Structure and Infrared Emissivity Properties of the MAO Coatings Formed on TC4 Alloys in K2ZrF6-Based Solution
Abstract
:1. Introduction
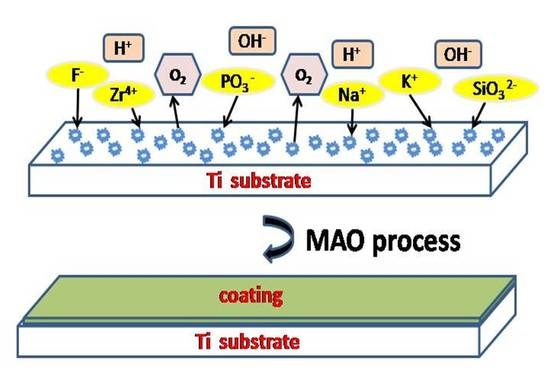
2. Materials and Methods
3. Results
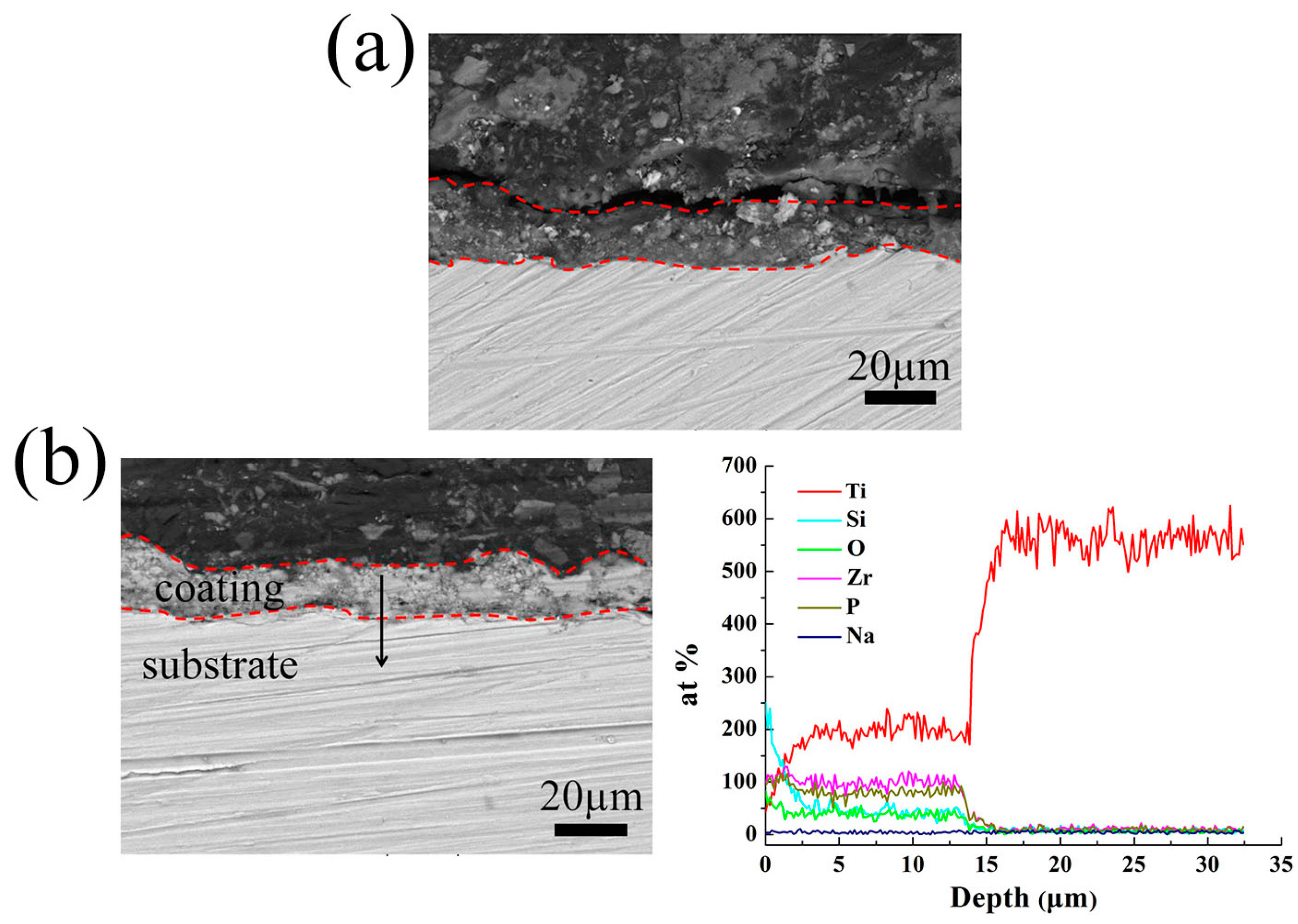
3.1. Surface and Cross-Section Microstructure
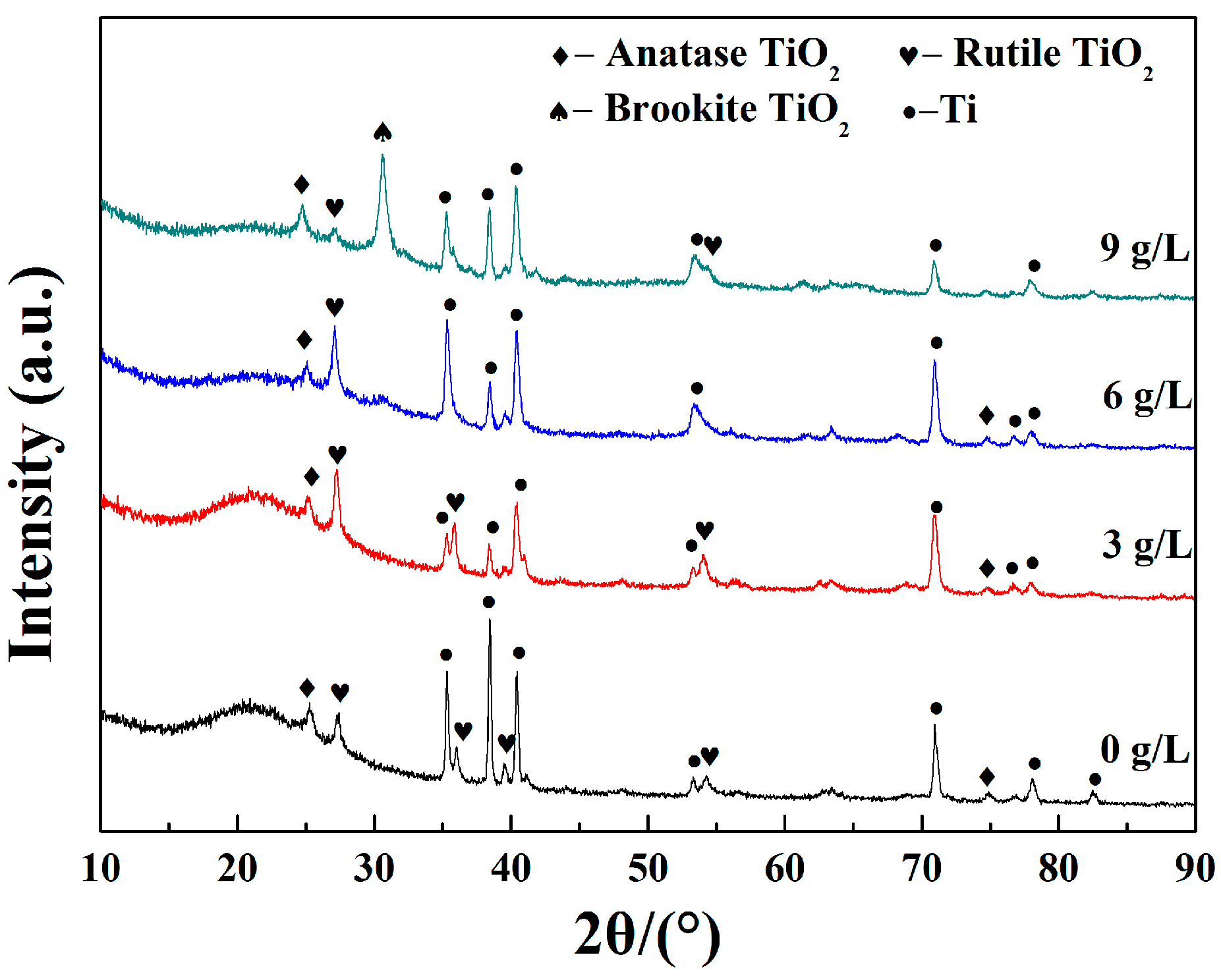
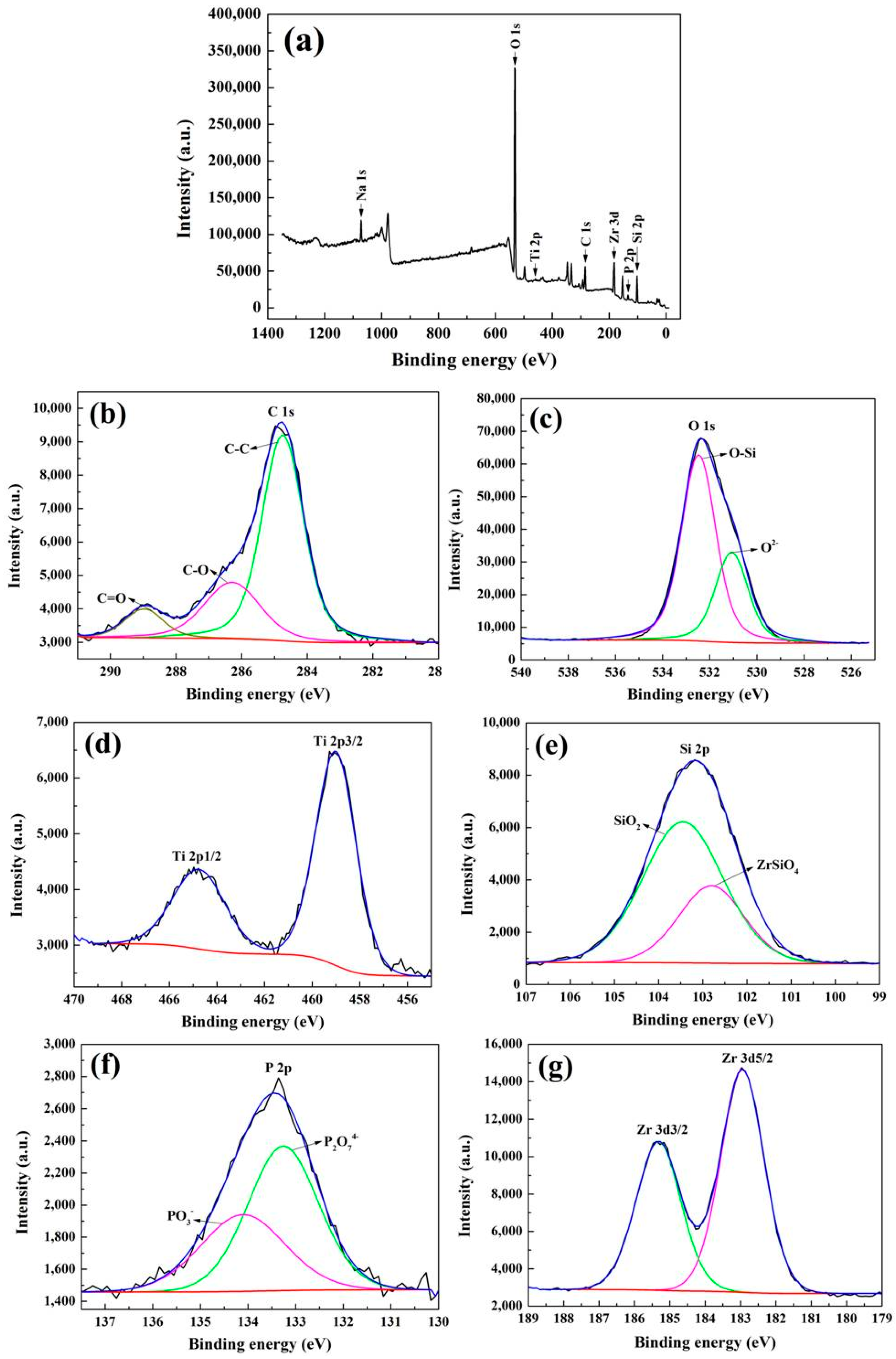
3.2. Phase Composition
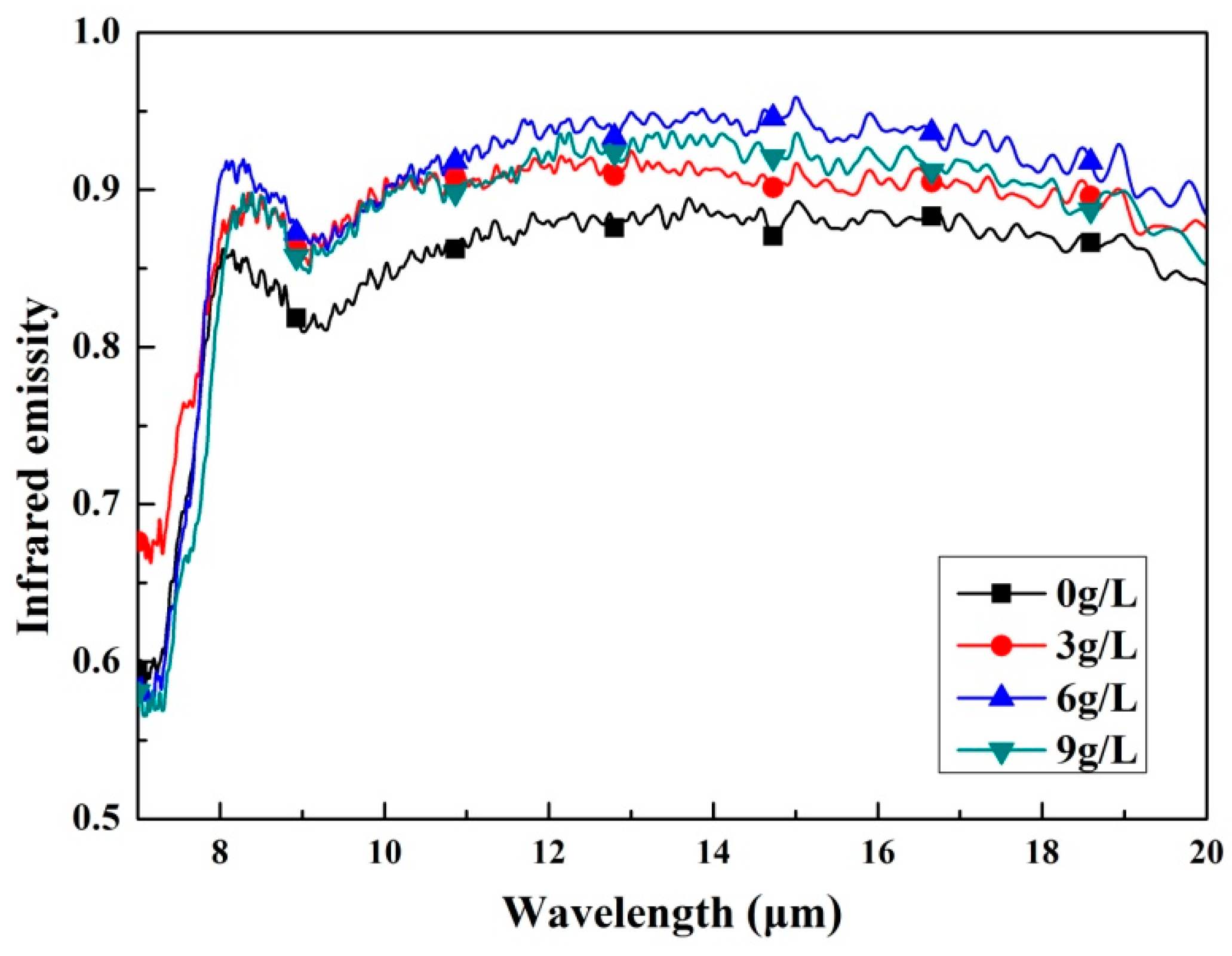
3.3. Infrared Emissivity Characterization
4. Discussion
5. Conclusions
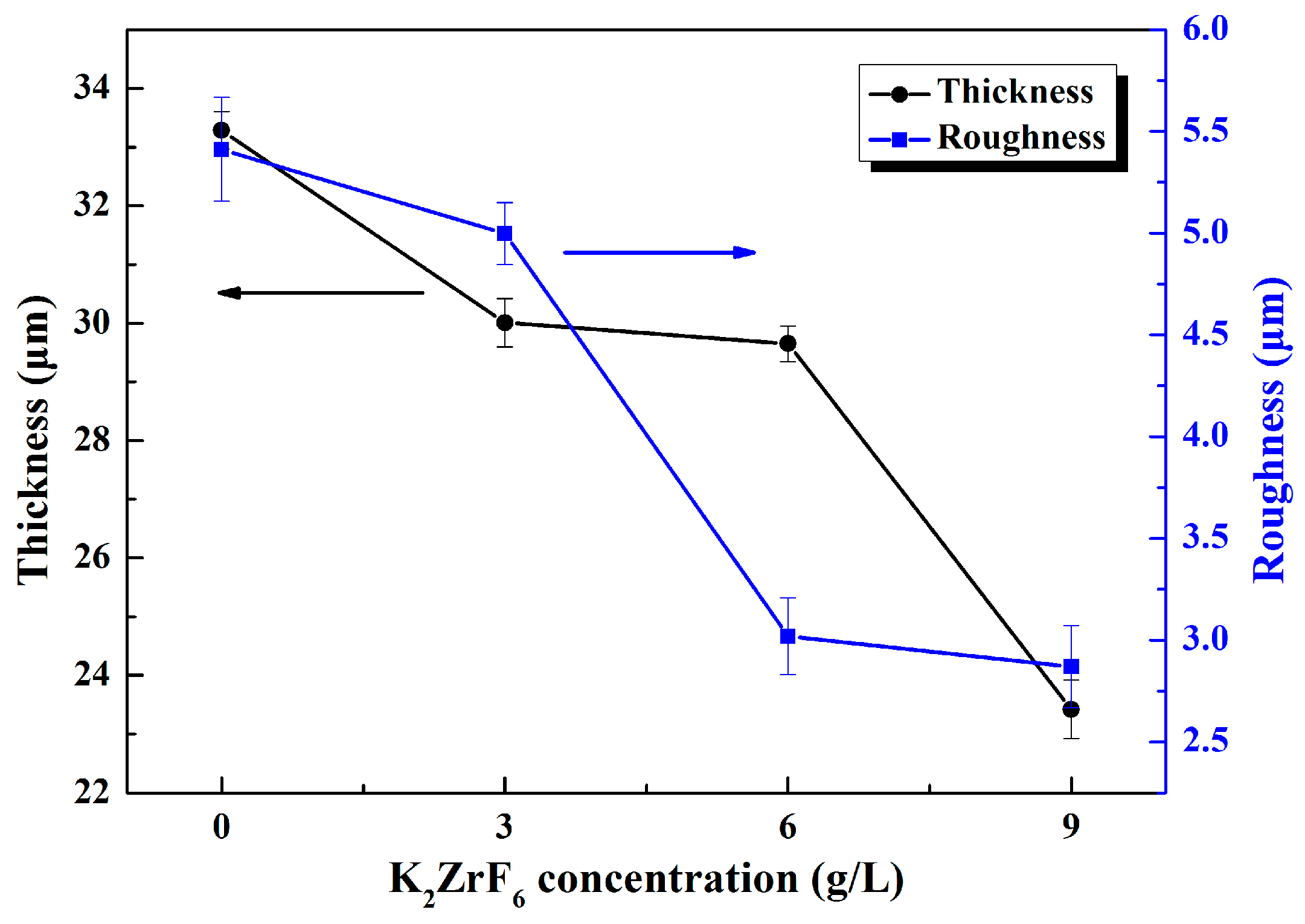
- With the increase of K2ZrF6 concentration, the surface morphologies of the coatings were changed to dense network structure and the number of discharge holes was significantly increased. The decrease ofthe thickness and the surface roughness also indicated that K2ZrF6 inhibited the deposition of silicate on the coatings surface.
- From the XRD analysis, the main phases in the coatings were asanatase-TiO2, rutile-TiO2 together with Ti phases. The absence of any Si-based, P-based and Zr-based species in the XRD peaks identification and the high-resolution spectra of XPS further confirmed that the elements Si, P and Zr existed in the form of amorphous phase.
- The infrared emissivity was drastically improved when the K2ZrF6 was added in the electrolyte. In addition, its highest value was found for the coating with the K2ZrF6 concentration of 6 g/L and the average could reach 0.89 at wavelength of 8–20 μm. It is considered that the doping of Zr4+ and the formation of amorphous ZrO2 enhanced the infrared emissivity of the coatings.