Doxorubicin Release Controlled by Induced Phase Separation and Use of a Co-Solvent
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Drug-Loaded Fiber Webs
2.3. Characterization
2.4. Solubility Parameter
2.5. Phase Separation
2.6. Loading Content (LC) and Loading Efficiency (LE)
2.7. In-Vitro Drug Release Analysis
3. Results
3.1. Drug Localization in the Drug-Carrier
3.2. Phase Separation Between PLA and PVP
3.3. Morphological Influence of Fibers Before and After Immersion in Water
3.4. Wettability of Drug-Loaded Webs
3.5. Loading Contents (LC) and Loading Efficiency (LE)
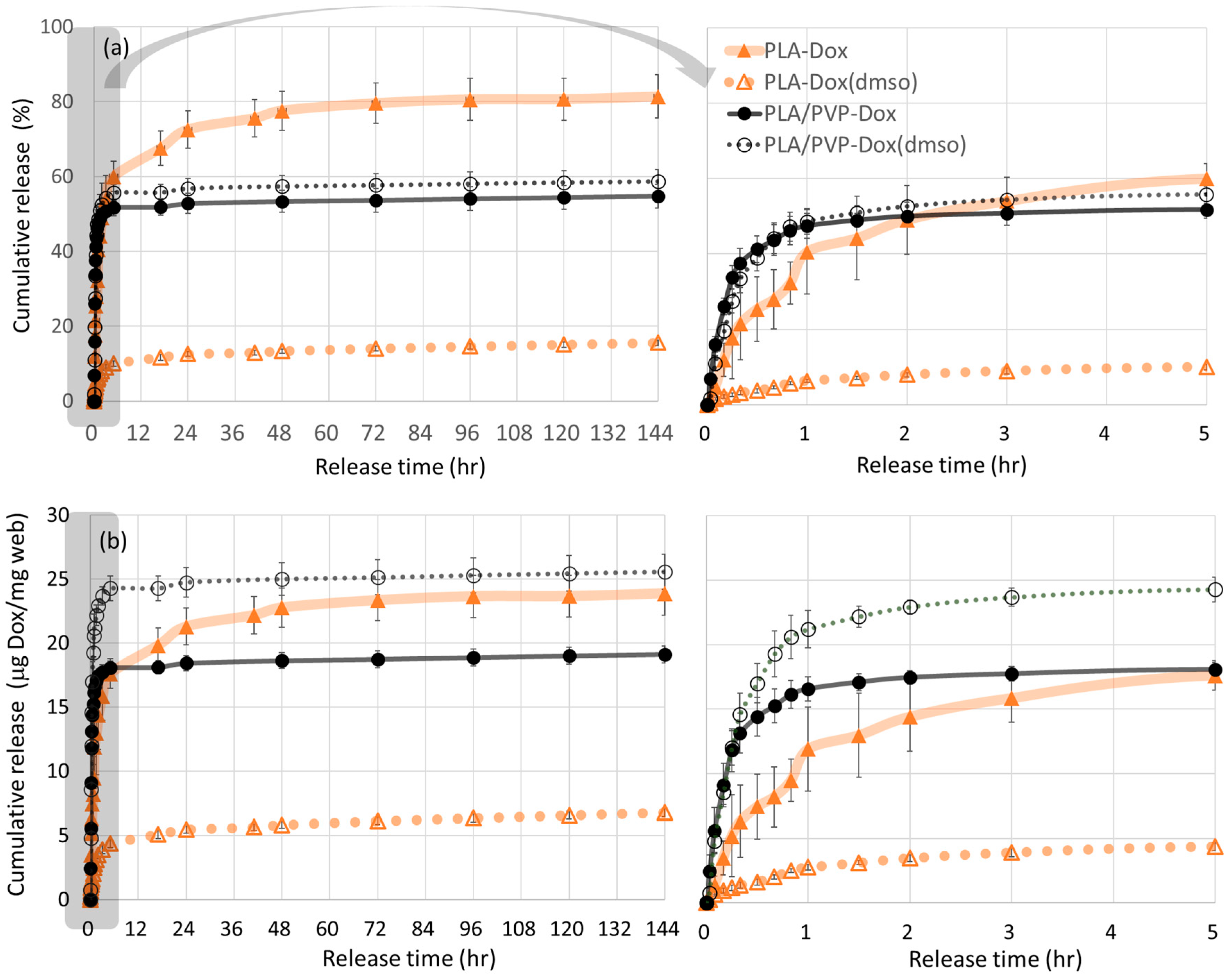
3.6. In-Vitro Dox Release
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Di, J.; Zhao, Y.; Yu, J. Fabrication of molecular sieve fibers by electrospinning. J. Mater. Chem. 2011, 21, 8511–8520. [Google Scholar] [CrossRef]
- Kim, J.S.; Reneker, D.H. Polybenzimidazole nanofiber produced by electrospinning. Polym. Eng. Sci. 1999, 39, 849–854. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y.N. Direct fabrication of composite and ceramic hollow nanofibers by electrospinning. Nano Lett. 2004, 4, 933–938. [Google Scholar] [CrossRef]
- Ji, S.; Li, Y.; Yang, M. Gas sensing properties of a composite composed of electrospun poly(methyl methacrylate) nanofibers and in situ polymerized polyaniline. Sens. Actuators B: Chem. 2008, 133, 644–649. [Google Scholar] [CrossRef]
- Truong, Y.B.; O’Bryan, Y.; McKelvie, I.D.; Kyratzis, I.L.; Humphries, W. Application of Electrospun Gas Diffusion Nanofibre-membranes in the Determination of Dissolved Carbon Dioxide. Macromol. Mater. Eng. 2013, 298, 590–596. [Google Scholar] [CrossRef]
