Blood Vessel Formation and Bone Regeneration Potential of the Stromal Vascular Fraction Seeded on a Calcium Phosphate Scaffold in the Human Maxillary Sinus Floor Elevation Model
Abstract
:1. Introduction
2. Materials and Methods
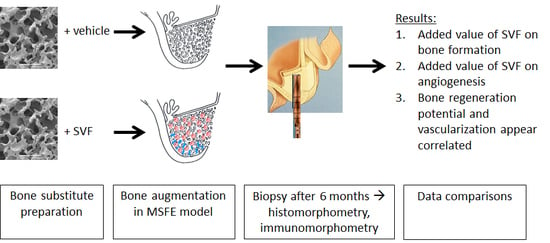
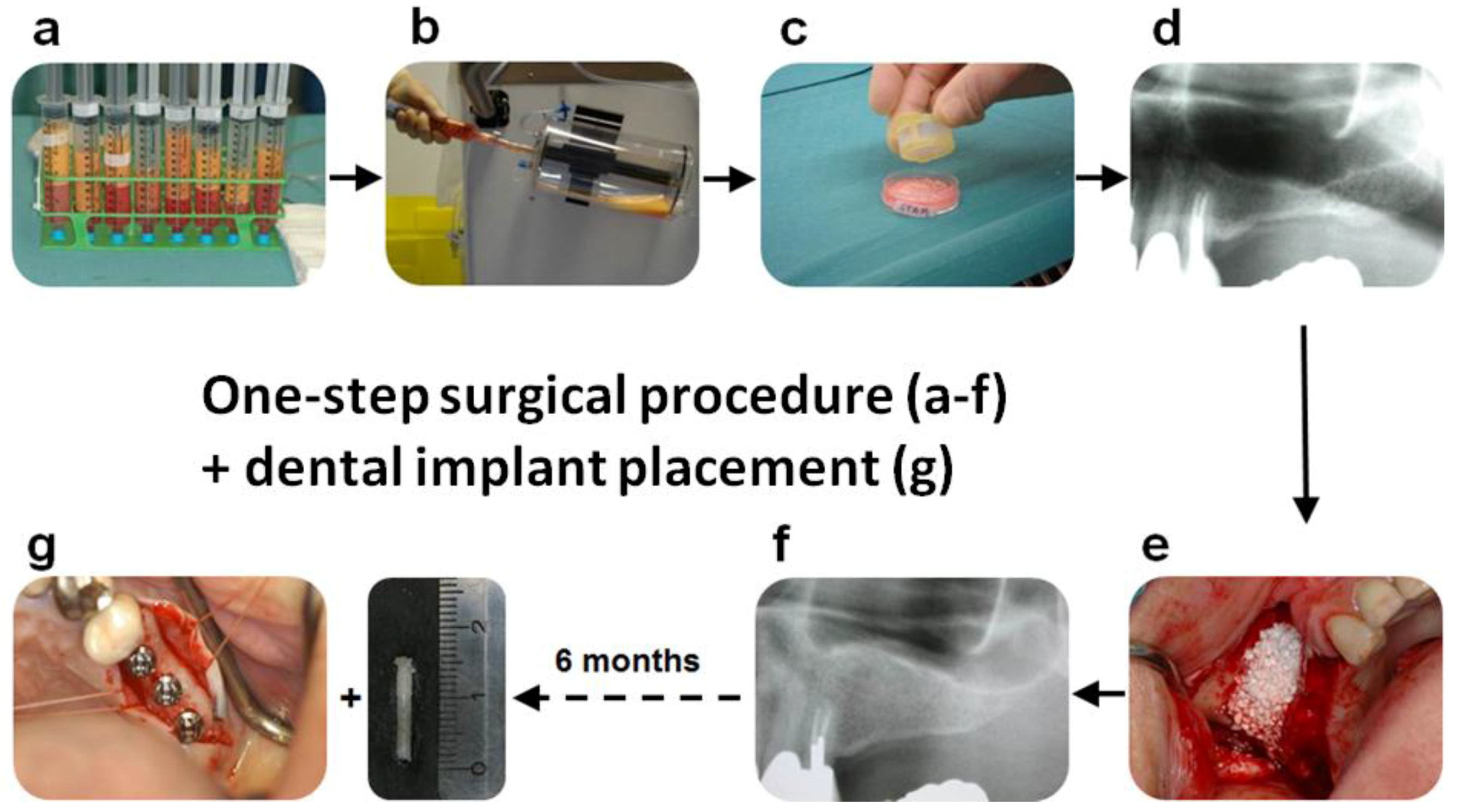
2.1. Clinical Study Outline
2.2. Biopsy Processing and Evaluation
2.3. Histology and Histomorphometry
2.4. Immunohistochemistry
2.5. Statistics
3. Results
3.1. Clinical Evaluation and Implant Survival
3.2. Data Analysis
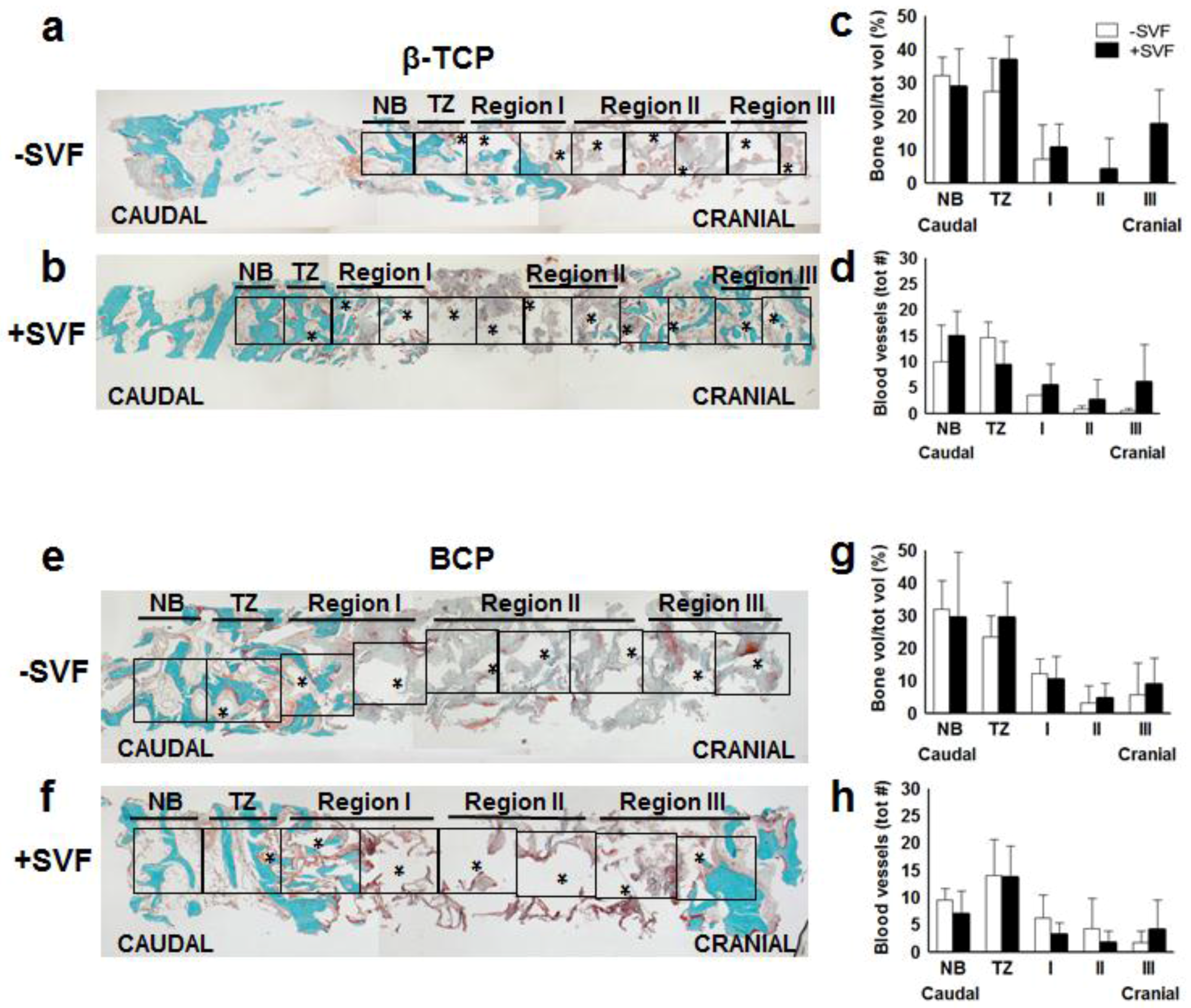
3.3. Quantitative Histomorphometric Evaluation
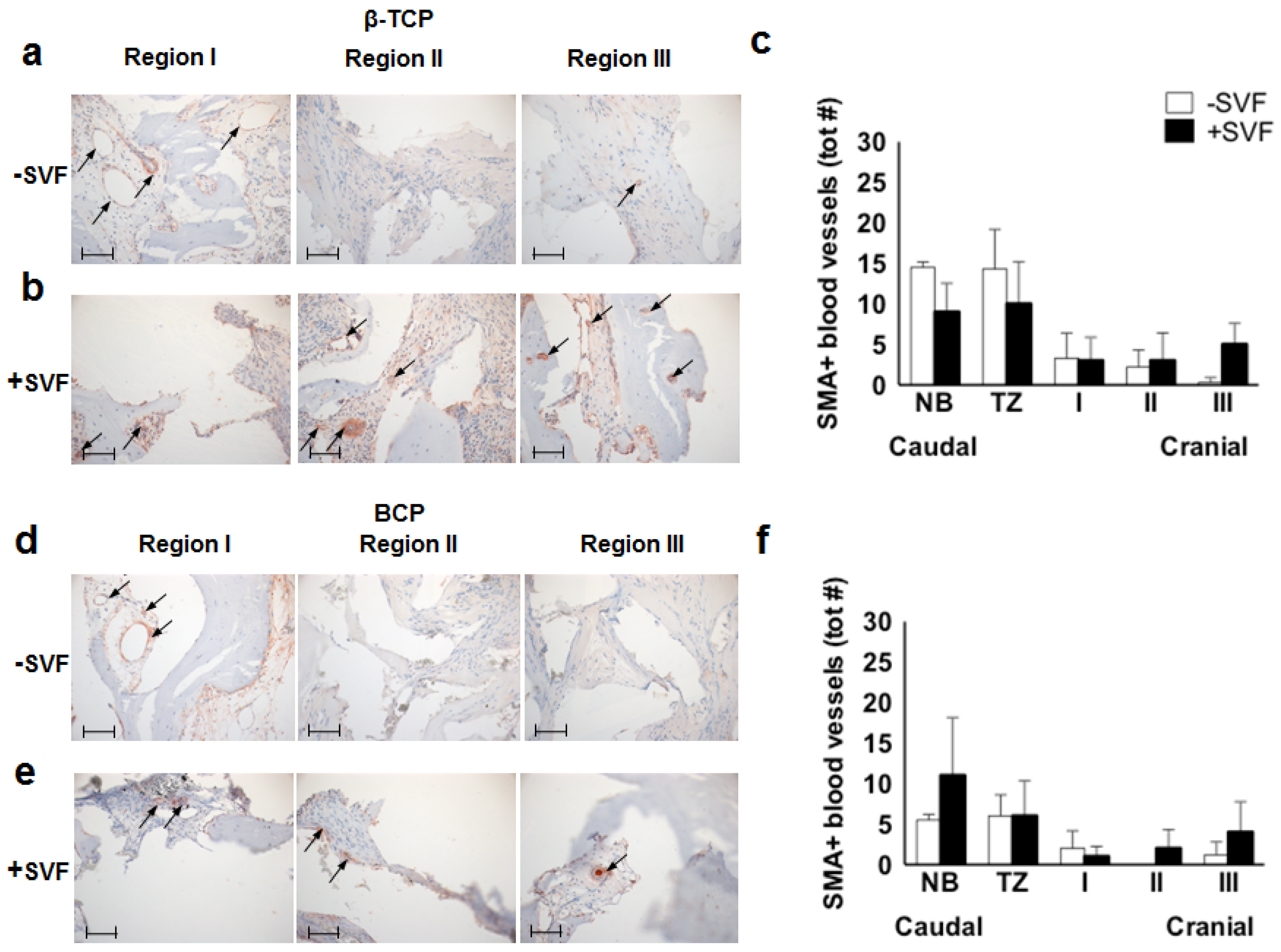
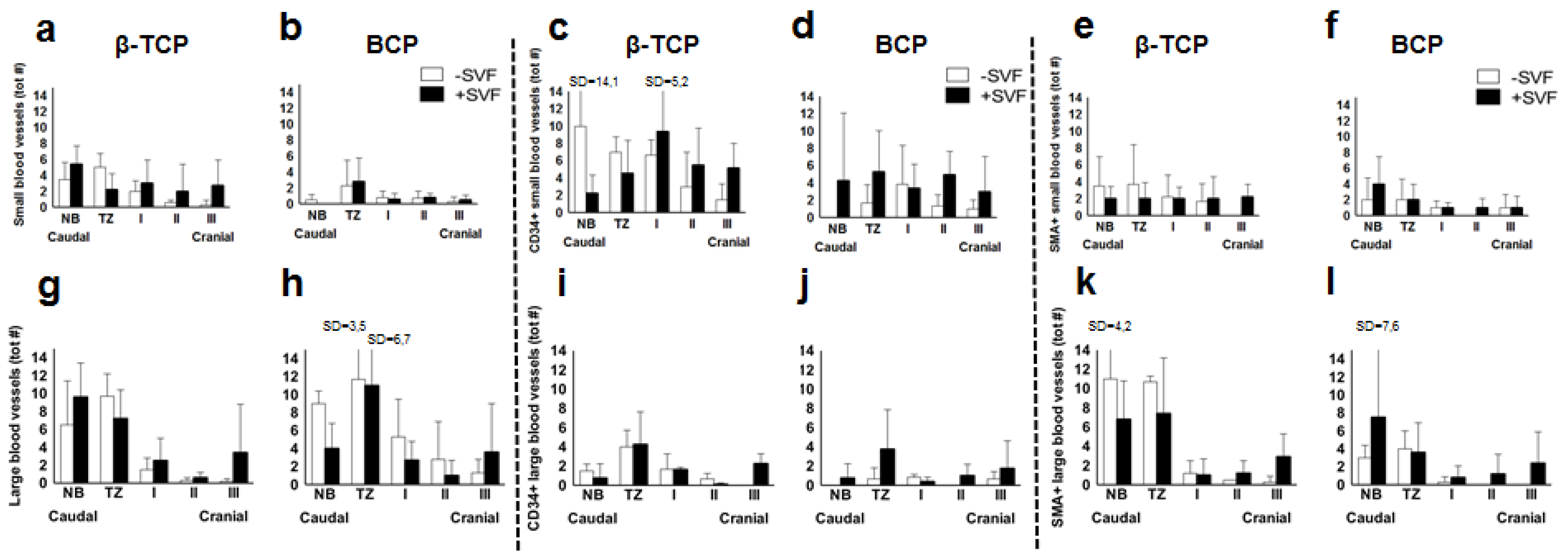
3.4. Immunohistochemistry
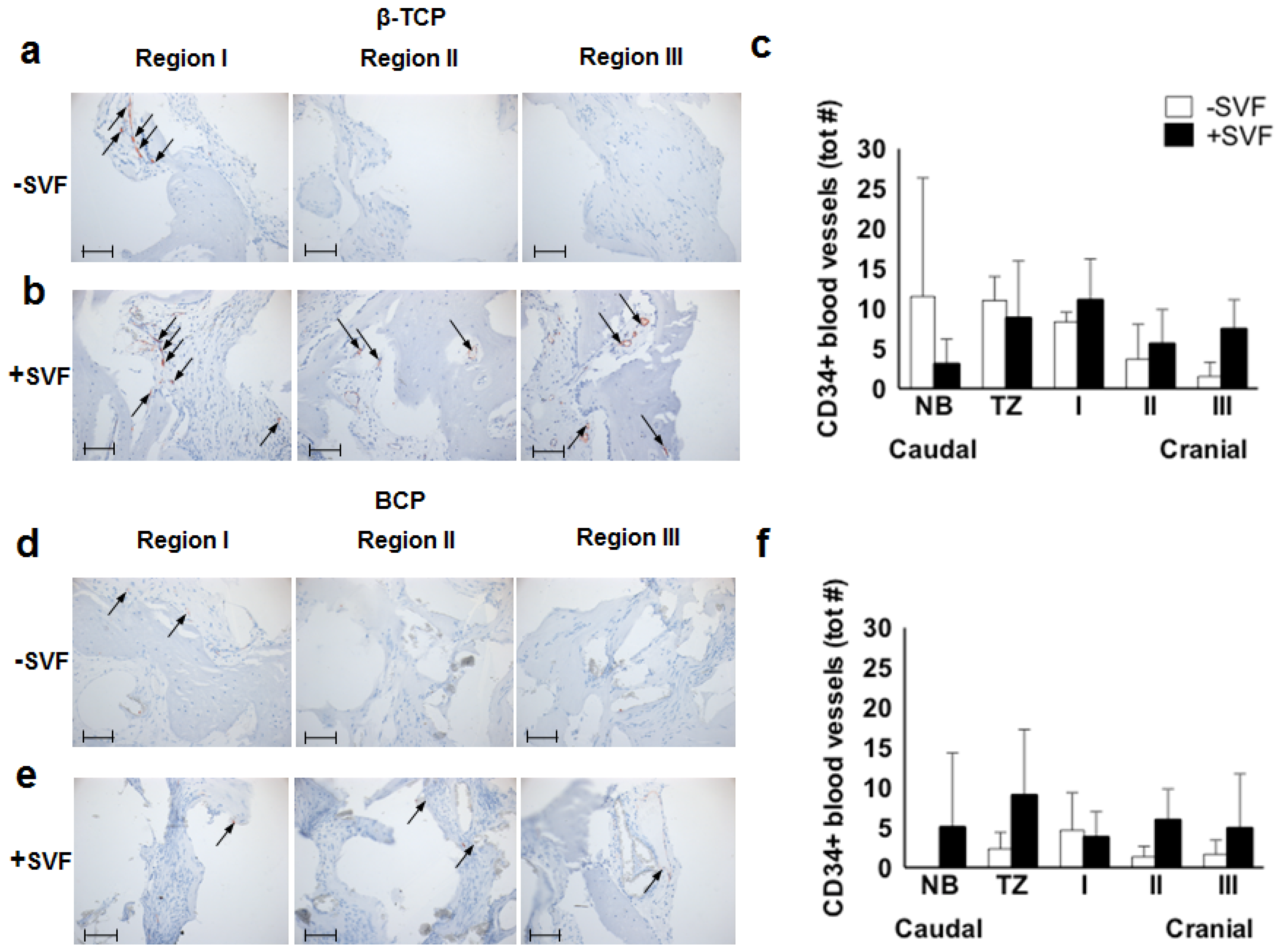
3.4.1. CD34+ Blood Vessels
3.4.2. SMA+ Blood Vessels
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tatum, H., Jr. Maxillary and sinus implant reconstructions. Dent. Clin. N. Am. 1986, 30, 207–229. [Google Scholar] [CrossRef] [PubMed]
- Zijderveld, S.A.; ten Bruggenkate, C.M.; van Den Bergh, J.P.; Schulten, E.A.J.M. Fractures of the iliac crest after split-thickness bone grafting for preprosthetic surgery: Report of 3 cases and review of the literature. J. Oral Maxillofac. Surg. 2004, 62, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Vamze, J.; Pilmane, M.; Skagers, A. Biocompatibility of pure and mixed hydroxyapatite and α-tricalcium phosphate implanted in rabbit bone. J. Mater. Sci. Mater. Med. 2015, 26, 73. [Google Scholar] [CrossRef] [PubMed]
- Al-Sanabani, J.S.; Madfa, A.A.; Al-Sanabani, F.A. Application of calcium phosphate materials in dentistry. Int. J. Biomater. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.; Abdallah, M.N.; Hanafi, A.A.; Misbahuddin, S.; Rashid, H.; Glogauer, M. Mechanisms of in vivo degradation and resorption of calcium phosphate based biomaterials. Materials 2015, 8, 7913–7925. [Google Scholar] [CrossRef] [PubMed]
