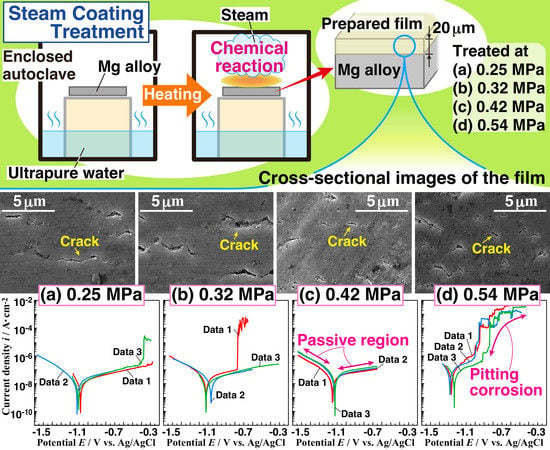
Effect of Vapor Pressure During the Steam Coating Treatment on Structure and Corrosion Resistance of the Mg(OH)2/Mg-Al LDH Composite Film Formed on Mg Alloy AZ61
Abstract
1. Introduction
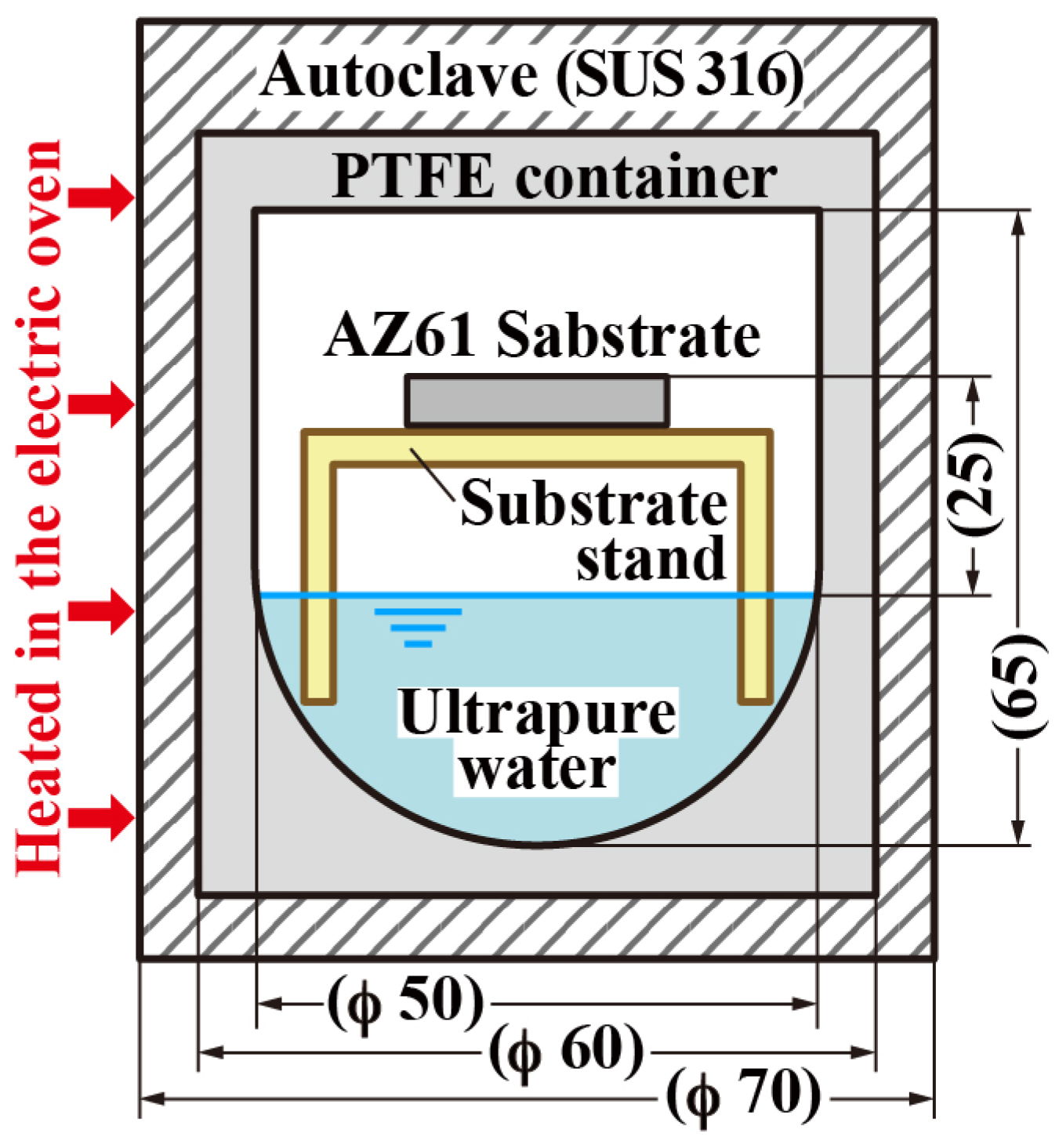
2. Methods and Materials
3. Results and Discussions
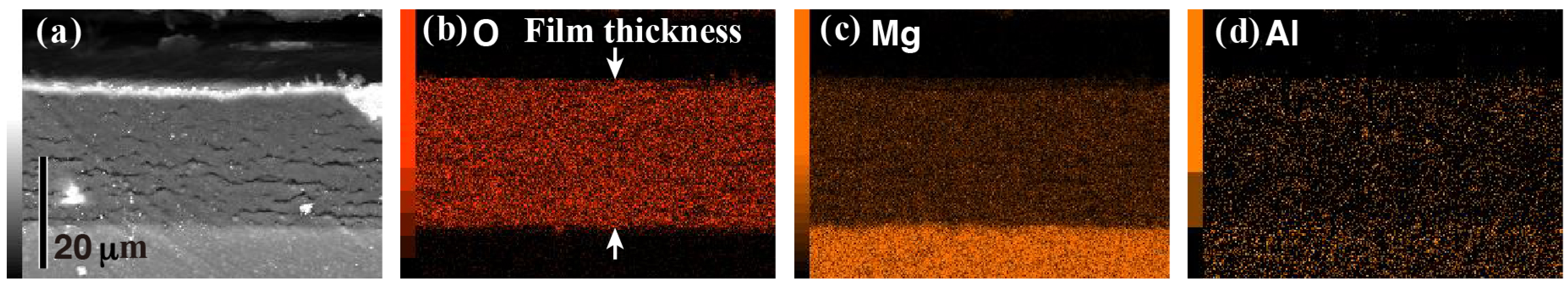
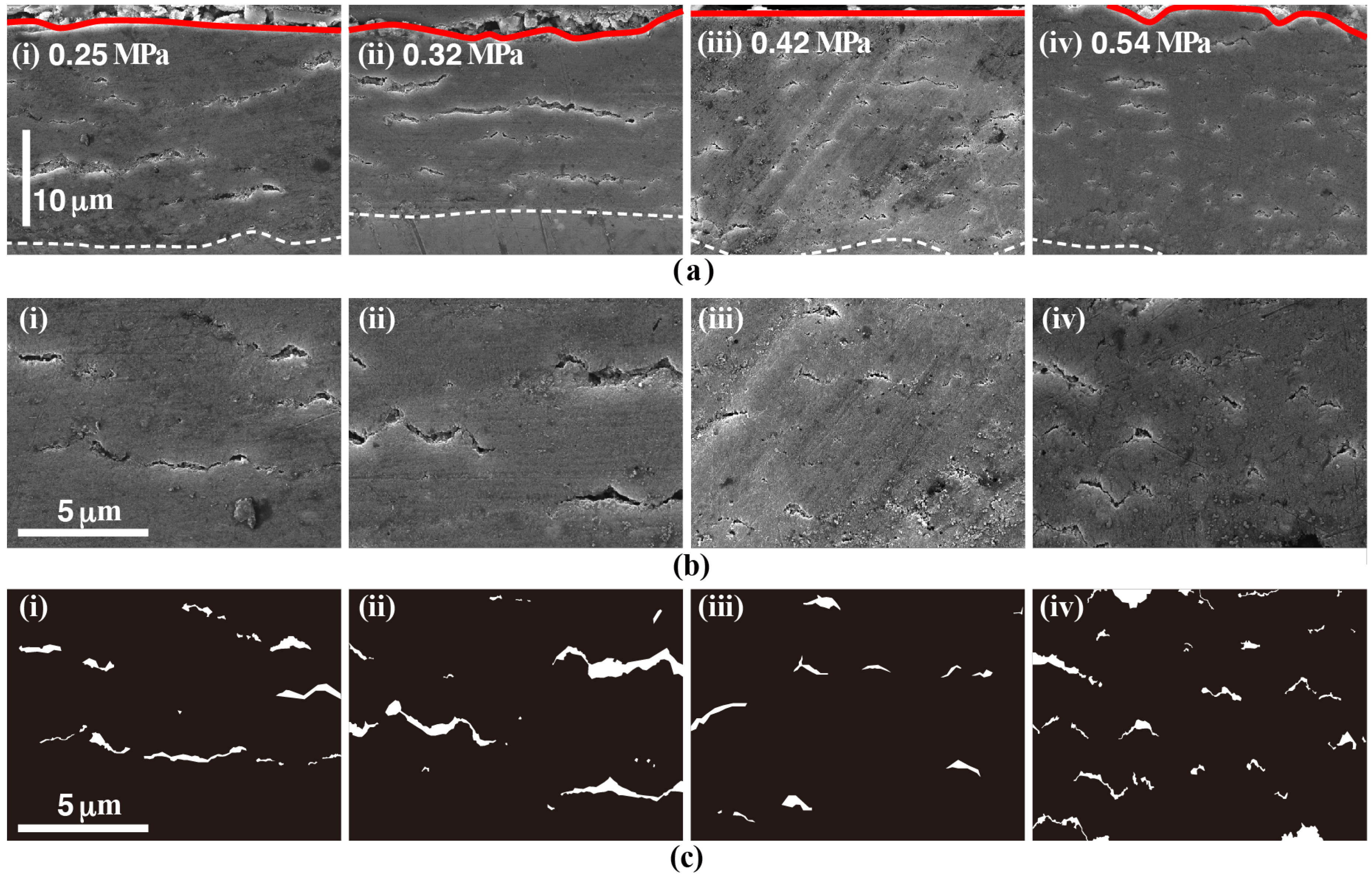
