Hydrothermal Synthesis of Co-Doped NiSe2 Nanowire for High-Performance Asymmetric Supercapacitors
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of Co@NiSe2
2.3. Assembly of Asymmetrical Supercapacitors
2.4. Material Characterization
2.5. Electrochemical Measurements
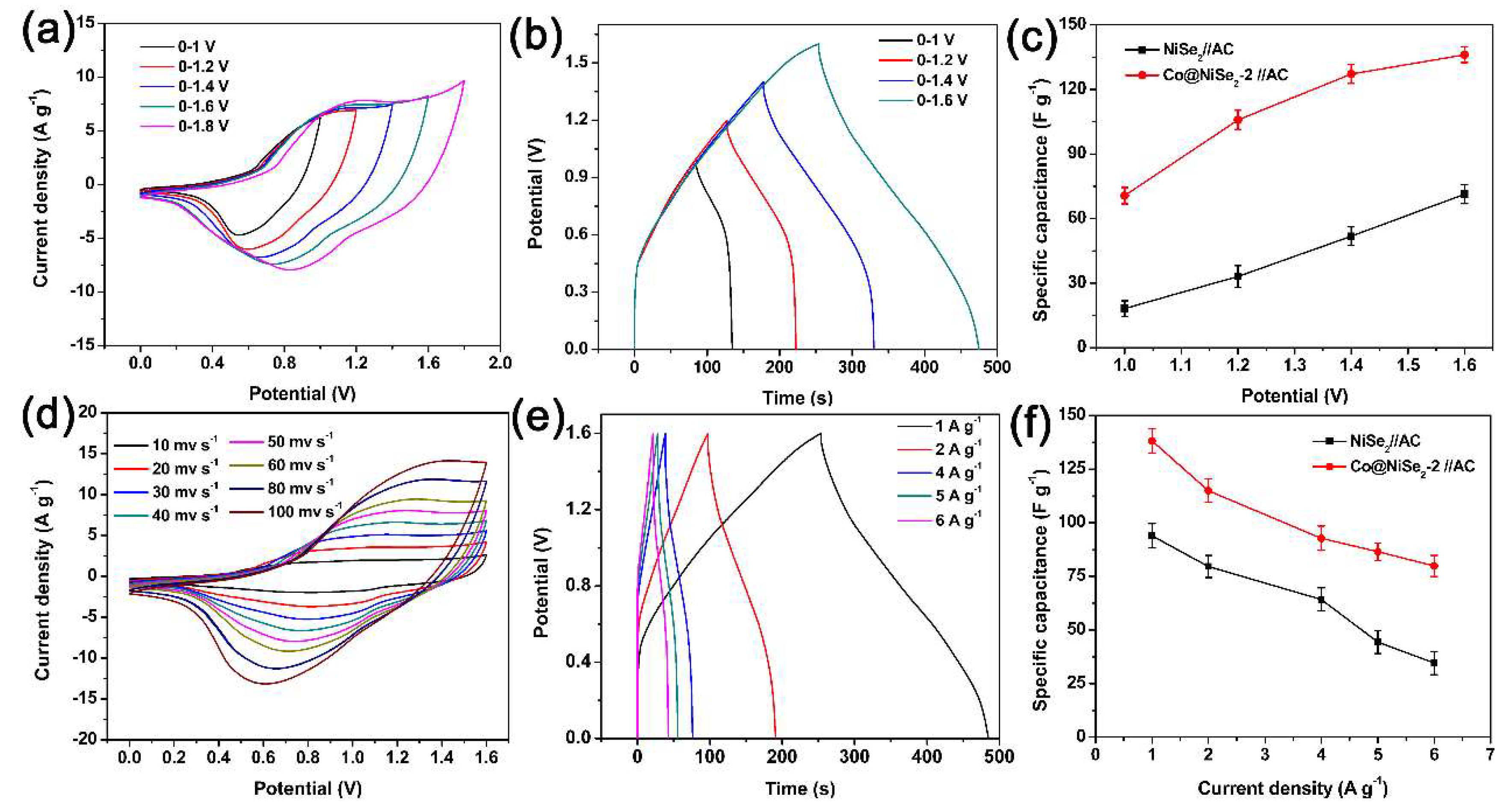
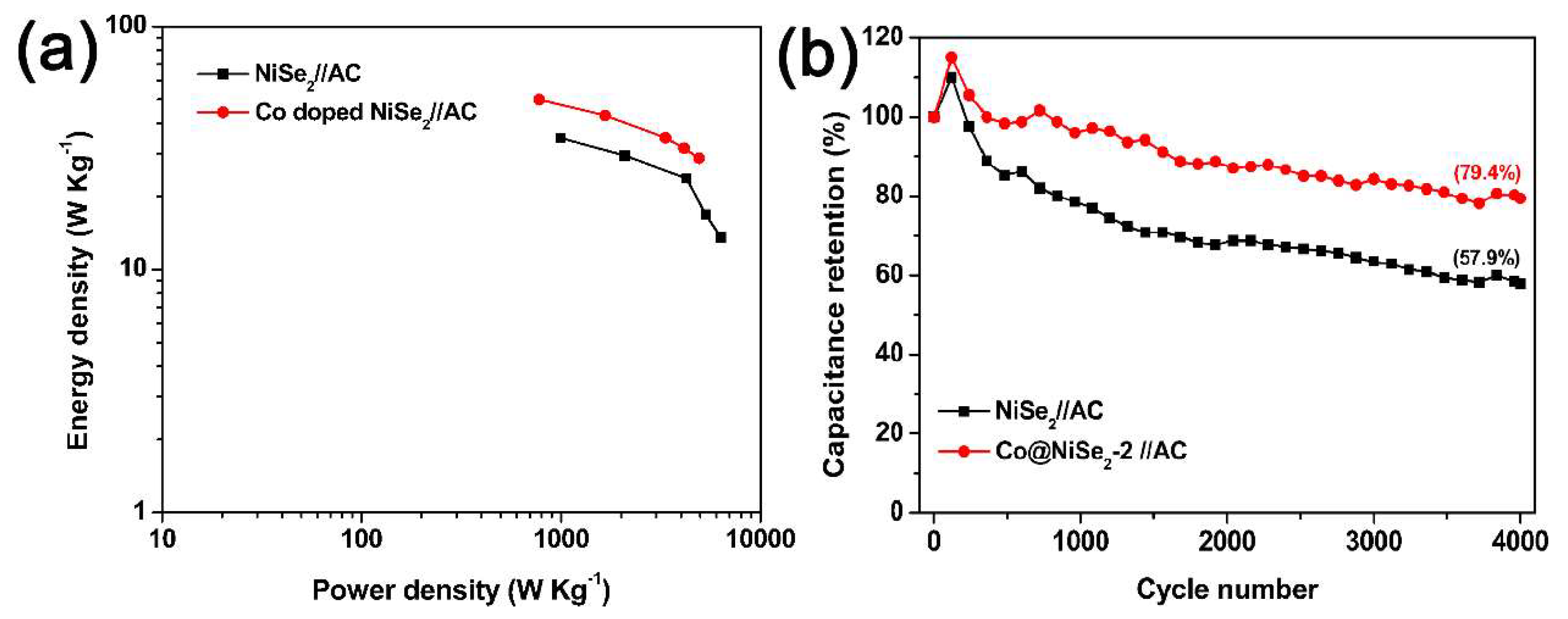
3. Results and Discussion
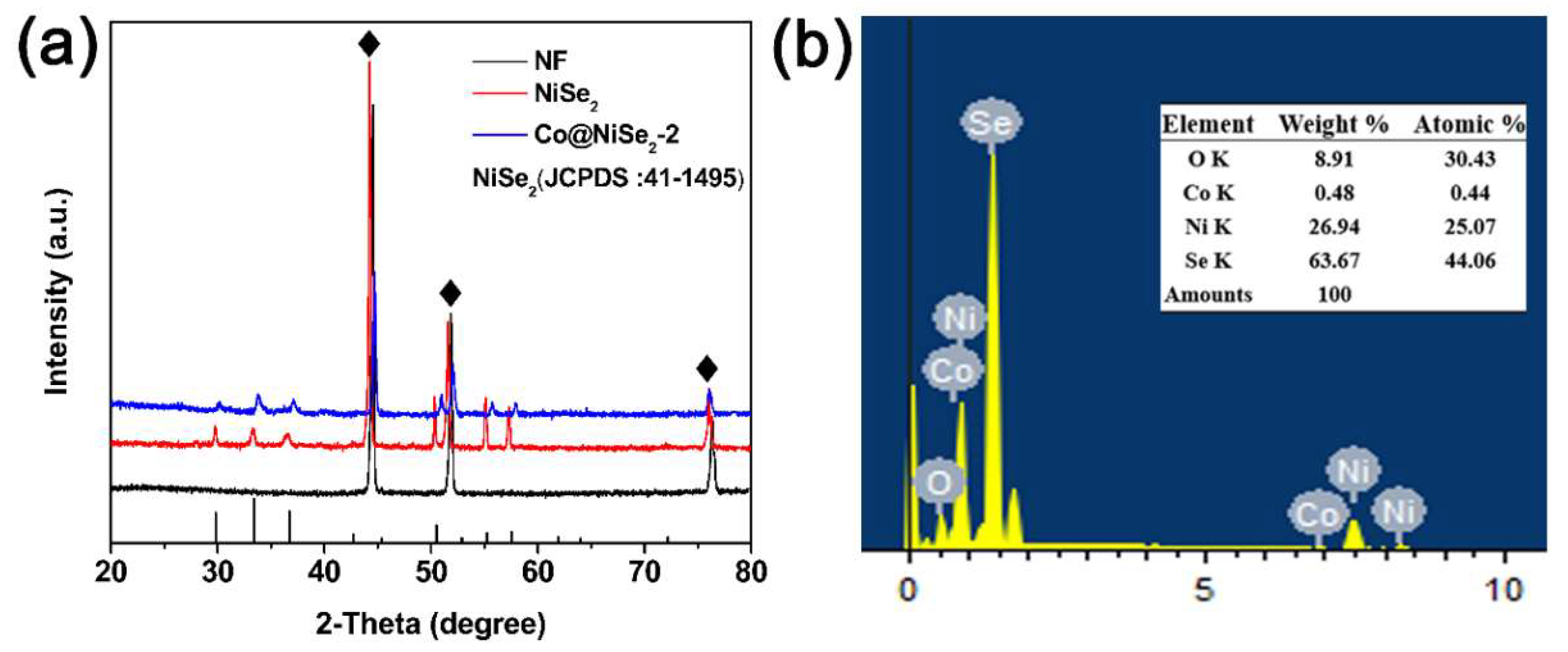
3.1. Characterization of Electrode Materials
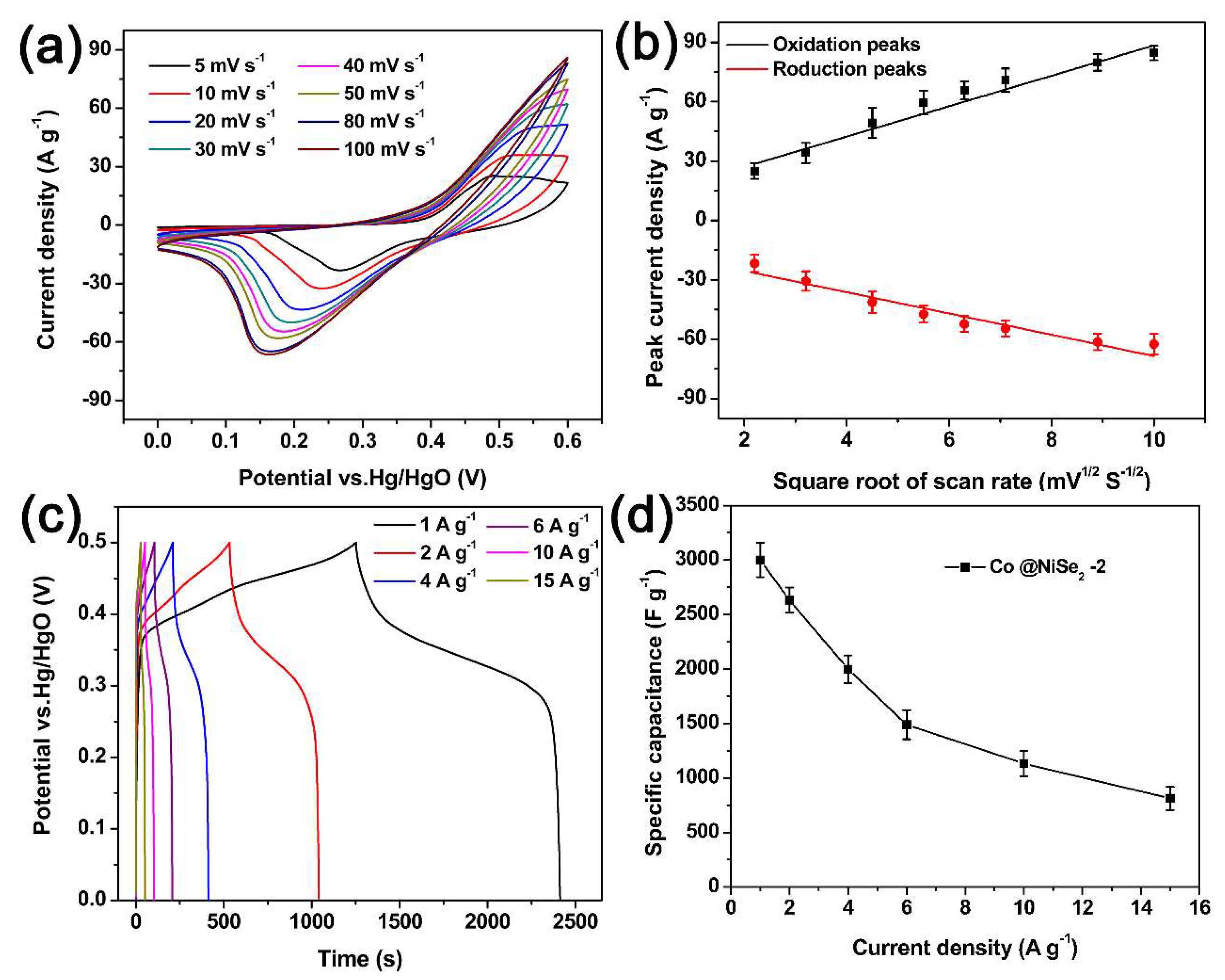
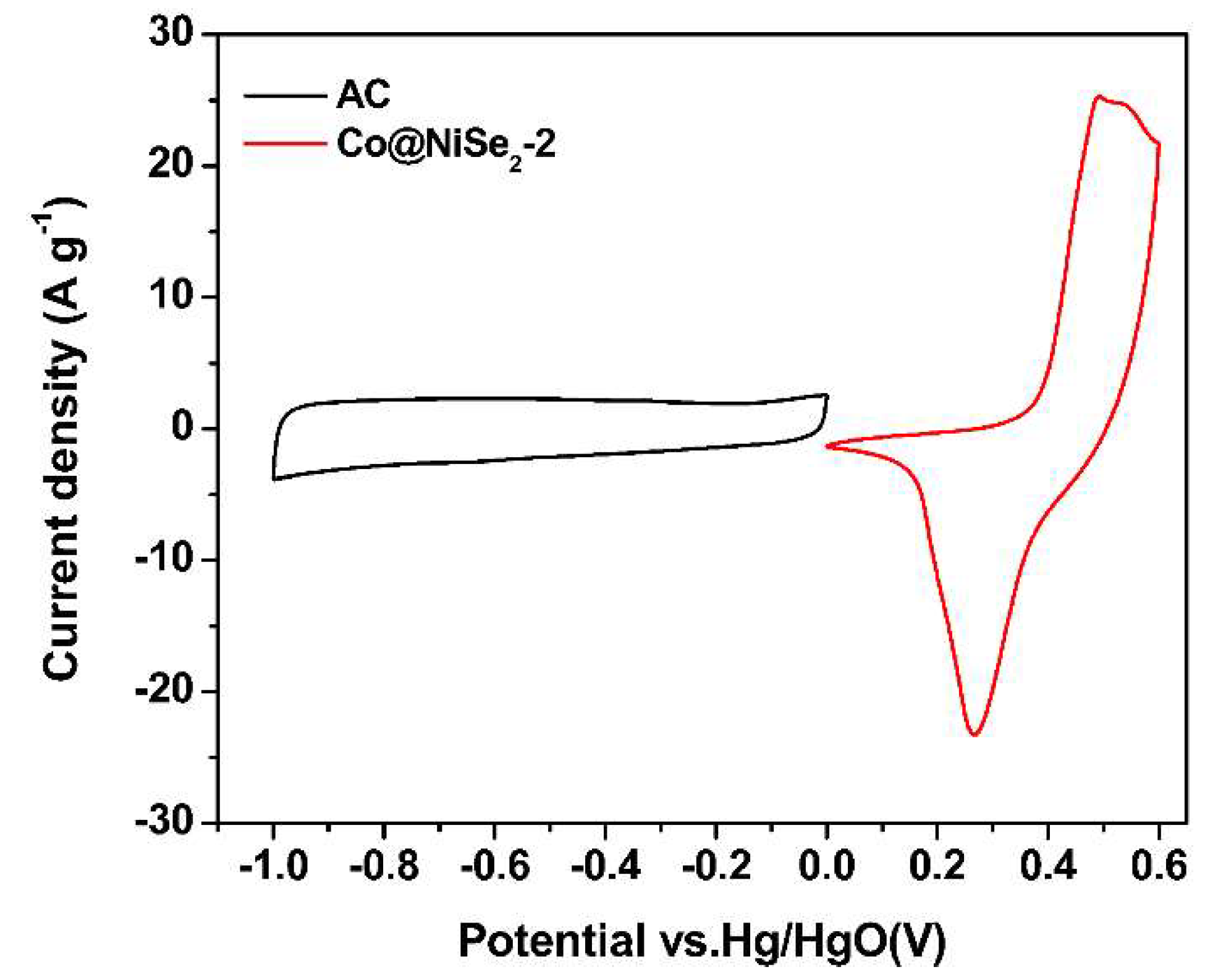
3.2. Electrochemical Properties of Electrode Materials
Electrochemical Tests of Electrode Materials in a Three-Electrode System
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bruce, P.G.; Freunberger, S.A.; Hardwick, L.J.; Tarascon, J.M. Li-O2 and Li-S batteries with high energy storage. Nat. Mater. 2011, 11, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zheng, S.; Xu, Y.; Xue, H.; Pang, H. Ruthenium based materials as electrode materials for supercapacitors. Chem. Eng. J. 2017, 333, 505–518. [Google Scholar] [CrossRef]
- Salanne, M.; Rotenberg, B.; Naoi, K.; Kaneko, K.; Taberna, P.L.; Grey, C.P.; Dunn, B.; Simon, P. Efficient storage mechanisms for building better supercapacitors. Nat. Energy 2016, 1, 16070. [Google Scholar] [CrossRef]
- Daraghmeh, A.; Hussain, S.; Servera, L.; Xuriguera, E.; Cornet, A.; Cirera, A. Impact of binder concentration and pressure on performance of symmetric CNFs based supercapacitors. Electrochim. Acta 2017, 245, 531–538. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, X.; Wang, Z.; Sun, F.; Dorrell, D.G. A review of supercapacitor modeling, estimation, and applications: A control/management perspective. Renew. Sustain. Energy Rev. 2017, 81, 1868–1878. [Google Scholar] [CrossRef]
