, F.; Khojasteh, A.; Tayebi, L. In vitro effect of graphene structures as an osteoinductive factor in bone tissue engineering: A systematic review. J. Biomed. Mater. Res. A 2018, 106, 2284–2343. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhu, X.; Qi, Z.; Wang, C.; Mao, X.; Zhu, C.; He, Z.; Li, M.; Tang, Z. Killing dental pathogens using antibacterial graphene oxide. ACS Appl. Mater. Interfaces 2015, 7, 5605–5611. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Jo, J.K.; Kim, D.A.; Patel, K.D.; Kim, H.W.; Lee, H.H. Nano-graphene oxide incorporated into PMMA resin to prevent microbial adhesion. Dent. Mater. 2018, 34, e63–e72. [Google Scholar] [CrossRef] [PubMed]
- Lukomska-Szymanska, M.; Konieczka, M.; Zarzycka, B.; Lapinska, B.; Grzegorczyk, J.; Sokolowski, J. Antibacterial Activity of Commercial Dentine Bonding Systems against E. faecalis-Flow Cytometry Study. Materials 2017, 10, 481. [Google Scholar] [CrossRef] [PubMed]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Hegde, H.M.; Bopanna, T.P. Enamel remineralization assessment after treatment with three different remineralizing agents using surface microhardness: An in vitro study. J. Conserv. Dent. 2014, 17, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Stookey, G.K.; Featherstone, J.D.; Rapozo-Hilo, M.; Schemehorn, B.R.; Williams, R.A.; Baker, R.A.; Barker, M.L.; Kaminski, M.A.; McQueen, C.M.; Amburgey, J.S.; et al. The Featherstone laboratory pH cycling model: A prospective, multi-site validation exercise. Am. J. Dent. 2011, 24, 322–328. [Google Scholar] [PubMed]
- Kim, Y.M.; Kim, D.H.; Song, C.W.; Yoon, S.Y.; Kim, S.Y.; Na, H.S.; Chung, J.; Kim, Y.I.; Kwon, Y.H. Antibacterial and remineralization effects of orthodontic bonding agents containing bioactive glass. Korean J. Orthod. 2018, 48, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Gunawidjaja, P.N.; Mathew, R.; Lo, A.Y.; Izquierdo-Barba, I.; Garcia, A.; Arcos, D.; Vallet-Regi, M.; Eden, M. Local structures of mesoporous bioactive glasses and their surface alterations in vitro: Inferences from solid-state nuclear magnetic resonance. Philos. Trans. A Math. Phys. Eng. Sci. 2012, 370, 1376–1399. [Google Scholar] [CrossRef] [PubMed]
- Olteanu, D.; Filip, A.; Socaci, C.; Biris, A.R.; Filip, X.; Coros, M.; Rosu, M.C.; Pogacean, F.; Alb, C.; Baldea, I.; et al. Cytotoxicity assessment of graphene-based nanomaterials on human dental follicle stem cells. Colloids Surf. B Biointerfaces 2015, 136, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, W.; Yue, C.; Zhang, T.; Li, P.; Xing, Z.; Chen, Y. A tough graphene nanosheet/hydroxyapatite composite with improved in vitro biocompatibility. Carbon 2013, 61, 105–115. [Google Scholar] [CrossRef]
- Brown, M.L.; Davis, H.B.; Tufekci, E.; Crowe, J.J.; Covell, D.A.; Mitchell, J.C. Ion release from a novel orthodontic resin bonding agent for the reduction and/or prevention of white spot lesions. An in vitro study. Angle Orthod. 2011, 81, 1014–1020. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Qi, X.; Boey, F.; Zhang, H. Graphene-based composites. Chem. Soc. Rev. 2012, 41, 666–686. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hu, M.; Zeng, T.H.; Wu, R.; Jiang, R.; Wei, J.; Wang, L.; Kong, J.; Chen, Y. Lateral dimension-dependent antibacterial activity of graphene oxide sheets. Langmuir 2012, 28, 12364–12372. [Google Scholar] [CrossRef] [PubMed]
- Kohda, N.; Iijima, M.; Kawaguchi, K.; Toshima, H.; Muguruma, T.; Endo, K.; Mizoguchi, I. Inhibition of enamel demineralization and bond-strength properties of bioactive glass containing 4-META/MMA-TBB-based resin adhesive. Eur. J. Oral Sci. 2015, 123, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Kvetková, L.; Duszová, A.; Hvizdoš, P.; Dusza, J.; Kun, P.; Balázsi, C. Fracture toughness and toughening mechanisms in graphene platelet reinforced Si3N4 composites. Scr. Mater. 2012, 66, 793–796. [Google Scholar] [CrossRef]
- Kun, P.; Tapasztó, O.; Wéber, F.; Balázsi, C. Determination of structural and mechanical properties of multilayer graphene added silicon nitride-based composites. Ceram. Int. 2012, 38, 211–216. [Google Scholar] [CrossRef]

| Sample | LV | BAG@GO | Significant | ||
|---|---|---|---|---|---|
| 1% | 3% | 5% | |||
| Mean (SD) | 3.8 (0.4) | 4.0 (0.0) | 3.8 (0.4) | 3.4 (0.5) | Not significant |
| Median, Q1–Q3 | 4, 4–4 | 4, 4–4 | 4, 4–4 | 3, 3–4 | |
| Min.–max. | 3–4 | 4–4 | 3–4 | 3–4 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-M.; Yoo, K.-H.; Yoon, S.-Y.; Kim, I.-R.; Park, B.-S.; Son, W.-S.; Ko, C.-C.; Son, S.-A.; Kim, Y.-I. Enamel Anti-Demineralization Effect of Orthodontic Adhesive Containing Bioactive Glass and Graphene Oxide: An In-Vitro Study. Materials 2018, 11, 1728. https://doi.org/10.3390/ma11091728
Lee S-M, Yoo K-H, Yoon S-Y, Kim I-R, Park B-S, Son W-S, Ko C-C, Son S-A, Kim Y-I. Enamel Anti-Demineralization Effect of Orthodontic Adhesive Containing Bioactive Glass and Graphene Oxide: An In-Vitro Study. Materials. 2018; 11(9):1728. https://doi.org/10.3390/ma11091728
Chicago/Turabian StyleLee, Seung-Min, Kyung-Hyeon Yoo, Seog-Young Yoon, In-Ryoung Kim, Bong-Soo Park, Woo-Sung Son, Ching-Chang Ko, Sung-Ae Son, and Yong-Il Kim. 2018. "Enamel Anti-Demineralization Effect of Orthodontic Adhesive Containing Bioactive Glass and Graphene Oxide: An In-Vitro Study" Materials 11, no. 9: 1728. https://doi.org/10.3390/ma11091728
APA StyleLee, S.-M., Yoo, K.-H., Yoon, S.-Y., Kim, I.-R., Park, B.-S., Son, W.-S., Ko, C.-C., Son, S.-A., & Kim, Y.-I. (2018). Enamel Anti-Demineralization Effect of Orthodontic Adhesive Containing Bioactive Glass and Graphene Oxide: An In-Vitro Study. Materials, 11(9), 1728. https://doi.org/10.3390/ma11091728





