On the Role of Processing Parameters in Producing Recycled Aluminum AA6061 Based Metal Matrix Composite (MMC-AlR) Prepared Using Hot Press Forging (HPF) Process
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation
2.2. Hot Press Forging Process
2.3. Hardness Test
2.4. Density Measurement
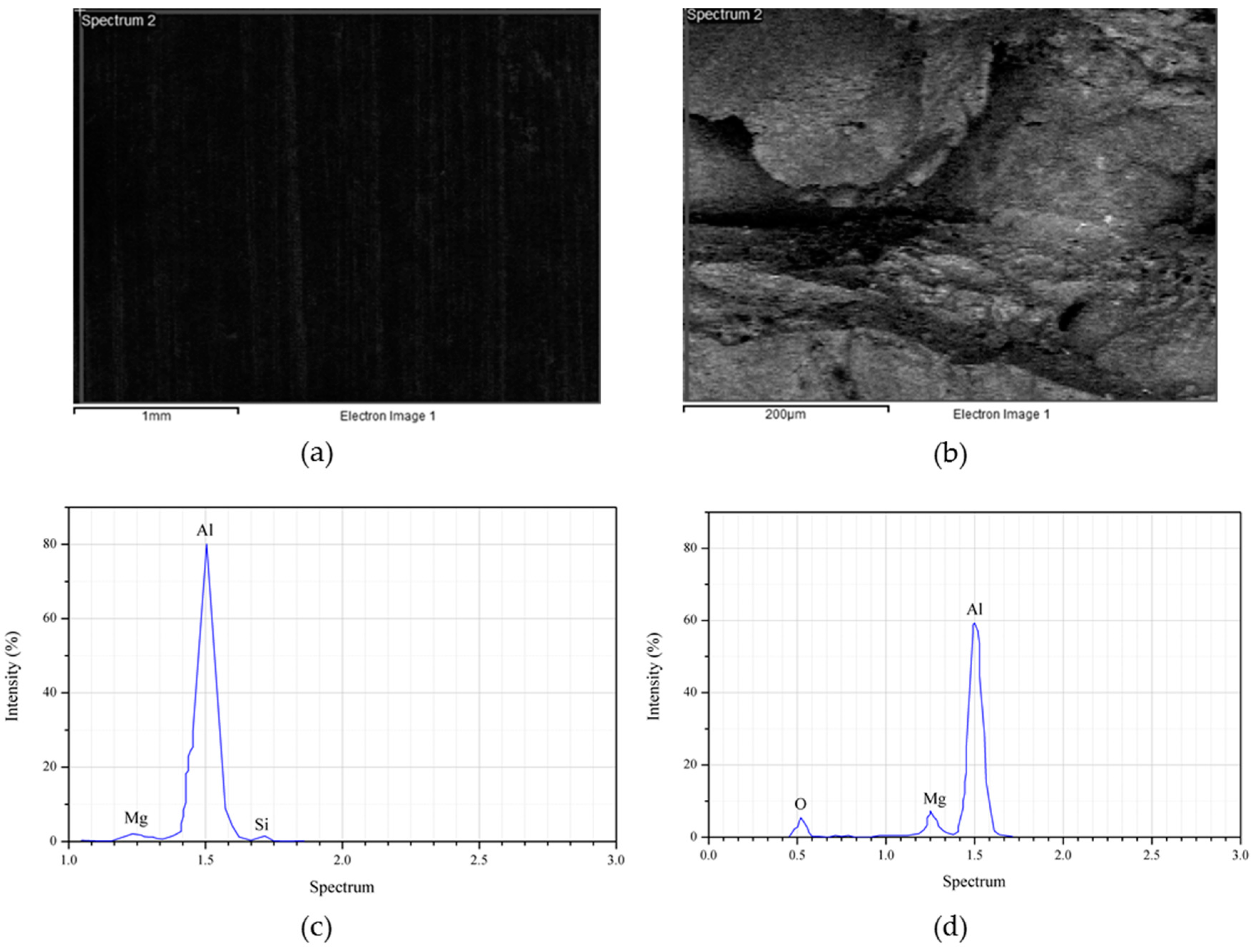
