Microwave-Assisted Rapid Preparation of Nano-ZnO/Ag Composite Functionalized Polyester Nonwoven Membrane for Improving Its UV Shielding and Antibacterial Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of ZnO–Ag Composite Coated Polyester Nonwoven
2.3. Characterizations
2.4. Washing Stability
2.5. Ultraviolet Resistant Properties
2.6. Antibacterial Properties
3. Results and Discussion
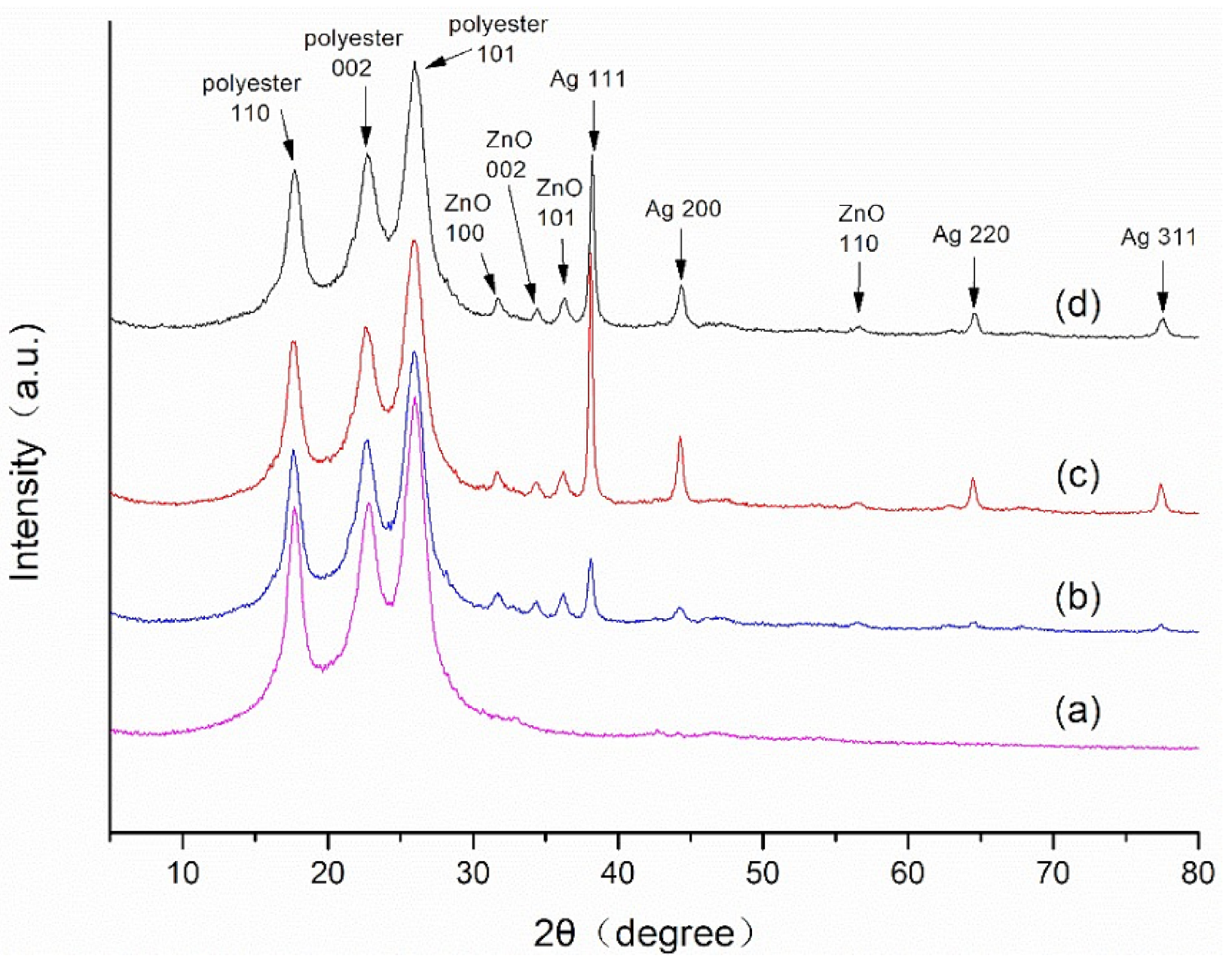
3.1. Crystalline Structure Analysis
3.2. Surface Morphology Analysis
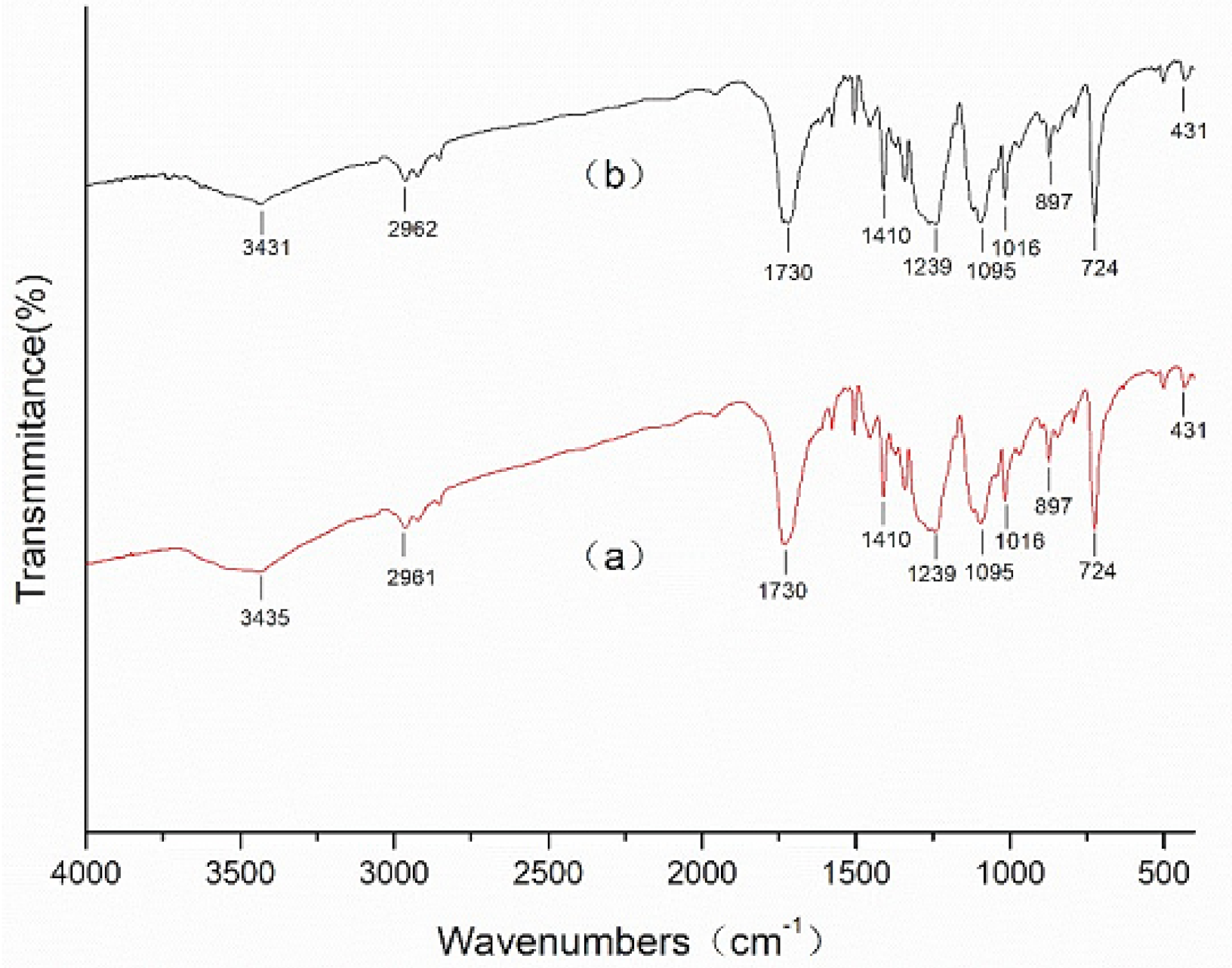
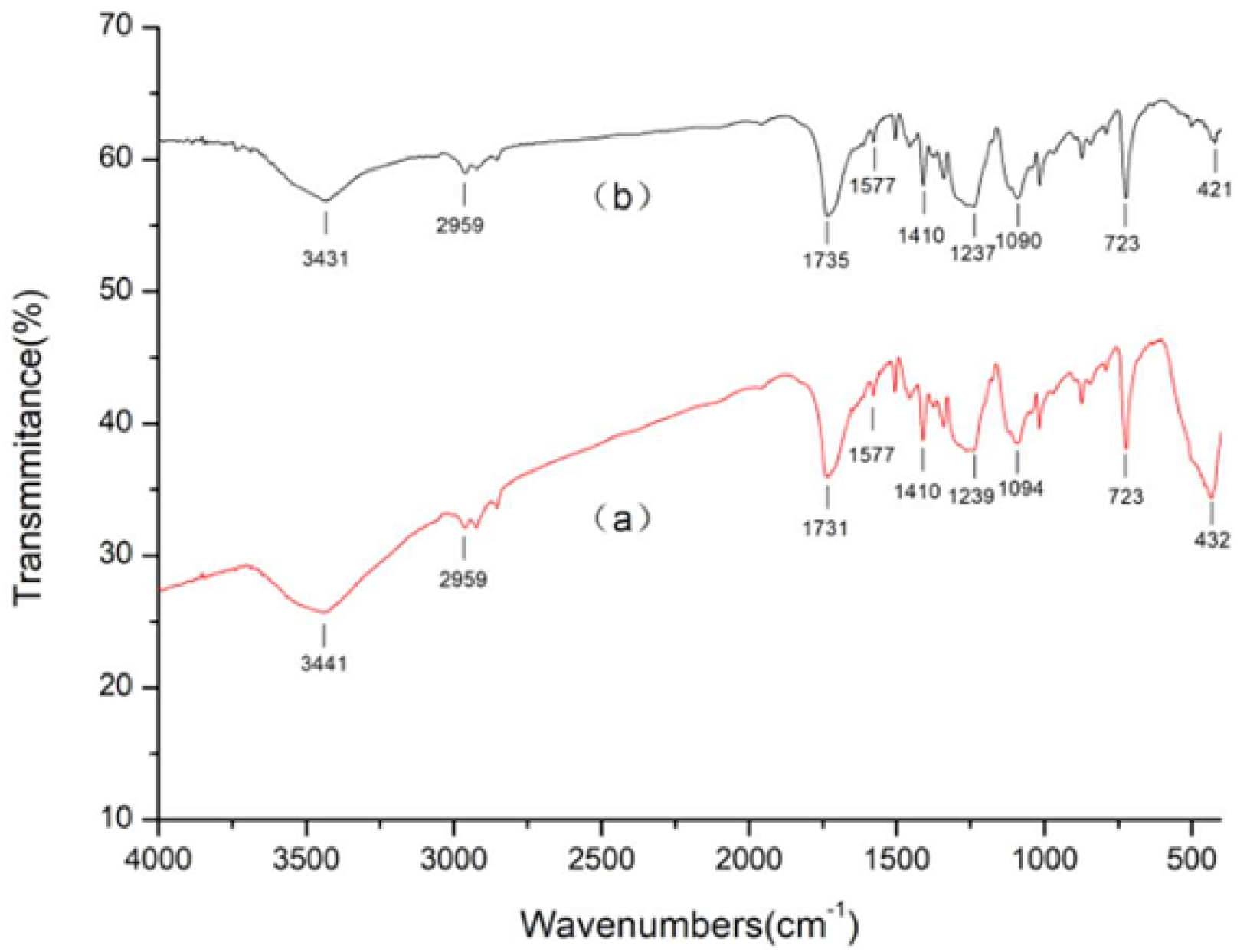
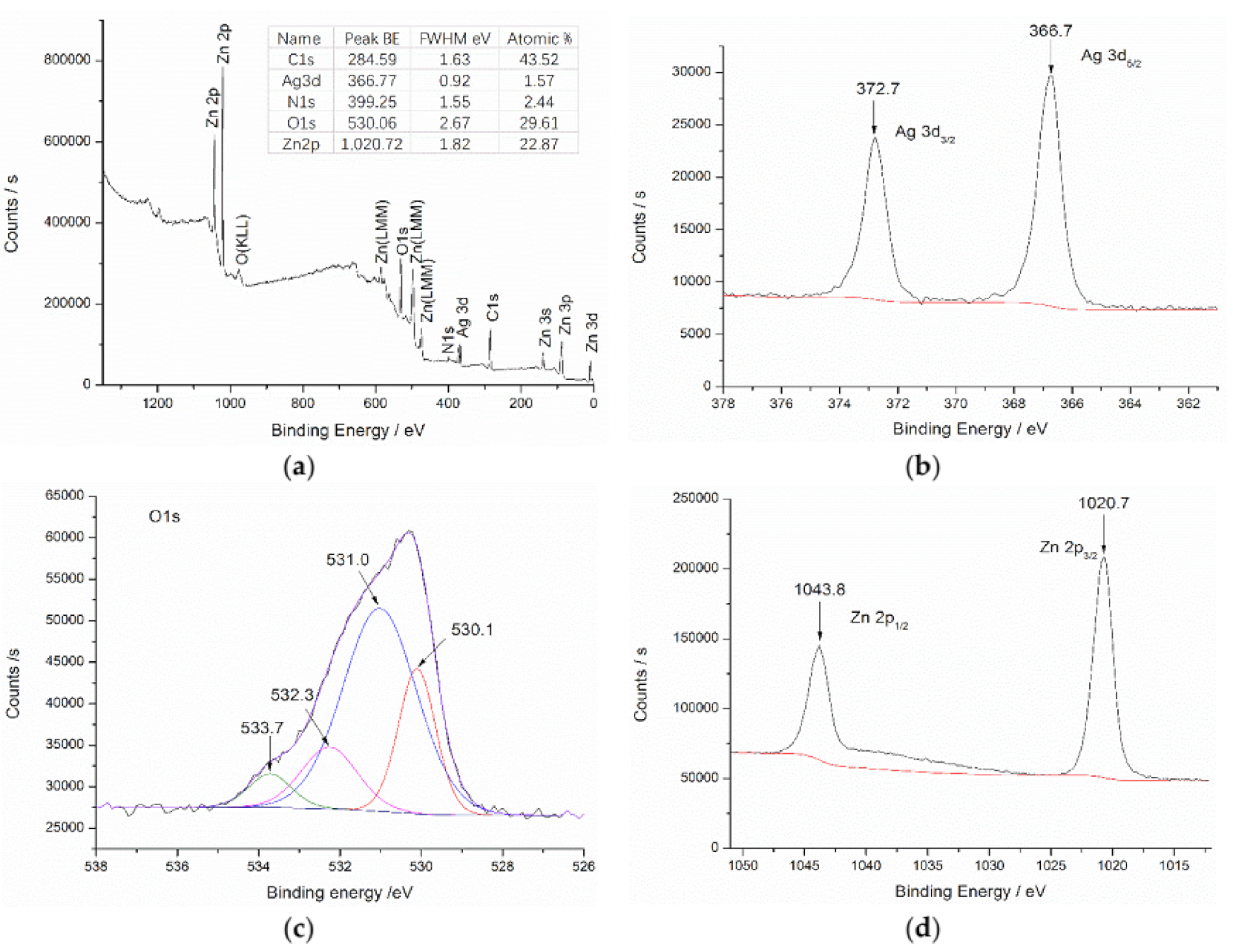
3.3. Surface Chemistry Analysis
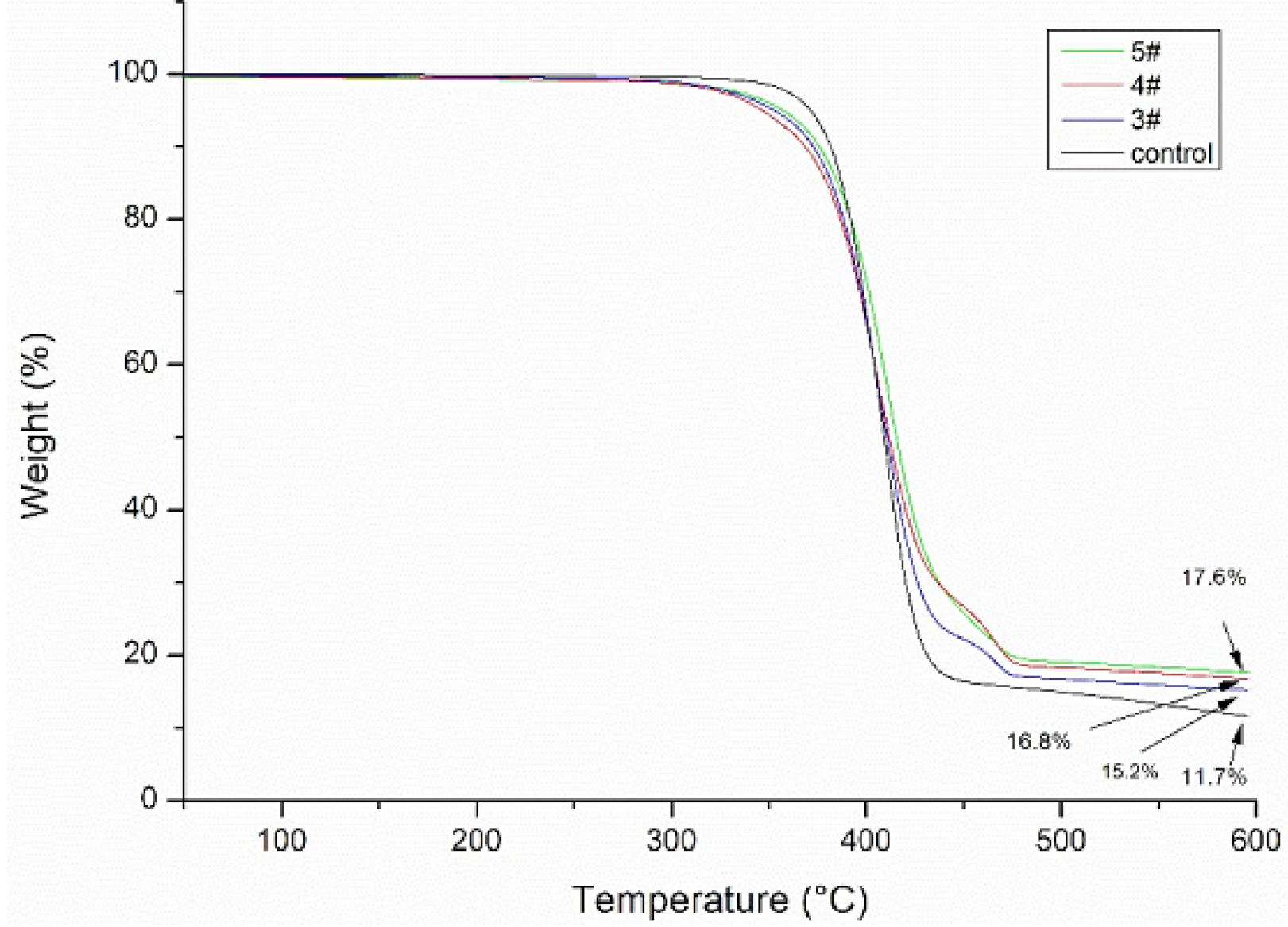
3.4. Thermogravimetric Analyses
3.5. Washing Stability Properties
3.6. Ultraviolet Resistant Properties
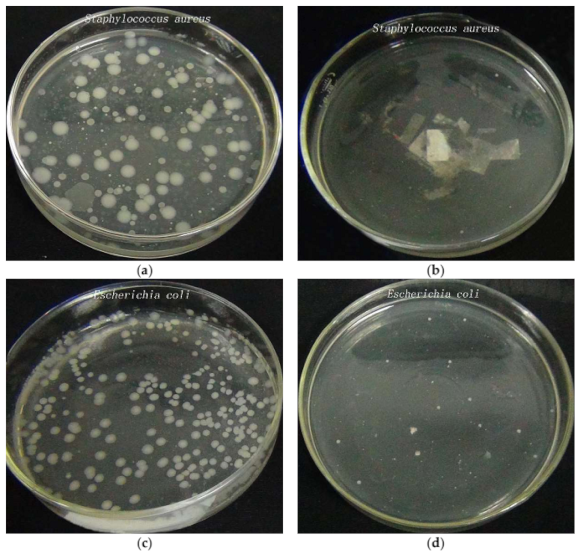
3.7. Antibacterial Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yu, L.; Zhang, S.; Liu, W.; Zhu, X.; Chen, X.; Chen, X. Improving the flame retardancy of pet fabric by photo-induced grafting. Polym. Degrad. Stab. 2010, 95, 1934–1942. [Google Scholar] [CrossRef]
- Ibănescu, M.; Muşat, V.; Textor, T.; Badilita, V.; Mahltig, B. Photocatalytic and antimicrobial Ag/ZnO nanocomposites for functionalization of textile fabrics. J. Alloys Compd. 2014, 610, 244–249. [Google Scholar] [CrossRef]