3.1. Morphologies of the Anticorrosive Films
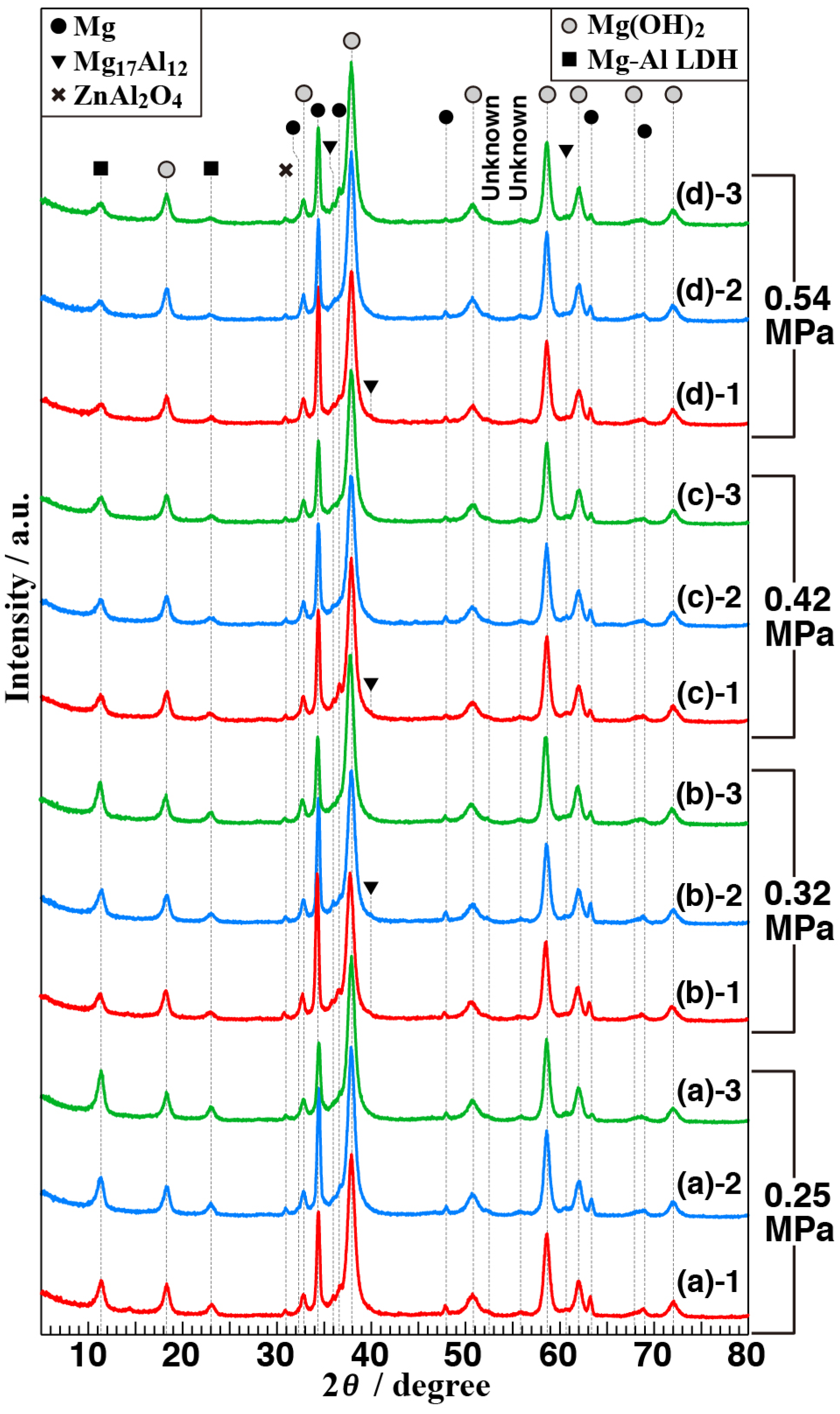
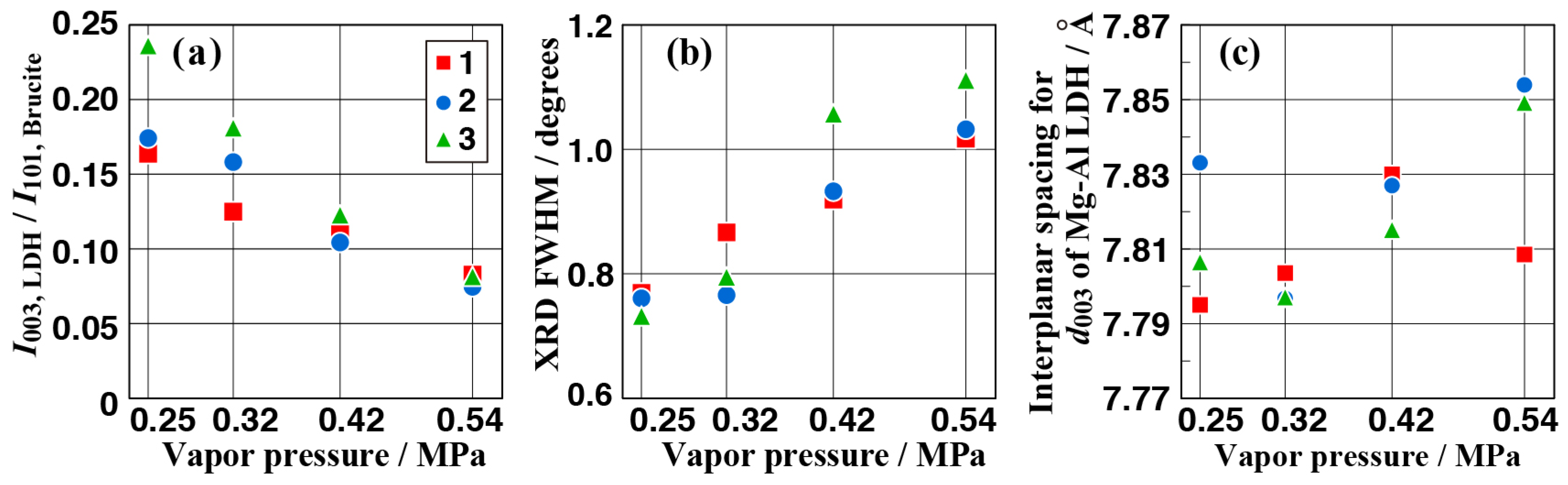
3.2. Crystal Phases of the Anticorrosive Film
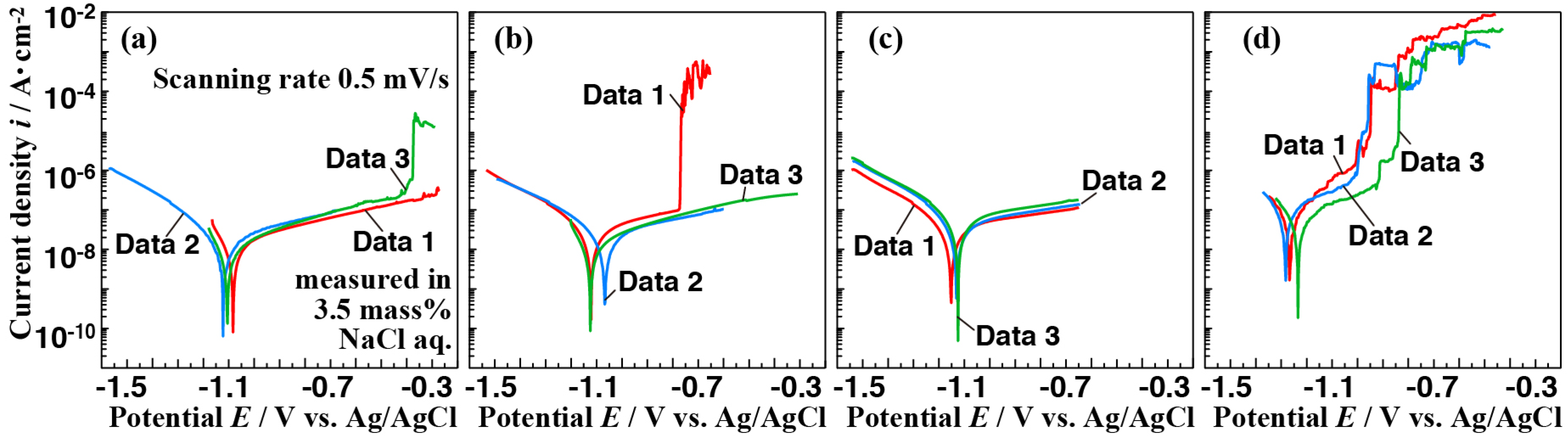