- Zerbo, I.R.; Zijderveld, S.A.; de Boer, A.; Bronckers, A.L.J.J.; de Lange, G.; ten Bruggenkate, C.M.; Burger, E.H. Histomorphometry of human sinus floor augmentation using a porous beta-tricalcium phosphate: A prospective study. Clin. Oral Implants Res. 2004, 15, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Zijderveld, S.A.; Giltaij, L.R.; Bergh, J.P.; Smit, T.H. Pre-clinical and clinical experiences with BMP-2 and BMP-7 in sinus floor elevation surgery. A comparison. J. Musculoskelet. Res. 2002, 6, 43–54. [Google Scholar] [CrossRef]
- Zijderveld, S.A.; Zerbo, I.R.; van den Bergh, J.P.; Schulten, E.A.J.M.; ten Bruggenkate, C.M. Maxillary sinus floor augmentation using a beta-tricalcium phosphate (Cerasorb) alone compared to autogenous bone grafts. Int. J. Oral Maxillofac. Implants 2005, 20, 432–440. [Google Scholar] [PubMed]
- Li, Y.; Jiang, T.; Zheng, L.; Zhao, J. Osteogenic differentiation of mesenchymal stem cells (MSCs) induced by three calcium phosphate ceramic (CaP) powders: A comparative study. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 80, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Urquia Edreira, E.R.; Hayrapetyan, A.; Wolke, J.G.; Croes, H.J.; Klymov, A.; Jansen, J.A.; van den Beucken, J.J. Effect of calcium phosphate ceramic substrate geometry on mesenchymal stromal cell organization and osteogenic differentiation. Biofabrication 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Crivellato, E. “Sprouting angiogenesis”, a reappraisal. Dev. Biol. 2012, 372, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Logsdon, E.A.; Finley, S.D.; Popel, A.S.; Mac Gabhann, F. A systems biology view of blood vessel growth and remodelling. J. Cell. Mol. Med. 2014, 18, 1491–1508. [Google Scholar] [CrossRef] [PubMed]