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tang, H.; Sun, Q.; Yi, C.G.; Jiang, Z.H.; Wang, F.P. High emissivity coatings on titanium alloy prepared by micro-arc oxidation for high temperature application. J. Mater. Sci. 2012, 47, 2162–2168. [Google Scholar] [CrossRef]
- Tang, H.; Sun, Q.; Xin, T.Z.; Yi, C.G.; Jiang, Z.H.; Wang, F.P. Influence of Co(CH3COO)2 concentration on thermal emissivity of coatings formed on titanium alloy by micro-arc oxidation. Curr. Appl. Phys. 2012, 12, 284–290. [Google Scholar] [CrossRef]
- Rodríguez-Barrero, S.; Fernández-Larrinoa, J.; Azkona, I.; Lacalle, L.N.L.D.; Polvorosa, R. Enhanced Performance of Nanostructured Coatings for Drilling by Droplet Elimination. Mater. Manuf. Process. 2014, 31, 1–10. [Google Scholar] [CrossRef]
- Polvorosa, R.; Suárez, A.; Lacalle, L.N.L.D.; Cerrillo, I.; Wretland, A.; Veiga, F. Tool wear on nickel alloys with different coolant pressures: Comparison of Alloy 718 and Waspaloy. J. Manuf. Process. 2017, 26, 44–56. [Google Scholar] [CrossRef]
- Fernández-Abia, A.I.; Barreiro, J.; Lacalle, L.N.L.D.; González-Madruga, D. Effect of mechanical pre-treatments in the behaviour of nanostructured PVD-coated tools in turning. Int. J. Adv. Manuf. Technol. 2014, 73, 1119–1132. [Google Scholar] [CrossRef]
- Ge, Y.L.; Wang, Y.M.; Zhang, Y.F.; Guo, L.X.; Jia, D.C.; Ouyang, J.H.; Zhou, Y. The improved thermal radiation property of SiC doped micro arc oxidation ceramic coating formed on niobium metal for metal thermal protective system. Surf. Coat. Technol. 2017, 309, 880–886. [Google Scholar] [CrossRef]