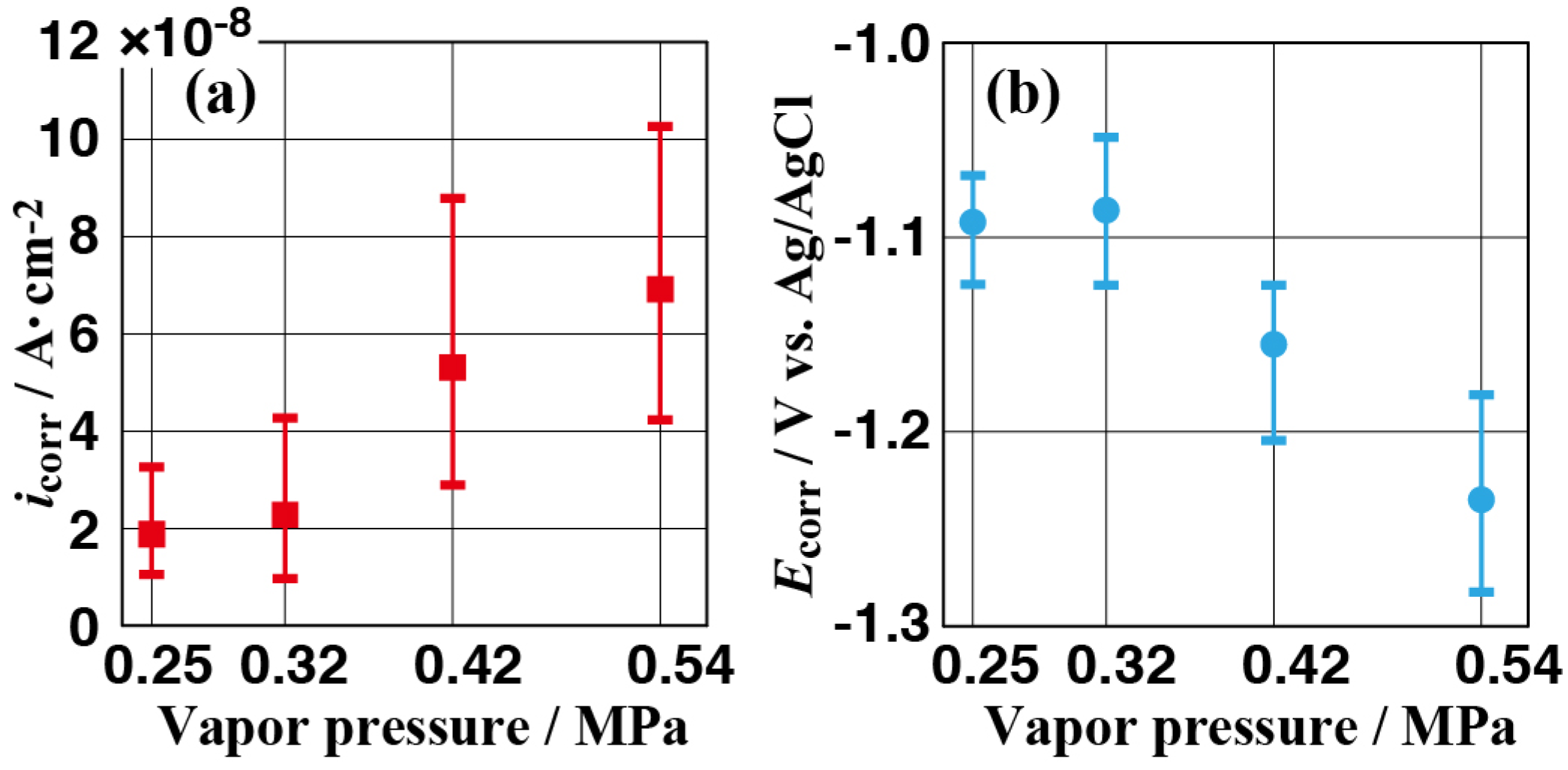
3.3. Corrosion Resistance of the Films
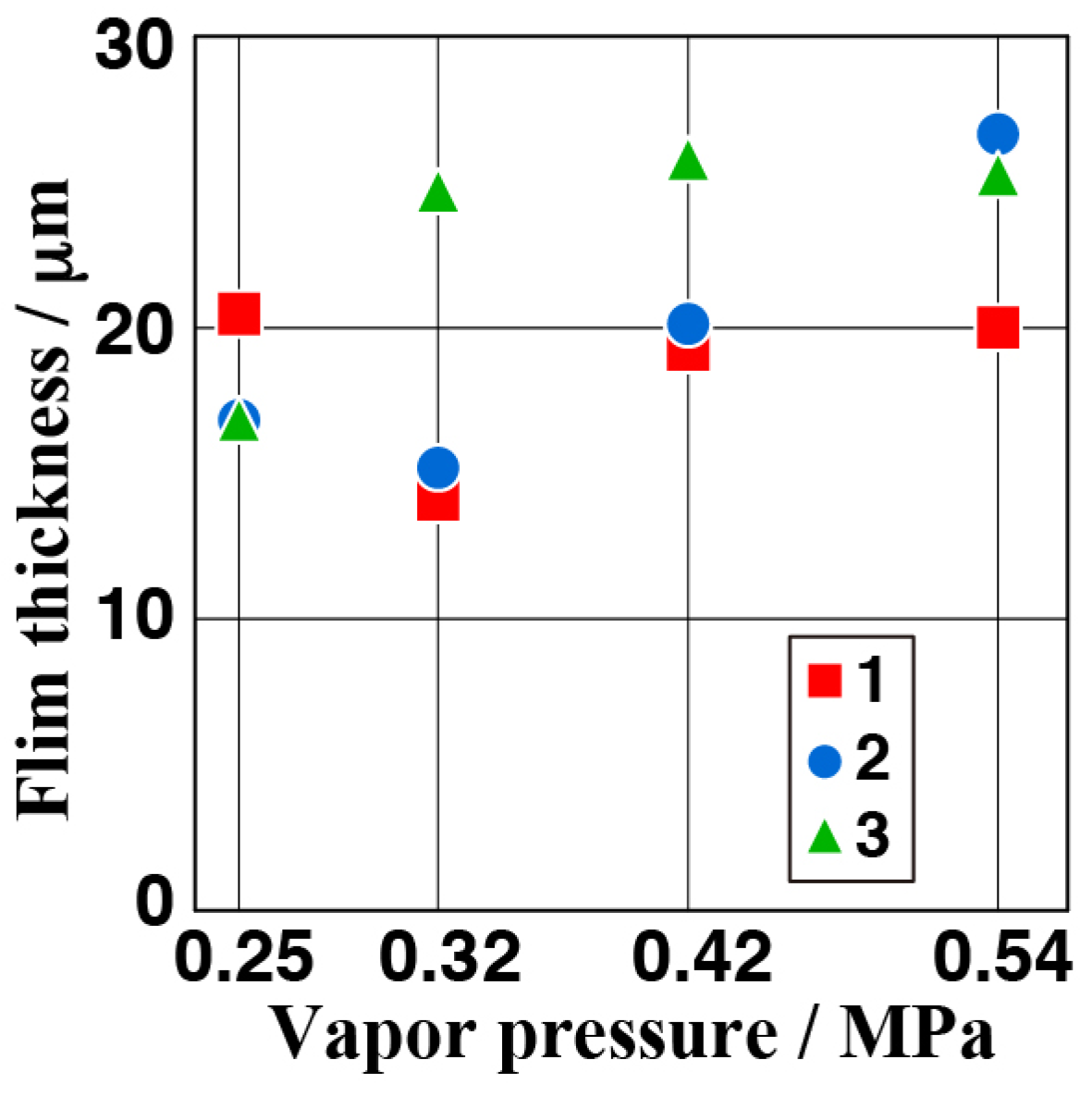
3.4. Effect of Vapor Pressure on the Structure and the Corrosion Resistance of the Film
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- United States Geological Survey, Bureau of Mines Minerals Yearbook. 2018. Available online: http://minerals.usgs.gov/minerals/pubs/ usbmmyb.html (accessed on 26 June 2018).
