1. Introduction
There is an increased global concern associated with the dependency on fossil feedstocks as the main source for energy as well as for various products, not only due to its harmful effect on the environment, but also the issue of possible supply insecurity. World energy consumption from 1971 to 2009 displays an average energy usage growth per year of approximately 100 million ton oil equivalent, or about 1.5% each year. A major part of this usage, about 80%, is supplied by fossil fuels, implying a profound dependency on fossil resources. In case of EU, the energy dependency data shows that about 64% of gas and 83% of oil demand in 2009 was imported, while these two fuels provide for about 60% of the of total EU energy need. Governments have established regulations to control the emissions and to reduce the amount of imported energy resources. The target for the EU climate policy is to reduce emissions down to 80% of the level of 1990, by 2020 [
1].
Natural gas is becoming increasingly important as a primary energy source and is used for production of heat and power for domestic and industrial purposes, as well as a chemical feedstock. It has also gained importance as a transportation fuel. However, natural gas is a fossil fuel contributing to CO
2 emissions. A suitable replacement for natural gas is bio-based Substitute Natural Gas (bio-SNG) produced from renewable resources. A feasible approach to produce bio-SNG is gasification of biomass, followed by methanation [
2]. At present, there are many on-going projects, aimed at demonstrating the production of methane using this route [
3,
4].
Previous system studies of biomass gasification-based methanation processes have shown that the overall biomass to bio-SNG efficiency (on LHV basis) using this route is around 64%–67%, depending on the technologies used in the conversion process [
5]. In a general synthesis gas to methane process, the CO and the H
2 in the synthesis gas are converted to methane, according to the chemical reaction in Equation (1), using a nickel catalyst [
6]. A clean producer gas from biomass gasification contains (after gas cleaning and conditioning) beside CO, also substantial amounts of CO
2. One possibility to further increase the conversion efficiency is to convert all carbon-containing components,
i.e., not only CO but also CO
2, by e.g., applying the Sabatier process. In this case, the bio-SNG production could be increased substantially, using the same amount of input raw feedstock, as pointed out by Mohseni
et al. [
7]. The Sabatier reaction is shown in Equation (2) [
6].
The producer gas from biomass gasification varies considerably in composition and contains normally between 15%–60% and 20%–25% of hydrogen and carbon monoxide, respectively, when oxygen or steam blown gasification technologies are used [
8]. This implies that the amount of hydrogen is not enough even for the general methanation process in Equation (1), which requires a H
2:CO ratio of at least 3:1. To achieve this ratio in a general process, part of the CO is shifted to H
2 using the water gas shift (WGS) process according to Equation (3).
An additional source of hydrogen is needed to convert all CO and CO
2 and a feasible source is hydrogen produced from electrolysis powered by wind and solar energy. Hydrogen from electrolysis has the benefit of being flexible in its operation and it could also be used as an approach to store intermittently produced electricity for future use [
9]. The electricity, needed for the electrolysis, is approximately 4.3 kWh per produced Nm
3 of hydrogen [
10]. The amount of hydrogen for the methanation of the CO and CO
2 content in the gas produced by a 5 MW
fuel gasifier will require a power input of approximately 7 MW, corresponding to, e.g. 6–10 wind power plants, assuming a plant size of around 2.5–4 MW and a normal average load of 30% during a year. This implies rather small to medium scale gasifier systems to be operated in conjunction with a few wind power plants or a farm.
In gasification of solid fuels, such as biomass and waste, to produce a synthesis gas, the fuel is partially oxidized in an atmosphere of air, oxygen and/or steam at elevated temperatures, typically between 600 and 1200 °C, to generate a valuable product gas, consisting mainly of H
2, CO, CO
2, CH
4, and light hydrocarbons (C
2 + C
3), tar and other impurities, as such H
2S, CS
2, COS, AsH
3, PH
3, HCl, N
2, NH
3, HCN, particulates and alkali salts [
11]. These components have to be removed before utilizing the gas, depending on the application of interest. The type of cleaning process to be used to achieve the required cleanness of the gas depends on downstream application, the composition of the raw gas and the size of the process [
12,
13]. The downstream cleaning process includes many steps where some steps, such as particulate removal, using cyclones and different filters, are common for several types of gasifiers and other, such as sorption processes, using different sorbents, or physical and chemical washing processes, generally depends on the size of the overall plant. Impurities of particular interest for removal are tars and sulfur compounds. In the case of small to medium scale gasifiers, a major challenge is how to design an effective gas upgrading and cleaning process with the ability to produce a clean gas in as few steps as possible.
The aim of the present study was to investigate the possibility to adapt and develop existing large scale gas cleaning technology for a SNG production plant, based on a medium scale (50 MW
thermal) biomass gasifier. The study specifically focuses on the WoodRoll
® gasification process [
14], producing a relatively clean gas without tar, where the main issues are removal of sulfur and other impurities in the raw produced gas. The pre-requisite of the gas cleaning process was that it should remove all impurities, in particular sulfur compounds, but simultaneously keep as much CO
2 as possible in the gas prior to methanation.