- Wang, W.; Liu, W.Y.; Zeng, Y.X.; Han, Y.; Yu, M.H.; Lu, X.H.; Tong, Y.X. A novel exfoliation strategy to significantly boost the energy storage capability of commercial carbon cloth. Adv. Mater. 2015, 27, 3572–3578. [Google Scholar] [CrossRef] [PubMed]
- Cakici, M.; Reddy, K.R.; Alonso, M.F. Advanced electrochemical energy storage supercapacitors based on the flexible carbon fiber fabric-coated with uniform coral-like MnO2 structured electrodes. Chem. Eng. J. 2017, 309, 151–158. [Google Scholar] [CrossRef]
- Hulicova, J.D.; Puziy, A.M.; Poddubnaya, O.I.; Suarez, G.F.; Juan, M.D.; Lu, G.Q. Highly Stable Performance of Supercapacitors from Phosphorus-Enriched Carbons. J. Am. Chem. Soc. 2009, 131, 5026–5027. [Google Scholar] [CrossRef] [PubMed]
- Futaba, D.N.; Hata, K.; Yamada, T.; Hiraoka, T.; Hayamizu, Y.; Kakudate, Y.; Tanaike, O.; Hatori, H.; Yumura, M.; Iijima, S. Shape-engineerable and highly densely packed single-walled carbon nanotubes and their application as super-capacitor electrodes. Nat. Mater. 2006, 12, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.G.; Yang, M.; Hong, W.H.; Choi, J.W.; Huh, Y.S. 3D Macroporous Graphene Frameworks for Supercapacitors with High Energy and Power Densities. ACS Nano 2012, 6, 4020–4028. [Google Scholar] [CrossRef] [PubMed]
- Naderi, H.R.; Nasab, A.S.; Nasrabadi, M.R.; Ganjali, M.R. Decoration of nitrogen-doped reduced graphene oxide with cobalt tungstate nanoparticles for use in high-performance supercapacitors. Appl. Surf. Sci. 2017, 423, 1025–1034. [Google Scholar] [CrossRef]