- Nakatsugawa, I.; Umehara, H.; Akimoto, M.; Shimamura, K.; Ono, S.; Takaya, M.; Tsubota, T. Corrosion and surface treatment of magnesium alloy. In Handbook of Advanced Magnesium Technology (in Japanese), 2nd ed.; Editorial Committee of Handbook of Advanced Magnesium Technology, Ed.; Kallos Publishing Co., Ltd.: Tokyo, Japan, 2000; pp. 311–352. ISBN 4-87432-014-7. [Google Scholar]
- Mordike, B.L.; Ebert, T. Magnesium properties—Applications—Potential. Mater. Sci. Eng. A 2001, 302, 37–45. [Google Scholar] [CrossRef]
- Wang, X.J.; Xu, D.K.; Wu, R.Z.; Chen, X.B.; Peng, Q.M.; Jin, L.; Xin, Y.C.; Zhang, Z.Q.; Liu, Y.; Chen, X.H.; et al. What is going on in magnesium alloys? J. Mater. Sci. Technol. 2018, 34, 245–247. [Google Scholar] [CrossRef]
- Kumar, D.S.; Sasanka, C.T.; Ravindra, K.; Suman, K.N.S. Magnesium and its alloys in automotive applications—A review. Am. J. Mater. Sci. Technol. 2015, 4, 12–30. [Google Scholar] [CrossRef]
- Pollock, T.M. Weight loss with magnesium alloys. Science 2010, 328, 986–987. [Google Scholar] [CrossRef] [PubMed]
- Aghion, E.; Bronfin, B.; Eliezer, D. The role of the magnesium industry in protecting the environment. J. Mater. Process. Technol. 2001, 117, 381–385. [Google Scholar] [CrossRef]
- Metroke, T.L.; Kachurina, O.; Knobbe, E.T. Spectroscopic and corrosion resistance characterization of GLYMO–TEOS Ormosil coatings for aluminum Aaloy corrosion inhibition. Prog. Org. Coat. 2002, 44, 295–305. [Google Scholar] [CrossRef]
- Takai, M.; Takaya, M. Influence of conversion coating on magnesium and aluminum alloys by adhesion method. Mater. Trans. 2008, 49, 1065–1070. [Google Scholar] [CrossRef]
- Sakuragi, N.; Yamamoto, S.; Koide, Y. A self-assembled monolayers assisted solid-state conversion of boehmite particles to aluminum oxide film. J. Am. Chem. Soc. 2007, 129, 10048–10049. [Google Scholar] [CrossRef] [PubMed]
- Kulinich, S.A.; Farzaneh, M.; Du, X.W. Growth of corrosion-resistant manganese oxide coatings on an aluminum alloy. Inorg. Mater. 2007, 43, 956–963. [Google Scholar] [CrossRef]
- He, T.; Wang, Y.C.; Zhang, Y.J.; Lv, Q.; Xu, T.; Liu, T. Super-hydrophobic surface treatment as corrosion protection for aluminum in seawater. Corros. Sci. 2009, 51, 1757–1761. [Google Scholar] [CrossRef]
- Konieczny, J.; Dobrzański, L.A.; Labisz, K.; Duszczyk, J. The influence of cast method and anodizing parameters on structure and layer thickness of aluminium alloys. J. Mater. Process. Technol. 2004, 157–158, 718–723. [Google Scholar] [CrossRef]
- Saenz de Miera, M.; Curioni, M.; Skeldon, P.; Thompson, G.E. Modelling the anodizing behavior of aluminium alloys in sulphuric acid through alloy analogues. Corros. Sci. 2008, 50, 3410–3415. [Google Scholar] [CrossRef]
- Twite, R.L.; Bierwagen, G.P. Review of alternatives to chromate for corrosion protection of aluminum aerospace alloys. Prog. Org. Coat. 1998, 33, 91–100. [Google Scholar] [CrossRef]
- Tallman, D.E.; Spinks, G.; Dominis, A.; Wallace, G.G. Electroactive conducting polymers for corrosion control. J. Solid State Electr. 2002, 6, 73–84. [Google Scholar] [CrossRef]
- Kure, K.; Konno, Y.; Tsuji, E.; Skeldon, P.; Thompson, G.E.; Habazaki, H. Formation of self-organized nanoporous anodic films on Type 304 stainless steel. Electrochem. Commun. 2012, 21, 1–4. [Google Scholar] [CrossRef]
- Song, Y.K.; Mansfeld, F. Development of a molybdate–phosphate–silane–silicate (MPSS) coating process for electrogalvanized steel. Corros. Sci. 2006, 48, 154–164. [Google Scholar] [CrossRef]
- Hara, M.; Ichino, R.; Okido, M.; Wada, N. Corrosion protection property of colloidal silicate film on galvanized steel. Surf. Coat. Technol. 2003, 169–170, 679–681. [Google Scholar] [CrossRef]
