Recent Advances in Osmotic Energy Generation via Pressure-Retarded Osmosis (PRO): A Review
Abstract
:1. Introduction
2. Theoretical Background
2.1. Characteristics of Osmosis-Driven Processes
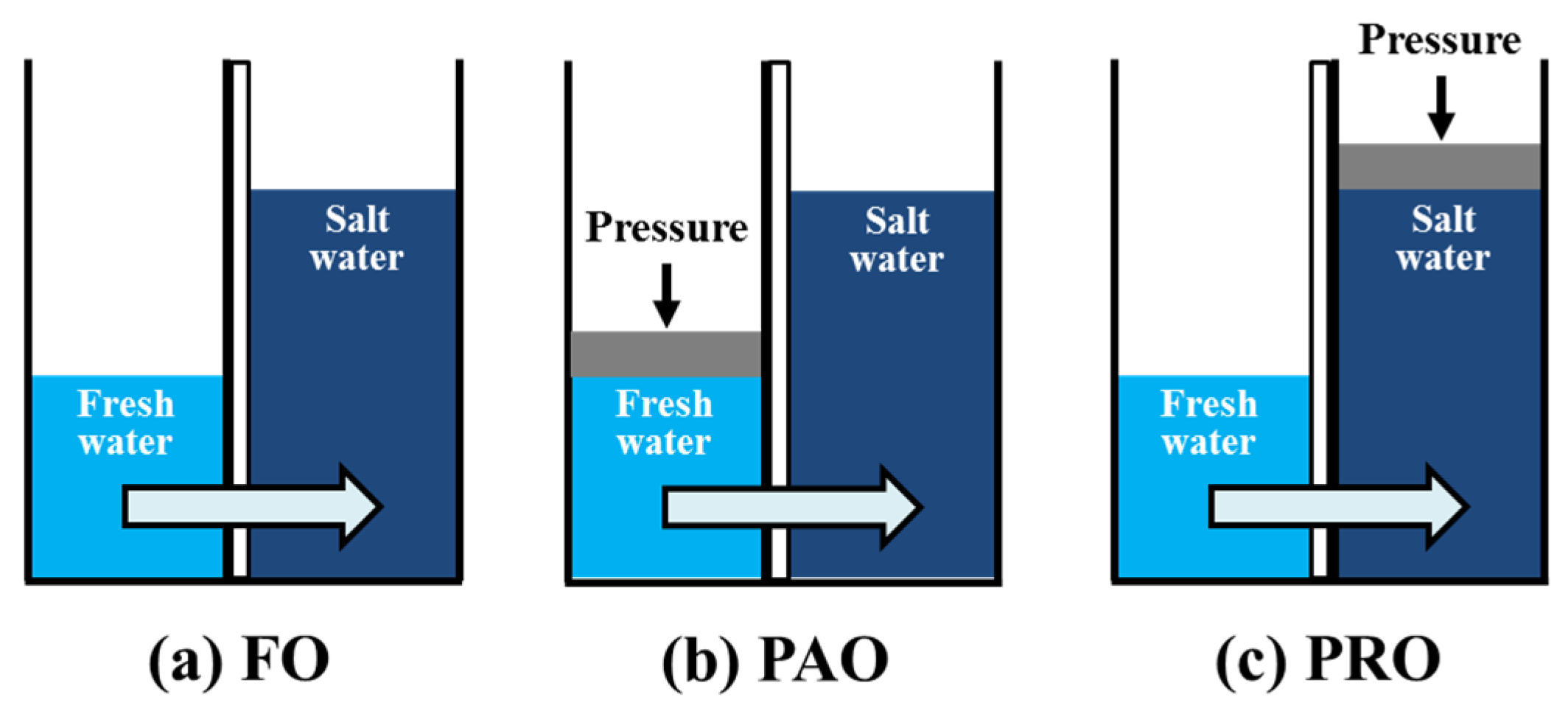
2.2. Water Flux and Power Density
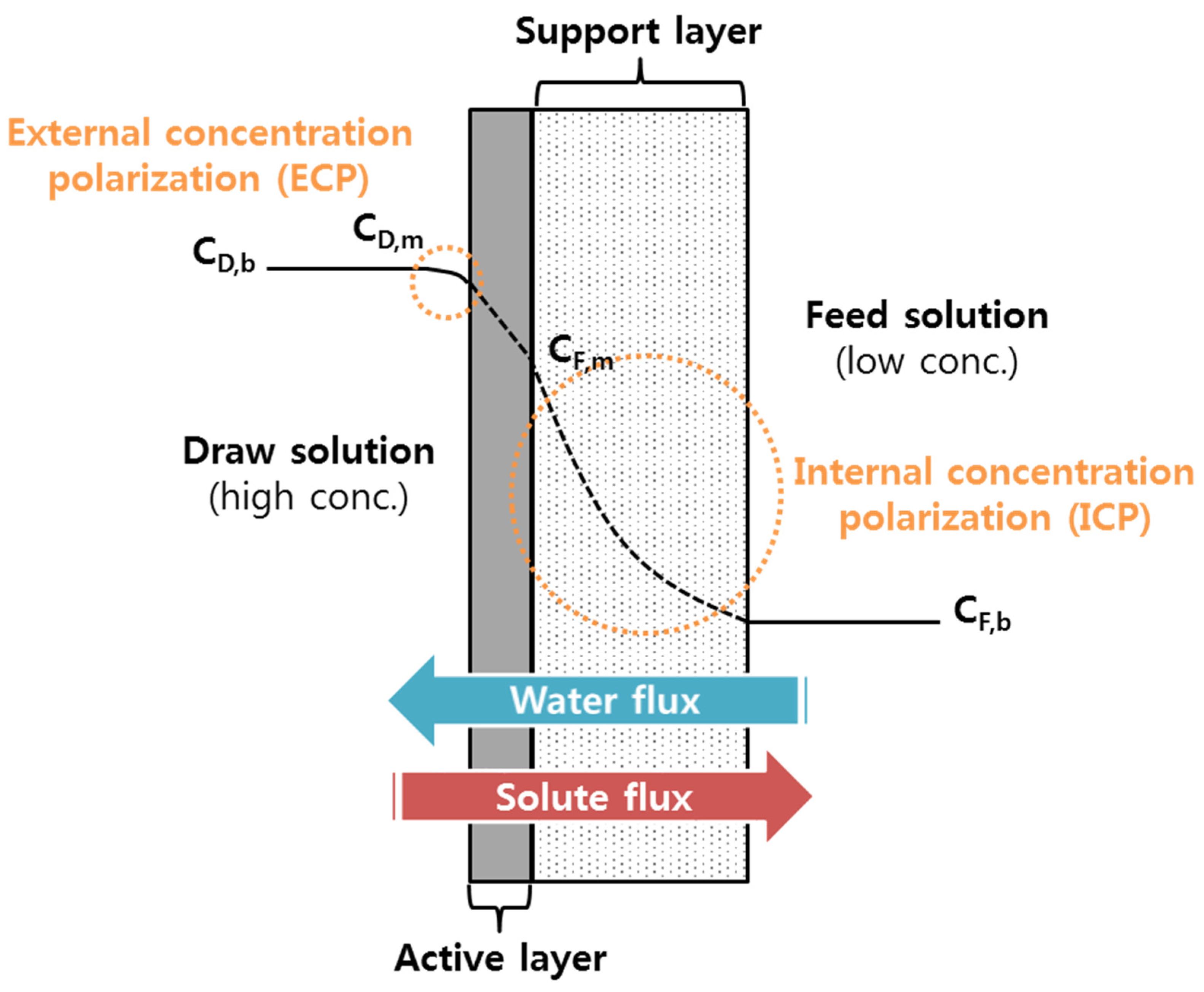
2.3. Concentration Polarizations and Reverse Solute Flux
3. Membrane Developments in PRO
3.1. Flat-Sheet Membranes
| Name | Material Active/Support | Draw Solution | Feed Solution | Pressure (Bar) | Power Density (W/m2) | Ref. |
|---|---|---|---|---|---|---|
| PA-PSf TFC | PA/PSf flat-sheet | 0.5 M NaCl | 40 mM NaCl | 12 | 10.0 | [23] |
| PA-PI TFC | PA/PI flat-sheet | 1.0 M NaCl | DI water | 15 | 12 | [24] |
| PAN-TFC | PA/PDA coated PAN flat-sheet | 3.5 wt % NaCl | DI water | 10 | 2.6 | [25] |
| TFC-PRO | Modified PA with SDS/PI flat sheet | 1.0 M NaCl | DI water | 22.0 | 18.09 | [26] |
| TNC-PRO | PA/modified PAN nanofiber | 1.06 M NaCl (Seawater brine) | 80 mM NaCl (Synthetic brackish) | 15.2 | 15.2 | [27] |
| TNC-PRO | PA/modified PAN nanofiber | 1.06 M NaCl (Seawater brine) | 0.9 mM NaCl (Synthetic river) | 15.2 | 21.3 | [27] |
