Preparation and Characterization of PVA Alkaline Solid Polymer Electrolyte with Addition of Bamboo Charcoal
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of the PVA-BC-KOH Electrolyte Membranes
2.3. Morphology Investigation
2.4. XRD Analysis
2.5. Moisture Content Measurement
2.6. Thermal Analysis
2.7. Measurement of Ionic Conductivity
2.8. Measurement of the Electrochemical Stability Window
2.9. The Assembly of Ni-MH Battery
3. Results and Discussions
3.1. Morphology
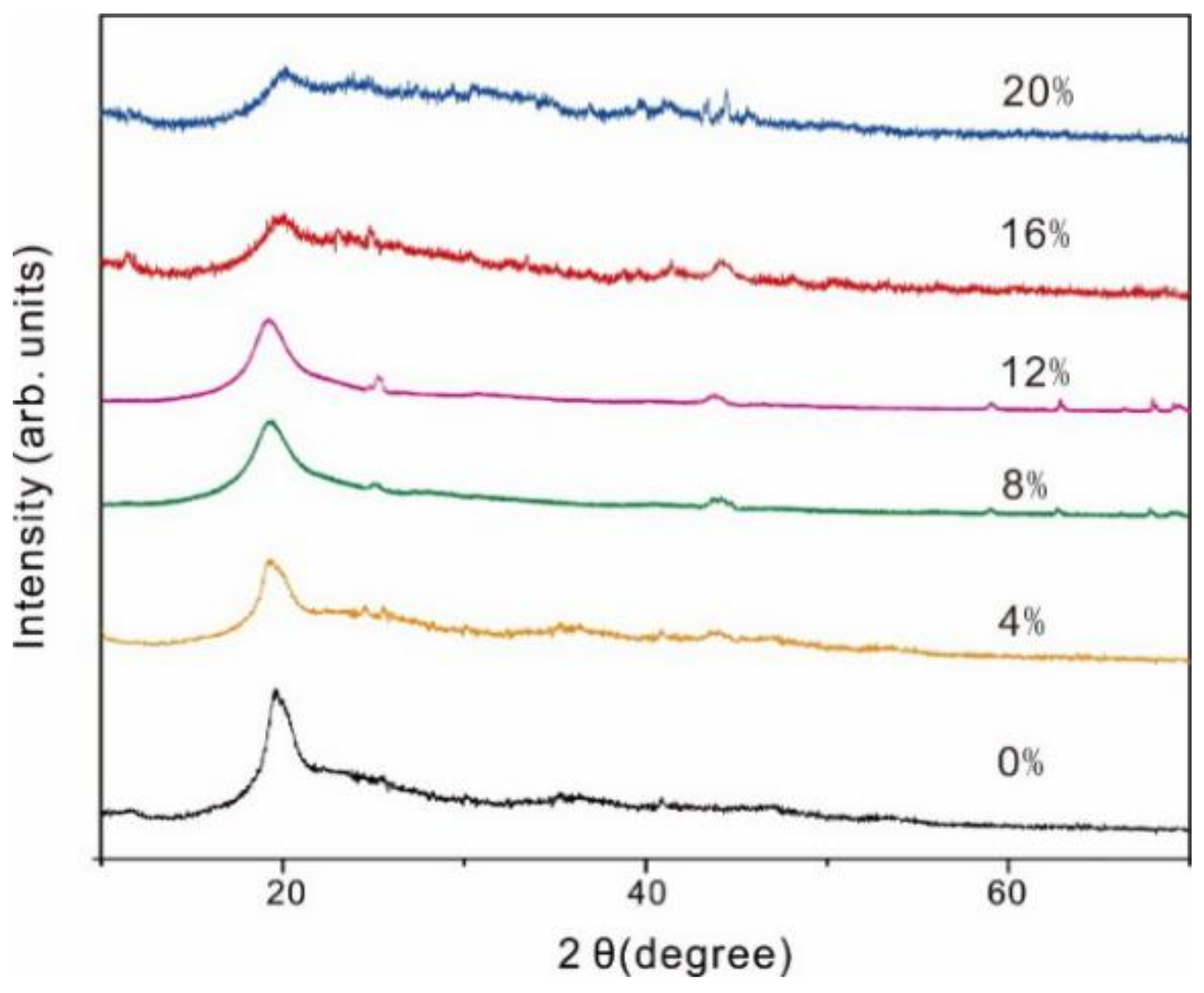
3.2. XRD Analysis
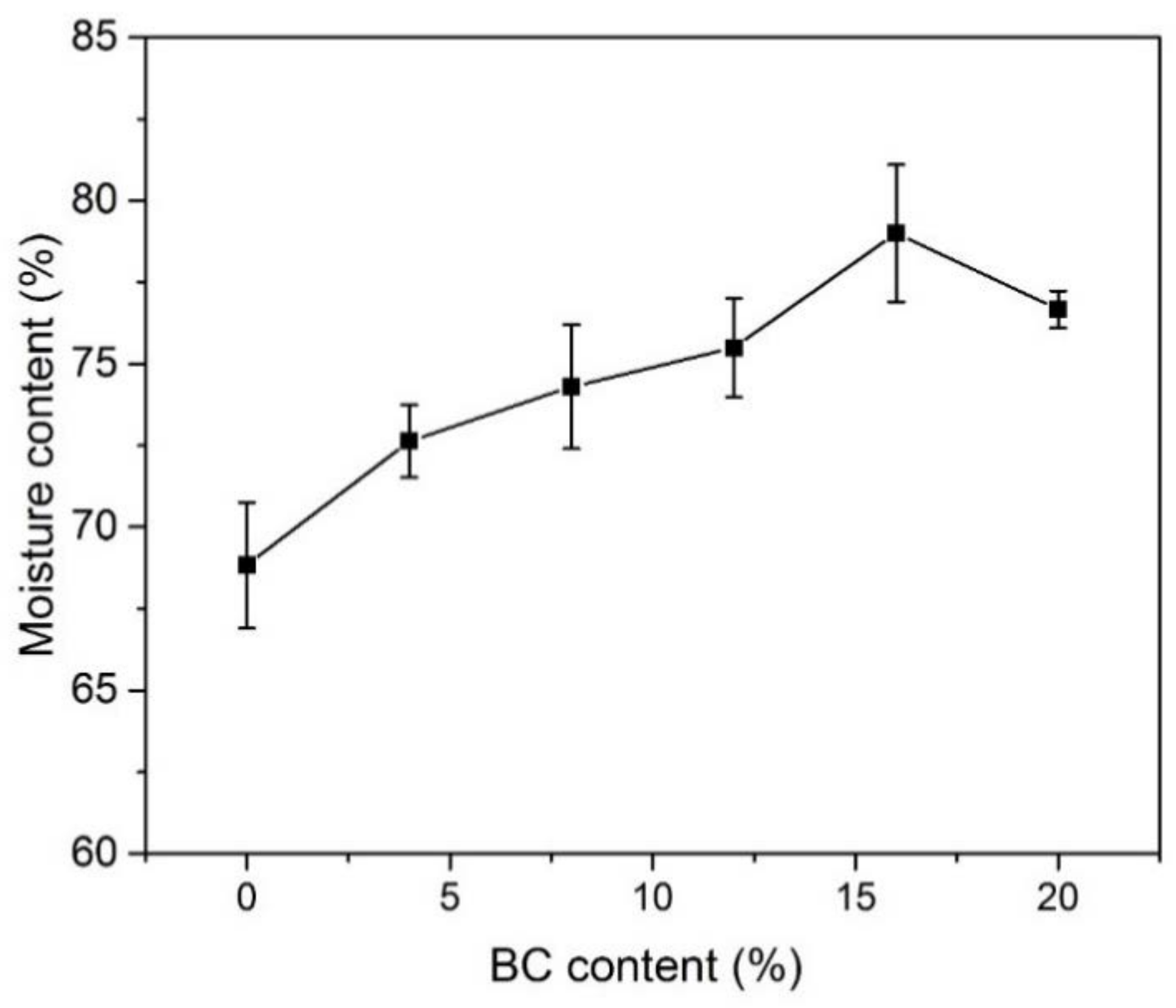
3.3. Moisture Content
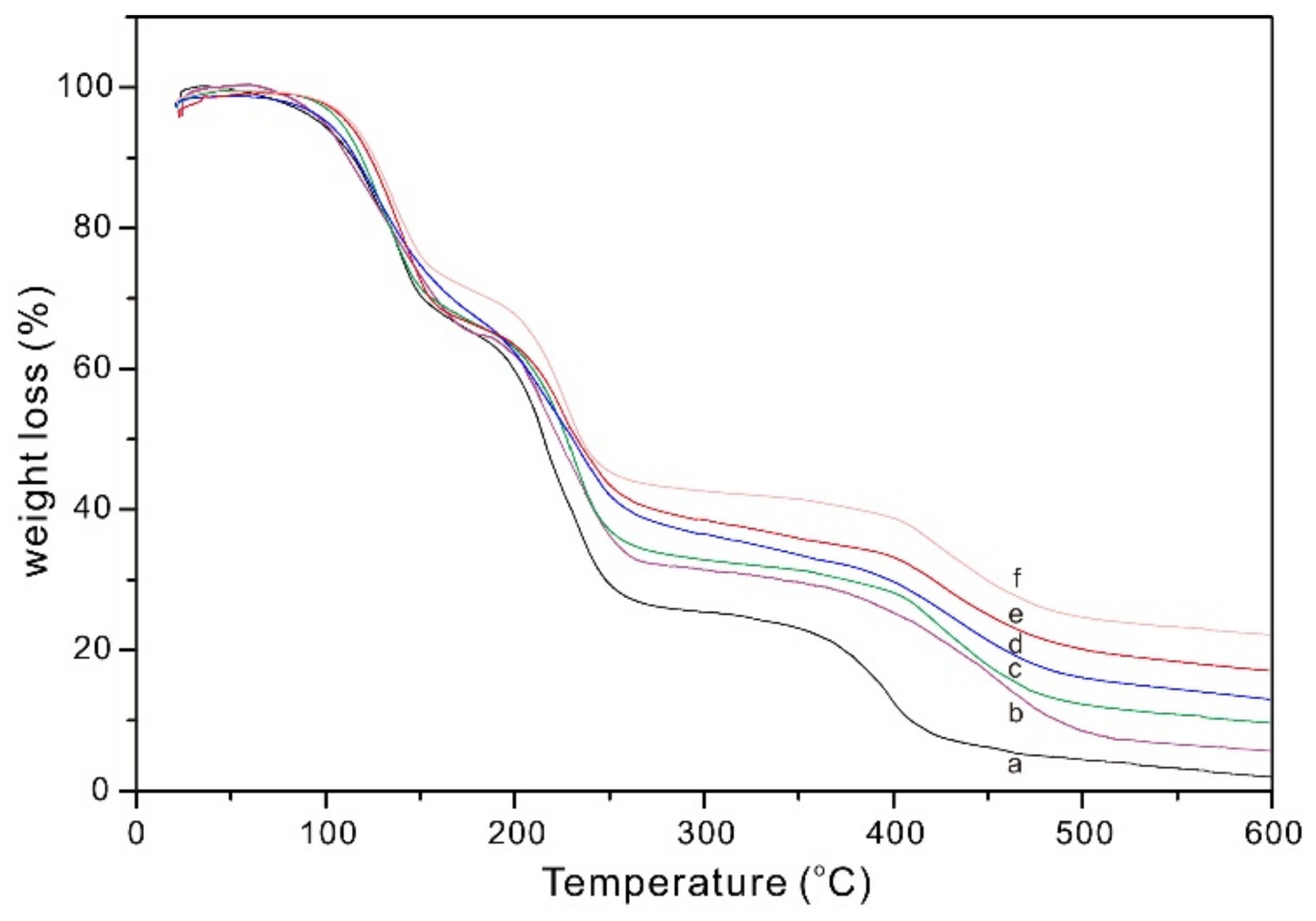
3.4. Thermal Analysis
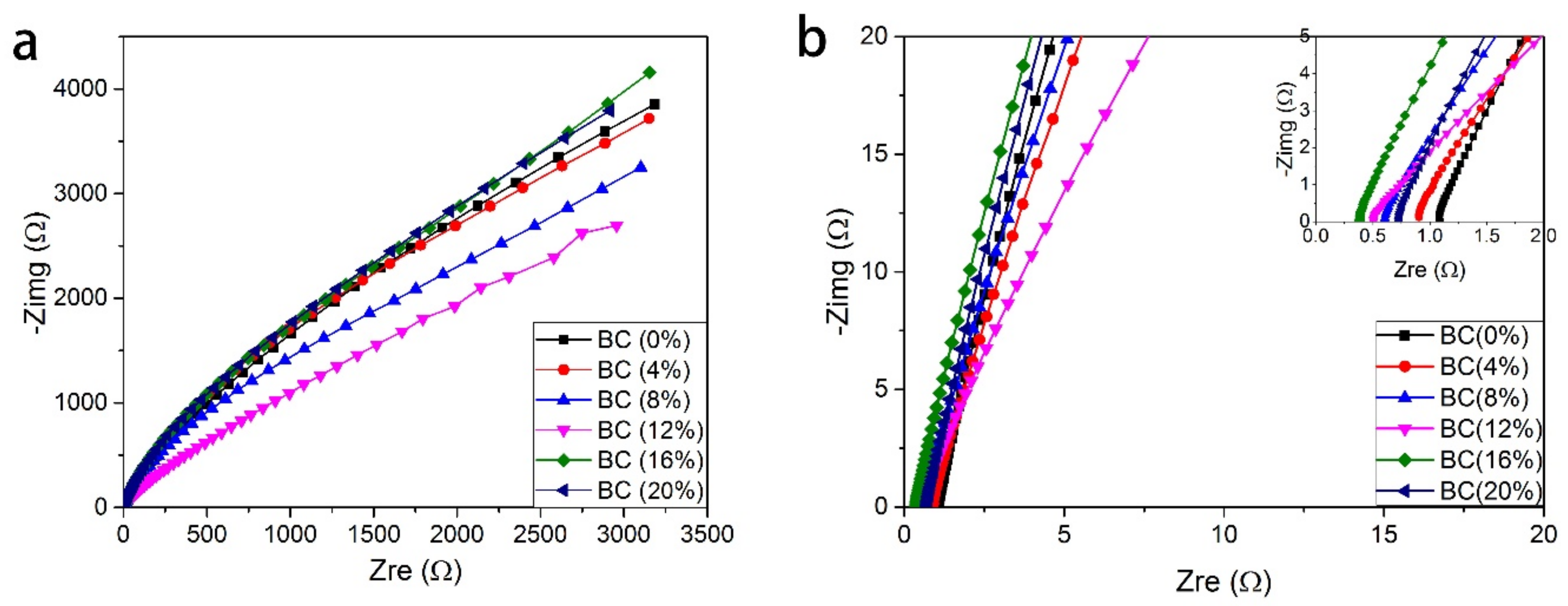
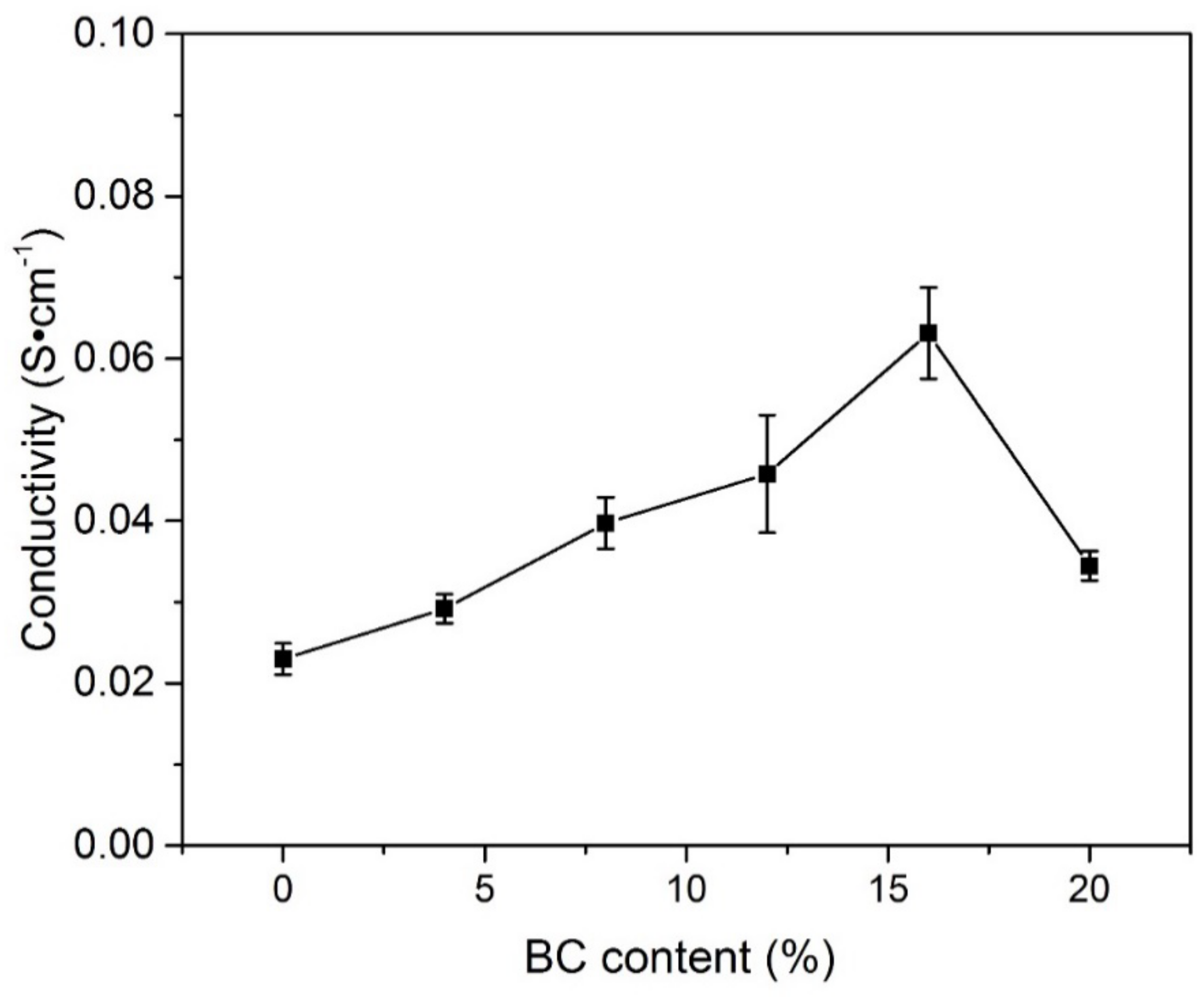
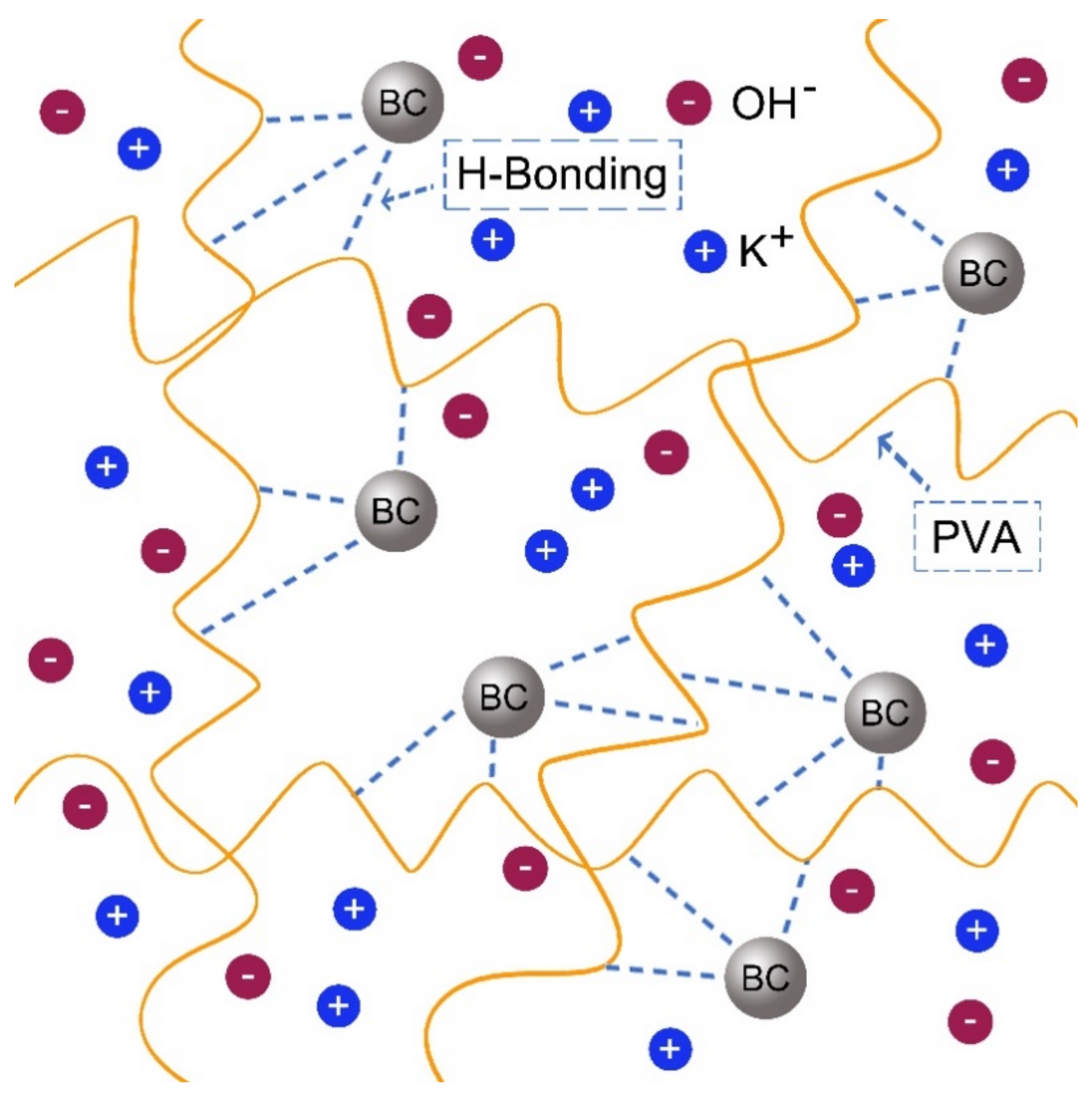
3.5. The Influence of BC Content on Ionic Conductivity
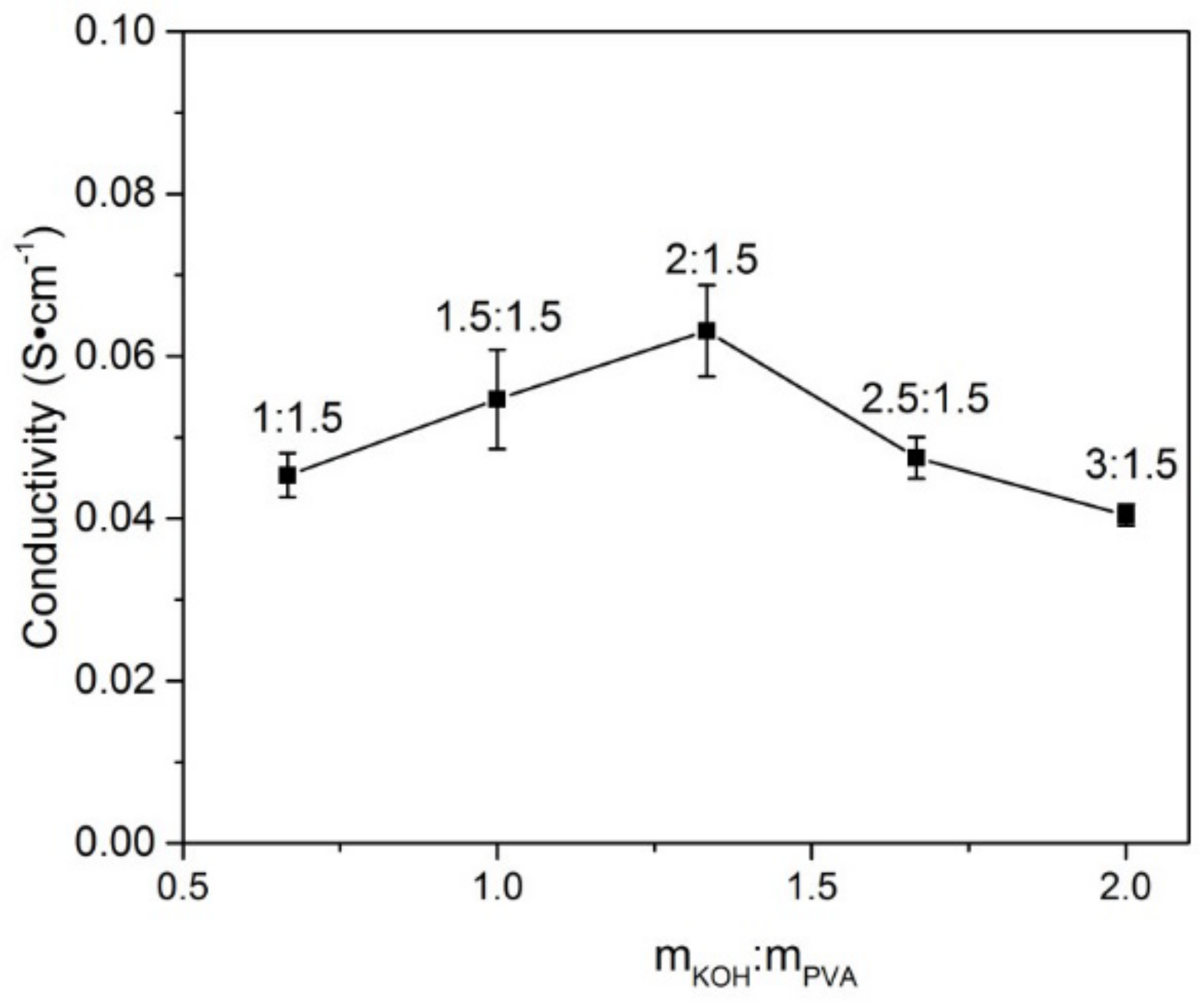
3.6. The Influence of Content of KOH on Ionic Conductivity
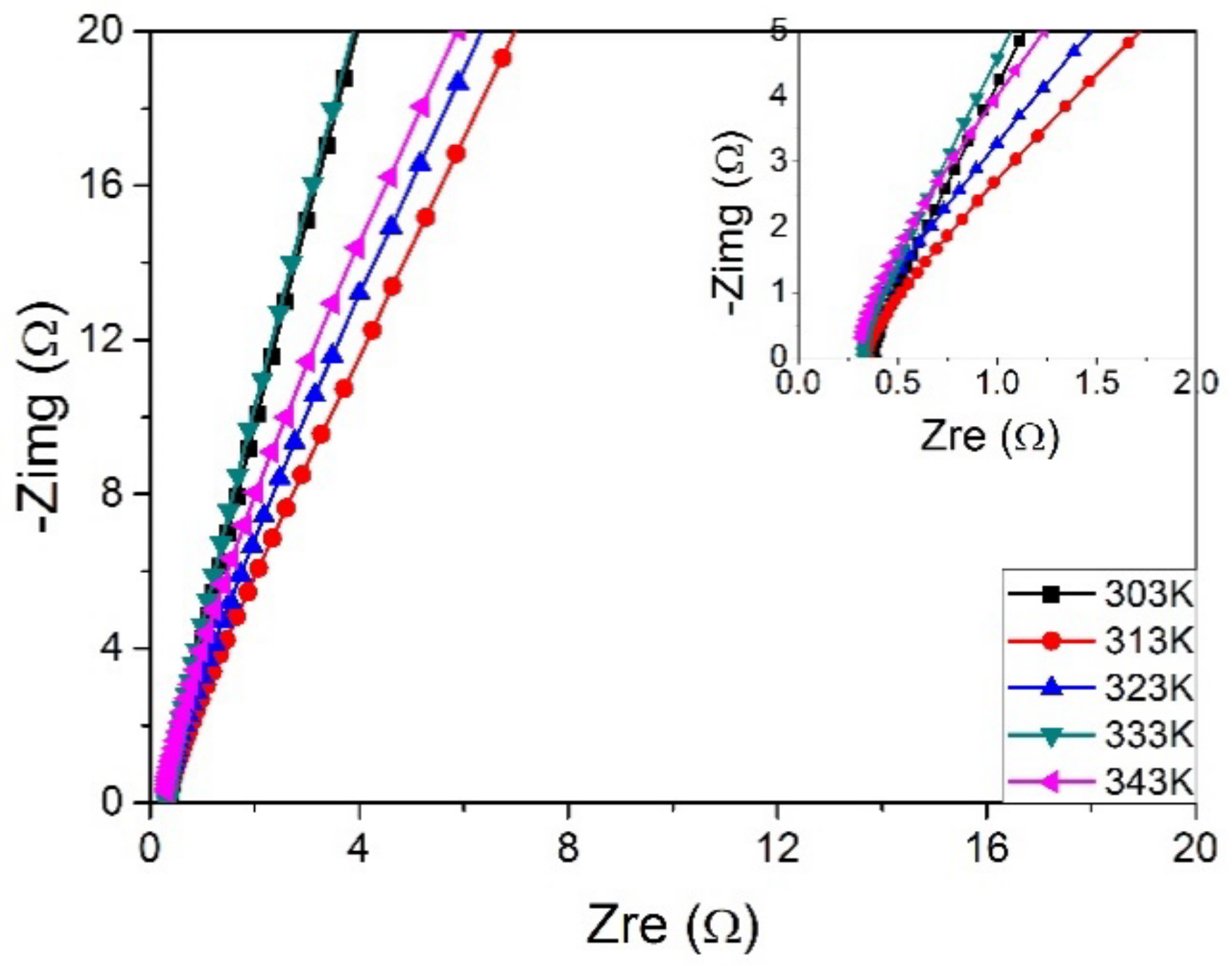
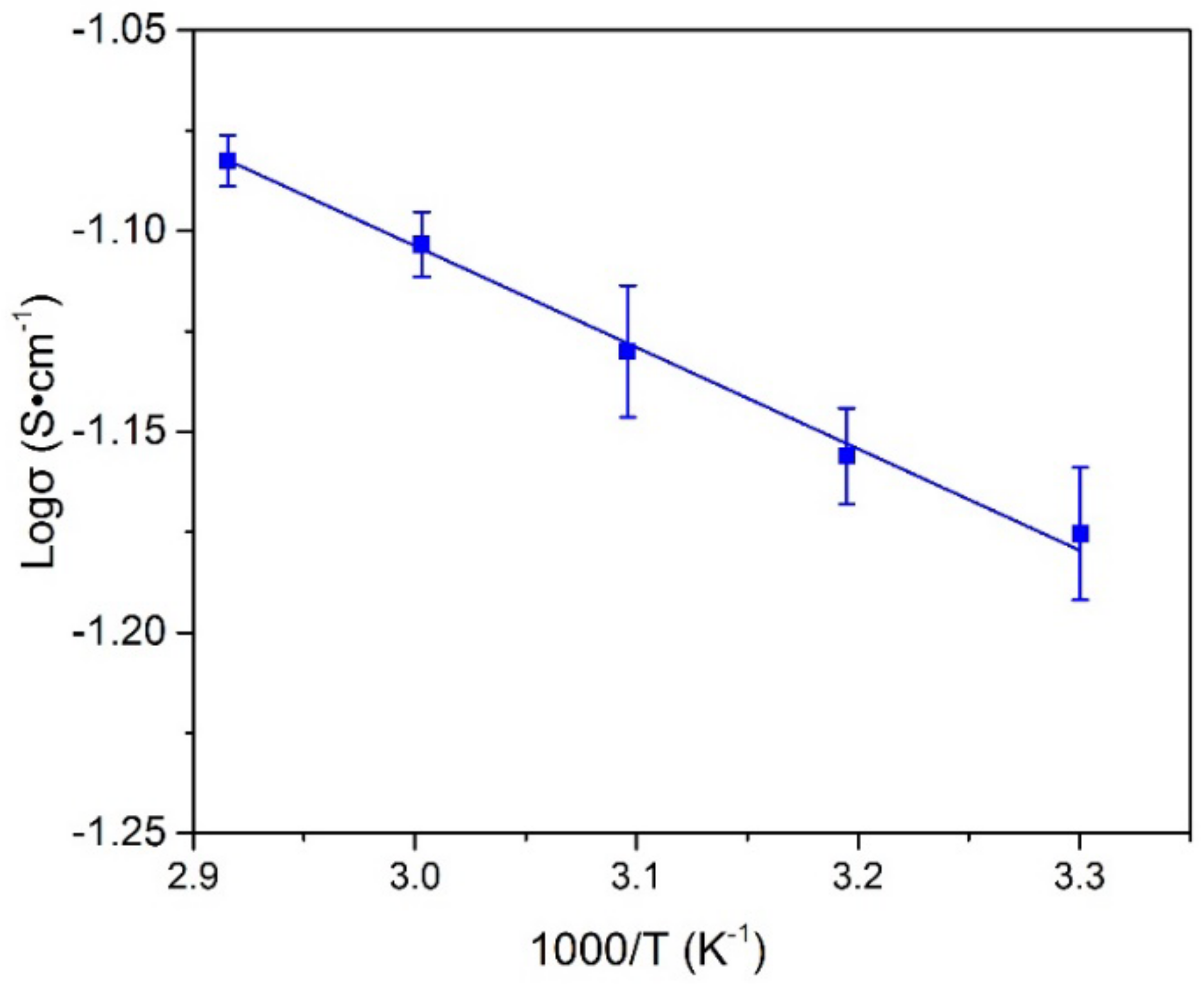
3.7. Influence of Temperature on the Ionic Conductivity
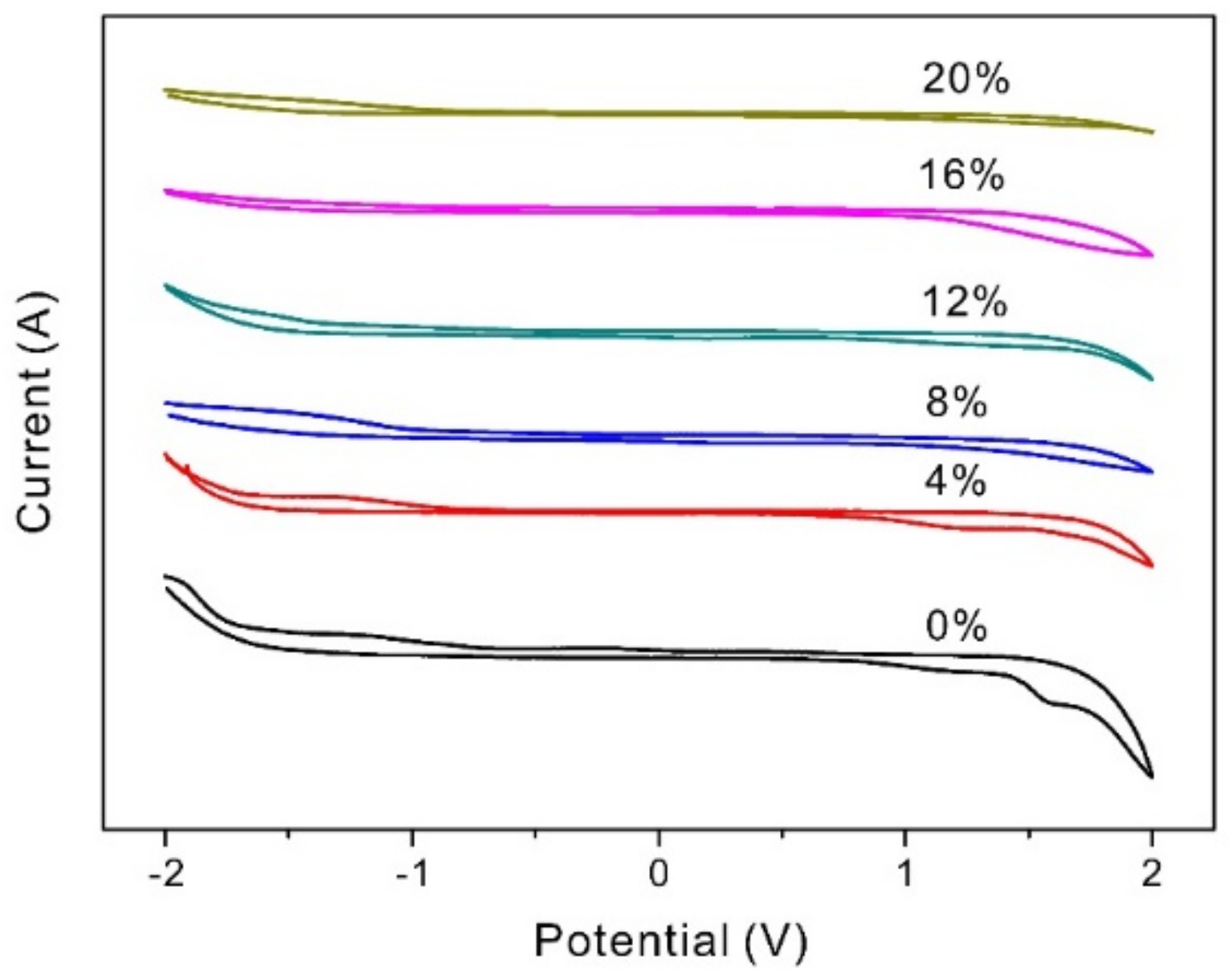
3.8. The Electrochemical Stability Windows
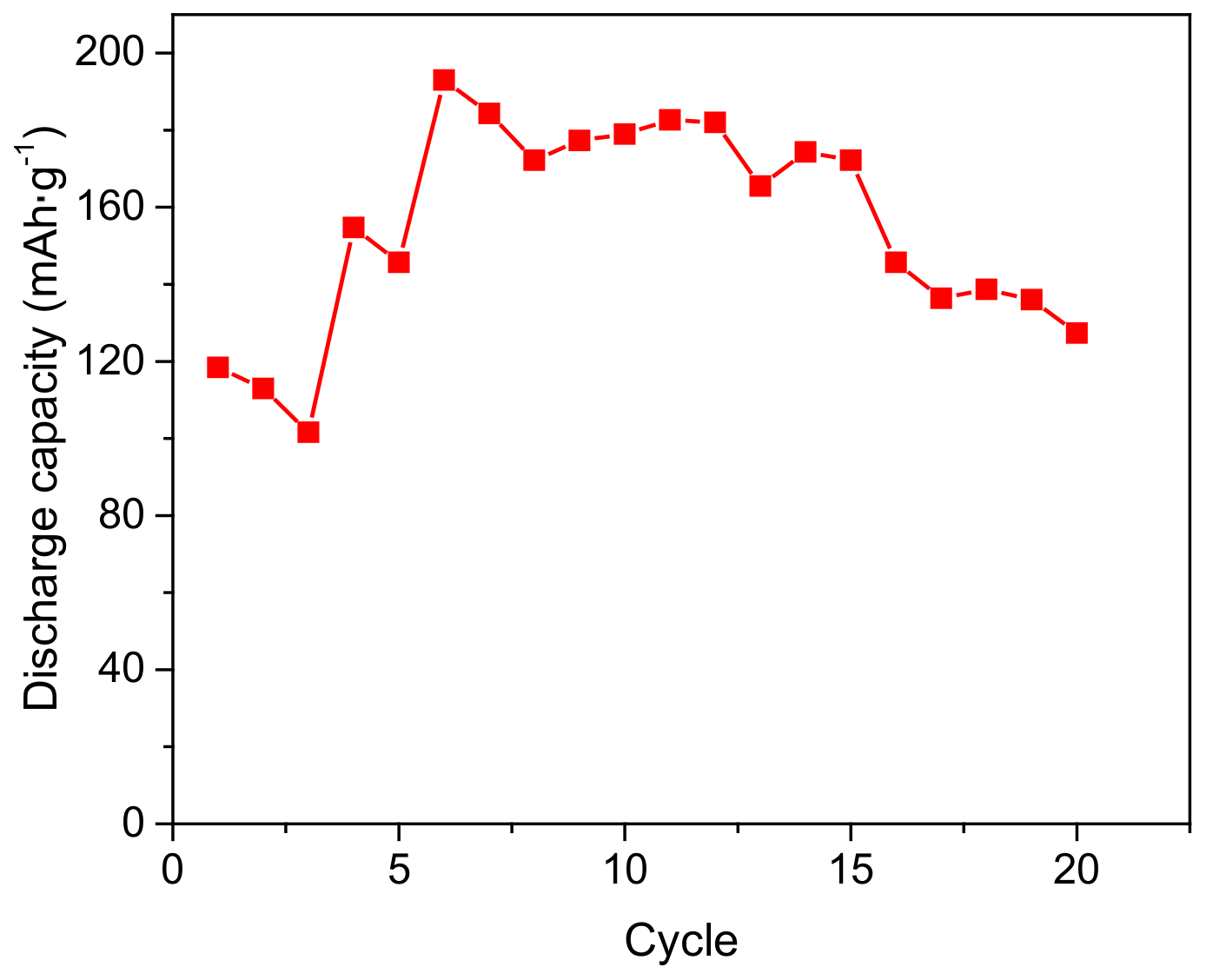
3.9. The Applications in Ni-MH Battery
