Effect of CNT/PDMS Nanocomposites on the Dynamics of Pioneer Bacterial Communities in the Natural Biofilms of Seawater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. The Primer Coat
2.1.2. Silicone-Based Matrix System
2.1.3. Carbon Nanotubes (CNTs)
2.1.4. Preparation of the PDMS-Based Composites (PCs)
2.2. Panel Preparation
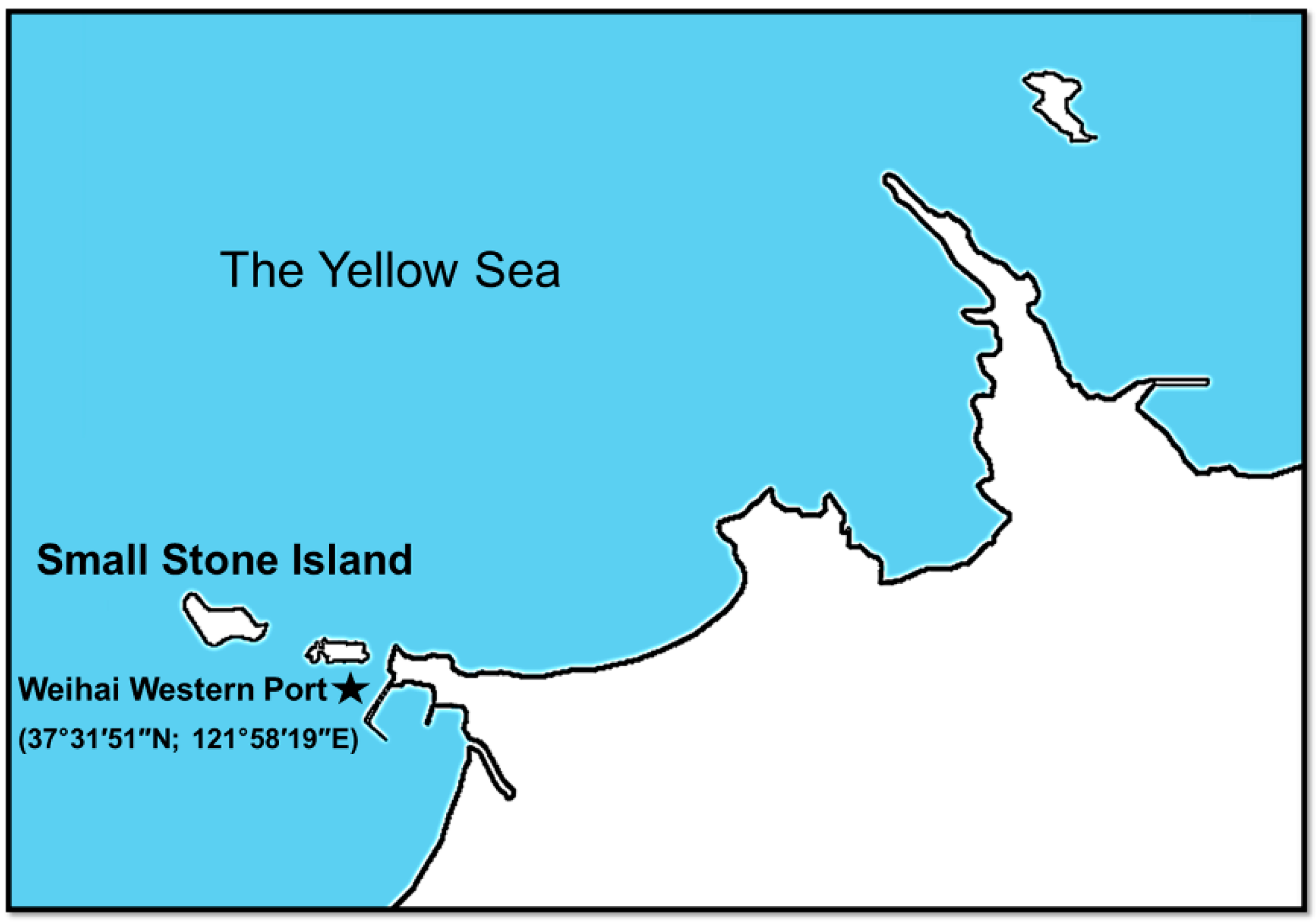
2.3. Seawater Exposure Assays
2.4. Sampling
2.5. SSCP
2.6. Data Analysis
3. Results and Discussion
3.1. Fouling and Surface Evaluation
3.2. SSCP Patterns of the Bacterial Biofilm Communities
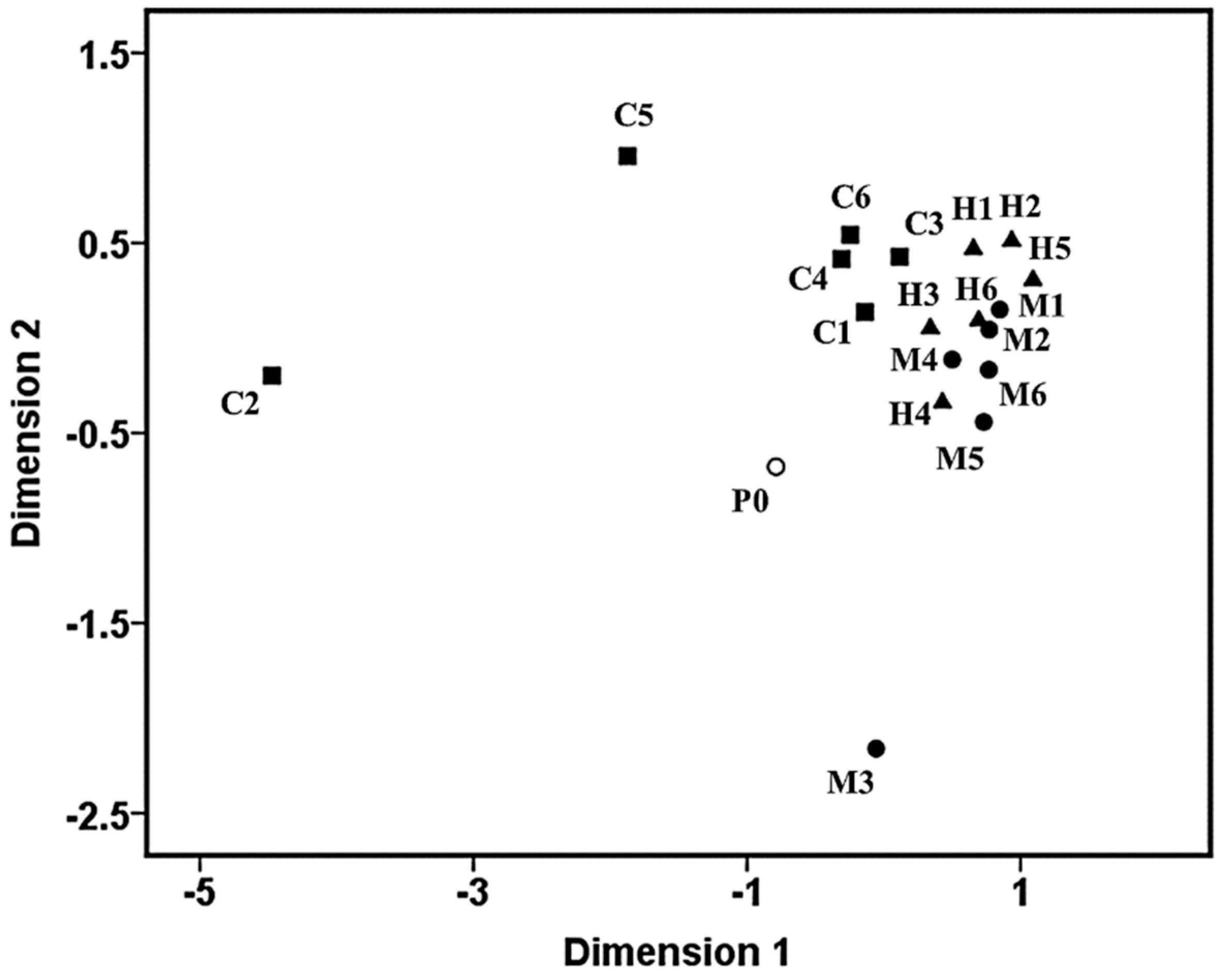
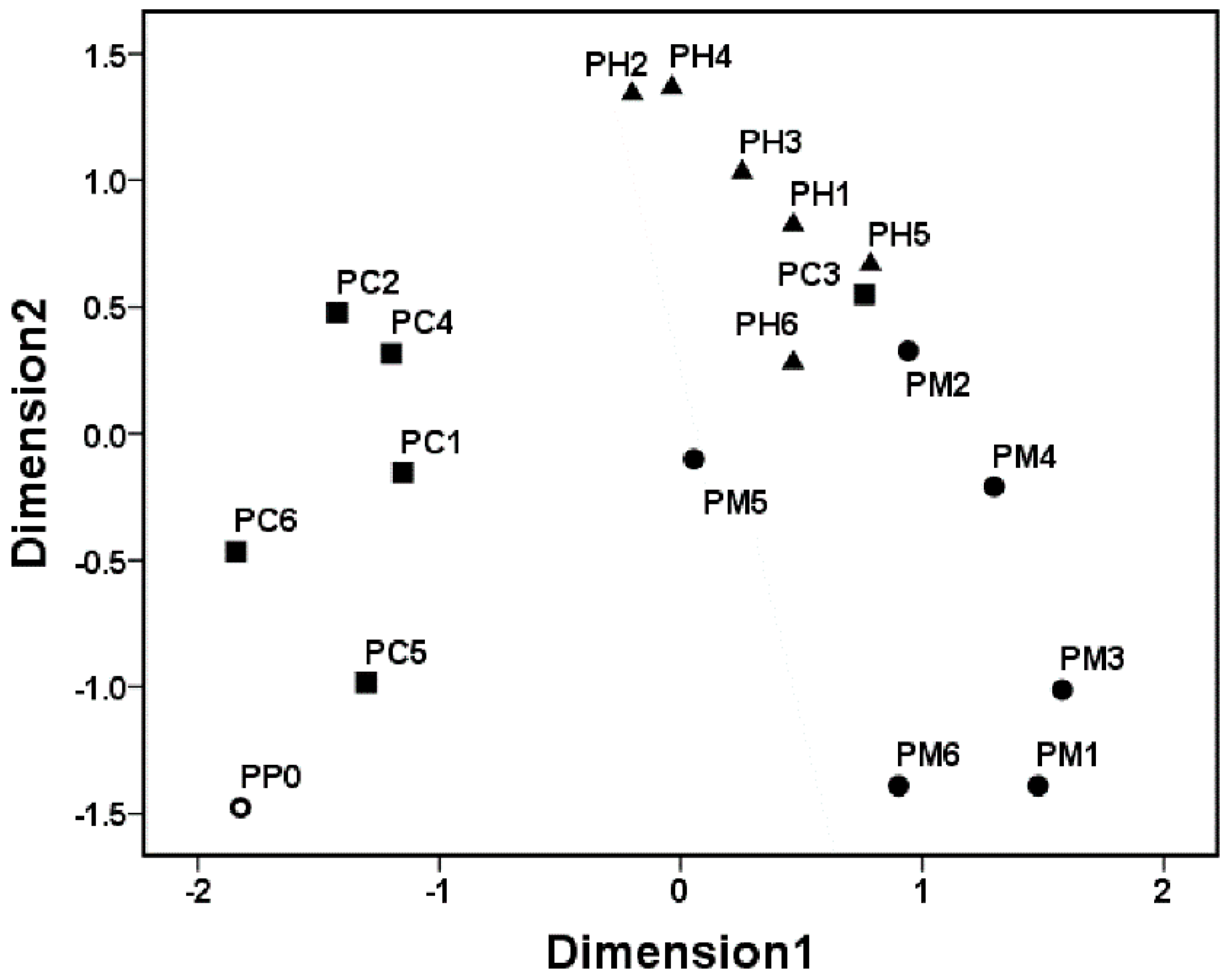
3.3. Clustering Patterns of the Pioneer Bacterial Communities
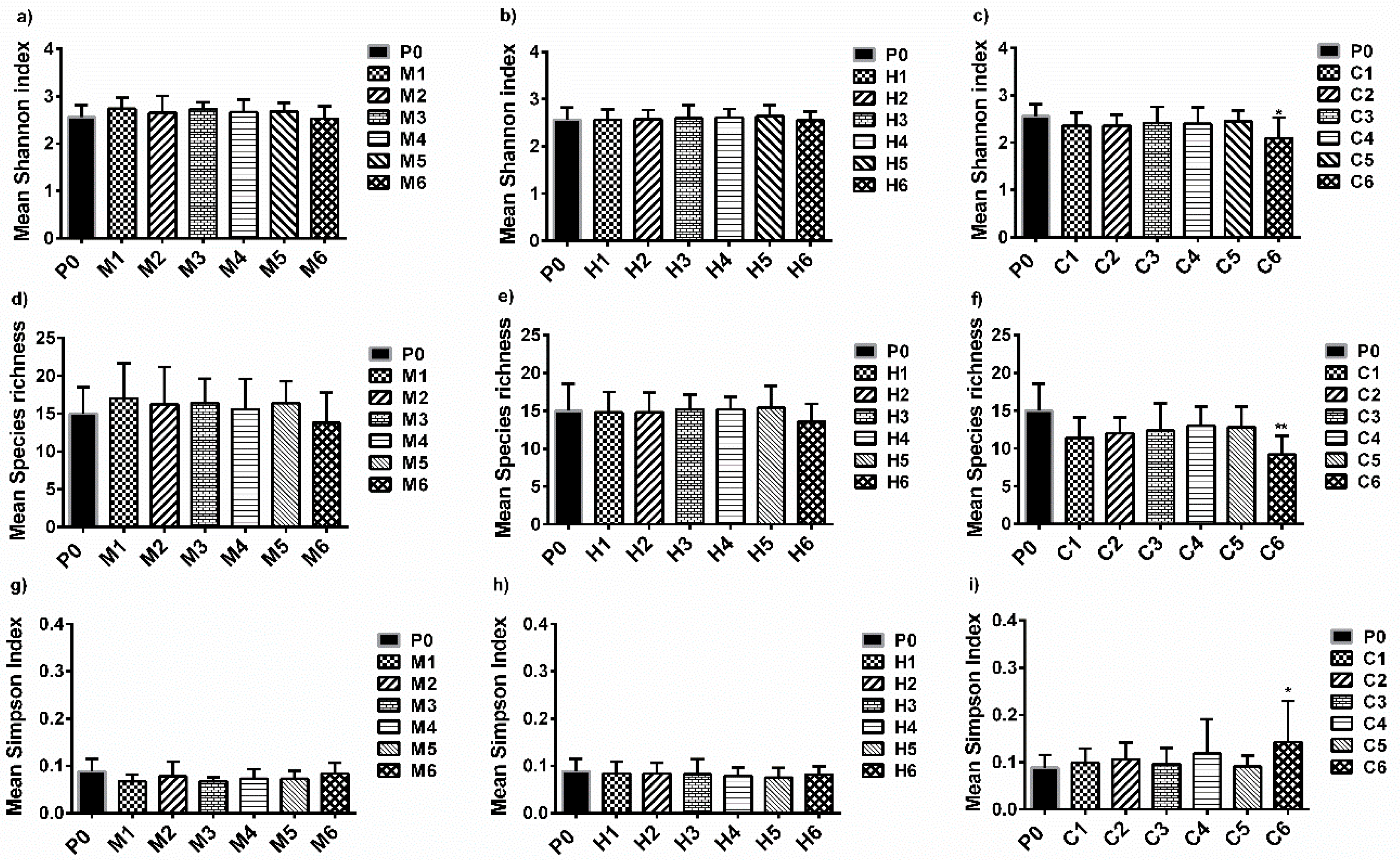
3.4. Analysis of Pioneer Bacterial Communities in the Natural Biofilms
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fitridge, I.; Dempster, T.; Guenther, J.; de Nys, R. The impact and control of biofouling in marine aquaculture: A review. Biofouling 2012, 28, 649–669. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.; Bendick, J.; Holm, E.; Hertel, W. Economic impact of biofouling on a naval surface ship. Biofouling 2011, 27, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Salta, M.; Wharton, J.A.; Blache, Y.; Stokes, K.R.; Briand, J.F. Marine biofilms on artificial surfaces: Structure and dynamics. Environ. Microbiol. 2013, 15, 2879–2893. [Google Scholar] [CrossRef] [PubMed]
- Hoiby, N. A short history of microbial biofilms and biofilm infections. APMIS 2017, 125, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B. Approaches to prevention, removal and killing of biofilms. Int. Biodeterior. Biodegrad. 2003, 51, 249–253. [Google Scholar] [CrossRef]