The WoodRoll
® gasification process is divided into three main steps;
drying, pyrolysis and
gasification [
14] A separation of the processes enables production of a syngas with reduced content of impurities and condensable heavy fractions and also minimizes the nitrogen dilution. As the process is divided in stages a higher degree of fuel flexibility is possible as mixing and purification can be handled in the most efficient stage of operation. The first step, the dryer is indirectly heated in a rotary drum with heat exchange tubes. With proper control of heating and mixing in the reactor the particular process is well proven for a variety of materials. A hot gas reactor is supplying the heat by burning pyrolysis gas from subsequent steps. The dried fuel is then transferred to a pyrolysis reactor. In this second step, the fuel pyrolyzed in a similar rotary drum at much higher temperature (400 °C) producing char and pyrolysis gas, consisting of permanent gases with condensable tars and heavier fractions held in gas phase. Heat is mainly generated from the flue gas from the gasification stage downstream. The char is cooled and grinded to the required size. In the third step, an entrained gasifier, where radiant heat burners supply the heat, combusting the pyrolysis gas, the grinded char and superheated steam are mixed. The steam gasification occurs at a temperature of 1,100 °C. The superheated steam is produced in a cooling step of the produced raw gas after the gasifier. The raw product gas produced in the WoodRoll
® process has typically a gas composition consisting of 16%–20% CO, 20%–24% CO
2, 55%–65% H
2 and almost no or small amounts of methane. The subsequent sections start with a short review of gas cleaning for removing sulfur and other impurities, followed by methodology and a description of the modeling results. Finally, a short discussion about applying the gas cleaning process is presented.
2. Gas Cleaning Sulfur and Other Impurities
The specific requirements for a synthesis gas, concerning an acceptable amount of sulfur contaminants are somewhat diverse, with requirements as low as <0.01 ppmv to <1 ppmv reported in the literature [
15].
Table 1 outlines the requirement for the major components of a synthesis gas from biomass gasification, considered for further upgrading in an end-user application [
15,
16]. To allow for later comparisons the acceptable amount of contaminants in the synthesis gas is given in both mg/Nm
3 and ppm. These impurities affect the methanation process negatively, reducing the expected lifetime of the catalyst in the process, due to e.g., sulfur poisoning, alkali salt deposition and carbon formation [
17].
Gas cleaning processes for removal of sulfur compounds, such as H
2S and CO
2, and generally denoted as acid gases (AG), as well as other compounds of halides and nitrogen compounds are most readily performed by means of a physical and chemical washing process at plant scales larger than approximately 20 MW
fuel [
18]. In a physical and chemical process, the gas is washed with a suitable solvent, physical or chemical, in a packed (structured or unstructured) column or tray column at a temperature where the impurity absorption is efficient.
Table 1.
Acceptable amount of contaminants in a synthesis gas entering an end user application [
16].
Table 1.
Acceptable amount of contaminants in a synthesis gas entering an end user application [16].
| Contaminant | Removal level |
|---|
| Particles | Essentially complete | - |
| Tars | Below dewpoint | - |
| Na + K | <10 ppbv | - |
| (HCl + HBr + HF) | <10 ppbv | <0.015 mg/Nm3 (a) |
| NH3 + HCN | <1 ppmv | <0.7 mg/Nm3 (b) |
| (H2S + COS + CS2) | <10 ppbv to <1 ppmv [14] | <0.014 mg/Nm3 to <1.4 mg/Nm3 (c) |
Physical solvents tend to be favored over chemical solvents when the concentration of acid gases or other impurities is very high. Unlike chemical solvents, physical solvents are non-corrosive, requiring only carbon steel construction. The regeneration of chemical solvents is achieved by the application of heat, whereas physical solvents can often be stripped of impurities by reducing the pressure without the application of heat. The absorption loading capacity for a physical or chemical solvent is proportional to the volumetric gas flow rate or the removed acid gases, respectively [
18]. The solvent used with the absorbed AG and other impurities is transferred to a regeneration unit for purification and release of the acid gases.
General criteria for selection of the appropriate AG removal process configuration are among others given by the demand of the required gas purity, the raw gas composition,
i.e., some solvents do not adsorb COS, and selectivity towards desirable, undesirable gas components and solvent loss [
18]. A suitable solvent is essential for an efficient process to optimize the removal of undesired components while not removing desired components of the gas. Two typical solvents for chemical removal of AG are amines and potassium carbonate. Amines used for this purpose are monoethylamine (MEA), diethylamine (DEA), methyldiethylamine (MDEA) and diisopropyl amine (DIPA). MDEA is widely used due to its high selectivity for H
2S, but a drawback is the large fraction of CO
2 slipping through unabsorbed [
18,
19].
Examples of physical processes are the Purisol
® process; a typical process for selective removal of H
2S with CO
2 remaining in the treated gas, using N-methyl-2-pyrrolidone (NMP) as the solvent [
20]. COS is not as soluble as H
2S, but is hydrolyzed [
21]. Another solvent example, suitable for selective removal of H
2S, is the Selexol
® process, using dimethyl ethers of polyethylene glycol (DEPG) as the solvent [
22,
23]. The Purisol
® process is especially suitable for the cleaning of high-pressure, high CO
2 synthesis gas for gas turbine integrated gasification combined cycle (IGCC) systems [
24].
Another possible physical solvent is methanol, as used in the Rectisol
® process, an absorption process with high removal efficiency towards COS, H
2S and CO
2, as well as other impurities like HCN and NH
3 [
25]. A typical example of a Rectisol
® process, including WGS, is illustrated in
Figure 1. The main advantage of using methanol as a solvent is the lower costs of the solvent, compared to the amine-based solvents. Another is its high removal efficiency towards COS compared to other solvents [
18].
