Biofuels from the Fresh Water Microalgae Chlorella vulgaris (FWM-CV) for Diesel Engines
Abstract
: This work aims to investigate biofuels for diesel engines produced on a lab-scale from the fresh water microalgae Chlorella vulgaris (FWM-CV). The impact of growing conditions on the properties of biodiesel produced from FWM-CV was evaluated. The properties of FWM-CV biodiesel were found to be within the ASTM standards for biodiesel. Due to the limited amount of biodiesel produced on the lab-scale, the biomass of dry cells of FWM-CV was used to yield emulsified water fuel. The preparation of emulsion fuel with and without FWM-CV cells was conducted using ultrasound to overcome the problems of large size microalgae colonies and to form homogenized emulsions. The emulsified water fuels, prepared using ultrasound, were found to be stable and the size of FWM-CV colonies were effectively reduced to pass through the engine nozzle safely. Engine tests at 3670 rpm were conducted using three fuels: cottonseed biodiesel CS-B100, emulsified cottonseed biodiesel water fuel, water and emulsifier (CS-E20) and emulsified water containing FWM-CV cells CS-ME20. The results showed that the brake specific fuel consumption (BSFC) was increased by about 41% when the engine was fueled with emulsified water fuels compared to CS-B100. The engine power, exhaust gas temperature, NOx and CO2 were significantly lower than that produced by CS-B100. The CS-ME20 produced higher power than CS-E20 due to the heating value improvement as a result of adding FWM-CV cells to the fuel.1. Introduction
The use of fossil fuels in various industries, such as transportation, has been reported to be one of the main factors causing high CO2 levels in the atmosphere which is resulting in global warming. Research into the use of biodiesel in Compression Ignition (CI) engines has recently increased due to the depletion of fossil fuels, high fuel prices and pollution problems associated with exhaust gas emissions. Developing alternative fuels is an essential step towards solving the problems associated with fossil fuels. It is important that alternative fuels be renewable, have less impact on the environment and be produced and used by currently available technology. The natural biodiesel resources such as oil crops and waste cooking oil are not sufficient to cover the global transportation fuel demand [1]. Therefore, exploring other potential sources for alternative fuels is a necessity [2]. Microalgae are regarded as a promising alternative fuel for IC engines [3,4].
Microalgae are unicellular photosynthetic organisms that use light energy and carbon dioxide (CO2), with relatively higher photosynthetic efficiency [5]. Chlorella vulgaris is one of the most attractive algae species for producing biofuels owing to its fast growth and easy cultivation. However, it is yet to be commercially viable due to its low lipid content [6]. Therefore, increasing the lipid content in this species is an important research area that needs to be addressed. The dual requirements of maximizing biomass and lipid production are difficult to achieve. Widjaja et al. [7] reported that various research claims that lipid storage in many microalgae was enhanced under environmental stress. Increasing the lipid content under stress conditions could affect the biomass productivity. The productivity of biomass and the productivity of lipid content of C. vulgaris can both be enhanced if specific culture conditions are applied [8]. The lipid content in C. vulgaris can be increased by up to 56.6% of the dry biomass weight, by adding 1.2 × 10−5 mol·L−1 FeCl3 [6]. The lipid content of C. vulgaris is significantly affected by the variation in the growing conditions. For example, Converti et al. [9] stated that the lipid content of microalgae decreased from 14.71% to 5.90% when the growing temperature increased from 25 to 30 °C.
One method of harnessing microalgae is by producing biodiesel from its oil. Biodiesel fuel gives a comparable engine performance and emission to petroleum diesel [10]. Biodiesel from microalgae oil has received significant attention recently as it is renewable, environmentally friendly and represents the ability to convert CO2 to oil [11]. Microalgae oil contains high values of palmitic acid, and the concentration of linoleic acid met the requirements of the European legislation for biodiesel [9]. Microalgae biofuels are non-toxic, highly bio-degradable, contain no sulphur and the leftover materials (after extracting the oil) can be used for ethanol production or as soil fertilizer [12]. Microalgae have high biomass and high lipid productivities per unit of area in comparison with other crops [13,14]. Chisti [1] and Ayhan [15] reported that the demand for fuel in the transportation industry can only be covered by microalgae as a renewable source. It was reported that microalgae can produce the same amount of biodiesel (for 30% w/w oil content) compared to rapeseed or soybean crops using around 49- to 132-times less land [16]. Furthermore, microalgae are non-edible and can grow under various conditions in which there is no significant impact on the human food supply chain [7,17–19].
The properties of biodiesel depend on its fatty acids (FA) composition [20]. The biodiesel fuel properties are the outcome of its individual fatty ester's properties and structure such as chain length, degree of unsaturation and branching of the chain. Those parameters of the fatty acid esters influence cetane number, heat of combustion, cold flow viscosity and exhaust emissions [21]. Ramírez-Verduzco, et al. [22] estimated density, viscosity, cetane number, and the higher heating value for tallow and soybean biodiesel using a developed empirical equation. They found that the increase in the number of double bonds in the fatty acid methyl esters (FAMEs) causes a reduction in the values of cetane numbers, viscosity and the higher heating values.
The other way of utilizing microalgae or its constituents is by producing emulsion fuels. Emulsion fuels is a term usually used to describe mixtures of diesel and/or biodiesel with water [23]. Owing to the differences in the physical and chemical properties of the mixture components (i.e., water, diesel or biodiesel), emulsifiers are normally used to facilitate the interaction between the mixture components and prolong the stability of the emulsion. According to Fayyad, et al. [24], emulsion fuels were first brought to attention in 1931 by Joseph Vance, and are still not very well known and accepted by consumers. Emulsified water with microalgae in diesel and biodiesel is regarded as a potential fuel in terms of renewability, emission reduction, economy and it is adaptable to the current technology. However, there are some problems associated with the direct use of microalgae slurry-biodiesel emulsion in diesel engine represented by:
The presence of large sized microalgae aggregates which causes injection blockage or damage.
High viscosity which increases the resistance to fuel flow.
The low stability of the emulsion that results in the settlement of the heavy phase of the emulsion in fuel passages.
This study has been designed to address the issues associated with the use of microalgae (using C. vulgaris as a model) for fuel production. The effect of various growing conditions on biodiesel properties produced from FWM-CV will be investigated. The use of ultrasound for breaking down microalgae colonies for the purpose of preventing injector blockage and producing homogenous stable emulsions will be addressed. Finally, the impact on diesel engine performance and exhaust gas emissions of adding FWM-CV cells to the emulsified water fuel will be studied.
2. Materials and Methods
2.1. Microalgae Strain and Medium
A culture of fresh water microalgae C. vulgaris FWM-CV (CCAP 211/11) was obtained from the Commonwealth Scientific and Industrial Research Organization (CSIRO), Centre for Marine and Atmospheric Research (C.M.A.R) in Hobart, Tasmania.
MBL Medium Woods-Hole [25] adapted for freshwater algae was used. The stock solutions were prepared from the chemicals presented in the Table 1 and Milli-Q water. The stock solutions were stored in the refrigerator at 4 °C.
To prepare MBL medium, one mL of each stock solution (1–11) was added to one litre of Milli-Q water. The pH was adjusted to 7.2 using hydrochloric acid. The media was autoclaved at 121 °C (15 PSI) for 15 min. The stock solution of the iron stressor (FeCl3 per EDTA) was prepared by dissolving 0.324 g of EDTA and 0.1947 g of FeCl3 in 200 mL of Milli-Q water then autoclaved at 121 °C for 15 min. One mL of the iron stressor solution was added to each litre of media to reach the amount of 1.2 × 10−5 mol·L−1.