- Bergers, G.; Song, S. The role of pericytes in blood-vessel formation and maintenance. Neuro Oncol. 2005, 7, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Colton, C.K. Implantable biohybrid artificial organs. Cell Transplant. 1995, 4, 415–436. [Google Scholar] [CrossRef] [PubMed]
- Moioli, E.K.; Clark, P.A.; Chen, M.; Dennis, J.E.; Erickson, H.P.; Gerson, S.L.; Mao, J.J. Synergistic actions of hematopoietic and mesenchymal stem/ progenitor cells in vascularizing bioengineered tissues. PLoS ONE 2008, 3, e3922. [Google Scholar] [CrossRef] [PubMed]
- Farré-Guasch, E.; Martí-Pagès, C.; Hernádez-Alfaro, F.; Klein-Nulend, J.; Casals, N. Buccal fat pad, an oral access source of human adipose stem cells with potential for osteochondral tissue engineering: An in vitro study. Tissue Eng. Part C Methods 2010, 16, 1083–1094. [Google Scholar] [CrossRef] [PubMed]
- Aust, L.; Devlin, B.; Foster, S.J.; Halvorsen, Y.D.; Hicok, K.; du Laney, T.; Sen, A.; Willingmyre, G.D.; Gimble, J.M. Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy 2004, 6, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Helder, M.N.; Knippenberg, M.; Klein-Nulend, J.; Wuisman, P.I.J.M. Stem cells from adipose tissue allow challenging new concepts for regenerative medicine. Tissue Eng. 2007, 13, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Agata, H.; Asahina, I.; Watanabe, N.; Ishii, Y.; Kubo, N.; Ohshima, S.; Yamazaki, M.; Tojo, A.; Kagami, H. Characteristic change and loss of in vivo osteogenic abilities of human bone marrow stromal cells during passage. Tissue Eng. Part A 2010, 16, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Izadpanah, R.; Kaushal, D.; Kriedt, C.; Tsien, F.; Patel, B.; Dufour, J.; Bunnell, B.A. Long-term in vitro expansion alters the biology of adult mesenchymal stem cells. Cancer Res. 2008, 68, 4229–4238. [Google Scholar] [CrossRef] [PubMed]
