The Potential of Using Immobilized Xylanases to Enhance the Hydrolysis of Soluble, Biomass Derived Xylooligomers
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate Pre-treatment and Water Soluble Component/Oligosaccharide Production
2.2. Sodium Alginate Beads Production and Activation
2.3. Enzyme Immobilization
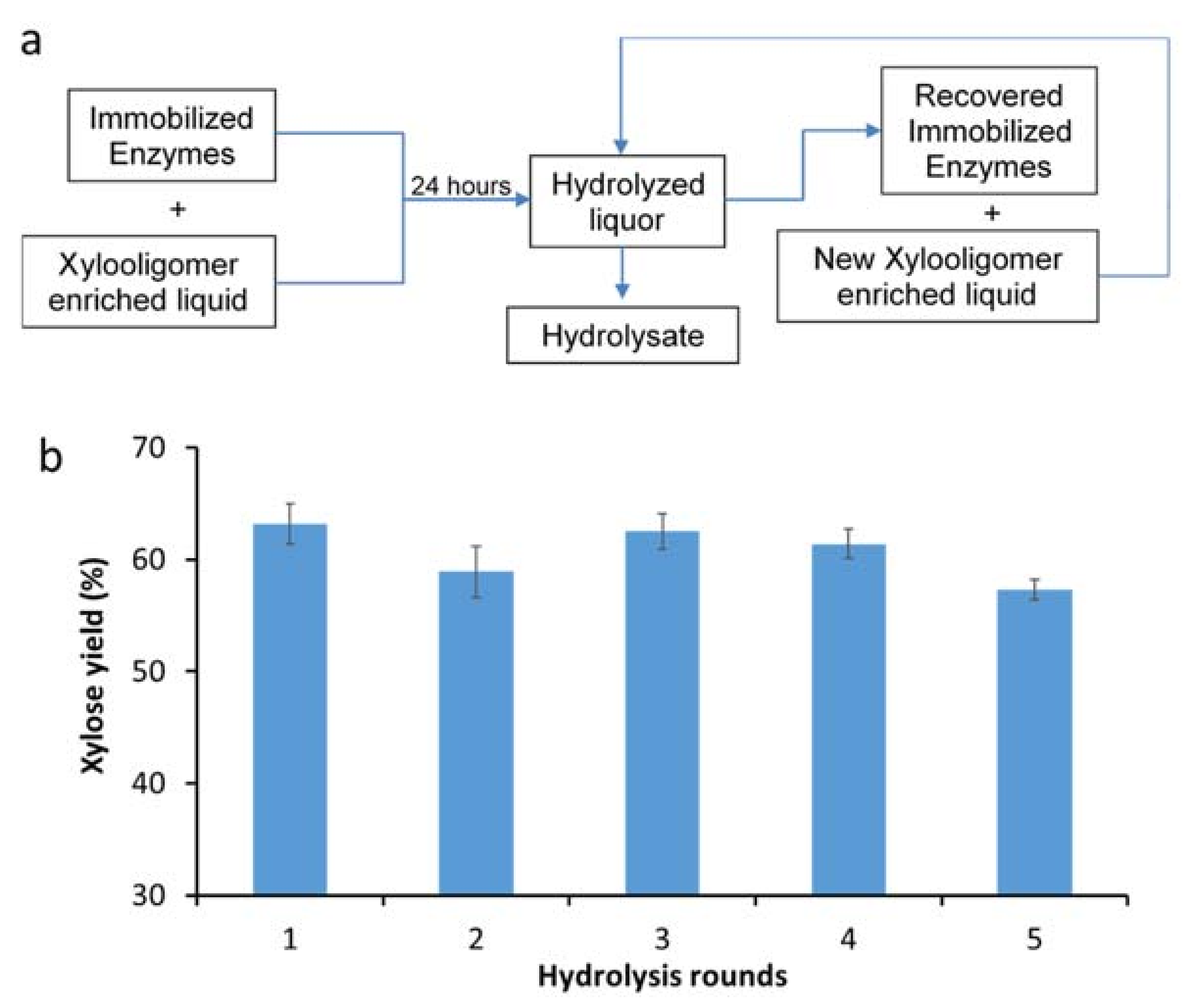
2.4. Enzyme Hydrolysis and Recycle
2.5. Enzyme Activity Assay
3. Results and Discussion
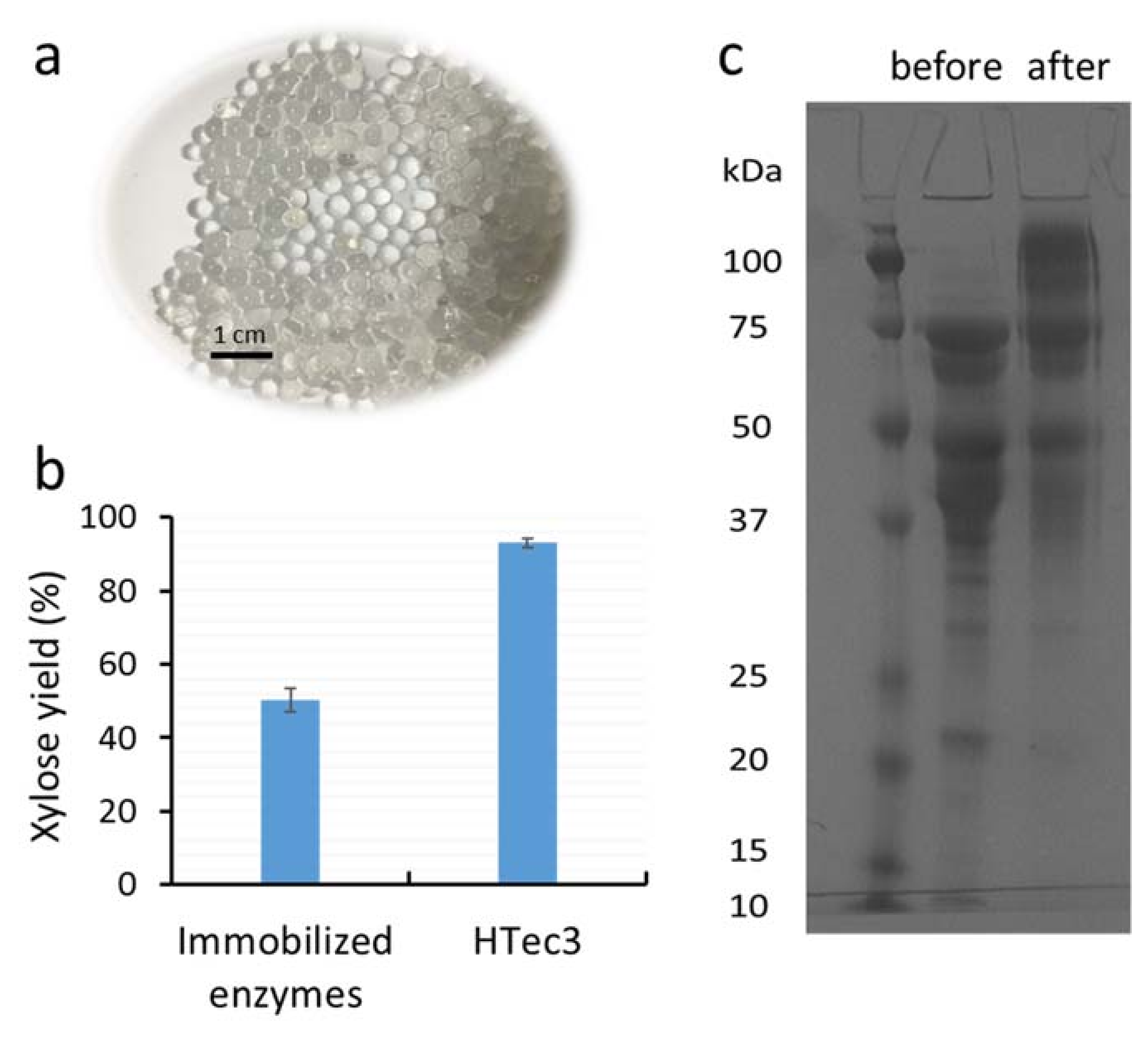
3.1. Covalent Surface Immobilization of Xylanases on Alginate Beads for Xylooligomer Hydrolysis
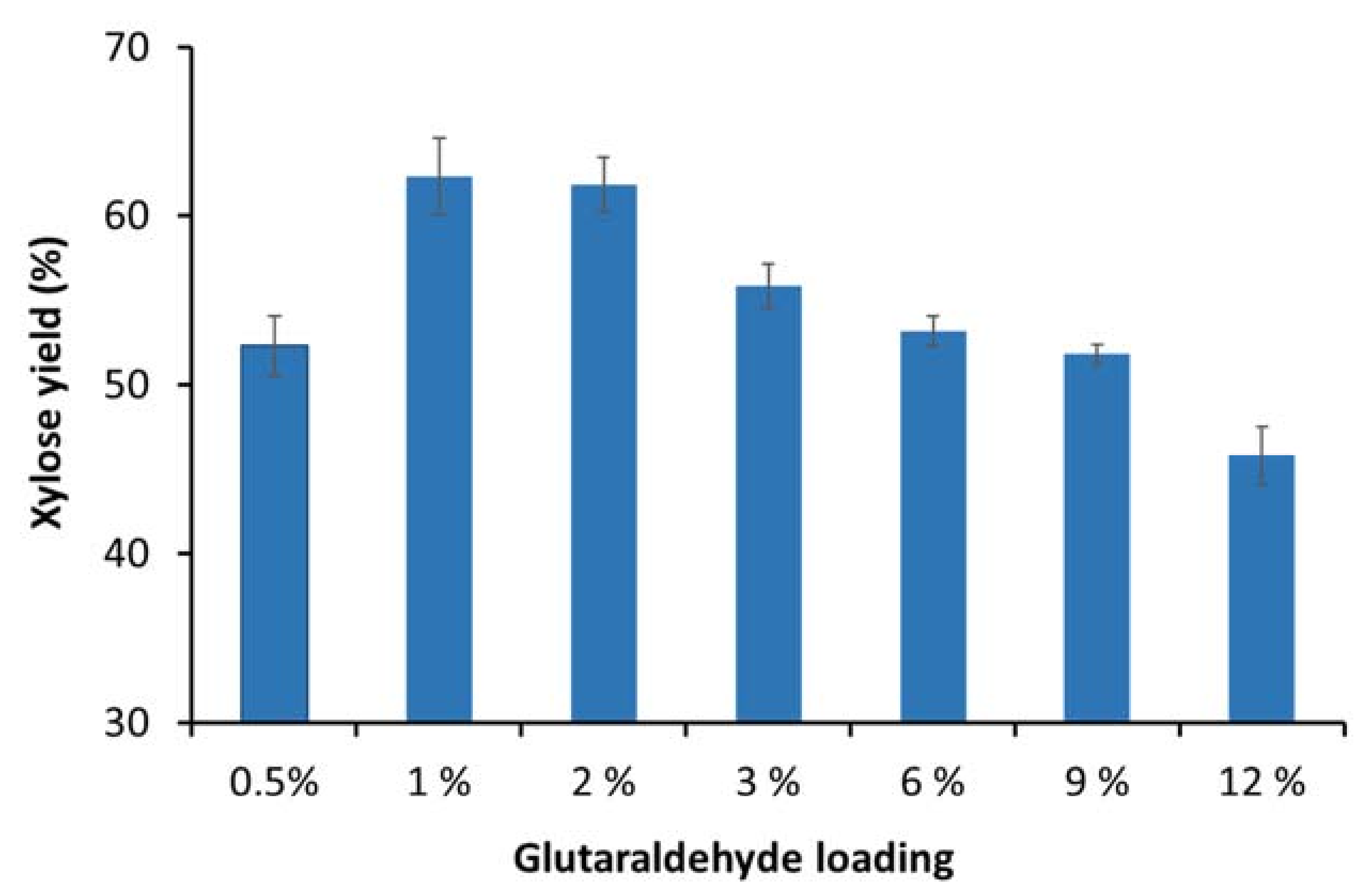
3.2. Optimization of the Glutaraldehyde Loading Concentration for Enzyme Immobilization
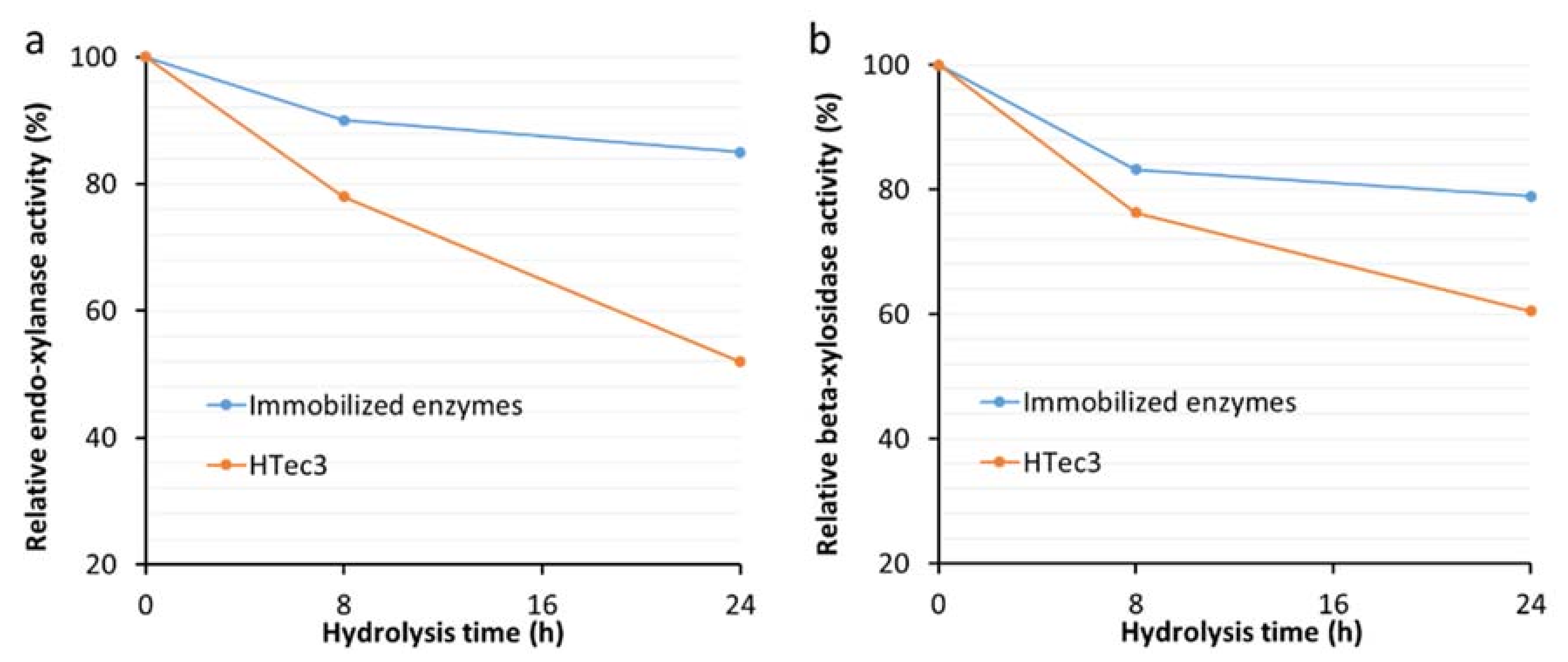
3.3. The Stability of Major Xylanases Before and After Immobilization
3.4. Assessing the Recyclability of Immobilized Xylanases over Multiple Rounds of Hydrolysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sims, R.E.H.; Mabee, W.; Saddler, J.N.; Taylor, M. An overview of second generation biofuel technologies. Bioresour. Technol. 2010, 101, 1570–1580. [Google Scholar] [CrossRef] [PubMed]
- Ragauskas, A.; Williams, C.; Davison, B.; Britovsek, G.; Cairney, J.; Eckert, C.; Frederick, W.; Hallett, J.; Leak, D.; Liotta, C.; et al. The path forward for biofuels and biomaterials. Science 2006, 311, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Alvira, P.; Tomas-Pejo, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Arantes, V.; Pribowo, A.; Gourlay, K.; Saddler, J.N. Substrate factors that influence the synergistic interaction of AA9 and cellulases during the enzymatic hydrolysis of biomass. Energy Environ. Sci. 2014, 7, 2308–2315. [Google Scholar] [CrossRef]
- Shekiro, J.; Kuhn, E.M.; Selig, M.J.; Nagle, N.J.; Decker, S.R.; Elander, R.T. Enzymatic conversion of xylan residues from dilute acid-pretreated corn stover. Appl. Biochem. Biotechnol. 2012, 168, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Kumar, R.; Tsai, Y.; Elander, R.T.; Wyman, C.E. Xylose yields and relationship to combined severity for dilute acid post-hydrolysis of xylooligomers from hydrothermal pretreatment of corn stover. Green Chem. 2015, 17, 394–403. [Google Scholar] [CrossRef]
- Hu, J.; Davies, J.; Mok, Y.K.; Gene, B.; Lee, Q.F.; Arato, C.; Saddler, J.N. Enzymatic hydrolysis of industrial derived xylo-oligomers to monomeric sugars for potential chemical/biofuel production. ACS Sustain. Chem. Eng. 2016, 12, 7130–7136. [Google Scholar] [CrossRef]
- Madakbas, S.; Danis, Ö.; Demir, S.; Kahraman, M.V. Xylanase immobilization on functionalized polyaniline support by covalent attachment. Starch 2013, 65, 146–150. [Google Scholar] [CrossRef]
- Pal, A.; Khanum, F. Covalent immobilization of xylanase on the surface of alginate-glutaraldehyde beads decreases the ‘catalytic efficiency’ but provides ‘low temperature stabilization’ effect. J. Biochem. Technol. 2012, 3, 409–413. [Google Scholar]