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Yang, Y.; Liu, X.; Dai, Z.; Yuan, F.; Bando, Y.; Golberg, D.; Wang, X. In situ electrochemistry of rechargeable battery materials: Status report and perspectives. Adv. Mater. 2017, 29, 1606922. [Google Scholar] [CrossRef] [PubMed]
- Sebastián, D.; Baglio, V. Advanced materials in polymer electrolyte fuel cells. Materials 2017, 10, 1163. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.; Lee, J.H.; Kim, T.Y.; Han, U.K.; Lee, J.H. High-performance triboelectric nanogenerators based on solid polymer electrolytes with asymmetric pairing of ions. Adv. Energy Mater. 2017, 7, 1700289. [Google Scholar] [CrossRef]
- Novitski, D.M.; Kosakian, A.; Weissbach, T.; Secanell, M.; Holdcroft, S. Electrochemical reduction of dissolved oxygen in alkaline solid polymer electrolyte films. J. Am. Chem. Soc. 2016, 138, 15465–15472. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Bao, J.J.; Chen, T.T.; Zou, B.K.; Wen, Z.Y.; Guo, X.X.; Chen, C.H. Solid polymer electrolyte based on thermoplastic polyurethane and its application in all-solid-state lithium ion batteries. Solid State Ion. 2017, 309, 15–21. [Google Scholar] [CrossRef]