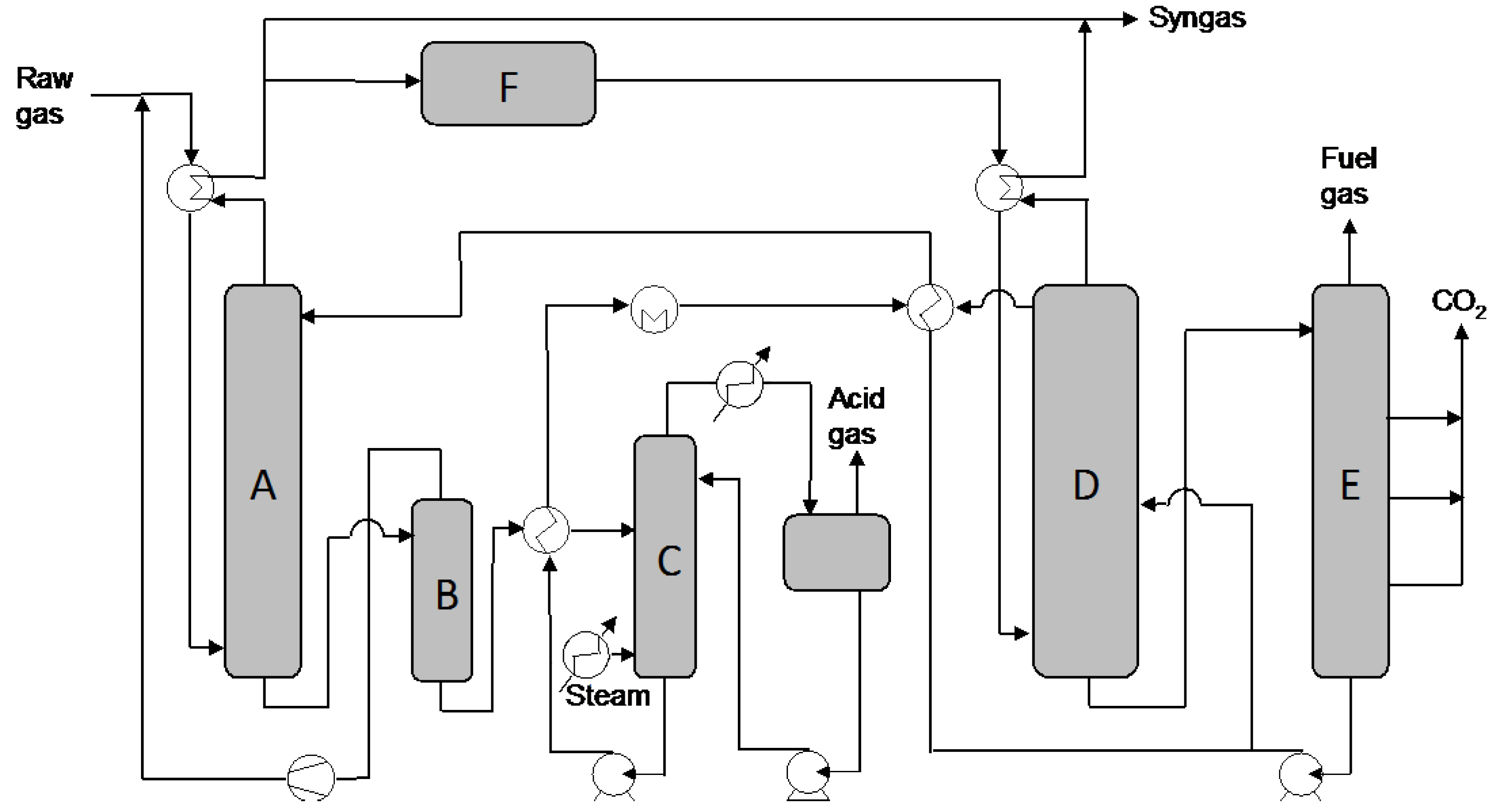
Figure 1.
A schematic flow diagram of typical example of a selective Rectisol
® process for AG removal.
A: H
2S absorber;
B: H
2S flash column;
C: hot regenerator;
D: CO
2 absorber;
E: CO
2 flash tower;
F: WGS. Adapted from Higman and van der Burgt [
13].
Figure 1.
A schematic flow diagram of typical example of a selective Rectisol
® process for AG removal.
A: H
2S absorber;
B: H
2S flash column;
C: hot regenerator;
D: CO
2 absorber;
E: CO
2 flash tower;
F: WGS. Adapted from Higman and van der Burgt [
13].