- Kumar, P.; Gupta, A.; Dhakate, S.R.; Mathur, R.B.; Nagar, S.; Gupta, V.K. Covalent immobilization of xylanase produced from Bacillus pumilus SV-85S on electrospun polymethyl methacrylate nanofiber membrane. Biotechnol. Appl. Biochem. 2013, 60, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Kuhad, R.C. Immobilization of xylanase from Bacillus pumilus strain MK001 and its application in production of xylo-oligosaccharides. Appl. Biochem. Biotechnol. 2007, 142, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Naby, M.A. Immobilization of Aspergillus niger NRC 107 xylanase and β-xylosidase, and properties of the immobilized enzymes. Appl. Biochem. Biotechnol. 1992, 38, 69–81. [Google Scholar] [CrossRef]
- Bura, R.; Bothast, R.J.; Mansfield, S.D.; Saddler, J.N. Optimization of SO2-catalyzed steam pretreatment of corn fiber for ethanol production. Appl. Biochem. Biotechnol. 2003, 105, 319–335. [Google Scholar] [CrossRef]
- Hu, J.; Arantes, V.; Saddler, J.N. The enhancement of enzymatic hydrolysis of lignocellulosic substrates by the addition of accessory enzymes such as xylanase: Is it an additive or synergistic effect? Biotechnol. Biofuels 2011, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.; Hilterhaus, L. Evaluation of immobilized enzymes for industrial applications. Chem. Soc. Rev. 2013, 42, 6236–6249. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.C.; Bothast, R.J. Production, purification, and characterization of a highly glucose-tolerant novel beta-glucosidase from Candida peltata. Appl. Environ. Microbiol. 1996, 62, 3165–3170. [Google Scholar] [PubMed]
- Hu, J.; Arantes, V.; Pribowo, A.; Saddler, J.N. The synergistic action of accessory enzymes enhances the hydrolytic potential of a “cellulase mixture” but is highly substrate specific. Biotechnol. Biofuels 2013, 6, 112. [Google Scholar] [CrossRef] [PubMed]
- Semenova, M.V.; Drachevskaya, M.I.; Sinitsyna, O.A.; Gusakov, A.V.; Sinitsyn, A.P. Isolation and properties of extracellular β-xylosidases from fungi Aspergillus japonicus and Trichoderma reesei. Biochemistry 2009, 74, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Shallom, D.; Shoham, Y. Microbial hemicellulases. Curr. Opin. Microbiol. 2003, 6, 219–228. [Google Scholar] [CrossRef]
- Mohamad, N.R.; Marzuki, N.H.C.; Buang, N.A.; Huyop, F.; Wahab, R.A. An overview of technologies for immobilization of enzymes and surface analysis techniques for immobilized enzymes. Biotechnol. Equip. 2015, 29, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Saha, B. Hemicellulose bioconversion. J. Ind. Microbiol. Biotechnol. 