Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Blends Preparation
2.3. Characterization
3. Results and Discussion
3.1. Reactive Plasticization of PLA/PBAT and PLA/PBS Blends by Using EJ400
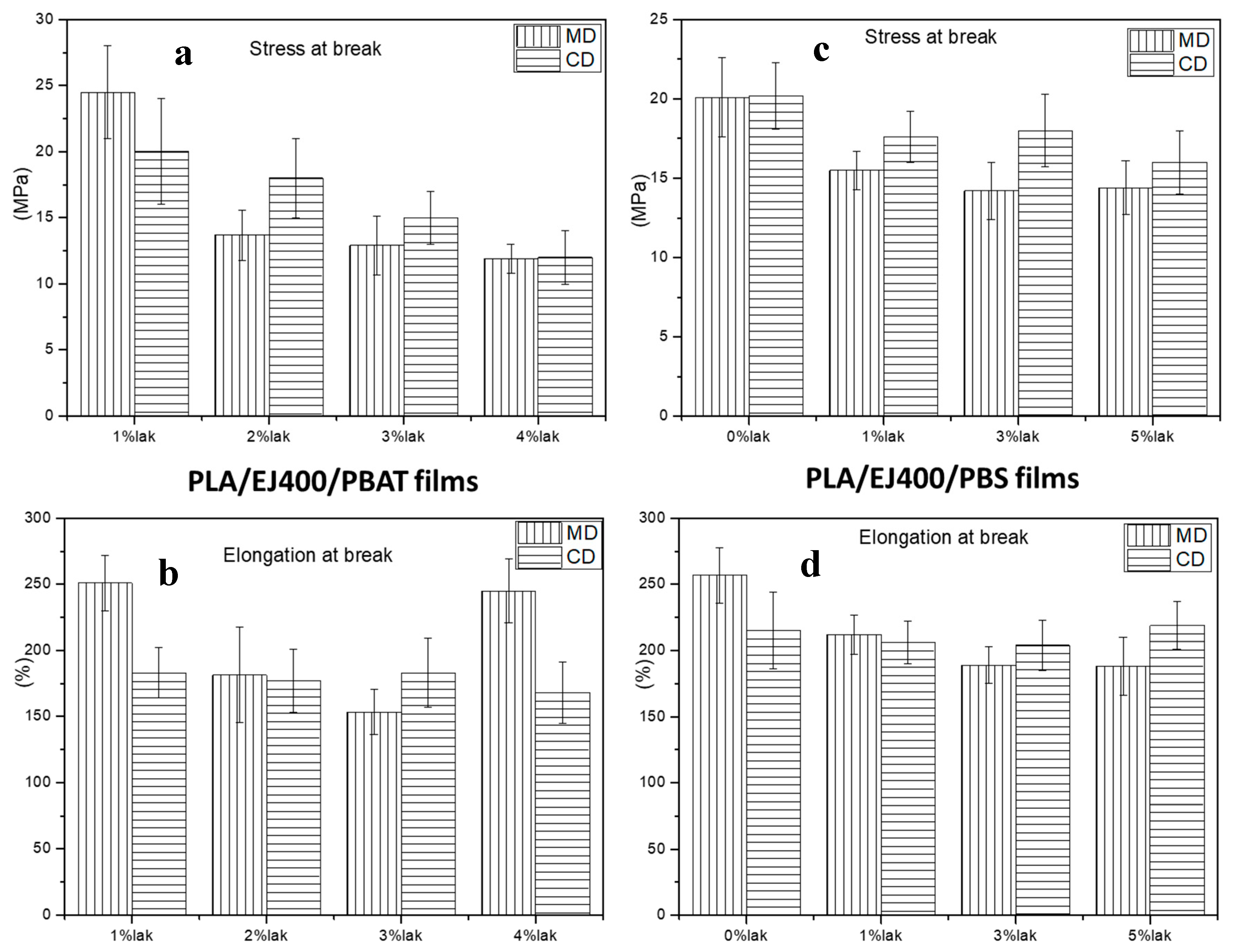
3.2. Mechanical Properties of Plasticized and Nucleated Blown Extruded Films
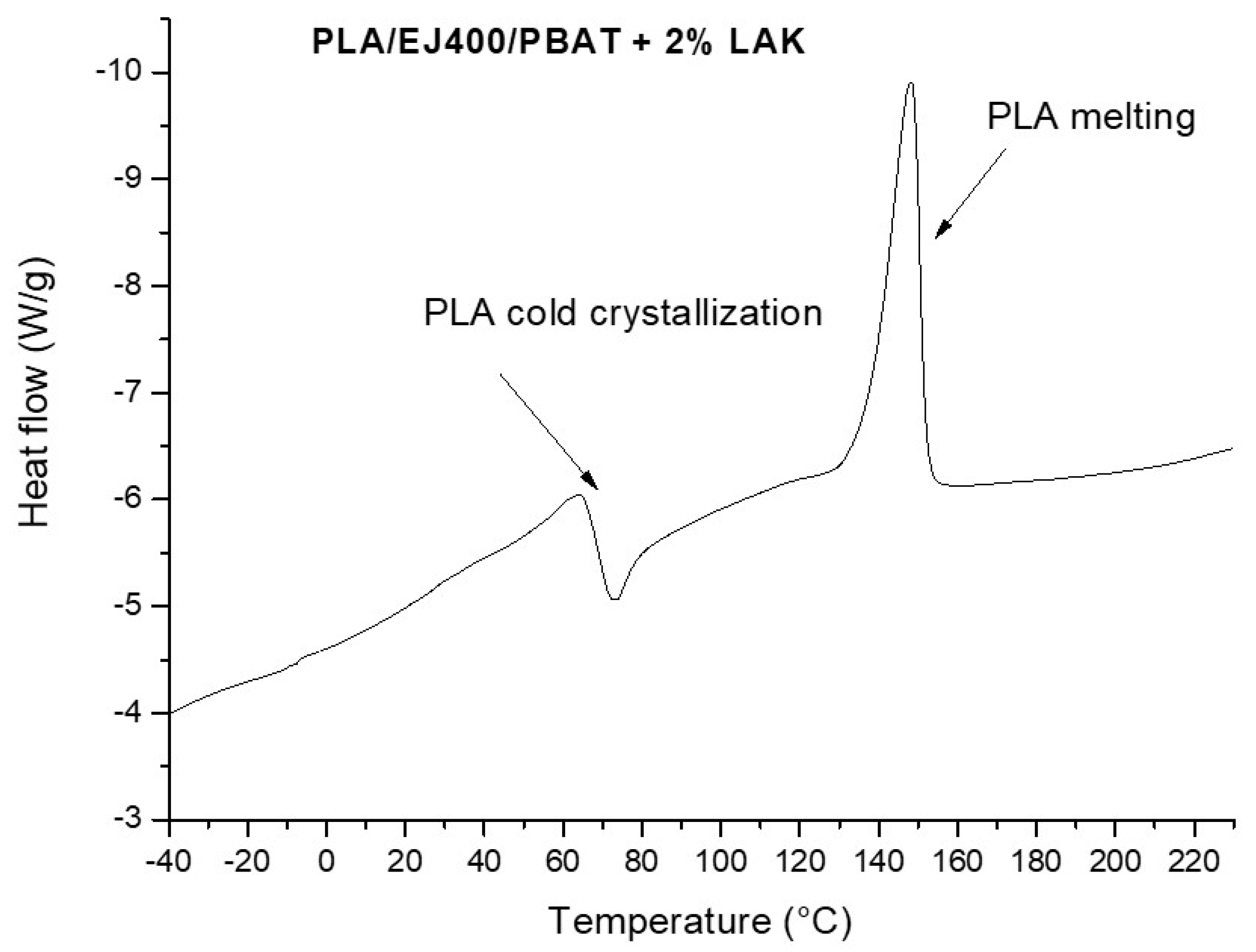
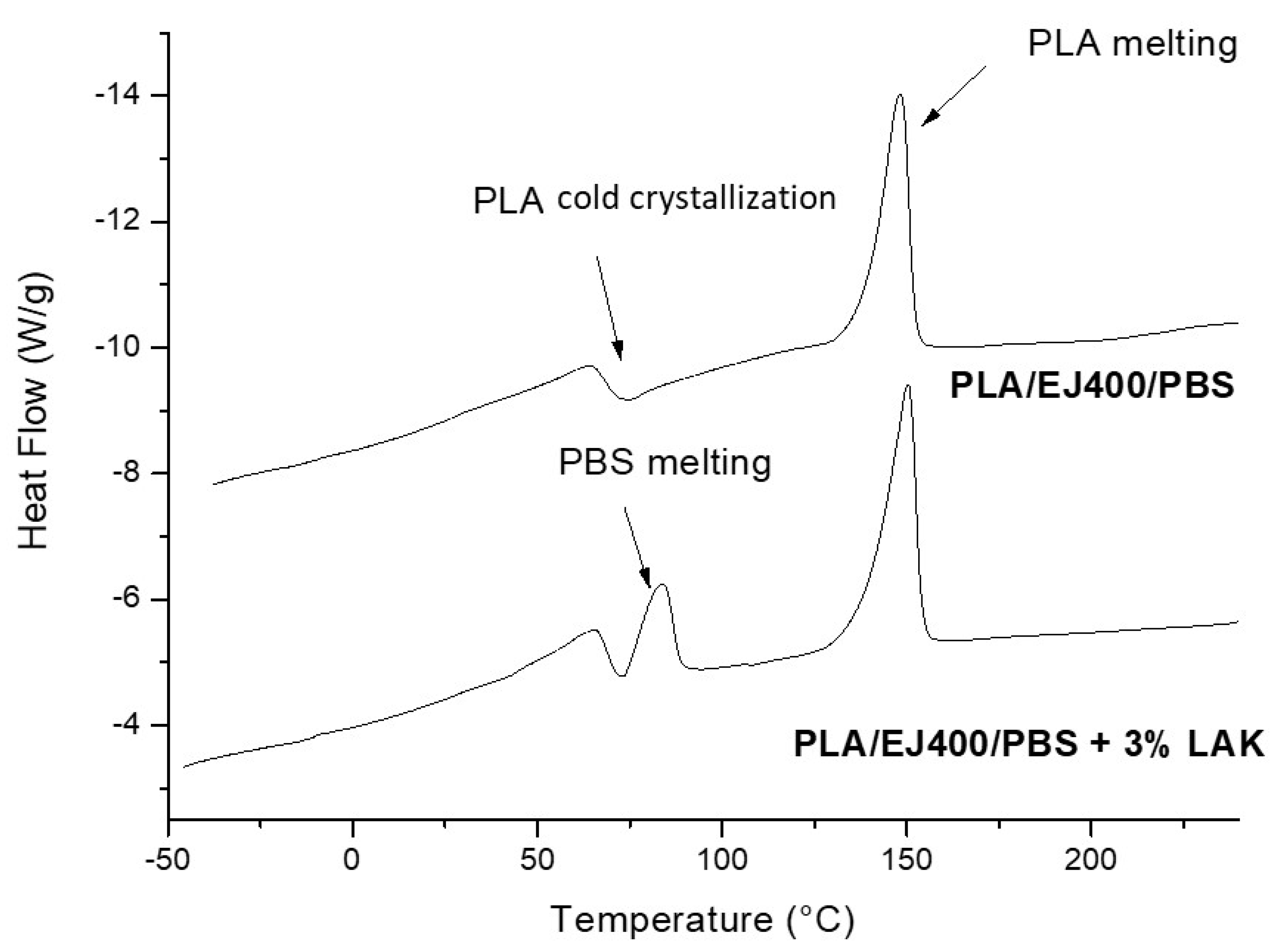
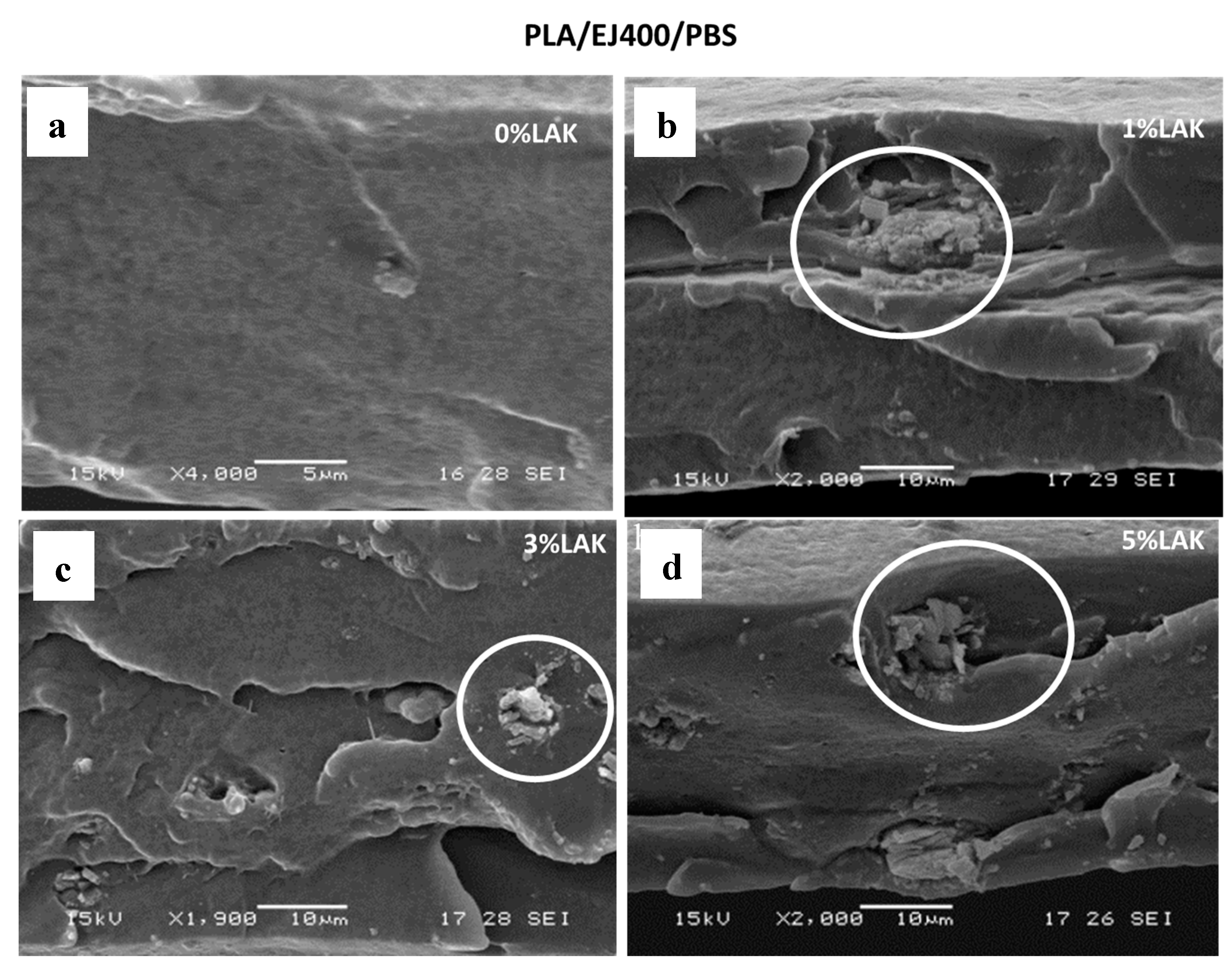
3.3. Thermal and Morphological Features of Plasticized and Nucleated Blown Extruded Films
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siracusa, V.; Rocculi, P.; Romani, S.; Rosa, M.D. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Sorrentino, A.; Gorrasi, G.; Vittoria, V. Potential perspectives of bio-nanocomposites for food packaging applications. Trends Food Sci. Technol. 2007, 18, 84–95. [Google Scholar] [CrossRef]
