Alkali-Silica Reactivity of High Density Aggregates for Radiation Shielding Concrete
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
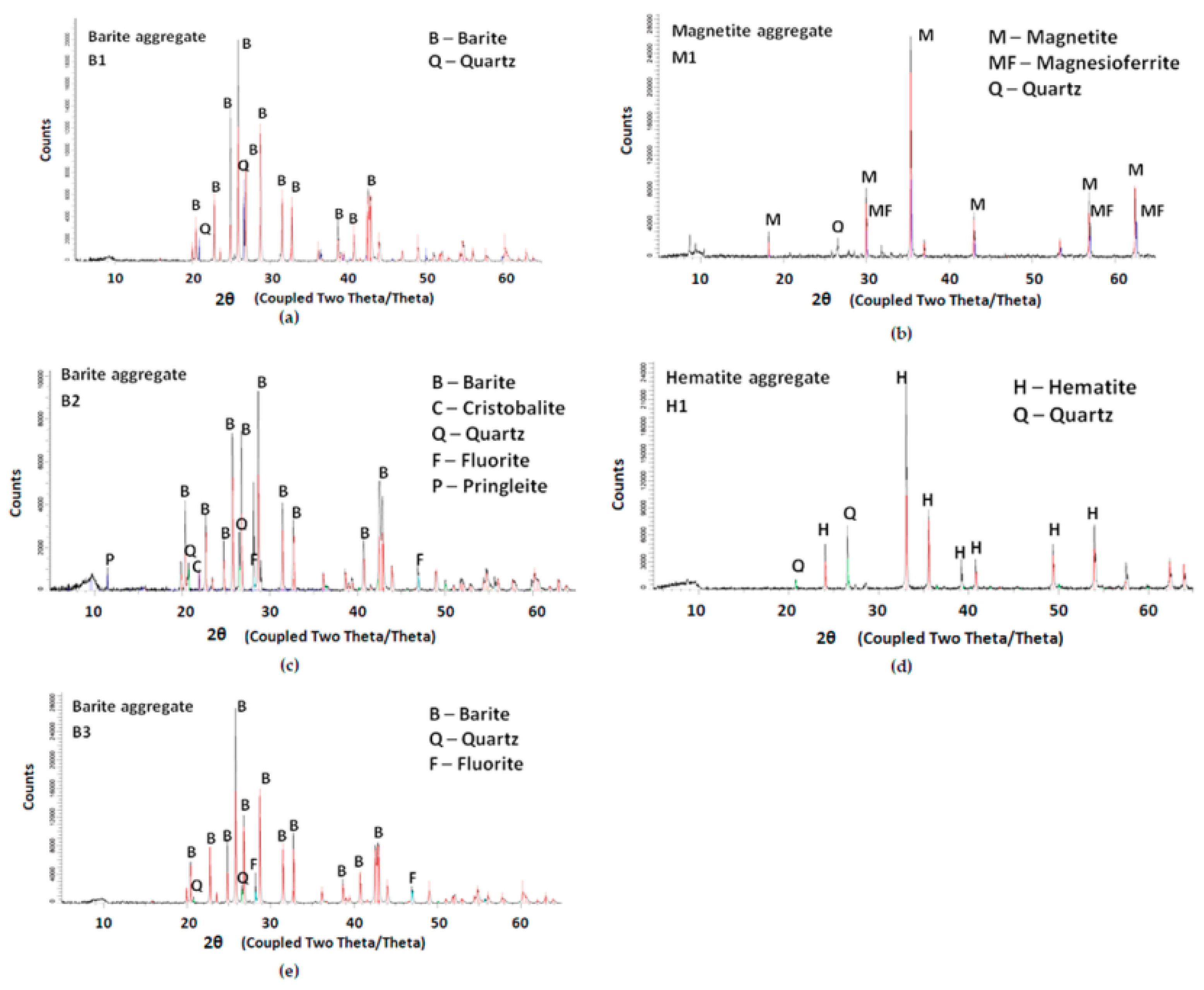
2.2. XRD Analysis
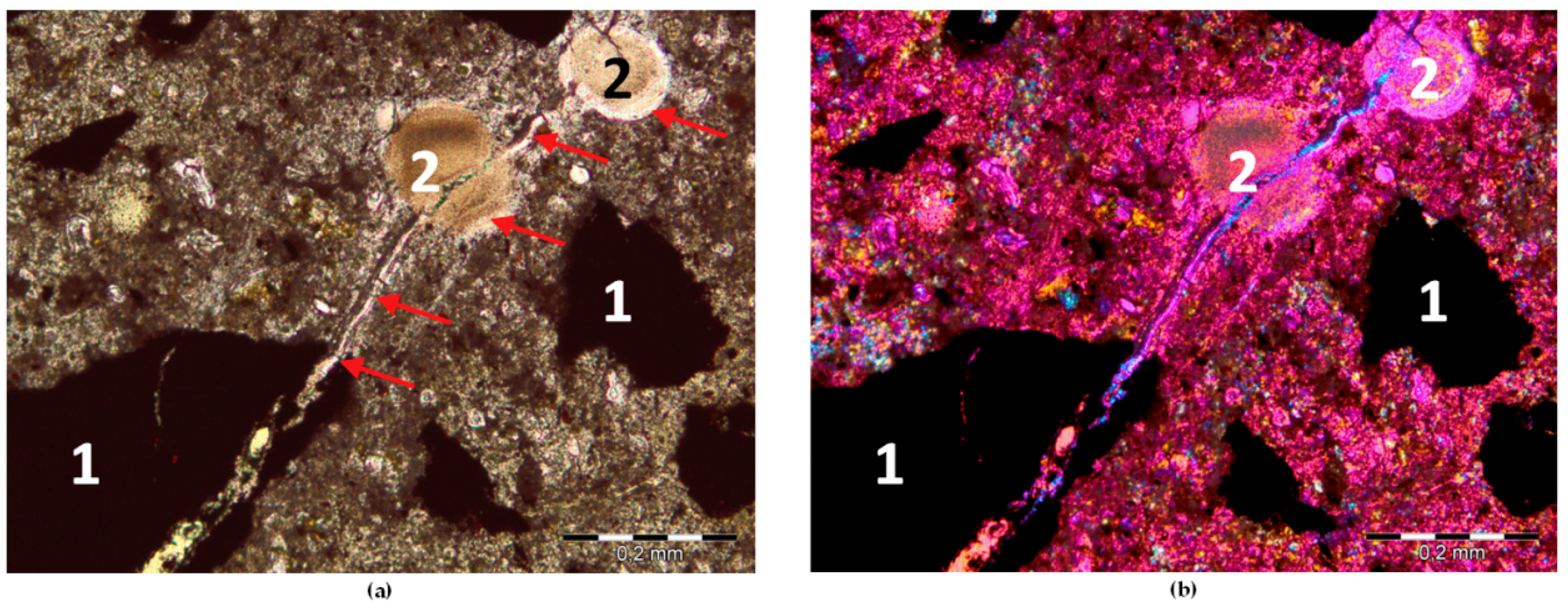
2.3. Thin Section
2.4. ASTM C114
2.5. ASTM C1260 Accelerated Mortar Bar Test
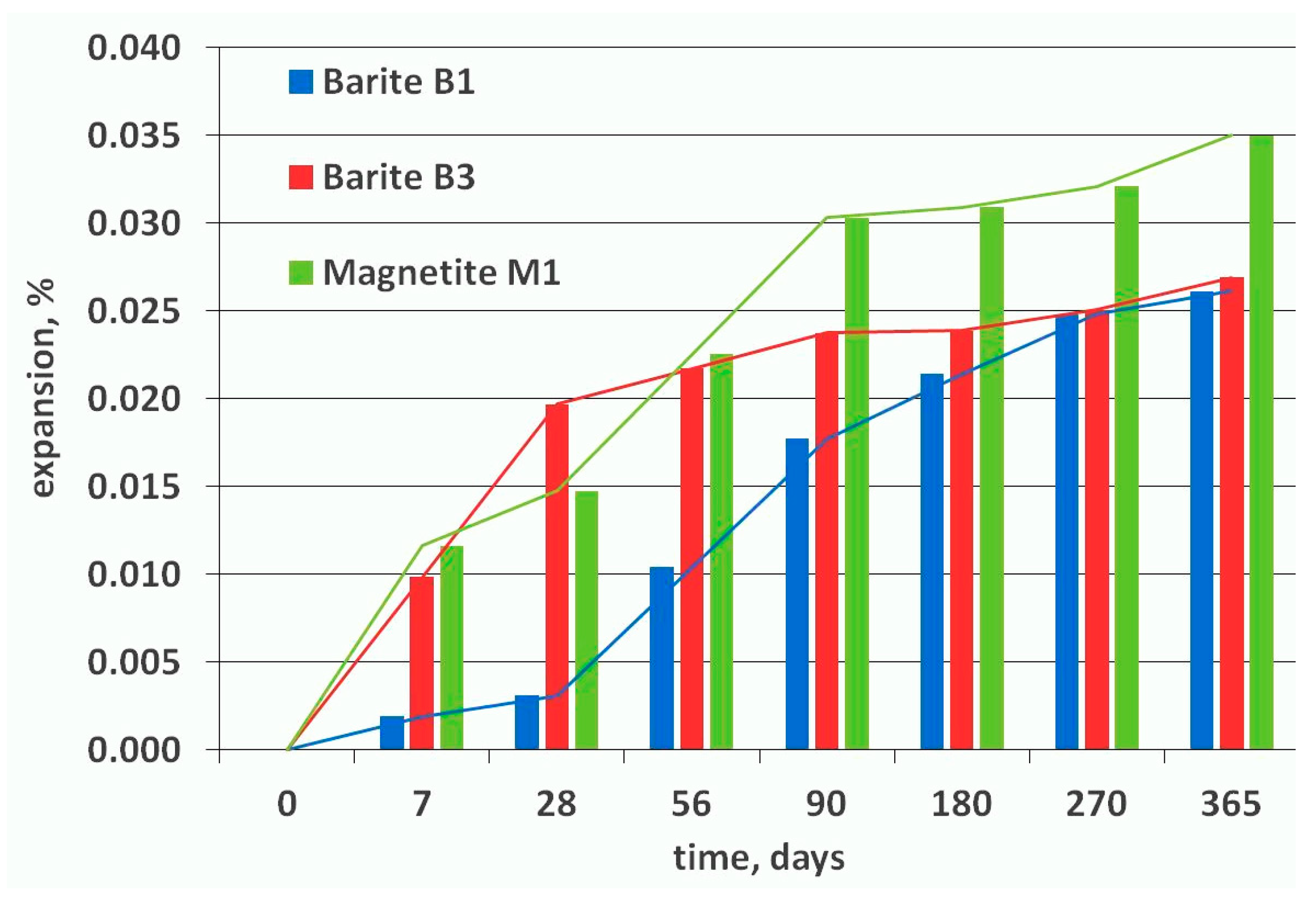
2.6. ASTM C1293 Concrete Prism Expansion Test
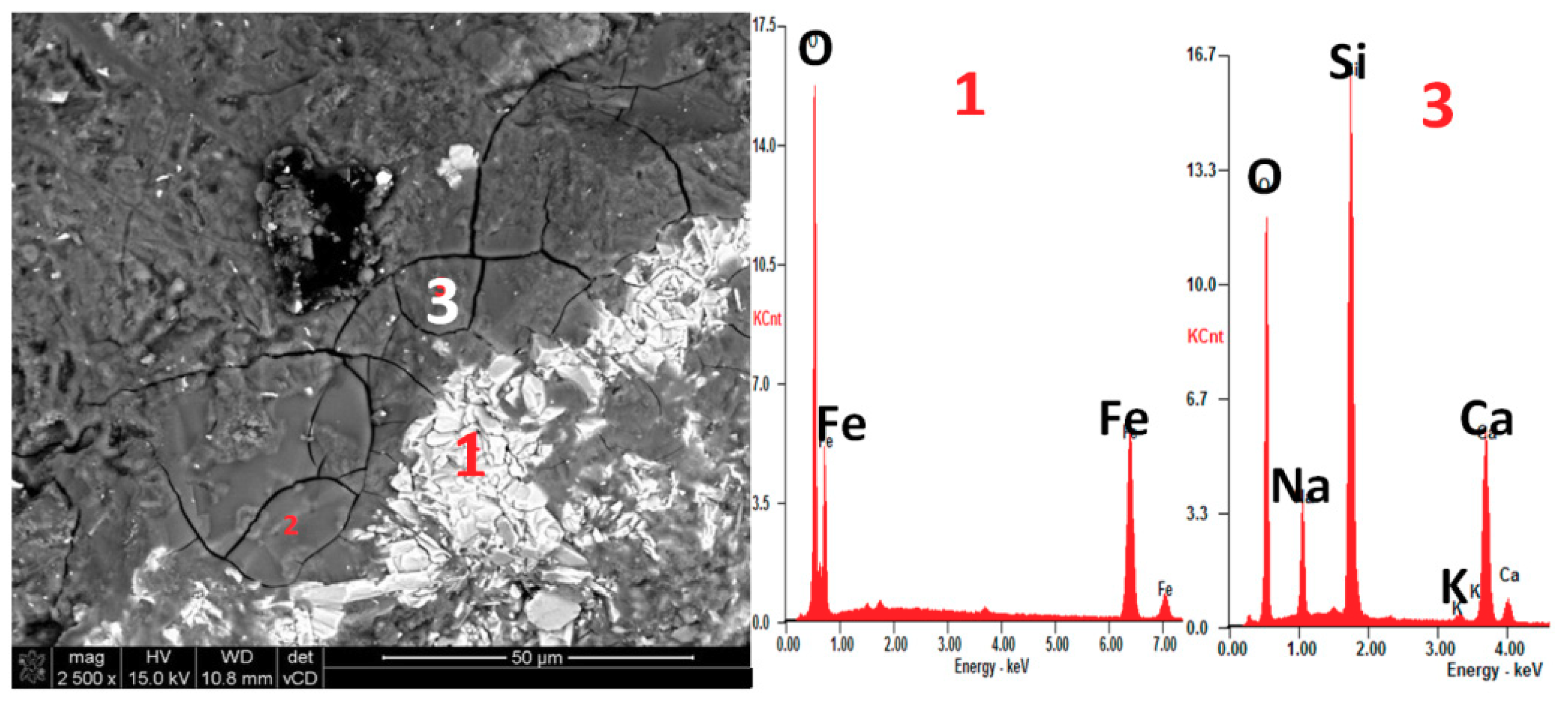