- Dang, H.Y.; Lovell, C.R. Microbial surface colonization and biofilm development in marine environments. Microbiol. Mol. Biol. Rev. 2016, 80, 91–138. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Jia, R.; Li, Y.; Gu, T. Advances in the treatment of problematic industrial biofilms. World J. Microbiol. Biotechnol. 2017, 33, 97. [Google Scholar] [CrossRef] [PubMed]
- Riga, E.K.; Vohringer, M.; Widyaya, V.T.; Lienkamp, K. Polymer-based surfaces designed to reduce biofilm formation: From antimicrobial polymers to strategies for long-term applications. Macromol. Rapid Commun. 2017, 38, 1700216. [Google Scholar] [CrossRef] [PubMed]
- Gule, N.P.; Begum, N.M.; Klumperman, B. Advances in biofouling mitigation: A review. Crit. Rev. Environ. Sci. Technol. 2016, 46, 535–555. [Google Scholar] [CrossRef]
- Selim, M.S.; Shenashen, M.A.; El-Safty, S.A.; Higazy, S.A.; Selim, M.M.; Isago, H.; Elmarakbi, A. Recent progress in marine foul-release polymeric nanocomposite coatings. Prog. Mater. Sci. 2017, 87, 1–32. [Google Scholar] [CrossRef]
- Nir, S.; Reches, M. Bio-inspired antifouling approaches: The quest towards non-toxic and non-biocidal materials. Curr. Opin. Biotechnol. 2016, 39, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Stupak, M.E.; García, M.T.; Pérez, M.C. Non-toxic alternative compounds for marine antifouling paints. Int. Biodeterior. Biodegrad. 2003, 52, 49–52. [Google Scholar] [CrossRef]
- Lejars, M.; Margaillan, A.; Bressy, C. Fouling release coatings: A nontoxic alternative to biocidal antifouling coatings. Chem. Rev. 2012, 112, 4347–4390. [Google Scholar] [CrossRef] [PubMed]
