Effect of Coal Rank on Various Fluid Saturations Creating Mechanical Property Alterations Using Australian Coals
Abstract
:1. Introduction
2. Methodology
2.1. Samples Used for Testing
2.2. Samples Preparation for Testing
2.2.1. Water Saturation
2.2.2. N2 Saturation
2.3. Experimental Procedure
2.3.1. Unconfined Compressive Strength (UCS) Tests
2.3.2. Acoustic Emission (AE) Tests
2.3.3. Scanning Electron Microscopy (SEM) Analysis
3. Experimental Results and Discussion
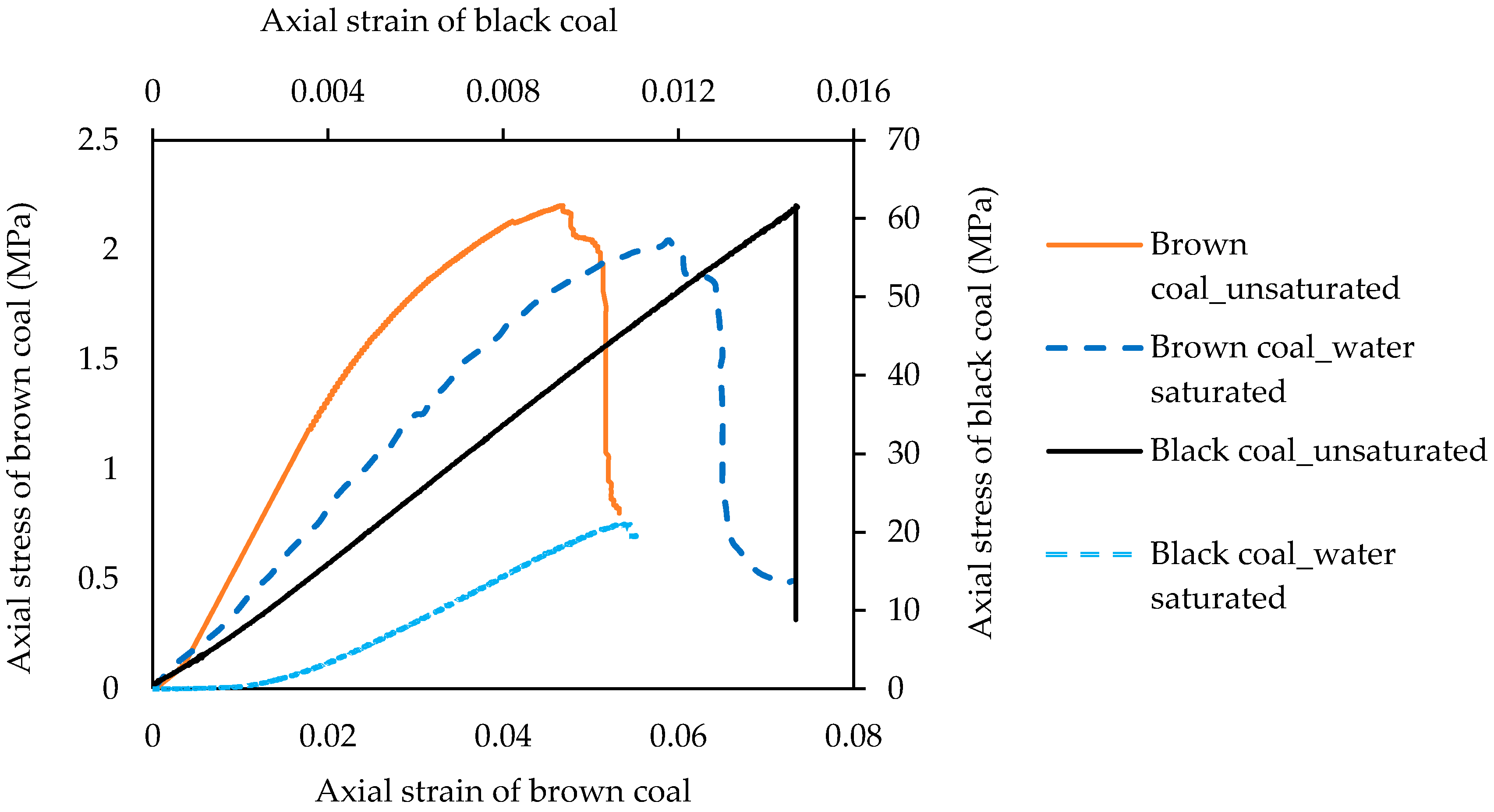
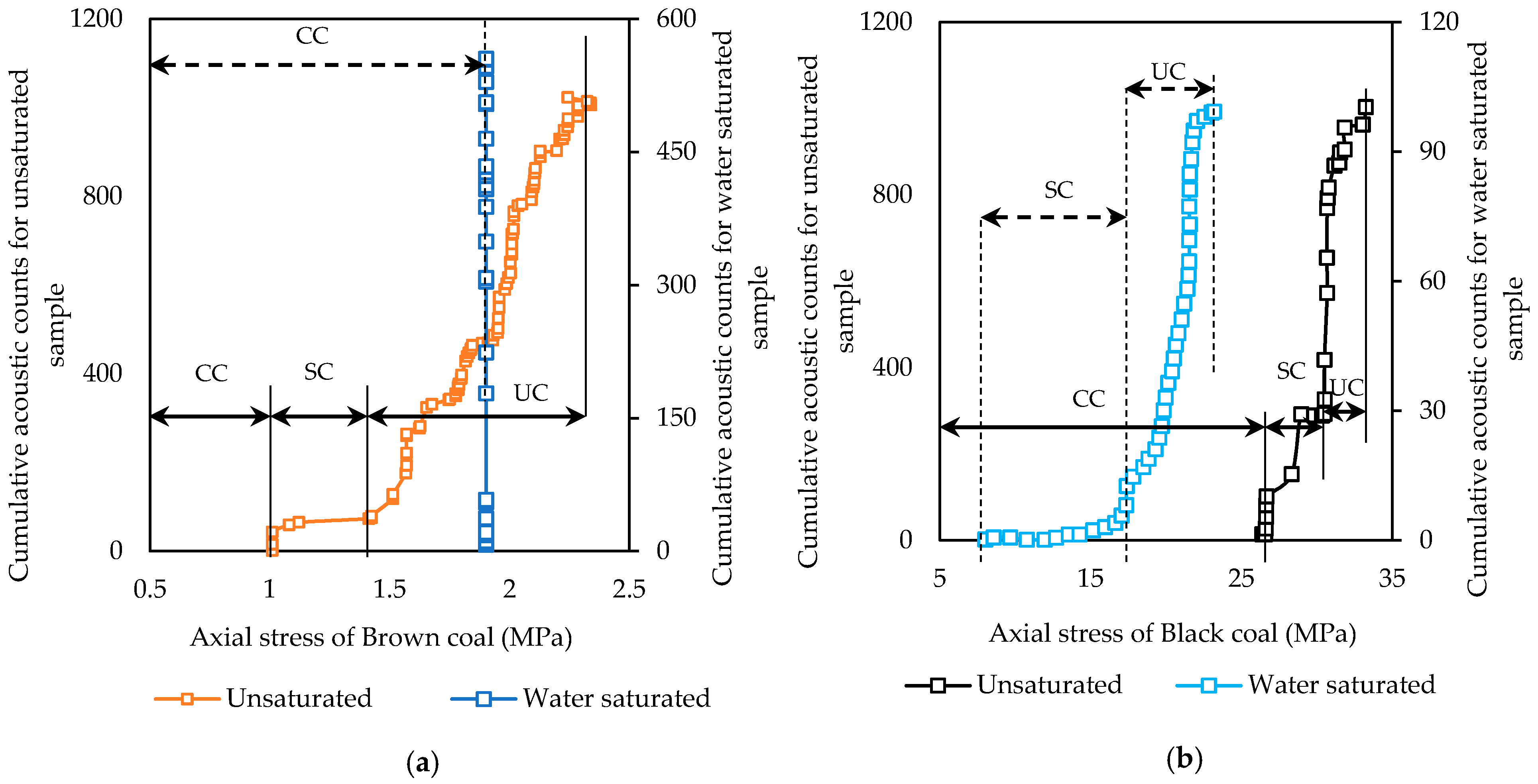
3.1. Effect of Coal Rank on Water Saturation Effect on Geomechanical Properties in Coal
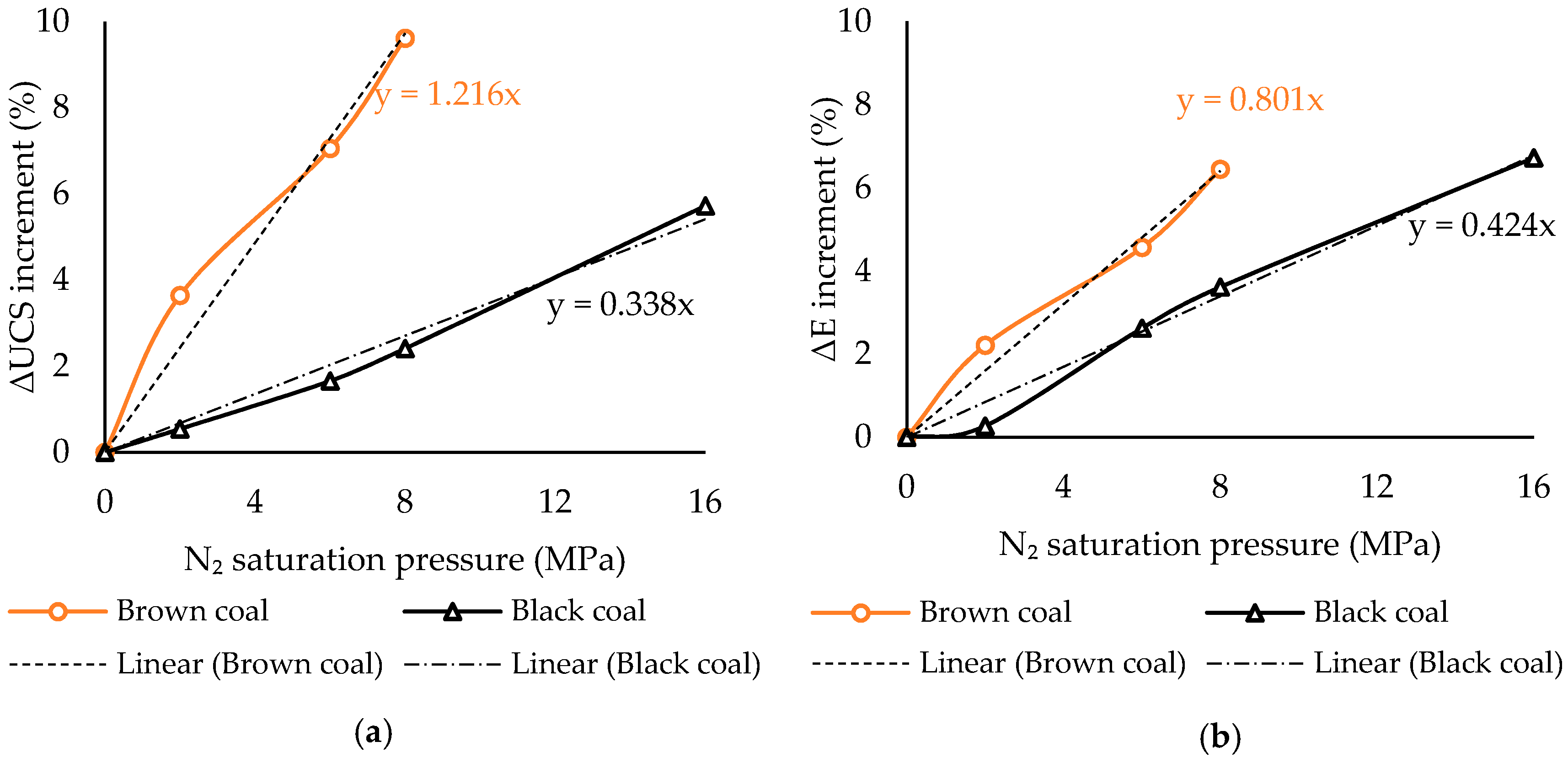