- Yuan, A.; Zhao, J. Composite alkaline polymer electrolytes and its application to nickel–metal hydride batteries. Electrochim. Acta 2006, 51, 2454–2462. [Google Scholar] [CrossRef]
- Iwakura, C.; Nohara, S.; Furukawa, N.; Inoue, H. The possible use of polymer gel electrolytes in nickel/metal hydride battery. Solid State Ion. 2002, 148, 487–492. [Google Scholar] [CrossRef]
- Geng, M.; Northwood, D.O. Development of advanced rechargeable Ni/MH and Ni/Zn batteries. Int. J. Hydrog. Energy 2003, 28, 633–636. [Google Scholar] [CrossRef]
- Albert, A.; Lochner, T.; Schmidt, T.J.; Gubler, L. Stability and degradation mechanisms of radiation-grafted polymer electrolyte membranes for water electrolysis. ACS Appl. Mater. Interfaces 2016, 8, 15297. [Google Scholar] [CrossRef] [PubMed]
- Fattah, N.F.A.; Ng, H.M.; Mahipal, Y.K.; Numan, A.; Ramesh, S.; Ramesh, K. An approach to solid-state electrical double layer capacitors fabricated with graphene oxide-doped, ionic liquid-based solid copolymer electrolytes. Materials 2016, 9, 450. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.F.; Arof, A.K. Ionic conductivity in PEO-KOH polymer electrolytes and electrochemical cell performance. Phys. Status Solidi 2005, 202, 2494–2500. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Mangani, I.R.; Vasanthi, R.; Selladurai, S. Ionic conductivity and battery characteristic studies on PEO + NaClO3, polymer electrolyte. Ionics 2001, 7, 88–93. [Google Scholar] [CrossRef]
- Yang, C.C.; Lin, S.J.; Hsu, S.T. Synthesis and characterization of alkaline polyvinyl alcohol and poly (epichlorohydrin) blend polymer electrolytes and performance in electrochemical cells. J. Power Sour. 2003, 122, 210–218. [Google Scholar] [CrossRef]
- Virya, A.; Lian, K. Polyacrylamide-lithium chloride polymer electrolyte and its applications in electrochemical capacitors. Electrochem. Commun. 2017, 74, 33–37. [Google Scholar] [CrossRef]
- Liao, G.M.; Yang, C.C.; Hu, C.C. Optimal loading of quaternized chitosan nanofillers in functionalized polyvinyl alcohol polymer membrane for effective hydroxide ion conduction and suppressed alcohol transport. Polymer 2018, 138, 65–74. [Google Scholar] [CrossRef]
- Lewandowski, A.; Skorupska, K.; Malinska, J. Novel poly (vinyl alcohol)-KOH-H2O alkaline polymer electrolyte. Solid State Ion. 2000, 133, 265–271. [Google Scholar] [CrossRef]
- Li, J.; Lian, K. A comparative study of tetraethylammonium hydroxide polymer electrolytes for solid electrochemical capacitors. Polymer 2016, 99, 140–146. [Google Scholar] [CrossRef]
