Hot Water Pretreatment of Boreal Aspen Woodchips in a Pilot Scale Digester
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Hot Water Extraction
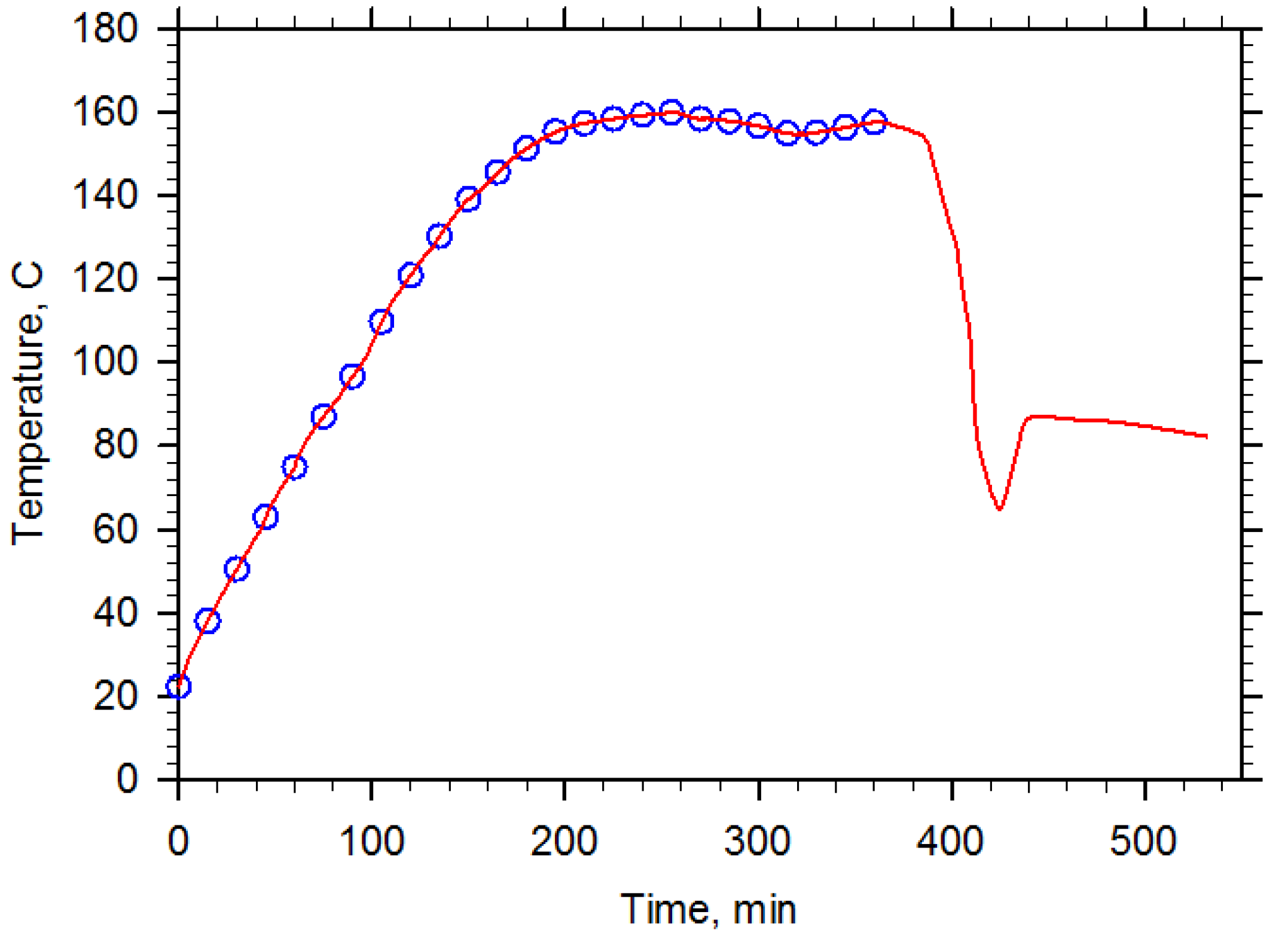
2.3. NMR Analysis
3. Results and Discussions
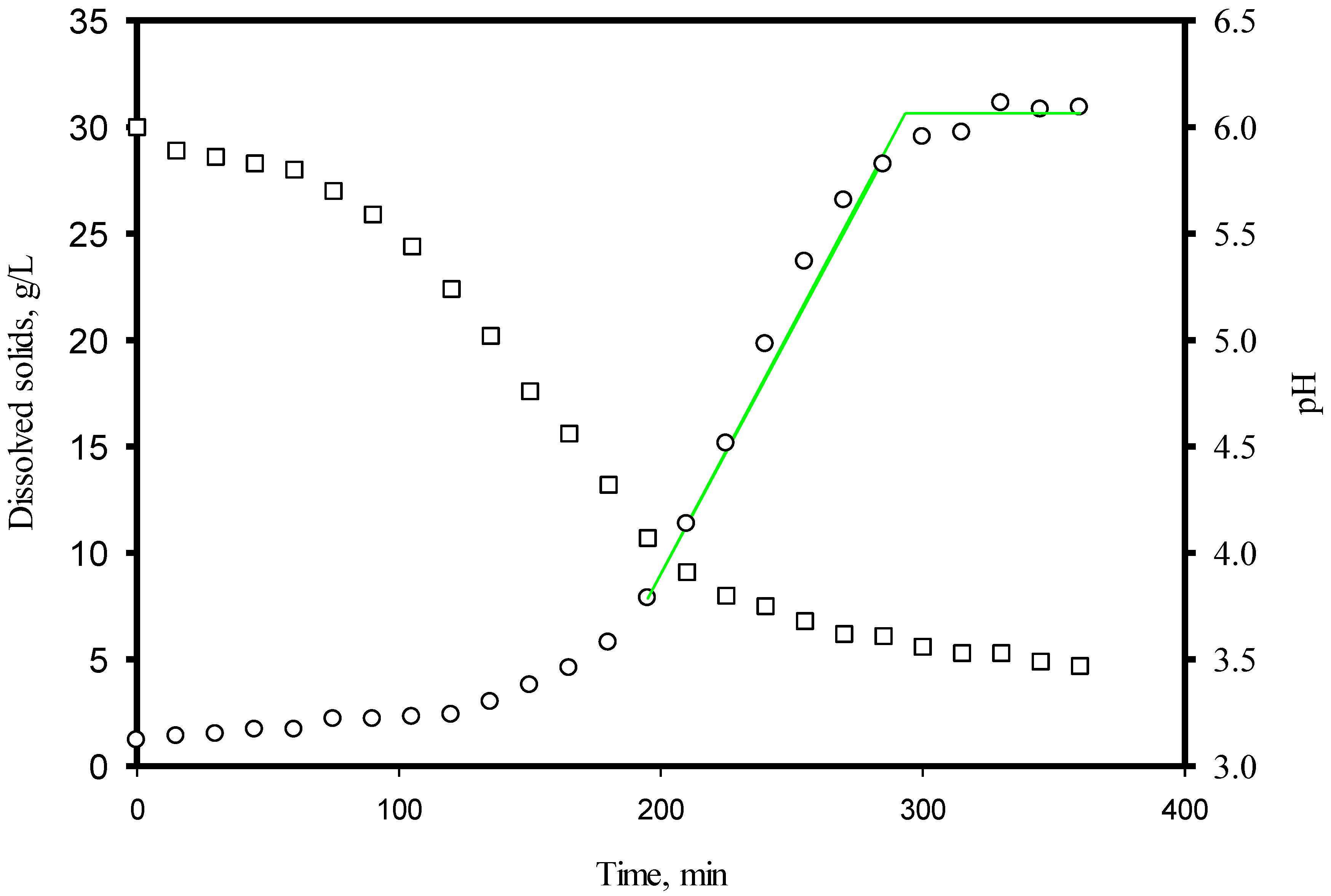
3.1. Dissolved Solids and pH
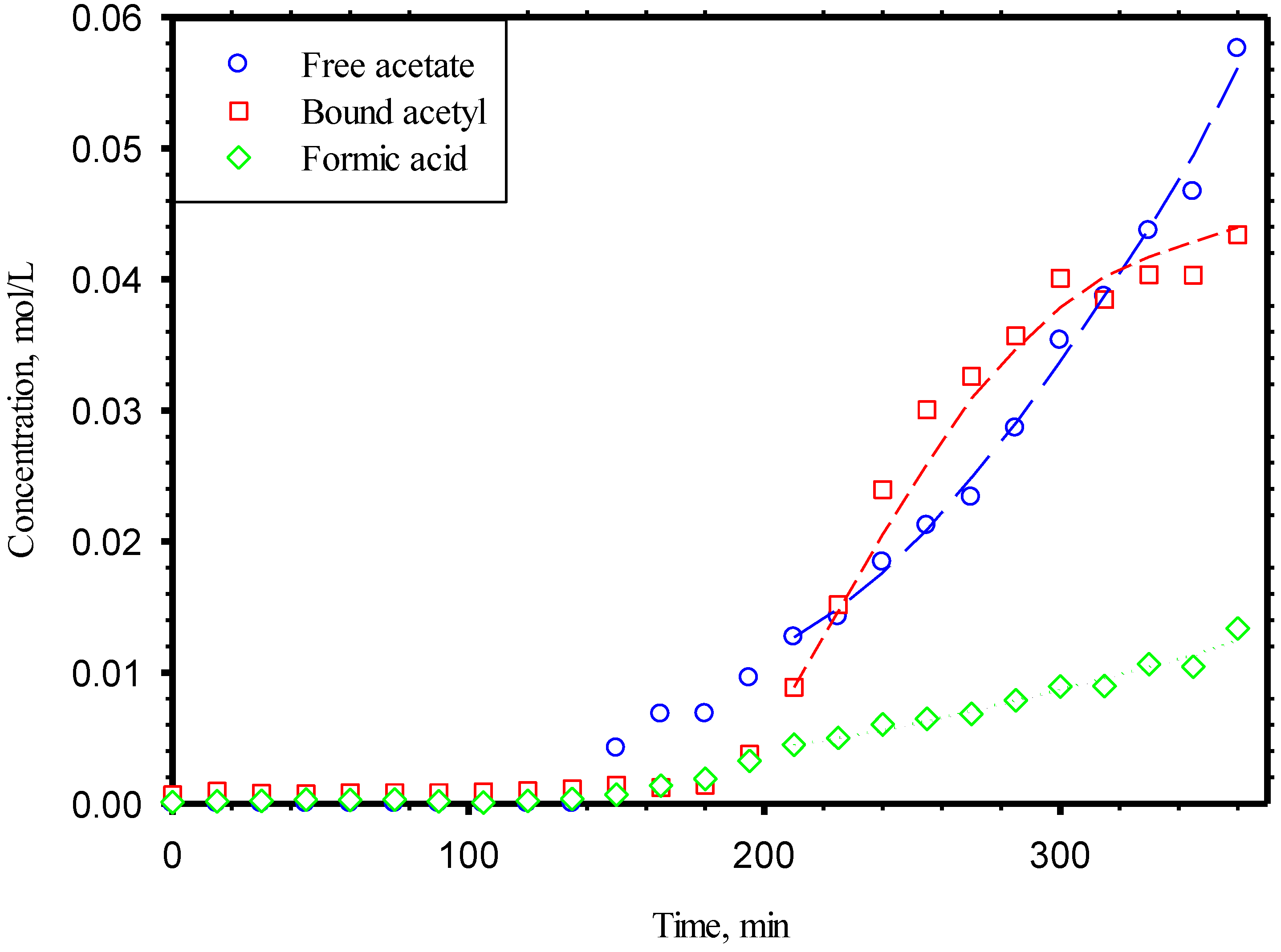
3.2. Acids
| Parameters | Temp = 160 °C |
|---|---|
| kEAcOP, min−1 | 1.17 × 10−4 |
| bHAcOP | 10 |
| kHAcOH, min−1 | 1.49 × 10−4 |
| bHAcOP | 1.03 × 105 |
| kEFOH, min−1 | 1.7 × 10−6 |
| bHF | 9.29 × 104 |
| C+o | 1.27 × 10−3 |
| KW | 2.84 × 10−12 |
| NOAC | 3.16 |
3.3. Xylose, Xylooligomer, and Furfural
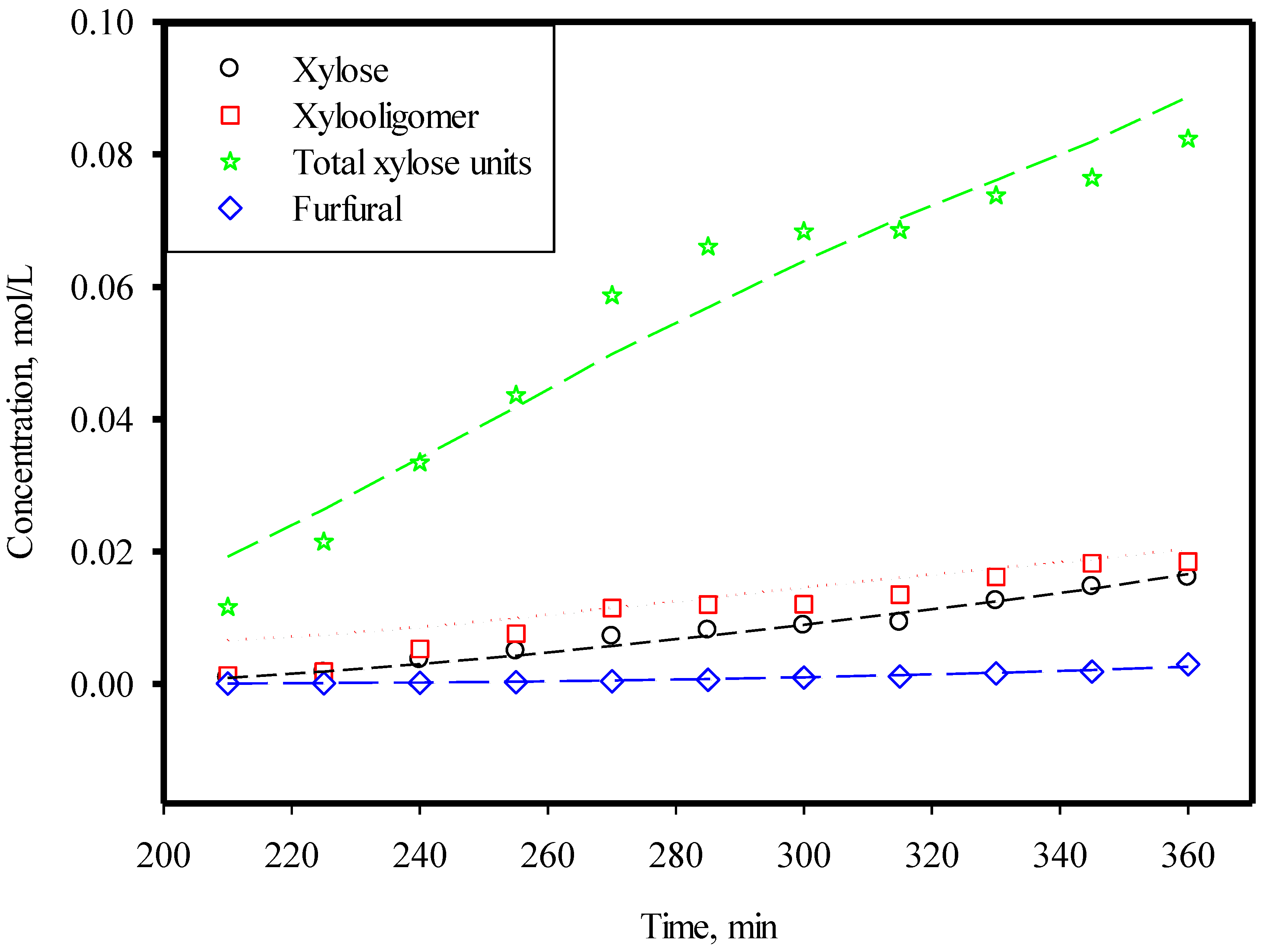
| Parameters | Temp = 160 °C |
|---|---|
| kEX, min−1 | 1.07 × 10−3 |
| kEX, min−1 | 4.95 × 10−3 |
| kDX, min−1 | 2.4 × 10−3 |
| bEX | 100 |
| bDX | 2.86 × 10−9 |
| N0 | 15 |
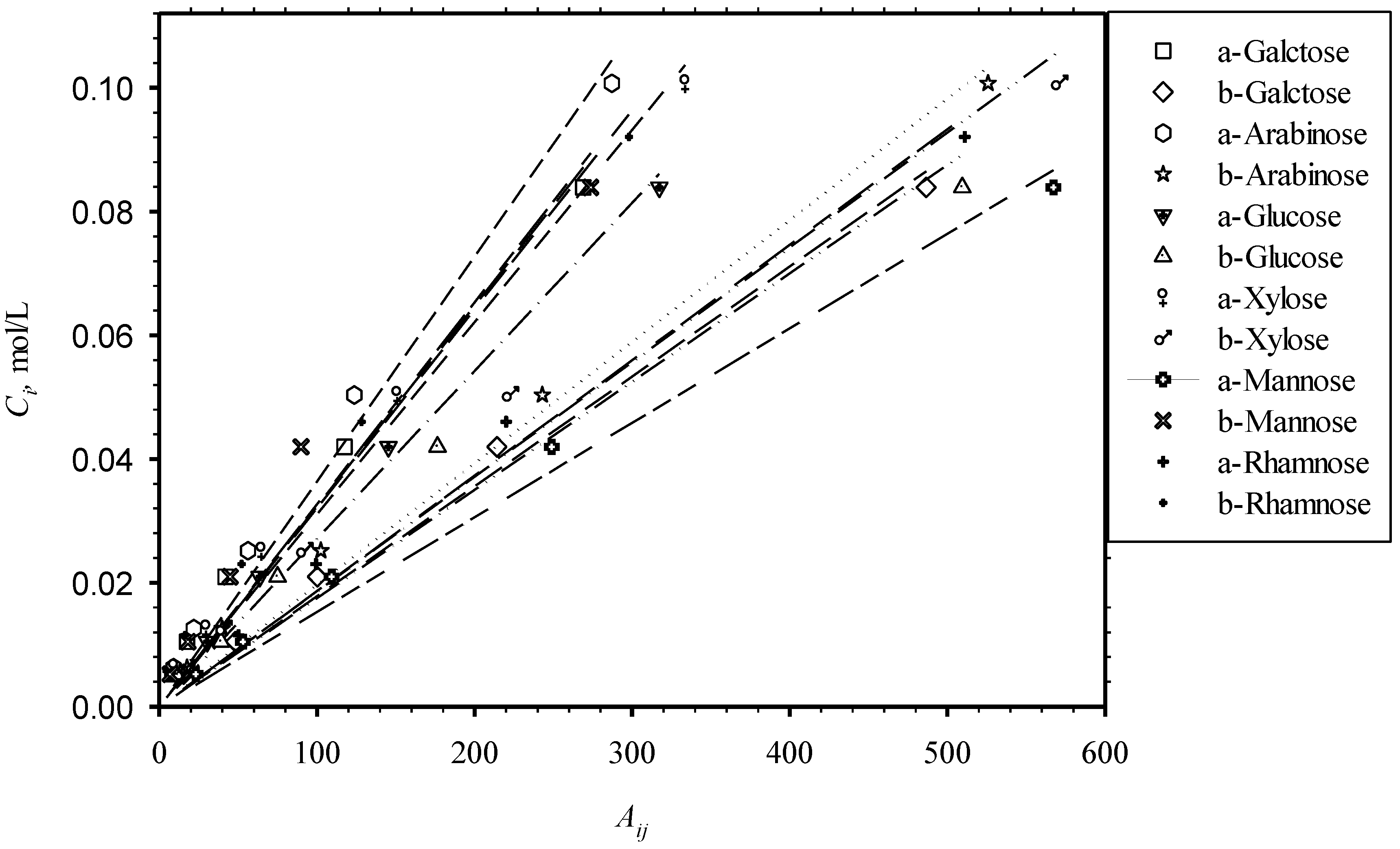
3.4. Minor Monosaccharides
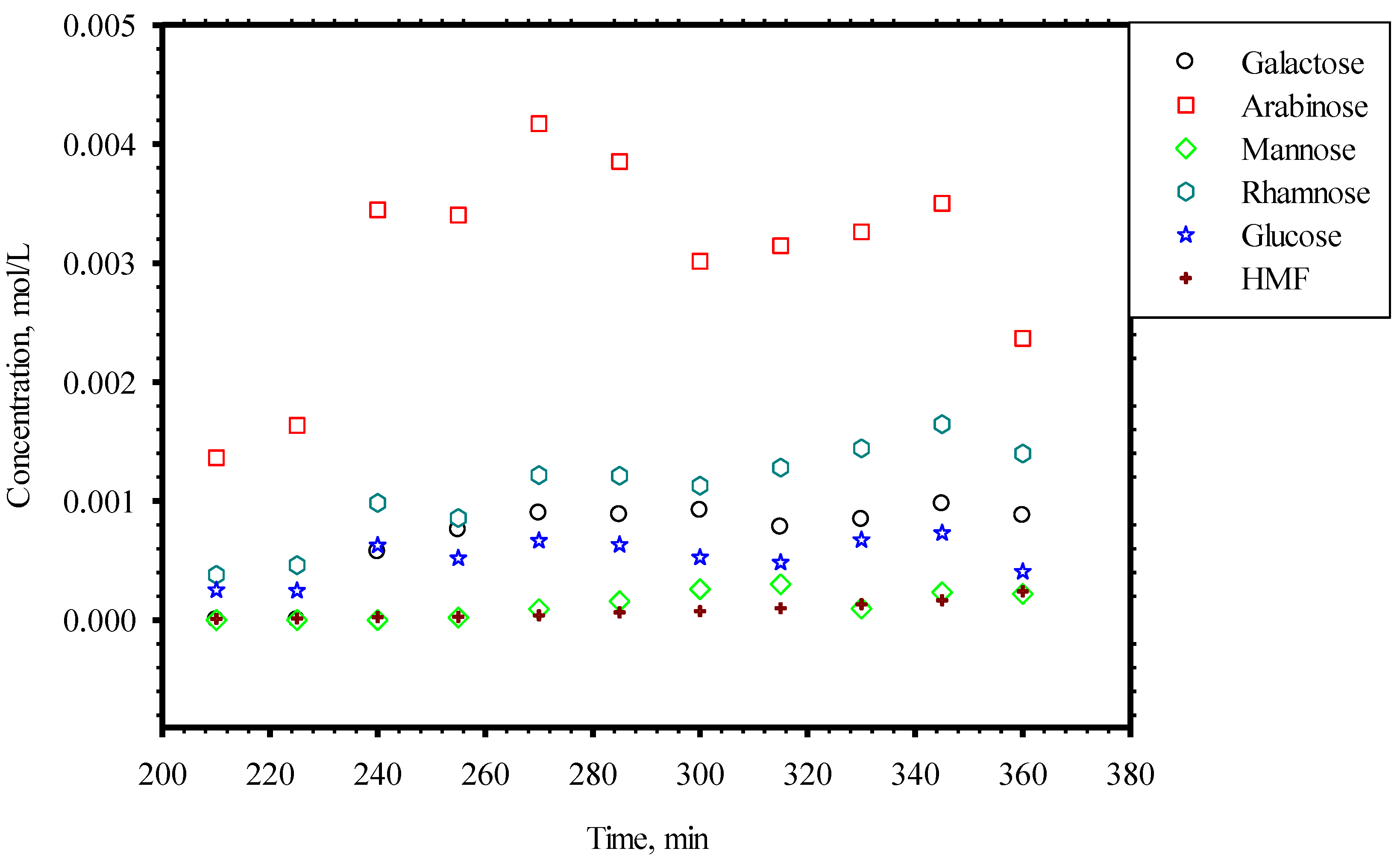
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cara, C.; Ruiz, E.; Oliva, J.M.; Sáez, F.; Castro, E. Conversion of olive tree biomass into fermentable sugars by dilute acid pretreatment and enzymatic saccharification. Bioresour. Technol. 