The solubility of CO
2 and H
2S is dependent on partial pressure and temperature [
26]. A packed absorber is most commonly used and the raw gas enters counter-currently to the solvent [
27]. In the Rectisol
® process, gas components such as CO
2, H
2S, COS, HCN and NH
3 as well as nickel and iron carbonyls are absorbed physically and removed from a gas. These components are then desorbed from the solvent in a regeneration process. The raw gas has to be cooled before entering the plant. Trace components like hydrogen cyanide and ammonia are removed in a prewash step where cold methanol is used. The sulfur compounds are removed by using carbon dioxide-loaded solvent, which generally results in a residual content of sulfur below 0.1 ppm. In the regeneration process, CO and H
2 are recovered by, firstly, flashing at medium pressure, and secondly by heating to boiling temperature and stripping with methanol vapor. In gas mixtures with relatively high hydrogen sulfide, the hot regeneration produces a Claus gas with enough hydrogen sulfide content for further treatment. In cases with low H
2S content or with high content of carbon dioxide, an additional step like stripping and reabsorption is needed. The final content of CO
2 in the syngas can be around 3% [
28,
29].
5. Modeling of the Gas Cleaning Process
The selection of an appropriate absorption solvent for the process was based on an extensive survey, as stated above. The chemical absorption solvents were ruled out, since physical solvents generally are more favorable when the concentration of acid gases or in particular if other impurities is high. Also, physical solvents are, unlike chemical, non-corrosive, and therefore require less expensive process construction materials, such as carbon steel construction [
24].
Table 2 shows the relative solubility of selected gas compounds, compared to CO
2, in the three solvents DEPG, NMP and methanol, which are used in the Selexol
®, the Purisol
® and the Rectisol
® processes, respectively. Based on this comparison, the relative difference between the different compounds for each solution is similar for all solutions, except for NH
3 and COS, where a higher relative solubility is obtained in case of methanol. This higher efficiency for removal of NH
3, also valid for other impurities, such as HCN and COS [
25], is one reason for preferring methanol as the solvent for the gas cleaning process. Another parameter supporting methanol and the Rectisol
® process are its ability to remove sulfur to below 0.01 ppmv H
2S, compared to e.g., Selexol
® where 1 ppmv is a typical level [
31].
Table 2.
Relative solubility of gas compounds, compared to hydrogen sulfide, in the three solvents DEPG, NMP and methanol (adopted from [
24]) DEPG and NMP is calculated using values at 25 °C but in case of methanol −25 °C is presented due to the high volatility of methanol.
Table 2.
Relative solubility of gas compounds, compared to hydrogen sulfide, in the three solvents DEPG, NMP and methanol (adopted from [24]) DEPG and NMP is calculated using values at 25 °C but in case of methanol −25 °C is presented due to the high volatility of methanol.
| Gas | DEPG at 25 °C | NMP at 25 °C | MeOH at −25 °C |
|---|
| H2 | 0.013 | 0.0064 | 0.0054 |
| CO | 0.028 | 0.021 | 0.02 |
| CO2 | 1 | 1 | 1 |
| CH4 | 0.066 | 0.072 | 0.051 |
| NH3 | 4.8 | - | 23.2 |
| COS | 2.3 | 2,7 | 3.9 |
| H2S | 8.8 | 10.2 | 7.06 |
Other important aspects, influencing favoring methanol, are the flexibility and ease in reconfiguring the process [
24], and that the solvent is both inexpensive and abundant. A drawback of using methanol, as a solvent, is, however, the low operating temperature at −60 to −75 °C, much below the methanation step ranges [
31]. In this case Selexol
®, using DEPG as solvent, operating at ambient temperature would be a better choice. However, the superior removal of sulfur compounds and other impurities by the solvent methanol, compared to other physical solvents, outweigh this drawback at the present stage of analysis.
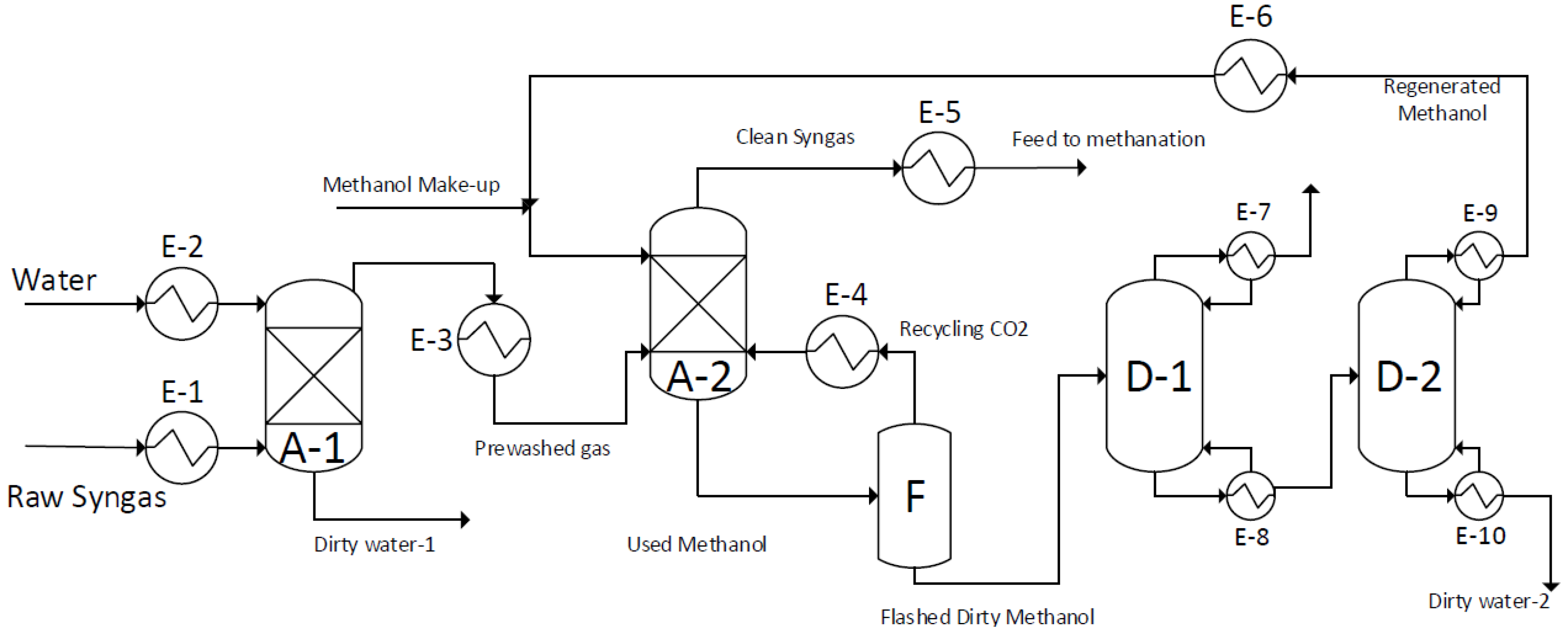
5.1. The Modified Rectisol® Process
The configuration of the concluding gas cleaning and upgrading process for the 50 MW scale is displayed in
Figure 2. This process is based on Rectisol
® process and is designed to the keep the carbon dioxide in the main stream while removing other impurities to the lowest possible concentrations. The process mainly includes the following major parts, as indicated in