- Yang, C.C. Chemical composition and XRD analyses for alkaline composite PVA polymer electrolyte. Mater. Lett. 2004, 58, 33–38. [Google Scholar] [CrossRef]
- Ngai, K.S.; Ramesh, S.; Ramesh, K.; Juan, J.C. A review of polymer electrolytes: Fundamental, approaches and applications. Ionics 2016, 22, 1259–1279. [Google Scholar] [CrossRef]
- Pan, W.H.; Lue, S.J.; Chang, C.M.; Liu, Y.L. Alkali doped polyvinyl alcohol/multi-walled carbon nano-tube electrolyte for direct methanol alkaline fuel cell. J. Membr. Sci. 2011, 376, 225–232. [Google Scholar] [CrossRef]
- Hema, M.; Tamilselvi, P.; Pandaram, P. Conductivity enhancement in SiO2 doped PVA: PVDF nanocomposite polymer electrolyte by gamma ray irradiation. Nucl. Instrum. Methods Phys. Res. Sect. B 2017, 403, 13–20. [Google Scholar] [CrossRef]
- More, S.; Dhokne, R.; Moharil, S. Structural properties and temperature dependence dielectric properties of PVA-Al2O3, composite thin films. Polym. Bull. 2017, 19, 1–15. [Google Scholar]