- Eduok, U.; Faye, O.; Szpunar, J. Recent developments and applications of protective silicone coatings: A review of PDMS functional materials. Prog. Org. Coat. 2017, 111, 124–163. [Google Scholar] [CrossRef]
- Zhang, H.B.; Chiao, M. Anti-fouling coatings of poly(dimethylsiloxane) devices for biological and biomedical applications. J. Med. Biol. Eng. 2015, 35, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Eduok, U.; Suleiman, R.; Gittens, J.; Khaled, M.; Smith, T.J.; Akid, R.; El Ali, B.; Khalil, A. Anticorrosion/antifouling properties of bacterial spore-loaded sol-gel type coating for mild steel in saline marine condition: A case of thermophilic strain of bacillus licheniformis. RSC Adv. 2015, 5, 93818–93830. [Google Scholar] [CrossRef]
- Eduok, U.; Khaled, M.; Khalil, A.; Suleiman, R.; El Ali, B. Probing the corrosion inhibiting role of a thermophilic bacillus licheniformis biofilm on steel in a saline axenic culture. RSC Adv. 2016, 6, 18246–18256. [Google Scholar] [CrossRef]
- Kochkodan, V.; Hilal, N. A comprehensive review on surface modified polymer membranes for biofouling mitigation. Desalination 2015, 356, 187–207. [Google Scholar] [CrossRef]
- Jhaveri, J.H.; Murthy, Z.V.P. Nanocomposite membranes. Desalin. Water Treat. 2016, 57, 26803–26819. [Google Scholar] [CrossRef]
- Lyszcz, M.; Galazka, A. Genetic differentiation methods of microorganisms in the soil—Plant system. Postepy Mikrobiol. 2017, 56, 341–352. [Google Scholar]