2.2. Culture Conditions
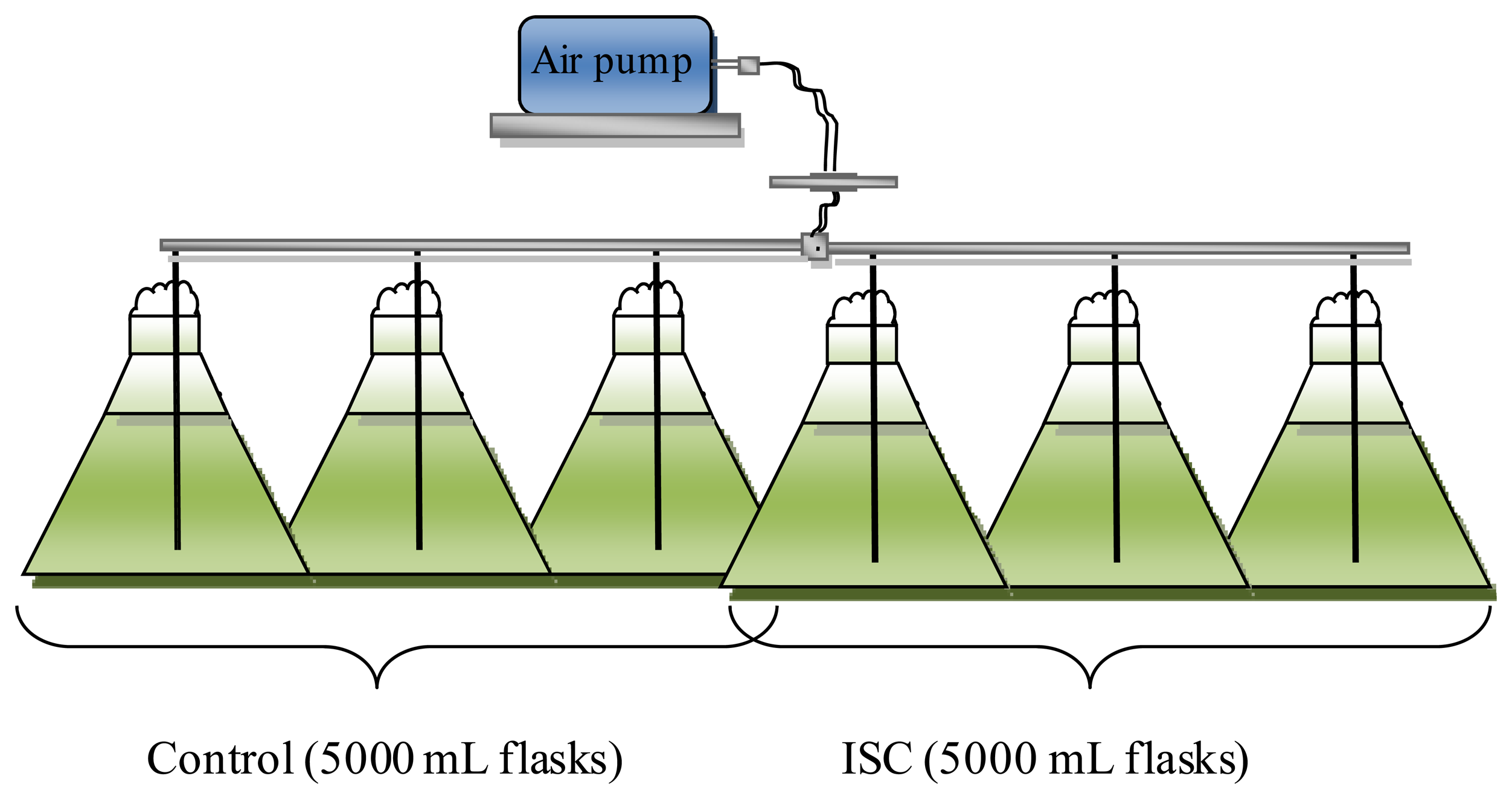
The FWM-CV was grown at the laboratories of the University of Southern Queensland (USQ) using sterilized MBL medium. The culture temperature was fixed at 22 ± 3 °C. Fluorescent light was used to supply constant light intensity for the culture which was not less than 2500 lux on a 16:8 light to dark cycle [26]. For the emulsion fuel experiment, the microalgae was grown in 5 L flasks. The cultures were supplied with air using an air pump to generate large, slow bubbles to mix the culture and increased the contact of the culture with air and the medium. The air supply system was autoclaved and a 0.2 micron filter was used to prevent culture contamination.
2.3. Growth Monitoring
The growth of the culture was measured using two different methods. The first method was using a Neubauer haemocytometer and light microscopy to measure the cell density. The second method was the use of UV/Vis spectrophotometer (JENWAY 6705, Staffordshire, UK) to monitor the growth curve by measuring the optical density (OD) of the culture at 515 nm. This wavelength was selected based on preliminary tests for measuring the maximum absorbance at wide range of wavelengths. There are also some other researchers [22,23] who used wavelengths close to 515 nm (i.e., 505 nm [27], 525 nm [28]) for measuring the growth of C. vulgaris. The OD is a simple and efficient method for measuring the growth curve. A linear regression equation was derived to describe the relationship between the optical density and the cell density.
2.4. Microalgae Harvesting
The microalgae cells were harvested using a Beckman Avanti J-251 high speed centrifuge (Beckman Coulter, chaska, MN, USA) at 8000 rpm for 10 min. The samples were then transferred to pre-weighed Petri dishes. In order to determine the dry weight of the FWM-CV, the resulting biomasses were freeze dried. Lyophilised cells were stored in desiccators until the time of oil extraction.
Another linear equation was derived to describe the relationship between the optical density (OD) at 515 nm and the dry weight of microalgae (Mdw) in milligrams. Three samples of 100 mL of microalgae culture in different growth phases were harvested using the centrifuge after measuring the optical density. The samples were then freeze dried and weighed to deriving the relationship between the OD and the Mdw.
2.5. Biodiesel Production from FWM-CV
Biodiesel production and a lipid enhancement test was conducted to study the effect of adding iron to the culture medium at the late exponential growth phase on microalgae lipid content and the FAME components [6]. The culture was divided into two parts each part consists of three samples as replications (Figure 1). In the first part, the samples were kept as a control for 75.5 days. While in the second part, a new medium was added to the culture in the ratio of 3:1 (v/v) on day 35, and after three days, 1.2 × 10−5 FeCl3/EDTA per liter was added to the culture. This sample is referred to as Iron Supplemented Culture (ISC).
2.6. Lipid Extraction
The Folch [29] method for lipid extraction was used in this work. Each sample of dried microalgae was mixed with a solution of (2:1) chloroform (analytical grade, BDH Chemicals, Victoria, Australia) and methanol (analytical grade, Ajax Chemicals, Victoria, Australia). Widjaja et al. [7] has used chloroform and methanol (2:1, v/v) to extract the oil from microalgae C. vulgaris. The solution volume was 20 times greater than the volume of biomass in order to obtain an adequate volume to continue the experimental analysis. The mixture was then shaken for 20 min using a Bioline BL4600 orbital shaker (Maharashtra, India) at 150 rpm. The solution containing chloroform-methanol and lipid was separated from the biomass by gravity filtration using Machery-Nagel 615MN filter papers (Düren, Germany). This solution was collected in centrifuge tubes and then the lipid was rinsed in the test tube by chloroform to minimize lipid losses. These tubes were re-filled by volume of Milli-Q water equal to 20% of the total volume of the sample. To separate the chloroform-methanol phase, the samples were centrifuged at 3000 rpm for 10 min. The upper phase was discarded by siphoning using a Pasture pipette and the lower phase containing lipids was evaporated under vacuum in a rotary evaporator. The samples were dried and left at the room temperature for one day for further drying, then weighed to determine the lipid content.
