Gasification of a Dried Sewage Sludge in a Laboratory Scale Fixed Bed Reactor
Abstract
:1. Introduction
2. Experimental
2.1. Apparatus
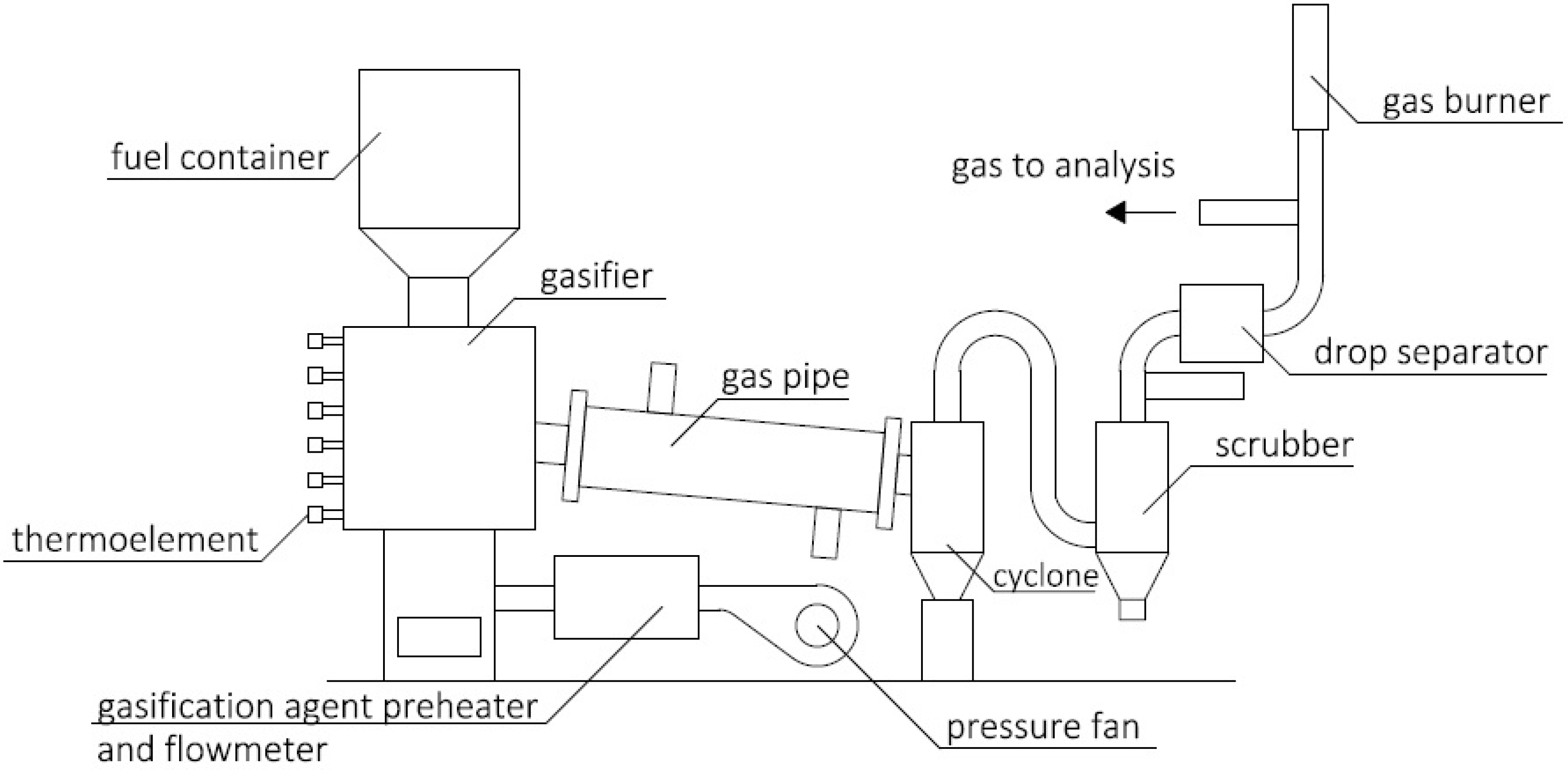
2.2. Fuel Properties

| Element/Parameter | Sewage Sludge | |
|---|---|---|
| SS1 | SS2 | |
| Proximate Analysis | % (as received) | |
| Moisture | 5.30 | 5.30 |
| Volatile matter | 51.00 | 49.00 |
| Ash | 36.50 | 44.20 |
| Ultimate Analysis | % (dry basis) | |
| C | 31.79 | 27.72 |
| H | 4.36 | 3.81 |
| N | 4.88 | 3.59 |
| O (by difference) | 20.57 | 18.84 |
| S | 1.67 | 1.81 |
| F | 0.013 | 0.003 |
| Cl | 0.22 | 0.03 |
| Calorific Value | ||
| Higher heating value (HHV), MJ/kg dry basis | 14.05 | 11.71 |
| Lower heating value (LHV), MJ/kg dry basis | 12.96 | 10.75 |
| Compound | Sewage Sludge | |
|---|---|---|
