Screen-Printed Electrodes: New Tools for Developing Microbial Electrochemistry at Microscale Level
Abstract
:1. Introduction
2. Results and Discussion
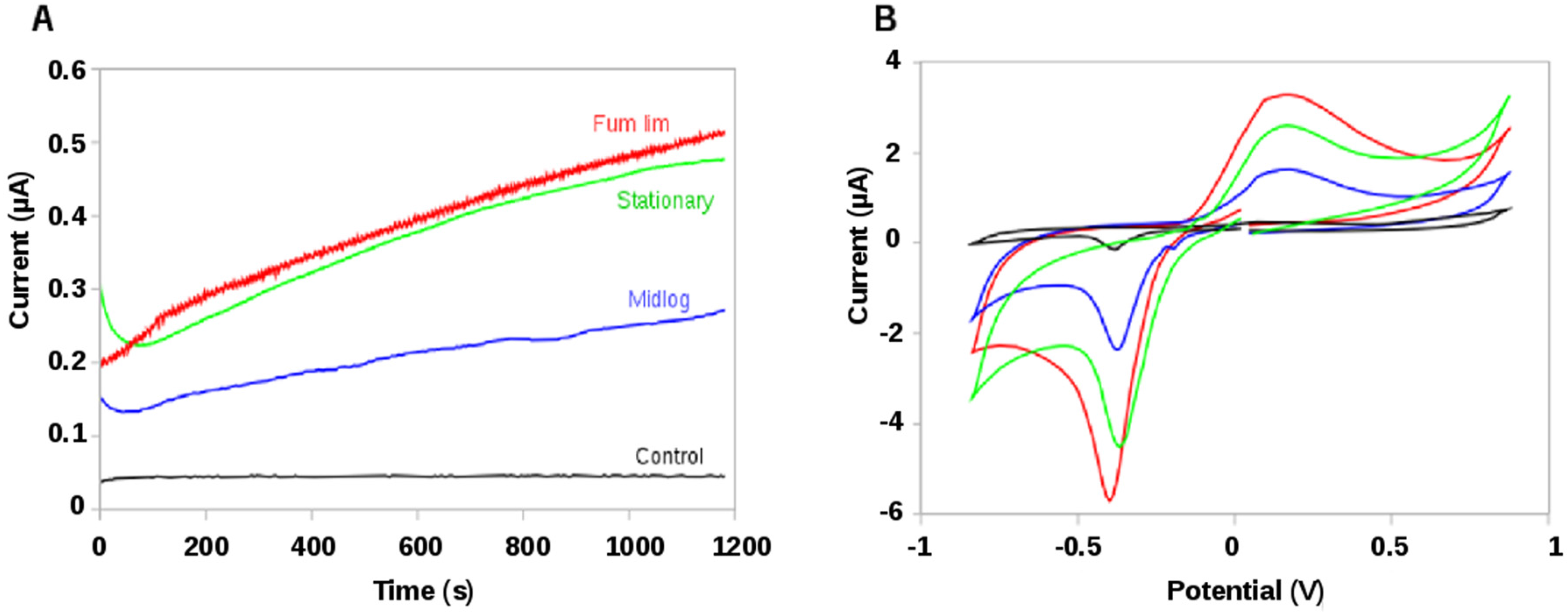
2.1. Screen-Printed Electrodes (SPEs): Testing Electroactivity in Geobacter
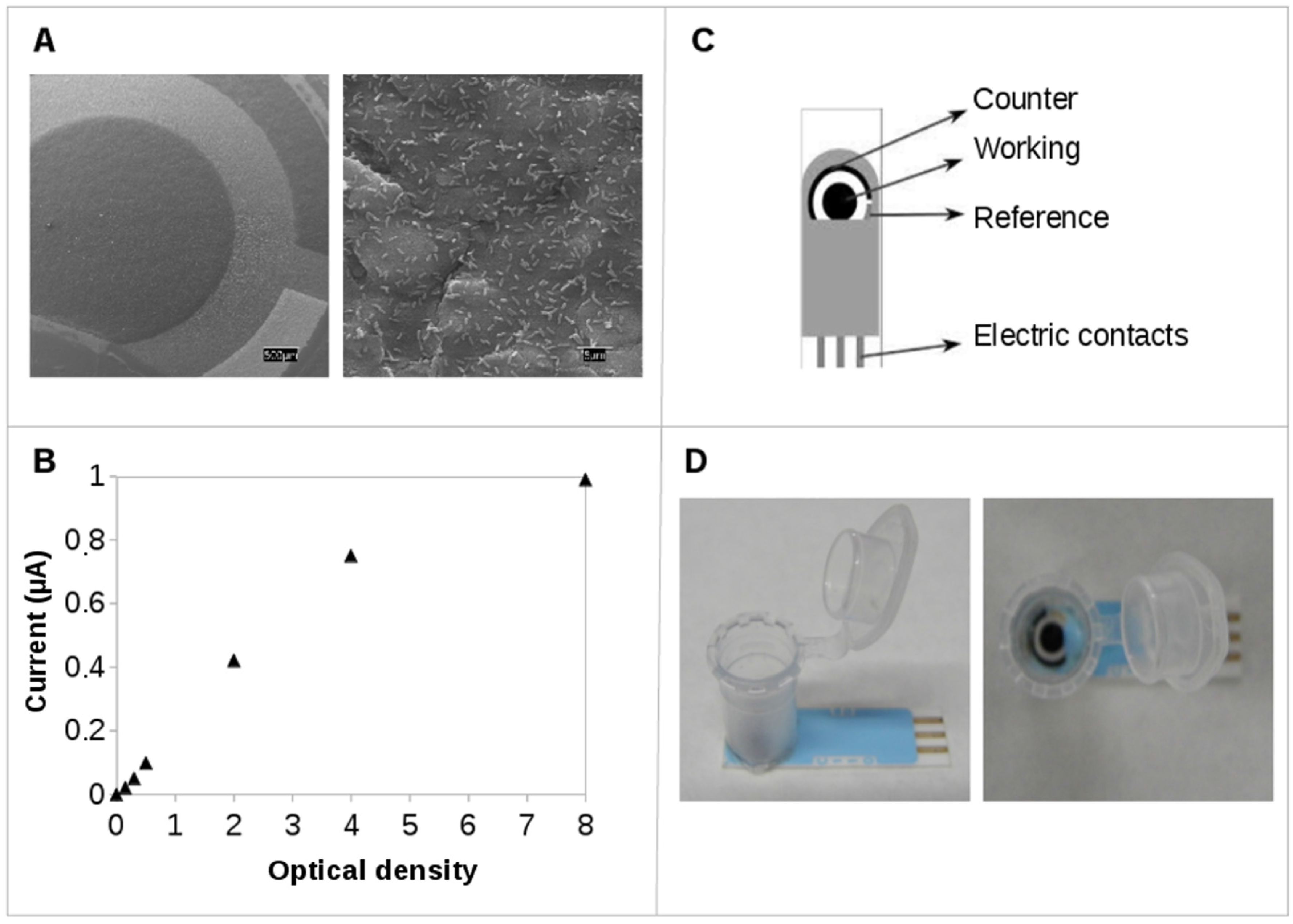
2.2. Practical Applications
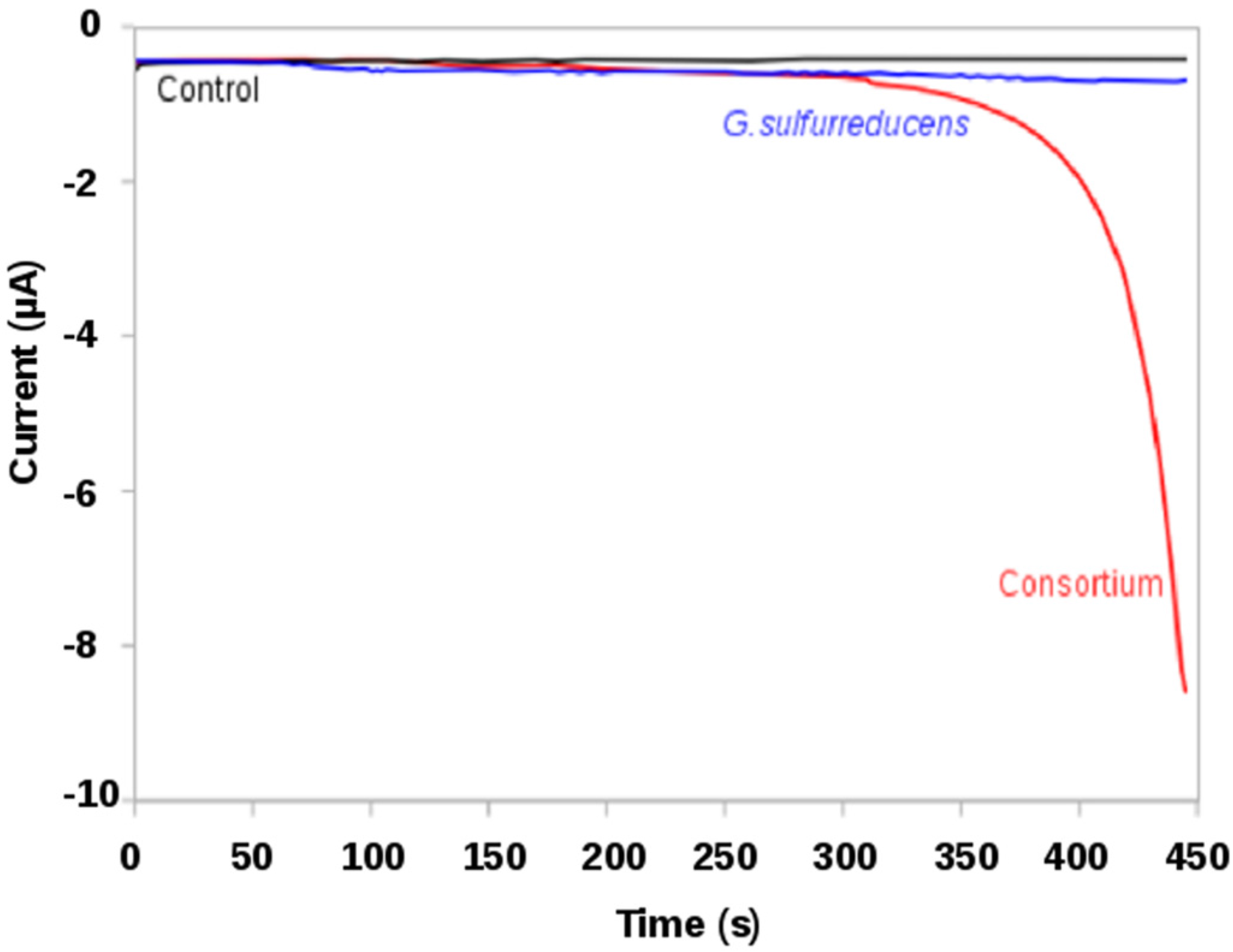
2.2.1. Screening of Metabolic Features from a Microbial Consortium: Screen-Printed Electrodes (SPEs) Acting as Electron Donor
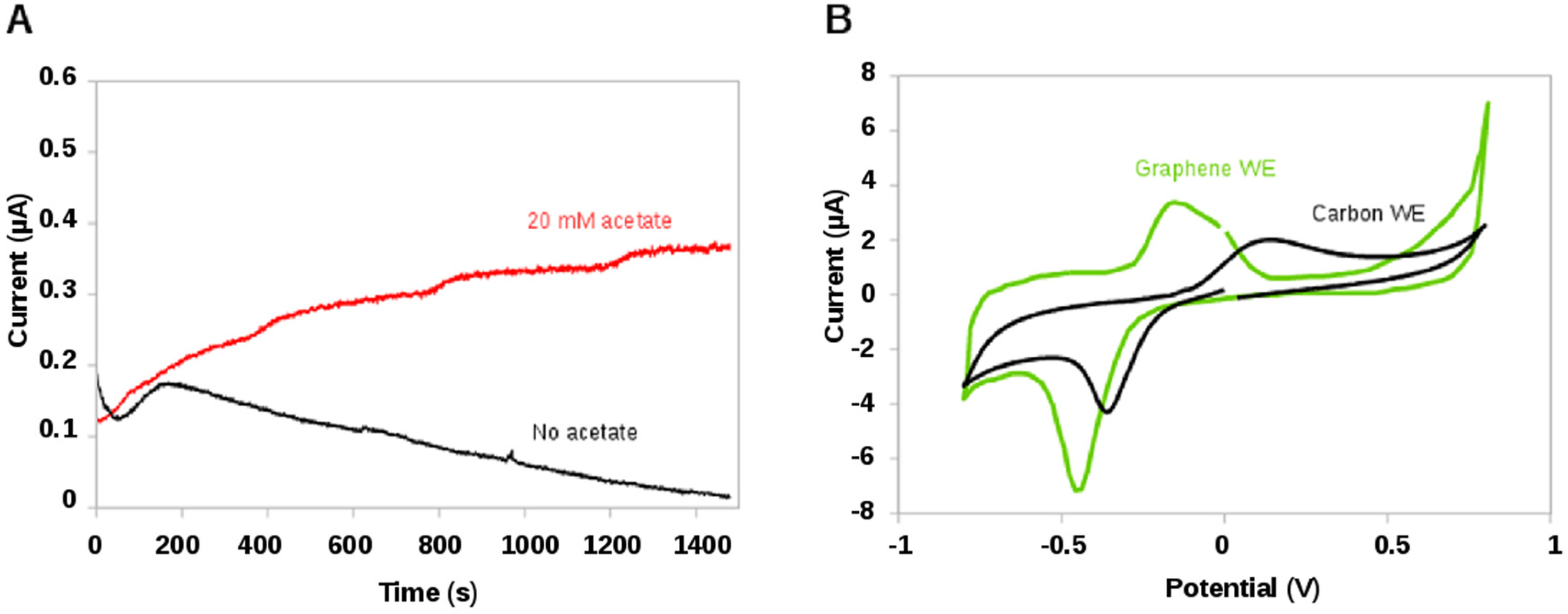
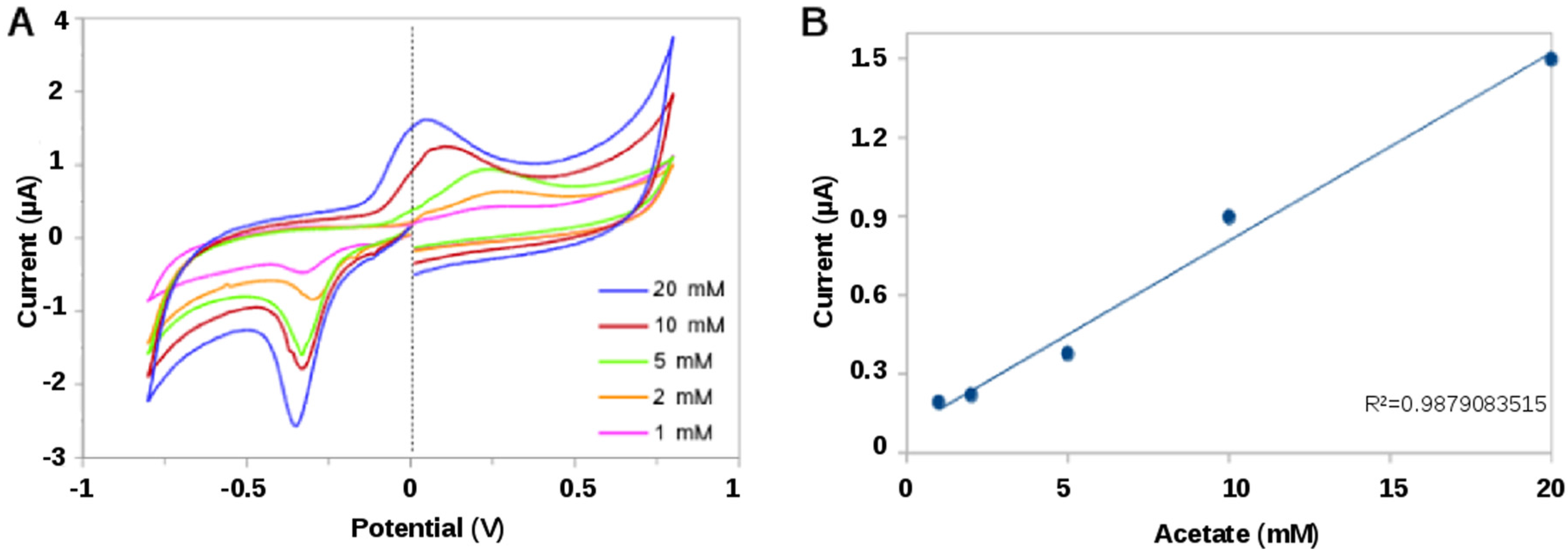
2.2.2. Bioelectrochemical Sensing of Acetate
2.2.3. Effects of a Background of Real Urban Wastewater

3. Experimental Section
3.1. Bacterial Culture
3.2. Electrochemical Analysis
3.3. Perfomance of the Acetate Biosensor Assay
3.4. Scanning Electron Microscopy (SEM)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bond, D.R.; Lovley, D.R. Electricity production by Geobacter sulfurreducens attached to electrodes. Appl. Environ. Microbiol. 2003, 69, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Park, H.S.; Hyun, M.S.; Chang, I.S.; Kim, M.; Kim, B.H. A mediator-less microbial fuel cell using a metal reducing bacterium, Shewanella putrefaciens. Enzym. Microb. Technol. 2002, 30, 145–152. [Google Scholar] [CrossRef]
- Rodrigo, J.; Boltes, K.; Esteve-Nuñez, A. Microbial-electrochemical bioremediation and detoxification of dibenzothiophene-polluted soil. Chemosphere 2014, 101, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Schröder, U.; Harnisch, F.; Angenent, L.T. Microbial electrochemistry and technology: Terminology and classification. Energy Environ. Sci. 2015, 8, 513–519. [Google Scholar] [CrossRef]