2.7. Transesterification
The oil extracted from FWM-CV was converted to biodiesel using transesterification. The transesterification was conducted by heating the lipid to 48 °C. At the same time 0.45 g of NaOH was added to 11 mL of methanol and shaken. Due to low lipid weight, the amount of this mixture was increased to be around 20 times greater than the lipid weight and then added to the lipid samples for 40 min. After 10 h, the oil phase was separated to another flask and centrifuged to remove the glycerin.
2.8. Fatty Acid Methyl Esters (FAMEs) Analysis and Physical Properties
The FAMEs and their percentage in FWM-CV biodiesel for the control and the ISC were identified using gas chromatography-mass spectrometry (Shimadzu GC-2010, Brisbane, Australia). The density, cetane number, kinematic viscosity and the heating value of biodiesels from the control and ISC were calculated from their FAMEs and their percentage.
2.9. Statistical Analysis
The Analysis of Variance one way ANOVA was performed using SPSS (Version 19, IBM, Armonk, NY, USA) to study the significance of the change in microalgae biomass and lipid productivity. The parameters are microalgae dry weight g·L−1, biomass productivity mg·L−1·day−1, lipid content % and lipid productivity mg·L−1·day−1 for the control ISC samples. Choix, et al. [30] used ANOVA to analyse the difference in the variable of volumetric productivity and growth rate of microalgae.
2.10. Growing of FWM-CV for the Emulsified Water Fuel Experiment
Fresh culture of FWM-CV was grown using sterilised 15 L water bottles (see Figure 2) to be used for water emulsified fuel test. Each bottle was filled with 8 L of medium and one g·L−1 of glucose was added to enhance growth.
2.11. FWM-CV Emulsion Fuel
In order to study the properties of the emulsified water in cottonseed biodiesel and the effect of using FWM-CV as an additive, three fuels were prepared. The fuels were: cottonseed biodiesel 100% (CS-B100), emulsified water 20% in cottonseed biodiesel (CS-E20) and emulsified water 20% containing FWM-CV in cottonseed biodiesel (CS-ME20). The cottonseed biodiesel CS-B100 was obtained from the Queensland University of Technology (QUT).
The CS-E20 and CS-ME20 fuel was prepared at the engine lab of the University of Southern Queensland (USQ). An ultrasonic horn reactor (Dr. Hielscher, Model UIP 500, Germany) with variable input power and a fixed frequency of 55 kHz was used for the CS-E20 and CS-ME20 fuel preparation (Figure 3). The emulsion fuels were prepared following the same procedure for both CS-E20 and CS-ME20, except that the water in CS-ME20 is supplemented with FWM-CV. The fuel was prepared based on a volumetric ratio of 79.2% of CS-B100, 19.8% water and 1% emulsifier.
Based on observations of the FWM-CV during the growing process and pre-testing of fuel preparation, the issue with the size of the microalgae colonies was detected. To overcome this, the emulsion preparation was performed in two stages; (a) breaking down the aggregates of FWM-CV and (b) mixing the pre-sonicated FWM-CV in water with CS-B100 and surfactant using ultrasound.
2.11.1. Disintegration of FWM-CV Aggregates by Sonication
Ultrasound is an effective method for de-agglomeration of microalgae clusters [31], it has therefore been adopted in this study for the purpose of breaking down the aggregates of FWM-CV prior to mixing the slurry with biodiesel and surfactant. 200 mL of FWM-CV slurry was treated with ultrasound for 5 min. The ultrasonic power intensity applied in this experiment was 21.52 W·cm−2. The temperature of the microalgae slurry during sonication treatment was monitored using A DiGi-Sense type K thermocouple (Coleparmer Chatswood, Australia). The maximum temperature reached in this experiment was 62 °C. Such a high temperature together with the mechanical effects of ultrasound forms a valuable tool for rupturing cells [32] and releases the lipid content into the slurry. The release of the lipids into the slurry may make the final emulsified water more combustible.
The efficiency of ultrasound in breaking down FWM-CV aggregates was evaluated by comparing the microscopic photos of the untreated and treated microalgae colonies in water. FWM-CV aggregates and cells in the untreated and treated slurries were visualized using a Motic Stereo-microscope (AIS Australian Instrument Services, Bayswater, Australia). Photos of the microalgae cells and aggregates were captured using a CC12 camera which was attached to the microscope and the images were analysed using the AnalySIS software (AIS Australian Instrument Services, Bayswater, Australia).
2.11.2. Producing Emulsified Water (with and without FWM-CV) in Biodiesel
The pre-sonicated water containing FWM-CV was mixed with cottonseed biodiesel and surfactant in a 300 mL Pyrex beaker. The total volume of the emulsion was 250 mL made-up of 19.8% volumetric percentage of microalgae slurry (49.5 mL), 79.2% volumetric percentage of cottonseed biodiesel (198 mL) and 1% of surfactant (2.5 mL). The dry weight percentage of microalgae biomass in the slurry was 2 g·L−1, so the net dry biomass of microalgae in o liter of CS-ME20 is approximately 0.4 g (≈20% of the total dry biomass). Surfactants are normally added to the emulsion to prevent the separation between the emulsion constituents and maintain stable emulsion for certain period of time [33]. Triton X-100 was used as a surfactant in this study as recommended by Scragg et al. [34]. The mixture of water containing microalgae, biodiesel and surfactant was treated in batches with ultrasound. The batch volume was 250 mL. Each batch was treated with an ultrasonic power intensity of 24.17 W·cm−2 for 10 min.
2.11.3. Emulsion Physical Properties Measurements
The physical properties of the emulsified water fuel significantly affect the fluid dynamics in the injection and combustion process. Since fuel prices and fuel consumption are normally based on the volume, the heating value of the fuels is usually normalised by mass unit, therefore determining the fuel density is essential. The density was measured for all the fuels using volumetric and weighing measurements (Labco pipette, Brisbane, Australia) and Explorer OHAUS E12140 balance, (Melbourne, Australia). The density of each fuel was taken as the average of ten readings at 20 °C.
The viscosity of the fuels was measured using a BROOKFIELD Viscometer DV-II+Pro EXTRA (Middleboro, MA, USA). The viscometer was connected to a Brookfield temperature controller. The suitable spindle and rotational speed to obtain a torque percentage within the working range of the device. The viscosity readings were recorded when all the measurement parameters are stable.
The surface tension and heat of combustion were measured at the Queensland University of Technology (QUT). A KSV Sigma 702 Tensiometer (Castle Hill, Australia) equipped with Du Nouy ring for interfacial tension was used for surface tension measurements. Interfacial tension ring was set in contact with fuel sample. The force required for pulling the ring from the sample surface corresponds to the surface tension of the sample. The measurements were repeated at least three times to minimize the experimental errors and the average was presented.
The heat of combustion (Higher Heating Value) of the fuel samples was measured using a model 6200 oxygen bomb calorimeter. The heat of combustion is determined by measuring the amount of heat produced from burning fuel sample with oxygen under constant volume to obtain complete combustion. The heat capacity of bomb calorimeter was evaluated by burning a standard material with known heat capacity (benzoic acid). The heat of combustion of the sample was then determined from the heat capacity of the bomb calorimeter and the temperature rise of the heat sink (water) when the fuel sample is burned.