- Li, O.L.; Tsunakawa, M.; Shimada, Y.; Nakamura, K.; Nishinaka, K.; Ishizaki, T. Corrosion resistance of composite oxide film prepared on Ca-added flame-resistant magnesium alloy AZCa612 by micro-arc oxidation. Corros. Sci. 2017, 125, 99–105. [Google Scholar] [CrossRef]
- Chong, K.Z.; Shih, T.S. Conversion-coating treatment for magnesium alloys by a permanganate-phosphate solution. Mater. Chem. Phys. 2003, 80, 191–200. [Google Scholar] [CrossRef]
- Sharma, A.K.; Rani, R.U.; Mayanna, S.M. Thermal studies on electrodeposited black oxide coating on magnesium alloys. Thermochim. Acta 2001, 376, 67–75. [Google Scholar] [CrossRef]
- Montemora, M.F.; Ferreira, M.G.S. Electrochemical study of modified bis-[triethoxysilylpropyl] tetrasulfide silane films applied on the AZ31 Mg alloy. Electrochim. Acta 2007, 52, 7486–7495. [Google Scholar] [CrossRef]
- Blawert, C.; Dietzel, W.; Ghali, E.; Song, G. Anodizing treatments for magnesium alloys and their effect on corrosion resistance in various environments. Adv. Eng. Mater. 2006, 8, 511–533. [Google Scholar] [CrossRef]
- Zeng, R.C.; Cui, L.Y.; Jiang, K.; Liu, R.; Zhao, B.D.; Zheng, Y.F. In vitro corrosion and cytocompatibility of a microarc oxidation coating and poly(L-lactic acid) composite coating on Mg−1Li−1Ca alloy for orthopedic implants. Appl. Mater. Interfaces 2016, 8, 10014–10028. [Google Scholar] [CrossRef] [PubMed]
- Duan, G.Q.; Yang, L.X.; Liao, S.G.; Zhang, C.Y.; Lu, X.P.; Yang, Y.; Zhang, B.; Wei, Y.; Zhang, T.; Yu, B.X.; et al. Designing for the chemical conversion coating with high corrosion resistance and low electrical contact resistance on AZ91D magnesium alloy. Corros. Sci. 2018, 135, 197–206. [Google Scholar] [CrossRef]
- Kendig, M.; Jeanjaquet, S.; Addison, R.; Waldrop, J. Role of hexavalent chromium in the inhibition of corrosion of aluminum alloys. Surf. Coat. Technol. 2001, 140, 58–66. [Google Scholar] [CrossRef]
- Meng, Q.; Frankel, G.S. Effect of copper content on chromate conversion coating protection of 7xxx-T6 alminum alloys. Corrosion 2004, 60, 897–905. [Google Scholar] [CrossRef]
- Rocco, A.M.; Nogueira, T.M.C.; Simao, R.A.; Lima, W.C. Evaluation of chromate passivation and chromate conversion coating on 55% Al–Zn coated steel. Surf. Coat. Technol. 2004, 179, 135–144. [Google Scholar] [CrossRef]
- Ishizaki, T.; Chiba, S.; Watanabe, K.; Suzuki, H. Corrosion resistance of Mg-Al layered double hydroxide container-containing magnesium hydroxide films formed directly on magnesium alloy by chemical-free steam coating. J. Mater. Chem. A 2013, 1, 8968–8977. [Google Scholar] [CrossRef]
- Kamiyama, N.; Panomsuwan, G.; Yamamoto, E.; Sudare, T.; Saito, N.; Ishizaki, T. Effect of treatment time in the Mg(OH)2/Mg-Al LDH composite film formed on Mg alloy AZ31 by steam coating on the corrosion resistance. Surf. Coat. Technol. 2016, 286, 172–177. [Google Scholar] [CrossRef]
- Ishizaki, T.; Kamiyama, N.; Watanabe, K.; Serizawa, A. Corrosion resistance of Mg(OH)2/Mg–Al layered double hydroxide composite film formed directly on combustion-resistant magnesium alloy AMCa602 by steam coating. Corros. Sci. 2015, 92, 76–84. [Google Scholar] [CrossRef]
- Nakamura, K.; Tsunakawa, M.; Shimada, Y.; Serizawa, A.; Ishizaki, T. Formation mechanism of Mg-Al layered double hydroxide-containing magnesium hydroxide films prepared on Ca-added flame-resistant magnesium ally by steam coating. Surf. Coat. Technol. 2016, 328, 436–443. [Google Scholar] [CrossRef]
- Ke, C.; Wu, Y.; Qiu, Y.; Duan, J.H.; Birbilis, N.; Chen, X.B. Influence of surface chemistry on the formation of crystalline hydroxide coatings on Mg alloys in liquid water and steam systems. Corros. Sci. 2016, 113, 145–159. [Google Scholar] [CrossRef]
- Kozawa, T.; Yanagisawa, K.; Yoshida, A.; Onda, A.; Suzuki, Y. Preparation of β-CaSiO3 powder by water vapor-assisted solid-state reaction. J. Ceram. Soc. Jpn. 2013, 121, 103–105. [Google Scholar] [CrossRef]
