Compositional Dependence of Phase Selection in CoCrCu0.1FeMoNi-Based High-Entropy Alloys
Abstract
1. Introduction
2. Materials and Methods
3. Results
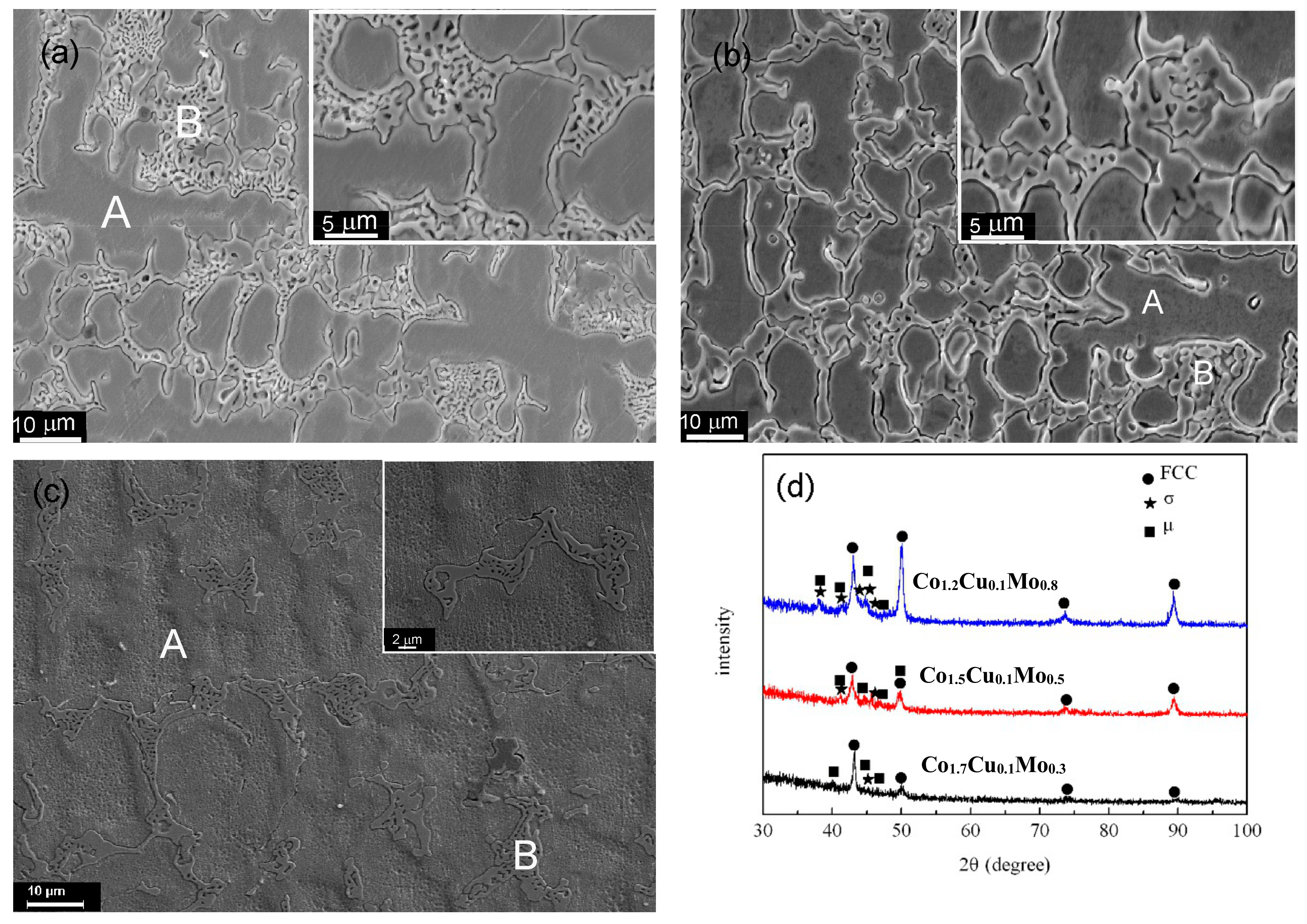
3.1. CoaCrCu0.1FeMo2−aNi Alloys
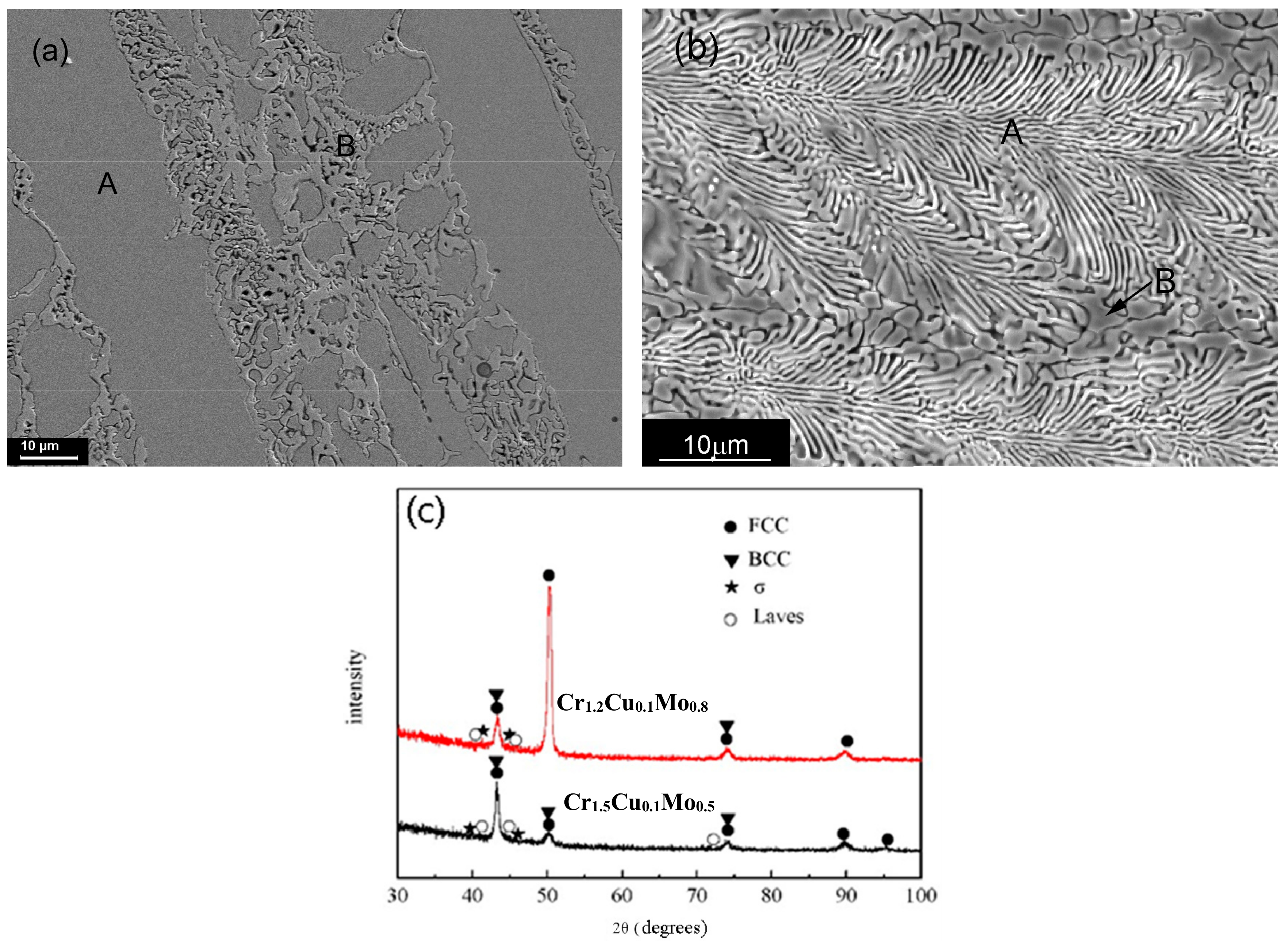
3.2. CoCrbCu0.1FeMo2−bNi Alloys
3.3. CoCrCu0.1FecMo2−cNi Alloys
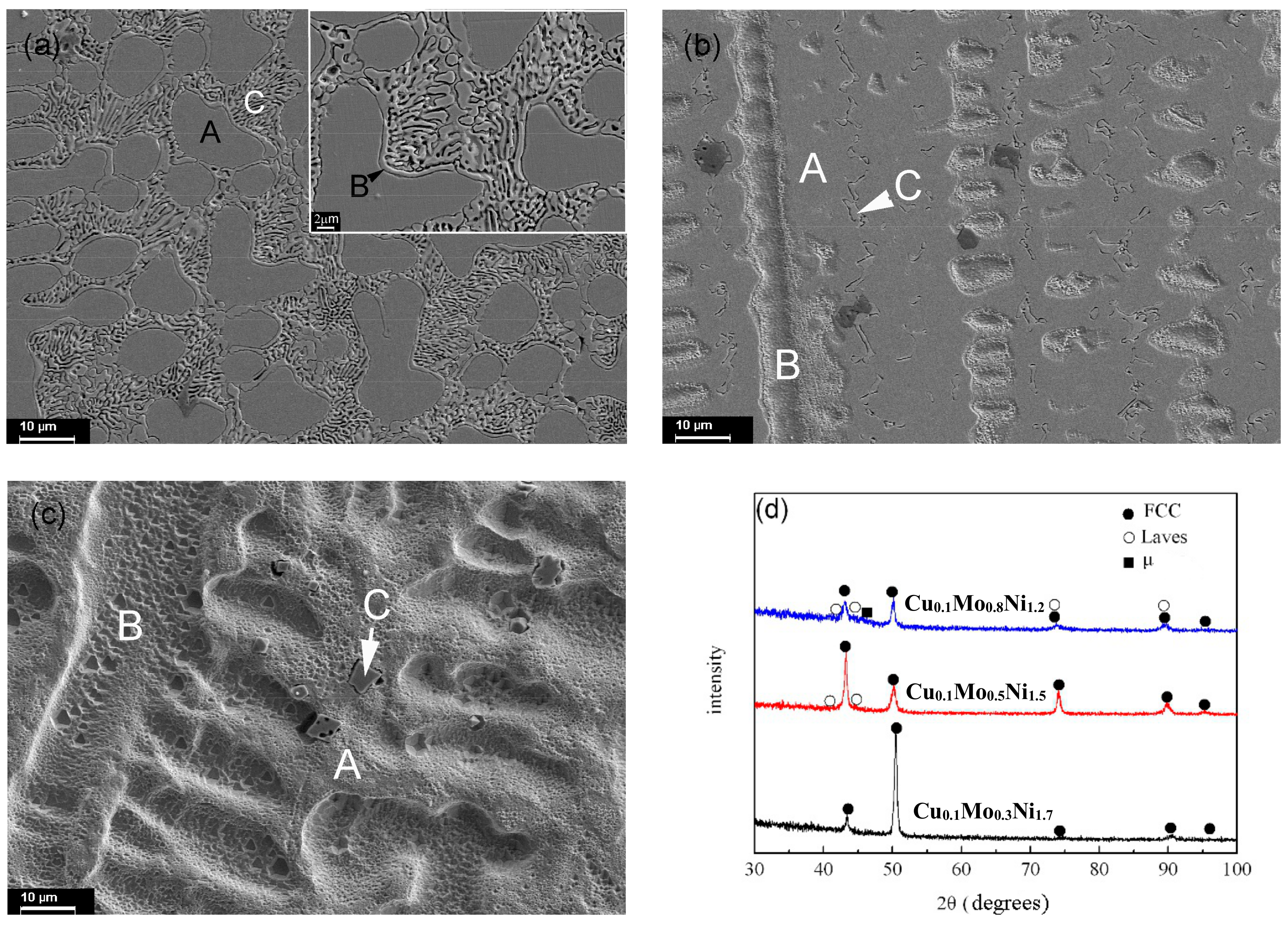
3.4. CoCrCu0.1FeMo2−dNid Alloys
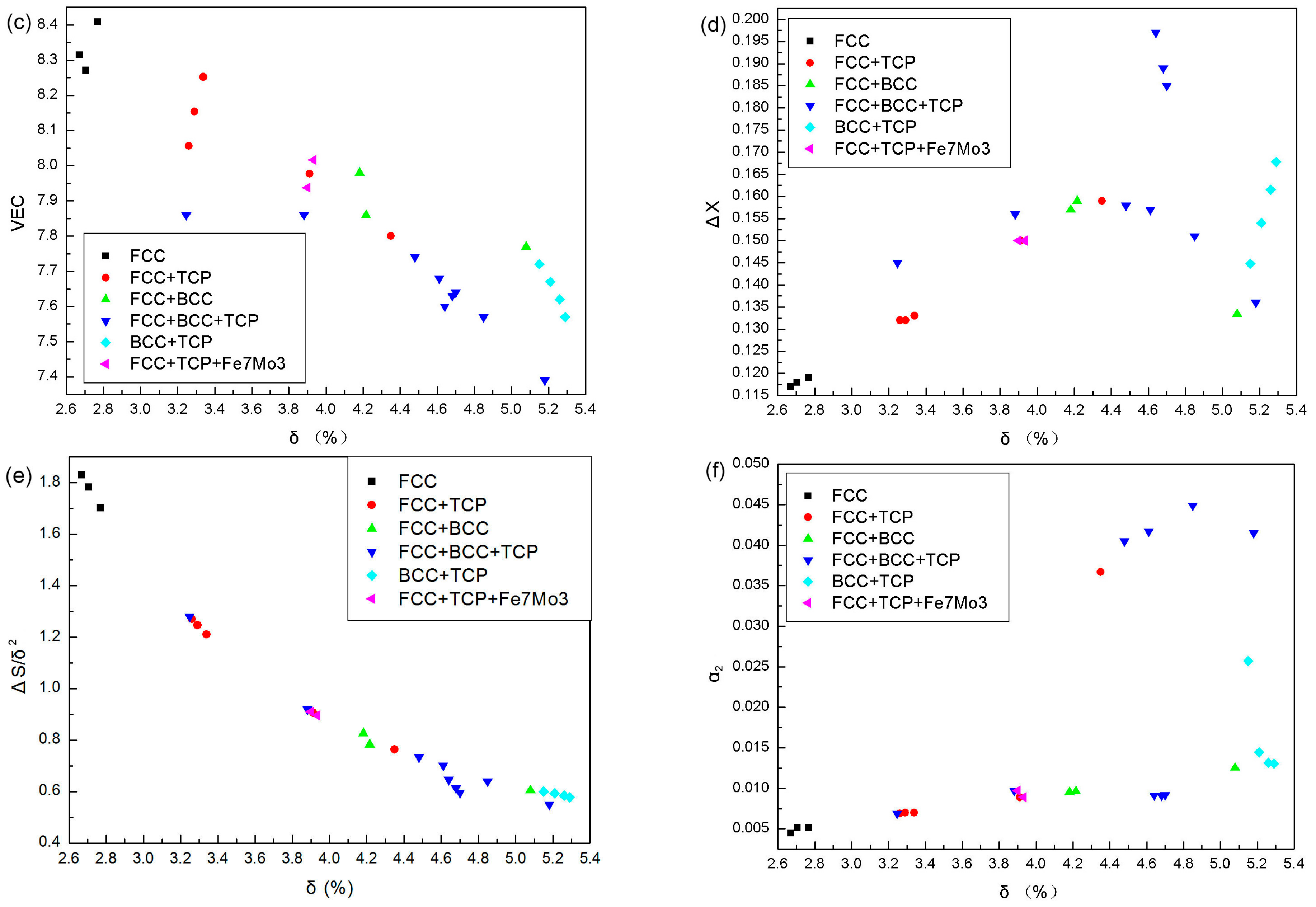
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cantor, B.; Chang, I.T.H.; Knight, P.; Vincent, A.J.B. Microstructure development in equiatomic multicomponent alloys. Mater. Sci. Eng. A 2004, 375, 213–218. [Google Scholar] [CrossRef]
- Miracle, D.B.; Senkov, O.N. A critical review of high entropy alloys and related concepts. Acta Mater. 2017, 122, 488–511. [Google Scholar] [CrossRef]
- Gorsse, S.; Miracle, D.B.; Senkov, O.N. Mapping the world of complex concentrated alloys. Acta Mater. 2017, 135, 177–187. [Google Scholar] [CrossRef]
- Senkov, O.N.; Rao, S.; Chaput, K.J.; Woodward, C. Compositional effect on microstructure and properties of NbTiZr-based complex concentrated alloys. Acta Mater. 2017, 122, 488–511. [Google Scholar] [CrossRef]
- Yurchenko, N.Y.; Stepanov, N.D.; Zherebtsov, S.V.; Tikhonovsky, M.A.; Salishchev, G.A. Structure and mechanical properties of B2 ordered refractory AlNbTiVZrx (x = 0–1.5) high-entropy alloys. Mater. Sci. Eng. A 2017, 704, 82–90. [Google Scholar] [CrossRef]
- Stepanov, N.D.; Shaysultanov, D.G.; Chernichenko, R.S.; Ikornikov, D.M.; Zherebtsov, S.V. Mechanical properties of a new high entropy alloy with a duplex ultra-fine grained structure. Mater. Sci. Eng. A 2018, 728, 82–90. [Google Scholar] [CrossRef]
- Yurchenko, N.Y.; Stepanov, N.D.; Gridneva, A.O.; Mishunin, M.V.; Salishchev, G.A.; Zherebtsov, S.V. Effect of Cr and Zr on phase stability of refractory Al-Cr-Nb-Ti-V-Zr high-entropy alloys. J. Alloys Compd. 2018, 757, 403–414. [Google Scholar] [CrossRef]