2008, 99, 1869–1876. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Lin, L.; Liu, T.; Liu, S. Dilute sulfuric acid hydrolysis of sugar maple wood extract at atmospheric pressure. Bioresour. Technol. 2010, 101, 3586–3594. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.W.; Lamb, K.A.; Overend, R.P. Comparison of natural hemicellulose and chemically acetylated xylan as substrates for the determination of acetyl-xylan esterase activity in Aspergilli. Enzym. Microb. Technol. 1990, 12, 127–131. [Google Scholar] [CrossRef]
- Romaní, A.; Garrote, G.; Alonso, J.L.; Parajó, J.C. Experimental assessment on the enzymatic hydrolysis of hydrothermally pretreated eucalyptus globulus wood. Ind. Eng. Chem. Res. 2010, 49, 4653–4663. [Google Scholar] [CrossRef]
- Borrega, M.; Nieminen, K.; Sixta, H. Degradation kinetics of the main carbohydrates in birch wood during hot water extraction in a batch reactor at elevated temperatures. Bioresour. Technol. 2011, 102, 10724–10732. [Google Scholar] [CrossRef] [PubMed]
- Nabarlatz, D.; Ebringerová, A.; Montané, D. Autohydrolysis of agricultural by-products for the production of xylo-oligosaccharides. Carbohydr. Polym. 2007, 69, 20–28. [Google Scholar] [CrossRef]