Figure 2:
A-1: A water scrubber
A-2: A methanol wash
F: A flash tank for used methanol
D-1: A flashed methanol distiller
D-2: A dehydration of cleaned methanol
A simplified description of the process is as follows; the first step (E-1) is the cooling and pressurization of the raw syngas to make the gases more soluble in liquids. The working temperature and pressure is normally varied between −30 to 0 °C and above 25 bar, respectively. Although such working conditions are energy demanding and expensive, the conditions are less energy demanding than a conventional Rectisol
® processes, operating at temperatures between −50 and −20 °C and pressures of 30 to 45 bars to meet the cleaning requirements. This step consists of five to six condensers with intercooling in which water condensate is removed from the raw syngas. For the sake of simplicity, this is not shown in
Figure 2.
Figure 2.
An Aspen process flow diagram of the modified Rectisol® process for the 50 MW scale gasifier.
Figure 2.
An Aspen process flow diagram of the modified Rectisol® process for the 50 MW scale gasifier.
In the second step, the water scrubber (A-1), the water removes HCl, NH
3 and a part of HCN. This step makes the regeneration of the used methanol easier and more efficient as discussed in more detail below. The methanol wash (A-2) basically removes the major impurities, such as H
2S, COS, CS
2, and HCN, producing a clean gas with only traces of impurities left in the gas. At the same time approximately half of the carbon dioxide is removed from the raw syngas and dissolved in the used methanol. The clean outlet gas from the methanol wash contains a certain amount of methanol,
ca. 3 g/Nm
3, as shown in
Table 2. In a general gas cleaning process, aimed at a pure syngas production, a downstream water scrubber is applied to remove the methanol in the outlet gas. In the present study, a certain amount of methanol in the cleaned outlet gas is acceptable. Therefore, a water scrubber is not included in the present process. However, part of the methanol in the cleaned outlet gas will condense in the gas cooler (E-5). To reduce operating costs, the methanol used in the methanol (A-2) should be regenerated. The used methanol passes through a flash tank (F) to reduce the pressure. This allows most of dissolved gases, together with a small volume of methanol, to escape from the liquid to the gas phase. Therefore, the outlet streams of the flash tank (F), consists of a liquid stream of partially cleaned methanol and a gas stream of a mixture of methanol, carbon dioxide and impurities. The gas is recycled to the methanol wash (A-2), via compression and cooling step (E-8) to preserve the carbon dioxide in the final clean syngas. The flashed methanol liquid stream is distillated in the flashed methanol distiller (D-1), where the heavy key bottom products of the column goes to a further step of dehydration distillation (D-2) to provide with a clean methanol in the light key distillate products of the column. Regenerated methanol is cooled and pressurized (E-6) and recycled to the methanol wash (A-2).
5.2. The Gas Quality and Process Performance
The process described above is able to remove the impurities to a content as required or close to required for a synthesis gas for further upgrading to methane, comparing numbers in
Table 1 with results in
Table 3. The Aspen Plus modeling results in
Table 3, show that the content of impurities, such as H
2S, COS, CS
2, HCl, NH
3 and HCN, is substantially decreased comparing the raw syngas with the clean syngas after the methanol wash. The concentration of H
2S, CS
2, HCl, NH
3 and HCN, and the concentration level 0.01 mg/Nm
3 for COS are, as obtained by the modeling, acceptable for further upgrading in a methanation process. Nevertheless, this result is based on a not experimentally verified modeling and should be treated as such. In any case, a guard bed, located between the cleaning process and the Sabatier reactor, to further lower the content of COS, may be included if needed. Simultaneously, as the acid gases are removed, up to 92% of the original CO
2 is preserved in the final cleaned syngas stream. The high carbon dioxide content in the final syngas is achieved by recycling of CO
2 from the flash tank (F) to the methanol wash (A-2) via the compression step. Without this recycling, the amount of the CO
2 saving would not have exceeded 60% of the CO
2 entering the process.