- Wang, Z.J.; Guo, S.; Liu, C.T. Phase Selection in High-Entropy Alloys: From Nonequilibrium to Equilibrium. JOM 2014, 66, 1966–1972. [Google Scholar] [CrossRef]
- Lu, Z.P.; Wang, H.; Chen, M.W.; Baker, I.; Yeh, J.W.; Liu, C.T.; Nieh, T.G. An assessment on the future development of high-entropy alloys: Summary from a recent workshop. Intermetallics 2015, 66, 67–76. [Google Scholar] [CrossRef]
- Lu, Y.; Dong, Y.; Guo, S.; Jiang, L.; Kang, H.; Wang, T.; Wen, B.; Wang, Z.; Jie, J.; Cao, Z.; et al. A promising new class of high-entropy alloys: Eutectic high-entropy alloys. Sci. Rep. 2014, 4, 6200. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Ng, C.; Liu, C.T. Anomalous solidification microstructures in Co-free AlxCrCuFeNi2 High-Entropy Alloys. J. Alloys Compd. 2013, 557, 77–81. [Google Scholar] [CrossRef]
- Liu, N.; Wu, P.H.; Peng, Z.; Xiang, H.F.; Chen, C.; Wang, X.J.; Zhang, J. Microstructure, phase stability and properties of CoCr0.5CuxFeyMoNi compositionally complex alloys. Mater. Sci. Technol. 2017, 133, 210–214. [Google Scholar] [CrossRef]
- Wu, P.H.; Peng, Z.; Liu, N.; Niu, M.; Zhu, Z.X.; Wang, X.J. The Effect of Mn Content on the Microstructure and Properties of CoCrCu0.1Fe0.15Mo1.5MnxNi near Equiatomic Alloys. Mater. Trans. 2016, 57, 5–8. [Google Scholar] [CrossRef]
- Jiang, L.; Cao, Z.Q.; Jie, J.C.; Zhang, J.J.; Lu, Y.P.; Wang, T.M.; Li, T.J. Effect of Mo and Ni elements on microstructure evolution and mechanism properties of the CoFeNixVMoy high entropy alloys. J. Alloys Compd. 2015, 649, 585–590. [Google Scholar] [CrossRef]
- He, F.; Wang, Z.J.; Cheng, P.; Wang, Q.; Li, J.J.; Dang, Y.Y.; Wang, J.C.; Liu, C.T. Designing eutectic high entropy alloys of CoCrFeNiNbx. J. Alloys Compd. 2015, 656, 284–289. [Google Scholar] [CrossRef]