- Rozendal, R.A.; Hamelers, H.V.M.; Rabaey, K.; Keller, J.; Buisman, C.J.N. Towards practical implementation of bioelectrochemical wastewater treatment. Trends Biotechnol. 2008, 26, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Clauwaert, P.; Aelterman, P.; Pham, T.H.; de Schamphelaire, L.; Carballa, M.; Rabaey, K.; Verstraete, W. Minimizing losses in bio-electrochemical systems: The road to applications. Appl. Microbiol. Biotechnol. 2008, 79, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Busalmen, J.P.; Esteve-Nuñez, A.; Feliu, J.M. Whole cell electrochemistry of electricity-producing microorganisms evidence an adaptation for optimal exocellular electron transport. Environ. Sci. Technol. 2008, 42, 2445–2450. [Google Scholar] [CrossRef] [PubMed]
- Kuzume, A.; Zhumaev, U.; Li, J.; Fu, Y.; Fueeg, M.; Esteve-Nunez, A.; Wandlowski, T. An in-situ surface electrochemistry approach toward whole-cell studies: Charge transfer between Geobacter sulfurreducens and electrified metal/electrolyte interfaces through linker molecules. Electrochim. Acta 2013, 112, 933–942. [Google Scholar] [CrossRef]
- Kuzume, A.; Zhumaev, U.; Li, J.; Fu, Y.; Füeg, M.; Estévez, M.; Borjas, Z.; Wandlowski, T.; Esteve-Nuñez, A. An in situ surface electrochemistry approach towards whole-cell studies: The structure and reactivity of a Geobacter sulfurreducens submonolayer on electrified metal/electrolyte interfaces. Phys. Chem. Chem. Phys. 2014, 16, 22229–22236. [Google Scholar] [CrossRef] [PubMed]
- Schrott, G.D.; Bonanni, P.S.; Robuschi, L.; Esteve-Nuñez, A.; Busalmen, J.P. Electrochemical insight into the mechanism of electron transport in biofilms of Geobacter sulfurreducens. Electrochim. Acta 2011, 56, 10791–10795. [Google Scholar] [CrossRef]