| PAN-mTFC (RO-like) | PA/PAN nanofiber | 0.5 M NaCl | DI water | 10.3 | 8 | [28] |
| PAN-pTFC (NF-like) | PA/PAN nanofiber | 0.5 M NaCl | DI water | 8.6 | 6.2 | [28] |
| TFC-PRO | PA/CNTs-PEI composite nanofiber | 1.0 M NaCl | DI water | 16.9 | 17.3 | [29] |
| TFN-PRO | PA/CNTs-PES | 0.5 M NaCl | DI water | 6 | 1.65 | [31] |
3.2. Hollow-Fiber Membranes
| Name | Material Active/Support | Selective Layer | Draw Solution | Feed Solution | Pressure (Bar) | Power Density (W/m2) | Ref. |
|---|---|---|---|---|---|---|---|
| PES-TFC | PA/PES | Lumen side | 1.0 M NaCl | 10 mM NaCl | 8.4 | 11.0 | [32] |
| PES-TFC | PA/PES | Lumen side | 1.0 M NaCl | 40 mM NaCl (Waste water brine) | 9.0 | 10.6 | [32] |
| TFC-PEI | PA/PEI | Lumen side | 1.0 M NaCl | 10 mM NaCl | 15.1 | 20.9 | [33] |
| TFC-PEI | PA/PEI | Lumen side | 1.0 M NaCl | 10 mM NaCl | 15.1 | 18.7 | [33] |
| PBI-PAN | PBI-POSS/PAN | Outer layer | 1.0 M NaCl | 10 mM NaCl | 7.0 | 2.5 | [34] |
| TFC-PI | PA/PI | Outer layer | 1.0 M NaCl | DI water | 20.0 | 7.6 (=13.7 (1)) | [35] |
| TFC-PI | PA/PI | Lumen side | 1.0 M NaCl | 10 mM NaCl | 15.0 | 14.4 | [36] |