- Hong, Z.L.; Ouyang, J.H.; Liu, Z.G.; Wang, Y.M. Microstructure, thermal shock resistance and thermal emissivity of plasma sprayed LaMAl11O19 (M = Mg, Fe) coatings for metallic thermal protection systems. Appl. Surf. Sci. 2013, 271, 52–59. [Google Scholar] [CrossRef]
- Li, Z.; Zheng, F.; Gong, H.Q.; Hu, P.F.; Song, S.L.; Zhen, Q. Study on ZrSiO4-aluminosilicate glass coating with high infrared emissivity and anti-oxidation properties. Compos. Commun. 2017, 4, 16–19. [Google Scholar] [CrossRef]
- Tang, H.; Xin, T.Z.; Sun, Q.; Yi, C.G.; Jiang, Z.H.; Wang, F.P. Influence of FeSO4 concentration on thermal emissivity of coatings formed on titanium alloy by micro-arc oxidation. Appl. Surf. Sci. 2011, 257, 10839–10844. [Google Scholar] [CrossRef]
- Karbowniczek, J.; Muhaffel, F.; Cempura, G.; Cimenoglu, H.; Filemonowicz, A.C. Influence of electrolyte composition on microstructure, adhesion and bioactivity of micro-arc oxidation coatings produced on biomedical Ti6Al7Nb alloy. Surf. Coat. Technol. 2017, 321, 97–107. [Google Scholar] [CrossRef]
- Wang, Y.H.; Liu, Z.G.; Ouyang, J.H.; Wang, Y.M.; Zhou, Y. Influence of electrolyte compositions on structure and high-temperature oxidation resistance of microarc oxidation coatings formed on Ti2AlNb alloy. J. Alloys Compd. 2015, 647, 431–437. [Google Scholar] [CrossRef]
- Qiao, L.P.; Lou, J.; Zhang, S.F.; Qu, B.; Chang, W.H.; Zhang, R.F. The entrance mechanism of calcium and phosphorus elements into micro arc oxidation coatings developed on Ti6Al4V alloy. Surf. Coat. Technol. 2016, 285, 187–196. [Google Scholar] [CrossRef]