- Miao, L.; Zhu, D.Z.; Zhao, Y.H.; Liu, M.X.; Duan, H.; Xiong, W.; Zhu, Q.J.; Li, L.C.; Gan, L.H. Design of carbon materials with ultramicro-, supermicro- and mesopores using solvent- and self-template strategy for supercapacitors. Microporous Mesoporous Mater. 2017, 253, 1–9. [Google Scholar] [CrossRef]
- Yan, J.; Ren, C.E.; Maleski, K.; Hatter, C.B.; Anasori, B.; Urbankowski, P.; Sarycheva, A.; Gogotsi, Y. Flexible MXene/Graphene Films for Ultrafast Supercapacitors with Outstanding Volumetric Capacitance. Adv. Funct. Mater. 2017, 27, 1701264. [Google Scholar] [CrossRef]
- Simon, P.; Gogotsi, Y. Materials for electrochemical capacitors. Nat. Mater. 2008, 7, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.Q.; Liu, G.J.; Wu, J.H.; Liu, L.; Lin, J.M.; Wei, Y.L. Asymmetric supercapacitor based on graphene oxide/polypyrrole composite and activated carbon electrodes. Electrochim. Acta 2014, 137, 26–33. [Google Scholar] [CrossRef]
- Fan, L.Q.; Liu, G.J.; Zhang, C.Y.; Wu, J.H.; Wei, Y.L. Facile one-step hydrothermal preparation of molybdenum disulfide/carbon composite for use in supercapacitor. Int. J. Hydrogen Energy 2015, 40, 10150–10157. [Google Scholar] [CrossRef]
- You, B.; Sun, Y.J. Hierarchically porous nickel sulfide multifunctional superstructures. Adv. Energy Mater. 2016, 6, 1502333–1502341. [Google Scholar] [CrossRef]
- Dong, X.; Wang, K.; Zhao, C.; Qian, X.; Chen, S.; Li, Z.; Liu, H.; Dou, S. Direct synthesis of RGO/Cu2O composite films on Cu foil for supercapacitors. J. Alloys Compd. 2014, 586, 745–753. [Google Scholar] [CrossRef]
- Wang, S.L.; Li, W.; Xin, L.P.; Wu, M.; Long, Y.; Huang, H.; Lou, X. Facile synthesis of truncated cube-like NiSe2 single crystals for high-performance asymmetric supercapacitors. Chem. Eng. J. 2017, 330, 1334–1341. [Google Scholar] [CrossRef]