| TFC-PI | PA/PI | Lumen side | 1.0 M NaCl | 40 mM NaCl | 15.0 | 10.6 | [36] |
| TFC-PI | PA/PI | Lumen side | 1.0 M Na-Fe-Ca | DI water | 12.0 | 16.2 | [37] |
| TFC-P84 | PA/P84 | Lumen side | 1.0 M NaCl | DI water | 21.0 | 12.0 | [38] |
| PDA-TFC | PA/PDA-PES | Outer layer | 0.6 M NaCl | DI water | 7.0 | 3.0 | [39] |
| PDA-TFC | PA-TBP/PDA-PES | Outer layer | 0.6 M NaCl | DI water | 8.0 | 3.9 | [40] |
| PES-TFC | PA/PES | Lumen side | 1.0 M NaCl | DI water | 20.0 | 24.3 | [41] |
| PES-TFC | PA/PES | Outer layer | 0.6 M NaCl | DI water | 6.0 | 1.6 | [42] |
4. Applications in PRO
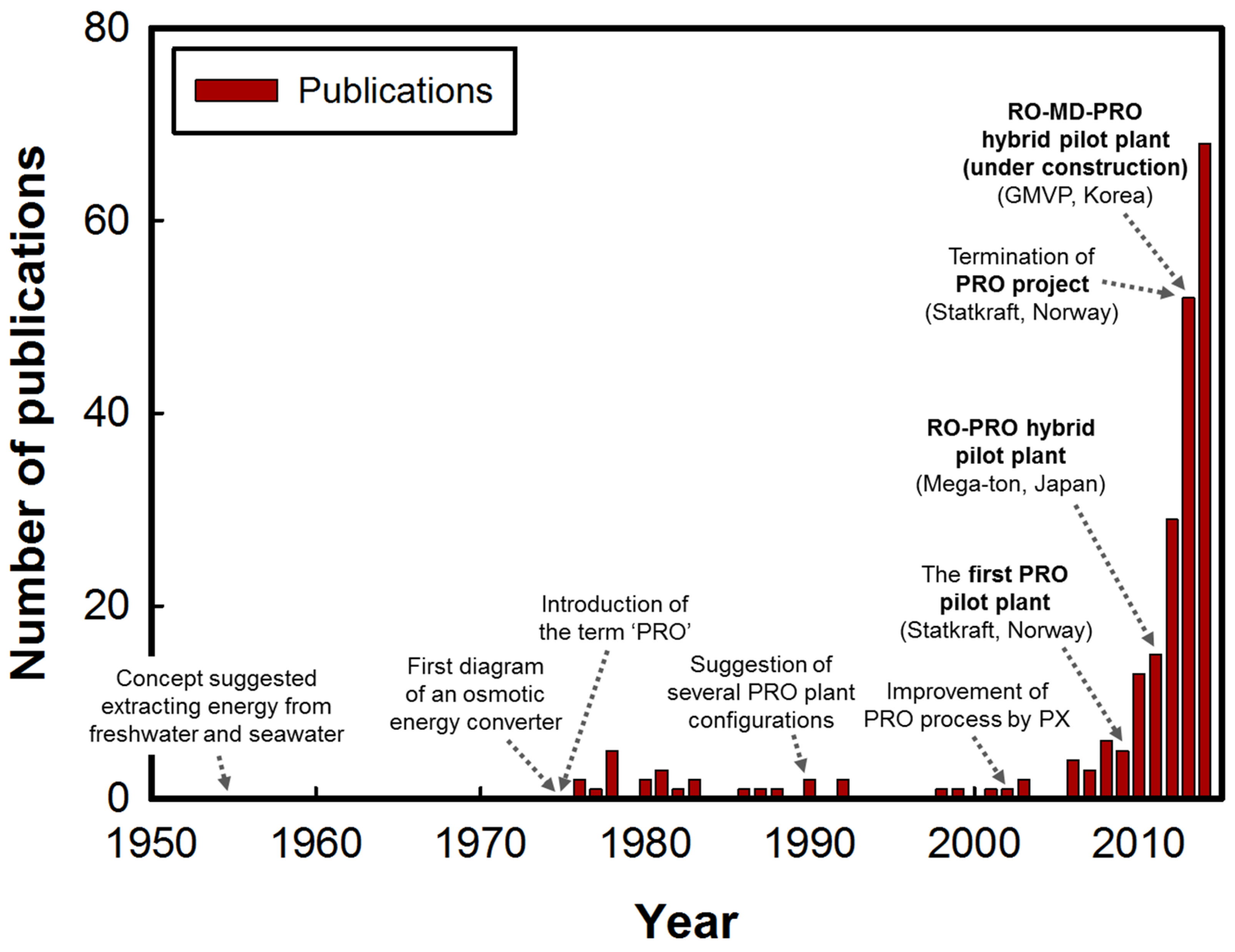
4.1. Stand-Alone PRO Processes
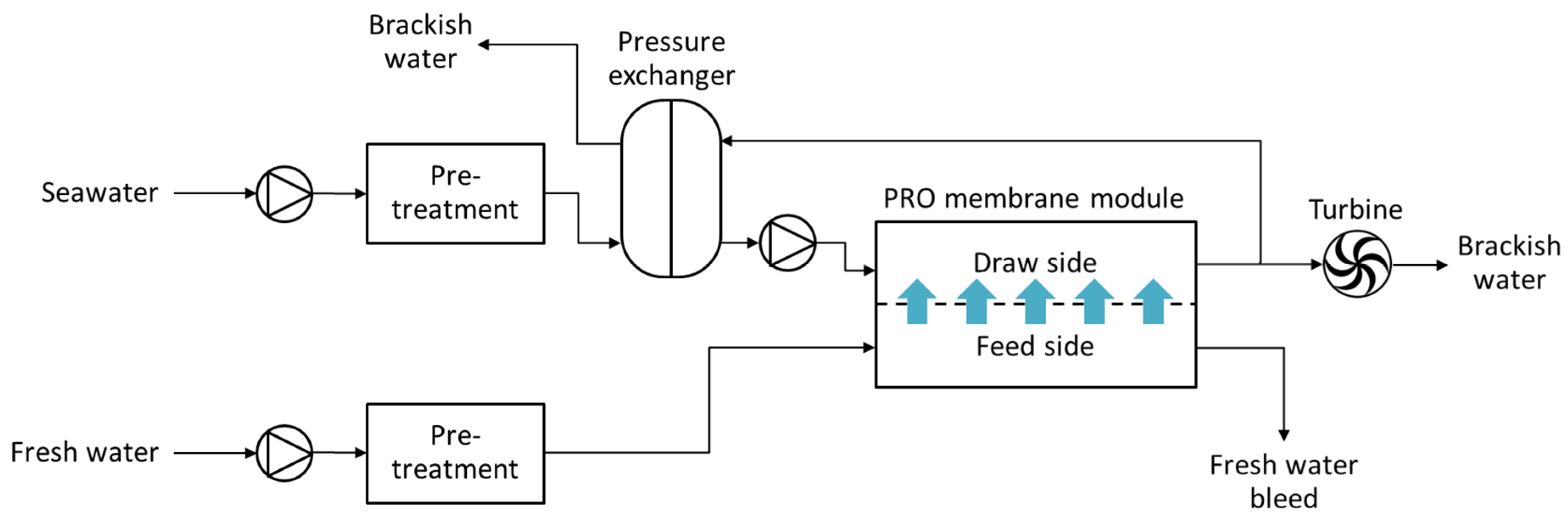
4.2. PRO-Hybrid Processes
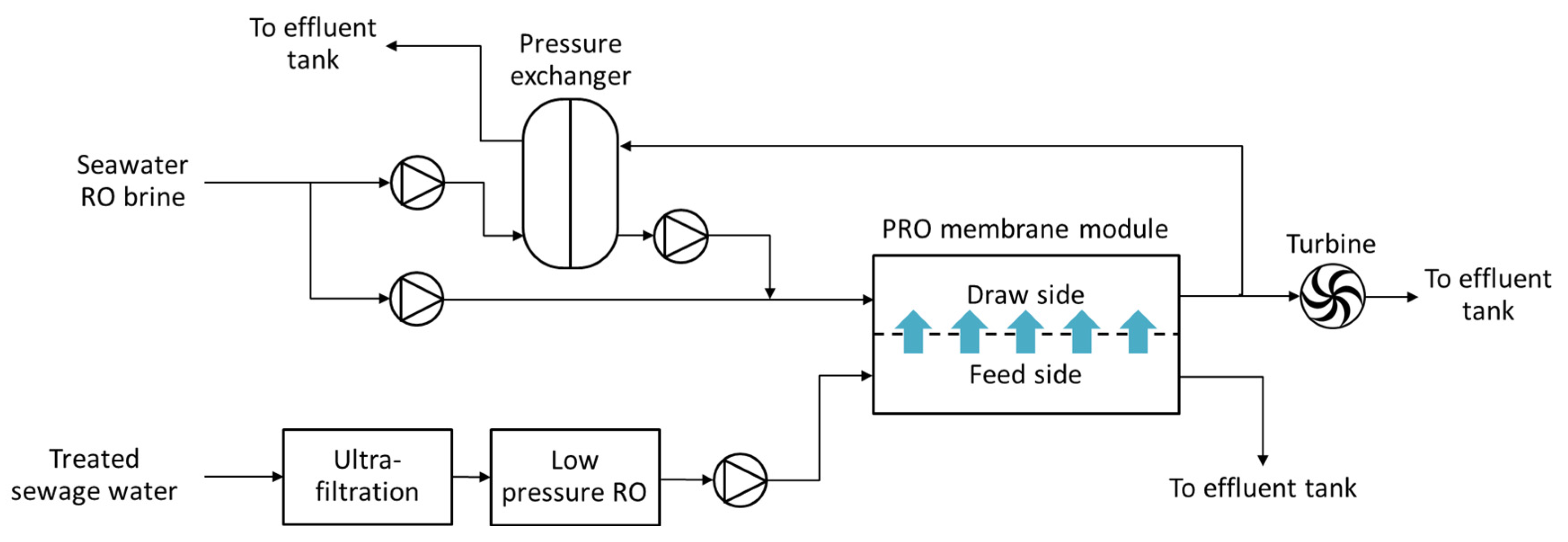
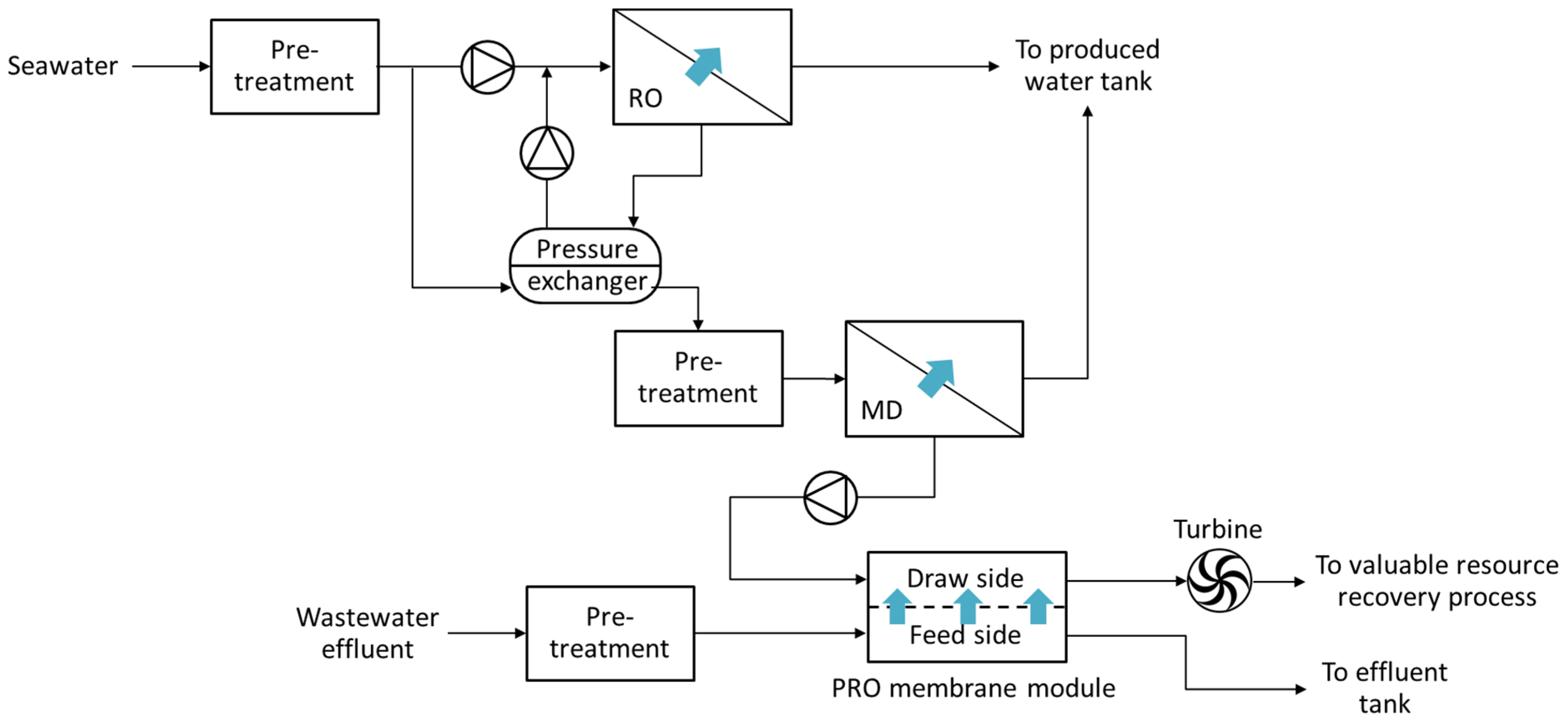
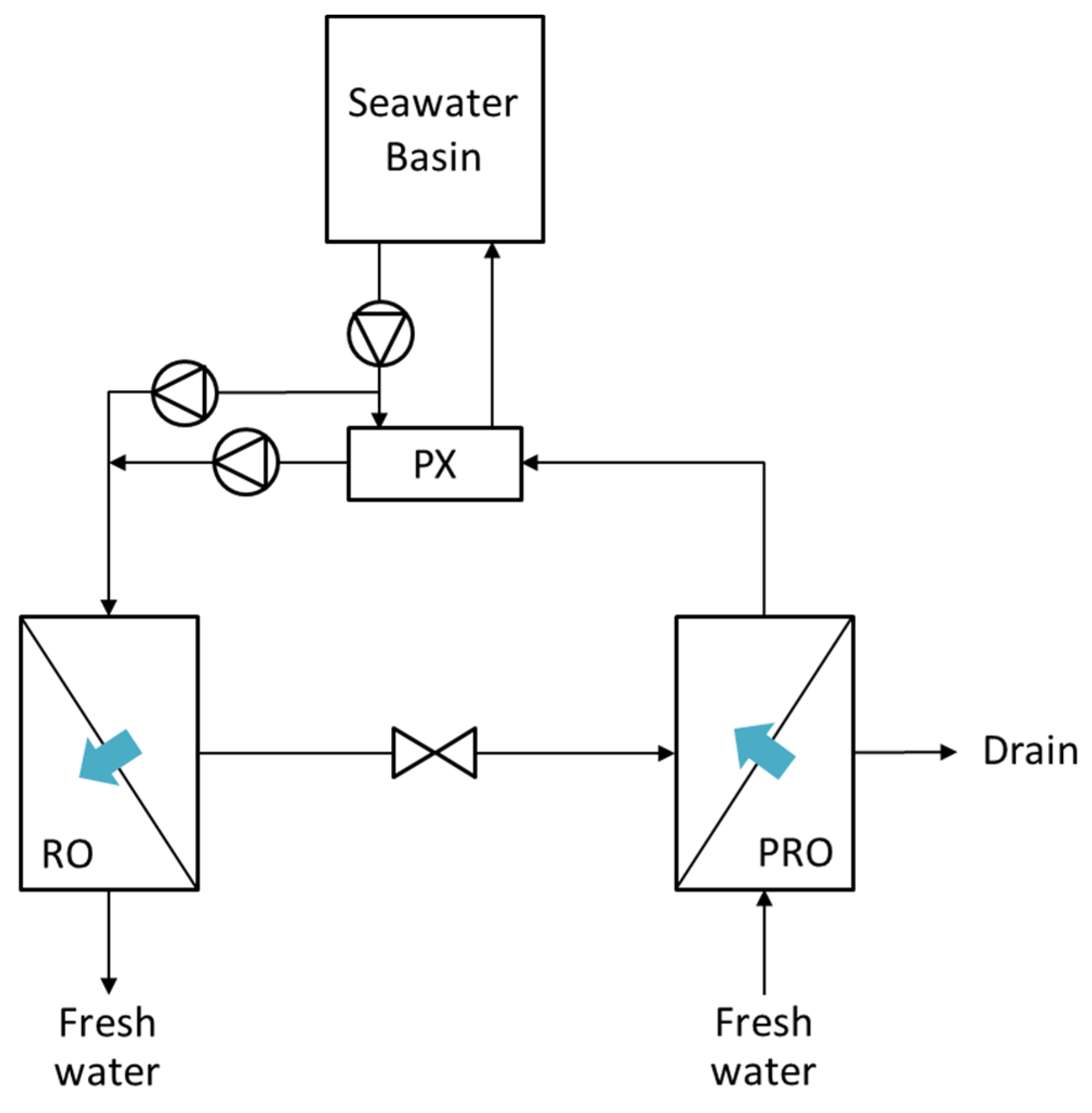
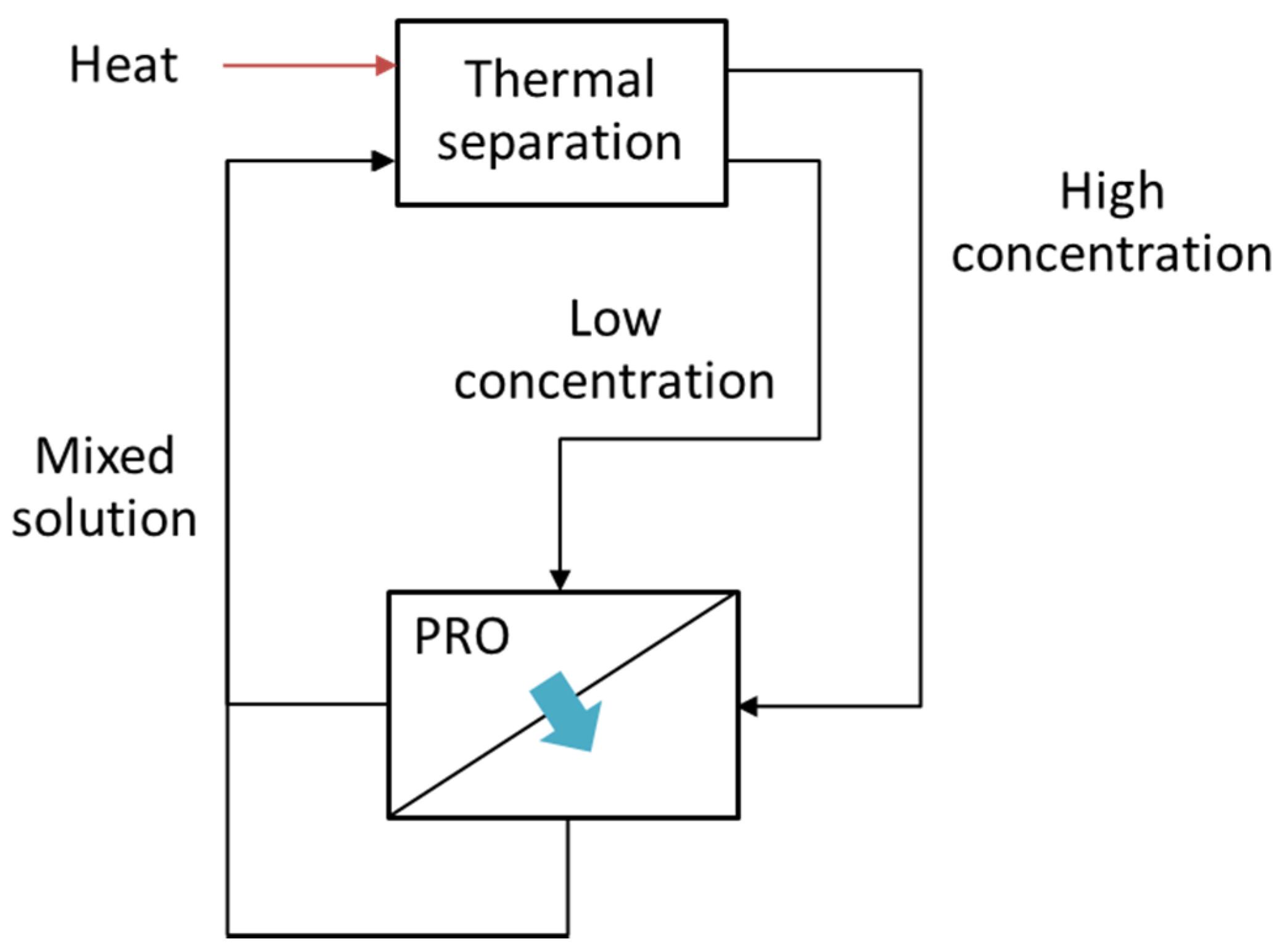
5. Numerical Studies Regarding PRO
| Author (Year) | Remarks | Ref. |
|---|---|---|
| Loeb (1976) |
| [54] |
| Lee et al. (1981) |
| [8] |
| McCutcheon and Elimelech (2006) |
| [21] |
| Yip et al. (2011) |
| [20] |
| Sivertsen et al. (2012) |
| [55] |
| Kim et al. (2013) |
| [56] |