- Carmona-Martínez, A.A.; Harnisch, F.; Kuhlicke, U.; Neu, T.R.; Schröder, U. Electron transfer and biofilm formation of Shewanella putrefaciens as function of anode potential. Bioelectrochemistry 2013, 93, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Busalmen, J.P.; Esteve-Núñez, A.; Berná, A.; Feliu, J.M. C-type cytochromes wire electricity-producing bacteria to electrodes. Angew. Chem. Int. Ed. 2008, 47, 4874–4877. [Google Scholar] [CrossRef] [PubMed]
- Harnisch, F.; Rabaey, K. The diversity of techniques to study electrochemically active biofilms highlights the need for standardization. ChemSusChem 2012, 5, 1027–1038. [Google Scholar] [CrossRef] [PubMed]
- Schröder, U. Editorial: Microbial Fuel Cells and Microbial Electrochemistry: Into the Next Century! ChemSusChem 2012, 5, 959. [Google Scholar] [CrossRef] [PubMed]
- Davila, D.; Esquivel, J.P.; Sabate, N.; Mas, J. Silicon-based microfabricated microbial fuel cell toxicity sensor. Biosens. Bioelectron. 2011, 26, 2426–2430. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Y.; LeDuc, P.R.; Gregory, K.B. Microbial electricity generation via microfluidic flow control. Biotechnol. Bioeng. 2011, 108, 2061–2069. [Google Scholar] [CrossRef] [PubMed]
- Fraiwan, A.; Dai, C.; Sidhu, N.K.; Rastogi, A.; Choi, S. A micro-sized microbial fuel cell with electrochemical sensing functionality. In Proceedings of the 2014 9th IEEE International Conference on Nano/Micro Engineered and Molecular Systems (NEMS), Waikiki Beach, HI, USA, 13–16 April 2014.
- Hayat, A.; Marty, J.L. Disposable screen printed electrochemical sensors: Tools for environmental monitoring. Sensors 2014, 14, 10432–10453. [Google Scholar] [CrossRef] [PubMed]