- Yuan, X.; Xu, W.; Huang, F.; Chen, D.; Wei, Q. Polyester fabric coated with Ag/ ZnO composite film by magnetron sputtering. Appl. Surf. Sci. 2016, 390, 863–869. [Google Scholar] [CrossRef]
- Ho Kim, J.; Hwan Lee, J.; Kim, S.-W.; Yoo, Y.-Z.; Seong, T.-Y. Highly flexible ZnO/Ag/ZnO conducting electrode for organic photonic devices. Ceram. Int. 2015, 41, 7146–7150. [Google Scholar] [CrossRef]
- Wei, Y.; Ke, L.; Kong, J.; Liu, H.; Jiao, Z.; Lu, X.; Du, H.; Sun, X.W. Enhanced photoelectrochemical water-splitting effect with a bent ZnO nanorod photo anode decorated with Ag nanoparticles. Nanotechnology 2012, 23, 235401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhao, Y.; Jia, Z.; Qin, Z.; Chu, L.; Yang, J.; Zhang, J.; Huang, W.; Li, X.A. High stable, transparent and conductive ZnO/Ag/ZnO nanofilm electrodes on rigid/flexible substrates. Energies 2016, 9, 443. [Google Scholar] [CrossRef]
- Seung, W.; Gupta, M.K.; Lee, K.Y.; Shin, K.S.; Lee, J.H.; Kim, T.Y.; Kim, S.; Lin, J.; Kim, J.H.; Kim, S.W. Nanopatterned textile-based wearable triboelectric nanogenerator. ACS Nano. 2015, 9, 3501–3509. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, D.-S.; Kim, S.-K.; Yoo, Y.-Z.; Hwan Lee, J.; Kim, S.-W.; Seong, T.-Y. Highly flexible Al-doped ZnO/Ag/Al-doped ZnO multilayer films deposited on PET substrates at room temperature. Ceram. Int. 2016, 42, 3473–3478. [Google Scholar] [CrossRef]