- Beigbeder, A.; Mincheva, R.; Pettitt, M.E.; Callow, M.E.; Callow, J.A.; Claes, M.; Dubois, P. Marine fouling release silicone/carbon nanotube nanocomposite coatings: On the importance of the nanotube dispersion state. J. Nanosci. Nanotechnol. 2010, 10, 2972–2978. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhang, Z. New anti-biofouling carbon nanotubes-filled polydimethylsiloxane composites against colonization by pioneer eukaryotic microbes. Int. Biodeterior. Biodegrad. 2016, 110, 147–154. [Google Scholar] [CrossRef]
- Cavas, L.; Yildiz, P.G.; Mimigianni, P.; Sapalidis, A.; Nitodas, S. Reinforcement effects of multiwall carbon nanotubes and graphene oxide on PDMS marine coatings. J. Coat. Technol. Res. 2018, 15, 105–120. [Google Scholar] [CrossRef]
- Mahdavi, M.R.; Delnavaz, M.; Vatanpour, V.; Farahbakhsh, J. Effect of blending polypyrrole coated multi-walled carbon nanotube on desalination performance and antifouling property of thin film nanocomposite nanofiltration membranes. Sep. Purif. Technol. 2017, 184, 119–127. [Google Scholar] [CrossRef]
- Yang, J.-L.; Shen, P.-J.; Liang, X.; Li, Y.-F.; Bao, W.-Y.; Li, J.-L. Larval settlement and metamorphosis of the mussel Mytilus coruscus in response to monospecific bacterial biofilms. Biofouling 2013, 29, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Oliva, M.; Martinelli, E.; Galli, G.; Pretti, C. PDMS-based films containing surface-active amphiphilic block copolymers to combat fouling from barnacles B. Amphitrite and B. Improvisus. Polymer 2017, 108, 476–482. [Google Scholar] [CrossRef]
- Beigbeder, A.; Labruyere, C.; Viville, P.; Pettitt, M.E.; Callow, M.E.; Callow, J.A.; Bonnaud, L.; Lazzaroni, R.; Dubois, P. Surface and fouling-release properties of silicone/organomodified montmorillonite coatings. J. Adhes. Sci. Technol. 2011, 25, 1689–1700. [Google Scholar] [CrossRef]
