Abstract
As one of the carbon capture and utilization (CCU) technologies, mineral carbonation which has been introduced to reduce the carbon dioxide (CO2) concentration in the atmosphere is a technology that makes it possible to capture CO2 and recycle byproducts as resources. However, existing mineral carbonation requires additional energy and costs, as it entails high temperature and high pressure reaction conditions. This study compared two processes which electrolyze NaCl and CaCl2 solution to produce CO2 absorbent needed to generate CaCO3, and which were conducted at room temperature and pressure unlike existing mineral carbonation. As a result, high-purity calcite was obtained through Process 1 using NaCl solution, and aragonite and portlandite were obtained in addition to calcite through Process 2 (two steps) using CaCl2 solution.
1. Introduction
Carbon dioxide (CO2) accounts for more than 88% of greenhouse gas emissions and has contributed more than 55% to the greenhouse effect [1,2]. In order to solve the climate change and environmental problems caused by CO2, a number of studies have been carried out actively [1,3,4].
Until now, intensive research has been conducted on carbon capture and storage (CCS) technologies to store the CO2 underground or on the seabed and carbon capture and utilization (CCU) technologies to capture CO2 and convert it to be reused. However, it has become known that there are several problems in the practical application of these techniques [5]. In the case of CCS, excessive energy consumption throughout the entire process and the possibility of leakage of absorbent have been pointed out as the biggest problems [6]. CCU technology was introduced as a partial alternative to these problems. Among the CCU technologies, mineral carbonation is one of the most discussed techniques for treating large amounts of CO2. However, the magnesium-based mineral carbonation process is also complicated and should be conducted in high temperature and high pressure conditions, so the facility where it is conducted needs to be equipped for the high risk group [7,8,9]. For this reason, this study introduces a mineral carbonation process undertaken with less energy by using electrolysis technology with a porous ceramic membrane as ion channel and separator. In this process, divalent metal cations are combined with CO2 to form carbonate minerals [10]. Generally, CO2 reacts with the alkaline absorbent solutions. In the existing carbonation process, magnesium hydroxide was primarily used to produce the alkaline solution, because the Mg-containing minerals contain about 40% Mg2+, while the Ca-containing minerals contain about 10%–15% Ca2+ [11]. However, the existing magnesium-based carbonation process has the aforementioned problems. According to Nduagu [12] and Khoo et al. [13], extracting magnesium oxide or hydroxide from serpentine requires heating to a temperature of 500 °C or higher, and high temperature reaction conditions are needed again in the hydration process after extraction [14].
To reduce these problems, our previous study and this study have introduced a process of electrolyzing chloride salts present in seawater and producing alkaline solution, which is a CO2 absorbent. Our process does not extract oxides and hydroxides from rocks, but uses electrolysis at room pressure and room temperature to obtain hydroxides. More specifically, in our previous report [15], a calcium carbonation process using NaCl solution was introduced. The entire process is divided into three steps: electrolysis of NaCl (sodium hydroxide formation), reaction with CO2 (sodium bicarbonate formation), and carbonation (generation of calcium carbonate). Through this process, we were able to obtain CaCO3 in the form of calcite in a stable manner. However, since NaCl solution is used for NaCl process, Ca2+ should be added to the carbonation process in order to replace Na+. In addition, the process of precipitating the carbonate after this substitution process is complex and requires considerable time.
Therefore, in this study, in order to simplify process and increase energy efficiency, CaCl2 in seawater was used to make alkaline solution, so that the process of Ca2+ addition was eliminated and the process was reduced to two steps, unlike in our previous study. Since the substitution process is not required, the carbonation process is simpler than the NaCl solution process. Additionally, it was expected that more products could be obtained because of the high content of calcium ions contained in the carbonation process. Precipitates would also be formed more quickly through this process, and the consumption of energy would be decreased. This present work focuses on the comparison of the characterization and yield of final products for our previous and proposed processes.
2. Materials and Methods
2.1. Materials
NaCl (99.5%) and CaCl2 (95.9%) were purchased from Samchun Chemical Korea and Kanto Co., Tokyo, Japan, respectively, and used as received. Pure CO2 (99.0%) gas was purchased from Samheung, Gyeonggi-do, Korea, and was used without any pretreatment. Pure CaCO3 (≥99.0%, Sigma-Aldrich, St. Louis, MO, USA) was used to compare with the final CaCO3 product from the two processes. Ultra-pure water prepared from AquaMax 311 (YL Instrument Co. Ltd, Anyang, Korea) was used in all processes.