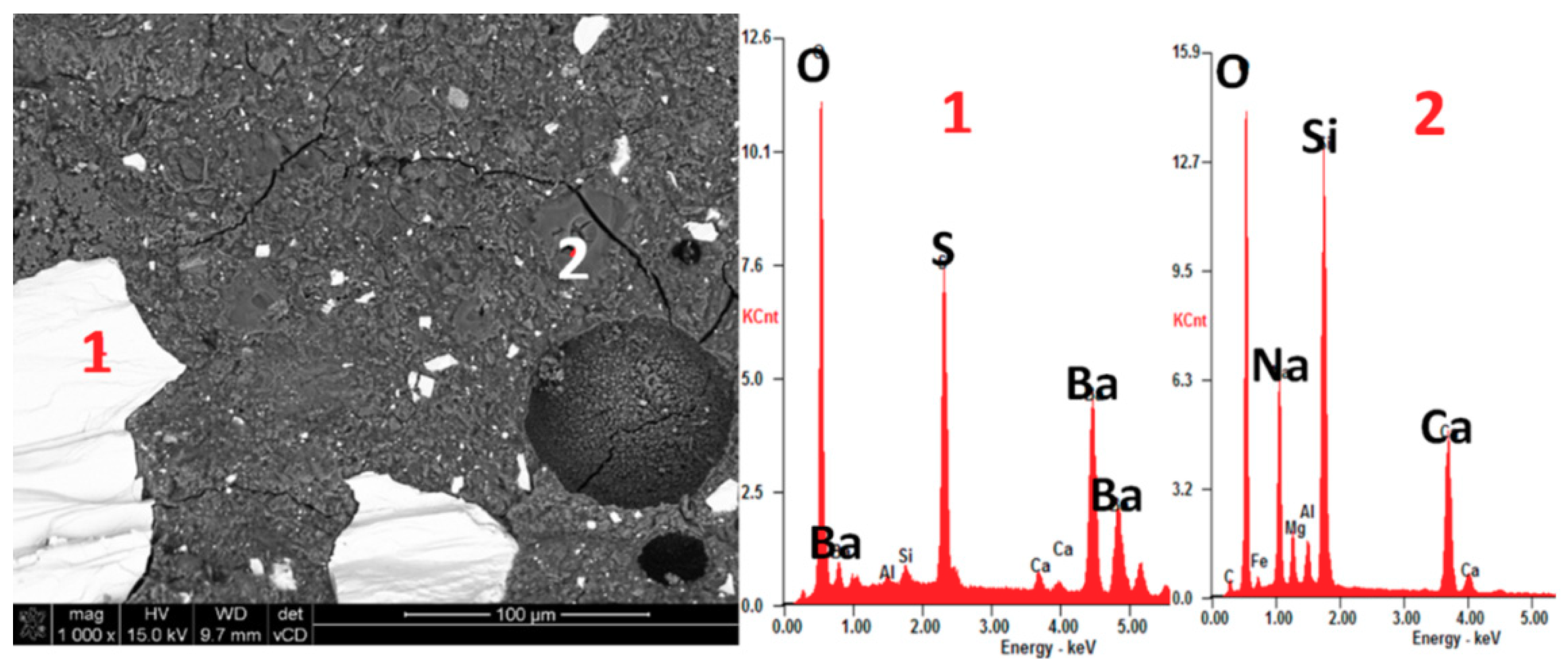
2.7. SEM-EDX Investigation
2.8. Evaluation of Quartz Size in Aggregate Grains
3. Results
3.1. XRD and Petrographic Analysis
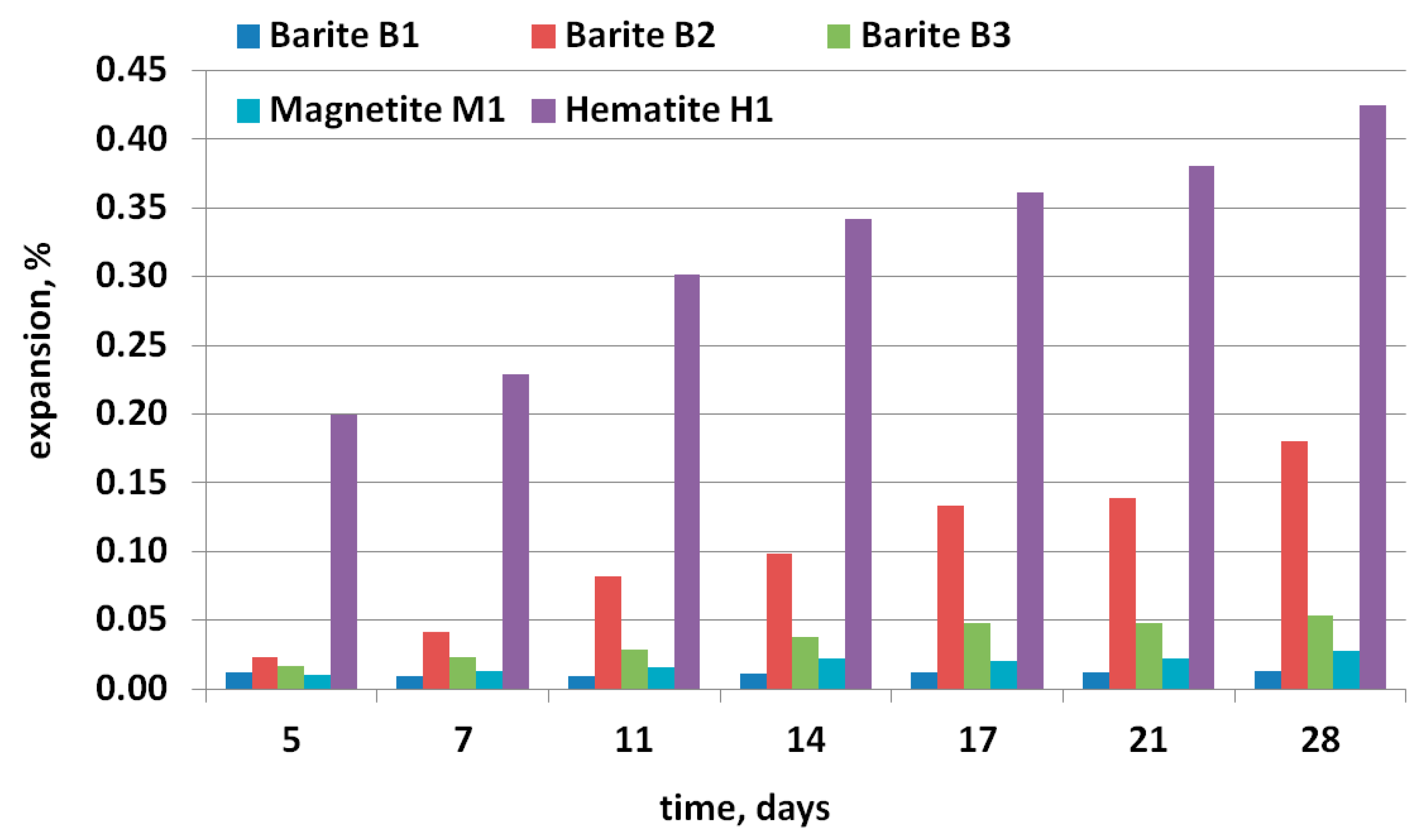
3.2. Expansion Tests and Post-Mortem Microstructure Evaluation
4. Discussion
5. Conclusions
- (1)
- The thin section petrographic analysis and XRD analysis showed that the reactive silica in cristobalite form was present in barite aggregate (B2).
- (2)
- The microcrystalline form of quartz, considered reactive, was identified in hematite aggregate (H1) and barite aggregate (B2).
- (3)
- Reactive quartz grains sized 10 to 60 μm were detected in both high-density aggregates, but the content of the microcrystalline quartz was only 0.13% for magnetite aggregate (M1) and 2.67% for hematite aggregate (H1).
- (4)
- According to the ASTM C1260 test, the highest expansion (0.34%) of mortar bars was found for hematite (H1) aggregate, and the smallest (0.03%) for specimens with magnetite (M1) aggregate.
- (5)
- The presence of ASR gel in mortar bars with hematite (H1) and barite (B2) aggregate was confirmed in post-mortem analysis using SEM. The characteristic ASR composition of Si-Ca-Na gel was proven by the SEM-EDS analysis.
- (6)
- Results obtained from the short-term (ASTM C1260) and the long-term (ASTM C1293) test methods permitted the selection of the high-density weight aggregates from among the options available, with no practical reactivity, required for applications in structures of major importance.
- (7)
- The analysis of thin sections showed that aggregates that come from different mining sites, such as barite, vary significantly between in terms of mineral composition. It is important to consider not only the type of a rock as a criterion for its potential for reactivity, but also its mineralogical composition.