- Zhu, J.M.; Zhang, H.F.; Fu, H.M.; Wang, A.M.; Li, H.; Hu, Z.Q. Microstructures and compressive properties of multicomponent AlCoCrFeNiMox alloys. Mater. Sci. Eng. A 2010, 527, 6975–6979. [Google Scholar] [CrossRef]
- Zhu, J.M.; Fu, H.M.; Zhang, H.F.; Wang, A.M.; Li, H.; Hu, Z.Q. Microstructures and compressive properties of multicomponent AlCoCrCuFeNiMox alloys. J. Alloys Compd. 2010, 497, 52–56. [Google Scholar] [CrossRef]
- He, F.; Wang, Z.J.; Zhu, M.; Li, J.J.; Dang, Y.Y.; Wang, J.C. The phase stability of Ni2CrFeMox multi-principal-component alloys with medium configurational entropy. Mater. Des. 2015, 85, 1–6. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Juan, C.C.; Wang, W.R.; Sheu, T.S.; Chen, S.K.; Yeh, J.W. On the superior hot hardness and softening resistance of AlCoCrxFeMo0.5Ni high-entropy alloys. Mater. Sci. Eng. A 2011, 528, 3581–3588. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Sheu, T.S.; Wang, W.R.; Tang, W.Y.; Chen, S.K.; Yeh, J.W. Microstructure and Mechanical Properties of New AlCoxCrFeMo0.5Ni High-Entropy Alloys. Adv. Eng. Mater. 2010, 12, 44–49. [Google Scholar] [CrossRef]
- Liu, W.H.; Lu, Z.P.; He, J.Y.; Luan, J.H.; Wang, Z.J.; Liu, B.; Liu, Y.; Chen, M.W.; Liu, C.T. Ductile CoCrFeNiMox high entropy alloys strengthened by hard intermetallic phases. Acta Mater. 2016, 116, 332–342. [Google Scholar] [CrossRef]
- Li, X.C.; Dou, D.; Zheng, Z.Y.; Li, J.C. Microstructure and Properties of FeAlCrNiMox High-Entropy Alloys. J. Mater. Eng. Perform. 2016, 25, 2164–2169. [Google Scholar] [CrossRef]
- Tian, L.H.; Xiong, W.; Liu, C.; Lu, S.; Fu, M. Microstructure and wear behavior of atmospheric plasma-sprayed AlCoCrFeNiTi high-entropy alloy coating. J. Mater. Eng. Perform. 2016, 25, 5513–5521. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, Y.J.; Lin, J.P.; Chen, G.L.; Liaw, P.K. Solid-solution phase formation rules for multi-component alloys. Adv. Eng. Mater. 2008, 10, 534–538. [Google Scholar] [CrossRef]