| SS1 | SS2 | |
| Polycyclic aromatic hydrocarbons (PAHs) | Concentration, μg/kg dry basis | |
| acenaphthene | n.d. | 80.84 |
| phenanthrene | 511.12 | n.d. |
| anthracene | 200.03 | n.d. |
| benzo(a)fluoranthene | 44.78 | 126.48 |
| pyrene | 187.22 | 123.86 |
| benzo(a)anthracene | n.d. | 35.15 |
| chrysene | 108.14 | 23.79 |
| benzo(b)fluoranthene | 700.51 | 53.62 |
| benzo(a)pyrene | n.d. | 46.11 |
| dibenzo(a,h)anthracene | 101.54 | n.d. |
| benzo(g,h,i)perylene | 209.44 | n.d. |
| indeno(1,2,3-cd)pyrene | 370.62 | 131.48 |
| Sum | 2433.40 | 621.33 |
| Pesticides | Concentration, μg/kg dry basis | |
| heptachlor | 4.14 | n.d. |
| aldrin | 3.13 | 1.28 |
| endrin | 11.58 | n.d. |
| Sum | 18.85 | 1.28 |
| pesticides and polychlorinated biphenyls (PCBs) | Concentration, μg/kg dry basis | |
| 2,2′,5,5′-PCB | 9.75 | 7.90 |
| 2,2′,4,5,5′-PCB | 33.33 | n.d. |
| 2,2′,4,4′,5,5′-PCB | 23.78 | 4.57 |
| Sum | 66.86 | 12.47 |
| Heavy metals | Concentration, mg/kg dry basis | |
| Zn | 920.90 | 991.20 |
| Cu | 495.30 | 183.16 |
| Pb | 119.30 | 59.97 |
| Ni | 103.67 | 18.90 |
| Cr | 180.53 | 584.53 |
| Cd | 6.47 | 3.24 |
| As | 4.19 | 3.94 |
| Hg | 0.99 | 0.96 |
| Se | 9.84 | 1.70 |
| Sum | 1841.19 | 1847.6 |
3. Results and Discussion
3.1. Influence of the Air Ratio on the Syngas Composition
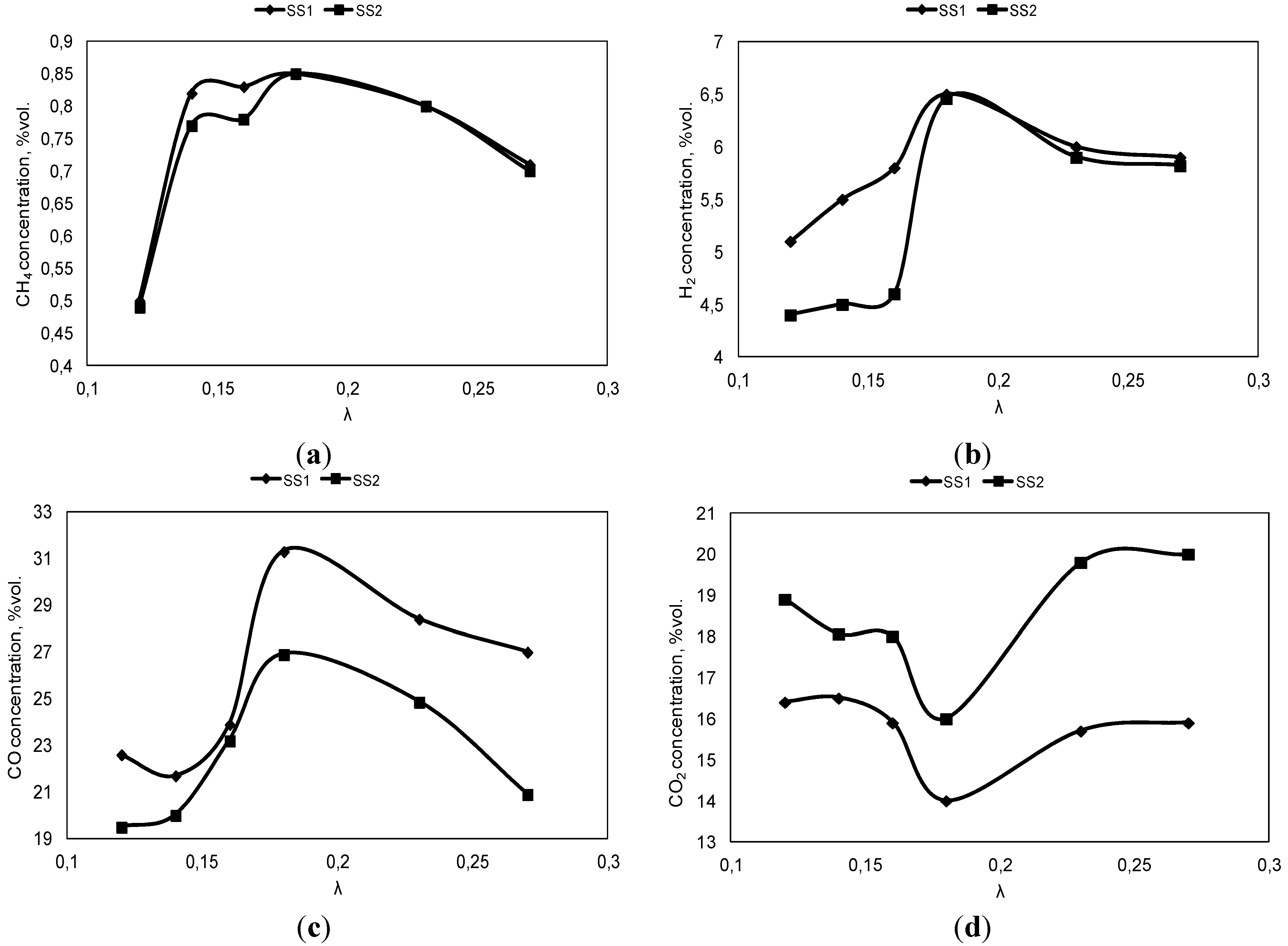
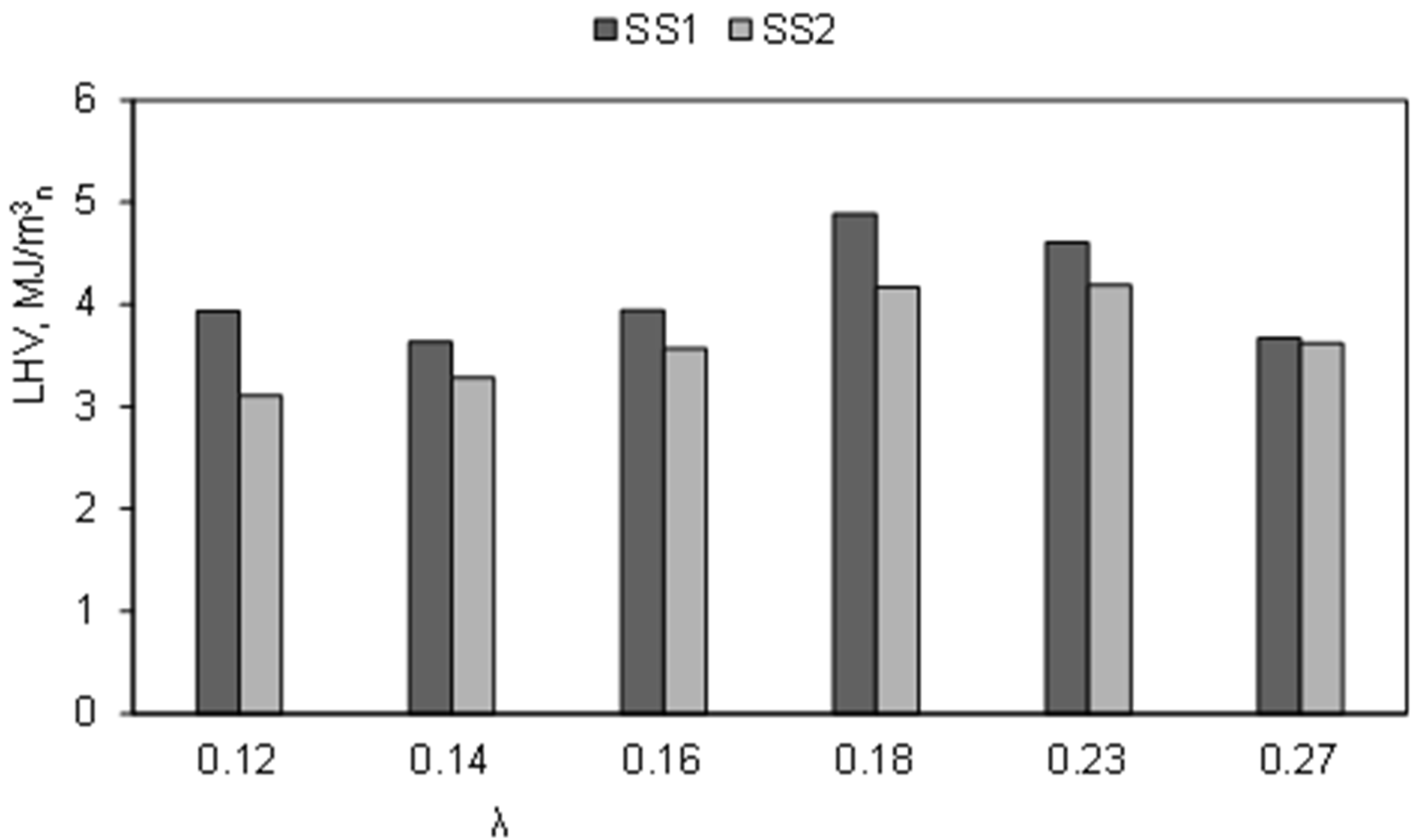
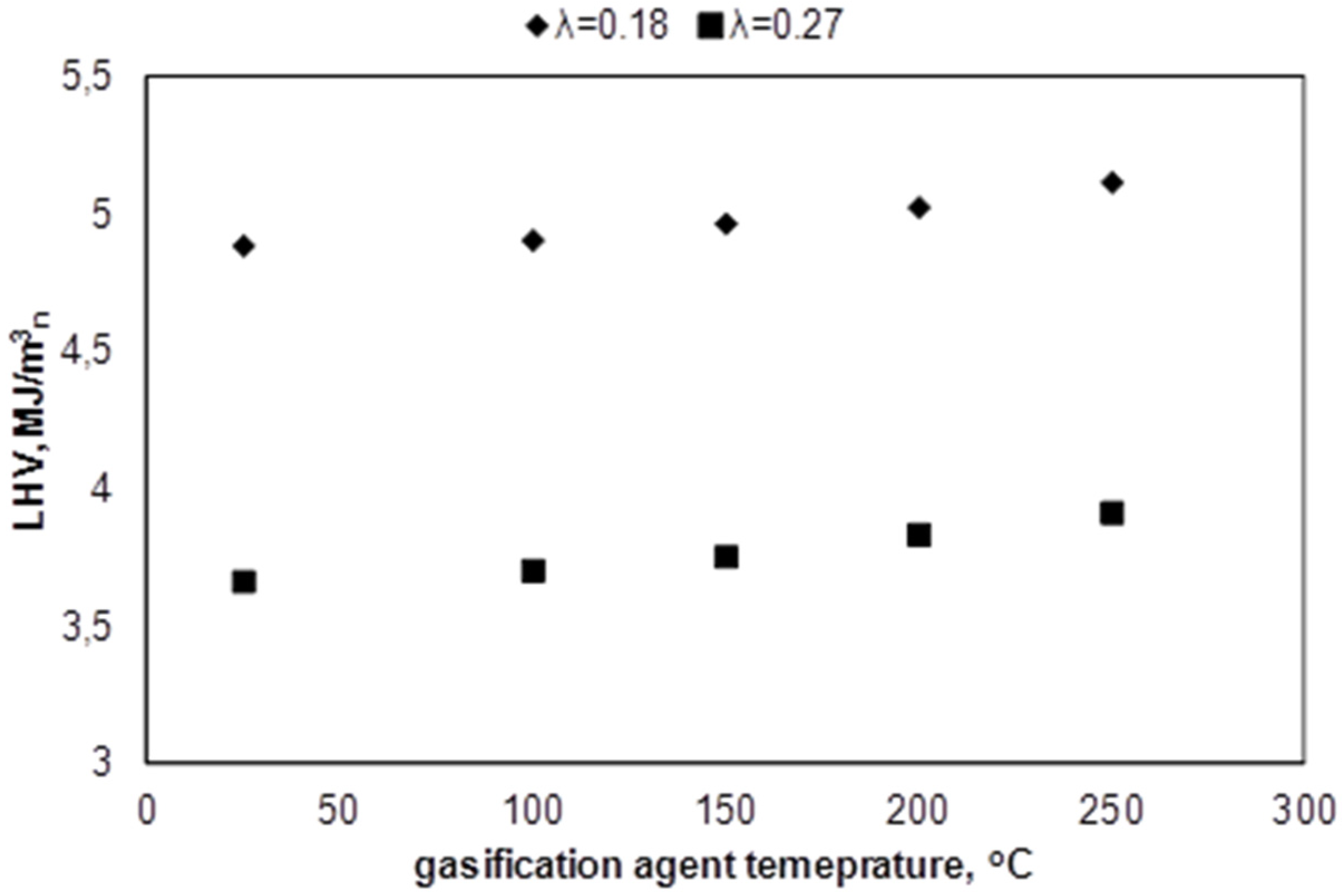
3.2. Influence of the Temperature of Gasification Agent on Syngas Composition
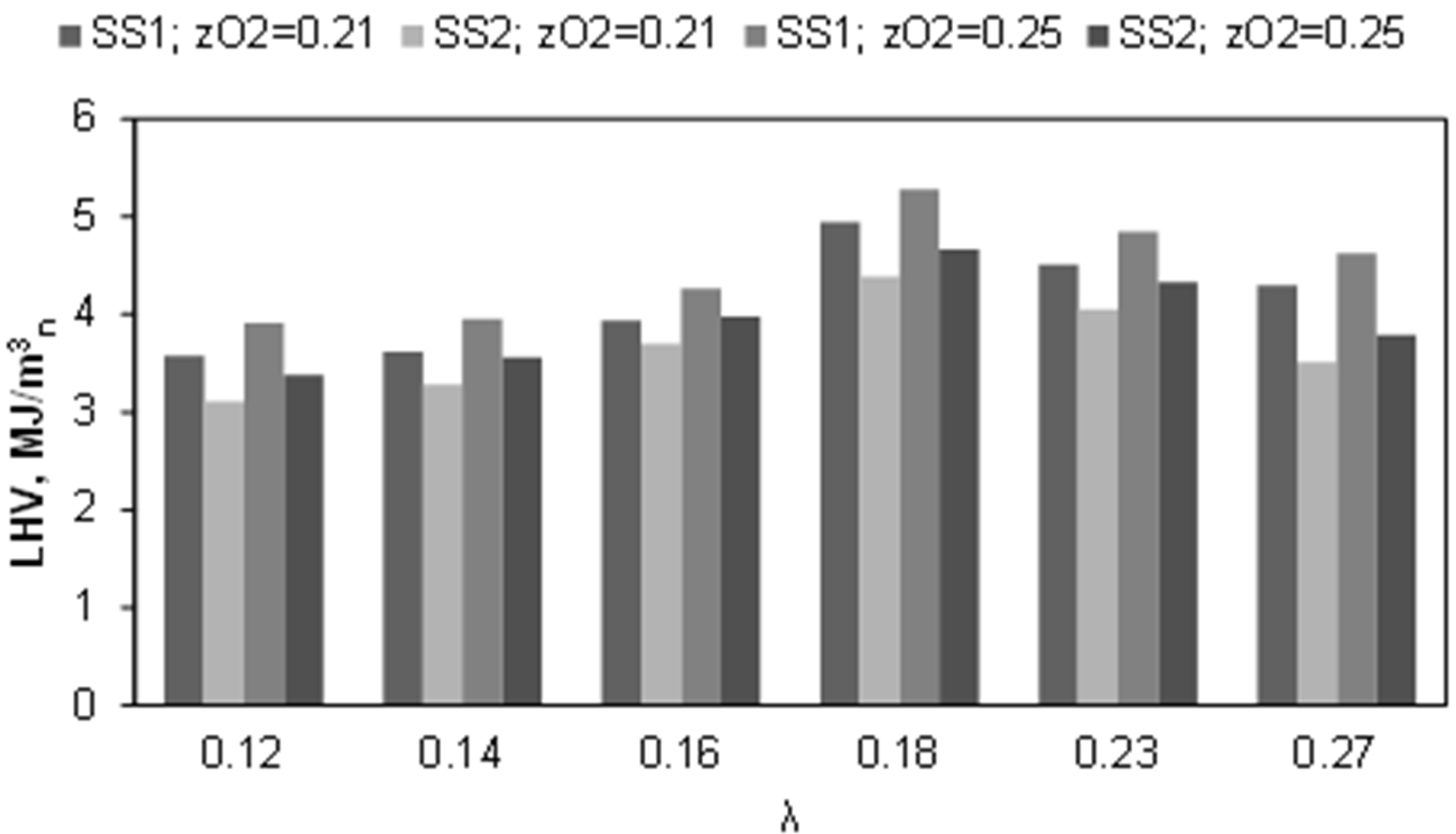