2.5. Crystal Structure Analysis
3. Results and Discussion
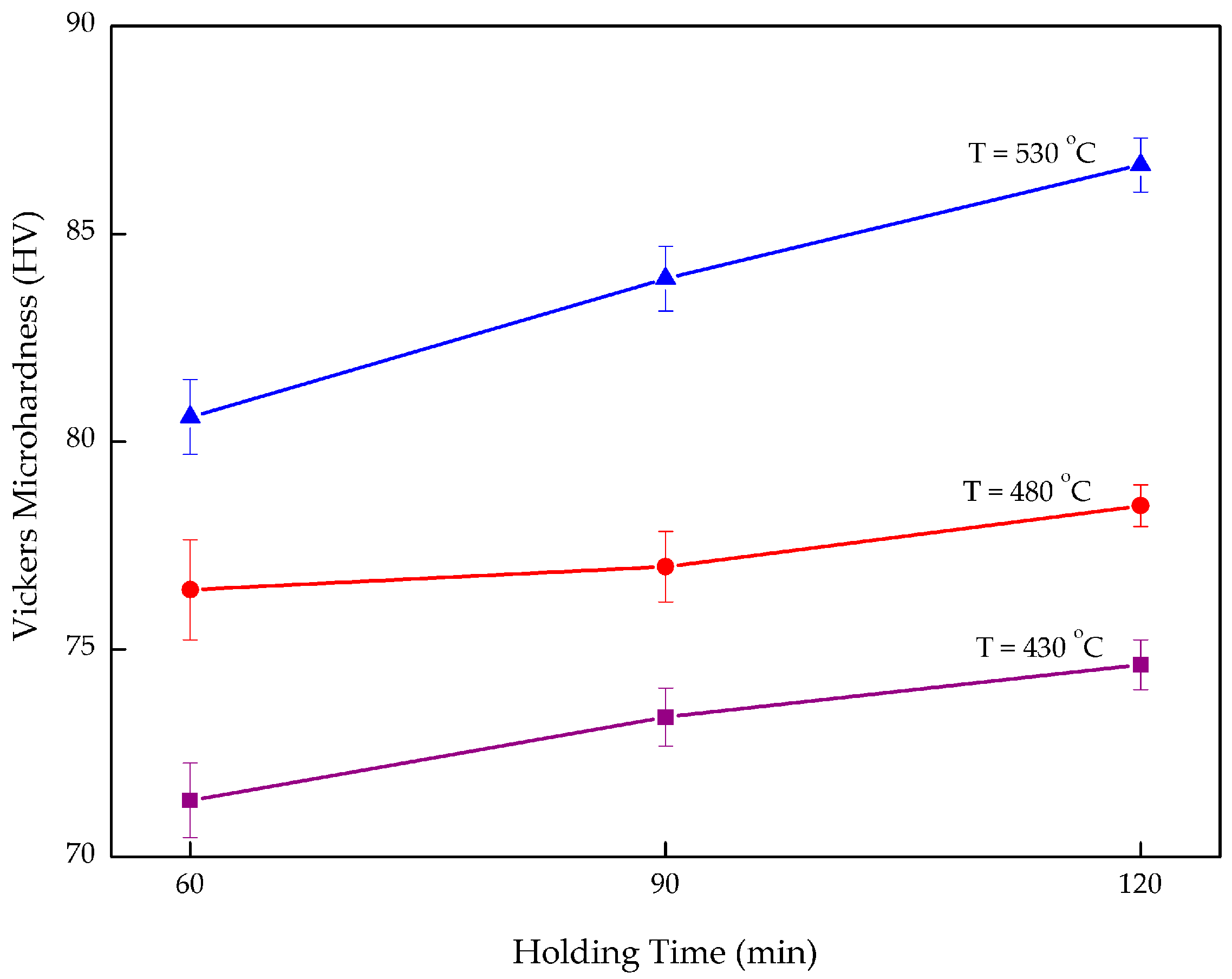
3.1. Vickers Microhardness
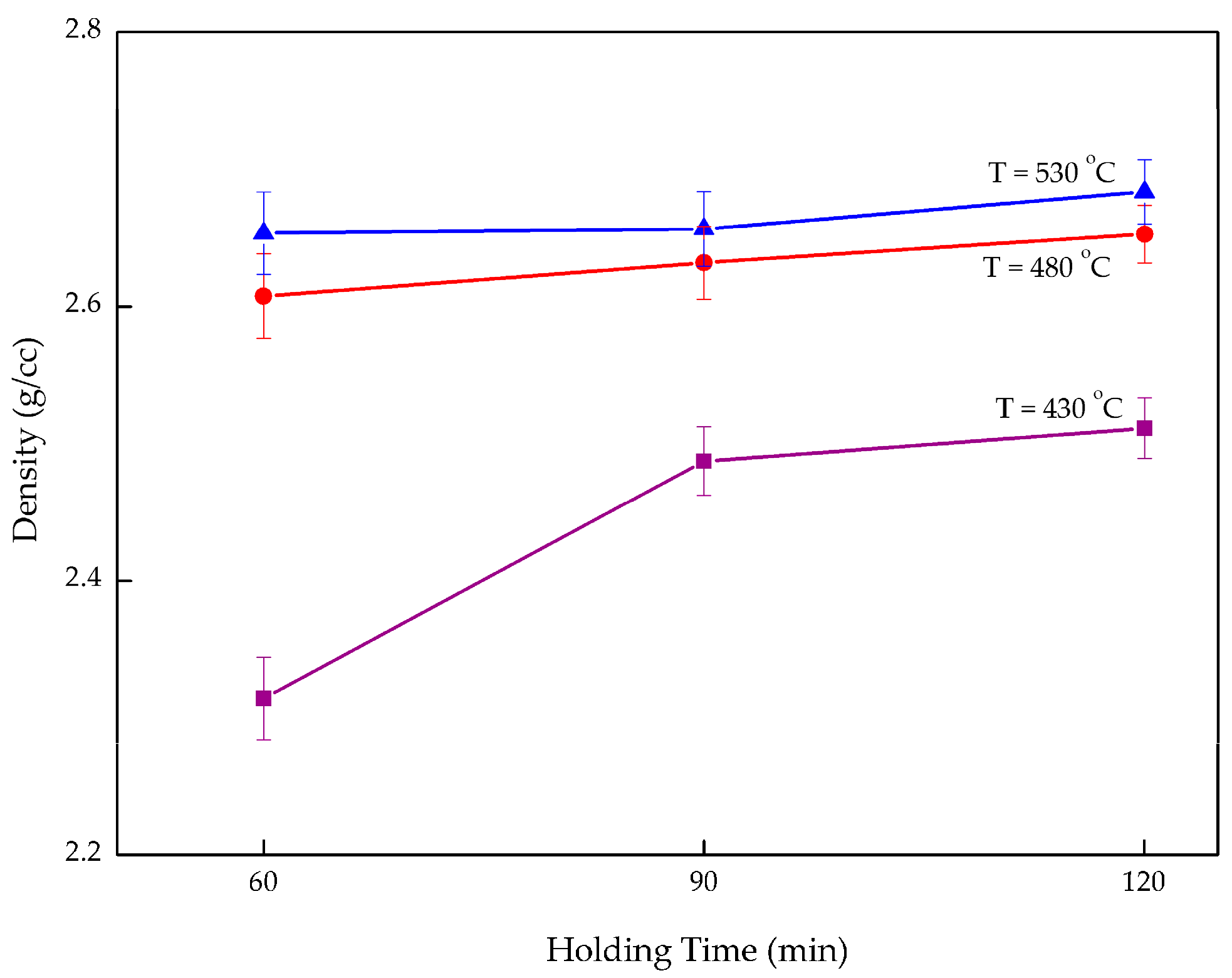
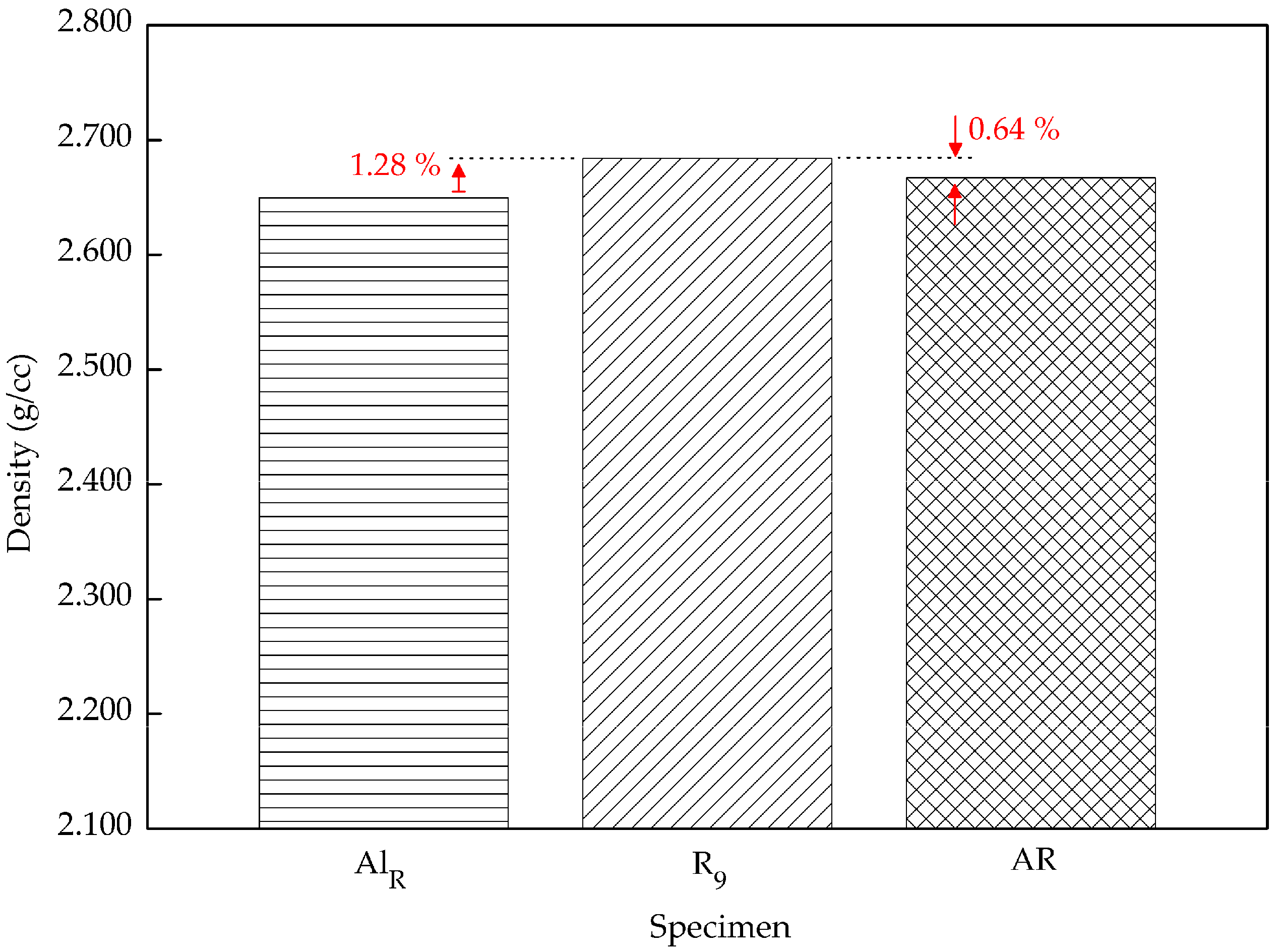
3.2. Density
4. Conclusions
- An increase in both microhardness and density was seen as both temperature and holding time increased. Nevertheless, temperature had a stronger effect the MMC-AlR performance, compared to the holding time. The microhardness increased 4.94–10.45%, from 7.360 to 86.656 HV0.3, by increasing the operating temperature from 430 to 530 °C. The density increased 0.95–12.71%, from 2.314 to 2.684 g/cc, by increasing the operating temperature from 430 to 530 °C.