Author Contributions
Funding
Conflicts of Interest
References
- Torrenti, J.M.; Nahas, G. Durability and Safety of Concrete Structures in the Nuclear Context. In Proceedings of the 6th International Conference on Concrete under Severe Conditions (CONSEC’10), Merida, Yucatan, Mexico, 7–9 June 2010; pp. 3–18. [Google Scholar]
- Glinicki, M.A. Długotrwała Funkcjonalność Betonu w Konstrukcjach Osłonowych Elektrowni Jądrowych (Longterm Performance of Concrete in Shielding Structures of Nuclear Power Plants); Institute of Fundamental Technological Research Polish Academy of Sciences: Warszawa, Poland, 2015; p. 64. [Google Scholar]
- Rosseel, T.M.; Maruyama, I.; Le Pape, Y.; Kontani, O.; Giorla, A.B.; Remec, I.; Wall, J.J.; Sircar, M.; Andrade, C.; Ordonez, M. Review of the Current State of Knowledge on the Effects of Radiation on Concrete. J. Adv. Concr. Technol. 2016, 14, 368–383. [Google Scholar] [CrossRef] [Green Version]
- Pignatelli, I.; Kumar, A.; Field, K.G.; Wang, B.; Yu, Y.; Le Pape, Y.; Bauchy, M.; Sant, G. Direct Experimental Evidence for Differing Reactivity Alterations of Minerals Following Irradiation: The Case of Calcite and Quartz. Sci. Rep. 2016, 6, 20155. [Google Scholar] [CrossRef] [PubMed]
- Saouma, V.E.; Hariri-Ardebili, M.A. A Proposed Aging Management Program for Alkali Silica Reactions in a Nuclear Power Plant. Nucl. Eng. Des. 2014, 277, 248–264. [Google Scholar] [CrossRef]
- ASTM. Standard Guide for Reducing the Risk of Deleterious Alkali-Aggregate Reaction in Concrete; ASTM C1778; ASTM: West Conshohocken, PA, USA, 2016. [Google Scholar]
- Recommendations for the Prevention of Damage by Alkali-Aggregate Reactions in New Concrete Structures, State-of-the-Art Report of the RILEM Technical Committee 219-ACS; Springer: Dordrecht, The Netherlands, 2016; p. 168.
- Kurtis, K.E.; Xi, Y.; Glinicki, M.A.; Provis, J.; Giannini, E.R.; Fu, T. Can We Design Concrete to Survive Nuclear Environments? Concr. Int. 2017, 39, 29–35. [Google Scholar]
- Glinicki, M.A.; Antolik, A.; Gawlicki, M. Evaluation of Compatibility of Neutron-Shielding Boron Aggregates with Portland Cement in Mortar. Construct. Build. Mater. 2018, 164, 731–738. [Google Scholar] [CrossRef]
- Lee, S.Y.; Daugherty, A.M.; Broton, D.J. Assessing Aggregates for Radiation-Shielding Concrete, Methods for Petrographic Examination of High-Density and Boron-Bearing Aggregates. Concr. Int. 2013, 35, 31–38. [Google Scholar]
- Ichikawa, T.; Koizumi, H. Possibility of Radiation-Induced Degradation of Concrete by Alkali-Silica Reaction of Aggregates. J. Nucl. Sci. Technol. 2002, 39, 880–884. [Google Scholar] [CrossRef]
- ACI 221R-96. Guide for Use of Normal Weight and Heavyweight Aggregates in Concrete; ACI Committee 221; ACI: Farmington Hills, MI, USA, 2001. [Google Scholar]
- ACI 304.3R-96. Heavyweight Concrete: Measuring, Mixing, Transporting, and Placing; ACI Committee 304; ACI: Farmington Hills, MI, USA, 1997. [Google Scholar]
- PN-C-84088-06. Barite—Barite for Special Construction; British Geological Survey: Nottingham, UK, 1983. (In Polish) [Google Scholar]
- ASTM C1260. Standard Test Method for Potential Alkali Reactivity of Aggregates (Mortar-Bar Method); ASTM International: West Conshohocken, PA, USA, 2014. [Google Scholar]
- ASTM C1293. Standard Test Method for Determination of Length Change of Concrete Due to Alkali-Silica Reaction; ASTM International: West Conshohocken, PA, USA, 2018. [Google Scholar]
- Hagelia, P.; Fernandes, I. On the AAR Susceptibility of Granitic and Quartzitic Aggregates in View of Petrographic Characteristics and Accelerated Testing. In Proceedings of the 14th Conference on Alkali-Aggregate Reaction in Concrete, Austin, TX, USA, 20–25 May 2012; p. 10. [Google Scholar]
- Bérubé, M.A.; Duchesne, J.; Dorion, J.F.; Rivest, M. Laboratory Assessment of Alkali Contribution by Aggregates to Concrete and Application to Concrete Structures Affected by Alkali-Silica Reactivity. Cem. Concr. Res. 2002, 32, 1215–1227. [Google Scholar] [CrossRef]