| Naguib et al. (2015) |
| [57] |
| Author | Process Type | Remarks | Ref. |
|---|---|---|---|
| Naguib et al. | PRO |
| [57] |
| Lin et al. | PRO |
| [58] |
| Banchik et al. | PRO |
| [59] |
| Mingheng Li | PRO |
| [60] |
| Reimund et al. | PRO |
| [61] |
| He et al. | PRO (single-stage) |
| [62] |
| He et al. | PRO (dual-stage) |
| [63] |
| Altaee and Hilal | PRO (dual-stage) |
| [64,65] |
| Kim et al. | RO-PRO hybrid |
| [56] |
| He et al. | RO-PRO hybrid |
| [66] |
| Almansoori et al. | RO-PRO hybrid |
| [67] |
| Lee et al. | MD-PRO hybrid |
| [68] |
| He et al. | RO-PRO hybrid, RO-PRO-solar hybrid |
| [69] |
6. Fouling and Cleaning in PRO Processes
6.1. Membrane Fouling in PRO
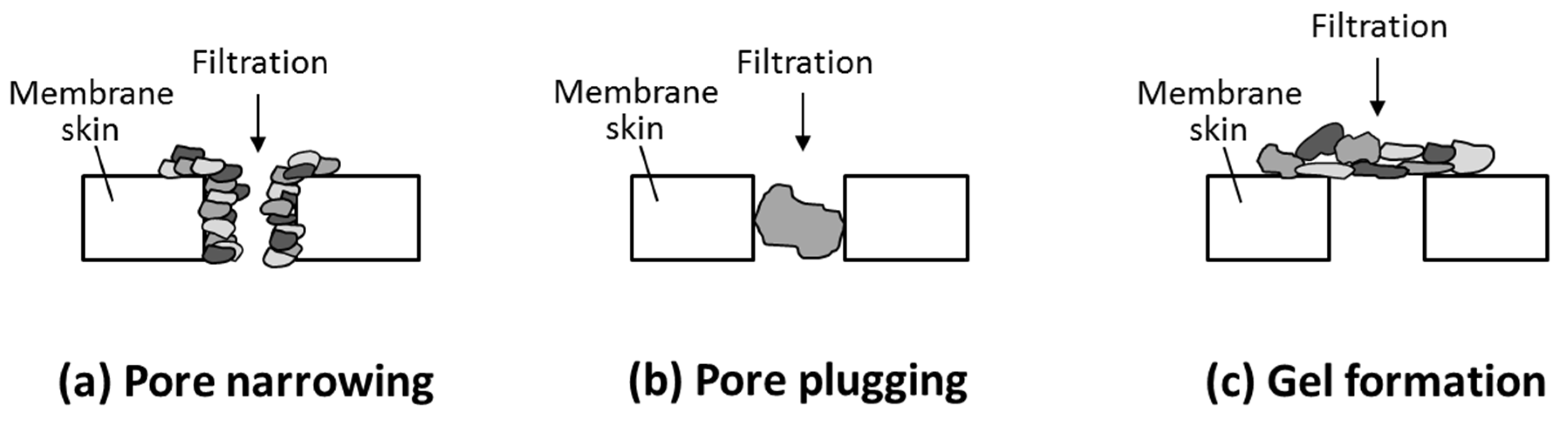
6.2. Membrane Cleaning in PRO
| Fouling | Feed solution (Concentration) | Draw Solution (Concentration) | Foulant | Membrane Type | Cleaning Method | Ref. |
|---|---|---|---|---|---|---|
| Organic | 10 mM NaCl | CaCl2/NaCl/MgCl2/Synthetic Seawater (SW)/Synthetic SW desalination brine (SWBr) | Alginate/Humic acid | CTA | - | [72] |
| Organic | Freshwater (Mostadmark water work in Norway) | Synthetic salt water (NaCl + CaCl2 + DI water) | NOM | CTA/TFC | - | [73] |
| Organic | Synthetic river water (NaCl + NaHCO3 + CaCl2 + DI water) | Synthetic seawater (NaCl solution) | SRNOM | TFC | OBW | [74] |
| Scaling | Feed solution with bulk gypsum | Synthetic solution (CaCl2/NaCl/Na2SO4) | Calcium sulfate dihydrate (gypsum) | CTA | - | [75] |
| Organic fouling | 10 mM NaCl | 1.2 M NaCl | Alginate | CTA | - | [76] |
| Organic, inorganic, combined | Synthetic wastewater | Synthetic seawater and its RO brine | Alginate, BSA, SRNOM | CTA | Physical flushing, OBW, Pressure-assisted OBW | [77] |
| Organic | Wastewater retentate from NEWater plant | SWBr | Effluent organic matter | TFC-PES hollow-fiber membrane | - | [78] |
7. Concluding Remarks
- Development of high-performance PRO membrane module. Outstanding performances in terms of power density have been already achieved by hand-casting membranes. However, to maintain the high performance from small-scale membranes to modules is considered a critical issue. In addition, further enhancement of the PRO membrane and module can be achievable for discovering the new materials or optimizing the membrane parameters [83].