- Sarma, B.; Sarma, B.K. Role of residual stress and texture of ZnO nanocrystals on electro-optical properties of ZnO/Ag/ZnO multilayer transparent conductors. J. Alloys Compd. 2018, 734, 210–219. [Google Scholar] [CrossRef]
- Rajabi, M.; Ghorbani, M. Performance evaluation of nanogenerators based on Ag doped ZnO nanorods. Sens. Actuators A Phys. 2017, 266, 338–344. [Google Scholar] [CrossRef]
- Mirzaei, A.; Neri, G. Microwave-assisted synthesis of metal oxide nanostructures for gas sensing application: A review. Sens. Actuators B 2016, 237, 749–775. [Google Scholar] [CrossRef]
- Li, Y.; Hou, Y.; Zou, Y. Microwave assisted fabrication of nano-ZnO assembled cotton fibers with excellent UV blocking property and water-wash durability. Fibers Polym. 2012, 13, 185–190. [Google Scholar] [CrossRef]
- Tran Thi, V.H.; Lee, B.-K. Development of multifunctional self-cleaning and UV blocking cotton fabric with modification of photoactive ZnO coating via microwave method. J. Photochem. Photobiol. A 2017, 338, 13–22. [Google Scholar] [CrossRef]
- Peng, L.; Guo, R.; Lan, J.; Jiang, S.; Lin, S. Microwave-assisted deposition of silver nanoparticles on bamboo pulp fabric through dopamine functionalization. Appl. Surf. Sci. 2016, 386, 151–159. [Google Scholar] [CrossRef]
- Rehan, M.; Mowafi, S.; Abdelmoez Aly, S.; Elshemy, N.S.; Haggag, K. Microwave-heating for in-situ Ag NPs preparation into viscose fibers. Eur. Polym. J. 2017, 86, 68–84. [Google Scholar] [CrossRef]
- Poortavasoly, H.; Montazer, M.; Harifi, T. Aminolysis of polyethylene terephthalate surface along with in situ synthesis and stabilizing ZnO nanoparticles using triethanolamine optimized with response surface methodology. Mater. Sci. Eng. C 2016, 58, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Poortavasoly, H.; Montazer, M.; Harifi, T. Simultaneous synthesis of nano silver and activation of polyester producing higher tensile strength aminohydroxylated fiber with antibacterial and hydrophilic properties. RSC Adv. 2014, 4, 46250–46256. [Google Scholar] [CrossRef]
- Textiles—Evaluation for Solar Ultraviolet Radiation Protective Properties; Standardization Administration of the People’s Republic of China: Beijing, China, 2009; GB/T 18830.
- Feng, Y.; Shen, J.; Liu, X.; Zhao, Y.; Liang, X.; Huang, J.; Min, J.; Wang, L.; Shi, W. The preparation and optical properties of Ag and Ag/ZnO composite structure. J. Sol Gel Sci. Technol. 2016, 79, 98–106. [Google Scholar] [CrossRef]