- Saha, K.; Butola, B.S.; Joshi, M. Drug-Loaded Polyurethane/Clay Nanocomposite Nanofibers for Topical Drug- Delivery Application. J. Appl. Polym. Sci. 2014, 131, 40230. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, Y.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Composit. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Electrospinning of collagen nanofibers. Biomacromolecules 2002, 3, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Li, W.J.; Laurencin, C.T.; Caterson, E.J.; Tuan, R.S.; Ko, F.K. Electrospun nanofibrous structure: A novel scaffold for tissue engineering. J. Biomed. Mater. Res. 2002, 60, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Sill, T.J.; von Recum, H.A. Electro spinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef] [PubMed]
- Becker, T.; Kipke, D.; Brandon, T. Calcium alginate gel: A biocompatible and mechanically stable polymer for endovascular embolization. J. Biomed. Mater. Res. 2001, 54, 76–86. [Google Scholar] [CrossRef]
- Orive, G.; Ponce, S.; Hernandez, R.; Gascon, A.; Igartua, M.; Pedraz, J. Biocompatibility of microcapsules for cell immobilization elaborated with different type of alginates. Biomaterials 2002, 23, 3825–3831. [Google Scholar] [CrossRef]
- Dhandayuthapani, B.; Krishnan, U.M.; Sethuraman, S. Fabrication and characterization of chitosan-gelatin blend nanofibers for skin tissue engineering. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2010, 94, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Chi, C.; Chen, Y.; Chen, K.; Chen, P.; Yao, C. Evaluation of proanthocyanidin-crosslinked electrospun gelatin nanofibers for drug delivering system. Mater. Sci. Eng. C: Mater. Biol. Appl. 2012, 32, 2476–2483. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, Y.; Ramakrishna, S.; Lim, C. Electrospinning and mechanical characterization of gelatin nanofibers. Polymer 2004, 45, 5361–5368. [Google Scholar] [CrossRef]