- MMC-AlR was much stronger than AlR when compared to the AR. MMC-AlR hardness is only 10.21% less, while AlR had 16.85% of differentiation from AR. MMC-AlR density increased 0.63%, while AlR was recorded as having 0.64% of differentiation from AR.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gronostajski, J.; Matuszak, A. The recycling of metals by plastic deformation: An example of recycling of aluminum and its alloys chips. J. Mater. Process. Technol. 1999, 92, 35–41. [Google Scholar] [CrossRef]
- Samuel, M. A new technique for recycling aluminum scrap. J. Mater. Process. Technol. 2003, 135, 117–124. [Google Scholar] [CrossRef]
- Rahim, S.N.A.; Lajis, M.A.; Ariffin, S. A Review on Recycling Aluminum Chips by Hot Extrusion Process. Procedia CIRP 2015, 26, 761–766. [Google Scholar] [CrossRef]
- Shamsudin, S.; Lajis, M.A.; Zhong, Z.W.; Ahmad, A.; Wagiman, A. Weld strength in solid-state recycling of aluminum chips. Mater. Werkst. 2017, 48, 290–298. [Google Scholar] [CrossRef]
- Duflou, J.R.; Tekkaya, A.E.; Haase, M.; Welo, T.; Vanmeensel, K.; Kellens, K.; Dewulf, W.; Paraskevas, D. Environmental assessment of solid state recycling routes for aluminum alloys: Can solid state processes significantly reduce the environmental impact of aluminum recycling? CIRP Ann.-Manuf. Technol. 2015, 64, 37–40. [Google Scholar] [CrossRef]
- Lajis, M.A.; Khamis, S.S.; Yusuf, N.K. Optimization of Hot Press Forging Parameters in Direct Recycling of Aluminum Chip (AA 6061). Key Eng. Mater. 2014, 622–623, 223–230. [Google Scholar] [CrossRef]
- Chawla, N.; Chawla, K.K. Metal Matrix Composites; Wiley Online Library: Berlin, Germany, 2006. [Google Scholar]
- Clyne, T.W.; Withers, P.J. An Introduction to Metal Matrix Composites; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Sajjadi, S.A.; Torabi Parizi, M.; Ezatpour, H.R.; Sedghi, A. Fabrication of A356 composite reinforced with micro and nano Al2O3 particles by a developed compocasting method and study of its properties. J. Alloys Compd. 2012, 511, 226–231. [Google Scholar] [CrossRef]
- Ibrahim, I.A.; Mohamed, F.A.; Lavernia, E.J. Particulate reinforced metal matrix composites—A review. J. Mater. Sci. 1991, 26, 1137–1156. [Google Scholar] [CrossRef]
- Corrochano, J.; Cerecedo, C.; Valcárcel, V.; Lieblich, M.; Guitián, F. Whiskers of Al2O3 as reinforcement of a powder metallurgical 6061 aluminum matrix composite. Mater. Lett. 2008, 62, 103–105. [Google Scholar] [CrossRef]
- Xia, Z.; Ellyin, F.; Meijer, G. Mechanical Behavior Of Al2O3-Particle-Reinforced 6061 Aluminum Alloy Under Uniaxial And Multiaxial Cyclic Loading. Compos. Sci. Technol. 1997, 3538, 237–248. [Google Scholar] [CrossRef]