2.2. Electrolysis Device
The electrolysis device is shown in Figure 1. Each of the units is the same as those used before [15]. The anode (carbon) is located in the center of a round ceramic membrane. The electrodes of the cathode (stainless) were installed at the same distance around the anode. The mean pore size of the porous ceramic membrane was 0.2 μm, which acts as an ion channel when the electrolytic process is started. In other words, ion separation occurs between the inner and outer reactor during the electrolysis process. After the electrolytic process, fine CO2 bubbles (100–400 μm) were injected from the generator (ANGEL AQUA, Changwon, Korea). At this time, this ceramic wall plays as an ion separator.
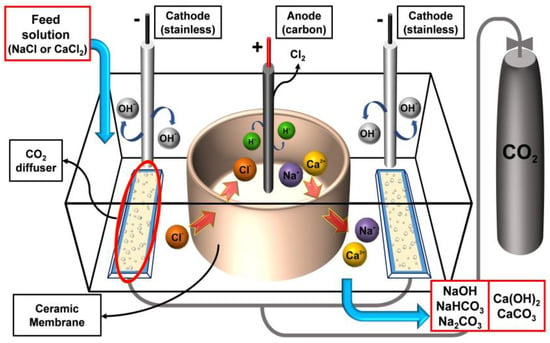
Figure 1.
Schematic diagram of the electrolysis device.
2.3. NaCl Solution Process: Process 1
In the case of the NaCl solution process (Process 1), the experiment to generate CaCO3 was carried out in three steps, preparing 2%–6% NaCl solution based on the concentration of coastal seawater and deep seawater. Depending on 1–4 A of current intensity, electrolysis of NaCl solution was required to react for 10–15 min at 25 °C and 1 bar, and voltage was about 8–24 V. Characterization of the product was reported in our earlier paper [15], and chemical reaction of each step was summarized as follows:
Step 1:
Step 2:
Step 3:
2.4. CaCl2 Solution Process: Process 2
In the case of the CaCl2 solution process (Process 2), the experiment to generate CaCO3 was performed in two steps, as 0.5%–2% CaCl2 is based on the concentration of seawater. All other parameters are the same as in Process 1. The reaction time was stopped at the point where there is almost no variation in pH, at around 11.6. Under the same conditions, the reaction time of electrolysis in Process 2 took 5 min less than in Process 1.
In Step 1, an alkaline calcium hydroxide solution used as an absorbent of CO2 was produced through electrolysis, and the reaction is as follows:
In Step 2, calcium hydroxide solution was formed by a ceramic membrane at the negative electrode side after electrolysis. CaCO3 was precipitated by blowing CO2 at constant flow rate (2 L·min−1). The precipitation reaction is as follows:
A side reaction as in Equation (5b) occurs when the pH value is higher than 12.
2.5. Characteristics of Precipitates
The final product CaCO3 was obtained by GF/C film (Whatman® Glass microfiber filters, Whatman, Clifton, NJ, USA) filtration and drying for 24 h at 80 °C. Fourier transform infrared (FT-IR) spectra were obtained with a Spectrum 100 FT-IR Spectrometer, PerkinElmer connected attenuated total reflection (ATR) tool (PerkinElmer, Norwalk, CT, USA). X-ray diffraction (XRD) was measured using a Rigaku Ultima IV (XRD, Ultima IV, RIGAKU, Tokyo, Japan) using a Cu Kα X-ray 40 kV/30 mA, and the range was 2θ = 15°–70°. The shape and particle size of the fully dried samples were investigated by field emission scanning electron microscopy (FE-SEM, JEOL-7800F, Tokyo, Japan).