- Xu, Y.J.; Yao, Z.P.; Jia, F.Z.; Wang, Y.L.; Jiang, Z.H.; Bu, H.T. Preparation of PEO ceramic coating on Ti alloy and its high temperature oxidation resistance. Curr. Appl. Phys. 2010, 10, 698–702. [Google Scholar] [CrossRef]
- Ezhilselvi, V.; Nithin, J.; Balaraju, J.N.; Subramanian, S. The influence of current density on the morphology and corrosion properties of MAO coatings on AZ31B magnesium alloy. Surf. Coat. Technol. 2016, 288, 221–229. [Google Scholar] [CrossRef]
- Zhang, X.M.; Chen, D.F.; Gong, X.Z.; Yang, S.Q.; Tian, X.B. Modulation effects of K2ZrF6 additive on microstructure and heat resistance of micro-arc oxide coatings fabricated on LY12 aluminum alloy. J. Inorg. Mater. 2010, 25, 865–870. [Google Scholar] [CrossRef]
- Liang, J.; Guo, B.G.; Tian, J.; Liu, H.W.; Zhou, J.F.; Xu, T. Effect of potassium fluoride in electrolytic solution on the structure and properties of microarc oxidation coatings on magnesium alloy. Appl. Surf. Sci. 2005, 252, 345–351. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, L.; Cai, W.; Shan, A.; Jiang, Z.H. Effects of fluoride on the structure and properties of microarc oxidation coating on aluminium alloy. J. Alloys Compd. 2010, 505, 188–193. [Google Scholar] [CrossRef]
- Kang, M.L.; Ko, Y.G.; Dong, H.S. Microstructural characteristics of oxide layers formed on Mg–9 wt % Al–1 wt % Zn alloy via two-step plasma electrolytic oxidation. J. Alloys Compd. 2014, 615, S418–S422. [Google Scholar] [CrossRef]
- Tsunekawa, S.; Aoki, Y.; Habazaki, H. Two-step plasma electrolytic oxidation of Ti–15V–3Al–3Cr–3Sn for wear-resistant and adhesive coating. Surf. Coat. Technol. 2011, 205, 4732–4740. [Google Scholar] [CrossRef]