2.12. Engine Test
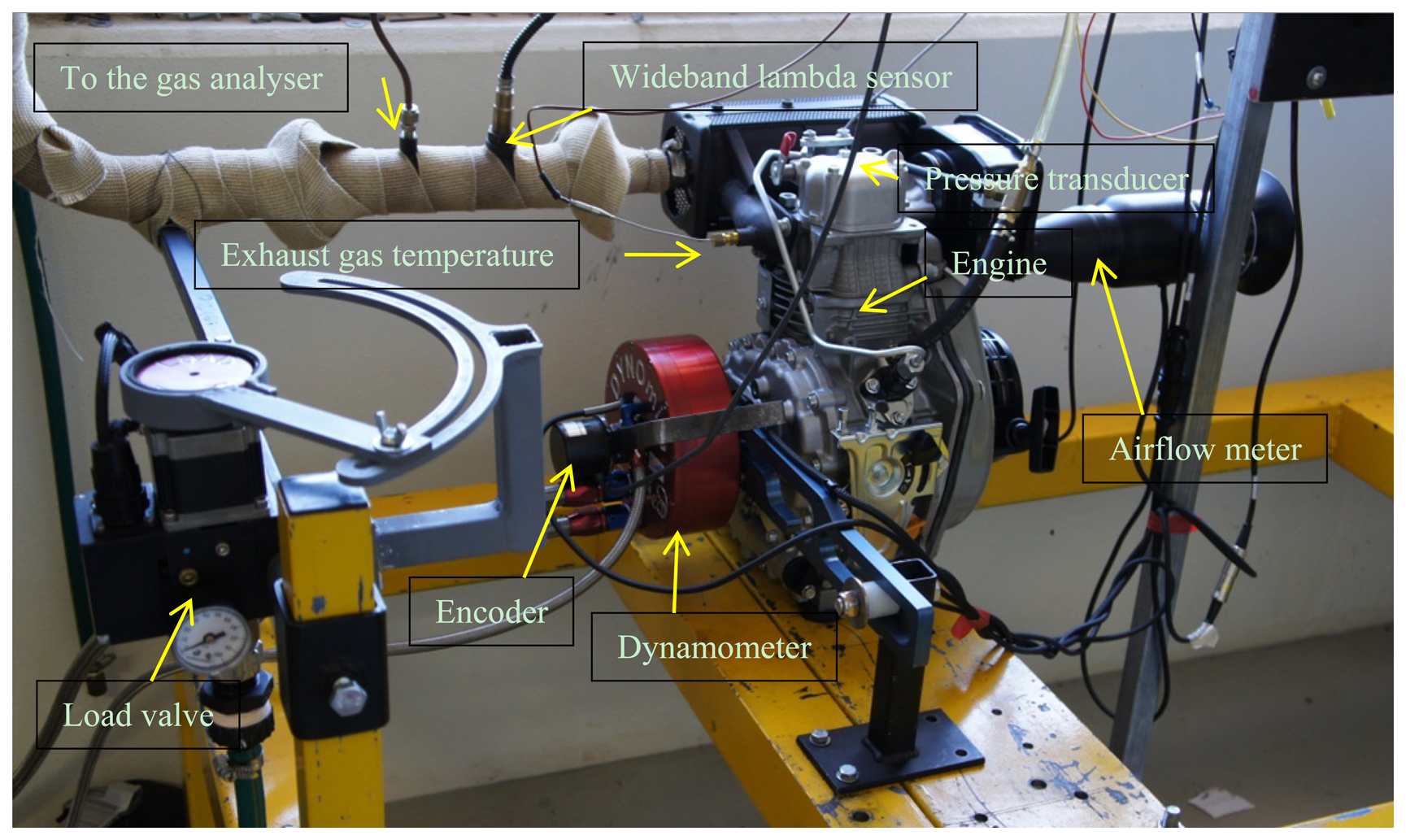
An engine test was performed using a single-cylinder air-cooled Yanmar L48N6 diesel engine (Yanmar Italy S.P.A, Cassano Magnago VA, Italy). The data that will be discussed in this work is only for the maximum output power at the engine speed of 3670 rpm. The engine test bed is presented in Figure 4. A Land-and-Sea Water-brake dynamometer (Concord, NH, USA) was used to load the engine and to measure the engine speed and break power. The exhaust gas emission was measured using a CODA Gas analyser (Hamilton, Australia).
3. Results and Discussion
3.1. Bimass Productivity
Microlgae biomass production is an essential indicator for biodiesel production at a specific lipid content. Monitoring the cultures growth rate using microscopy and haemocytometry is a slow method for a large number of samples, therefore finding an easy and effective way to have an indication for the microalgae growth would be valuable. The optical density measured using a spectrophotometer gives an indication for cell density and it is an easy method. A linear regression equation was derived for FWM-CV in MBL as given in Equation (1):
Cell density per volume of media could be an unsuitable indication for biomass and biodiesel production. Thus, another linear correlation was derived for FWM-CV in MBL between the optical density and the dry weight of microalgae as shown in Equation (2), where ƒMdw is the dry weight of microalgae in milligram (Mdw):
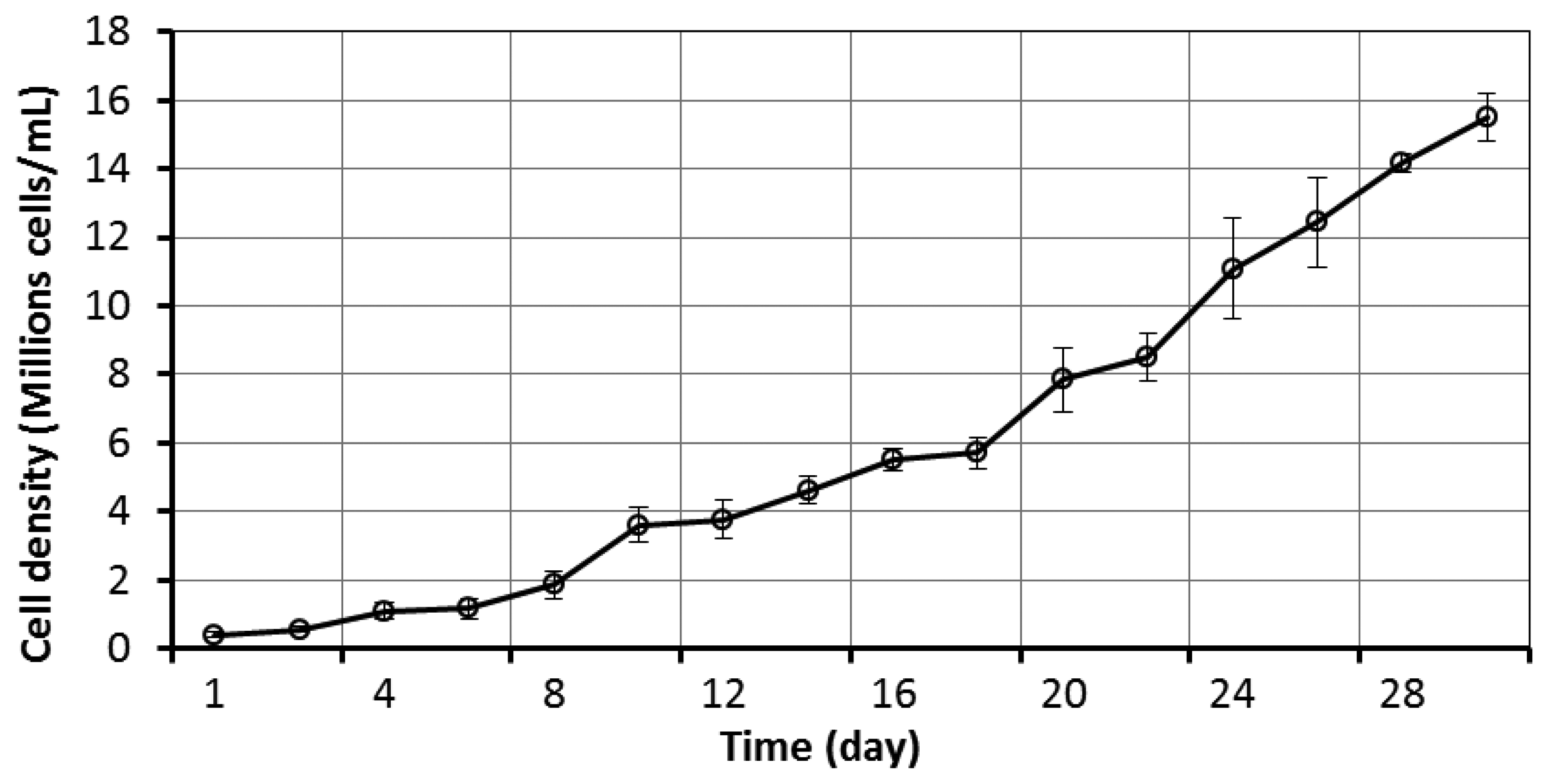
Figure 5 shows the growth curve of FWM-CV in MBL for thirty two days. This figure shows that the cell density in the first week is relatively slow due to the low inoculation ratio (small number of cells is added to the new media). In the next three weeks, the growth curve turns to be exponential. In the period of 28 to 32 days, the growth started to slow and the late exponential growth rate was identified to shift to the second stage.