- Guo, S.; Liu, C.T. Phase stability in high entropy alloys: Formation of solid-solution phase or amorphous phase. Prog. Nat. Sci. 2011, 21, 433–446. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, X.; Liaw, P.K. Alloy Design and Properties Optimization of High-Entropy Alloys. JOM 2012, 64, 830–838. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, Y. Prediction of high-entropy stabilized solid-solution in multicomponent alloys. Mater. Chem. Phys. 2012, 132, 233–238. [Google Scholar] [CrossRef]
- Yang, X.; Chen, S.Y.; Cotton, J.D.; Zhang, Y. Phase stability of low-density, multiprincipal component alloys containing aluminum, magnesium, and lithium. JOM 2014, 10, 2009–2020. [Google Scholar] [CrossRef]
- Wang, Z.J.; Huang, Y.H.; Yang, Y.; Wang, J.C.; Liu, C.T. Atomic-size effect and solid solubility of multicomponent alloys. Scr. Mater. 2015, 94, 28–31. [Google Scholar] [CrossRef]
- Tsai, M.H.; Tsai, K.Y.; Tsai, C.W.; Lee, C.; Juan, C.C.; Yeh, J.W. Criterion for sigma phase formation in Cr- and V-containing high-entropy alloys. Mater. Res. Lett. 2013, 1, 207–212. [Google Scholar] [CrossRef]
- Dong, Y.; Lu, Y.; Jiang, L.; Wang, T.; Li, T. Effect of electro-negativity on the stability of topologically close-packed phase in high entropy alloys. Intermetallics 2014, 52, 105–109. [Google Scholar] [CrossRef]
- Wang, Z.J.; Qiu, W.F.; Yang, Y.; Liu, C.T. Atomic-size and lattice-distortion effects in newly developed high-entropy alloys with multiple principal elements. Intermetallics 2015, 64, 63–69. [Google Scholar] [CrossRef]
- Wu, P.H.; Liu, N.; Yang, W.; Zhu, Z.X.; Lu, Y.P.; Wang, X.J. Microstructure and Solidification Behavior of Multi-component CoCrCuxFeMoNi High-entropy alloys. Mater. Sci. Eng. A 2015, 642, 142–149. [Google Scholar] [CrossRef]