- Mazahery, A.; Abdizadeh, H.; Baharvandi, H.R. Development of high-performance A356/nano-Al2O3 composites. Mater. Sci. Eng. A 2009, 518, 61–64. [Google Scholar] [CrossRef]
- Akbulut, H.; Durman, M. Temperature dependent strength analysis of short fiber reinforced Al-Si metal matrix composites. Mater. Sci. Eng. A 1999, 262, 214–226. [Google Scholar] [CrossRef]
- Lajis, M.A.; Ahmad, A.; Yusuf, N.K.; Azami, A.H.; Wagiman, A. Mechanical properties of recycled aluminum chip reinforced with alumina (Al2O3) particle. Mater. Werkst. 2017, 48, 306–310. [Google Scholar] [CrossRef]
- Przestacki, D. Conventional and laser assisted machining of composite A359/20SiCp. Procedia CIRP 2014, 14, 229–233. [Google Scholar] [CrossRef]
- Przestacki, D.; Jankowiak, M. Surface roughness analysis after laser assisted machining of hard to cut materials. J. Phys. Conf. Ser. 2014, 483, 12019. [Google Scholar] [CrossRef]
- Przestacki, D.; Kukliński, M.; Bartkowska, A. Influence of laser heat treatment on microstructure and properties of surface layer of Waspaloy aimed for laser-assisted machining. Int. J. Adv. Manuf. Technol. 2017. [Google Scholar] [CrossRef]
- Durrant, G.; Scott, V.D. The effect of forging on the properties and microstructure of saffil fibre reinforced aluminum. Compos. Sci. Technol. 1993, 49, 153–164. [Google Scholar] [CrossRef]
- Yusuf, N.K.; Lajis, M.A.; Daud, M.I.; Noh, M.Z. Effect of Operating Temperature on Direct Recycling Aluminum Chips (AA6061) in Hot Press Forging Process. Appl. Mech. Mater. 2013, 315, 728–732. [Google Scholar] [CrossRef]
- Ahmad, A.; Lajis, M.A.; Yusuf, N.K.; Wagiman, A. Hot Press Forging as The Direct Recycling Technique of Aluminum—A Review. ARPN J. Eng. Appl. Sci. 2016, 11, 2258–2265. [Google Scholar]
- Yusuf, N.K. A New Approach of Direct Recycling Aluminum Alloy Chips (AA6061) in Hot Press Forging Process. Ph.D. Thesis, Universiti Tun Hussein Onn Malaysia, Parit Raja, Malaysia, May 2013. [Google Scholar]
- ASTM 131. Standard Practice for Cleaning of Materials and Components by Ultrasonic Techniques; ASTM International: West Conshohocken, PA, USA, 2012. [Google Scholar]
- ASTM E92. Standard Test Method for Vickers Hardness of Metallic Materials; ASTM International: West Conshohocken, PA, USA, 2004. [Google Scholar]
- Ingel, R.P.; Iii, D.L. Lattice Parameters and Density for Y203-Stabilized ZrO2. J. Am. Ceram. Soc. 1986, 69, 325–332. [Google Scholar] [CrossRef]
- Daoud, A. Wear performance of 2014 Al alloy reinforced with continuous carbon fibers manufactured by gas pressure infiltration. Mater. Lett. 2004, 58, 3206–3213. [Google Scholar] [CrossRef]