3.2. Rank Effect on N2 Saturation-Created Geomechanical Alterations in Coal
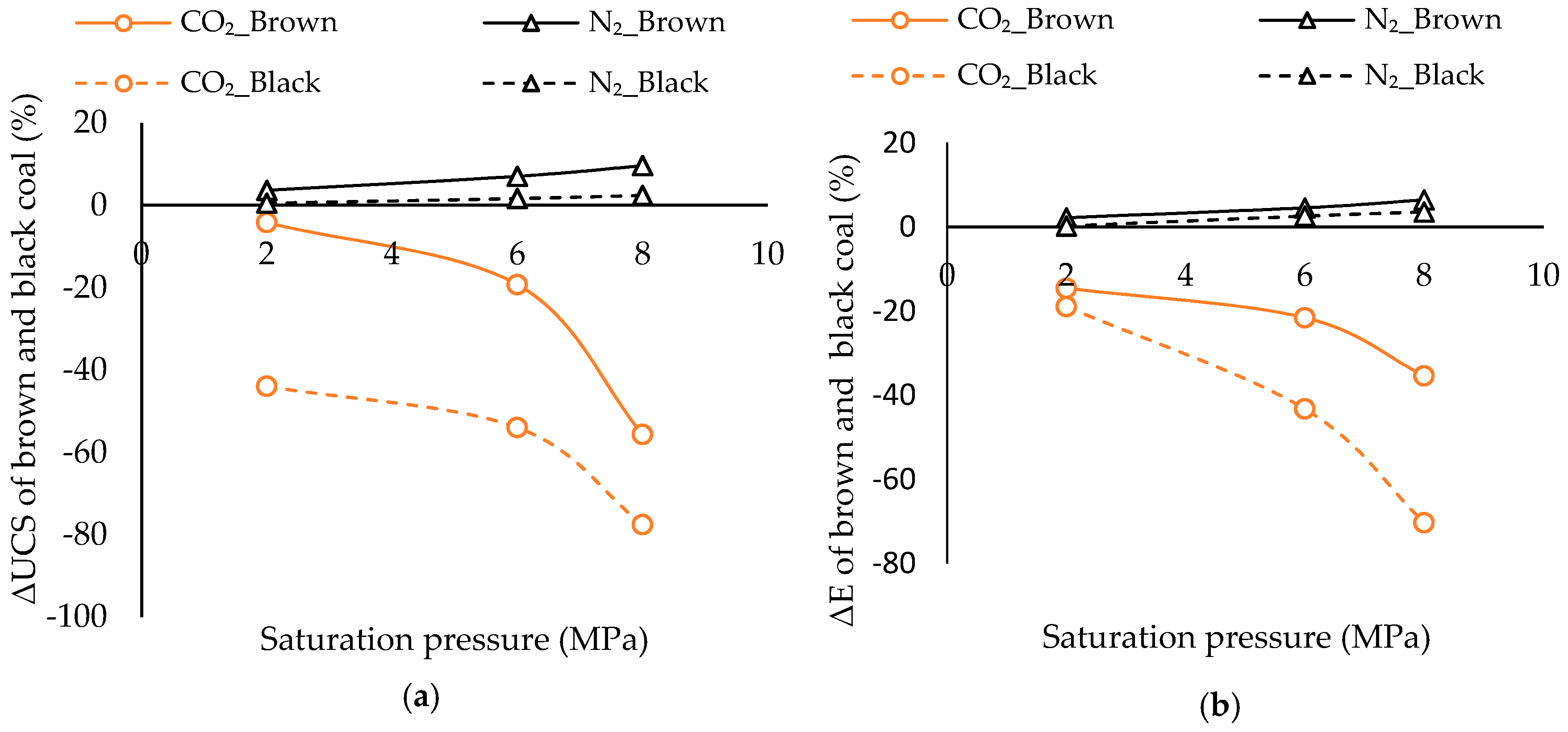
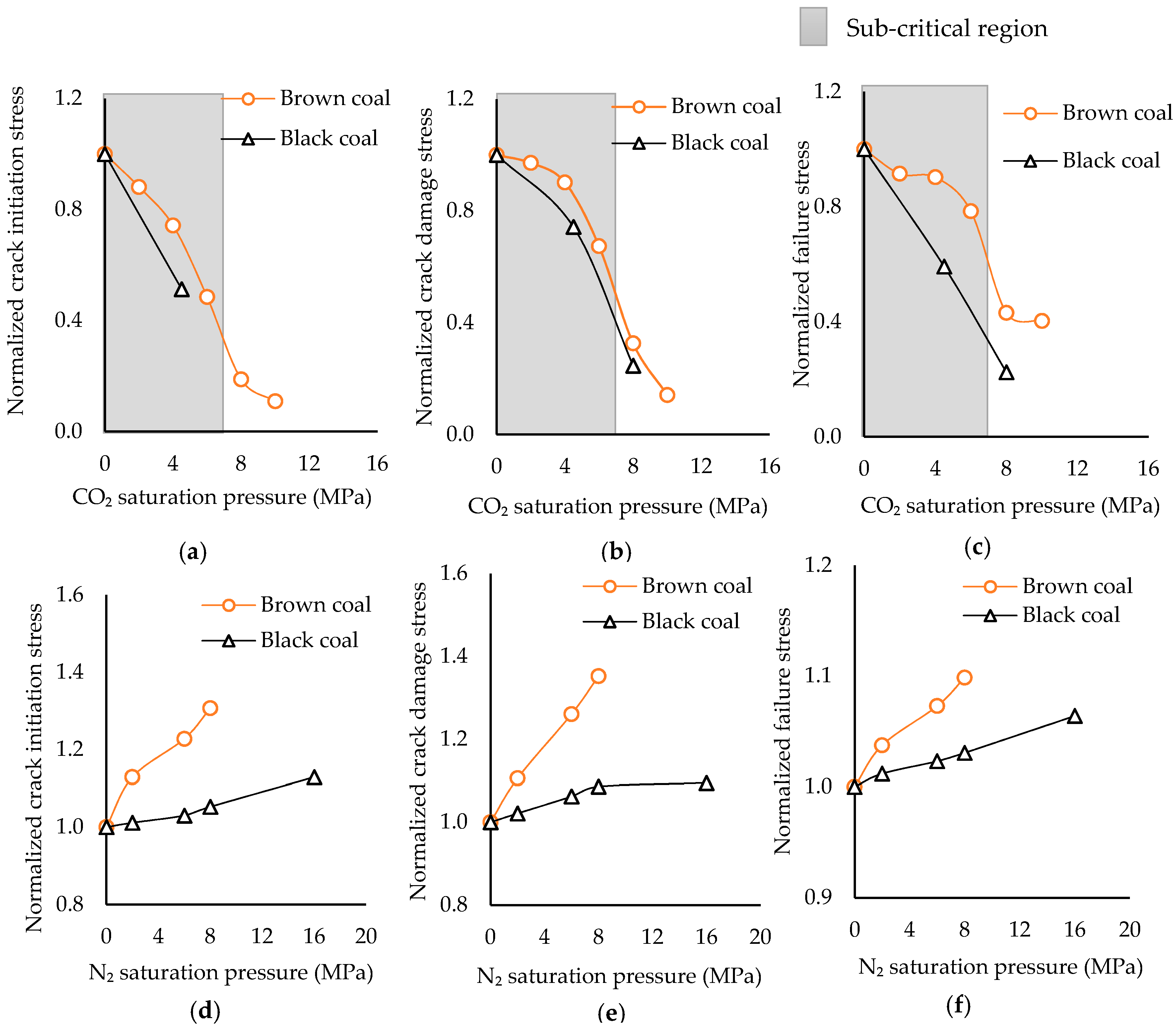
3.3. How Significant is Water and N2 Saturation on Coal Strength Compared to That of CO2
3.3.1. Comparison of Variation of Coal’s Mechanical Properties
3.3.2. Comparison of Fracture Propagation
4. Conclusions and Suggestions for Future Research
4.1. Conclusions Drawn from the Present Study
- Water saturation causes a significant strength reduction in coal regardless of rank, because the moisture penetrating into the coal mass causes its fractures to expand, decreasing the surface energy of the coal mass, and eventually causing the strength to be reduced.