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Jacquot, M.; Desobry, S. Poly-Lactic Acid: Production, applications, nanocomposites, and release studies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 552–571. [Google Scholar] [CrossRef]
- Bautista, I.E.; Jover, J.; Gutierrez, M.; Corpas, J.F.; Cremades, R.; Fontiveros, O.; Vega, J.F. Preparation of crayfish chitin by in situ lactic acid production. Process Biochem. 2001, 37, 229–234. [Google Scholar] [CrossRef]
- Bogaert, J.C.; Coszach, P. Poly(lactic acids): A potential solution to plastic waste dilemma. Macromol. Symp. 2000, 153, 287–303. [Google Scholar] [CrossRef]
- Auras, R.; Harte, B.; Selke, S. An Overview of Polylactides as Packaging Materials. Macromol. Biosci. 2004, 4, 835–864. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, M.; Whiteman, N. Polylactide, a New Thermoplastic for Extrusion Coating. In Proceedings of the 2000 Polymers, Laminations and Coatings Conference, Chicago, IL, USA, 27–31 August 2000; pp. 631–635. [Google Scholar]
- Migliaresi, C.; de Lollis, A.; Fambri, L.; Cohn, D. The effect of thermal history on the crystallinity of different molecular weight PLLA biodegradable polymers. Clin. Mater. 1991, 8, 111–118. [Google Scholar] [CrossRef]
- Miyata, T.; Masuku, T. Crystallization behaviour of poly(l-lactide). Polymer 1998, 39, 5515–5521. [Google Scholar] [CrossRef]
- Jin, T.; Zhang, H. Biodegradable Polylactic Acid Polymer with Nisin for Use in Antimicrobial Food Packaging. J. Food Sci. 2008, 73, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Coltelli, M.B.; Della Maggiore, I.; Bertoldo, M.; Bronco, S.; Signori, F.; Ciardelli, F. Poly(lactic acid) properties as a consequence of poly(butylene adipate-co-terephthalate) blending and acetyl tributyl citrate plasticization. J. Appl. Polym. Sci. 2008, 110, 1250–1262. [Google Scholar] [CrossRef]