- Rubio, D.; Garcia, S.; Paz, M.F.; De la Cueva, T.; Lopez-Fernandez, L.A.; Lloyd, A.C.; Garcia-Castro, J.; Bernad, A. Molecular characterization of spontaneous mesenchymal stem cell transformation. PLoS ONE 2008, 3, e1398. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.H.; Koh, Y.J.; Han, J.; Sung, H.K.; Jong Lee, H.; Morisada, T.; Schwendener, R.A.; Brekken, R.A.; Kang, G.; Oike, Y.; et al. Angiogenic role of LYVE-1-positive macrophages in adipose tissue. Circ. Res. 2007, 100, e47–e57. [Google Scholar] [CrossRef] [PubMed]
- Overman, J.R.; Farré-Guasch, E.; Helder, M.N.; ten Bruggenkate, C.M.; Schulten, E.A.J.M.; Klein-Nulend, J. Short (15 min) bone morphogenetic protein-2 treatment stimulates osteogenic differentiation of human adipose stem cells seeded on calcium phosphate scaffolds in vitro. Tissue Eng. Part A 2013, 19, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Overman, J.R.; Helder, M.N.; ten Bruggenkate, C.M.; Schulten, E.A.J.M.; Klein-Nulend, J.; Bakker, A.D. Growth factor gene expression profiles of bone morphogenetic protein-2-treated human adipose stem cells seeded on calcium phosphate scaffolds in vitro. Biochimie 2013, 95, 2304–2313. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Kim, D.H.; Song, H.R.; Kang, W.H.; Kim, H.J.; Lim, H.C.; Cho, D.W.; Bae, J.H. Repair of rabbit ulna segmental bone defect using freshly isolated adipose-derived stromal vascular fraction. Cytotherapy 2012, 14, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Prins, H.J.; Schulten, E.A.J.M.; Ten Bruggenkate, C.M.; Klein-Nulend, J.; Helder, M.N. Bone regeneration using the freshly isolated autologous stromal vascular fraction of adipose tissue in combination with calcium phosphate ceramics. Stem Cells Transl. Med. 2016, 5, 1362–1374. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.J.; Koh, B.I.; Kim, H.; Joo, H.J.; Jin, H.K.; Jeon, J.; Choi, C.; Lee, D.H.; Chung, J.H.; Cho, C.H.; et al. Stromal vascular fraction from adipose tissue forms profound vascular network through the dynamic reassembly of blood endothelial cells. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1141–1150. [Google Scholar] [CrossRef]
- Rubina, K.; Kalinina, N.; Efimenko, A.; Rubina, K.; Kalinina, N.; Efimenko, A.; Lopatina, T.; Melikhova, V.; Tsokolaeva, Z.; Sysoeva, V.; et al. Adipose stromal cells stimulate angiogenesis via promoting progenitor cell differentiation, secretion of angiogenic factors, and enhancing vessel maturation. Tissue Eng. Part A 2009, 15, 2039–2050. [Google Scholar] [CrossRef] [PubMed]
- Madonna, R.; De Caterina, R. In vitro neovasculogenic potential of resident adipose tissue precursors. Am. J. Physiol. Cell Physiol. 2008, 295, 1271–1280. [Google Scholar] [CrossRef] [PubMed]
- Oostlander, A.E.; Bravenboer, N.; Sohl, E.; Holzmann, P.J.; van der Woude, C.J.; Dijkstra, G.; Stokkers, P.C.; Oldenburg, B.; Netelenbos, J.C.; Hommes, D.W.; et al. Histomorphometric analysis reveals reduced bone mass and bone formation in patients with quiescent Crohn’s disease. Dutch Initiative on Crohn and Colitis (ICC). Gastroenterology 2011, 140, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Schulten, E.A.J.M.; Prins, H.J.; Overman, J.R.; Helder, M.N.; ten Bruggenkate, C.M.; Klein-Nulend, J. A novel approach revealing the effect of a collagenous membrane on osteoconduction in maxillary sinus floor elevation with beta-tricalcium phosphate. Eur. Cells Mater. 2013, 25, 215–228. [Google Scholar] [CrossRef]