- Lu, Y.; Jiang, H.; Guo, S.; Wang, T.; Cao, Z.; Li, T. A new strategy to design eutectic high-entropy alloys using mixing enthalpy. Intermetallics 2017, 9, 124–128. [Google Scholar] [CrossRef]
- Wu, P.H. Microstructure and Solidification Behavior of Multi-Component CoCrCuxFeMoNi High-Entropy Alloys. Master’s Thesis, Jiangsu University of Science and Technology, Zhenjiang, China, 15 June 2016. [Google Scholar]
- Singh, A.K.; Kumar, N.; Dwivedi, A.; Subramaniam, A. A geometrical parameter for the formation of disordered solid solutions in multi-component alloys. Intermetallics 2014, 52, 105–109. [Google Scholar] [CrossRef]


| Alloys | Co | Cr | Cu | Fe | Mo | Ni |
|---|---|---|---|---|---|---|
| Co1.2CrCu0.1FeMo0.8Ni | 23.53 | 19.61 | 1.96 | 19.61 | 15.69 | 19.61 |
| Co1.5CrCu0.1FeMo0.5Ni | 29.41 | 19.61 | 1.96 | 19.61 | 9.8 | 19.61 |
| Co1.7CrCu0.1FeMo0.3Ni | 33.33 | 19.61 | 1.96 | 19.61 | 5.88 | 19.61 |
| CoCr1.2Cu0.1FeMo0.8Ni | 19.61 | 23.53 | 1.96 | 19.61 | 15.69 | 19.61 |
| CoCr1.5Cu0.1FeMo0.5Ni | 19.61 | 29.41 | 1.96 | 19.61 | 9.8 | 19.61 |
| CoCrCu0.1Fe1.2Mo0.8Ni | 19.61 | 19.61 | 1.96 | 23.53 | 15.69 | 19.61 |
| CoCrCu0.1Fe1.5Mo0.5Ni | 19.61 | 19.61 | 1.96 | 29.41 | 9.8 | 19.61 |
| CoCrCu0.1Fe1.7Mo0.3Ni | 19.61 | 19.61 | 1.96 | 33.33 | 5.88 | 19.61 |
| CoCrCu0.1Fe Mo0.8Ni1.2 | 19.61 | 19.61 | 1.96 | 19.61 | 15.69 | 23.53 |
| CoCrCu0.1Fe Mo0.5Ni1.5 | 19.61 | 19.61 | 1.96 | 19.61 | 9.8 | 29.41 |
| CoCrCu0.1Fe Mo0.3Ni1.7 | 19.61 | 19.61 | 1.96 | 19.61 | 5.88 | 33.33 |
| Alloy | Value | Region | Co | Cr | Cu | Fe | Mo | Ni |
|---|---|---|---|---|---|---|---|---|
| CoaCrCu0.1FeMo2−aNi | a = 1.2 | Content | 23.55 | 18.37 | 1.84 | 19.73 | 17.93 | 18.58 |
| A | 24.17 | 18.46 | 1.85 | 20.45 | 14.32 | 20.75 | ||
| B | 19.32 | 18.01 | 0.43 | 14.57 | 36.10 | 11.57 | ||
| a = 1.5 | Content | 27.92 | 20.60 | 1.8 | 19.63 | 9.5 | 19.55 | |
| A | 20.45 | 27.40 | 1.68 | 21.25 | 8.93 | 20.29 | ||
| B | 17.03 | 33.13 | 0.82 | 16.70 | 19.91 | 12.41 | ||