- Sathya, S.; Murthy, P.S.; Das, A.; Sankar, G.G.; Venkatnarayanan, S.; Pandian, R.; Sathyaseelan, V.S.; Pandiyan, V.; Doble, M.; Venugopalan, V.P. Marine antifouling property of PMMA nanocomposite films: Results of laboratory and field assessment. Int. Biodeterior. Biodegrad. 2016, 114, 57–66. [Google Scholar] [CrossRef]
- Noguer, A.C.; Olsen, S.M.; Hvilsted, S.; Kiil, S. Field study of the long-term release of block copolymers from fouling-release coatings. Prog. Org. Coat. 2017, 112, 101–108. [Google Scholar] [CrossRef]
- Eichner, C.A.; Erb, R.W.; Timmis, K.N.; Wagner-Dobler, I. Thermal gradient gel electrophoresis analysis of bioprotection from pollutant shocks in the activated sludge microbial community. Appl. Environ. Microbiol. 1999, 65, 102–109. [Google Scholar] [PubMed]
- Wiens, J.J.; Donoghue, M.J. Historical biogeography, ecology and species richness. Trends Ecol. Evol. 2004, 19, 639–644. [Google Scholar] [CrossRef] [PubMed]
- He, F.L.; Hu, X.S. Hubbell’s fundamental biodiversity parameter and the simpson diversity index. Ecol. Lett. 2005, 8, 386–390. [Google Scholar] [CrossRef]
- Camps, M.; Barani, A.; Gregori, G.; Bouchez, A.; Le Berre, B.; Bressy, C.; Blache, Y.; Briand, J.F. Antifouling coatings influence both abundance and community structure of colonizing biofilms: A case study in the northwestern mediterranean sea. Appl. Environ. Microbiol. 2014, 80, 4821–4831. [Google Scholar] [CrossRef] [PubMed]
- Casse, F.; Swain, G.W. The development of microfouling on four commercial antifouling coatings under static and dynamic immersion. Int. Biodeterior. Biodegrad. 2006, 57, 179–185. [Google Scholar] [CrossRef]