- Wen, Q.; Dong, L.; Sun, X.; Zhuang, J.; Chen, Z. Preparation of multistage sheet-cluster zno photoanode via a solid state reaction and its property in dsscs. Chem. Res. Chin. Univ. 2016, 32, 437–442. [Google Scholar] [CrossRef]
- Liu, H.R.; Shao, G.X.; Zhao, J.F.; Zhang, Z.X.; Zhang, Y.; Liang, J.; Liu, X.G.; Jia, H.S.; Xu, B.S. Worm-like Ag/ZnO core–shell heterostructural composites: Fabrication, characterization, and photocatalysis. J. Phys. Chem. C 2012, 116, 16182–16190. [Google Scholar] [CrossRef]
- Allahyarzadeh, V.; Montazer, M.; Nejad, N.H.; Samadi, N. In situ synthesis of nano silver on polyester using NaOH/Nano TiO2. J. Appl. Polym. Sci. 2013, 129, 892–900. [Google Scholar] [CrossRef]
- El-Saftawy, A.A.; Elfalaky, A.; Ragheb, M.S.; Zakhary, S.G. Electron beam induced surface modifications of pet film. Radiat. Phys. Chem. 2014, 102, 96–102. [Google Scholar] [CrossRef]
- Kadam, A.N.; Bhopate, D.P.; Kondalkar, V.V.; Majhi, S.M.; Bathula, C.D.; Tran, A.-V.; Lee, S.-W. Facile synthesis of Ag-ZnO core–shell nanostructures with enhanced photocatalytic activity. Ind. Eng. Chem. 2018, 61, 78–86. [Google Scholar] [CrossRef]
- Sharma, S.K.; Ghodake, G.S.; Kim, D.Y.; Kim, D.-Y.; Thakur, O.P. Synthesis and characterization of hybrid Ag-ZnO nanocomposite for the application of sensor selectivity. Curr. Appl. Phys. 2018, 18, 377–383. [Google Scholar] [CrossRef]