- Liu, S. Woody biomass: Niche position as a source of sustainable renewable chemicals and energy and kinetics of hot-water extraction/hydrolysis. Biotechnol. Adv. 2010, 28, 563–582. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, S.J. Pretreatment technologies for biological and chemical conversion of woody biomass. Tappi J. 2012, 11, 9–16. [Google Scholar]
- Zhao, Y.L.; Wang, Y.; Zhu, J.Y.; Ragauskas, A.; Deng, Y.L. Enhanced enzymatic hydrolysis of spruce by alkaline pretreatment at low temperature. Biotechnol. Bioeng. 2008, 99, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Fatehi, P.; Ni, Y. Removal of inhibitors from pre-hydrolysis liquor of kraft-based dissolving pulp production process using adsorption and flocculation processes. Bioresour. Technol. 2012, 116, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Shupe, A.M.; Liu, S. Ethanol fermentation from hydrolysed hot-water wood extracts by pentose fermenting yeasts. Biomass Bioenergy 2012, 39, 31–38. [Google Scholar] [CrossRef]
- Yoon, S.H.; van Heiningen, A. Kraft pulping and papermaking properties of hot-water pre-extracted loblolly pine in an integrated forest products biorefinery. Tappi J. 2008, 7, 22–27. [Google Scholar]