- Mo, X.; Xu, C.; Kotaki, M.; Ramakrishna, S. Electrospun P(LLA-CL) nanofiber: A biomimetic extracellular matrix for smooth muscle cell and endothelial cell proliferation. Biomaterials 2004, 25, 1883–1890. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Murugan, R.; Ramakrishna, S.; Wang, X.; Ma, Y.; Wang, S. Fabrication of nano-structured porous PLLA scaffold intended for nerve tissue engineering. Biomaterials 2004, 25, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Sebe, I.; Szabo, P.; Kallai-Szabo, B.; Zelko, R. Incorporating small molecules or biologics into nanofibers for optimized drug release: A review. Int. J. Pharm. 2015, 494, 516–530. [Google Scholar] [CrossRef] [PubMed]
- Bashur, C.A.; Dahlgren, L.A.; Goldstein, A.S. Effect of fiber diameter and orientation on fibroblast morphology and proliferation on electrospun poly(D,L-lactic-co-glycolic acid) meshes. Biomaterials 2006, 27, 5681–5688. [Google Scholar] [CrossRef] [PubMed]
- Gentsch, R.; Pippig, F.; Schmidt, S.; Cernoch, P.; Polleux, J.; Boerner, H.G. Single-Step Electrospinning to Bioactive Polymer Nanofibers. Macromolecules 2011, 44, 453–461. [Google Scholar] [CrossRef]
- Liao, S.; Murugan, R.; Chan, C.K.; Ramakrishna, S. Processing nanoengineered scaffolds through electrospinning and mineralization, suitable for biomimetic bone tissue engineering. J. Mech. Behav. Biomed. Mater. 2008, 1, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tan, X.; Luo, T.; Shi, Y.; Yang, Y.; Liu, L. Preparation and characterization of electrospun PLA/PU bilayer nanofibrous membranes for controlled drug release applications. Integr. Ferroelectr. 2017, 179, 104–119. [Google Scholar] [CrossRef]
- Lv, J.; Yin, X.; Zeng, Q.; Dong, W.; Liu, H.; Zhu, L. Preparation of carboxymethyl chitosan nanofibers through electrospinning the ball-milled nanopowders with poly (lactic acid) and the blood compatibility of the electrospun NCMC/PLA mats. J. Polym. Res. 2017, 24, 60. [Google Scholar] [CrossRef]