- Scatto, M.; Salmini, E.; Castiello, S.; Coltelli, M.B.; Conzatti, L.; Stagnaro, P.; Andreotti, L.; Bronco, S. Plasticized and nanofilled poly(lactic acid)-based cast films: Effect of plasticizer and organoclay on processability and final properties. J. Appl. Pol. Sci. 2013, 127, 4947–4956. [Google Scholar] [CrossRef]
- Farsetti, S.; Cioni, B.; Lazzeri, A. Physico-Mechanical Properties of Biodegradable Rubber Toughened Polymers. Macromol. Symp. 2011, 301, 82–89. [Google Scholar] [CrossRef]
- Signori, F.; Coltelli, M.B.; Bronco, S.; Ciardelli, F. Thermal degradation of poly(lactic acid) (PLA) and poly(butylene adipate-co-terephthalate) (PBAT) and their blends upon melt processing. Polym. Degrad. Stab. 2009, 94, 74–82. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Bronco, S.; Chinea, C. The effect of free radical reactions on structure and properties of poly(lactic acid) (PLA) based blends. Polym. Degrad. Stab. 2010, 95, 332–341. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Toncelli, C.; Ciardelli, F.; Bronco, S. Compatible blends of biorelated polyesters through catalytic transesterification in the melt. Polym. Degrad. Stab. 2011, 96, 982–990. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Signori, F.; Toncelli, C.; Escrig Rondan, C.; Bronco, S.; Ciardelli, F. Biodegradable and Compostable PLA-based Formulations to Replace Plastic Disposable Commodities. In Proceedings of the International Conference (Bio)Degradable Polymers from Renewable Resources, Vienna, Austria, 18–21 November 2007; Abstract Book; p. 53. Available online: http://www.ifpan.edu.pl/paj/polymer/polymers-abs-book.pdf (accessed on 11 January 2018).