Table 2 shows the summary of ANOVA and descriptive statistic of FWM-CV dry weight (g·L−1), biomass productivity (mg·L−1·day−1), lipid content (%) and lipid productivity (mg·L−1·day−1) for the control and the ISC samples.
The results in Table 2 are comparable to the results of Lee et al. [35] and Mata et al. [17]. Table 2 demonstrates that the dry weight of FWM-CV is significantly different between the control and the ISC. The dry weight of 0.89 (g·L−1) produced by ISC is significantly higher than that of the control sample. This difference is justified by the growing time and the new fresh medium added to the ISC. The dry weight of the control and ISC are 0.636 g·L−1 and 0.885 g·L−1 respectively which are both greater than Lee et al. [34] results due to the significant difference in culturing time.
To illuminate the differences in culture time, the biomass productivity was measured in mg·L−1·day−1 (dividing the biomass dry weight by the sample volume and the growth time) which gives a better indication of productivity. ANOVA test in Table 2 presents highly significant differences in the biomass productivity between the control and the ISC samples. The ISC is significantly surpassed the control by yielding 14.76 mg·L−1·day−1 due the new medium added in day 38 which offers more nutrients. Table 2 also shows that the biomass productivity of FWM-CV in MBL is lower than the results of Lee et al. [35] and Mata et al. [17]. The reason for this difference is due to the low concentration of FWM-CV culture at the start of growth which requires a longer time to reach the same concentration in Lee et al. [35] and Mata et al. [17] and the medium type that does not provide the required nutrient for fast growing.
Table 2 presents a comparison of biomass and lipid productivities of FWM-CV. It illustrates that the lipid content and the lipid productivity are significantly changed during the experiment. The results of ISC sample shows that the lipid content and the lipid productivity were significantly increased from 8.9% to 19.3% and from 0.74 mg·L−1·day−1 to 2.19 mg·L−1·day−1 respectively as shown in Table 2. This increment is found to be significantly lower than the results obtained by Liu et al. [6]. The control samples give lower lipid content compared to the results obtained by Lee et al. [35] and in the lower range of Mata et al. [17]. The possible reasons for the low results are the longer growing time, the growing media not being rich in nutrients for lipid production and the low percentage of carbon sources in autotrophic growing conditions present. The efficiency of the lipid extraction was also low and there was potential for loss of lipid in the glassware when transferring product.
3.2. FWM-CV Biodiesel
3.2.1. FAMEs Components
The differences in FAME components in FWM-CV biodiesel for the control sample and ISC samples, which have been compared with the components reported by Harwood [36], are illustrated in Table 3. The results demonstrate that adding FeCl3 to the medium in the ISC sample caused a significant difference in the FAMEs of FWM-CV. The percentages of some FAMEs are increased and others are decreased in comparison with the control sample and the components reported by Harwood [36]. In the ISC sample there is a significant reduction in the percentage of palmitic acid methyl ester (C17H34O2) 4.5% and α-linoleic acid methyl ester (C19H32O2) 2.5% as compared to the results of the control samples and Harwood [36]. Conversely, there is a significant increase in the percentage of palmatoleic acid methyl ester (C17H32O2), stearic acid methyl ester (C19H38O2) and linoleic acid (C19H34O2) which are 29.5%, 31.5% and 29.6% respectively. Such changes affect the properties of biodiesel in terms of density, cetane number, kinematic viscosity and heat of combustion.
3.2.2. Physicochemical Properties Analyses
Table 4 illustrates the physical properties of the common FAMEs as reported in the literature. Those properties are used to calculate the physical properties for the FAMEs in the FWM-CV. From Table 4, it can be seen that the highest density value is 0.901 g·cm−3 for α-linoleic acid methyl ester (C19H32O2), while the lower density is for stearic acid methyl ester (C19H38O2) 0.864 g·cm−3. Those values show an agreement with Ramírez-Verduzco et al. [22] who found that for the same carbon number, increasing the number of double bonds increases the density.
Viscosity is one of the most important physical fuel parameters. Biodiesel normally has a higher viscosity than diesel [37]. Table 4 shows that the saturated FAMEs stearic acid methyl ester (C19H38O2) and palmitic acid methyl ester (C17H34O2) have a higher kinematic viscosity (5.85 mm2·s−1 and 4.38 mm2·s−1 respectively) compared to the unsaturated FAMEs. Similarly, the saturated FAMEs also showed the higher cetane number than unsaturated FAMEs. The highest cetane numbers are 86.9 and 74.5 for stearic acid methyl ester (C19H38O2) and palmitic acid methyl ester (C17H34O2) respectively.
The heat of combustion is another important fuel property as it determines the energy of the fuel. Mehta and Anand [38] found a high degree of correlation between the number of double bonds and the lower heating value of the FAMEs, for example with C18 compounds, the lower heating value decreases with an increase in the number of double bonds. This can be seen in Table 4 which presents the highest heat of combustion in the saturated FAME stearic acid methyl ester (C19H38O2) 40.1 MJ·kg−1.
The kinematic viscosity, cetane number and heating value of the FAMEs in FWM-CV show a good agreement with Ramírez-Verduzco et al. [22] findings of the kinematic viscosity, cetane number and higher heating value increase when the number of double bonds decrease.
Table 5 represents the calculated physical properties of the biodiesel FWM-CV based on the FAME percentages in the samples. It shows that the density of the control and ISC samples are higher than the density of diesel fuel, and are in the same range of biodiesel standards. The control sample presents higher density than the ISC sample because of the higher percentage of α-linoleic acid methyl ester (C19H32O2) 13.29% which has a density of 0.901 g·cm−3 [40].
Table 5 also displays that the kinematic viscosity of biodiesel from ISC samples is slightly higher than that of the control sample. This increase is due to the high percentage of stearic acid methyl ester (C19H38O2) 31.50% in the ISC sample which has a viscosity of 5.85 mm2 s−1 (at 40 °C) [22]. That is however the percentage of palmitic acid methyl ester (C17H34O2) in the ISC sample is lower than the control sample, 4.52% and 23.74% respectively.
The heat of combustion of the control and ISC samples is relatively close. Biodiesel from FWM-CV presents a heat of combustion lower than diesel and in the range of biodiesel standards.
3.3. Emulsion Fuel Properties
It can be seen in Figure 6 that the injector orifice diameter expressed by equivalent circle diameter is 193.698 μm while the average sample of FWM-CV colonies diameter is averaged 2011 μm as presented in Figure 7. The size of the aggregates is much higher than the injector orifice size which would lead to orifice blockage and possible injector damage or inconsistency in the injector performance. It can be clearly seen from Figures 7 and 8 that ultrasound treatment has successfully disintegrated the microalgae aggregates into dispersed cells in the water. This in turn reduces the chances of nozzle blockage due to the entrapment of large microalgae aggregate into nozzle orifice.
It can also be observed from Figure 8 that the maximum aggregate size is 47.281 μm, which is less than the injector orifice diameter. The small cells size enhances the emulsion stability since the larger aggregated tend to precipitate faster.