Among the impurities, COS displays the smallest solubility in methanol. Since water was also unable to remove COS from the gas stream, a larger number of equilibrium stages in the methanol wash (A-2) and greater methanol flow rate were employed. However, this implies a larger loss of CO2.
Therefore, to achieve a selective removal of H2S and more importantly COS, compared to CO2, a delicate design optimization for the methanol wash (A-2) was performed ensuring enough time for the solvent to absorb the H2S and COS, while keeping CO2 in the gas stream. The result of this design optimization renders the optimum solvent flow rate in the column, and number of equilibrium stages of column (A-2). The CO2 still dissolved is separated from the dirty methanol in the flash tank (F), together with impurities and traces of methanol. This gas stream is compressed and recycled back to the methanol wash as described above.
Specifications for the recycled streams are listed in
Table 4. The pressure and temperature of the recycled CO
2 stream should be increased from 1.5 bar to above 36 bar and reduced to 248 K, respectively, as indicated in
Table 3. A compressor normally has an outlet temperature limitation and therefore it is not possible to achieve the conditions needed with only one compressor step [
32]. A series of compressors with inter-cooler should be employed to fulfill the desired pressure, as well as a final cooler step to reduce the temperature of the compressed gas to the desired temperature of 248 K.
Table 3.
Flow specifications of the product gas and its constituent substances at different steps in the process as obtained from the ASPEN calculations. The specified raw gas is the main gas components in the gas obtained from the WoodRoll® gasifier gasifying wood together with estimated values for other important components.
Table 3.
Flow specifications of the product gas and its constituent substances at different steps in the process as obtained from the ASPEN calculations. The specified raw gas is the main gas components in the gas obtained from the WoodRoll® gasifier gasifying wood together with estimated values for other important components.
| | Raw Product Gas | After Water Scrubber | After Methanol Wash |
|---|
| Composition (%) | | | |
| Water | 19 | 7.53 × 10−5 | 0 |
| Carbon monoxide | 18 | 17.68 | 17.97 |
| Carbon dioxide | 15 | 22.03 | 20.74 |
| Hydrogen | 48 | 60.20 | 61.18 |
| Contaminants (mg/Nm3) | | | |
| Hydrogen sulfide | 5.97 × 102 * | 7.20 × 103 | ≈0 |
| Carbonyl sulfide | 4.64 × 102 * | 5.60 × 103 | 0.01 |
| Carbon disulfide | 7.46 × 102 * | 9.10 × 103 | 0 |
| Hydrogen chloride | 3.42 × 102 * | 0 | 0 |
| Ammonia | 1.49 × 102 * | 0 | 0 |
| Hydrogen cyanide | 9.39 × 102 * | 7.60 × 102 | 0 |
| Methanol | 0 | 0 | 3.08 × 103 |
| Total flow (kmol/s) | 0.18 | 0.15 | 0.15 |
| Total flow (kg/s) | 2.92 | 2.38 | 2.27 |
| Total flow (m3/s) | 6.22 | 0.12 | 0.09 |
| Temperature (K) | 413 | 251 | 267 |
| Pressure (bar) | 1 | 25 | 35 |
Table 4.
Flow specifications of the recycled gas at different steps in the process.
Table 4.
Flow specifications of the recycled gas at different steps in the process.
| Composition (%) | After Dirty Methanol Flash | Compressed Cooled Gas |
|---|
| H2O | 5.02 × 10−7 | 5.02 × 10−7 |
| CO | 8.46 × 10−3 | 8.46 × 10−3 |
| CO2 | 94.69 | 94.69 |
| H2 | 0.01 | 0.01 |
| Total flow (kmol/s) | 0.06 | 0.06 |
| Total flow (kg/s) | 2.38 | 2.38 |
| Total flow (m3/s) | 0.83 | 3.09 × 10−3 |
| Temperature (K) | 273 | 248 |
| Pressure (bar) | 1.5 | 36 |
The loss of methanol should be kept at a minimum due to the costs. From the detailed results, shown in
Table 5, calculated by Aspen Plus, the make-up of methanol to be added to the process to account for losses can be calculated to 10 kg/h. This loss is of the order of magnitude to be expected for a plant of the size addressed as compared to a conventional Rectisol
® process [
18].
Table 5.
Methanol flow specifications.
Table 5.
Methanol flow specifications.