- Gozdziewska, M.; Cichowicz, G.; Markowska, K.; Zawada, K.; Megiel, E. Nitroxide-coated silver nanoparticles: Synthesis, surface physicochemistry and antibacterial activity. RSC Adv. 2015, 5, 58403–58415. [Google Scholar] [CrossRef]
- Bazant, P.; Kuritka, I.; Munster, L.; Kalina, L. Microwave solvothermal decoration of the cellulose surface by nanostructured hybrid Ag/ZnO particles: A joint XPS, XRD and SEM study. Cellulose 2015, 22, 1275–1293. [Google Scholar] [CrossRef]
- Sanaee, Z.; Mohajerzadeh, S.; Zand, K.; Gard, F.S.; Pajouhi, H. Minimizing permeability of pet substrates using oxygen plasma treatment. Appl. Surf. Sci. 2011, 257, 2218–2225. [Google Scholar] [CrossRef]
- Gerenser, L.J. Photoemission investigation of silver/poly(ethylene terephthalate) interfacial chemistry: The effect of oxygen-plasma treatment. J. Vac. Sci. Technol. A 1990, 8, 3682–3691. [Google Scholar] [CrossRef]
- Krystosiak, P.; Tomaszewski, W.; Megiel, E. High-density polystyrene-grafted silver nanoparticles and their use in the preparation of nanocomposites with antibacterial properties. J. Colloid Interface Sci. 2017, 498, 9–21. [Google Scholar] [CrossRef] [PubMed]