- Yokohara, T.; Yamaguchi, M. Structure and properties for biomass-based polyester blends of PLA and PBS. Eur. Polym. J. 2008, 44, 677–685. [Google Scholar] [CrossRef]
- Supthanyakula, R.; Kaabbuathongb, N.; Chirachanchai, S. Random poly(butylene succinate-co-lactic acid) as a multi-functional additive for miscibility, toughness, and clarity of PLA/PBS blends. Polymer 2016, 105, 1–9. [Google Scholar] [CrossRef]
- Jin, F.L.; Pang, Q.Q.; Zhang, T.Y.; Park, S.J. Synergistic reinforcing of poly(lactic acid)-based systems by polybutylene succinate and nano-calcium carbonate. J. Ind. Eng. Chem. 2015, 32, 77–84. [Google Scholar] [CrossRef]
- Tcharkhtchi, A.; Abdallah-Elhirtsi, S.; Ebrahimi, K.; Fitoussi, J.; Shirinbayan, M.; Farzaneh, S. Some New Concepts of Shape Memory Effect of Polymers. Polymers 2014, 6, 1144–1163. [Google Scholar] [CrossRef]
- Bechthold, I.; Bretz, K.; Kabasci, S.; Kopitzky, R. Succinic Acid: A New Platform Chemical for Biobased Polymers from Renewable Resources. Chem. Eng. Technol. 2008, 31, 647–654. [Google Scholar] [CrossRef]
- Harmsen, P.; Hackmann, M. Green Building Blocks for Biobased Plastics, Biobased Processes and Market Development; Wageningen UR Food and Biobased Research: Wageningen, The Netherlands, 2013; ISBN 978-94-6173-610-9. [Google Scholar]
- Shakoor, A.; Thomas, N.L. Talc as a nucleating agent and reinforcing filler in poly(lactic acid) composites. Polym. Eng. Sci. 2014, 54, 64–70. [Google Scholar] [CrossRef]
- Shi, X.; Zhang, G.; Phuong, T.V.; Lazzeri, A. Synergistic Effects of Nucleating Agents and Plasticizers on the Crystallization Behavior of Poly(lactic acid). Molecules 2015, 20, 1579–1593. [Google Scholar] [CrossRef] [PubMed]
- Fehri, M.K.; Cinelli, P.; Coltelli, M.B.; Anguillesi, I.; Lazzeri, A. Thermal Properties of Plasticized Poly(lactic acid)(PLA) Containing Nucleating agent. Int. J. Chem. Eng. Appl. 2016, 7, 85–88. [Google Scholar] [CrossRef]
- Fehri, M.K.; Mugoni, C.; Cinelli, P.; Anguillesi, I.; Coltelli, M.B.; Fiori, S.; Montorsi, M.; Lazzeri, A. Composition dependence of the synergistic effect of nucleating agent and plasticizer in poly(lactic acid): A Mixture Design study. Expr. Polym. Lett. 2016, 10, 274–288. [Google Scholar] [CrossRef]
- Raffa, P.; Coltelli, M.B.; Savi, S.; Bianchi, S.; Castelvetro, V. Chain extension and branching of poly(ethylene terephthalate) (PET) with di- and multifunctional epoxy or isocyanate additives: An experimental and modelling study. React. Funct. Polym. 2012, 72, 50–60. [Google Scholar] [CrossRef]
- Lazzeri, A.; Phuong, V.T.; Cinelli, P. Copolymers Based on Reactive Polyesters and Plasticisers for the Manufacture of Transparent, Biodegradable Packaging Film. WO 2013164743 A1, 7 November 2013. [Google Scholar]
- Wang, S.; Pang, S.; Xu, N.; Pan, L.; Lin, Q. In situ compatibilization of polylactide/thermoplastic polyester elastomer blends using a multifunctional epoxide compound as a processing agent. J. Appl. Polym. Sci. 2016, 133, 43424. [Google Scholar] [CrossRef]
- Al-Itry, R.; Lamnawar, K.; Maazouz, A. Improvement of thermal stability, rheological and mechanical properties of PLA, PBAT and their blends by reactive extrusion with functionalized epoxy. Polym. Degrad. Stab. 2012, 97, 1898–1914. [Google Scholar] [CrossRef]