- Hesabi, Z.R.; Simchi, A.; Reihani, S.M.S. Structural evolution during mechanical milling of nanometric and micrometric Al2O3 reinforced Al matrix composites. Mater. Sci. Eng. A 2006, 428, 159–168. [Google Scholar] [CrossRef]
- Prabhu, Y.T.; Rao, K.V. X-Ray Analysis by Williamson-Hall and Size-Strain Plot Methods of ZnO Nanoparticles with Fuel Variation. World J. Nano Sci. Eng. 2014, 4, 21–28. [Google Scholar] [CrossRef]
- Li, B.; Sun, Z.; Hou, G.; Ding, F.; Hu, P.; Yuan, F. The effects of alumina reinforcement and nickel activated sintering on nanosized tungsten matrix. J. Alloys Compd. 2017, 692, 420–426. [Google Scholar] [CrossRef]
- Sriraman, K.R.; Ganesh Sundara Raman, S.; Seshadri, S.K. Influence of crystallite size on the hardness and fatigue life of steel samples coated with electrodeposited nanocrystalline Ni-W alloys. Mater. Lett. 2007, 61, 715–718. [Google Scholar] [CrossRef]
- Firestein, K.L.; Corthay, S.; Steinman, A.E.; Matveev, A.T.; Kovalskii, A.M.; Sukhorukova, I.V.; Golberg, D.; Shtansky, D.V. High-strength aluminum-based composites reinforced with BN, AlB2 and AlN particles fabricated via reactive spark plasma sintering of Al-BN powder mixtures. Mater. Sci. Eng. A 2017, 681, 1–9. [Google Scholar] [CrossRef]
- Kallip, K.; Leparoux, M.; AlOgab, K.A.; Clerc, S.; Deguilhem, G.; Arroyo, Y.; Kwon, H. Investigation of different carbon nanotube reinforcements for fabricating bulk AlMg5 matrix nanocomposites. J. Alloys Compd. 2015, 646, 710–718. [Google Scholar] [CrossRef]
- Hansen, N.; Huang, X.; Hughes, D.A. Microstructural evolution and hardening parameters. Mater. Sci. Eng. A 2001, 317, 3–11. [Google Scholar] [CrossRef]
- Hansen, N.; Huang, X.; Winther, G. Grain orientation, deformation microstructure and flow stress. Mater. Sci. Eng. A 2008, 494, 61–67. [Google Scholar] [CrossRef]
- Al-Mosawi, B.T.; Wexler, D.; Calka, A. Characterization and mechanical properties of α-Al2O3 particle reinforced aluminum matrix composites, synthesized via uniball magneto-milling and uniaxial hot pressing. Adv. Powder Technol. 2016, 28, 1054–1064. [Google Scholar] [CrossRef]
- Nguyen, Q.B.; Tun, K.S.; Chan, J.; Kwok, R.; Kuma, J.V.M.; Gupta, M. Enhancing strength and hardness of AZ31B through simultaneous addition of nickel and nano-Al2O3 particulates. Mater. Sci. Eng. A 2011, 528, 888–894. [Google Scholar] [CrossRef]
- Nagaral, M. Effect of Al2O3 Particles on Mechanical and Wear Properties of 6061al Alloy Metal Matrix Composites. J. Mater. Sci. Eng. 2013, 2, 2–5. [Google Scholar] [CrossRef]
- Rahimian, M.; Ehsani, N.; Parvin, N.; Baharvandi, H.R. The effect of sintering temperature and the amount of reinforcement on the properties of Al-Al2O3 composite. Mater. Des. 2009, 30, 3333–3337. [Google Scholar] [CrossRef]