- Zhang, W.; Liu, X.; Wang, D.; Jin, Y. Effects of bamboo charcoal on fouling and microbial diversity in a flat-sheet ceramic membrane bioreactor. Bioresour. Technol. 2017, 243, 1020–1026. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.A.; Xu, C.; Shi, M.; Song, G.; Lang, X. The high performance of tungsten carbides/porous bamboo charcoals supported Pt catalysts for methanol electrooxidation. J. Power Sour. 2013, 242, 273–279. [Google Scholar] [CrossRef]
- Yang, C.C. Alkaline direct methanol fuel cell based on a novel anion-exchange composite polymer membrane. J. Appl. Electrochem. 2012, 42, 305–317. [Google Scholar] [CrossRef]
- Xiao, W.; Li, X.H.; Wang, Z.X.; Guo, H.J.; Wang, J.X.; Huang, S.L.; Gan, L. Physicochemical properties of a novel composite polymer electrolyte doped with vinyltrimethoxylsilane-modified nano-La2O3. J. Rare Earth 2012, 30, 1034–1040. [Google Scholar] [CrossRef]
- Zhang, J.; Han, H.; Wu, S.; Xu, S.; Yang, Y.; Zhou, C.; Zhao, X. Conductive carbon nanoparticles hybrid PEO/P(VDF-HFP)/SiO2, nanocomposite polymer electrolyte type dye sensitized solar cells. Solid State Ionics 2007, 178, 1595–1601. [Google Scholar] [CrossRef]
- Wu, Q.; Zhang, J.; Sang, S. Preparation of alkaline solid polymer electrolyte based on PVA-TiO2-KOH-H2O and its performance in Zn-Ni battery. J. Phys. Chem. Solids 2008, 69, 2691–2695. [Google Scholar] [CrossRef]