- Selection of adequate pre-treatments with considerations of feed water characteristics. Frequently preferred feed solutions such as wastewater effluent and river water contain various organic and inorganic pollutants, and consequently cause severe membrane fouling. In particular, for the commercial-scale plants, appropriate pretreatments corresponding to water quality characteristics are carefully considered.
- Optimization of the process to enhance the economic feasibility. The efficient configuration of the PRO or PRO-hybrid processes should be suggested site-specific characteristics such as a plant location, plant capacity, and types of available feed and draw solutions. In addition, optimal operating conditions in terms of the hydraulic pressure, flow rate, temperature, and pH need to be further investigated in order to increase the energy generation of the designed process.
- Improvement of model-based economic analysis. Numerical studies to assess the commercial viability of the PRO process mostly have a critical limitation, i.e., excluding the cost for pre-treatments. To increase the reliability of feasibility studies, the models need to be carefully considered with the effects of all components such as pre-treatments, pumps, membrane modules, PX and hydro-turbine. Furthermore, an indicator that is focused on water and energy co-generation processes is highly required to fairly evaluate the efficiency of the PRO-hybrid processes.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mehio Sibai, A.; Nasreddine, L.; Mokdad, A.H.; Adra, N.; Tabet, M.; Hwalla, N. Nutrition transition and cardiovascular disease risk factors in middle east and north africa countries: Reviewing the evidence. Ann. Nutr. Metab. 2010, 57, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Hoffert, M.I.; Caldeira, K.; Benford, G.; Criswell, D.R.; Green, C.; Herzog, H.; Jain, A.K.; Kheshgi, H.S.; Lackner, K.S.; Lewis, J.S.; et al. Advanced technology paths to global climate stability: Energy for a greenhouse planet. Science 2002, 298, 981–987. [Google Scholar] [CrossRef] [PubMed]
- REN21. Renewables 2014: Global status report. Available online: http://www.ren21.net/ (accessed on 13 July 2015).
- La Mantia, F.; Pasta, M.; Deshazer, H.D.; Logan, B.E.; Cui, Y. Batteries for efficient energy extraction from a water salinity difference. Nano Lett. 2011, 11, 1810–1813. [Google Scholar] [CrossRef] [PubMed]
- Post, J.W.; Hamelers, H.V.; Buisman, C.J. Energy recovery from controlled mixing salt and fresh water with a reverse electrodialysis system. Environ. Sci. Technol. 2008, 42, 5785–5790. [Google Scholar] [CrossRef] [PubMed]
- Pattle, R. Production of electric power by mixing fresh and salt water in the hydroelectric pile. Nature 1954, 174, 660. [Google Scholar] [CrossRef]
- Loeb, S. Method and Apparatus for Generating Power Utilizing Pressure-Retarded-Osmosis. U.S. Patent 4193267 A, 1975. [Google Scholar]
- Lee, K.; Baker, R.; Lonsdale, H. Membranes for power generation by pressure-retarded osmosis. J. Membr. Sci. 1981, 8, 141–171. [Google Scholar] [CrossRef]
- Skilhagen, S.E.; Dugstad, J.E.; Aaberg, R.J. Osmotic power—power production based on the osmotic pressure difference between waters with varying salt gradients. Desalination 2008, 220, 476–482. [Google Scholar] [CrossRef]
- Saito, K.; Irie, M.; Zaitsu, S.; Sakai, H.; Hayashi, H.; Tanioka, A. Power generation with salinity gradient by pressure retarded osmosis using concentrated brine from swro system and treated sewage as pure water. Desalin. Water Treat. 2012, 41, 114–121. [Google Scholar] [CrossRef]
- GMVP. Md/pro Hybrid Desalination Demonstration Plant. Available online: http://www.globalmvp.org (accessed on 10 August 2015).
- Maisonneuve, J.; Pillay, P.; Laflamme, C.B. Osmotic power potential in remote regions of Quebec. Renew. Energy 2015, 81, 62–70. [Google Scholar] [CrossRef]
- Naghiloo, A.; Abbaspour, M.; Mohammadi-Ivatloo, B.; Bakhtari, K. Modeling and design of a 25 MW osmotic power plant (PRO) on bahmanshir river of Iran. Renew. Energy 2015, 78, 51–59. [Google Scholar] [CrossRef]
- Helfer, F.; Lemckert, C. The power of salinity gradients: An australian example. Renew. Sustain. Energy Rev. 2015, 50, 1–16. [Google Scholar] [CrossRef]
- Helfer, F.; Lemckert, C.; Anissimov, Y.G. Osmotic power with pressure retarded osmosis: Theory, performance and trends—A review. J. Membr. Sci. 2014, 453, 337–358. [Google Scholar] [CrossRef]
- Altaee, A.; Sharif, A. Pressure retarded osmosis: Advancement in the process applications for power generation and desalination. Desalination 2015, 356, 31–46. [Google Scholar] [CrossRef]
- Sahebi, S.; Phuntsho, S.; Kim, J.E.; Hong, S.; Shon, H.K. Pressure assisted fertiliser drawn osmosis process to enhance final dilution of the fertiliser draw solution beyond osmotic equilibrium. J. Membr. Sci. 2015, 481, 63–72. [Google Scholar] [CrossRef]
- Soltanieh, M.; Gill, W.N. Review of reverse osmosis membranes and transport models. Chem. Eng. Commun. 1981, 12, 279–363. [Google Scholar]
- Lonsdale, H.; Merten, U.; Riley, R. Transport properties of cellulose acetate osmotic membranes. J. Appl. Polym. Sci. 1965, 9, 1341–1362. [Google Scholar] [CrossRef]
- Yip, N.Y.; Elimelech, M. Performance limiting effects in power generation from salinity gradients by pressure retarded osmosis. Environ. Sci. Technol. 2011, 45, 10273–10282. [Google Scholar] [CrossRef] [PubMed]
- McCutcheon, J.R.; Elimelech, M. Influence of concentrative and dilutive internal concentration polarization on flux behavior in forward osmosis. J. Membr. Sci. 2006, 284, 237–247. [Google Scholar] [CrossRef]
- Phillip, W.A.; Yong, J.S.; Elimelech, M. Reverse draw solute permeation in forward osmosis: Modeling and experiments. Environ. Sci. Technol. 2010, 44, 5170–5176. [Google Scholar] [PubMed]
- Yip, N.Y.; Tiraferri, A.; Phillip, W.A.; Schiffman, J.D.; Hoover, L.A.; Kim, Y.C.; Elimelech, M. Thin-film composite pressure retarded osmosis membranes for sustainable power generation from salinity gradients. Environ. Sci. Technol. 2011, 45, 4360–4369. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Zhang, S.; Li, X.; Chung, T.-S. High performance thin film composite pressure retarded osmosis (PRO) membranes for renewable salinity-gradient energy generation. J. Membr. Sci. 2013, 440, 108–121. [Google Scholar] [CrossRef]
- Zhang, S.; Fu, F.; Chung, T.-S. Substrate modifications and alcohol treatment on thin film composite membranes for osmotic power. Chem. Eng. Sci. 2013, 87, 40–50. [Google Scholar] [CrossRef]