- García, J.C.; Zamudio, M.A.M.; Pérez, A.; López, F.; Colodette, J.L. Search for optimum conditions of Paulownia autohydrolysis process and influence in pulping process. Environ. Process Sustain. Energy 2011, 30, 92–101. [Google Scholar] [CrossRef]
- Jun, A.; Tschirner, U.W.; Tauer, Z. Hemicellulose extraction from aspen chips prior to kraft pulping utilizing kraft white liquor. Biomass Bioenergy 2012, 37, 229–236. [Google Scholar] [CrossRef]
- Lu, H.; Hu, R.; Ward, A.; Amidon, T.E.; Liang, B.; Liu, S. Hot-water extraction and its effect on soda pulping of aspen woodchips. Biomass Bioenergy 2012, 39, 5–13. [Google Scholar] [CrossRef]
- Liu, S.; Lu, H.; Hu, R.; Shupe, A.; Lin, L.; Liang, B. A sustainable woody biomass biorefinery. Biotechnol. Adv. 2012, 30, 785–810. [Google Scholar] [CrossRef] [PubMed]
- Conner, A. Kinetic modeling of hardwood prehydrolysis. Part I. Xylan removal by water prehydrolysis. Wood Fiber Sci. 1984, 16, 268–277. [Google Scholar]
- Garrote, G.; Dominguez, H.; Parajo, J.C. Kinetic modelling of corncob autohydrolysis. Process Biochem. 2001, 36, 571–578. [Google Scholar] [CrossRef]