- Huang, K.J.; Zhang, J.Z.; Fan, Y. Preparation of layered MoSe2 nanosheets on Ni-foam substrate with enhanced supercapacitor performance. Mater. Lett. 2015, 152, 244–247. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Pan, A.Q.; Wang, Y.P.; Cao, X.X.; Zhou, Z.; Zhu, T.; Liang, S.; Cao, G. Self-templated synthesis of N-doped CoSe2/C double-shelled dodecahedra for high-performance supercapacitors. Energy Storage Mater. 2017, 8, 28–34. [Google Scholar] [CrossRef]
- Wang, Y.G.; Song, Y.F.; Xia, Y.Y. Electrochemical capacitors: Mechanism, materials systems, characterization and applications. Chem. Soc. Rev. 2016, 45, 5925–5950. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.L.; Cui, S.Z.; Hou, H.W.; Chen, W.H.; Mi, L. Hierarchical ternary Ni-Co-Se nanowires for high-performance supercapacitor device design. Dalton Trans. 2016, 45, 19458–19465. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Wu, J.H.; Fan, L.Q.; Xu, K.Q.; Zhong, X.; Lin, Y.Z.; Lin, J.M. Improvement of the performance for quasi-solid-state supercapacitor by using PVA-KOH-KI polymer gel electrolyte. Electrochim. Acta 2011, 56, 6881–6886. [Google Scholar] [CrossRef]
- Yang, H.Z. Estimation of Supercapacitor Charge Capacity Bounds Considering Charge Redistribution. Chem. Eng. J. 2018, 345, 186–195. [Google Scholar] [CrossRef]
- Yan, J.; Fan, Z.; Wei, S.; Ning, G.Q.; Wei, T.; Zhang, Q.; Zhang, R.; Zhi, L.; Wei, F. Advanced Asymmetric Supercapacitors Based on Ni(OH)2 /Graphene and Porous Graphene Electrodes with High Energy Density. Adv. Funct. Mater. 2012, 22, 2632–2641. [Google Scholar] [CrossRef]
- Yan, J.; Wang, Q.; Wei, T.; Fan, Z. Recent Advances in Design and Fabrication of Electrochemical Supercapacitors with High Energy Densities. Adv. Energy Mater. 2014, 4, 1300816. [Google Scholar] [CrossRef]