- Cui, Y.; Liu, X.-Y.; Chung, T.-S. Enhanced osmotic energy generation from salinity gradients by modifying thin film composite membranes. Chem. Eng. J. 2014, 242, 195–203. [Google Scholar] [CrossRef]
- Song, X.; Liu, Z.; Sun, D.D. Energy recovery from concentrated seawater brine by thin-film nanofiber composite pressure retarded osmosis membranes with high power density. Energy Environ. Sci. 2013, 6, 1199–1210. [Google Scholar] [CrossRef]
- Bui, N.-N.; McCutcheon, J.R. Nanofiber supported thin-film composite membrane for pressure-retarded osmosis. Environ. Sci. Technol. 2014, 48, 4129–4136. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Wang, R.; Goh, K.; Liao, Y.; Fane, A.G. Synthesis and characterization of high-performance novel thin film nanocomposite pro membranes with tiered nanofiber support reinforced by functionalized carbon nanotubes. J. Membr. Sci. 2015, 486, 151–160. [Google Scholar] [CrossRef]
- Gerstandt, K.; Peinemann, K.-V.; Skilhagen, S.E.; Thorsen, T.; Holt, T. Membrane processes in energy supply for an osmotic power plant. Desalination 2008, 224, 64–70. [Google Scholar] [CrossRef]
- Son, M.; Park, H.; Liu, L.; Choi, H.; Kim, J.H.; Choi, H. Thin-film nanocomposite membrane with cnt positioning in support layer for energy harvesting from saline water. Chem. Eng. J. 2015, 284, 68–77. [Google Scholar] [CrossRef]
- Chou, S.; Wang, R.; Shi, L.; She, Q.; Tang, C.; Fane, A.G. Thin-film composite hollow fiber membranes for pressure retarded osmosis (PRO) process with high power density. J. Membr. Sci. 2012, 389, 25–33. [Google Scholar] [CrossRef]
- Chou, S.; Wang, R.; Fane, A.G. Robust and high performance hollow fiber membranes for energy harvesting from salinity gradients by pressure retarded osmosis. J. Membr. Sci. 2013, 448, 44–54. [Google Scholar] [CrossRef]
- Fu, F.-J.; Zhang, S.; Sun, S.-P.; Wang, K.-Y.; Chung, T.-S. Poss-containing delamination-free dual-layer hollow fiber membranes for forward osmosis and osmotic power generation. J. Membr. Sci. 2013, 443, 144–155. [Google Scholar] [CrossRef]
- Sun, S.-P.; Chung, T.-S. Outer-selective pressure-retarded osmosis hollow fiber membranes from vacuum-assisted interfacial polymerization for osmotic power generation. Environ. Sci. Technol. 2013, 47, 13167–13174. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Chung, T.S. Robust and high performance pressure retarded osmosis hollow fiber membranes for osmotic power generation. AIChE J. 2014, 60, 1107–1119. [Google Scholar] [CrossRef]
- Han, G.; Ge, Q.; Chung, T.-S. Conceptual demonstration of novel closed-loop pressure retarded osmosis process for sustainable osmotic energy generation. Appl. Energy 2014, 132, 383–393. [Google Scholar] [CrossRef]
- Li, X.; Chung, T.-S. Thin-film composite P84 co-polyimide hollow fiber membranes for osmotic power generation. Appl. Energy 2014, 114, 600–610. [Google Scholar] [CrossRef]
- Ingole, P.G.; Choi, W.; Kim, K.H.; Park, C.H.; Choi, W.K.; Lee, H.K. Synthesis, characterization and surface modification of pes hollow fiber membrane support with polydopamine and thin film composite for energy generation. Chem. Eng. J. 2014, 243, 137–146. [Google Scholar] [CrossRef]
- Ingole, P.G.; Kim, K.H.; Park, C.H.; Choi, W.K.; Lee, H.K. Preparation, modification and characterization of polymeric hollow fiber membranes for pressure-retarded osmosis. RSC Adv. 2014, 4, 51430–51439. [Google Scholar] [CrossRef]
- Zhang, S.; Sukitpaneenit, P.; Chung, T.-S. Design of robust hollow fiber membranes with high power density for osmotic energy production. Chem. Eng. J. 2014, 241, 457–465. [Google Scholar] [CrossRef]
- Ingole, P.G.; Choi, W.; Kim, K.-H.; Jo, H.-D.; Choi, W.-K.; Park, J.-S.; Lee, H.-K. Preparation, characterization and performance evaluations of thin film composite hollow fiber membrane for energy generation. Desalination 2014, 345, 136–145. [Google Scholar] [CrossRef]
- Loeb, S. Large-scale power production by pressure-retarded osmosis, using river water and sea water passing through spiral modules. Desalination 2002, 143, 115–122. [Google Scholar] [CrossRef]
- Neumann, F. Report of the Meeting on Salinity Gradient Power Generation; Institute for Infrastructure, Environment and Innovation (IMI): Brussels, Belgium, 2012; pp. 1–61. [Google Scholar]
- Statkraft. Available online: http://www.statkraft.com (accessed on 20 November 2012).
- Kim, J.; Lee, J.; Kim, J.H. Overview of pressure-retarded osmosis (PRO) process and hybrid application to sea water reverse osmosis process. Desalin. Water Treat. 2012, 43, 193–200. [Google Scholar] [CrossRef]
- Kurihara, M.; Hanakawa, M. Mega-ton water system: Japanese national research and development project on seawater desalination and wastewater reclamation. Desalination 2013, 308, 131–137. [Google Scholar] [CrossRef]
- Achilli, A.; Prante, J.L.; Hancock, N.T.; Maxwell, E.B.; Childress, A.E. Experimental results from RO-PRO: A next generation system for low-energy desalination. Environ. Sci. Technol. 2014, 48, 6437–6443. [Google Scholar] [CrossRef] [PubMed]
- Sarp, S.; In-Ho, Y.; Park, Y.G. Membrane based desalination apparatus with osmotic energy recovery and membrane based desalination method with osmotic energy recovery. Patent WO 2014129724 A1, 2013. [Google Scholar]
- Prante, J.L.; Ruskowitz, J.A.; Childress, A.E.; Achilli, A. RO-PRO desalination: An integrated low-energy approach to seawater desalination. Appl. Energy 2014, 120, 104–114. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; McGinnis, R.L.; Elimelech, M. A novel ammonia—Carbon dioxide forward (direct) osmosis desalination process. Desalination 2005, 174, 1–11. [Google Scholar] [CrossRef]
- Logan, B.E.; Elimelech, M. Membrane-based processes for sustainable power generation using water. Nature 2012, 488, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Shaulsky, E.; Boo, C.; Lin, S.; Elimelech, M. Membrane-based osmotic heat engine with organic solvent for enhanced power generation from low-grade heat. Environ. Sci. Technol. 2015, 49, 5820–5827. [Google Scholar] [CrossRef] [PubMed]
- Loeb, S. Production of energy from concentrated brines by pressure-retarded osmosis: I. Preliminary technical and economic correlations. J. Membr. Sci. 1976, 1, 49–63. [Google Scholar] [CrossRef]
- Sivertsen, E.; Holt, T.; Thelin, W.; Brekke, G. Modelling mass transport in hollow fibre membranes used for pressure retarded osmosis. J. Membr. Sci. 2012, 417, 69–79. [Google Scholar] [CrossRef]