| a = 1.7 | Content | 33.63 | 19.57 | 1.75 | 19.8 | 6.00 | 19.25 | |
| A | 34.86 | 17.83 | 1.61 | 20.30 | 6.09 | 19.31 | ||
| B | 30.34 | 20.33 | 1.90 | 19.79 | 8.98 | 18.66 | ||
| CoCrbCu0.1FeMo2−bNi | b = 1.2 | Content | 19.32 | 25.69 | 1.85 | 20.44 | 12.36 | 20.34 |
| A | 18.68 | 30.11 | - | 20.20 | 9.53 | 21.47 | ||
| B | 22.51 | 22.99 | 1.77 | 17.02 | 17.53 | 18.17 | ||
| b = 1.5 | Content | 20.02 | 29.45 | 1.51 | 19.23 | 10.36 | 19.43 | |
| A | 15.50 | 26.95 | - | 18.31 | 24.98 | 14.25 | ||
| B | 16.76 | 24.25 | - | 19.83 | 19.13 | 20.03 | ||
| CoCrCu0.1FecMo2−cNi | c = 1.2 | Content | 15.33 | 21.76 | 1.83 | 23.53 | 17.92 | 19.63 |
| A | 15.70 | 21.66 | - | 20.57 | 29.21 | 12.86 | ||
| B | 14.95 | 22.12 | 0.40 | 20.93 | 29.95 | 11.65 | ||
| c = 1.5 | Content | 17.71 | 21.65 | 1.94 | 29.41 | 10.82 | 18.47 | |
| A | 17.63 | 19.24 | 2.25 | 31.42 | 10.49 | 18.95 | ||
| B | 15.51 | 21.98 | 0.34 | 23.16 | 27.19 | 11.83 | ||
| C | 15.94 | 22.96 | 0.62 | 22.66 | 24.96 | 12.85 | ||
| c = 1.7 | Content | 20.8 | 19.98 | 1.64 | 33.30 | 6.05 | 18.23 | |
| A | 18.95 | 19.89 | 1.58 | 32.19 | 8.45 | 18.94 | ||
| B | 22.54 | 20.36 | 1.89 | 34.23 | 2.81 | 18.16 | ||
| CoCrCu0.1FeMo2−dNid | d = 1.2 | Content | 19.37 | 19.63 | 1.36 | 19.47 | 16.64 | 23.53 |
| A | 19.49 | 18.93 | 1.20 | 21.82 | 13.50 | 25.06 | ||
| B | 16.80 | 22.33 | 0.67 | 16.58 | 27.68 | 15.95 | ||
| C | 17.03 | 21.79 | 0.45 | 13.97 | 32.56 | 14.20 | ||
| d = 1.5 | Content | 19.79 | 19.68 | 1.96 | 19.16 | 10.37 | 29.04 | |
| A | 19.09 | 19.13 | 2.15 | 17.77 | 13.22 | 28.65 | ||
| B | 21.82 | 20.07 | 1.71 | 21.02 | 3.28 | 32.10 | ||
| C | 18.28 | 22.86 | 0.67 | 15.67 | 21.82 | 20.70 | ||
| d = 1.7 | Content | 20.82 | 19.67 | 1.53 | 19.74 | 6.25 | 31.89 | |
| A | 17.80 | 21.90 | 1.84 | 18.78 | 8.06 | 31.62 | ||
| B | 21.38 | 19.25 | 1.65 | 19.93 | 4.60 | 33.18 | ||