- Taleat, Z.; Khoshroo, A.; Mazloum-Ardakani, M. Screen-printed electrodes for biosensing: A review (2008–2013). Microchim. Acta 2014, 181, 865–891. [Google Scholar] [CrossRef]
- Gómez-Mingot, M.; Iniesta, J.; Montiel, V.; Kadara, R.O.; Banks, C.E. Screen printed graphite macroelectrodes for the direct electron transfer of cytochrome c. Analyst 2011, 136, 2146–2150. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.-L.; Lien, C.-H.; Vijayakumar, P.S.; Hsieh, P.-H.; Zen, J.-M. Electrochemical regulation of microbial growth on disposable screen printed carbon electrodes. Electrochim. Acta 2012, 82, 103–108. [Google Scholar] [CrossRef]
- Tran, H.V.; Piro, B.; Reisberg, S.; Huy Nguyen, L.; Dung Nguyen, T.; Duc, H.T.; Pham, M.C. An electrochemical ELISA-like immunosensor for miRNAs detection based on screen-printed gold electrodes modified with reduced graphene oxide and carbon nanotubes. Biosens. Bioelectron. 2014, 62, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Asturias-Arribas, L.; Asuncion Alonso-Lomillo, M.; Dominguez-Renedo, O.; Julia Arcos-Martinez, M. Cytochrome P450 2D6 based electrochemical sensor for the determination of codeine. Talanta 2014, 129, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Esteve-Núñez, A.; Busalmen, J.P.; Berná, A.; Gutiérrez-Garrán, C.; Feliu, J.M. Opportunities behind the unusual ability of Geobacter sulfurreducens for exocellular respiration and electricity production. Energy Environ. Sci. 2011, 4, 2066–2069. [Google Scholar] [CrossRef]
- Sudhakara Prasad, K.; Arun, A.B.; Rekha, P.D.; Young, C.-C.; Chang, J.-L.; Zen, J.-M. A microbial sensor based on direct electron transfer at Shewanella sp. drop-coated screen-printed carbon electrodes. Electroanalysis 2009, 21, 1646–1650. [Google Scholar] [CrossRef]