- Maloney, M.T.; Chapman, T.W.; Baker, A.J. Dilute acid hydrolysis of paper birch: Kinetics studies of xylan and acetyl-group hydrolysis. Biotechnol. Bioeng. 1985, 27, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Nabarlatz, D.; Farriol, X.; Montane, D. Kinetic modeling of the autohydrolysis of lignocellulosic biomass for the production of hemicellulose-derived ligosaccharides. Ind. Eng. Chem. Res. 2004, 43, 4124–4131. [Google Scholar] [CrossRef]
- Nabarlatz, D.; Farriol, X.; Montané, D. Autohydrolysis of almond shells for the production of xylo-oligosaccharides: Product characteristics and reaction kinetics. Ind. Eng. Chem. Res. 2005, 44, 7746–7755. [Google Scholar] [CrossRef]
- Morinelly, J.E.; Jensen, J.R.; Browne, M.; Co, T.B.; Shonnard, D.R. Kinetic characterization of xylose monomer and oligomer concentrations during dilute acid pretreatment of lignocellulosic biomass from forests and switchgrass. Ind. Eng. Chem. Res. 2009, 48, 9877–9884. [Google Scholar] [CrossRef]
- Zhuang, X.; Yuan, Z.; Ma, L.; Wu, C.; Xu, M.; Xu, J.; Zhu, S.; Qi, W. Kinetic study of hydrolysis of xylan and agricultural wastes with hot liquid water. Biotechnol. Adv. 2009, 27, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Mittal, A.; Scott, G.; Amidon, T.; Kiemle, D.; Stipanovic, A. Quantitative analysis of sugars in wood hydrolyzates with 1H NMR during the autohydrolysis of hardwoods. Bioresour. Technol. 2009, 100, 6398–6406. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.J. Chemical reactions on surfaces during woody biomass hydrolysis. J. Bioprocess Eng. Biorefinery 2013, 2, 125–142. [Google Scholar] [CrossRef]
- Kim, Y.; Kreke, T.; Mosier, N.S.; Ladisch, M.R. Severity factor coefficients for subcritical liquid hot water pretreatment of hardwood chips. Biotechnol. Bioeng. 2014, 111, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Mittal, A.; Chatterjee, S.G.; Scott, G.M.; Amidon, T.E. Modeling xylan solubilization during autohydrolysis of sugar maple and aspen wood chips: Reaction kinetics and mass transfer. Chem. Eng. Sci. 2009, 64, 3031–3041. [Google Scholar] [CrossRef]
- Kiemle, D.J.; Stipanovic, A.J.; Mayo, K.E. Proton NMR methods in the compositional characterization of polysaccharides. In Hemicelluloses: Science and Technology; Gatenholm, P., Tenhanen, M., Eds.; American Chemical Society Publication: Washington, DC, USA, 2004; Volume 864, pp. 122–139. [Google Scholar]