- Yang, C.C. Study of alkaline nanocomposite polymer electrolytes based on PVA–ZrO2–KOH. Mater. Sci. Eng. B 2006, 131, 256–262. [Google Scholar] [CrossRef]
- Mohamad, A.A.; Arof, A.K. Plasticized alkaline solid polymer electrolyte system. Mater. Lett. 2007, 61, 3096–3099. [Google Scholar] [CrossRef]
- Yang, C.C.; Li, Y.J.; Liou, T.H. Preparation of novel poly(vinyl alcohol)/SiO2 nanocomposite membranes by a sol–gel process and their application on alkaline DMFCs. Desalination 2011, 276, 366–372. [Google Scholar] [CrossRef]
- Li, B.; Lu, X.; Yuan, J.; Zhu, Y.; Li, L. Alkaline poly(vinyl alcohol)/poly(acrylic acid) polymer electrolyte membrane for Ni-MH battery application. Ionics 2015, 21, 141–148. [Google Scholar] [CrossRef]
- Popa, S.; Lliescu, S.; Llia, G.; Plesu, N.; Popa, A.; Visa, A.; Macarie, L. Solid polymer electrolytes based on phosphorus containing polymers for lithium polymer batteries. Eur. Polym. J. 2017, 94, 286–298. [Google Scholar] [CrossRef]
- Fan, L.D.; Chen, J.; Qin, G.; Wang, L.B.; Hu, X.Y.; Shen, Z.S. Preparation of PVA-KOH-Halloysite nanotube alkaline solid polymer electrolyte and its application in Ni-MH battery. Int. J. Electrochem. Sci. 2017, 12, 5142–5156. [Google Scholar] [CrossRef]
