An Evaluation of Wetting and Adhesion of Three Bioceramic Root Canal Sealers to Intraradicular Human Dentin
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Preparation
2.2. Measurements
2.3. Calculation of Wetting and Adhesion
2.4. Statistical Analysis
3. Results
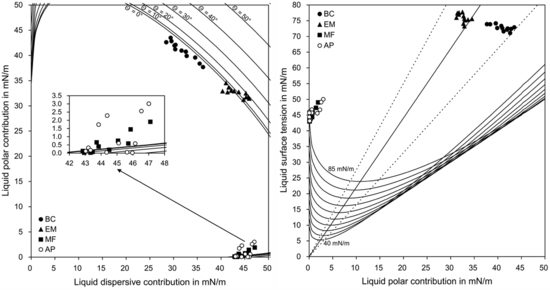
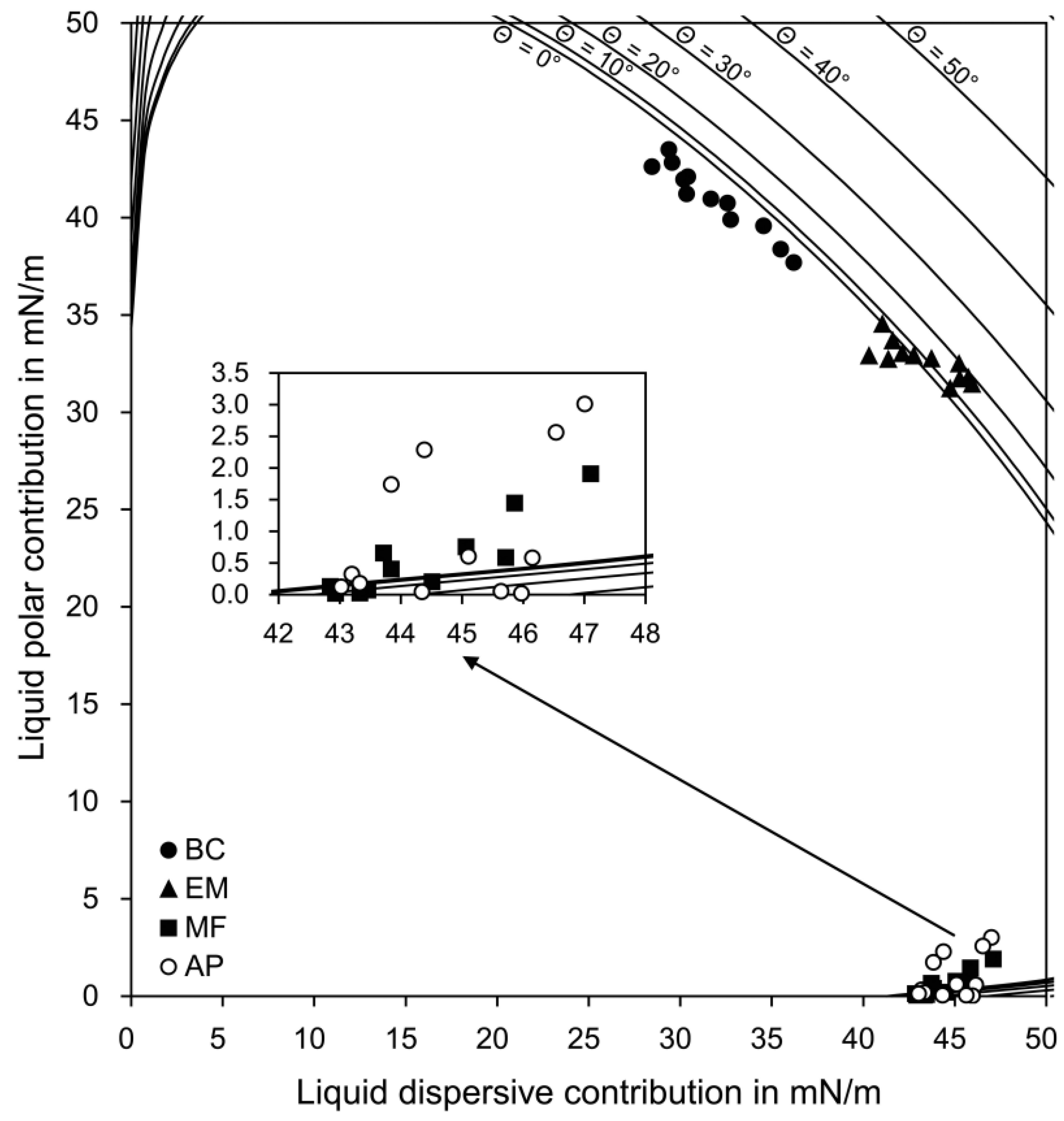
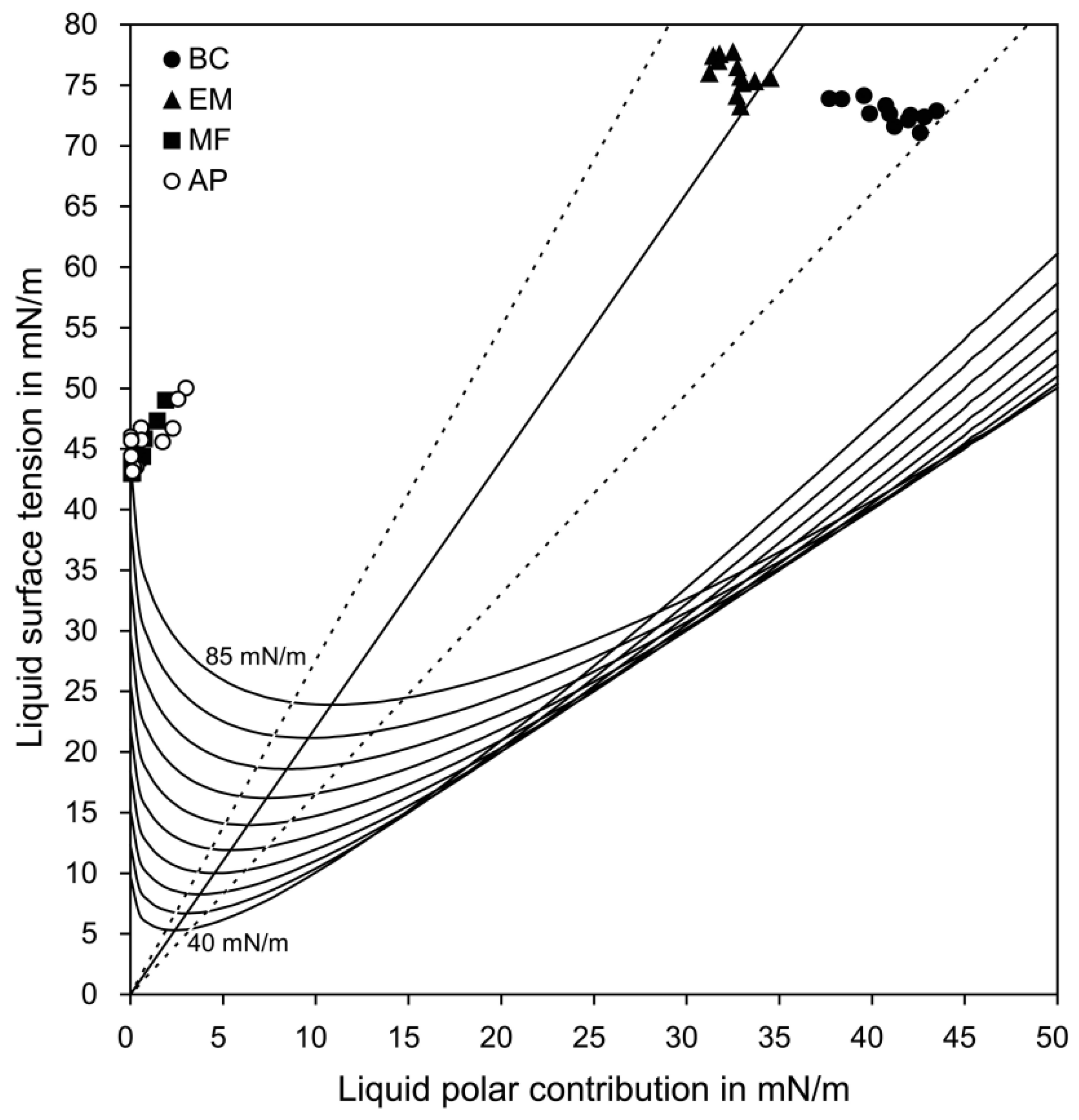
3.1. Surface Energy Parameters
3.2. Wetting and Adhesion
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Assmann, E.; Scarparo, R.K.; Bottcher, D.E.; Grecca, F.S. Dentin bond strength of two mineral trioxide aggregate-based and one epoxy resin-based sealers. J. Endod. 2012, 38, 219–221. [Google Scholar] [CrossRef] [PubMed]
- Dentistry-Root Canal Sealing Materials; ISO 6876:2012; International Organization for Standardization (ISO): Geneva, Switzerland, 2012.