3. Results
3.1. Fourier Transform Infrared Analysis
Figure 2 shows the IR spectra measured in the 1800–600 cm−1 range of the respective samples. All CaCO3 products from Process 1 and Process 2 were confirmed as calcite, and the peaks were 1418, 876, and 713 cm−1. Interestingly, the IR spectrum of CaCO3 obtained from Process 2 was observed for new peaks. The value of the peak observed in the 1500–1400 cm−1 region and the 700–650 cm−1 region is the peak of CaCO3 as aragonite (respectively, 1465 cm−1, C–O, stretching mode; 692 cm−1, C–O, out-plane bending), and the peak in the 850–800 cm−1 region is the value of the peak only as vaterite and aragonite (848 cm−1, C–O, in-plane bending) [16,17]. In addition, the peak (1080 cm−1) in the 1100–1000 cm−1 region is the stretching mode of the O–C–O functional group.
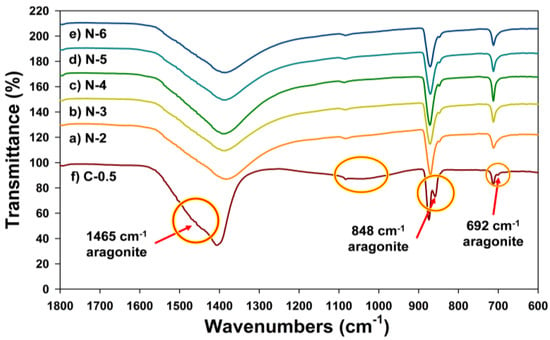
Figure 2.
Fourier transform infrared (FT-IR) spectrum of the precipitated particles through Process 1 and Process 2. (a) N-2; (b) N-3; (c) N-4; (d) N-5; (e) N-6; and (f) C-0.5 (range of 1800–600 cm−1). Product particles formed using CaCl2 0.5% solution are termed as C-0.5, while product particles formed using NaCl are termed as N-X, where X is the NaCl concentration.
3.2. X-ray Diffraction Analysis
XRD patterns were analyzed for more accurate compositional analysis of the generated particles and the results are shown in Figure 3. The product of Process 1 is that characteristic peaks of calcite only appear through our previous studies. In XRD pattern analysis of the products of Process 2, it was possible to confirm the two-theta values for aragonite (26.3, 27.2) and for Ca(OH)2 (34.1, 47.1), as well as the two-theta value for calcite (29.3, 39.3, 43.1) [18].
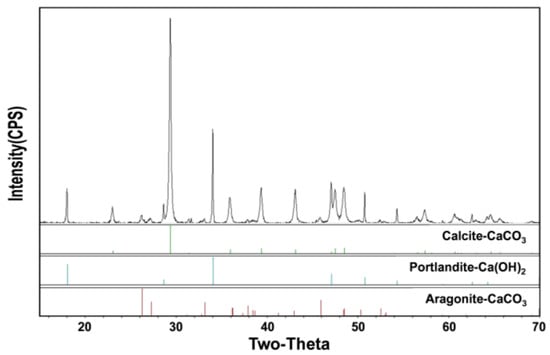
Figure 3.
The X-ray diffraction (XRD) spectra of the produced particles from Process 2.
According to the research of Ma et al. [19], crystalline forms of CaCO3 are greatly influenced by the pH of the water soluble matrix (WSM), and it has been reported that aragonite crystal forms can be obtained at low pH levels (pH 5.5–9.0). For Process 2, after forming a Ca(HCO3)2, CaCO3 was precipitated by adjusting the pH in the last step. Thus, as reported in the case where the precipitation starts from a low pH level, aragonite crystals can be obtained. However, as the pH level gradually increases, it is determined that crystalline calcite was also generated, as well as aragonite [20]. Furthermore, the Ca(OH)− which exists in the form of hydroxide ions is converted into Ca(OH)2 and precipitates by pH control [21]. For Process 1, after adjusting the pH to 12 or higher by using NaOH solution, precipitation was started through substitution reaction with the calcium ions. Thus, if the precipitation is started at a high pH, it is possible to obtain only the hexahedral form of calcite precipitation [20,22].
3.3. Field Emission Scanning Electron Microscopy Image Analysis
FE-SEM was used to confirm other characteristics of other formed particles. Figure 4 is the SEM image of formed particles pictured at ×10,000 magnification. From Figure 4, numerous differences were observed in the size and shape of formed particles. The size of formed particles obtained from Process 1 is 3–4 μm, regardless of the concentration of feed solution, and the particles formed were cubic calcite. On the other hand, formed particles obtained from Process 2 were smaller than 1 μm, and it was observed that several shapes of particles exist. When the CaCO3 crystals are formed at high pH, crystal nucleation proceeds rapidly and affects particle size [19].