- Parfitt, A.M.; Drezner, M.K.; Glorieux, F.H.; Kanis, J.A.; Malluche, H.; Meunier, P.J.; Ott, S.M.; Recker, R.R. Bone histomorphometry: Standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 1987, 2, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, J.; Zhu, X.D.; Tang, Z.R.; Yang, X.; Tan, Y.F.; Fan, Y.J.; Zhang, X.D. Enhanced effect of β-tricalcium phosphate phase on neovascularization of porous calcium phosphate ceramics: In vitro and in vivo evidence. Acta Biomater. 2015, 11, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Pusztaszeri, M.P.; Seelentag, W.; Bosman, F.T. Immunohistochemical expression of endothelial markers CD31, CD34, von Willebrand Factor, and Fli-1 in normal human tissues. J. Histochem. Cytochem. 2006, 54, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Brey, E.M.; McIntire, L.V.; Johnston, C.M.; Reece, G.P.; Patrick, C.W., Jr. Three-dimensional, quantitative analysis of desmin and smooth muscle alpha actin expression during angiogenesis. Ann. Biomed. Eng. 2004, 32, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Amir, L.R.; Becking, A.G.; Jovanovic, A.; Perdijk, F.B.; Everts, V.; Bronckers, A.L.J.J. Formation of new bone during vertical distraction osteogenesis of the human mandible is related to the presence of blood vessels. Clin. Oral Implants Res. 2006, 17, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Roux, B.M.; Cheng, M.; Brey, E.M. Engineering clinically relevant volumes of vascularized bone. J. Cell. Mol. Med. 2015, 19, 903–914. [Google Scholar] [CrossRef] [PubMed]
- Planat-Benard, V.; Silvestre, J.S.; Cousin, B.; André, M.; Nibbelink, M.; Tamarat, R.; Clergue, M.; Manneville, C.; Saillan-Barreau, C.; Duriez, M.; et al. Plasticity of human adipose lineage cells toward endothelial cells: Physiological and therapeutic perspectives. Circulation 2004, 109, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Vergroesen, P.P.; Kroeze, R.J.; Helder, M.N.; Smit, T.H. The use poly(L-lactide-co-caprolactone) as a scaffold for adipose stem cells in bone tissue engineering: Application in a spinal fusion model. Macromol. Biosci. 2011, 14, 722–730. [Google Scholar] [CrossRef] [PubMed]