- Cintra, L.T.A.; Benetti, F.; de Azevedo Queiroz, Í.O.; Ferreira, L.L.; Massunari, L.; Bueno, C.R.E.; de Oliveira, S.H.P.; Gomes-Filho, J.E. Evaluation of the cytotoxicity and biocompatibility of new resin epoxy-based endodontic sealer containing calcium hydroxide. J. Endod. 2017, 43, 2088–2092. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Lozano, F.J.; García-Bernal, D.; Oñate-Sánchez, R.E.; Ortolani-Seltenerich, P.S.; Forner, L.; Moraleda, J.M. Evaluation of cytocompatibility of calcium silicate-based endodontic sealers and their effects on the biological responses of mesenchymal dental stem cells. Int. Endod. J. 2017, 50, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Loushine, B.A.; Bryan, T.E.; Looney, S.W.; Gillen, B.M.; Loushine, R.J.; Weller, R.N.; Pashley, D.H.; Tay, F.R. Setting properties and cytotoxicity evaluation of a premixed bioceramic root canal sealer. J. Endod. 2011, 37, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Candeiro, G.T.; Correia, F.C.; Duarte, M.A.; Ribeiro-Siqueira, D.C.; Gavini, G. Evaluation of radiopacity, pH, release of calcium ions, and flow of a bioceramic root canal sealer. J. Endod. 2012, 38, 842–845. [Google Scholar] [CrossRef] [PubMed]
- Candeiro, G.T.M.; Moura-Netto, C.; D'Almeida-Couto, R.S.; Azambuja-Júnior, N.; Marques, M.M.; Cai, S.; Gavini, G. Cytotoxicity, genotoxicity and antibacterial effectiveness of a bioceramic endodontic sealer. Int. Endod. J. 2016, 49, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Kwak, S.W.; Ha, J.H.; Lee, W.; Kim, H.C. Physicochemical properties of epoxy resin-based and bioceramic-based root canal sealers. Bioinorg. Chem. Appl. 2017, 2017, 2582849. [Google Scholar] [CrossRef] [PubMed]
- Ballal, N.V.; Tweeny, A.; Khechen, K.; Prabhu, K.N.; Satyanarayan; Tay, F.R. Wettability of root canal sealers on intraradicular dentine treated with different irrigating solutions. J. Dent. 2013, 41, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Dogan Buzoglu, H.; Calt, S.; Gümüsderelioglu, M. Evaluation of the surface free energy on root canal dentine walls treated with chelating agents and NaOCl. Int. Endod. J. 2007, 40, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Al-Haddad, A.; Che Ab Aziz, Z.A. Bioceramic-Based Root Canal Sealers: A Review. Int. J. Biomater. 2016, 2016, 9753210. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.S. Adhesive dentistry and endodontics. Part 2: bonding in the root canal system-the promise and the problems: A review. J. Endod. 2006, 32, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Grégoire, G.; Dabsie, F.; Dieng-Sarr, F.; Akon, B.; Sharrock, P. Solvent composition of one-step self-etch adhesives and dentine wettability. J. Dent. 2011, 39, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Kim, Y.K.; Kim, K.H.; Kwon, T.Y. Shear bond strengths of various luting cements to zirconia ceramic: surface chemical aspects. J. Dent. 2011, 39, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.M.; Min, B.K.; Son, J.S.; Kim, K.H.; Kwon, T.Y. Durability of resin bond strength to dental noble metal–ceramic alloys conditioned with novel mercapto silane-based primer systems. J. Adhes. Sci. Technol. 2016, 30, 506–519. [Google Scholar] [CrossRef]