3.3.1. Density and Viscosity
The densities of the CS-B100, CS-E20 and CS-ME20 are presented in Table 6. It can be seen from Table 6 that the variations in the density values is insignificant and the differences are in the error range. Emulsion fuels contain some degree of fine air bubbles as a result of the mixing process. The fine air bubbles reduce the density which is expected to be higher due to the presence of water in the emulsion. This in turn results in close density values for all fuels.
Table 6 also presents the dynamic viscosity values for CS-B100, CS-E20 and CS-ME20 measured at 25 °C and 40 °C. The viscosity of the emulsion fuels is extremely higher than that of the CS-B100.
The surface tension of CS-ME20, 31.675 mN−1·m is slightly lower than that of the CS-E20 which was 32.105 mN·m−1. The difference in the surface tension between CS-ME20 and CS-E20 can be ascribed to the additional FWM-CV cells and fragments of cells that lower the surface tension of the fuel. This lower surface tension can help in performing finer droplet size during the fuel injection process.
In general, Table 6 shows that emulsion fuels with and without microalgae cells have a lower heat of combustion as compared to cottonseed biodiesel. The lower value of heat of combustion for the emulsion fuels is expected as a result of the water content in the emulsion fuels. However, the heat of combustion of the emulsion was slightly enhanced (increased from 30.18 to 31.07 MJ·kg−1) by adding FWM-CV to it.
3.3.2. Fuel Stability
One of the most important characteristics of the emulsion fuel is the stability. The layers separation causes many problems such as abnormal combustion or even engine stoppage due to the injection of a high percentage of water. The stability of the CS-E20 and the CS-ME20 fuel was monitored after four hours and after two days as illustrated in Figure 9a,b respectively. It is clear from Figure 9 that both fuels are very stable for more than two days. The enhancement of the emulsion fuel homogeneity is mainly a result of the presence of the emulsifier and the effective mixing method using ultrasound.
3.3.3. Injector Nozzle Test
Nozzle test is an important experimental evaluation to determine the feasibility of emulsion as alternative fuel in diesel engines. Samples of the emulsified water fuels were tested using nozzle tester and high speed camera Model SA3 (Photron, Tokyo, Japan) with the capability of capturing 1000 frame per second video. Samples of the injection process of the CS-B100 and CS-ME20 is presented in Figure 10. It can be observed from Figure 10 that the spray pattern of both CS-B100 and CS-ME20 is relatively consistent and no problem with fuel pattern was recognized. The CS-ME20 shows a spray angle of 18° which is slightly wider than that of the CS-B100 (16°). This difference may be explained by the presence of water and microalgae cells that formed fine droplets in the CS-ME20.
3.4. Engine Test
Figure 11 presents the percentage of several determinants of engine performance with CS-ME20 and CS-E20 fuels as compared to CS-B100 as a base line at engine speed of 3670 rpm. It can be seen from this figure that the brake power (BP) dropped by 24.1% when CS-E20 used. This drop can be justified by the 20% water content that reduced the lower heating value associated with the higher viscosity. In comparison, the reduction in the brake power of CS-ME20 was less at a percentage of 15.18%. The less reduction in the BP of CS-ME20 as opposed to CS-E20 is attributed to the improved heating value of the fuel that was caused by the added FWM-CV cells. It can also be due to the fuel spray angle 18° (see Figure 10) which indicates finer droplet size resulting in better combustion. These results agree with Fahd et al. [45] and Koc and Abdullah [46] in the power reduction in the emulsified water fuel due to the lower heating value.
The brake specific fuel consumption (BSFC) for CS-E20 and CS-ME20 increased by 41.8% and 41.2%, respectively due to the higher fuel injected to maintain the engine at the same speed and the lower power produced with the emulsion fuels that is mainly ascribed to the water content of the fuels.
Figure 11 shows that the exhaust gas temperature of CS-E20 and CS-ME20 is lower than that of CS-B100 by 24.5% and 9.6% respectively. The decrease in the exhaust gas temperature of the emulsion fuels is due to the lower heating value and the water content, which absorbed some heat in the combustion process [45,46]. The reduction in the exhaust gas temperature significantly reduces the NOx emission from the emulsion fuels which dropped by 71.9% and 60.4% for CS-E20 and CS-ME20 respectively. The exhaust gas temperature from CS-ME20 is higher than the exhaust gas temperature from CS-E20 by 19.7% due to the higher calorific value that results from the presence of FWM-CV cells. The reduction in the NOx level showed very strong agreement with the reduction found by Scragg, Morrison and Shales [34], when compared with biodiesel fuel from rapeseed with emulsion fuel containing microalgae slurry. The emulsion fuels of CS-E20 and CS-ME20 produce significantly lower CO2 than CS-B100 by 19.9% and 11.6% due to the better combustion that allows more CO to be converted to CO2. This finding is in a good agreement with [34].
4. Conclusions
In this work, fresh water C. vulgaris microalgae (FWM-CV) were grown to monitor the growth curve, biomass productivity, lipid content, fatty acid methyl ester (FAME) components and to evaluate the physical properties of the resulting microalgae biodiesel fuel. To increase the lipid content, iron was added to the media as a stressor treatment. Adding iron to the culture medium has a positive influence on the lipid content and gives different fatty acid methyl ester (FAME) components, which led to different biodiesel properties. The results demonstrate that the physical properties of FWM-CV biodiesel and the conventional diesel are close. The ISC presents higher kinematic viscosity and higher heat of combustion compared to the control and diesel. There is no significant difference in density and heat of combustion between the control and ISC. The kinematic viscosity and the density of FAME from FWM-CV are within the range of ASTM standards for biodiesel. Cetane number is higher than conventional diesel.
Emulsified water fuel in cottonseed biodiesel was prepared using ultrasound. The emulsified water fuel was prepared with and without adding FWM-CV cells to enhance the fuel heating value. Using ultrasound effectively broke down the colonies of FWM-CV to individual cells to prevent nozzle blockage.
The engine test at 3670 rpm indicated an increase in the BSFC with emulsified water fuels, while the emulsified water fuels presented lower results than CS-B100 in terms of engine power, exhaust gas temperature, NOx and CO2 emissions due to the water content that affected the fuel properties. Adding FWM-CV cells to the emulsified water fuel enhanced the fuel properties of CS-ME20 and increased the engine power by 8.93% compared to the CS-E20.