Figure 4.
Scanning electron microscopy (SEM) image of the precipitated particles through Process 1 and Process 2 (×10,000 magnification). (a) N-2; (b) N-3; (c) N-4; (d) N-5; (e) N-6; and (f) C-0.5.
According to the research of Westin and Rasmuson [23], the growth rate of calcite was 4.29 × 10−6 mol·m−2·s−1 and that of aragonite was 1.31 × 10−6 mol·m−2·s−1 under the same reaction conditions. In the case of Process 2, the intensity of calcite peak was strong, as shown in IR and XRD spectra. It was assumed that numerous calcites whose growth rates were faster in three times were formed. Additionally, in the case of Process 2, in which a crystalline product is formed with an additional product through a side reaction, it is confirmed that the size of the particles is smaller compared to the products of Process 1, due to a hindrance of growth between particles.
3.4. Conversion Yield of Precipitate
Figure 5 shows the average conversion rate of formed precipitates in two processes. The rates for the two processes were calculated by the rate of theoretical value and measured value for overall Ca2+ contained in used solution in the final step, which is the precipitation procedure. In Process 1, conversion rate increased as the concentration of feed solution increased. However, it was confirmed that the average conversion rate was 77.7% in 5% solution, and it was decreased in 6% solution. In Process 2, the average conversion rate of precipitates was 90.0% in 0.5% solution, which is more than 10% higher than the highest value shown in Process 1. The reason for this difference is that Na+ and Ca2+ of the formed absorbents reacted with different amount of CO32−, although the overall amount of supplied CO2 gas was the same in all experiments. In addition, the reason that conversion rate was decreased in high concentration CaCl2 solution was because precipitates were not dramatically increased compared to the increased amount of Ca2+.
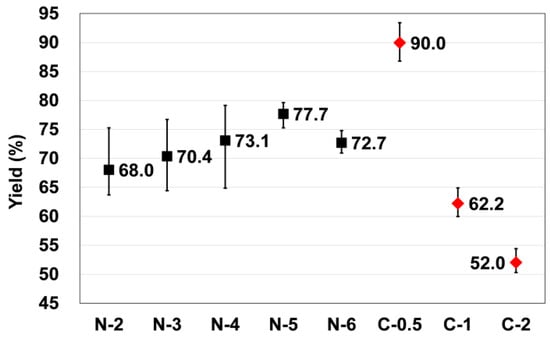
Figure 5.
Conversion rate of precipitates for each concentration feed solution used in Process 1 and Process 2.
4. Conclusions
In this study, attempts to minimize the problems of existing CCS techniques were made by using Na+ and Ca2+ contained in sea water, and two processes for capturing CO2 and producing carbonate were compared and analyzed. The process of the two methods that we have presented can be compared in three major ways.
First, it is possible to be approached from the energy perspective. Both processes form precipitates in a thermodynamically stable form. Additionally, the overall process uses reaction conditions of room temperature and pressure. This demonstrates that CaCO3 can be produced quickly and stably using less energy than conventional methods requiring high-temperature and high-pressure reaction conditions, which was proven through structural analysis using FT-IR spectra. Further, the energy consumption of Process 2 was small compared to Process 1, through simplification to a two-step process and reduced process time of electrolysis reaction. In addition, the carbonation process is an exothermic reaction, and does not require additional energy [24].
Second, it is possible to be approached by added value of formed precipitates. The result of SEM imaging and XRD pattern analysis of the precipitate produced showed that CaCO3 produced by Process 1 could be used as chemical material without any purification process after it was produced with high purity calcite. This has also been proven through our previous studies. On the other hand, in the precipitate produced by Process 2, aragonite—another crystal form of CaCO3—was identified along with portlandite. Aragonite precipitate is a raw material used widely, not only in plastic, rubber, paint, and paper, but also in feeds and pharmaceuticals. If only aragonite CaCO3 is to be formed, 50–70 °C temperature should be used as reaction conditions [25]. It is also possible to weakly control the size of precipitates by controlling pH. Through this process, purity and shape control of aragonite can be applied as raw material in various fields.