- Zong, X.; Kim, K.; Fang, D.; Ran, S.; Hsiao, B.; Chu, B. Structure and process relationship of electrospun bioabsorbable nanofiber membranes. Polymer 2002, 43, 4403–4412. [Google Scholar] [CrossRef]
- Kenawy, E.; Bowlin, G.; Mansfield, K.; Layman, J.; Simpson, D.; Sanders, E.; Wnek, G. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Controll. Release 2002, 81, 57–64. [Google Scholar] [CrossRef]
- Zeng, J.; Chen, X.; Xu, X.; Liang, Q.; Bian, X.; Yang, L.; Jing, X. Ultrafine fibers electrospun from biodegradable polymers. J. Appl. Polym. Sci. 2003, 89, 1085–1092. [Google Scholar] [CrossRef]
- Zeng, J.; Aigner, A.; Czubayko, F.; Kissel, T.; Wendorff, J.; Greiner, A. Poly(vinyl alcohol) nanofibers by electrospinning as a protein delivery system and the retardation of enzyme release by additional polymer coatings. Biomacromolecules 2005, 6, 1484–1488. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Luu, Y.; Chang, C.; Fang, D.; Hsiao, B.; Chu, B.; Hadjiargyrou, M. Incorporation and controlled release of a hydrophilic antibiotic using poly(lactide-co-glycolide)-based electrospun nanofibrous scaffolds. J. Controll. Release 2004, 98, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Wang, C. Electrospun micro- and nanofibers for sustained delivery of paclitaxel to treat C6 glioma in vitro. Pharm. Res. 2006, 23, 1817–1826. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.; Sparer, R.; Hobot, C.; Dang, K. Adjusting drug diffusivity using miscible polymer blends. J. Controll. Release 2005, 102, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Yang, L.; Liang, Q.; Zhang, X.; Guan, H.; Xu, X.; Chen, X.; Jing, X. Influence of the drug compatibility with polymer solution on the release kinetics of electrospun fiber formulation. J. Controll. Release 2005, 105, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Wei, Z.; Zhao, H.; Liu, T.; Dong, A.; Zhang, J. Electrospinning of Ibuprofen-Loaded Composite Nanofibers for Improving the Performances of Transdermal Patches. J. Nanosci. Nanotechnol. 2013, 13, 3855–3863. [Google Scholar] [CrossRef] [PubMed]