2003, 30, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, H.R.; Pedersen, S.; Jorgensen, C.T.; Meyer, A.S. Enzymatic hydrolysis of wheat arabinoxylan by a recombinant “minimal” enzyme cocktail containing beta-xylosidase and novel endo-1,4-β-xylanase and α -(l)-arabinofuranosidase activities. Biotechnol. Prog. 2007, 23, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Saddler, J.N. Why does GH10 xylanase have better performance than GH11 xylanase for the deconstruction of pretreated biomass? Biomass Bioenergy 2018, 110, 13–16. [Google Scholar] [CrossRef]
- Zhai, R.; Hu, J.; Saddler, J.N. What are the major components in steam pretreated lignocellulosic biomass that inhibit the efficacy of cellulase enzyme mixtures? ACS Sustain. Chem. Eng. 2016, 4, 3429–3436. [Google Scholar] [CrossRef]
| Immobilization Method | Immobilization Support | Immobilization Yield (%) | Recyclability | Reference |
|---|---|---|---|---|
| Covalent binding | Polyaniline via glutaraldehyde | N/A | >72% of its original activity after 15 recycling rounds | [8] |
| Glutaraldehyde activated alginate beads | >91 | >85% of its original activity after five recycling rounds | [9] | |
| Polymethyl methacrylate (PMMA) nanofiber membrane (NFM) activated with glutaraldehyde | 90 | ~80% of its original activity after 11 recycling rounds | [10] | |
| HP-20 (styrene-divynilbenzene adsorbent resin) with glutaraldehyde | 42 | ~70% of its original activity after 11 recycling rounds | [11] | |
| Ionic binding | Q-sepharose | 45 | ~70% of its original activity after 11 recycling rounds | [11] |