Acknowledgments
The authors are grateful to the financial and technical support of the Iraqi government and the University of Southern Queensland. Thanks are due to Dr. R.J. Brown and M.A. Islam, at the Queensland University of Technology for providing cottonseed biodiesel and facilitating the measurements of surface tension and heat of combustion of the used fuels.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar]
- Yusaf, T.; Baker, P.; Hamawand, I.; Noor, M.M. Effect of compress natural gas mixing on the engine performance and emissions. Int. J. Automot. Mech. Eng. 2013, 8, 1438–1451. [Google Scholar]
- Singh, J.; Gu, S. Commercialization potential of microalgae for biofuels production. Renew. Sustain. Energy Rev. 2010, 14, 2596–2610. [Google Scholar]
- Mustafa, B. Potential alternatives to edible oils for biodiesel production—A review of current work. Energy Convers. Manag. 2011, 52, 1479–1492. [Google Scholar]
- Miao, X.; Wu, Q. Biodiesel production from heterotrophic microalgal oil. Bioresour. Technol. 2006, 97, 841–846. [Google Scholar]
- Liu, Z.-Y.; Wang, G.-C.; Zhou, B.-C. Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresour. Technol. 2008, 99, 4717–4722. [Google Scholar]
- Widjaja, A.; Chien, C.-C.; Ju, Y.-H. Study of increasing lipid production from fresh water microalgae Chlorella vulgaris. J. Taiwan Inst. Chem. Eng. 2009, 40, 13–20. [Google Scholar]
- Lv, J.-M.; Cheng, L.-H.; Xu, X.-H.; Zhang, L.; Chen, H.-L. Enhanced lipid production of Chlorella vulgaris by adjustment of cultivation conditions. Bioresour. Technol. 2010, 101, 6797–6804. [Google Scholar]
- Converti, A.; Casazza, A.A.; Ortiz, E.Y.; Perego, P.; del Borghi, M. Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem. Eng. Process. Process Intensif. 2009, 48, 1146–1151. [Google Scholar]
- Al-lwayzy, S.H.; Yusaf, T.; Jensen, T. Evaluating tractor performance and exhaust gas emissions using biodiesel from cotton seed oil. IOP Mater. Sci. Eng. 2012, 36, 012042. [Google Scholar]
- Hossain Sharif, A.B.M.; Salleh, A.; Nasrulhaq, A.; Chowdhury, P.; Naqiudden, M. Biodiesel fuel production from algae as renewable energy. Am. J. Biochem. Biotechnol. 2008, 4, 250–245. [Google Scholar]
- Demirbas, A.; Fatih Demirbas, M. Importance of algae oil as a source of biodiesel. Energy Convers. Manag. 2011, 52, 163–170. [Google Scholar]
- Demirbas, A. Importance of biodiesel as transportation fuel. Energy Policy 2007, 35, 4661–4670. [Google Scholar]
- Amin, S. Review on biofuel oil and gas production processes from microalgae. Energy Convers. Manag. 2009, 50, 1834–1840. [Google Scholar]
- Demirbas, A. Use of algae as biofuel sources. Energy Convers. Manag. 2010, 51, 2738–2749. [Google Scholar]
- Balat, M.; Balat, H. Progress in biodiesel processing. Appl. Energy 2010, 87, 1815–1835. [Google Scholar]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar]
- Al-lwayzy, S.; Yusaf, T. Chlorella protothecoides microalgae as an alternative fuel for tractor diesel engines. Energies 2013, 6, 766–783. [Google Scholar]
- Hamawand, I.; Yusaf, T.; Hamawand, S. Growing algae using water from coal seam gas industry and harvesting using an innovative technique: A review and a potential. Fuel 2014, 117 Part A, 422–430. [Google Scholar]
- Knothe, G. “Designer” biodiesel: Optimizing fatty ester composition to improve fuel properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar]
- Knothe, G. Dependence of biodiesel fuel properties on the structure of fatty acid alkyl esters. Fuel Process. Technol. 2005, 86, 1059–1070. [Google Scholar]
- Ramírez-Verduzco, L.F.; Rodríguez-Rodríguez, J.E.; Jaramillo-Jacob, A.D.R. Predicting cetane number, kinematic viscosity, density and higher heating value of biodiesel from its fatty acid methyl ester composition. Fuel 2012, 91, 102–111. [Google Scholar]
- Lif, A.; Holmberg, K. Water-in-diesel emulsions and related systems. Adv. Colloid Interface Sci. 2006, 123–126, 231–239. [Google Scholar]
- Fayyad, S.M.; Abu-Ein, S.; Al-Marahleh, G.; Al-Momani, W.; Al-Momani, M.; Abulghanam, Z.; Badran, O.; Abu-Rahmah, T. Experimental emulsified diesel and benzen investigation. Res. J. Appl. Sci. Eng. 2010, 2, 268–273. [Google Scholar]
- Nichols, H.W. Growth Media—Freshwater. In Handbook of Phycological Methods, Culture Methods and Growth Measurements; Stein, J., Ed.; Cambridge University Press: Cambridge, UK, 1973; pp. 7–24. [Google Scholar]
- Cheirsilp, B.; Torpee, S. Enhanced growth and lipid production of microalgae under mixotrophic culture condition: Effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 2012, 110, 510–516. [Google Scholar]
- Aravinthan, V.; Story, N.; Yusaf, T. Nutrient removal of nursery and municipal wastewater using Chlorella vulgaris microalgae for lipid extraction. Desalination Water Treat. 2013, 52, 727–736. [Google Scholar]
- Harris, P.; McCabe, B.K.; Pittaway, P. Growth and characterisation of Chlorella vulgaris under increased lipid culture regimes. Proceedings of the 2010 Southern Region Engineering Conference (SREC 2010), Toowoomba, Australia, 11–12 November 2010.
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Choix, F.J.; de-Bashan, L.E.; Bashan, Y. Enhanced accumulation of starch and total carbohydrates in alginate-immobilized Chlorella spp. Induced by Azospirillum brasilense: II. Heterotrophic conditions. Enzyme Microb. Technol. 2012, 51, 300–309. [Google Scholar]
- Halim, R.; Harun, R.; Danquah, M.K.; Webley, P.A. Microalgal cell disruption for biofuel development. Appl. Energy 2012, 91, 116–121. [Google Scholar]
- Al-Juboori, R.A.; Yusaf, T. Identifying the optimum process parameters for ultrasonic cellular disruption of E. coli. Int. J. Chem. React. Eng. 2012, 10, 1–32. [Google Scholar]
- Benter, M.M.; Gilmour, I.A.; Arnoux, L. Biomass-oil slurry fuels: An investigation into their preparation and formulation. Biomass Bioenergy 1997, 12, 253–261. [Google Scholar]
- Scragg, A.H.; Morrison, J.; Shales, S.W. The use of a fuel containing Chlorella vulgaris in a diesel engine. Enzyme Microb. Technol. 2003, 33, 884–889. [Google Scholar]
- Lee, J.Y.; Yoo, C.; Jun, S.Y.; Ahn, C.Y.; Oh, H.M. Comparison of several methods for effective lipid extraction from microalgae. Bioresour. Technol. 2010, 101, S75–S77. [Google Scholar]
- Harwood, J. Membrane Lipids in Algae. In Lipids in Photosynthesis: Structure, Function and Genetics; Paul-André, S., Norio, M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; pp. 53–64. [Google Scholar]
- Knothe, G.; Steidley, K.R. Kinematic viscosity of biodiesel fuel components and related compounds. Influence of compound structure and comparison to petrodiesel fuel components. Fuel 2005, 84, 1059–1065. [Google Scholar]
- Mehta, P.S.; Anand, K. Estimation of a lower heating value of vegetable oil and biodiesel fuel. Energy Fuels 2009, 23, 3893–3898. [Google Scholar]