- Zupancic, S.; Sinha-Ray, S.; Sinha-Ray, S.; Kristl, J.; Yarin, A.L. Long-Term Sustained Ciprofloxacin Release from PMMA and Hydrophilic Polymer Blended Nanofibers. Mol. Pharm. 2016, 13, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Zupancic, S.; Sinha-Ray, S.; Sinha-Ray, S.; Kristl, J.; Yarin, A.L. Controlled Release of Ciprofloxacin from Core-Shell Nanofibers with Monolithic or Blended Core. Mol. Pharm. 2016, 13, 1393–1404. [Google Scholar] [CrossRef] [PubMed]
- Khansari, S.; Duzyer, S.; Sinha-Ray, S.; Hockenberger, A.S.; Yarin, A.L.; Pourdeyhimi, B. Two-Stage Desorption-Controlled Release of Fluorescent Dye and Vitamin from Solution-Blown and Electrospun Nanofiber Mats Containing Porogens. Mol. Pharm. 2013, 10, 4509–4526. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, P.S.; Lochee, Y.; Jhurry, D.; Bhaw-Luximon, A.; Bowlin, G.L. Characterization of Electrospun Novel Poly(ester-ether) Copolymers: 1,4-Dioxan-2-one and D,L-3-Methyl-1,4-dioxan-2-one. J. Eng. Fibers Fabr. 2011, 6, 60–69. [Google Scholar]
- Meng, Z.X.; Zheng, W.; Li, L.; Zheng, Y.F. Fabrication, characterization and in vitro drug release behavior of electrospun PLGA/chitosan nanofibrous scaffold. Mater. Chem. Phys. 2011, 125, 606–611. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Feng, Y.; Li, J.; Lim, C.; Ramakrishna, S. Coaxial electrospinning of (fluorescein isothiocyanate-conjugated bovine serum albumin)-encapsulated poly(epsilon-caprolactone) nanofibers for sustained release. Biomacromolecules 2006, 7, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yan, K.; Lin, Y.; Hsieh, P.C.H. Biodegradable Core/Shell Fibers by Coaxial Electrospinning: Processing, Fiber Characterization, and Its Application in Sustained Drug Release. Macromolecules 2010, 43, 6389–6397. [Google Scholar] [CrossRef]