- Zhang, W.Y.; Gui, Z.Y.; Lu, C.; Cheng, S.J.; Cai, D.X.; Gao, Y. Improving transparency of incompatible polymer blends by reactive compatibilization. Mater. Lett. 2013, 92, 68–70. [Google Scholar] [CrossRef]
- Arruda, L.C.; Magaton, M.; Bretas, R.E.S.; Ueki, M. Influence of chain extender on mechanical, thermal and morphological properties of blown films of PLA/PBAT blends. Polym. Test. 2015, 43, 27–37. [Google Scholar] [CrossRef]
- Saeidlou, S.; Huneault, M.A.; Li, H.; Park, C.B. Poly(lactic acid) crystallization. Prog. Polym. Sci. 2012, 37, 1657–1677. [Google Scholar] [CrossRef]
- Chen, P.; Wang, W.; Wang, Y.; Yu, K.; Zhou, H.; Wang, X.; Mi, J. Crystallization-induced microcellular foaming of poly(lactic acid) with high volume expansion ratio. Polym. Degrad. Stabil. 2017, 144, 231–240. [Google Scholar] [CrossRef]
- You, J.; Lou, L.; Yu, W.; Zhou, C. The Preparation and Crystallization of Long Chain Branching Polylactide Made by Melt Radicals Reaction. J. Appl. Polym. Sci. 2013, 1959–1970. [Google Scholar] [CrossRef]
- Mallet, B.; Lamnawar, K.; Maazouz, A. Improvement of blown film extrusion of poly(lactic acid): Structure–Processing–Properties relationships. Polym. Eng. Sci. 2014, 54, 840–857. [Google Scholar] [CrossRef]
- Jariyasakoolroj, P.; Tashiro, K.; Wang, H.; Yamamoto, H.; Chinsirikul, W.; Kerddonfag, N.; Chirachanchai, S. Isotropically small crystalline lamellae induced by high biaxial-stretching rate as a key microstructure for super-tough polylactide film. Polymer 2015, 68, 234–245. [Google Scholar] [CrossRef]
- Niaounakis, M. Biopolymers: Processing and Products; Elsevier: Waltham, MA, USA, 2015; p. 19. [Google Scholar]
- Aliotta, L.; Cinelli, P.; Coltelli, M.B.; Righetti, M.C.; Gazzano, M.; Lazzeri, A. Effect of nucleating agents on crystallinity and properties of poly(lactic acid) (PLA). Eur. Polym. J. 2017, 93, 822–832. [Google Scholar] [CrossRef]
- Pérez-Camargo, R.A.; Fernández-d’Arlas, B.; Cavallo, D.; Debuissy, T.; Pollet, E.; Avérous, L.; Müller, A.J. Tailoring the Structure, Morphology, and Crystallization of Isodimorphic Poly(butylene succinate-ran-butylene adipate) Random Copolymers by Changing Composition and Thermal History. Macromolecules 2017, 50, 597–608. [Google Scholar] [CrossRef]
- Cardinaels, R.; Moldenaers, P. Polymer Morphology; Guo, Q., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 2016; Volume 19, pp. 348–373. [Google Scholar]
- Andreasson, E.; Mehmood, N.; Kao-Walter, S. Trouser Tear Tests of Two Thin Polymer Films. In Proceedings of the 13th International Conference on Fracture, Beijing, China, 16–21 June 2013; Available online: http://www.gruppofrattura.it/ocs/index.php/ICF/icf13/paper/viewFile/11465/10844 (accessed on 22 December 2017).
- Quero, E.; Müller, A.J.; Signori, F.; Coltelli, M.B.; Bronco, S. Isothermal Cold-Crystallization of PLA/PBAT Blends with and without the Addition of Acetyl Tributyl Citrate. Macromol. Chem. Phys. 2012, 213, 36–48. [Google Scholar] [CrossRef]
- Bhatia, A.; Gupta, R.; Bhattacharya, S.; Choi, H. Compatibility of biodegradable poly(lactic acid) (PLA) and poly(butylene succinate) (PBS) blends for packaging application. Korea-Aust. Rheol. J. 2007, 19, 125–131. [Google Scholar]



| Blown Extrusion Parameters | Values |