- Briand, J.F.; Djeridi, I.; Jamet, D.; Coupe, S.; Bressy, C.; Molmeret, M.; Le Berre, B.; Rimet, F.; Bouchez, A.; Blache, Y. Pioneer marine biofilms on artificial surfaces including antifouling coatings immersed in two contrasting french mediterranean coast sites. Biofouling 2012, 28, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.L.; Maki, J.S.; Rittschof, D.; Teo, S.L.M. Early marine bacterial biofilm on a copper-based antifouling paint. Int. Biodeterior. Biodegrad. 2013, 83, 71–76. [Google Scholar] [CrossRef]
- Yang, J.L.; Li, Y.F.; Guo, X.P.; Liang, X.; Xu, Y.F.; Ding, D.W.; Bao, W.Y.; Dobretsov, S. The effect of carbon nanotubes and titanium dioxide incorporated in PDMS on biofilm community composition and subsequent mussel plantigrade settlement. Biofouling 2016, 32, 763–777. [Google Scholar] [CrossRef] [PubMed]
- Ling, G.C.; Low, M.H.; Erken, M.; Longford, S.; Nielsen, S.; Poole, A.J.; Steinberg, P.; McDougald, D.; Kjelleberg, S. Micro-fabricated polydimethyl siloxane (PDMS) surfaces regulate the development of marine microbial biofilm communities. Biofouling 2014, 30, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.-D. Microbiological deterioration and degradation of synthetic polymeric materials: Recent research advances. Int. Biodeterior. Biodegrad. 2003, 52, 69–91. [Google Scholar] [CrossRef]


| CNTs | Hydroxyl Content % (w/w) | Carboxyl Content % (w/w) | Diameter (nm) | Length (μm) | SSA (m2/g) | PC Sets | PC Names |
|---|---|---|---|---|---|---|---|
| F1 | _ | _ | 10–20 | 30–100 | >165 | M | M1 |
| F2 | _ | _ | 8–15 | ~50 | >233 | M | M2 |
| F3 | _ | _ | 10–20 | 10–30 | >200 | M | M3 |
| F4 | _ | _ | 20–30 | 10–30 | >110 | M | M4 |
| F5 | _ | _ | 30–50 | 10–20 | >60 | M | M5 |