| C | 2.44 | 88.74 | - | 3.85 | 0.68 | 4.29 |
| Alloy | δ (%) | VEC | ΔX | ΔH (kJ·mol−1) | ΔS (J·K−1·mol−1) | α2 | ΔS/δ2 | Phases | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Co1.2CrCu0.1FeMo0.8Ni | 3.912 | 7.977 | 0.150 | −0.878 | 13.853 | 0.0089 | 0.9052 | FCC + σ + μ | |
| Co1.5CrCu0.1FeMo0.5Ni | 3.291 | 8.154 | 0.132 | −0.807 | 13.492 | 0.0070 | 1.2457 | FCC + σ + μ | |
| Co1.7CrCu0.1FeMo0.3Ni | 2.705 | 8.271 | 0.118 | −0.740 | 13.037 | 0.0051 | 1.7817 | FCC | |
| CoCr1.2Cu0.1FeMo0.8Ni | 3.882 | 7.860 | 0.156 | −0.897 | 13.853 | 0.0097 | 0.9192 | FCC + BCC + Laves | |
| CoCr1.5Cu0.1FeMo0.5Ni | 3.247 | 7.860 | 0.145 | −0.882 | 13.492 | 0.0069 | 1.2797 | FCC + BCC + σ + Laves | |
| CoCrCu0.1Fe1.2Mo0.8Ni | 3.900 | 7.938 | 0.150 | −0.847 | 13.853 | 0.0097 | 0.9108 | FCC + Laves + Fe7Mo3 | |
| CoCrCu0.1Fe1.5Mo0.5Ni | 3.260 | 8.056 | 0.132 | −0.745 | 13.492 | 0.0069 | 1.2695 | FCC + Laves | |
| CoCrCu0.1Fe1.7Mo0.3Ni | 2.669 | 8.314 | 0.117 | −0.670 | 13.037 | 0.0045 | 1.8301 | FCC | |
| CoCrCu0.1FeMo0.8Ni1.2 | 3.933 | 8.016 | 0.150 | −0.923 | 13.853 | 0.0089 | 0.8956 | FCC + Laves + Fe7Mo3 | |
| CoCrCu0.1FeMo0.5Ni1.5 | 3.339 | 8.252 | 0.133 | −0.907 | 13.492 | 0.0070 | 1.2102 | FCC + Laves | |
| CoCrCu0.1FeMo0.3Ni1.7 | 2.768 | 8.408 | 0.119 | −0.869 | 13.037 | 0.0051 | 1.7016 | FCC | |
| Al0.1CoCrCu0.1FeMo0.9Ni | 4.35 | 7.80 | 0.159 | −1.116 | 14.45 | 0.0367 | 0.7636 | FCC + Laves | [35] |
| Al0.2CoCrCu0.1FeMo0.8Ni | 4.48 | 7.74 | 0.158 | −1.322 | 14.73 | 0.0405 | 0.7339 | FCC + BCC + Laves + σ | [35] |
| Al0.3CoCrCu0.1FeMo0.7Ni | 4.61 | 7.68 | 0.157 | −1.523 | 14.91 | 0.0417 | 0.7016 | FCC + BCC + Laves + σ | [35] |
| Al0.5CoCrCu0.1FeMo0.5Ni | 4.85 | 7.57 | 0.151 | −1.915 | 15.05 | 0.0449 | 0.6398 | FCC + BCC+ Laves + σ | [35] |
| Al0.8CoCrCu0.1FeMo0.2Ni | 5.18 | 7.39 | 0.136 | −2.474 | 14.73 | 0.0415 | 0.5490 | FCC + BCC + σ | [35] |