| Patient Number | Gender, Age (Years) | Body Mass Index | Unilateral/Bilateral | Control/Study Side | Graft Material | Dental Implant Positions |
|---|---|---|---|---|---|---|
| 1 | ♀, 58 | 24.5 | bilateral | Control study | β-TCP | 14,15,16 24,25,26 |
| 2 | ♀, 46 | 30.4 | bilateral | Study control | β-TCP | 14,15,16 24,25,26 |
| 3 | ♂, 69 | 35.8 | bilateral | control study | β-TCP | 14,15,16 25,26,27 |
| 4 | ♀, 59 | 31.2 | unilateral | study | β-TCP | 24,25,26 |
| 5 | ♂, 64 | 24.8 | unilateral | study | β-TCP | 15,16 |
| 6 | ♀, 57 | 24.2 | bilateral | study control | BCP | 14,15,16 24,26 |
| 7 | ♂, 56 | 28.7 | bilateral | study control | BCP | 15,16,17 25,26,27 |
| 8 | ♀, 51 | 30.9 | unilateral | study | BCP | 14,15,16 |
| 9 | ♂, 52 | 32.1 | unilateral | study | BCP | 23,25,26 |
| 10 | ♀, 51 | 30.3 | bilateral | study control | BCP | 15,16 25,26 |
| % of Total Blood Vessels | |||||||||
| - | Size of blood vessels | Region I | Region II | Region III | Total | Region I | Region II | Region III | Total |
| −SVF | +SVF | ||||||||
| β-TCP | Small | 68 | 26 | 6 | 100 | 40 | 13 | 47 | 100 |
| Large | 69 | 20 | 11 | 100 | 47 | 10 | 43 | 100 | |
| BCP | Small | 43 | 37 | 20 | 100 | 29 | 43 | 28 | 100 |
| Large | 67 | 18 | 15 | 100 | 59 | 7 | 34 | 100 | |
| % of Total CD34+ Blood Vessels | |||||||||
| - | Size of blood vessels | Region I | Region II | Region III | Total | Region I | Region II | Region III | Total |
| −SVF | +SVF | ||||||||
| β-TCP | Small | 70 | 19 | 11 | 100 | 50 | 25 | 25 | 100 |
| Large | 76 | 24 | 0 | 100 | 40 | 0 | 60 | 100 | |
| BCP | Small | 70 | 14 | 16 | 100 | 30 | 51 | 19 | 100 |
| Large | 69 | 0 | 31 | 100 | 18 | 42 | 40 | 100 | |
| % of Total SMA+ Blood Vessels | |||||||||