- Rahimian, M.; Parvin, N.; Ehsani, N. Investigation of particle size and amount of alumina on microstructure and mechanical properties of Al matrix composite made by powder metallurgy. Mater. Sci. Eng. A 2010, 527, 1031–1038. [Google Scholar] [CrossRef]
- Fultz, B.; Howe, J.M. Transmission Electron Microscopy and Diffractometry of Materials; Springer: Berlin, Germany, 2007; ISBN 978-3-642-29760-1. [Google Scholar]
- Deb, A.K.; Chatterjee, P.; Sen Gupta, S.P. An X-ray diffraction study on dislocation microstructure of as-prepared Al-Al2O3 composites. Acta Mater. 2004, 52, 2755–2764. [Google Scholar] [CrossRef]
- Endler, I.; Höhn, M.; Schmidt, J.; Scholz, S.; Herrmann, M.; Knaut, M. Ternary and quarternary TiSiN and TiSiCN nanocomposite coatings obtained by Chemical Vapor Deposition. Surf. Coat. Technol. 2013, 215, 133–140. [Google Scholar] [CrossRef]
- Serban, N.; Cojocaru, V.-D.; Butu, M. Mechanical behavior and microstructural development of 6063-T1 aluminum alloy processed by equal-channel angular pressing (ECAP): Pass number influence. JOM 2012, 64, 607–614. [Google Scholar] [CrossRef]
- Serban, N.; Ghiban, N.; Cojocaru, V.-D. Mechanical behavior and microstructural development of 6063-T1 aluminum alloy processed by equal-channel angular pressing (ECAP): Die channel angle influence. JOM 2013, 65, 1411–1418. [Google Scholar] [CrossRef]
- Humphreys, F.J.; Hatherly, M. Chapter 4: The Structure and Energy of Grain. Recryst. Relat. Annealing Phenom. 2004, 91–119. [Google Scholar]
- Balog, M.; Poletti, C.; Simancik, F.; Walcher, M.; Rajner, W. The effect of native Al2O3 skin disruption on properties of fine Al powder compacts. J. Alloys Compd. 2011, 509, 235–238. [Google Scholar] [CrossRef]
- Yusuf, N.K.; Lajis, M.A.; Ahmad, A. Hot Press as a Sustainable Direct Recycling Technique of Aluminum: Mechanical Properties and Surface Integrity. Materials 2017, 10, 902. [Google Scholar] [CrossRef] [PubMed]
- Ceschini, L.; Minak, G.; Morri, A. Forging of the AA2618/20vol.% Al2O3p composite: Effects on microstructure and tensile properties. Compos. Sci. Technol. 2009, 69, 1783–1789. [Google Scholar] [CrossRef]
- Shen, Y.-L.; Chawla, N. On the correlation between hardness and tensile strength in particle reinforced metal matrix composites. Mater. Sci. Eng. A 2001, 297, 44–47. [Google Scholar] [CrossRef]
- Kume, Y.; Ota, S.; Kobashi, M.; Kanetake, N. Upgrading Property of A-Fe Alloys through the Microstructure Refinement by Compressive Torsion Processing. Mater. Sci. Forum 2014, 794, 802–806. [Google Scholar] [CrossRef]
- Kume, Y.; Kobashi, M.; Kanetake, N. Microstructure Refinement of Al-Si Alloy Using Compressive Torsion Processing. Mater. Sci. Forum 2006, 519–521, 1441–1446. [Google Scholar] [CrossRef]