- Mai, L.Q.; Minhas-Khan, A.; Tian, X.; Hercule, K.M.; Zhao, Y.-L.; Lin, X.; Xu, X. Synergistic interaction between redox-active electrolyte and binder-free functionalized carbon for ultrahigh supercapacitor performance. Nat. Commun. 2013, 4, 2923–2925. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jiang, J.; Li, H.; Zhao, X.S. A high-performance asymmetric supercapacitor fabricated with graphene-based electrodes. Energy Environ. Sci. 2011, 4, 4009–4015. [Google Scholar] [CrossRef]
- Tu, Q.M.; Fan, L.Q.; Pan, F.; Huang, J.L.; Gu, Y.; Lin, J.-M.; Huang, M.-L.; Huang, Y.-F.; Wu, J.-H. Design of a novel redox-active gel polymer electrolyte with a dual-role ionic liquid for flexible supercapacitors. Electrochim. Acta 2018, 268, 562–568. [Google Scholar] [CrossRef]
- Saha, D.; Li, Y.C.; Bi, Z.H.; Chen, J.H.; Keum, J.K.; Hensley, D.K.; Grappe, H.A.; Meyer, H.M.; Dai, S.; Paranthaman, M.P.; et al. Studies on Supercapacitor Electrode Material from Activated Lignin-Derived Mesoporous Carbon. Am. Chem. Soc. 2014, 30, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.H.; Ghosh, S.; Pradhan, B.; Dalui, A.; Shrestha, L.K.; Acharya, S.; Ariga, K. Two-Dimensional (2D) Nanomaterials towards Electrochemical Nanoarchitectonics in Energy-Related Applications. Bull. Chem. Soc. Jpn. 2017, 90, 627–648. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, S.I.; Yoon, J.C.; Jang, J.H. Chemical Vapor Deposition of Mesoporous Graphene Nanoballs for Supercapacitor. ACS Nano 2013, 7, 6047–6055. [Google Scholar] [CrossRef] [PubMed]