- Shupe, A.M.; Kiemle, D.J.; Liu, S. Quantitative 2D HSQC NMR analysis of mixed wood sugars in hemicellulosic hydrolysate fermentation broth. J. Bioprocess Eng. Biorefinery 2012, 1, 93–100. [Google Scholar] [CrossRef]
- Yan, J.; Kiemle, D.; Liu, S. Quantification of xylooligomers in hot water wood extract by 1H–13C heteronuclear single quantum coherence NMR. Carbohydr. Polym. 2015, 117, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Qin, M.; Xu, C.; Chen, X. Hot water extraction of hemicelluloses from aspen wood chips of different sizes. BioResources 2013, 8, 5690–5700. [Google Scholar]
- Liu, S.J. A kinetic model on autocatalytic reactions in woody biomass hydrolysis. J. Biobased Mater. Bioenergy 2008, 2, 135–147. [Google Scholar] [CrossRef]
- Li, H.; Saeed, A.; Jahan, M.S.; Ni, Y.; van Heiningen, A. Hemicellulose removal from hardwood chips in the pre-hydrolysis step of the kraft-based dissolving pulp production process. J. Wood Chem. Technol. 2010, 30, 48–60. [Google Scholar] [CrossRef]
- Amidon, T.E.; Wood, C.D.; Shupe, A.M.; Wang, Y.; Graves, M.; Liu, S. Biorefinery: Conversion of woody biomass to chemicals, energy and materials. J. Biobased Mater. Bioenergy 2008, 2, 100–120. [Google Scholar] [CrossRef]