- Kim, J.; Park, M.; Snyder, S.A.; Kim, J.H. Reverse osmosis (RO) and pressure retarded osmosis (PRO) hybrid processes: Model-based scenario study. Desalination 2013, 322, 121–130. [Google Scholar] [CrossRef]
- Naguib, M.F.; Maisonneuve, J.; Laflamme, C.B.; Pillay, P. Modeling pressure-retarded osmotic power in commercial length membranes. Renew. Energy 2015, 76, 619–627. [Google Scholar] [CrossRef]
- Lin, S.; Straub, A.P.; Elimelech, M. Thermodynamic limits of extractable energy by pressure retarded osmosis. Energy Environ. Sci. 2014, 7, 2706–2714. [Google Scholar] [CrossRef]
- Banchik, L.D.; Sharqawy, M.H.; Lienhard, J.H. Limits of power production due to finite membrane area in pressure retarded osmosis. J. Membr. Sci. 2014, 468, 81–89. [Google Scholar] [CrossRef]
- Li, M. Analysis and optimization of pressure retarded osmosis for power generation. AIChE J. 2015, 61, 1233–1241. [Google Scholar] [CrossRef]
- Reimund, K.K.; McCutcheon, J.R.; Wilson, A.D. Thermodynamic analysis of energy density in pressure retarded osmosis: The impact of solution volumes and costs. J. Membr. Sci. 2015, 487, 240–248. [Google Scholar]
- He, W.; Wang, Y.; Shaheed, M.H. Energy and thermodynamic analysis of power generation using a natural salinity gradient based pressure retarded osmosis process. Desalination 2014, 350, 86–94. [Google Scholar] [CrossRef]
- He, W.; Wang, Y.; Shaheed, M.H. Enhanced energy generation and membrane performance by two-stage pressure retarded osmosis (PRO). Desalination 2015, 359, 186–199. [Google Scholar] [CrossRef]
- Altaee, A.; Sharif, A.; Zaragoza, G.; Hilal, N. Dual stage pro process for power generation from different feed resources. Desalination 2014, 352, 118–127. [Google Scholar] [CrossRef]
- Altaee, A.; Hilal, N. Design optimization of high performance dual stage pressure retarded osmosis. Desalination 2015, 355, 217–224. [Google Scholar] [CrossRef]
- He, W.; Wang, Y.; Sharif, A.; Shaheed, M.H. Thermodynamic analysis of a stand-alone reverse osmosis desalination system powered by pressure retarded osmosis. Desalination 2014, 352, 27–37. [Google Scholar] [CrossRef]
- Almansoori, A.; Saif, Y. Structural optimization of osmosis processes for water and power production in desalination applications. Desalination 2014, 344, 12–27. [Google Scholar] [CrossRef]
- Lee, J.-G.; Kim, Y.-D.; Shim, S.-M.; Im, B.-G.; Kim, W.-S. Numerical study of a hybrid multi-stage vacuum membrane distillation and pressure-retarded osmosis system. Desalination 2015, 363, 82–91. [Google Scholar] [CrossRef]
- He, W.; Wang, Y.; Shaheed, M.H. Stand-alone seawater RO (reverse osmosis) desalination powered by PV (photovoltaic) and PRO (pressure retarded osmosis). Energy 2015, 86, 423–435. [Google Scholar] [CrossRef]
- Thorsen, T.; Holt, T. The potential for power production from salinity gradients by pressure retarded osmosis. J. Membr. Sci. 2009, 335, 103–110. [Google Scholar] [CrossRef]
- Chung, T.-S.; Zhang, S.; Wang, K.Y.; Su, J.; Ling, M.M. Forward osmosis processes: Yesterday, today and tomorrow. Desalination 2012, 287, 78–81. [Google Scholar] [CrossRef]
- She, Q.; Wong, Y.K.W.; Zhao, S.; Tang, C.Y. Organic fouling in pressure retarded osmosis: Experiments, mechanisms and implications. J. Membr. Sci. 2013, 428, 181–189. [Google Scholar] [CrossRef]
- Thelin, W.R.; Sivertsen, E.; Holt, T.; Brekke, G. Natural organic matter fouling in pressure retarded osmosis. J. Membr. Sci. 2013, 438, 46–56. [Google Scholar] [CrossRef]
- Yip, N.Y.; Elimelech, M. Influence of natural organic matter fouling and osmotic backwash on pressure retarded osmosis energy production from natural salinity gradients. Environ. Sci. Technol. 2013, 47, 12607–12616. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Hou, D.; She, Q.; Tang, C.Y. Gypsum scaling in pressure retarded osmosis: Experiments, mechanisms and implications. Water Res. 2014, 48, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.; Kim, S.-H.; Kim, J.H. Impact of hydraulic pressure and pH on organic fouling in pressure retarded osmosis (PRO) process. Desalin. Water Treat. 2015. [Google Scholar] [CrossRef]
- Kim, D.I.; Kim, J.; Shon, H.K.; Hong, S. Pressure retarded osmosis (PRO) for integrating seawater desalination and wastewater reclamation: Energy consumption and fouling. J. Membr. Sci. 2015, 483, 34–41. [Google Scholar] [CrossRef]
- Wan, C.F.; Chung, T.-S. Osmotic power generation by pressure retarded osmosis using seawater brine as the draw solution and wastewater retentate as the feed. J. Membr. Sci. 2015, 479, 148–158. [Google Scholar] [CrossRef]
- Kimura, K.; Hane, Y.; Watanabe, Y.; Amy, G.; Ohkuma, N. Irreversible membrane fouling during ultrafiltration of surface water. Water Res. 2004, 38, 3431–3441. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, H.; Kimura, K.; Watanabe, Y. Mechanism involved in the evolution of physically irreversible fouling in microfiltration and ultrafiltration membranes used for drinking water treatment. Environ. Sci. Technol. 2007, 41, 6789–6794. [Google Scholar] [CrossRef] [PubMed]
- Boo, C.; Elimelech, M.; Hong, S. Fouling control in a forward osmosis process integrating seawater desalination and wastewater reclamation. J. Membr. Sci. 2013, 444, 148–156. [Google Scholar] [CrossRef]
- Qin, J.-J.; Liberman, B.; Kekre, K.A. Direct osmosis for reverse osmosis fouling control: Principles, applications and recent developments. Open Chem. Eng. J. 2009, 3, 8–16. [Google Scholar] [CrossRef]
- Sivertsen, E.; Holt, T.; Thelin, W.R.; Brekke, G. Iso-watt diagrams for evaluation of membrane performance in pressure retarded osmosis. J. Membr. Sci. 2015, 489, 299–307. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.; Jeong, K.; Park, M.J.; Shon, H.K.; Kim, J.H. Recent Advances in Osmotic Energy Generation via Pressure-Retarded Osmosis (PRO): A Review. Energies 2015, 8, 11821-11845. https://doi.org/10.3390/en81011821
Kim J, Jeong K, Park MJ, Shon HK, Kim JH. Recent Advances in Osmotic Energy Generation via Pressure-Retarded Osmosis (PRO): A Review. Energies. 2015; 8(10):11821-11845. https://doi.org/10.3390/en81011821
Chicago/Turabian StyleKim, Jihye, Kwanho Jeong, Myoung Jun Park, Ho Kyong Shon, and Joon Ha Kim. 2015. "Recent Advances in Osmotic Energy Generation via Pressure-Retarded Osmosis (PRO): A Review" Energies 8, no. 10: 11821-11845. https://doi.org/10.3390/en81011821
APA StyleKim, J., Jeong, K., Park, M. J., Shon, H. K., & Kim, J. H. (2015). Recent Advances in Osmotic Energy Generation via Pressure-Retarded Osmosis (PRO): A Review. Energies, 8(10), 11821-11845. https://doi.org/10.3390/en81011821