- Sengottaiyan, C.; Jayavel, R.; Bairi, P.; Shrestha, R.G.; Ariga, K.; Shrestha, L.K. Cobalt Oxide/Reduced Graphene Oxide Composite with Enhanced Electrochemical Supercapacitance Performance. Bull. Chem. Soc. Jpn. 2017, 90, 955–962. [Google Scholar] [CrossRef]
- Yu, W.; Jiang, X.; Ding, S.; Li, B.Q. Preparation and electrochemical characteristics of porous hollow spheres of NiO nanosheets as electrodes of supercapacitors. J. Power Sources 2014, 256, 440–448. [Google Scholar] [CrossRef]
- Wang, Y.; Mayorga-Martinez, C.C.; Pumera, M. Polyaniline/MoSX Supercapacitor by Electrodeposition. Bull. Chem. Soc. Jpn. 2017, 90, 847–853. [Google Scholar] [CrossRef]
- Guan, B.; Li, Y.; Yin, B.; Liu, K.; Wang, D.; Zhang, H.; Cheng, C. Synthesis of hierarchical NiS microflowers for high performance asymmetric supercapacitor. Chem. Eng. J. 2017, 308, 1165–1173. [Google Scholar] [CrossRef]
- Pang, H.; Wei, C.; Li, X.; Li, G.; Ma, Y.; Li, S.; Chen, J.; Zhang, J. Microwave-assisted synthesis of NiS2 nanostructures for supercapacitors and cocatalytic enhancing photocatalytic H2 production. Sci. Rep. 2014, 4, 3577. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Ji, X.; Jiang, J.; Yu, J.; Miao, L.; Bie, S.; Chen, H.; Ruan, Y. Hydrothermal synthesis of cobalt sulfide nanotubes: The size control and its application in supercapacitors. J. Power Sources 2013, 243, 396–402. [Google Scholar] [CrossRef]
- Zhou, P.; Fan, L.; Wu, J.; Gong, C.; Zhang, J.; Tu, Y. Facile hydrothermal synthesis of NiTe and its application as positive electrode material for asymmetric supercapacitor. J. Alloys Compd. 2016, 685, 384–390. [Google Scholar] [CrossRef]
- Dubal, D.P.; Lee, S.H.; Kim, J.G.; Kim, W.B.; Lokhande, C.D. Porous polypyrrole clusters prepared by electropolymerization for a high performance supercapacitor. J. Mater. Chem. 2012, 22, 3044–3052. [Google Scholar] [CrossRef]