- Constantiner, D.; Diamond, S. Alkali Release from Feldspars into Pore Solutions. Cem. Concr. Res. 2003, 33, 549–554. [Google Scholar] [CrossRef]
- ASTM C114. Standard Test Methods for Chemical Analysis of Hydraulic Cement; ASTM International: West Conshohocken, PA, USA, 2013. [Google Scholar]
- Jóźwiak-Niedźwiedzka, D.; Gibas, K.; Brandt, A.M.; Glinicki, M.A.; Dąbrowski, M.; Denis, P. Mineral Composition of Heavy Aggregates for Nuclear Shielding Concrete in Relation to Alkali-Silica Reaction. Procedia Eng. 2015, 108, 162–169. [Google Scholar] [CrossRef]
- Jóźwiak-Niedźwiedzka, D. Influence of Blended Cements on the Concrete Resistance to Carbonation. In Proceedings of the 10th International Symposium Brittle Matrix Composites BMC-10, Warsaw, Poland, 15–17 October 2012; pp. 125–134. [Google Scholar]
- Malvar, L.J.; Cline, G.D.; Burke, D.F.; Rollings, R.; Sherman, T.W.; Greene, J. Alkali silica reaction mitigation: State-of-the-Art and recommendations. ACI Mater. J. 2002, 99, 480–489. [Google Scholar]
- Alaejos, P.; Lanza, V. Influence of Equivalent Reactive Quartz Content on Expansion Due to Alkali Silica Reaction. Cem. Concr. Res. 2012, 42, 99–104. [Google Scholar] [CrossRef]
- Jóźwiak-Niedźwiedzka, D.; Jaskulski, R.; Glinicki, M.A. Application of Image Analysis to Identify Quartz Grains in Heavy Aggregates Susceptible to ASR in Radiation Shielding Concrete. Materials 2016, 9, 224. [Google Scholar] [CrossRef] [PubMed]
- Broekmans, M.A.T. Structural Properties of Quartz and Their Potential Role for ASR. Mater. Charact. 2004, 53, 129–140. [Google Scholar] [CrossRef]
- Jóźwiak-Niedźwiedzka, D.; Glinicki, M.A.; Gibas, K.; Jaskulski, R.; Denis, P.; Garbacik, A. Expansion Due to Alkali-Silica Reaction of Heavy Aggregates Used for Nuclear Shielding Concrete. In Proceedings of the 11th International Symposium Brittle Matrix Composites, BMC-11, Warsaw, Poland, 28–30 September 2015; pp. 353–360. [Google Scholar]
- Petrographic Atlas: Characterization of Aggregates Regarding Potential Reactivity to Alkalis. RILEM TC 219-ACS Recommended Guidance AAR-1.2 for Use with the RILEM AAR-1.1 Petrographic Examination Method; Springer: Dordrecht, The Netherlands, 2016; p. 198.
- Kubissa, W.; Glinicki, M.A.; Dąbrowski, M. Permeability Testing of Radiation Shielding Concrete Manufactured at Industrial Scale. Mater. Struct. 2018, 51, 1–15. [Google Scholar] [CrossRef]
- Standard Practice for Determining the Reactivity of Concrete Aggregates and Selecting Appropriate Measures for Preventing Deleterious Expansion in New Concrete Construction; AASHTO (American Association of State and Highway Transportation Officials): Washington, DC, USA, 2008; p. 2.
- Stark, D.; Morgan, B.; Okamoto, P.; Diamond, S. Eliminating or Minimizing Alkali-Silica Reactivity; Strategic Highway Research Program, National Research Council: Washington, DC, USA, 1993; p. 266. [Google Scholar]
- Guide Specification for Concrete Subject to Alkali-Silica Reactions; PCA Portland Cement Association: Skokie, IL, USA, 2007; p. 8.
- de Grosbois, M.; Fontaine, E. Evaluation of the Potential Alkali-Reactivity of Concrete Aggregates: Performance of Testing Methods and Producer’s Point of View. In Proceedings of the 11th International Conference on Alkali-Aggregate Reaction in Concrete, Quebec City, QC, Canada, 11–16 June 2002; pp. 267–276. [Google Scholar]
- Malvar, L.J.; Lenke, L.R. Alkali Silica Reaction Criteria for Accelerated Mortar Bar Tests Based on Field Performance Data. In Proceedings of the 3rd World of Coal Ash, WOCA, Lexington, KY, USA, 4–7 May 2009. [Google Scholar]
- Carles-Gibergues, A.; Cyr, M. Interpretation of Expansion Curves of Concrete Subjected to Accelerated Alkali-Aggregate Reaction (AAR) Tests. Cem. Concr. Res. 2002, 32, 691–700. [Google Scholar] [CrossRef]
- Li, C.; Ideker, J.H.; Thomas, M.D.A. Observations on Using Expanded Clay to Control the Expansion Caused by Alkali-Silica Reaction. In Proceedings of the 15th International Conference AAR, Sao Paulo, Brazil, 3–7 July 2016; p. 10. [Google Scholar]