- Einkhah, F.; Kang, M.L.; Sani, M.A.F.; Yoo, B.; Dong, H.S. Structure and corrosion behavior of oxide layer with Zr compounds on AZ31 Mg alloy processed by two-step plasma electrolytic oxidation. Surf. Coat. Technol. 2014, 238, 75–79. [Google Scholar] [CrossRef]
- Li, Q.B.; Yang, W.B.; Liu, C.C.; Wang, D.A.; Liang, J. Correlations between the growth mechanism and properties of micro-arc oxidation coatings on titanium alloy: Effects of electrolytes. Surf. Coat. Technol. 2017, 316, 162–170. [Google Scholar] [CrossRef]
- Ma, H.; Li, D.; Liu, C.; Huang, Z.; He, D.; Yan, Q.; Liu, P.; Nash, P.; Shen, D.J. An investigation of (NaPO3)6 effects and mechanisms during micro-arc oxidation of AZ31 magnesium alloy. Surf. Coat. Technol. 2015, 266, 151–159. [Google Scholar] [CrossRef]
- Zhu, X.L.; Chen, J.; Scheideler, L.; Reichl, R.; Geis-Gerstorfer, J. Effects of topography and composition of titanium surface oxides on osteoblast responses. Biomaterials 2004, 25, 4087–4103. [Google Scholar] [CrossRef] [PubMed]
- Dang, T.A.; Chou, C.N. Electron spectroscopy for chemical analysis of cool white phosphors coated with SiO2 thin film. J. Electrochem. Soc. 1996, 143, 302–305. [Google Scholar] [CrossRef]
- Li, S.J.; Yang, R.; Niinomi, M.; Hao, Y.L.; Cui, Y.Y. Formation and growth of calcium phosphate on the surface of oxidized Ti–29Nb–13Ta–4.6Zr alloy. Biomaterials 2004, 25, 2525–2532. [Google Scholar] [CrossRef] [PubMed]