3.3. Influence of the Gasification Agent Composition on Syngas Composition
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Tolis, A.; Rentizelas, A.; Aravossis, K.; Tatsiopoulos, I. Electricity and combined heat and power from municipal solid waste; theoretically optimal investment decision time and emissions trading implications. Waste Manag. Res. 2010, 28, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Cucchiella, F.; D’Adamo, I.; Gastaldi, M. Municipal waste management and recovery in an Italian region. Waste Manag. Res. 2012, 30, 1290–1298. [Google Scholar] [CrossRef] [PubMed]
- Sommer, M.; Ragossing, A. Energy from waste in Europe: An analysis and comparison of the EU27. Waste Manag. Res. 2011, 29, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Basu, P. Biomass Gasification and Pyrolysis: Practical Design and Theory; Elsevier Academic Press: Waltham, MA, USA, 2010. [Google Scholar]
- Basu, P. Combustion and Gasification in Fluidized Beds; Taylor & Francis Group/CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Werle, S. Possibility of NOx emission reduction from combustion process using sewage sludge gasification gas as an additional fuel. Arch. Environ. Prot. 2012, 38, 81–89. [Google Scholar] [CrossRef]
- Seggiani, M.; Puccini, M.; Raggio, G.; Vitolo, S. Effect of sewage sludge content on gas quality and solid residues produced by cogasification in an updraft gasifier. Waste Manag. 2012, 32, 1826–1834. [Google Scholar] [CrossRef] [PubMed]
- Phuphuakrat, T.; Nipattumakul, N.; Namioka, T.; Kerdsuwan, S. Characterization of tar content in the syngas produced in a downdraft type fixed bed gasification system from dried sewage sludge. Fuel 2010, 89, 2278–2284. [Google Scholar] [CrossRef]
- Midilli, A.; Dogru, M.; Howarth, C.R.; Ling, M.J.; Ayhan, T. Combustible gas production from sewage sludge with a downdraft gasifier. Energy Convers. Manag. 2001, 42, 155–172. [Google Scholar] [CrossRef]
- Dogru, M.; Midilli, A.; Howarth, C.R. Gasification of sewage sludge using a throated downdraft gasifier and uncertainty analysis. Fuel Proc. Technol. 2002, 75, 55–82. [Google Scholar] [CrossRef]
- Werther, J.; Ogada, T.; Borodulya, V.A.; Dikalenko, V.I. Devolatization and Combustion Characteristics of Wet Sewage Sludge in a Bubbling Fluidized Bed Furnace. In Proceedings of the Institute of Energy Conference, London, UK, 4–5 December 1995; pp. 149–158.