- This strength reduction is enhanced with increasing coal rank due to the well-developed cleats in mature coal which offer more loci for moisture to stay in the coal mass.
- The enhanced ductile nature of coal upon water adsorption delays crack initiation, regardless of rank. It is therefore necessary to have a lot of low injection pressure for coal seams with a greater degree of water saturation in terms of safety.
- In contrast to water saturation, N2 saturation slightly enhances coal strength, regardless of rank, and this increment increases with increasing N2 saturation pressures. This is possibly due to the ability of N2 to partially recover the coal seam gas (methane, CO2) adsorbed swelled areas and push out the adsorbed water from the coal mass.
- N2 saturation significantly slows crack initiation and propagation in coal regardless of rank. This is a favorable feature in terms of long-term stability in coal seams and can be used to strengthen weak seams after CO2 injection.
- The strength gain created by N2 saturation reduces with increasing rank, probably due to the lower level of moisture stored in high rank coal.
- The variation of mechanical properties of coal upon CO2 and N2 saturations shows quite different responses to increasing saturation pressure, as the CO2 phase condition may change from sub- to super-critical under greater pressures. In contrast, N2 does not undergo such phase change with increasing pressure, due to its much lower critical conditions.
- The present study was conducted under 35 °C temperature (N2 saturation) up to 8 MPa saturation pressure, which represents a pore pressure condition of an approximately 800 m deep coal seam [31]. As the study was conducted in an unconfined environment, the observed strength variations are expected to be lower in a confined environment under field conditions, because the confinement causes greater effective stress in the field, leading to a shrunken pore structure in coal seams with reduced gas flow performance through them.
4.2. Suggestions for Furture Research
- Methane is one of the major components in coal seam gas that plays an important role in the overall mechanical response of the coal mass. Hence, to understand the overall influence of coal seam gas on coal’s mechanical properties, future research is needed on methane saturation under various pressures for different rank coals.
- It is recommended to conduct further research on coal mass mechanical behavior under confinements for different saturation conditions to represent the real case scenarios existing in the field, where the coal mass has been exposed to lithostatic pressure conditions.
- The water saturation effect of the present study was conducted under atmospheric pressure conditions due to the unavailability of the necessary laboratory facilities. However, it is essential to investigate the water saturation effect under different pressure conditions, so that it can be clearly compared with the different pressure effects of N2 and CO2. This is a future research option.
- As found in the present study, coal mass mechanical behavior under various saturations varies with different coal types. However, the results of this study need to be generalized by testing various ranked coal samples taken from various coal basins around the world. Future experimentation is therefore recommended using a wider range of coal specimens taken from different basins in the world with diverse cleat structures and mineral compositions, in order to obtain better insight into the observed strength variations.