| Composition (%) | Fresh Methanol | Used Methanol | Regenerated Methanol |
|---|
| H2O | 0 | 2.82 × 10−5 | ≈0 |
| CO | 0 | 0.18 | ≈0 |
| CO2 | 0 | 33.53 | ≈0 |
| H2 | 0 | 2.04 × 10−4 | ≈0 |
| H2S | 0 | 1.14 × 10−3 | ≈0 |
| COS | 0 | 5.72 × 10−4 | ≈0 |
| CS2 | 0 | 1.14 × 10−4 | ≈0 |
| HCl | 0 | ≈0 | ≈0 |
| NH3 | 0 | ≈0 | ≈0 |
| HCN | 0 | 2.43 × 10−4 | ≈0 |
| CH3OH | 100 | 66.06 | ≈100 |
| Total flow (kmol/s) | 0.15 | 0.21 | 0.15 |
| Total flow (kg/s) | 4.71 | 7.20 | 4.71 |
| Total flow (m3/s) | 7.57 × 10−3 | 9.78 × 10−3 | 8.74 × 10−3 |
| Temperature (K) | 251 | 246 | 368 |
| Pressure (bar) | 35 | 35 | 3 |
The water scrubber is the key unit to reduce the loss of methanol by removing a part of the impurities. Results from different tested scenarios show that HCN could not be removed from the used methanol with conventional regeneration processes for physical solvents. In order to avoid extra costs of regeneration process or methanol make-up, a water wash column (A-1) was designed to remove a part of impurities, especially HCN. The required water flow rate is about 220 kg/h, and the used dirty water can be handled in a biological water treatment process as reported by Lurgi, the company, which is the license holder of the Rectisol
® process [
28]. The required amount of water should be compared with the amount used in the 50 MW
fuel gasification process, 6000–8000 kg/h, and also dirty condensed water, 300–500 kg/h, produced in the cooling of the syngas. These amounts are estimated from gasification tests at 500 kWfuel, using a reactive fuel in a WoodRoll
® pilot plant. The required water flow used in the modified Rectisol
® process is well below what is used in the gasification process and also lower than the amount of dirty condensed water produced. Since a water treatment plant is anyway needed for the gasification process, the additional costs of investment for the additional amount of dirty water will not be significant. At less reactive fuels, the amount of condensed water will be even higher from the gasification process.
5.3. Comparison with a Conventional Rectisol® Process
The study does not intend to provide with a detailed economic comparison with a convention Rectisol
® process or any other process, based on physical solvents, but only to highlight the main differences. A detailed description of both single-stage and two-stage (selective) Rectisol
® process is given in literature [
18,
29]. The single-stage removes contaminants together with CO
2 in a single methanol wash as shown in
Table 6, and the two-stage take advantage of two absorption columns, with methanol as solvent, to remove the contaminants.
Table 6 presents the flow specification of the two entering streams, the raw syngas and the methanol solvent, to the absorption column and the clean syngas leaving the column in a conventional single-stage Rectisol
® process. The molar flow rate ratio of the solvent to raw syngas can be estimate to be 1.21 in the case of a conventional single-stage Rectisol
®, while it is about 0.83 in the case of the suggested configuration. This implies a relative improvement of around 30% for the proposed process. Moreover, the less severe working conditions in terms of working pressure and temperature should be used in case of the suggested configuration. As shown in
Figure 1, the selective Rectisol
® process also separates CO
2 from the feed stream and delivers a separate stream mainly consisting of CO
2. Since the detailed calculations and specifications of conventional Rectisol
® process are kept confidential [
18,
29], it is not clear if the conventional selective Rectisol
® process can remove the impurities to less than 0.1 ppm in the produced CO
2 stream. Moreover, results of Aspen Plus modelling on the different scenarios for selective Rectisol
® process, together with previous works done on this matter [
18,
29] show that the production of a separate ultra-clean stream of CO
2 is hard to achieve in a selective Rectisol
® process. On the other hand, the suggested process configuration, shown in
Figure 2, with only one-step methanol wash, meets the cleaning demand for a downstream methanation. The main reason behind using two methanol wash units in selective Rectisol
® process is to achieve a selective cleaning of H
2S together with other contaminants, over CO
2 [
13,
18]. However, having two methanol wash units in a process also two regeneration units is needed as well.
Table 6.
Flow specifications of a single-stage Rectisol process [
29].
Table 6.
Flow specifications of a single-stage Rectisol process [29].
| Composition (%) | Raw Product Gas | Methanol | Purified Syngas |
|---|
| Carbon monoxide | 19.01 | 0 | 3.42 |
| Carbon dioxide | 34.20 | 0 | 27.58 |
| Hydrogen | 46.07 | 0 | 68.17 |
| Methane | 0.18 | 0 | 0.25 |
| Hydrogen sulfide | 0.13 | 0 | 0 |
| Methanol | 0 | 1 | 1.2 × 10−2 |
| Total flow (kmol/s) | 1.17 | 1.42 | 0.79 |
| Temperature (K) | 253 | 223 | 234 |
| Pressure (bar) | 34 | 44 | 33 |
The proposed gas cleaning process in
Figure 2 has only one methanol wash unit, suggesting this configuration could save costs both in investment and in the operation of the process. A conventional Rectisol
® process is normally equipped with a N
2 stripping column in the regeneration process of used methanol directly after the flash tank [
29], which is not shown in
Figure 2, as it is absent in the proposed process configuration. The reason for this is mainly the impact water scrubber (A-1) have on the quality of the gas fed to methanol wash (A-2),
i.e., the impurities is already removed from the gas by water scrubber (A-1) and therefore the regeneration of used methanol from methanol wash (A-2) requires less processing.