- Kozawa, T.; Yanagisawa, K.; Yoshida, A.; Suzuki, Y. Water vapor-assisted solid-state reaction for the synthesis of nanocrystalline BaZrO3 powder. J. Ceram. Soc. Jpn. 2013, 121, 308–312. [Google Scholar] [CrossRef]
- Din, R.U.; Jellesen, M.S.; Ambat, R. Performance comparison of steam-based and chromate conversion coatings on aluminum alloy 6060. Corrosion 2015, 71, 839–853. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, L.; Tang, A.; Chen, X.B.; Ma, Y.L.; Long, Y.; Peng, P.; Ding, X.X.; Pan, H.L.; Pan, F.S. Growth behavior of MgAl-layered double hydroxide films by conversion of anodic films on magnesium alloy AZ31 and their corrosion protection. Appl. Surf. Sci. 2018, 456, 419–429. [Google Scholar] [CrossRef]
- Hort, N.; Huang, Y.; Kainer, K.U. Intermetallics in magnesium alloys. Adv. Eng. Mater. 2006, 8, 235–240. [Google Scholar] [CrossRef]
- Feliu Jr., S.; Maffiotte, C.; Samaniego, A.; Galván, J.C.; Barranco, V. Effect of the chemistry and structure of the native oxide surface film on the corrosion properties of commercial AZ31 and AZ61 alloys. Appl. Surf. Sci. 2011, 257, 8558–8568. [Google Scholar] [CrossRef]
- El-Morsy, A.; Ismail, A.; Waly, M. Microstructural and mechanical properties evolution of magnesium AZ61 alloy processed through a combination of extrusion and thermomechanical processes. Mater. Sci. Eng. A 2008, 486, 528–533. [Google Scholar] [CrossRef]
- Czerwinski, F. The oxidation behaviour of an AZ91D magnesium alloy at high temperatures. Acta Mater. 2002, 50, 2639–2654. [Google Scholar] [CrossRef]
- Kannan, M.B.; Raja, V.S. Influence of Heat treatment and scandium addition on the electrochemical polarization behavior of Al-Zn-Mg-Cu-Zr alloy. Metall. Mater. Trans. A 2007, 38, 2843–2852. [Google Scholar] [CrossRef]
- Millange, F.; Walton, R.I.; O’Hare, D. Time-resolved in situ X-ray diffraction study of the liquid-phase reconstruction of Mg-Al-carbonate hydrotalcite-like compounds. J. Mater. Chem. 2000, 10, 1713–1720. [Google Scholar] [CrossRef]


| Sample | Vapor Pressure (MPa) | Temperature (°C) | Treatment Time (h:min) | Target Value of Film Thickness (m) |
|---|---|---|---|---|
| (A) (B) (C) (D) | 0.25, (0.27)* 0.32, (0.36)* 0.42, (0.48)* 0.54, (0.62)* | 130 140 150 160 | 9:30 5:30 4:15 3:20 | 20.0 |
| Sample | at. % | ||||
|---|---|---|---|---|---|
| C | O | Mg | Al | Zn | |
| (A) (B) (C) (D) | 4.22 6.60 4.80 5.26 | 65.55 63.19 64.75 64.47 | 28.50 28.29 27.75 27.97 | 1.68 1.82 2.68 2.25 | 0.05 0.10 0.02 0.04 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakamura, K.; Shimada, Y.; Miyashita, T.; Serizawa, A.; Ishizaki, T. Effect of Vapor Pressure During the Steam Coating Treatment on Structure and Corrosion Resistance of the Mg(OH)2/Mg-Al LDH Composite Film Formed on Mg Alloy AZ61. Materials 2018, 11, 1659. https://doi.org/10.3390/ma11091659
Nakamura K, Shimada Y, Miyashita T, Serizawa A, Ishizaki T. Effect of Vapor Pressure During the Steam Coating Treatment on Structure and Corrosion Resistance of the Mg(OH)2/Mg-Al LDH Composite Film Formed on Mg Alloy AZ61. Materials. 2018; 11(9):1659. https://doi.org/10.3390/ma11091659
Chicago/Turabian StyleNakamura, Kae, Yuta Shimada, Tomohiro Miyashita, Ai Serizawa, and Takahiro Ishizaki. 2018. "Effect of Vapor Pressure During the Steam Coating Treatment on Structure and Corrosion Resistance of the Mg(OH)2/Mg-Al LDH Composite Film Formed on Mg Alloy AZ61" Materials 11, no. 9: 1659. https://doi.org/10.3390/ma11091659
APA StyleNakamura, K., Shimada, Y., Miyashita, T., Serizawa, A., & Ishizaki, T. (2018). Effect of Vapor Pressure During the Steam Coating Treatment on Structure and Corrosion Resistance of the Mg(OH)2/Mg-Al LDH Composite Film Formed on Mg Alloy AZ61. Materials, 11(9), 1659. https://doi.org/10.3390/ma11091659