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AE | Acoustic emission |
| CC | Crack closure |
| CH4 | Methane |
| CO2 | Carbon dioxide |
| db | Dry basis |
| E | Young’s modulus |
| ECBM | Enhanced coal bed methane recovery |
| MCEM | Monash Center of Electron Microscopy |
| N2 | Nitrogen |
| SC | Stable crack propagation |
| SEM | Scanning electron microscopy |
| UC | Unstable crack propagation |
| UCS | Unconfined compressive strength |
| wb | Wet basis |
| σcd | Crack damage stress |
| σci | Crack initiation stress |
References
- Perera, M.S.A.; Ranjith, P.G. Carbon dioxide sequestration effects on coal’s hydro-mechanical properties: A review. Int. J. Energ. Res. 2012, 36, 1015–1031. [Google Scholar] [CrossRef]
- Ranathunga, A.S.; Perera, M.S.A.; Ranjith, P.G. Deep coal seams as a greener energy source: A review. J. Geophys. Eng. 2014, 11, 063001. [Google Scholar] [CrossRef]
- Vishal, V.; Singh, T.N. A laboratory investigation of permeability of coal to supercritical CO2. Geotech. Geol. Eng. 2015, 33, 1009–1016. [Google Scholar] [CrossRef]
- Xie, J.; Gao, M.; Yu, B.; Zhang, R.; Jin, W. Coal permeability model on the effect of gas extraction within effective influence zone. Geomech. Geophys. Geo-Energy Geo-Resources 2015, 1, 15–27. [Google Scholar] [CrossRef]
- Gale, J. Geological storage of CO2: What do we know, where are the gaps and what more needs to be done? Energy 2004, 29, 1329–1338. [Google Scholar] [CrossRef]
- Zhang, Y.; Gong, B.; Li, J.; Li, H. Discrete fracture modeling of 3D heterogeneous enhanced coalbed methane recovery with prismatic meshing. Energies 2015, 8, 6153–6176. [Google Scholar] [CrossRef]
- Hu, G.; Xu, J.; Zhang, F.; Zhao, C.; Qin, W.; Zhu, Y. Coal and coalbed methane co-extraction technology based on the ground movement in the yangquan coalfield, China. Energies 2015, 8, 6881–6897. [Google Scholar] [CrossRef]
- Aziz, N.I.; Ming-Li, W. The effect of sorbed gas on the strength of coal – an experimental study. Geotech. Geol. Eng. 1999, 17, 387–402. [Google Scholar] [CrossRef]
- Jasinge, D. An investigation of the effect of carbon dioxide sequestration on the behaviour of brown coal. Ph.D. Thesis, Monash University, Melbourne, Australia, 2010. [Google Scholar]
- Viete, D.R.; Ranjith, P.G. The effect of CO2 on the geomechanical and permeability behaviour of brown coal: Implications for coal seam CO2 sequestration. Int. J. Coal Geol. 2006, 66, 204–216. [Google Scholar] [CrossRef]
- White, C.M.; Smith, D.H.; Jones, K.L.; Goodman, A.L.; Jikich, S.A.; LaCount, R.B.; DuBose, S.B.; Ozdemir, E.; Morsi, B.I.; Schroeder, K.T. Sequestration of carbon dioxide in coal with enhanced coalbed methane recovery: A review. Energ. Fuel 2005, 19, 659–724. [Google Scholar] [CrossRef]
- Pan, Z.; Connell, L.D.; Camilleri, M. Laboratory characterisation of coal reservoir permeability for primary and enhanced coalbed methane recovery. Int. J. Coal Geol. 2010, 82, 252–261. [Google Scholar] [CrossRef]
- Gibbs, J.W. On the equilibrium of heterogeneous substances. Am. J. Sci. 1878, 16, 441–458. [Google Scholar] [CrossRef]
- Griffith, A.A. The phenomena of rupture and flow in solids. Phil. Trans. R. Soc. Lond. Ser. A 1921, 221, 163–198. [Google Scholar] [CrossRef]
- Perera, M.S.A.; Ranjith, P.G.; Viete, D.R. Effects of gaseous and super-critical carbon dioxide saturation on the mechanical properties of bituminous coal from the southern sydney basin. Appl. Energy 2013, 110, 73–81. [Google Scholar] [CrossRef]
- Ranathunga, A.S.; Perera, M.S.A.; Ranjith, P.G.; Bui, H. Super-critical CO2 saturation-induced mechanical property alterations in low rank coal: An experimental study. J. Supercrit. Fluids 2016, 109, 134–140. [Google Scholar] [CrossRef]
- Vishal, V.; Ranjith, P.; Singh, T. An experimental investigation on behaviour of coal under fluid saturation, using acoustic emission. J. Nat. Gas Sci. Eng. 2015, 22, 428–436. [Google Scholar] [CrossRef]
- Ates, Y.; Barron, K. The effect of gas sorption on the strength of coal. Min. Sci. Technol. 1988, 6, 291–300. [Google Scholar] [CrossRef]