Finally, it is possible to be approached from the perspective of CO2 reduction, as it relates to global warming. In this perspective, it was assumed that Process 2, for which the reaction conversion rate was 10% higher than Process 1, captured more CO2. Furthermore, the final product of Process 2 can process more CO2 than that of Process 1, and can be used as raw material for construction materials [26].
Thus, using Process 2 makes it possible to obtain a precipitate with a high added value more quickly and easily than Process 1. In addition, this study was processed as batch reaction. If precipitates are produced by continuous reactor, solutions left after reaction can be used again in the reaction by reprocessing. Therefore, it is possible to minimize environmental problems caused by waste liquor formed in the process. In addition, natural CaCO3 formation generally requires long reaction time, as stated in the previous study [15]. On the contrary, both processes we have proposed can reduce the reaction time to form CaCO3.
As mentioned earlier, conventional mineral carbonation is subject to the process of extracting the reaction components. According to Kakizawa et al. [27], it consumes 1.27 MW of energy in the process for extracting calcium ion from calcium silicate. However, in our studies, extraction process was excluded. In actual seawater application, a membrane enrichment system would be used, which would feed the reaction components through separation and concentration by the membrane.
More research is needed, but we hope to contribute to the reduction of atmospheric CO2 through carbonation of CO2 based on these considerations.
Acknowledgments
This work was supported by the Human Resources Program in Energy Technology of the Korea Institute of Energy Technology Evaluation and Planning (KETEP), granted financial resource from the Ministry of Trade, Industry & Energy, Republic of Korea. (No. 20154010200810).
Author Contributions
Hyun Sic Park, JunYoung Han and Byoung Ryul Min conceived and designed the experiments; Hyun Sic Park performed the experiments; Hyun Sic Park and JunYoung Han analyzed the data; Ju Sung Lee, Kwang-Mo Kim and Hyung Jun Jo contributed reagents/materials/analysis tools; Hyun Sic Park wrote first draft; Byoung Ryul Min revised the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yu, K.M.K.; Curcic, I.; Gabriel, J.; Tsang, S.C.E. Recent advances in CO2 capture and utilization. ChemSusChem 2008, 1, 893–899. [Google Scholar] [CrossRef] [PubMed]
- West, T.O.; Pena, N. Determining thresholds for mandatory reporting of greenhouse gas emissions. Environ. Sci. Technol. 2003, 37, 1057–1060. [Google Scholar] [CrossRef] [PubMed]
- Baciocchi, R.; Corti, A.; Costa, G.; Lombardi, L.; Zingaretti, D. Storage of carbon dioxide captured in a pilot-scale biogas upgrading plant by accelerated carbonation of industrial residues. Energy Procedia 2011, 4, 4985–4992. [Google Scholar] [CrossRef]
- Holloway, S. Underground sequestration of carbon dioxide a viable greenhouse gas mitigation option. Energy 2005, 30, 2318–2333. [Google Scholar] [CrossRef]
- Siefert, N.S.; Litster, S. Exergy and economic analyses of advanced IGCC-CCS and IGFC-CCS power plants. Appl. Energy 2013, 107, 315–328. [Google Scholar] [CrossRef]
- Damen, K.; Faaij, A.; Turkenburg, W. Health, safety and environmental risks of underground CO2 storage–overview of mechanisms and current knowledge. Clim. Chang. 2006, 74, 289–318. [Google Scholar] [CrossRef]
- Lackner, K.S.; Butt, D.P.; Wendt, C.H. Progress on binding CO2 in mineral substrates. Energy Convers. Manag. 1997, 38, S259–S264. [Google Scholar] [CrossRef]
- Fagerlund, J.; Nduagu, E.; Zevenhoven, R. Recent developments in the carbonation of serpentinite derived Mg(OH)2 using a pressurized fluidized bed. Energy Procedia 2011, 4, 4993–5000. [Google Scholar] [CrossRef]