- Mohamad, A.A.; Mohamed, N.S.; Yahya, M.Z.A.; Othman, R.; Ramesh, S.; Alias, Y.; Arof, A.K. Ionic conductivity studies of poly(vinyl alcohol) alkaline solid polymer electrolyte and its use in nickel-zinc cells. Solid State Ion. 2003, 156, 171–177. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, L.; Wang, M.; Zhang, Z.; Qin, G.; Hu, X.; Chen, Q. Preparation and Characterization of PVA Alkaline Solid Polymer Electrolyte with Addition of Bamboo Charcoal. Materials 2018, 11, 679. https://doi.org/10.3390/ma11050679
Fan L, Wang M, Zhang Z, Qin G, Hu X, Chen Q. Preparation and Characterization of PVA Alkaline Solid Polymer Electrolyte with Addition of Bamboo Charcoal. Materials. 2018; 11(5):679. https://doi.org/10.3390/ma11050679
Chicago/Turabian StyleFan, Lidan, Mengyue Wang, Zhen Zhang, Gang Qin, Xiaoyi Hu, and Qiang Chen. 2018. "Preparation and Characterization of PVA Alkaline Solid Polymer Electrolyte with Addition of Bamboo Charcoal" Materials 11, no. 5: 679. https://doi.org/10.3390/ma11050679
APA StyleFan, L., Wang, M., Zhang, Z., Qin, G., Hu, X., & Chen, Q. (2018). Preparation and Characterization of PVA Alkaline Solid Polymer Electrolyte with Addition of Bamboo Charcoal. Materials, 11(5), 679. https://doi.org/10.3390/ma11050679