- Wu, J.; Wang, N.; Zhao, Y.; Jiang, L. Electrospinning of multilevel structured functional micro-/nanofibers and their applications. J. Mater. Chem. A 2013, 1, 7290–7305. [Google Scholar] [CrossRef]
- Varabhas, J.S.; Chase, G.G.; Reneker, D.H. Electrospun nanofibers from a porous hollow tube. Polymer 2008, 49, 4226–4229. [Google Scholar] [CrossRef]
- Tran, C.; Kalra, V. Co-continuous nanoscale assembly of Nafion-polyacrylonitrile blends within nanofibers: A facile route to fabrication of porous nanofibers. Soft Matter 2013, 9, 846–852. [Google Scholar] [CrossRef]
- Cao, S.; Hu, B.; Liu, H. Synthesis of pH-responsive crosslinked poly[styrene-co-(maleic sodium anhydride)] and cellulose composite hydrogel nanofibers by electrospinning. Polym. Int. 2009, 58, 545–551. [Google Scholar] [CrossRef]
- Qi, M.; Li, X.; Yang, Y.; Zhou, S. Electrospun fibers of acid-labile biodegradable polymers containing ortho ester groups for controlled release of paracetamol. Eur. J. Pharm. Biopharm. 2008, 70, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Ebara, M.; Aoyagi, T. Temperature-responsive electrospun nanofibers for ‘on-off’ switchable release of dextran. Sci. Technol. Adv. Mater. 2012, 13, 064203. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Xu, L.; Yao, F.; Li, G.; Kang, E. Smart Nanofibers with a Photoresponsive Surface for Controlled Release. ACS Appl. Mater. Interfaces 2009, 1, 2424–2427. [Google Scholar] [CrossRef] [PubMed]