| - | Size of blood vessels | Region I | Region II | Region III | Total | Region I | Region II | Region III | Total |
| −SVF | +SVF | ||||||||
| β-TCP | Small | 52 | 48 | 0 | 100 | 30 | 24 | 46 | 100 |
| Large | 41 | 46 | 13 | 100 | 10 | 20 | 70 | 100 | |
| BCP | Small | 80 | 0 | 20 | 100 | 35 | 43 | 22 | 100 |
| Large | 75 | 0 | 25 | 100 | 22 | 19 | 59 | 100 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farré-Guasch, E.; Bravenboer, N.; Helder, M.N.; Schulten, E.A.J.M.; Ten Bruggenkate, C.M.; Klein-Nulend, J. Blood Vessel Formation and Bone Regeneration Potential of the Stromal Vascular Fraction Seeded on a Calcium Phosphate Scaffold in the Human Maxillary Sinus Floor Elevation Model. Materials 2018, 11, 161. https://doi.org/10.3390/ma11010161
Farré-Guasch E, Bravenboer N, Helder MN, Schulten EAJM, Ten Bruggenkate CM, Klein-Nulend J. Blood Vessel Formation and Bone Regeneration Potential of the Stromal Vascular Fraction Seeded on a Calcium Phosphate Scaffold in the Human Maxillary Sinus Floor Elevation Model. Materials. 2018; 11(1):161. https://doi.org/10.3390/ma11010161
Chicago/Turabian StyleFarré-Guasch, Elisabet, Nathalie Bravenboer, Marco N. Helder, Engelbert A. J. M. Schulten, Christiaan M. Ten Bruggenkate, and Jenneke Klein-Nulend. 2018. "Blood Vessel Formation and Bone Regeneration Potential of the Stromal Vascular Fraction Seeded on a Calcium Phosphate Scaffold in the Human Maxillary Sinus Floor Elevation Model" Materials 11, no. 1: 161. https://doi.org/10.3390/ma11010161
APA StyleFarré-Guasch, E., Bravenboer, N., Helder, M. N., Schulten, E. A. J. M., Ten Bruggenkate, C. M., & Klein-Nulend, J. (2018). Blood Vessel Formation and Bone Regeneration Potential of the Stromal Vascular Fraction Seeded on a Calcium Phosphate Scaffold in the Human Maxillary Sinus Floor Elevation Model. Materials, 11(1), 161. https://doi.org/10.3390/ma11010161