- Marsili, E.; Baron, D.B.; Shikhare, I.D.; Coursolle, D.; Gralnick, J.A.; Bond, D.R. Shewanella secretes flavins that mediate extracellular electron transfer. Proc. Natl. Acad. Sci. USA 2008, 105, 3968–3973. [Google Scholar] [CrossRef] [PubMed]
- Kotloski, N.J.; Gralnick, J.A. Flavin electron shuttles dominate extracellular electron transfer by Shewanella oneidensis. MBio 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R. Live wires: Direct extracellular electron exchange for bioenergy and the bioremediation of energy-related contamination. Energy Environ. Sci. 2011, 4, 4896–4906. [Google Scholar] [CrossRef]
- Maestro, B.; Ortiz, J.M.; Schrott, G.; Busalmen, J.P.; Climent, V.; Feliu, J.M. Crystallographic orientation and electrode nature are key factors for electric current generation by Geobacter sulfurreducens. Bioelectrochemistry 2014, 98, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Esteve-Núñez, A.; Sosnik, J.; Visconti, P.; Lovley, D.R. Fluorescent properties of c-type cytochromes reveal their potential role as an extracytoplasmic electron sink in Geobacter sulfurreducens. Environ. Microbiol. 2008, 10, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.; Helmus, R.A.; Stanley, B.A.; Zhu, J.; Liermann, L.J.; Brantley, S.L.; Tien, M. Survival during long-term starvation: Global proteomics analysis of Geobacter sulfurreducens under prolonged electron-acceptor limitation. J. Proteome Res. 2013, 12, 4316–4326. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.-H.R.; Hixson, K.K.; Giometti, C.S.; Stanley, A.; Esteve-Núñez, A.; Khare, T.; Tollaksen, S.L.; Zhu, W.; Adkins, J.N.; Lipton, M.S.; et al. The proteome of dissimilatory metal-reducing microorganism Geobacter sulfurreducens under various growth conditions. Biochim. Biophys. Acta 2006, 1764, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Castro Neto, A.H.; Guinea, F.; Peres, N.M.R.; Novoselov, K.S.; Geim, A.K. The electronic properties of graphene. Rev. Mod. Phys. 2009, 81, 109–162. [Google Scholar] [CrossRef]