- Nilsson, S.; Gómez-Barea, A.; Cano, D.F. Gasification reactivity of char from dried sewage sludge in a fluidized bed. Fuel 2012, 92, 346–353. [Google Scholar] [CrossRef]
- Sharma, A.K. Experimental investigations on a 20 kWe, solid biomass gasification system. Biomass Bioenergy 2011, 35, 421–428. [Google Scholar] [CrossRef]
- E4Tech. Review of Technologies for Gasification of Biomass and Wastes. Available online: http://www.ecolateral.org/gasificationnnfc090609.pdf (accessed on 23 July 2015).
- Wilk, R.K.; Werle, S. Biomass Gasification Installation, Mainly for Sewage Sludge. Poland Patent no P-397225, 2 December 2011. [Google Scholar]
- Werle, S.; Wilk, R.K. Analysis of use a sewage sludge derived syngas in the gas industry. Rynek Energii 2011, 4, 23–27. [Google Scholar]
- Solid Biofuels—Methods for Moisture Determining Using Drier Method. Part 3—Moisture Analysis in General Sample; Standards PN-EN 14774-3:2010; Polish Committee for Standardization: Warsaw, Poland, 2010.
- Solid Recovered Fuels—Determination of Volatile Content; Standards PN-EN 15402:2011; Polish Committee for Standardization: Warsaw, Poland, 2011.
- Solid Recovered Fuels—Determination of Ash Content; Standards PN-EN 15403:2011; Polish Committee for Standardization: Warsaw, Poland, 2011.
- Tomza-Marciniak, A.; Witczak, A. Distribution of endocrine-disrupting pesticides in water and fish from the Oder River, Poland. Acta Ichthyol. Piscat. 2010, 40, 1–9. [Google Scholar] [CrossRef]
- Kim, J.-W.; Mun, T.-Y.; Kim, J.-O.; Kim, J.-S. Air gasification of mixed plastic wastes using a two-stage gasifier for the production of producer gas with low tar and high caloric value. Fuel 2011, 90, 2266–2272. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Werle, S. Gasification of a Dried Sewage Sludge in a Laboratory Scale Fixed Bed Reactor. Energies 2015, 8, 8562-8572. https://doi.org/10.3390/en8088562
Werle S. Gasification of a Dried Sewage Sludge in a Laboratory Scale Fixed Bed Reactor. Energies. 2015; 8(8):8562-8572. https://doi.org/10.3390/en8088562
Chicago/Turabian StyleWerle, Sebastian. 2015. "Gasification of a Dried Sewage Sludge in a Laboratory Scale Fixed Bed Reactor" Energies 8, no. 8: 8562-8572. https://doi.org/10.3390/en8088562
APA StyleWerle, S. (2015). Gasification of a Dried Sewage Sludge in a Laboratory Scale Fixed Bed Reactor. Energies, 8(8), 8562-8572. https://doi.org/10.3390/en8088562