- Wang, H.; Casalongue, H.S.; Liang, Y.; Dai, H. Ni(OH)2 nanoplates grown on graphene as advanced electrochemical pseudocapacitor materials. J. Am. Chem. Soc. 2010, 132, 7472–7477. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ji, X.; Chen, H. The investigation of the electrochemically supercapacitive performances of mesoporous CuCo2S4. RSC Adv. 2016, 6, 84236–84241. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Lou, X.W. General Solution Growth of Mesoporous NiCo2O4 Nanosheets on Various Conductive Substrates as High-Performance Electrodes for Supercapacitors. Adv. Mater. 2013, 25, 976–979. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.S.; Wang, H.; Ji, S.; Linkov, V.; Wang, R. Core-shell structured Ni3S2@Co(OH)2 nano-wires grown on Ni foam as binder-free electrode for asymmetric supercapacitors. Chem. Eng. J. 2018, 345, 48–57. [Google Scholar] [CrossRef]


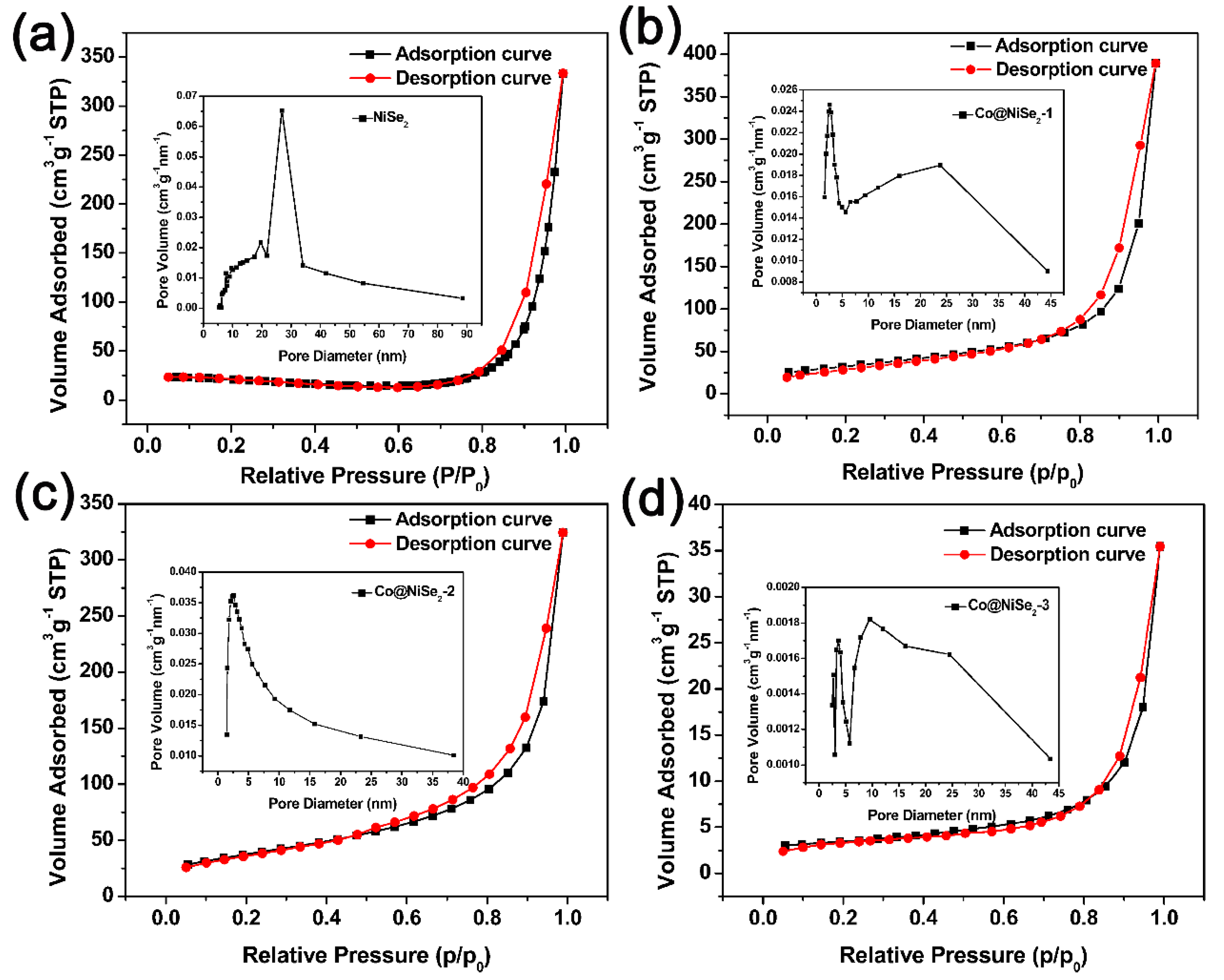

| Sample | BET (m2·g−1) | Pore Volume (cm3·g−1) | Pore Size (nm) |
|---|---|---|---|
| NiSe2 | 54.6 | 0.5 | >30 |
| Co@NiSe2-1 | 115.7 | 0.6 | >25 |
| Co@NiSe2-2 | 135.2 | 0.5 | >5 |
| Co@NiSe2-3 | 11.5 | 0.05 | >25 |
| Electrode Material | Electrolyte | Morphology Structure | Specific Capacitance (F·g−1) | Ref. |
|---|---|---|---|---|
| Co3O4/rGO | 2 M KOH | Cubic morphology | 487 F·g−1 at 5 mV·s−1 | [33] |
| NiO | 2 M KOH | Nanosheet hollow spheres | 600 F·g−1 at 10 A·g−1 | [34] |
| PANI/MoSX | 1 M H2SO4 | Nanoparticle | 48.64 mF·cm−2 at 56.62 μA·cm−2 | [35] |
| GO/PPy | 1 M Na2SO4 | Nanoparticle | 332.6 F·g−1 at 0.25 A·g−1 | [36] |
| NiS | 3 M KOH | Microflower | 1122.7 F·g−1 at 1 A·g−1 | [37] |