- Guo, W.; Cui, Y.; Song, H.; Sun, J. Layer-by-layer construction of graphene-based microbial fuel cell for improved power generation and methyl orange removal. Bioprocess Biosyst. Eng. 2014, 37, 1749–1758. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.G.; Hashmi, S.; Karthikeyan, C.; GhavamiNejad, A.; Vatankhah-Varnoosfaderani, M.; Stadler, F.J. Graphene oxide/carbon nanotube composite hydrogels-versatile materials for microbial fuel cell applications. Macromol. Rapid Commun. 2014, 35, 1861–1865. [Google Scholar] [CrossRef] [PubMed]
- Estevez-Canales, M.; Kuzume, A.; Borjas, Z.; Füeg, M.; Lovley, D.; Wandlowski, T.; Esteve-Núñez, A. A severe reduction in the cytochrome C content of Geobacter sulfurreducens eliminates its capacity for extracellular electron transfer. Environ. Microbiol. Rep. 2015, 7, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; Keller, J. Microbial fuel cell cathodes: From bottleneck to prime opportunity? Water Sci. Technol. 2008, 57, 655–659. [Google Scholar] [CrossRef] [PubMed]
- Caccavo, F.; Lonergan, D.J.; Lovley, D.R.; Davis, M.; Stolz, J.F.; McInerney, M.J. Geobacter sulfurreducens sp. nov., a hydrogen- and acetate-oxidizing dissimilatory metal-reducing microorganism. Appl. Environ. Microbiol. 1994, 60, 3752–3759. [Google Scholar] [PubMed]
- Nevin, K.P.; Woodard, T.L.; Franks, A.E.; Summers, Z.M.; Lovley, D.R. Microbial electrosynthesis: Feeding microbes electricity to convert carbon dioxide and water to multicarbon extracellular organic compounds. MBio 2010, 1. [Google Scholar] [CrossRef] [PubMed]