|---|---|
| Layflat width | 410 mm |
| Bubble diameter | 261 mm |
| Die diameter | 55 mm |
| Die gap | 0.8 mm |
| Film thickness | 0.05 mm |
| Blow-up ratio BUR | 4.75 |
| Draw down ratio DR | 3.37 |
| Forming ratio FR | 0.71 |
| Composition | Final Torque (N·cm) | Young Modulus (GPa) | Tensile Strength (MPa) | Elongation at Yield (%) | Stress at Break (MPa) | Elongation at Break (%) |
|---|---|---|---|---|---|---|
| PLA/PBAT 67/33 | 73 | 2.6 ± 0.2 | 38.8 ± 3.1 | 1.4 ± 0.5 | 31 ± 3 | 110 ± 20 |
| PLA/EJ400/PBAT 67/10/23 | 69 | 1.4 ± 0.1 | 28.8 ± 0.9 | 1.2 ± 0.2 | 29 ± 2 | 140 ± 10 |
| PLA/PBS 67/33 | 58 | 1.8 ± 0.2 | 45 ± 1 | 1.3 ± 0.5 | 32 ± 5 | 100 ± 20 |
| PLA/EJ400/PBS 67/10/23 | 55 | 2.7 ± 0.2 | 37 ± 2 | 0.9 ± 0.1 | 30 ± 4 | 150 ± 40 |
| LAK (wt. %) | MD | CD | ||||
|---|---|---|---|---|---|---|
| E’ (GPa) | TgPLA (°C) | TgPBS (°C) | E’ (GPa) | TgPLA (°C) | TgPBS (°C) | |
| 0 | 2.9 ± 0.1 | 35.1 ± 0.9 | −30 ± 2 | 4.2 ± 0.2 | 37 ± 2 | −31 ± 3 |
| 1 | 3.1 ± 0.3 | 32 ± 2 | −38.1 ± 0.9 | 3.4 ± 0.1 | 37.2 ± 0.7 | −33 ± 2 |
| 3 | 2.2 ± 0.1 | 33 ± 2 | −38 ± 2 | 2.2 ± 0.3 | 39 ± 1 | −31 ± 3 |
| 5 | 1.8 ± 0.2 | 31 ± 1 | −36 ± 3 | 2.6 ± 0.2 | 33.2 ± 0.9 | −33 ± 1 |
| LAK (wt. %) | MD | CD | ||||
|---|---|---|---|---|---|---|
| E’ (GPa) | TgPLA (°C) | TgPBAT (°C) | E’ (GPa) | TgPLA (°C) | TgPBAT (°C) | |
| 1 | 2.4 ± 0.2 | 35 ± 2 | −37 ± 1 | 4.1 ± 0.3 | 36.9 ± 0.7 | −36 ± 2 |
| 2 | 2.1 ± 0.1 | 35 ± 1 | −38 ± 2 | 5.3 ± 0.4 | 37 ± 2 | −33 ± 1 |
| 3 | 3.2 ± 0.3 | 35.1 ± 0.8 | −33 ± 2 | 3.4 ± 0.2 | 35 ± 1 | −33 ± 3 |
| 4 | 2.6 ± 0.1 | 35 ± 2 | −33.8 ± 0.7 | 2.2 ± 0.2 | 35.1 ± 0.9 | −33 ± 1 |
| % of LAK | ΔHc (J/g) | ΔHm (J/g) | Total ΔHPLA a (J/g) | Tc (°C) | Tm (°C) |
|---|---|---|---|---|---|
| 1 | 7.681 | 22.48 | 22.09 | 73.67 | 147.38 |
| 2 | 9.482 | 22.60 | 19.53 | 73.46 | 148.28 |
| 3 | 8.557 | 22.38 | 20.65 | 70.78 | 147.62 |
| 4 | 8.335 | 23.42 | 22.53 | 72.95 | 149.07 |
| % of LAK | ΔHcPLA b (J/g) | ΔHmPBS (J/g) | ΔHmPLA b (J/g) | Total ΔHPBS a (J/g) | Total ΔHPLA a (J/g) | TcPLA (°C) | TmPBS (°C) | TmPLA (°C) |
|---|---|---|---|---|---|---|---|---|
| 0 | 7.198 | - | 23.27 | - | 28.42 | 74.75 | - | 148.22 |
| 1 | 0.6645 | 7.626 | 24.96 | 37.24 | 34.52 | 73.70 | 84.29 | 148.93 |
| 3 | 0.9190 | 7.067 | 23.53 | 34.48 | 32.08 | 74.87 | 84.18 | 150.29 |
| 5 | 0 | 5.968 | 25.73 | 29.06 c | 35.63 | - | 83.90 | 148.92 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mallegni, N.; Phuong, T.V.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A. Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion. Materials 2018, 11, 148. https://doi.org/10.3390/ma11010148
Mallegni N, Phuong TV, Coltelli M-B, Cinelli P, Lazzeri A. Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion. Materials. 2018; 11(1):148. https://doi.org/10.3390/ma11010148
Chicago/Turabian StyleMallegni, Norma, Thanh Vu Phuong, Maria-Beatrice Coltelli, Patrizia Cinelli, and Andrea Lazzeri. 2018. "Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion" Materials 11, no. 1: 148. https://doi.org/10.3390/ma11010148
APA StyleMallegni, N., Phuong, T. V., Coltelli, M.-B., Cinelli, P., & Lazzeri, A. (2018). Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion. Materials, 11(1), 148. https://doi.org/10.3390/ma11010148