- Kaciulis, S.; Mattogno, G.; Napoli, A.; Bemporad, E.; Ferrari, F.; Montenero, A.; Gnappi, G. Surface analysis of biocompatible coatings on titanium. J. Electron Spectrosc. Relat. Phenom. 1998, 95, 61–69. [Google Scholar] [CrossRef]
- Santos, A.D.; Araujo, J.R.; Landi, S.M.; Kuznetsov, A.; Granjeiro, J.M.; Sena, L.A.D.; Achete, C.A. A study of the physical, chemical and biological properties of TiO2coatingsproduced by micro-arc oxidation in a Ca-P-based electrolyte. J. Mater. Sci. Mater. Med. 2014, 25, 1769–1780. [Google Scholar] [CrossRef] [PubMed]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-ray Photoelectron Spectroscopy; Perkin-Elmer Corporation: Eden Prairie, MN, USA, 1992. [Google Scholar]
- Dementjev, A.P.; Ivanova, O.P.; Vasilyev, L.A.; Naumkin, A.V.; Nemirovsky, D.M.; Shalaev, D.Y. Altered layer as sensitive initial chemical state indicator. J. Vac. Sci. Technol. A Vac. Surf. Films 1994, 12, 423–425. [Google Scholar] [CrossRef]
- Shokouhfar, M.; Allahkaram, S.R. Formation mechanism and surface characterization of ceramic composite coatings on pure titanium prepared by micro-arc oxidation in electrolytes containing nanoparticles. Surf. Coat. Technol. 2016, 291, 396–405. [Google Scholar] [CrossRef]
- Kok, W.H.; Sun, X.; Shi, L.; Wong, K.; Mitchell, K.A.R.; Foster, T. Formation of zinc phosphate coatings on AA6061 aluminum alloy. J. Mater. Sci. 2001, 36, 3941–3946. [Google Scholar] [CrossRef]
- Okazaki, Y.; Tateishi, T.; Ito, Y. Corrosion resistance of implant alloys in pseudo physiological solution and role of alloying elements in passive films. Mater. Trans. JIM 1997, 38, 78–84. [Google Scholar] [CrossRef]