- López-Heras, M.; Theodorou, I.G.; Leo, B.F.; Ryan, M.P.; Porter, A.E. Towards understanding the antibacterial activity of ag nanoparticles: Electron microscopy in the analysis of the materials-biology interface in the lung. Environ. Sci. Nano. 2015, 2, 312–326. [Google Scholar] [CrossRef]
- Shi, L.E.; Li, Z.H.; Zheng, W.; Zhao, Y.F.; Jin, Y.F.; Tang, Z.X. Synthesis, antibacterial activity, antibacterial mechanism and food applications of ZnO nanoparticles: A review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess 2014, 31, 173–186. [Google Scholar] [CrossRef] [PubMed]
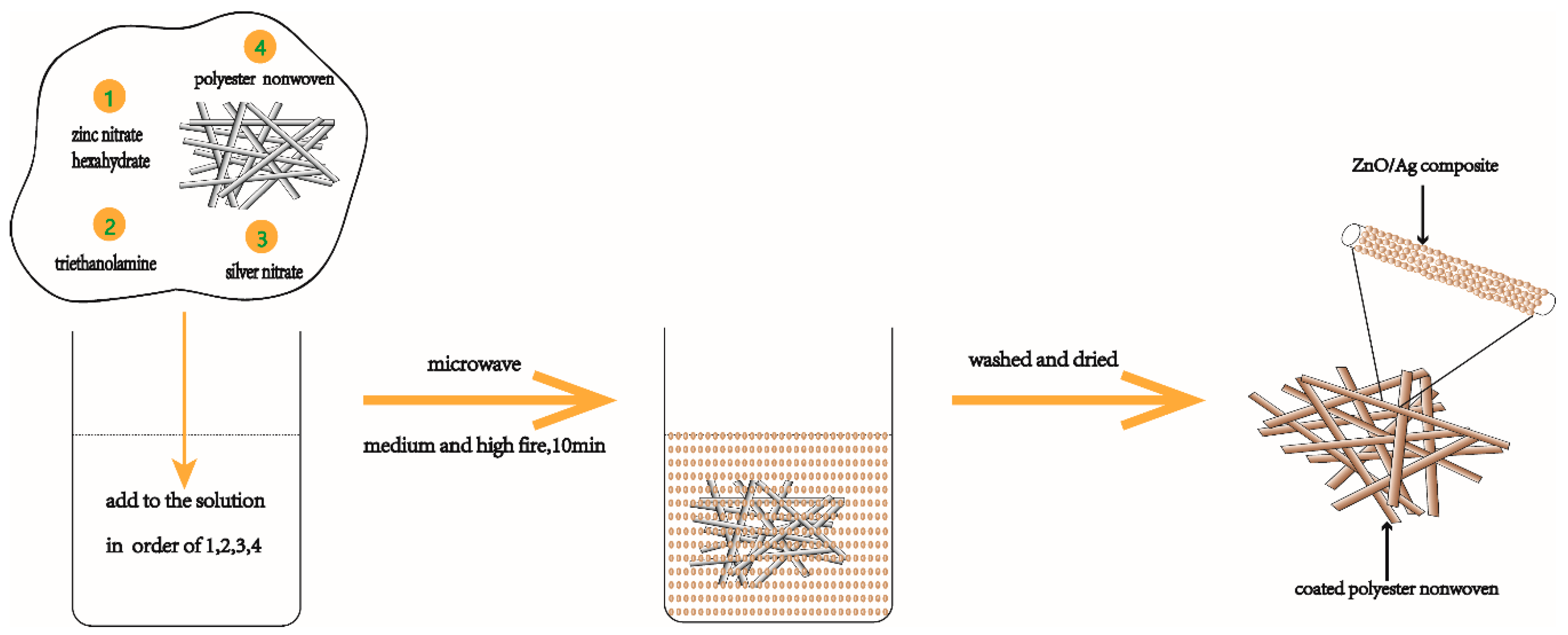


| Sample | H2O (mL) | Triethanolamine (mL) | Zn(NO3)2·6H2O (g) | AgNO3 (g) |
|---|---|---|---|---|
| Control | 0 | 0 | 0 | 0 |
| 1# | 80 | 20 | 0 | 0 |
| 2# | 80 | 20 | 2.97 | 0 |
| 3# | 80 | 20 | 2.97 | 0.1 |
| 4# | 80 | 20 | 2.97 | 0.2 |
| 5# | 80 | 20 | 2.97 | 0.3 |
| Sample | Treated Weight (g) | Treated Weight Gain (g) | 30 min | 60 min | 90 min | |||
|---|---|---|---|---|---|---|---|---|
| Weight Loss (g) | Weight Loss (%) | Weight Loss (g) | Weight Loss (%) | Weight Loss (g) | Weight Loss (%) | |||
| 3# | 0.2134 | 0.0152 | 0.0015 | 0.70 | 0.0045 | 2.11 | 0.0052 | 2.44 |
| 4# | 0.2213 | 0.0186 | 0.0052 | 2.35 | 0.0087 | 3.93 | 0.0092 | 4.16 |
| 5# | 0.2103 | 0.0162 | 0.0003 | 0.14 | 0.0021 | 1.00 | 0.0033 | 1.57 |