- Bose, S.K.; Barber, V.A.; Alves, E.F.; Kiemle, D.J.; Stipanovic, A.J.; Francis, R.C. An improved method for the hydrolysis of hardwood carbohydrates to monomers. Carbohydr. Polym. 2009, 78, 396–401. [Google Scholar] [CrossRef]
- Sjöström, E. Wood polysaccharides. In Wood Chemistry, 2nd ed.; Sjöström, E., Ed.; Academic Press: San Diego, CA, USA, 1993; pp. 51–70. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, J.; Liu, S. Hot Water Pretreatment of Boreal Aspen Woodchips in a Pilot Scale Digester. Energies 2015, 8, 1166-1180. https://doi.org/10.3390/en8021166
Yan J, Liu S. Hot Water Pretreatment of Boreal Aspen Woodchips in a Pilot Scale Digester. Energies. 2015; 8(2):1166-1180. https://doi.org/10.3390/en8021166
Chicago/Turabian StyleYan, Jipeng, and Shijie Liu. 2015. "Hot Water Pretreatment of Boreal Aspen Woodchips in a Pilot Scale Digester" Energies 8, no. 2: 1166-1180. https://doi.org/10.3390/en8021166
APA StyleYan, J., & Liu, S. (2015). Hot Water Pretreatment of Boreal Aspen Woodchips in a Pilot Scale Digester. Energies, 8(2), 1166-1180. https://doi.org/10.3390/en8021166