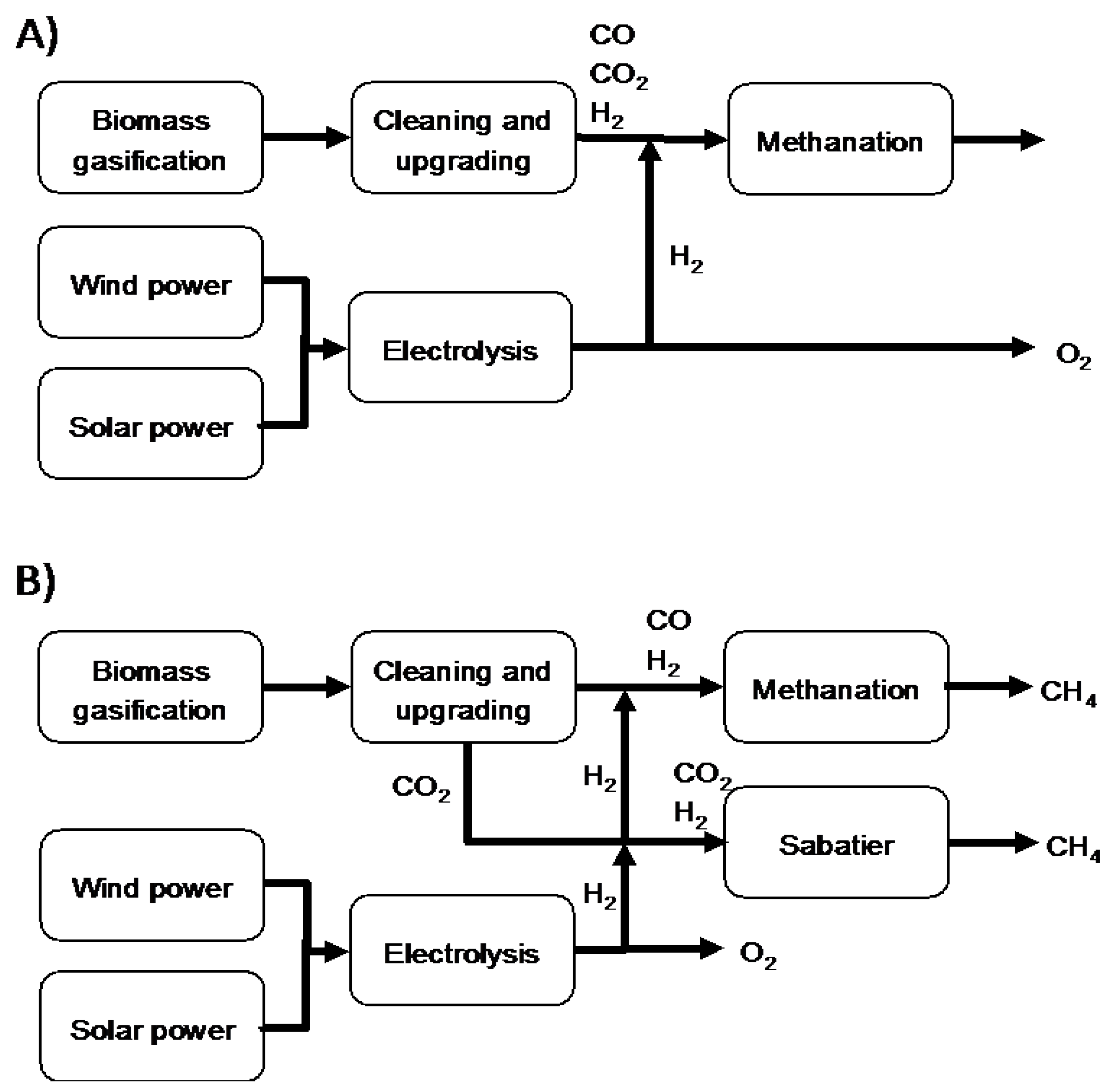
7. Future Work
Process integration of the modified process has not been considered in the present study. A techno-economic analysis, accounting for installation, and operational and maintenance costs, is proposed as a future work in order to evaluate the cost benefits for the gas cleaning process itself, as well as in the context of a complete system. For instance, the proposed gas cleaning process can be implemented in different gasification process scenarios. The general desire is to minimize the number of process steps with sustained performance, regarding robustness and yield, to achieve a more cost efficient process. Two examples of systems, aimed at production of methane or biogas, using additional hydrogen from wind or solar power, are illustrated in form of a tentative block diagram in
Figure 3. The general steps in the system includes gasification, gas cleaning and upgrading, consisting of several steps depending on the quality of the product gas and needs of the end user application, the solar or wind power plant, the electrolyzer, and finally the methanation step.
Depending on the cleaning and upgrading processes used, the production of the methane can be carried out in one or two process steps. A process where the impurities are removed, but CO, CO
2 together with hydrogen remains in the gas, suggests methanation in one process step. Using a conventional ultra-gas cleaning process, separating impurities together with CO
2, requires methane production in two separate process steps or in one step, providing simultaneous methanation of both CO
2 and CO is available [
33,
34]. Both processes would benefit from more efficient and simplified process in order to reduce costs. However, to achieve an overall optimized process efficiency the proposed gas cleaning process need to be integrated in the overall process and optimized as part of the overall process. This process integration is out of the scope of the present study, but will be investigated in future projects.
Figure 3.
Two simple block diagrams of processes for methane production from biomass including additional hydrogen from solar or wind power. (A) includes a single methanation step and (B) a methanation step and a Sabatier process converting the CO2.
Figure 3.
Two simple block diagrams of processes for methane production from biomass including additional hydrogen from solar or wind power. (A) includes a single methanation step and (B) a methanation step and a Sabatier process converting the CO2.