- Fagerlund, J.; Nduagu, E.; Romao, I.; Zevenhoven, R. CO2 fixation using magnesium silicate minerals part 1: Process description and performance. Energy 2012, 41, 184–191. [Google Scholar] [CrossRef]
- Oelkers, E.H.; Gislason, S.R.; Matter, J. Mineral carbonation of CO2. Elements 2008, 4, 333–337. [Google Scholar] [CrossRef]
- Goff, F.; Lackner, K. Carbon dioxide sequestering using ultramafic rocks. Environ. Geosci. 1998, 5, 89–101. [Google Scholar] [CrossRef]
- Nduagu, E. Mineral Carbonation: Preparation of Magnesium Hydroxide [Mg(OH)2] from Serpentinite Rock. Master’s (Eng) Thesis, Åbo Akademi University, Åbo, Finland, 2008. [Google Scholar]
- Khoo, H.; Bu, J.; Wong, R.; Kuan, S.; Sharratt, P. Carbon capture and utilization: Preliminary life cycle CO2, energy, and cost results of potential mineral carbonation. Energy Procedia 2011, 4, 2494–2501. [Google Scholar] [CrossRef]
- Stasiulaitiene, I.; Fagerlund, J.; Nduagu, E.; Denafas, G.; Zevenhoven, R. Carbonation of serpentinite rock from Lithuania and Finland. Energy Procedia 2011, 4, 2963–2970. [Google Scholar] [CrossRef]
- Park, H.S.; Lee, J.S.; Han, J.; Park, S.; Park, J.; Min, B.R. CO2 fixation by membrane separated NaCl electrolysis. Energies 2015, 8, 8704–8715. [Google Scholar] [CrossRef]
- Tlili, M.; Amor, M.B.; Gabrielli, C.; Joiret, S.; Maurin, G.; Rousseau, P. Characterization of caco3 hydrates by micro-raman spectroscopy. J. Raman Spectrosc. 2002, 33, 10–16. [Google Scholar] [CrossRef]
- Wu, G.; Wang, Y.; Zhu, S.; Wang, J. Preparation of ultrafine calcium carbonate particles with micropore dispersion method. Powder Technol. 2007, 172, 82–88. [Google Scholar] [CrossRef]
- McMurdie, H.F.; Morris, M.C.; Evans, E.H.; Paretzkin, B.; Wong-Ng, W.; Ettlinger, L.; Hubbard, C.R. Standard X-ray diffraction powder patterns from the jcpds research associateship. Powder Diffr. 1986, 1, 64–77. [Google Scholar] [CrossRef]
- Ma, Y.; Gao, Y.; Feng, Q. Effects of pH and temperature on CaCO3 crystallization in aqueous solution with water soluble matrix of pearls. J. Cryst. Growth 2010, 312, 3165–3170. [Google Scholar] [CrossRef]
- Stocks-Fischer, S.; Galinat, J.K.; Bang, S.S. Microbiological precipitation of CaCO3. Soil Biol. Biochem. 1999, 31, 1563–1571. [Google Scholar] [CrossRef]
- Ren, L.; Zhang, Y.; Bian, Y.; Liu, X.; Liu, C. Investigation of quartz flotation from decarburized vanadium-bearing coal. Physicochem. Probl. Miner. Process. 2015, 51, 755–767. [Google Scholar]
- Han, Y.S.; Hadiko, G.; Fuji, M.; Takahashi, M. Crystallization and transformation of vaterite at controlled pH. J. Cryst. Growth 2006, 289, 269–274. [Google Scholar]
- Westin, K.-J.; Rasmuson, A.C. Crystal growth of aragonite and calcite in presence of citric acid, DTPA, EDTA and pyromellitic acid. J. Colloid Interface Sci. 2005, 282, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Maroto-Valer, M.M. Integration of CO2 capture and mineral carbonation by using recyclable ammonium salts. ChemSusChem 2011, 4, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Colfen, H.; Qi, L. A systematic examination of the morphogenesis of calcium carbonate in the presence of a double-hydrophilic block copolymer. Chemistry 2001, 7, 106–116. [Google Scholar] [CrossRef]
- Iizuka, A.; Fujii, M.; Yamasaki, A.; Yanagisawa, Y. Development of a new CO2 sequestration process utilizing the carbonation of waste cement. Ind. Eng. Chem. Res. 2004, 43, 7880–7887. [Google Scholar] [CrossRef]
- Kakizawa, M.; Yamasaki, A.; Yanagisawa, Y. A new CO2 disposal process via artificial weathering of calcium silicate accelerated by acetic acid. Energy 2001, 26, 341–354. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).