- Coma, M.; Puig, S.; Pous, N.; Balaguer, M.D.; Colprim, J. Biocatalysed sulphate removal in a BES cathode. Bioresour. Technol. 2013, 130, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Clauwaert, P.; Rabaey, K.; Aelterman, P.; de Schamphelaire, L.; Pham, T.H.; Boeckx, P.; Boon, N.; Verstraete, W. Biological denitrification in microbial fuel cells. Environ. Sci. Technol. 2007, 41, 3354–3360. [Google Scholar] [CrossRef] [PubMed]
- Gregory, K.B.; Lovley, D.R. Remediation and recovery of uranium from contaminated subsurface environments with electrodes. Environ. Sci. Technol. 2005, 39, 8943–8947. [Google Scholar] [CrossRef] [PubMed]
- Aulenta, F.; Fina, A.; Potalivo, M.; Petrangeli Papini, M.; Rossetti, S.; Majone, M. Anaerobic transformation of tetrachloroethane, perchloroethylene, and their mixtures by mixed-cultures enriched from contaminated soils and sediments. Water Sci. Technol. 2005, 52, 357–362. [Google Scholar] [PubMed]
- Tront, J.M.; Fortner, J.D.; Plötze, M.; Hughes, J.B.; Puzrin, A.M. Microbial fuel cell biosensor for in situ assessment of microbial activity. Biosens. Bioelectron. 2008, 24, 586–590. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Jia, W.; Hou, C.; Lei, Y. Microbial biosensors: A review. Biosens. Bioelectron. 2011, 26, 1788–1799. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R.; Ueki, T.; Zhang, T.; Malvankar, N.S.; Shrestha, P.M.; Flanagan, K.A.; Aklujkar, M.; Butler, J.E.; Giloteaux, L.; Rotaru, A.-E.; et al. Geobacter: The Microbe’s Electric Physiology, Ecology, and Practical Applications; Academic Press: London, UK, 2011. [Google Scholar]