- Fournier, B.; Bérubé, M.A.; Frenette, J. Laboratory Investigations for Evaluating Potential Alkali-Reactivity of Aggregates and Selecting Preventive Measures Against Alkali–Aggregate Reactions (AAR). What Do They Really Mean? In Proceedings of the 11th International Conference on Alkali-Aggregate Reaction in Concrete, Quebec City, QC, Canada, 11–16 June 2000; pp. 287–296. [Google Scholar]
- Topçu, I.B. Properties of Heavyweight Concrete Produced with Barite. Cem. Concr. Res. 2003, 33, 815–822. [Google Scholar] [CrossRef]
- Castro, N.; Wigum, B.J. Assessment of the Potential Alkali-Reactivity of Aggregates for Concrete by Image Analysis Petrography. Cem. Concr. Res. 2012, 42, 1635–1644. [Google Scholar] [CrossRef]
- Sivathanu Pillai, C.; Santhakumar, A.R.; Poonguzhali, A.; Pujar, M.G.; Ashok Kumar, J.; Preetha, R.; Kamachi Mudali, U. Evaluation of Microstructural and Microchemical Aspects of High Density Concrete Exposed to Sustained Elevated Temperature. Constr. Build. Mater. 2016, 126, 453–465. [Google Scholar] [CrossRef]
- Gencel, O.; Brostow, W.; Ozel, C.; Filiz, M. Concretes Containing Hematite for Use as Shielding Barriers. Mater. Sci. 2010, 16, 249–256. [Google Scholar]
- Jones, A.E.K.; Clark, L.A. The Effects of ASR on the Properties of Concrete and the Implications for Assessment. Eng. Struct. 1998, 20, 785–791. [Google Scholar] [CrossRef]
- Pomaro, B.; Salomoni, V.A.; Gramegna, F.; Prete, G.; Majorana, C.E. Effects of High Energy Atomic Collisions and Material Behaviour of Concrete Barriers in Nuclear Physics Experiments. In Proceedings of the XVIII GIMC Conference, Syracuse, Italy, 21–24 September 2010. [Google Scholar]
- Rajabipour, F.; Giannini, E.; Dunant, C.; Ideker, J.H.; Thomas, M.D.A. Alkali-Silica Reaction: Current Understanding of the Reaction Mechanisms and the Knowledge Gaps. Cem. Concr. Res. 2015, 76, 130–146. [Google Scholar] [CrossRef]
- Tiecher, F.; Florindo, R.N.; Vieira, G.L.; Gomes, M.E.B.; Dal Molin, D.C.C.; Lermen, R.T. Influence of the Quartz Deformation Structures for the Occurrence of the Alkali–Silica Reaction. Materials 2018, 11, 1692. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Alam, M.S.; Ghafoori, N.; Sadiq, R. Role of Solution Concentration, Cement Alkali and Test Duration on Expansion of Accelerated Mortar Bar Test (AMBT). Mater. Struct. Constr. 2016, 49, 1955–1965. [Google Scholar] [CrossRef]



| Aggregate | LOI | Main Mineral Constituent | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | Na2O | K2O | BaO | Mn2O3 | ||
| Barite B1 | 1.10 | 4.88 | 0.15 | 0.26 | 2.39 | 0.14 | 30.23 | 0.04 | 0.00 | 59.26 | 0.06 |
| Barite B2 | 11.54 | 9.26 | 1.06 | 21.31 | 0.17 | 1.28 | 17.68 | 0.05 | 0.23 | 35.48 | 1.15 |
| Barite B3 | 2.05 | 3.56 | 0.73 | 5.52 | 1.58 | 1.05 | 27.59 | 0.04 | 0.01 | 57.23 | 0.04 |
| Hematite H1 | 0.50 | 9.83 | 0.68 | 86.74 | 0.02 | 0.13 | 0.00 | 0.03 | 0.27 | 0.16 | 1.62 |
| Magnetite M1 | −2.42 1 | 3.39 | 0.51 | 93.72 | 1.72 | 1.20 | 0.00 | 0.19 | 0.10 | 0.06 | 0.11 |
| Property | Magnetite M1 | Barite B1 | Barite B3 |
|---|---|---|---|
| slump (mm) | 50 | 130 | 50 |
| temperature (°C) | 22 | 22 | 22 |
| bulk density (kg/m3) | 2776 | 2696 | 2650 |
| air content (%) | 0.8 | 1.0 | 0.8 |
| fc28 (MPa) | 51.4 | 49.1 | 53.4 |
| fc90 (MPa) | 64.9 | 54.5 | 59.5 |
| Soluble Alkali | Hematite H1 | Magnetite M1 |
|---|---|---|
| Na2Osoluble | 4.0 | 105.0 |
| K2Osoluble | 1.5 | 162.0 |
| Na2Oeqv-soluble | 5.0 | 211.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jóźwiak-Niedźwiedzka, D.; Glinicki, M.A.; Gibas, K.; Baran, T. Alkali-Silica Reactivity of High Density Aggregates for Radiation Shielding Concrete. Materials 2018, 11, 2284. https://doi.org/10.3390/ma11112284
Jóźwiak-Niedźwiedzka D, Glinicki MA, Gibas K, Baran T. Alkali-Silica Reactivity of High Density Aggregates for Radiation Shielding Concrete. Materials. 2018; 11(11):2284. https://doi.org/10.3390/ma11112284
Chicago/Turabian StyleJóźwiak-Niedźwiedzka, Daria, Michał A. Glinicki, Karolina Gibas, and Tomasz Baran. 2018. "Alkali-Silica Reactivity of High Density Aggregates for Radiation Shielding Concrete" Materials 11, no. 11: 2284. https://doi.org/10.3390/ma11112284