- Huang, Z.; Zhou, W.; Tang, X.; Zhu, D.; Luo, F. Effects of substrate roughness on infrared-emissivity characteristics of Au films deposited on Ni alloy. Thin Solid Films 2011, 519, 3100–3106. [Google Scholar] [CrossRef]
- Cheng, X.D.; Min, J.; Zhu, Z.Q.; Ye, W.P. Preparation of high emissivity NiCr2O4 powders with a spinel structure by spray drying. Int. J. Miner. Metall. Mater. 2012, 19, 173–178. [Google Scholar] [CrossRef]
- Chen, X.B.; Liu, L.; Yu, P.Y.; Mao, S.S. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science 2011, 331, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Durdu, S.; Bayramoglu, S.; Demirtas, A.; Usta, M.; Ucisik, A.H. Characterization of AZ31 Mg Alloy coated by plasma electrolytic oxidation. Vacuum 2013, 88, 130–133. [Google Scholar] [CrossRef]
- Zhang, R.F.; Zhang, S.F.; Xiang, J.H.; Zhang, L.H.; Zhang, Y.Q.; Guo, S.B. Influence of sodium silicate concentration on properties of micro arc oxidation coatings formed on AZ91HP magnesium alloys. Surf. Coat. Technol. 2012, 206, 5072–5079. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Shin, S.H.; Lim, H.T.; Koo, B.H. Transformation of plasma electrolytic oxidation coatings from crater to cluster–based structure with increase in DC voltage and the role of ZrO2 nanoparticles. Surf. Coat. Technol. 2017, 311, 383–390. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Shin, S.H.; Hussain, I.; Koo, B.H. Structure and corrosion properties of the two-step PEO coatings formed on AZ91D Mg alloy in K2ZrF6-based electrolyte solution. Surf. Coat. Technol. 2016, 307, 484–490. [Google Scholar] [CrossRef]