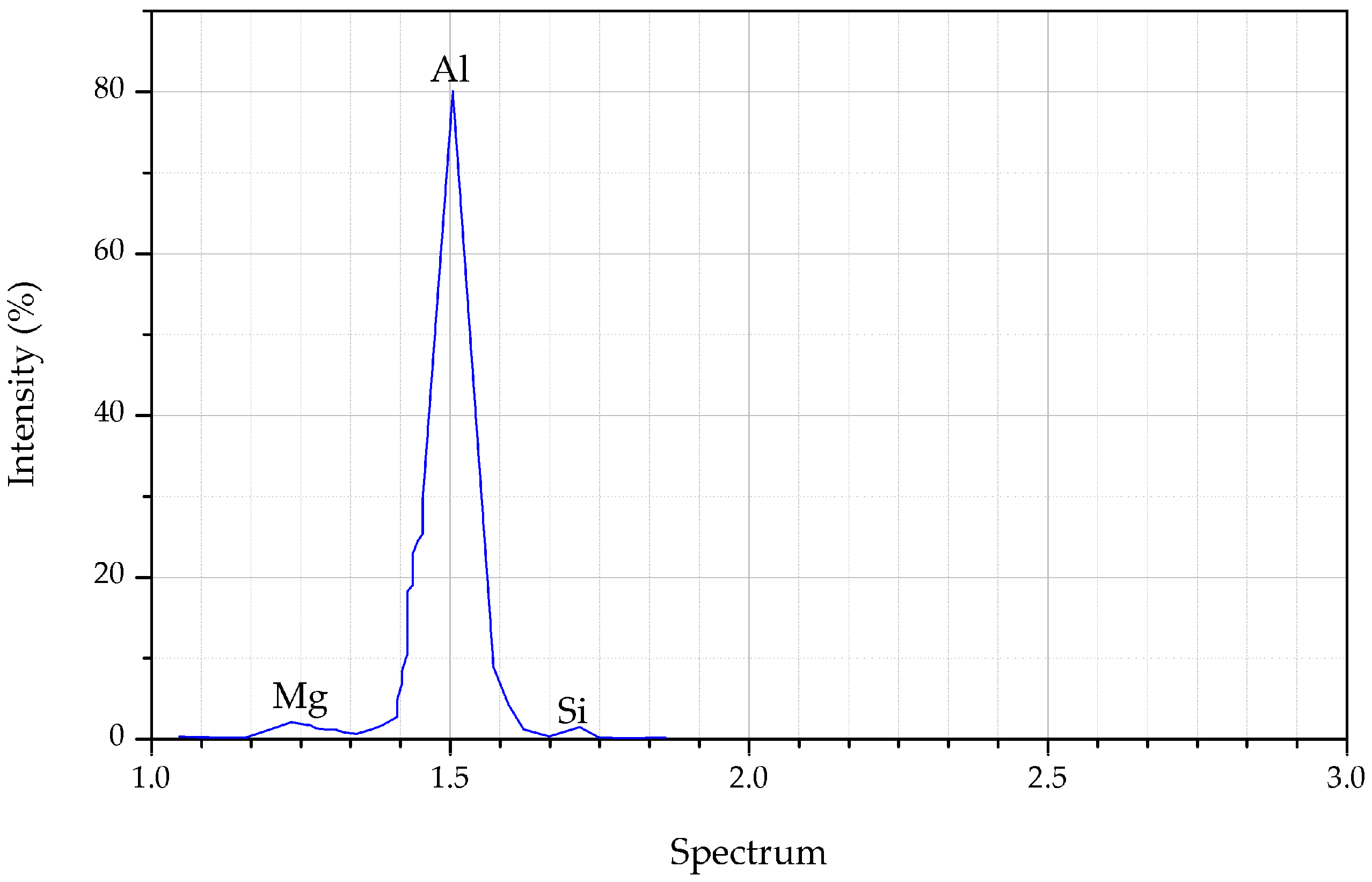



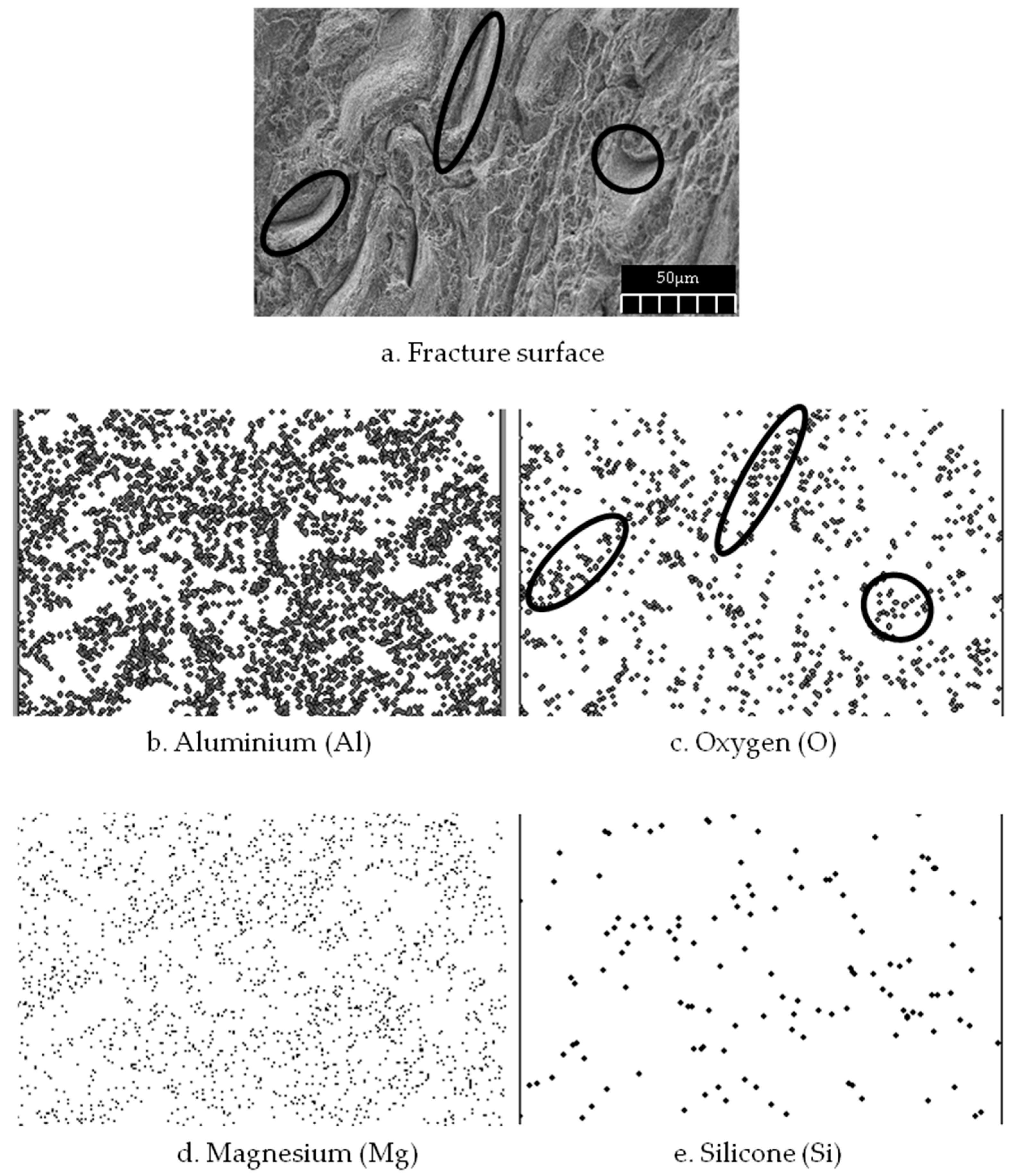
| Parameter | Nomenclature | Value |
|---|---|---|
| Matrix volume fraction | (%) | 98.0 |
| Reinforcement volume fraction | (%) | 2.0 |
| Matrix density | (g/cm3) | 2.667 |
| Reinforcement density | (g/cm3) | 3.916 |
| Matrix hardness | (HV) | 95.512 |
| Reinforcement hardness | (HV) | 2700 |
| Parameter | Value | Chip Morphology |
|---|---|---|
| Cutting Speed, v | 1100 m/min |  |
| Feed, f | 0.05 mm/tooth | |
| Depth of Cut, DOC | 1.0 mm |
| Temperature (°C) | Holding Time (min) | Specimen Designation |
|---|---|---|
| 430 | 60 | R1 |
| 430 | 90 | R2 |
| 430 | 120 | R3 |
| 480 | 60 | R4 |
| 480 | 90 | R5 |
| 480 | 120 | R6 |
| 530 | 60 | R7 |
| 530 | 90 | R8 |
| 530 | 120 | R9 |
| Specimen | Vickers Hardness (HV0.3) | Density (g/cc) |
|---|---|---|
| R1 | 71.360 | 2.314 |
| R2 | 73.367 | 2.487 |
| R3 | 74.625 | 2.512 |
| R4 | 76.432 | 2.608 |
| R5 | 76.988 | 2.632 |
| R6 | 78.454 | 2.653 |
| R7 | 80.592 | 2.654 |
| R8 | 83.921 | 2.657 |
| R9 | 86.656 | 2.684 |
| AR | 95.512 | 2.667 |
| Specimen |  |  |  |
|---|---|---|---|
| Number of Intercepts | 95 | 131 | 202 |
| Mean Intercepts length (mm) | 53.28 | 38.64 | 25.05 |
| G number | 5.17 | 6.09 | 7.34 |
| Average Grain Diameter (μm) | 60.07 | 43.62 | 28.33 |
| Specimen | Peak Height (cts) | d Spacing (Å) | Crystallite Size (Å) |
|---|---|---|---|
| R1 | 14,535 | 2.34677 | 739.9 |
| R9 | 10,452 | 2.34641 | 770.6 |
| AR | 9723 | 2.34527 | 785.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, A.; Lajis, M.A.; Yusuf, N.K. On the Role of Processing Parameters in Producing Recycled Aluminum AA6061 Based Metal Matrix Composite (MMC-AlR) Prepared Using Hot Press Forging (HPF) Process. Materials 2017, 10, 1098. https://doi.org/10.3390/ma10091098
Ahmad A, Lajis MA, Yusuf NK. On the Role of Processing Parameters in Producing Recycled Aluminum AA6061 Based Metal Matrix Composite (MMC-AlR) Prepared Using Hot Press Forging (HPF) Process. Materials. 2017; 10(9):1098. https://doi.org/10.3390/ma10091098
Chicago/Turabian StyleAhmad, Azlan, Mohd Amri Lajis, and Nur Kamilah Yusuf. 2017. "On the Role of Processing Parameters in Producing Recycled Aluminum AA6061 Based Metal Matrix Composite (MMC-AlR) Prepared Using Hot Press Forging (HPF) Process" Materials 10, no. 9: 1098. https://doi.org/10.3390/ma10091098
APA StyleAhmad, A., Lajis, M. A., & Yusuf, N. K. (2017). On the Role of Processing Parameters in Producing Recycled Aluminum AA6061 Based Metal Matrix Composite (MMC-AlR) Prepared Using Hot Press Forging (HPF) Process. Materials, 10(9), 1098. https://doi.org/10.3390/ma10091098