- Brady, B.; Brown, E. Rock Mechanics: For Underground Mining, 3rd ed.; Springer: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Jasinge, D.; Ranjith, P.; Choi, X.; Fernando, J. Investigation of the influence of coal swelling on permeability characteristics using natural brown coal and reconstituted brown coal specimens. Energy 2012, 39, 303–309. [Google Scholar] [CrossRef]
- Kiyama, T.; Nishimoto, S.; Fujioka, M.; Xue, Z.; Ishijima, Y.; Pan, Z.; Connell, L.D. Coal swelling strain and permeability change with injecting liquid/supercritical CO2 and N2 at stress-constrained conditions. Int. J. Coal Geol. 2011, 85, 56–64. [Google Scholar] [CrossRef]
- Perera, M.S.A.; Ranjith, P.G.; Choi, S.K.; Airey, D. The effects of sub-critical and super-critical carbon dioxide adsorption-induced coal matrix swelling on the permeability of naturally fractured black coal. Energy 2011, 36, 6442–6450. [Google Scholar] [CrossRef]
- Ranathunga, A.S.; Perera, M.S.A.; Ranjith, P.G.; Ju, Y.; Vishal, V.; De Silva, P. A macro-scale experimental study of sub-and super-critical CO2 flow behaviour in victorian brown coal. Fuel 2015, 158, 864–873. [Google Scholar] [CrossRef]
- Perera, M.S.A.; Ranjith, P.G.; Peter, M. Effects of saturation medium and pressure on strength parameters of Latrobe Valley brown coal: Carbon dioxide, water and nitrogen saturations. Energy 2012, 36, 6941–6947. [Google Scholar] [CrossRef]
- Ranjith, P.G.; Perera, M.S.A. Effects of cleat performance on strength reduction of coal in CO2 sequestration. Energy 2012, 45, 1069–1075. [Google Scholar] [CrossRef]
- Perera, M.S.A.; Ranjith, P.G.; Airey, D.W.; Choi, S.K. Sub- and super-critical carbon dioxide flow behavior in naturally fractured black coal: An experimental study. Fuel 2011, 90, 3390–3397. [Google Scholar] [CrossRef]
- ASTM D7012. Standard Test Method for Compressive Strength and Elastic Moduli of Intact Rock Core Specimens un-der Varying States of Stress and Temperatures; ASTM International: West Conshohocken, PA, USA, 2004. [Google Scholar]
- Vutukuri, V.S.; Lama, R.D.; Saluja, S.S. Handbook on Mechanical Properties of Rocks: Testing Techniques and Results; Trans Tech Publications: Bay Village, OH, USA, 1978; pp. 105–115. [Google Scholar]
- Kuvaev, N.; Panchenko, D. Effect of moisture on the dynamic strength of brown coals. J. Min. Sci. 1975, 11, 220–222. [Google Scholar] [CrossRef]
- Ranjith, P.G.; Jasinge, D.; Choi, S.K.; Mehic, M. The effect of CO2 saturation on mechanical properties of Australian black coal using acoustic emission. Fuel 2010, 89, 2110–2117. [Google Scholar] [CrossRef]
- Oldenburg, C.M. Geologic carbon sequestration: CO2 transport in depleted gas reservoirs. In Gas Transport in Porous Media; Springer: Dordrecht, The Netherlands, 2006; pp. 419–426. [Google Scholar]

| Temperature (°C) | Pressure (MPa) | CO2 Phase Condition | Coal Type | Coal Rank | UCS (MPa) | ΔUCS (%) | E (MPa) | ΔE (%) | Reference |
|---|---|---|---|---|---|---|---|---|---|
| 35 | 0 | - | Lignite 1 | Low | 2.40 | - | 41.60 | - | [16] |
| 33 | 0 | - | Bituminous 2 | High | 33.00 | - | 3700 | - | [15] |
| Room temperature | 0 | - | Bituminous 3 | High | 15.29 | - | 5340 | - | [17] |
| Room temperature | 1 | Gas | Lignite 1 | Low | 2.34 | 2.09 | 40.32 | 4.73 | [16] |
| 35 | 2 | Gas | Lignite 1 | Low | 2.25 | 5.86 | 35.08 | 17.11 | [16] |
| 35 | 3 | Gas | Lignite 1 | Low | 2.17 | 9.21 | 33.78 | 20.18 | [16] |
| Room temperature | 4 | Gas | Bituminous 3 | High | 12.62 | 17.60 | 3940 | 26.20 | [17] |
| 35 | 4 | Gas | Lignite 1 | Low | 2.15 | 10.04 | 33.65 | 20.49 | [16] |
| 33 | 4.5 | Gas | Bituminous 2 | High | 15.80 | 52.12 | 2300 | 37.84 | [15] |
| 35 | 6 | Gas | Lignite 1 | Low | 1.89 | 20.92 | 32.23 | 23.84 | [16] |
| 33 | 6 | Gas | Bituminous 2 | High | 14.30 | 56.67 | 2130 | 42.43 | [15] |
| 35 | 8 | Super critical | Lignite 1 | Low | 1.02 | 57.32 | 24.91 | 41.14 | [16] |