- Kim, H.J.; Bagheri, R.; Kim, Y.K.; Son, J.S.; Kwon, T.Y. Influence of curing mode on the surface energy and sorption/solubility of dental self-adhesive resin cements. Materials 2017, 10, 129. [Google Scholar] [CrossRef] [PubMed]
- Benetti, P.; Della Bona, A.; Kelly, J.R. Evaluation of thermal compatibility between core and veneer dental ceramics using shear bond strength test and contact angle measurement. Dent Mater. 2010, 26, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.H.; Kim, K.H.; Son, J.S.; Kwon, T.Y. Surface roughness effect on the solid equilibrium contact angle. J. Nanosci. Nanotechnol. 2017, 17, 4271–4274. [Google Scholar] [CrossRef]
- Owens, D.K.; Wendt, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Vivet, L.; Joudrier, A.L.; Bouttemy, M.; Vigneron, J.; Tan, K.L.; Morelle, J.M.; Etcheberry, A.; Chalumeau, L. Wettability and XPS analyses of nickel–phosphorus surfaces after plasma treatment: An efficient approach for surface qualification in mechatronic processes. Appl. Surf. Sci. 2013, 274, 71–78. [Google Scholar] [CrossRef]
- Zehnder, M. Root canal irrigants. J. Endod. 2006, 32, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Takimoto, M.; Ishii, R.; Iino, M.; Shimizu, Y.; Tsujimoto, A.; Takamizawa, T.; Ando, S.; Miyazaki, M. Influence of temporary cement contamination on the surface free energy and dentine bond strength of self-adhesive cements. J Dent. 2012, 40, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Son, J.S.; Kim, K.H.; Kwon, T.Y. Influence of surface energy parameters of dental self-adhesive resin cements on bond strength to dentin. J. Adhes. Sci. Technol. 2013, 27, 1778–1789. [Google Scholar] [CrossRef]
- Waters, M.G.; Williams, D.W.; Jagger, R.G.; Lewis, M.A. Adherence of Candida albicans to experimental denture soft lining materials. J. Prosthet. Dent. 1997, 77, 306–312. [Google Scholar] [CrossRef]
- Ström, G.; Fredriksson, M.; Stenius, P. Contact angles, work of adhesion, and interfacial tensions at a dissolving hydrocarbon surface. J. Colloid Interface Sci. 1987, 119, 352–361. [Google Scholar] [CrossRef]
- Anderson, L.J.; Easton, C.D.; Jacob, M.V. Compatibility of plasma-deposited linalyl acetate thin films with organic electronic device fabrication techniques. J. Mater. Sci. 2013, 48, 4851–4859. [Google Scholar] [CrossRef]
- Ahmad, J.; Bazaka, K.; Oelgemöller, M.; Jacob, M.V. Wetting, solubility and chemical characteristics of plasma-polymerized 1-isopropyl-4-methyl-1,4-cyclohexadiene thin films. Coatings 2014, 4, 527–552. [Google Scholar] [CrossRef]
- Zhou, H.M.; Shen, Y.; Zheng, W.; Li, L.; Zheng, Y.F.; Haapasalo, M. Physical properties of 5 root canal sealers. J. Endod. 2013, 39, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Voigt, M.M.; Mackenzie, R.C.I.; King, S.P.; Yau, C.P.; Atienzar, P.; Dane, J.; Keivanidis, P.E.; Zadrazil, I.; Bradley, D.D.C.; Nelson, J. Gravure printing inverted organic solar cells: The influence of ink properties on film quality and device performance. Sol. Energy Mater. Sol. Cells 2012, 105, 77–85. [Google Scholar] [CrossRef]
- Breschi, L.; Mazzoni, A.; Dorigo, E.D.S.; Ferrari, M. Adhesion to intraradicular dentin: A review. J. Adhes. Sci. Technol. 2009, 23, 1053–1083. [Google Scholar] [CrossRef]