- Abidian, M.; Kim, D.; Martin, D. Conducting-polymer nanotubes for controlled drug release. Adv. Mater. 2006, 18, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Wendorff, J.; Pietzonka, C.; Jia, Z.; Wang, G. Biocompatible and biodegradable polymer nanofibers displaying superparamagnetic properties. ChemPhysChem 2005, 6, 1461–1465. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Singh, H.; Hatton, T.; Rutledge, G. Field-responsive superparamagnetic composite nanofibers by electrospinning. Polymer 2004, 45, 5505–5514. [Google Scholar] [CrossRef]
- Chen, H.; Hsieh, Y. Ultrafine hydrogel fibers with dual temperature- and pH-responsive swelling behaviors. J. Polym. Sci. Part A: Polym. Chem. 2004, 42, 6331–6339. [Google Scholar] [CrossRef]
- Li, L.; Hsieh, Y. Ultra-fine polyelectrolyte hydrogel fibers from poly(acrylic acid)/poly(vinyl alcohol). Nanotechnology 2005, 16, 2852–2860. [Google Scholar] [CrossRef]
- Sanson, C.; Schatz, C.; Le Meins, J.; Soum, A.; Thevenot, J.; Garanger, E.; Lecommandoux, S. A simple method to achieve high doxorubicin loading in biodegradable polymersomes. J. Controll. Release 2010, 147, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Gondo, D.; Wada, T.; Kanehashi, S.; Nagai, K. Effects of various liquid organic solvents on solvent-induced crystallization of amorphous poly(lactic acid) film. J. Appl. Polym. Sci. 2013, 129, 1607–1617. [Google Scholar] [CrossRef]
- Li, L.; Jiang, Z.; Xu, J.; Fang, T. Predicting Poly(vinyl pyrrolidone)’s Solubility Parameter and Systematic Investigation of the Parameters of Electrospinning with Response Surface Methodology. J. Appl. Polym. Sci. 2014, 131, 40304. [Google Scholar] [CrossRef]
- Hansen, C.M. Hansen Solubility Parameters: A User’s Handbook, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Wenzel, R.N. Resistance of Solid Surfaces to Wetting by Water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
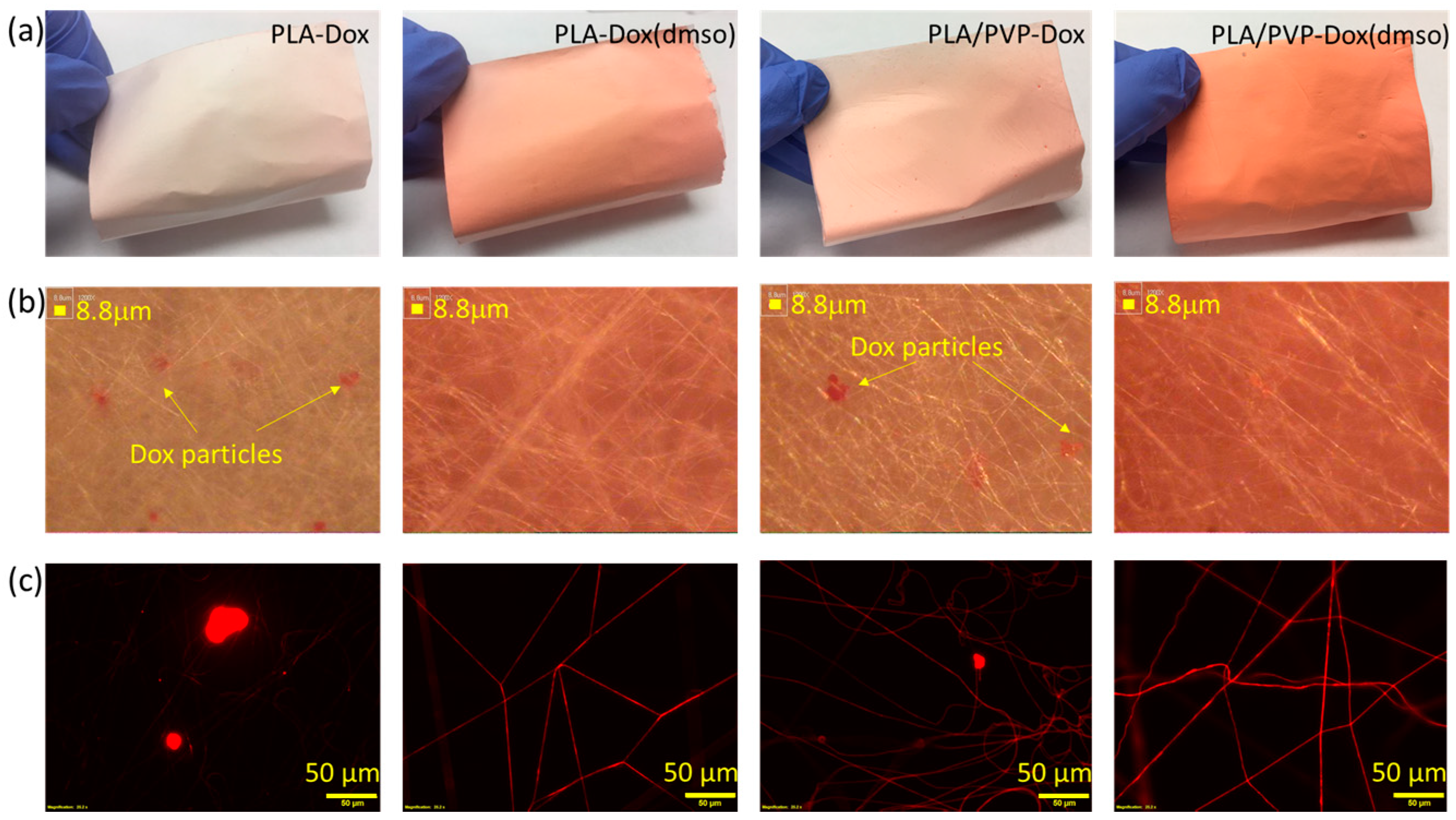
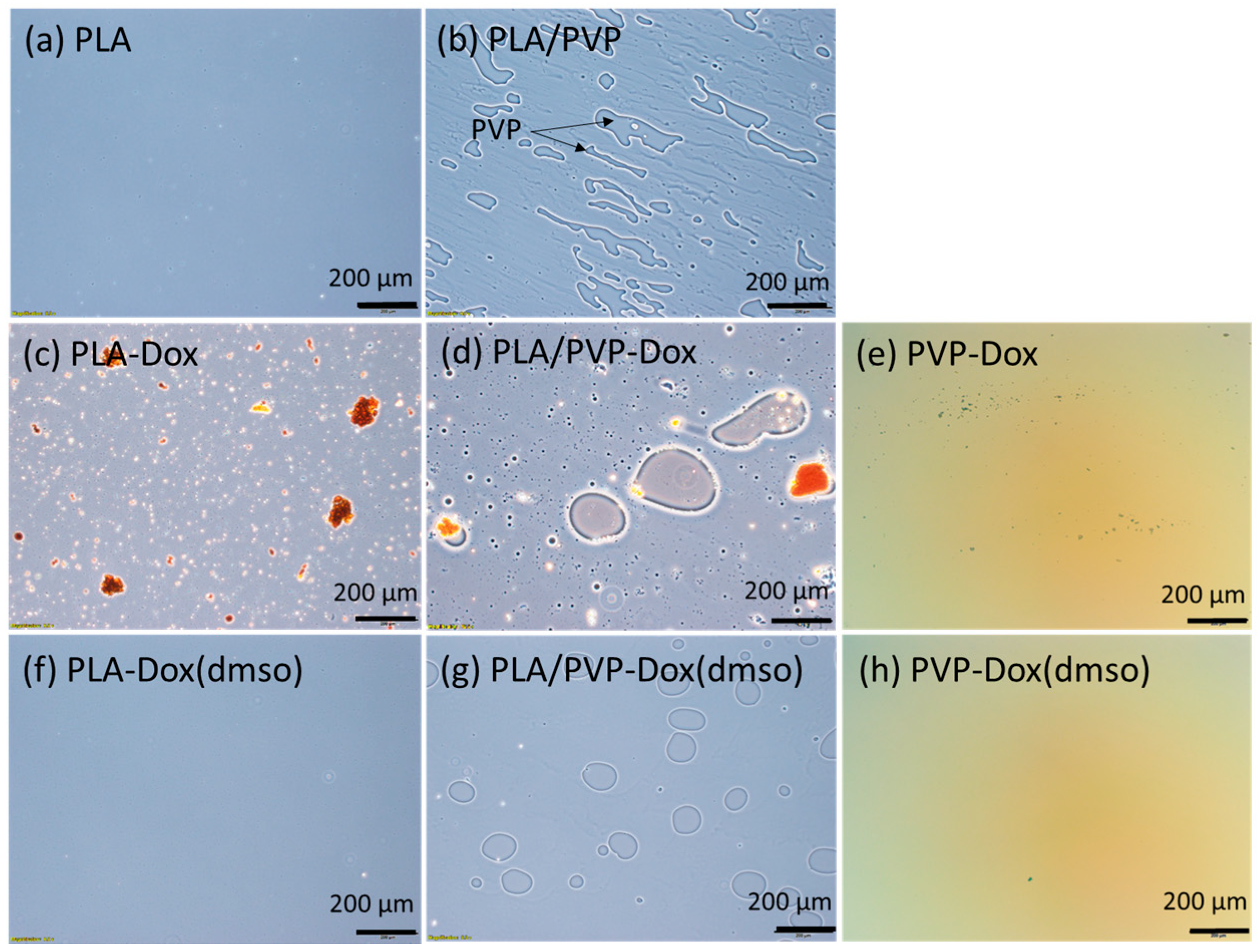



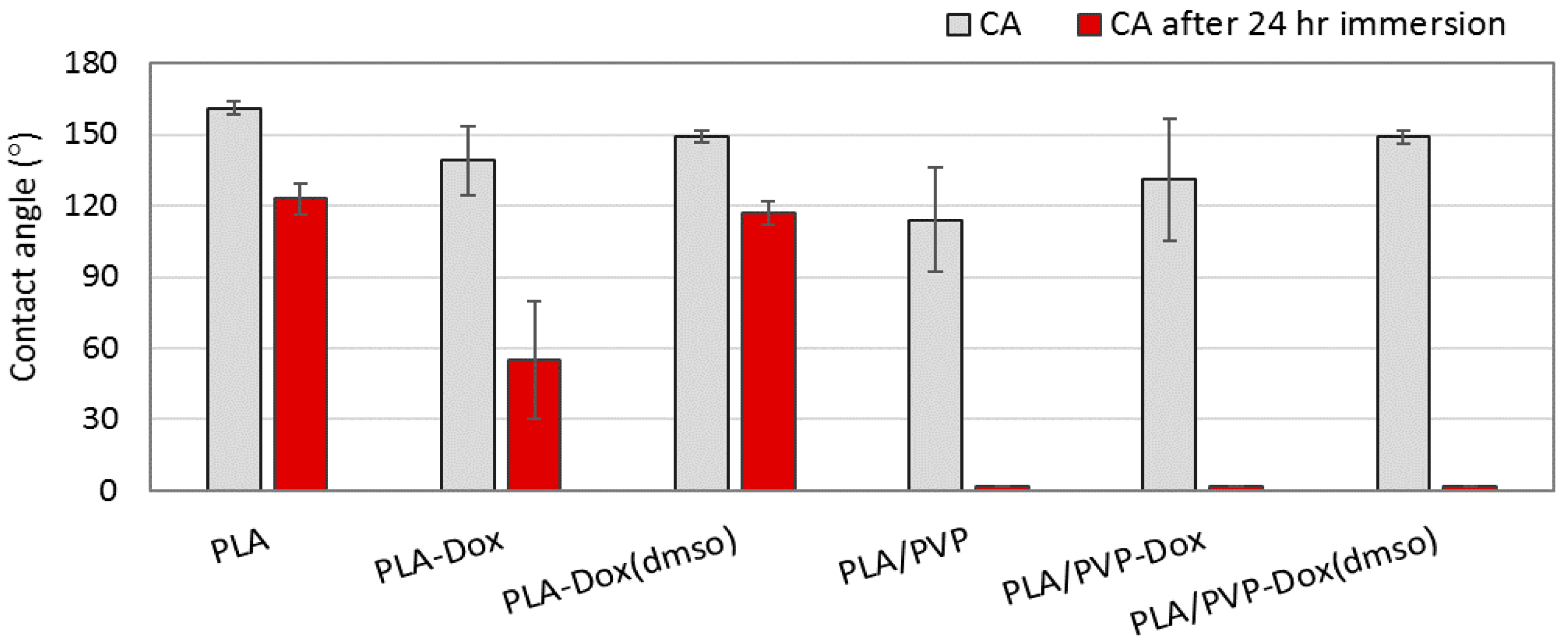
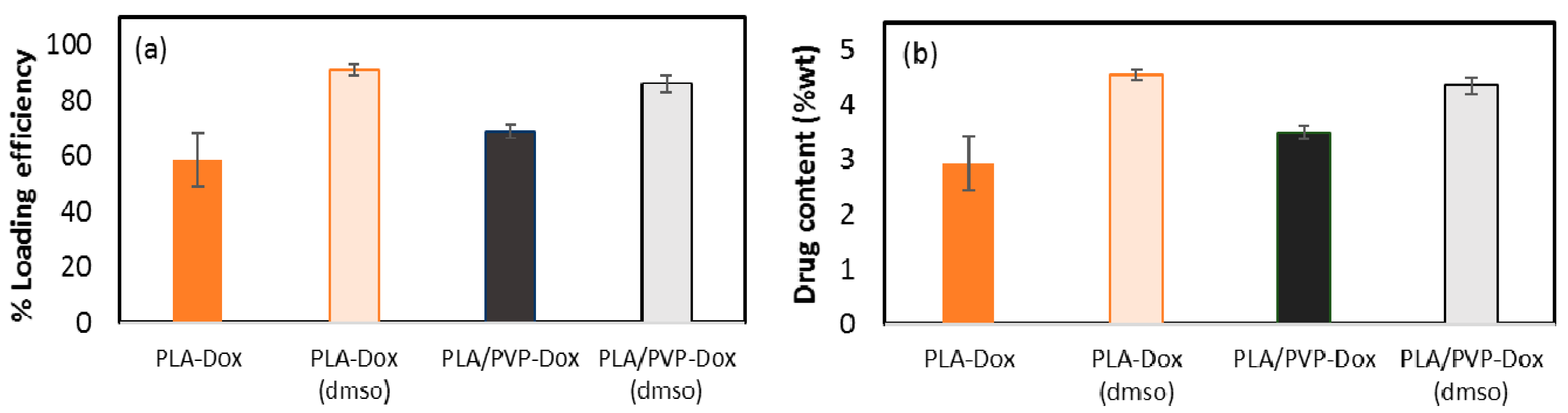
| Code | Description |