| 33 | 8 | Super critical | Bituminous 2 | High | 7.40 | 77.58 | 1100 | 70.27 | [15] |
| 35 | 10 | Super critical | Lignite 1 | Low | 0.93 | 61.09 | 23.42 | 44.69 | [16] |
| 33 | 12 | Super critical | Bituminous 2 | High | 7.30 | 77.88 | 1160 | 68.65 | [15] |
| 33 | 16 | Super critical | Bituminous 2 | High | 13.95 | 57.64 | 1570 | 57.58 | [15] |
| Physical Property | Brown Coal [16] | Black Coal [15] |
|---|---|---|
| Coal density (g/cm3) | 1.04–1.1 | 1.4–1.9 |
| Moisture content (% wb) 1 | 57–66 | 1–5 |
| Fixed carbon (% db) 1 | 48 | 60–71 |
| Ash yield (% db) 1 | 1.7 | 6–12 |
| Volatile matter content (% db) 1 | 50.3 | 10–25 |
| Vitrinite reflectance (%) | 0.0–0.4 2 | 1.1–1.5 2 |
| Saturation Fluid | Pressure (MPa) | Brown Coal | Black Coal | ||||||
|---|---|---|---|---|---|---|---|---|---|
| UCS (MPa) | ΔUCS (%) 3 | E (MPa) | ΔE (%) 3 | UCS (MPa) | ΔUCS (%) 3 | E (GPa) | ΔE (%) 3 | ||
| Unsaturated | - | 2.39 1 | - | 42.32 1 | - | 31.01 2 | - | 3.52 2 | - |
| - | 2.34 1 | - | 39.32 1 | - | 33.00 2 | - | 3.70 2 | - | |
| - | 2.36 1 | - | 42.16 1 | - | - | - | - | - | |
| Water | - | 2.07 | −14.64 | 35.46 | −16.21 | 21.01 | −36.33 | 2.24 | −39.46 |
| - | 2.01 | 35.17 | 20.89 | 2.14 | |||||
| - | 2.05 | 35.28 | 20.94 | 2.20 | |||||
| N2 | 2 | 2.43 | 1.58 | 42.52 | 0.47 | 33.39 | 1.18 | 3.71 | 0.27 |
| 2 | 2.39 | 41.97 | 32.17 | 3.54 | |||||
| 6 | 2.51 | 5.05 | 43.08 | 1.80 | 33.76 | 2.30 | 3.76 | 1.62 | |
| 6 | 2.48 | 43.01 | 32.89 | 3.64 | |||||
| 8 | 2.57 | 7.56 | 44.28 | 4.63 | 34.01 2 | 3.06 | 3.80 2 | 2.70 | |
| 8 | 2.54 | 44.16 | 33.20 2 | 3.71 2 | |||||
| 16 | - | - | - | - | 35.11 2 | 6.39 | 3.91 2 | 5.68 | |
| 16 | 33.30 2 | 3.71 2 | |||||||
| Coal Type | Sample Appearance Under Different Test Conditions | SEM Images 3 | Average Pore Size | |
|---|---|---|---|---|
| Natural samples | ||||
| Brown coal |  |  |  | ~0.9 μm |
| Before failure 1 | After failure 1 | |||
| Black coal |  |  |  | ~0.43 μm |
| Before failure 2 | After failure 2 | |||
| Water-saturated samples | ||||
| Brown coal |  |  |  | ~0.34 μm |
| Before failure | After failure | |||
| Black coal |  |  |  | ~0.12 μm |
| Before failure | After failure | |||
| N2 saturated samples | ||||
| Brown coal |  |  |  | ~0.62 μm |
| After saturation in 8 MPa N2 | After saturation in 8 MPa N22 | |||
| Black coal |  |  |  | ~0.23 μm |
| After failure | After failure 2 | |||
| Saturation Fluid | Saturation Pressure (MPa) | Brown Coal | Black Coal | ||||
|---|---|---|---|---|---|---|---|
| Crack Initiation Stress (σci, MPa) | Crack Damage Stress (σcd, MPa) | UCS (MPa) | Crack Initiation Stress (σci, MPa) | Crack Damage Stress (σcd, MPa) | UCS (MPa) | ||
| Unsaturated | - | 1.01 1 | 1.42 1 | 2.39 1 | 26.70 2 | 30.50 2 | 33.00 2 |
| Water | - | - | - | 2.06 | - | 17.15 | 20.94 |
| N2 | 2 | 1.14 | 1.57 | 2.43 | 27.10 | 29.38 | 33.39 |
| 6 | 1.24 | 1.79 | 2.51 | 27.53 | 30.43 | 33.76 | |
| 8 | 1.32 | 1.92 | 2.57 | 28.10 2 | 31.90 2 | 34.01 2 | |
| 16 | - | - | - | 30.14 2 | 32.16 2 | 35.11 2 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perera, M.S.A.; Ranathunga, A.S.; Ranjith, P.G. Effect of Coal Rank on Various Fluid Saturations Creating Mechanical Property Alterations Using Australian Coals. Energies 2016, 9, 440. https://doi.org/10.3390/en9060440
Perera MSA, Ranathunga AS, Ranjith PG. Effect of Coal Rank on Various Fluid Saturations Creating Mechanical Property Alterations Using Australian Coals. Energies. 2016; 9(6):440. https://doi.org/10.3390/en9060440
Chicago/Turabian StylePerera, Mandadige Samintha Anne, Ashani Savinda Ranathunga, and Pathegama Gamage Ranjith. 2016. "Effect of Coal Rank on Various Fluid Saturations Creating Mechanical Property Alterations Using Australian Coals" Energies 9, no. 6: 440. https://doi.org/10.3390/en9060440
APA StylePerera, M. S. A., Ranathunga, A. S., & Ranjith, P. G. (2016). Effect of Coal Rank on Various Fluid Saturations Creating Mechanical Property Alterations Using Australian Coals. Energies, 9(6), 440. https://doi.org/10.3390/en9060440