| Dowex-50W | 24.5–47.4 | 18% of its original activity after 40 days | [12] | |
| Physical adsorption | Chitin | 35 | <50% of its original activity after four recycling rounds | [11] |
| Tannin-chitosan | 37.7–69.3 | 33% of its original activity after 40 days | [12] | |
| Entrapment | Gelatin | 40 | <50% of its original activity after four recycling rounds | [11] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, J.; Davies, J.; Mok, Y.K.; Arato, C.; Saddler, J.N. The Potential of Using Immobilized Xylanases to Enhance the Hydrolysis of Soluble, Biomass Derived Xylooligomers. Materials 2018, 11, 2005. https://doi.org/10.3390/ma11102005
Hu J, Davies J, Mok YK, Arato C, Saddler JN. The Potential of Using Immobilized Xylanases to Enhance the Hydrolysis of Soluble, Biomass Derived Xylooligomers. Materials. 2018; 11(10):2005. https://doi.org/10.3390/ma11102005
Chicago/Turabian StyleHu, Jinguang, Joshua Davies, Yiu Ki Mok, Claudio Arato, and John N. Saddler. 2018. "The Potential of Using Immobilized Xylanases to Enhance the Hydrolysis of Soluble, Biomass Derived Xylooligomers" Materials 11, no. 10: 2005. https://doi.org/10.3390/ma11102005
APA StyleHu, J., Davies, J., Mok, Y. K., Arato, C., & Saddler, J. N. (2018). The Potential of Using Immobilized Xylanases to Enhance the Hydrolysis of Soluble, Biomass Derived Xylooligomers. Materials, 11(10), 2005. https://doi.org/10.3390/ma11102005