- Fernández-Abia, A.I.; Barreiro, J.; Lacalle, L.N.L.D.; Martínez-Pellitero, S. Behavior of austenitic stainless steels at high speed turning using specific force coefficients. Int. J. Adv. Manuf. Technol. 2012, 62, 505–515. [Google Scholar] [CrossRef]


| K2ZrF6 Concetration (g/L) | Element Content of MAO Coatings (at%) | |||||||
|---|---|---|---|---|---|---|---|---|
| O | Na | Si | P | Ti | K | F | Zr | |
| 0 | 71.59 | 1.41 | 19.29 | 2.31 | 5.39 | - | - | - |
| 3 | 71.97 | 0.82 | 18.62 | 2.54 | 5.58 | 0.12 | 0.10 | 0.25 |
| 6 | 72.94 | 0.30 | 12.15 | 4.44 | 7.19 | 0.10 | 0.34 | 2.52 |
| 9 | 72.44 | 0.30 | 10.81 | 4.71 | 6.60 | 0.10 | 1.17 | 3.87 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, G.; Li, Y.; Hu, D.; Xi, Z. Structure and Infrared Emissivity Properties of the MAO Coatings Formed on TC4 Alloys in K2ZrF6-Based Solution. Materials 2018, 11, 254. https://doi.org/10.3390/ma11020254
Gao G, Li Y, Hu D, Xi Z. Structure and Infrared Emissivity Properties of the MAO Coatings Formed on TC4 Alloys in K2ZrF6-Based Solution. Materials. 2018; 11(2):254. https://doi.org/10.3390/ma11020254
Chicago/Turabian StyleGao, Guangrui, Ying Li, Dan Hu, and Zhengping Xi. 2018. "Structure and Infrared Emissivity Properties of the MAO Coatings Formed on TC4 Alloys in K2ZrF6-Based Solution" Materials 11, no. 2: 254. https://doi.org/10.3390/ma11020254
APA StyleGao, G., Li, Y., Hu, D., & Xi, Z. (2018). Structure and Infrared Emissivity Properties of the MAO Coatings Formed on TC4 Alloys in K2ZrF6-Based Solution. Materials, 11(2), 254. https://doi.org/10.3390/ma11020254