| CoS | 6 M KOH | Nanotubes | 285 F·g−1 at 0.5 A·g−1 | [38] |
| NiTe | 3 M KOH | Nanorods | 804 F·g−1 at 1 A·g−1 | [39] |
| CoSe/C | 2 M KOH | Nanoparticles | 726 F·g−1 at 1 A·g−1 | [20] |
| NiSe2 | 4 M KOH | Nano cube | 1044 F·g−1 at 3 A·g−1 | [18] |
| MoSe2 | 6 M KOH | Nanosheets | 1114.3 F·g−1 at 1 A·g−1 | [19] |
| Co-doped NiSe2 | 3 M KOH | Nanowire | 3167.6 F·g−1 at 1 A·g−1 | This work |
| Sample | Rs (Ω·cm2) | Rct (Ω·cm2) | Zw (Ω·s−1/2·cm2) |
|---|---|---|---|
| NiSe2 | 1.14 | 1.45 | 0.0013 |
| Co@NiSe2-1 | 0.93 | 1.48 | 0.0026 |
| Co@NiSe2-2 | 0.72 | 0.82 | 0.0012 |
| Co@NiSe2-3 | 0.86 | 1.11 | 0.0015 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, Y.; Fan, L.-Q.; Huang, J.-L.; Geng, C.-L.; Lin, J.-M.; Huang, M.-L.; Huang, Y.-F.; Wu, J.-H. Hydrothermal Synthesis of Co-Doped NiSe2 Nanowire for High-Performance Asymmetric Supercapacitors. Materials 2018, 11, 1468. https://doi.org/10.3390/ma11081468
Gu Y, Fan L-Q, Huang J-L, Geng C-L, Lin J-M, Huang M-L, Huang Y-F, Wu J-H. Hydrothermal Synthesis of Co-Doped NiSe2 Nanowire for High-Performance Asymmetric Supercapacitors. Materials. 2018; 11(8):1468. https://doi.org/10.3390/ma11081468
Chicago/Turabian StyleGu, Yun, Le-Qing Fan, Jian-Ling Huang, Cheng-Long Geng, Jian-Ming Lin, Miao-Liang Huang, Yun-Fang Huang, and Ji-Huai Wu. 2018. "Hydrothermal Synthesis of Co-Doped NiSe2 Nanowire for High-Performance Asymmetric Supercapacitors" Materials 11, no. 8: 1468. https://doi.org/10.3390/ma11081468
APA StyleGu, Y., Fan, L.-Q., Huang, J.-L., Geng, C.-L., Lin, J.-M., Huang, M.-L., Huang, Y.-F., & Wu, J.-H. (2018). Hydrothermal Synthesis of Co-Doped NiSe2 Nanowire for High-Performance Asymmetric Supercapacitors. Materials, 11(8), 1468. https://doi.org/10.3390/ma11081468