|---|---|
| PLA | PLA web |
| PLA/PVP | PLA/PVP blend web (80:20 weight ratio) |
| PLA-Dox | Dox-loaded PLA web, Dox was mixed with PLA pre-spinning solution |
| PLA/PVP-Dox | Dox-loaded PLA/PVP web, Dox was mixed with PLA/PVP pre-spinning solution |
| PLA-Dox(DMSO) | Dox-loaded PLA web, Dox dissolved in DMSO was mixed with PLA pre-spinning solution |
| PLA/PVP-Dox(DMSO) | Dox-loaded PLA/PVP web, Dox dissolved in DMSO was mixed with PLA/PVP pre-spinning solution |
| Component | δt (MPa1/2) | δd (MPa1/2) | δp (MPa1/2) | δh (MPa1/2) |
|---|---|---|---|---|
| PLA [54] | 21.2 | 17.5 | 9.5 | 7.3 |
| PVP [55] | 24.3 | 18.8 | 13.4 | 7.5 |
| DMSO [56] | 26.7 | 18.4 | 16.4 | 10.2 |
| DCM [56] | 20.2 | 18.2 | 6.3 | 6.1 |
| DMF [56] | 24.9 | 17.4 | 13.7 | 11.3 |
| DCM/DMF * | 22.2 | 17.8 | 10.0 | 8.7 |
| R (MPa1/2) | DMSO | DCM | DMF | DCM/DMF |
|---|---|---|---|---|
| PLA | 7.7 | 3.7 | 5.8 | 1.6 |
| PVP | 4.1 | 7.3 | 4.7 | 4.1 |
| DMSO | 0 | 10.9 | 3.5 | 6.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.C.; Yuan, Y.; Choi, K.; Choi, S.-O.; Kim, J. Doxorubicin Release Controlled by Induced Phase Separation and Use of a Co-Solvent. Materials 2018, 11, 681. https://doi.org/10.3390/ma11050681
Park SC, Yuan Y, Choi K, Choi S-O, Kim J. Doxorubicin Release Controlled by Induced Phase Separation and Use of a Co-Solvent. Materials. 2018; 11(5):681. https://doi.org/10.3390/ma11050681
Chicago/Turabian StylePark, Seok Chan, Yue Yuan, Kyoungju Choi, Seong-O Choi, and Jooyoun Kim. 2018. "Doxorubicin Release Controlled by Induced Phase Separation and Use of a Co-Solvent" Materials 11, no. 5: 681. https://doi.org/10.3390/ma11050681
APA StylePark, S. C., Yuan, Y., Choi, K., Choi, S.-O., & Kim, J. (2018). Doxorubicin Release Controlled by Induced Phase Separation and Use of a Co-Solvent. Materials, 11(5), 681. https://doi.org/10.3390/ma11050681