| F6 | _ | _ | >50 | 10–20 | >40 | M | M6 |
| F7 | 5.58 | _ | <8 | 10–30 | >500 | H | H1 |
| F8 | 3.70 | _ | 8–15 | ~50 | >233 | H | H2 |
| F9 | 3.06 | _ | 10–20 | 10–30 | >200 | H | H3 |
| F10 | 1.76 | _ | 20–30 | ~30 | >110 | H | H4 |
| F11 | 1.06 | _ | 30–50 | ~20 | >60 | H | H5 |
| F12 | 0.71 | _ | >50 | ~20 | >40 | H | H6 |
| F13 | _ | 3.86 | <8 | ~30 | >500 | C | C1 |
| F14 | _ | 2.56 | 8–15 | ~50 | >233 | C | C2 |
| F15 | _ | 2.00 | 10–20 | 10–30 | >200 | C | C3 |
| F18 | _ | 1.23 | 20–30 | ~30 | >110 | C | C4 |
| F17 | _ | 0.73 | 30–50 | ~20 | >60 | C | C5 |
| F18 | _ | 0.64 | >50 | ~20 | >40 | C | C6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, Y.; Sun, Y.; Lang, Y.; Wang, L.; Liu, B.; Zhang, Z. Effect of CNT/PDMS Nanocomposites on the Dynamics of Pioneer Bacterial Communities in the Natural Biofilms of Seawater. Materials 2018, 11, 902. https://doi.org/10.3390/ma11060902
Ji Y, Sun Y, Lang Y, Wang L, Liu B, Zhang Z. Effect of CNT/PDMS Nanocomposites on the Dynamics of Pioneer Bacterial Communities in the Natural Biofilms of Seawater. Materials. 2018; 11(6):902. https://doi.org/10.3390/ma11060902
Chicago/Turabian StyleJi, Yubin, Yuan Sun, Yanhe Lang, Lei Wang, Bing Liu, and Zhizhou Zhang. 2018. "Effect of CNT/PDMS Nanocomposites on the Dynamics of Pioneer Bacterial Communities in the Natural Biofilms of Seawater" Materials 11, no. 6: 902. https://doi.org/10.3390/ma11060902
APA StyleJi, Y., Sun, Y., Lang, Y., Wang, L., Liu, B., & Zhang, Z. (2018). Effect of CNT/PDMS Nanocomposites on the Dynamics of Pioneer Bacterial Communities in the Natural Biofilms of Seawater. Materials, 11(6), 902. https://doi.org/10.3390/ma11060902