- Kim, Y.K.; Min, B.K.; Son, J.S.; Kim, K.H.; Kwon, T.Y. Influence of different drying methods on microtensile bond strength of self-adhesive resin cements to dentin. Acta Odontol. Scand. 2014, 72, 954–962. [Google Scholar] [CrossRef] [PubMed]
- Samuel, B.; Zhao, H.; Law, K.Y. Study of wetting and adhesion interactions between water and various polymer and superhydrophobic surfaces. J. Phys. Chem. C 2011, 115, 14852–14861. [Google Scholar] [CrossRef]

| Brand Name (Code) | Manufacturer | Composition | Batch Number |
|---|---|---|---|
| EndoSequence BC Sealer (BC) | Brasseler USA, Savannah, GA, USA | Zirconium oxide, calcium silicates, calcium phosphate monobasic, calcium hydroxide, filler, and thickening agents | 14004SP |
| Endoseal MTA (EM) | Maruchi, Wonju, Korea | Calcium silicates, calcium aluminates, calcium aluminoferrite, calcium sulfates, radiopacifier, and thickening agents | SEF670601 |
| MTA Fillapex (MF) | Angelus, Londrina, Brazil | Paste A: salicylate resin, bismuth trioxide, fumed silica; Paste B: fumed silica, titanium dioxide, mineral trioxide aggregate, and base resin | 35088 |
| AH Plus (AP) | Dentsply DeTrey GmbH, Konstanz, Germany | Epoxide paste: diepoxide, calcium tungstate, zirconium oxide, aerosol, pigment; Amine paste: 1-adamantane amine, N,N′-dibenzyl-5-oxa-nonandiamin-1,9, TCD-diamine, calcium tongstate, zirconium oxide, aerosil, and silicon oil | 1605000894 |
| Groups | σ (Total Surface Tension) | σd (Dispersive Component) | σp (Polar Component) | Polarity (%) |
|---|---|---|---|---|
| BC | 72.75 (0.93) a | 31.80 (2.54) a | 40.95 (1.80) a | 56.32 (3.00) a |
| EM | 75.91 (1.39) b | 43.32 (2.03) b | 32.59 (0.95) b | 42.96 (1.77) b |
| MF | 44.80 (1.94) c | 44.28 (1.38) b | 0.52 (0.61) c | 1.11 (1.25) c |
| AP | 45.84 (2.13) c | 44.88 (1.38) b | 0.96 (1.12) c | 2.01 (2.28) c |
| Groups | Number of Each Data Point (σp(σd)) Lying beneath Each Curve on the Wetting Envelope | Perpendicular Distance between Each Data Point (σ(σp)) and the Straight Line on the Isogram Diagram (mN/m) | ||||
|---|---|---|---|---|---|---|
| Θ = 0° | Θ = 10° | Θ = 20° | Θ = 30° | Θ = 40° | ||
| BC | 12 | 0 | 0 | 0 | 0 | 7.21 (1.92) a |
| EM | 5 | 2 | 5 | 0 | 0 | 1.74 (1.20) b |
| MF | 6 | 1 | 2 | 3 | 0 | 18.05 (0.33) c |
| AP | 7 | 1 | 1 | 1 | 2 | 18.08 (0.59) c |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, J.-H.; Kim, H.-C.; Kim, Y.K.; Kwon, T.-Y. An Evaluation of Wetting and Adhesion of Three Bioceramic Root Canal Sealers to Intraradicular Human Dentin. Materials 2018, 11, 1286. https://doi.org/10.3390/ma11081286
Ha J-H, Kim H-C, Kim YK, Kwon T-Y. An Evaluation of Wetting and Adhesion of Three Bioceramic Root Canal Sealers to Intraradicular Human Dentin. Materials. 2018; 11(8):1286. https://doi.org/10.3390/ma11081286
Chicago/Turabian StyleHa, Jung-Hong, Hyeon-Cheol Kim, Young Kyung Kim, and Tae-Yub Kwon. 2018. "An Evaluation of Wetting and Adhesion of Three Bioceramic Root Canal Sealers to Intraradicular Human Dentin" Materials 11, no. 8: 1286. https://doi.org/10.3390/ma11081286
APA StyleHa, J.-H., Kim, H.-C., Kim, Y. K., & Kwon, T.-Y. (2018). An Evaluation of Wetting and Adhesion of Three Bioceramic Root Canal Sealers to Intraradicular Human Dentin. Materials, 11(8), 1286. https://doi.org/10.3390/ma11081286