| AlCoCrCuFeNiMo0.2 | 5.08 | 7.77 | 0.133 | −4.47 | 15.6 | 0.0125 | 0.6045 | FCC + BCC | [16] |
| AlCoCrCuFeNiMo0.4 | 5.15 | 7.72 | 0.145 | −4.2 | 15.91 | 0.0257 | 0.5999 | BCC + α | [16] |
| AlCoCrCuFeNiMo0.6 | 5.21 | 7.67 | 0.154 | −3.95 | 16.08 | 0.0144 | 0.5924 | BCC + α | [16] |
| AlCoCrCuFeNiMo0.8 | 5.26 | 7.62 | 0.162 | −3.72 | 16.16 | 0.0131 | 0.5841 | BCC + α | [16] |
| AlCoCrCuFeNiMo | 5.29 | 7.57 | 0.168 | −3.51 | 16.18 | 0.0130 | 0.5782 | BCC +α | [16] |
| CoCrCu0.1Fe0.15NiMo1.5Mn0.05 | 4.70 | 7.64 | 0.185 | −4.61 | 13.14 | 0.0092 | 0.5948 | FCC + BCC + μ | [13] |
| CoCrCu0.1Fe0.15NiMo1.5Mn0.12 | 4.68 | 7.63 | 0.189 | −4.52 | 13.44 | 0.0091 | 0.6136 | FCC + BCC + μ | [13] |
| CoCrCu0.1Fe0.15NiMo1.5Mn0.3 | 4.64 | 7.60 | 0.197 | −4.29 | 13.90 | 0.0091 | 0.6456 | FCC + BCC + μ | [13] |
| CoCrCu0.1FeNiMo | 4.216 | 7.86 | 0.159 | −3.63 | 13.92 | 0.0097 | 0.7831 | FCC + BCC | [33] |
| CoCrCu0.3FeNiMo | 4.181 | 7.98 | 0.157 | −1.83 | 14.44 | 0.0096 | 0.8260 | FCC + BCC | [33] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, N.; Chen, C.; Chang, I.; Zhou, P.; Wang, X. Compositional Dependence of Phase Selection in CoCrCu0.1FeMoNi-Based High-Entropy Alloys. Materials 2018, 11, 1290. https://doi.org/10.3390/ma11081290
Liu N, Chen C, Chang I, Zhou P, Wang X. Compositional Dependence of Phase Selection in CoCrCu0.1FeMoNi-Based High-Entropy Alloys. Materials. 2018; 11(8):1290. https://doi.org/10.3390/ma11081290
Chicago/Turabian StyleLiu, Ning, Chen Chen, Isaac Chang, Pengjie Zhou, and Xiaojing Wang. 2018. "Compositional Dependence of Phase Selection in CoCrCu0.1FeMoNi-Based High-Entropy Alloys" Materials 11, no. 8: 1290. https://doi.org/10.3390/ma11081290
APA StyleLiu, N., Chen, C., Chang, I., Zhou, P., & Wang, X. (2018). Compositional Dependence of Phase Selection in CoCrCu0.1FeMoNi-Based High-Entropy Alloys. Materials, 11(8), 1290. https://doi.org/10.3390/ma11081290