- Speers, A.M.; Reguera, G. Electron donors supporting growth and electroactivity of Geobacter sulfurreducens anode biofilms. Appl. Environ. Microbiol. 2012, 78, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Henze, M. Biological Wastewater Treatment: Principles, Modelling and Design; IWA Publishing: London, UK, 2008. [Google Scholar]
- Lovley, D.R.; Phillips, E.J.P. Novel mode of microbial energy metabolism: Organic carbon oxidation coupled to dissimilatory reduction of iron or manganese. Appl. Environ. Microbiol. 1988, 54, 1472–1480. [Google Scholar] [PubMed]
- Esteve-Núñez, A.; Rothermich, M.; Sharma, M.; Lovley, D. Growth of Geobacter sulfurreducens under nutrient-limiting conditions in continuous culture. Environ. Microbiol. 2005, 7, 641–648. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estevez-Canales, M.; Berná, A.; Borjas, Z.; Esteve-Núñez, A. Screen-Printed Electrodes: New Tools for Developing Microbial Electrochemistry at Microscale Level. Energies 2015, 8, 13211-13221. https://doi.org/10.3390/en81112366
Estevez-Canales M, Berná A, Borjas Z, Esteve-Núñez A. Screen-Printed Electrodes: New Tools for Developing Microbial Electrochemistry at Microscale Level. Energies. 2015; 8(11):13211-13221. https://doi.org/10.3390/en81112366
Chicago/Turabian StyleEstevez-Canales, Marta, Antonio Berná, Zulema Borjas, and Abraham Esteve-Núñez. 2015. "Screen-Printed Electrodes: New Tools for Developing Microbial Electrochemistry at Microscale Level" Energies 8, no. 11: 13211-13221. https://doi.org/10.3390/en81112366
APA StyleEstevez-Canales, M., Berná, A., Borjas, Z., & Esteve-Núñez, A. (2015). Screen-Printed Electrodes: New Tools for Developing Microbial Electrochemistry at Microscale Level. Energies, 8(11), 13211-13221. https://doi.org/10.3390/en81112366