- Tong, D.; Hu, C.; Jiang, K.; Li, Y. Cetane number prediction of biodiesel from the composition of the fatty acid methyl esters. J. Am. Oil Chem. Soc. 2011, 88, 415–423. [Google Scholar]
- Bamgboye, A.I.; Hansen, A.C. Prediction of cetane number of biodiesel fuel from the fatty acid methyl ester (fame) composition. Int. Agrophys. 2008, 22, 21–29. [Google Scholar]
- Knothe, G.; Steidley, K.R. Kinematic viscosity of biodiesel components (fatty acid alkyl esters) and related compounds at low temperatures. Fuel 2007, 86, 2560–2567. [Google Scholar]
- Robert, L.; McCormick, R.L.; Graboski, M.S.; Alleman, T.L.; Herring, A.M.; Tyson, K.S. Impact of biodiesel source material and chemical structure on emissions of criteria pollutants from a heavy-duty engine. Environ. Sci. Technol. 2001, 35, 1742–1747. [Google Scholar]
- Meher, L.; Vidya Sagar, D.; Naik, S. Technical aspects of biodiesel production by transesterification—A review. Renew. Sustain. Energy Rev. 2006, 10, 248–268. [Google Scholar]
- Kahraman, B. Biodiesel as an alternative motor fuel: Production and policies in the European Union. Renew. Sustain. Energy Rev. 2008, 12, 542–552. [Google Scholar]
- Fahd, M.E.A.; Wenming, Y.; Lee, P.S.; Chou, S.K.; Yap, C.R. Experimental investigation of the performance and emission characteristics of direct injection diesel engine by water emulsion diesel under varying engine load condition. Appl. Energy 2013, 102, 1042–1049. [Google Scholar]
- Koc, A.B.; Abdullah, M. Performance and NOx emissions of a diesel engine fueled with biodiesel-diesel-water nanoemulsions. Fuel Process. Technol. 2013, 109, 70–77. [Google Scholar]
Abbreviations
| ANOVA | Analysis Of Variance |
| FWM-CV | Freshwater Microalgae Chlorella Vulgaris |
| OD | Optical Density |
| Mdw | Microalgae Dry Weight |
| Fames | Fatty Acid Methyl Esters |
| ISC | Iron Supplemented Culture |
| CS-B100 | Cottonseed Biodiesel 100% |
| CS-E20 | Cottonseed Biodiesel Emulsified Water 20% |
| CS-ME20 | Cottonseed Biodiesel Containing Emulsified FWM-CV Water 20% |
| MBL | Medium Woods Hole |
| ECD | Equivalent Circle Diameter |
| BSFC Brake | Specific Fuel Consumption |
| BP | Brake Power |








| Stock solutions | Concentration per litre of distilled water (g·L−1) | |||
|---|---|---|---|---|
| 1. CaCl2·2H2O | 36.76 | |||
| 2. MgSO4·7H2O | 36.97 | |||
| 3. NaHCO3 | 12.60 | |||
| 4. K2HPO4 | 8.71 | |||
| 5. NaNO3 | 85.01 | |||
| 6. Na2SiO3·9H2O | 28.42 | |||
| 7. Na2EDTA | 4.36 | |||
| 8. FeCl3·6H2O | 3.15 | |||
| 9. Metal Mix | CuSO4·5H2O | 0.01 | Each constituent was added separately to ∼750 mL of dH2O then completed to up to 1 L with dH2O | |
| ZnSO4·7H2O | 0.022 | |||
| CoCl2·6H2O | 0.01 | |||
| MnCl2·4H2O | 0.18 | |||
| Na2MoO4·2H2O | 0.006 | |||
| 10. Vitamin stock | Cyanocobalamin (Vitamin B12) | 0.0005 | ||
| Thiamine HCl (Vitamin B1) | 0.10 | |||
| Biotin | 0.0005 | |||
| 11. Tris stock | 250.0 | |||
| Properties | Stage two | F value | C. vulgaris [35] | C. vulgaris [17] | |||
|---|---|---|---|---|---|---|---|
| Control | ISC + | ||||||
| Means | SD | Means | SD | ||||
| Growing time (days) | 75.5 | - | 78.5 | - | - | 7 | - |
| Dry weight (g·L−1) | 0.636 | 0.06 | 0.89 | 0.08 | 41.64 *** | 0.5 | - |
| Biomass productivity (mg·L−1·day−1) | 10.598 | 0.97 | 14.76 | 1.33 | 13.39 *** | 74.2 | 20–200 |
| lipid content % | 8.94 | 0.82 | 19.27 | 1.70 | 88.16 *** | 14.96–5.58 | 5.0–58.0 |
| lipid productivity (mg·L−1·day−1) | 0.74 | 0.07 | 2.19 | 0.19 | 121.76 *** | 11.10–6.91 ++ | 11.2–40.0 |
+Iron supplemented culture in which new media and FeCl3 were added after 38 days++Calculated by the authors of this paper from the published data of lipid content, dry weight, lipid productivity, biomass productivity and growing time in [29]*** p (significance level) ≤ 0.001
| Fatty acids (FAs) | FAs formula | FAs of M-CV * (%) | FAME name | FAME formula | FAME of FWM-CV (%) | |
|---|---|---|---|---|---|---|
| Control | ISC | |||||
| Myristic acid (C14:0) | C14H28O2 | - | Myristic acid methyl ester (Methyl myristate) | C15H30O2 | - | 2.33 |
| Palmitic acid (C16:0) | C16H32O2 | 26 | Palmitic acid methyl ester (Methyl palmitate) | C17H34O2 | 23.74 | 4.52 |
| Palmatoleic acid (C16:1) | C16H30O2 | 8 | Palmatoleic acid methyl ester (Methyl palmitoleate) | C17H32O2 | 16.48 | 29.54 |
| Hexadecadienoic acid (C16:2) | - | 7 | - | - | - | - |
| Linolenic acid (C16:3) | - | 2 | - | - | - | - |
| Stearic acid (C18:0) | C18H36O2 | - | Stearic acid methyl ester (Methyl stearate) | C19H38O2 | 21.84 | 31.50 |
| Oleic acid (C18:1) | C18H34O2 | 2 | Oleic acid methyl ester (Methyl oleate) | C19H36O2 | - | - |
| Linoleic acid (C18:2) | C18H32O2 | 24 | Linoleic acid methyl ester (Methyl linoleate) | C19H34O2 | 24.64 | 29.59 |
| α-Linoleic acid (C18-3) | C18H30O2 | 20 | α-Linoleic acid methyl ester (Methyl gamma linolenate) | C19H32O2 | 13.29 | 2.52 |
*Fatty acids (FAs) in microalgae C. vulgaris (M-CV) [36].
| Fatty acid | Cetane number | Density (g·cm−3) | Kinematic viscosity (40 °C mm2s−1) | Heat of combustion (MJ·kg−1) |
|---|---|---|---|---|
| Myristic acid | 66.2 a,b,c | 0.867 d | 3.3 e | 38.9 d |
| Palmitic acid | 74.5 a,c | 0.865 d | 4.38 e | 39.45 d,g |
| Palmatoleic acid | 51.0 a,c | 0.869 d | 3.67 f | 39.30 d,g |
| Stearic acid | 86.9 a | 0.864 d | 5.85 e | 40.1 d,g |
| Linoleic acid | 38.2 a,b | 0.886 d | 3.65 e | 39.7 d,g |
| α-Linoleic acid | 22 b,c | 0.901 d | 3.14 e | 39.34 d,g |
| Samples | Density (kg·m−1) | Cetane number | Kinematic viscosity (40°C·mm2·s−1) | Heat of combustion (MJ·kg−1) |
|---|---|---|---|---|
| Control sample | 875.31 | 57.499 | 4.239 | 39.6 |
| ISC sample | 873.03 | 59.224 | 4.361 | 39.7 |
| Diesel | 838 c | 46.00 d | 1.9–4.1 a | 45.3–46.7 f |
| Biodiesel | 860–900 c | ≥47 e | 1.9–6.0 b | 39.3–39.8 f |
aStandard ASTM D957 method ASTM D445 [37]bStandard ASTM D6751 method ASTM D445 [37]c[5]dD613 ASTM method [42]e[43]fHigher heating value [44]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-lwayzy, S.H.; Yusaf, T.; Al-Juboori, R.A. Biofuels from the Fresh Water Microalgae Chlorella vulgaris (FWM-CV) for Diesel Engines. Energies 2014, 7, 1829-1851. https://doi.org/10.3390/en7031829
Al-lwayzy SH, Yusaf T, Al-Juboori RA. Biofuels from the Fresh Water Microalgae Chlorella vulgaris (FWM-CV) for Diesel Engines. Energies. 2014; 7(3):1829-1851. https://doi.org/10.3390/en7031829
Chicago/Turabian StyleAl-lwayzy, Saddam H., Talal Yusaf, and Raed A. Al-Juboori. 2014. "Biofuels from the Fresh Water Microalgae Chlorella vulgaris (FWM-CV) for Diesel Engines" Energies 7, no. 3: 1829-1851. https://doi.org/10.3390/en7031829
APA StyleAl-lwayzy, S. H., Yusaf, T., & Al-Juboori, R. A. (2014). Biofuels from the Fresh Water Microalgae Chlorella vulgaris (FWM-CV) for Diesel Engines. Energies, 7(3), 1829-1851. https://doi.org/10.3390/en7031829