| Sample | Average T(UVA) (%) | Average T(UVB) (%) | Average UPF |
|---|---|---|---|
| Control | 18.66 | 2.10 | 14.6 |
| 3# | 7.46 | 0.12 | 166.8 |
| 4# | 5.82 | 0.12 | 219.8 |
| 5# | 6.22 | 0.14 | 197.4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shao, D.; Wei, Q. Microwave-Assisted Rapid Preparation of Nano-ZnO/Ag Composite Functionalized Polyester Nonwoven Membrane for Improving Its UV Shielding and Antibacterial Properties. Materials 2018, 11, 1412. https://doi.org/10.3390/ma11081412
Shao D, Wei Q. Microwave-Assisted Rapid Preparation of Nano-ZnO/Ag Composite Functionalized Polyester Nonwoven Membrane for Improving Its UV Shielding and Antibacterial Properties. Materials. 2018; 11(8):1412. https://doi.org/10.3390/ma11081412
Chicago/Turabian StyleShao, Dongfeng, and Qufu Wei. 2018. "Microwave-Assisted Rapid Preparation of Nano-ZnO/Ag Composite Functionalized Polyester Nonwoven Membrane for Improving Its UV Shielding and Antibacterial Properties" Materials 11, no. 8: 1412. https://doi.org/10.3390/ma11081412
APA StyleShao, D., & Wei, Q. (2018). Microwave-Assisted Rapid Preparation of Nano-ZnO/Ag Composite Functionalized Polyester Nonwoven Membrane for Improving Its UV Shielding and Antibacterial Properties. Materials, 11(8), 1412. https://doi.org/10.3390/ma11081412





