An Experimental and Numerical Investigation of Fluidized Bed Gasification of Solid Waste
Abstract
: Gasification is a thermo-chemical process to convert carbon-based products such as biomass and coal into a gas mixture known as synthetic gas or syngas. Various types of gasification methods exist, and fluidized bed gasification is one of them which is considered more efficient than others as fuel is fluidized in oxygen, steam or air. This paper presents an experimental and numerical investigation of fluidized bed gasification of solid waste (SW) (wood). The experimental measurement of syngas composition was done using a pilot scale gasifier. A numerical model was developed using Advanced System for Process ENgineering (Aspen) Plus software. Several Aspen Plus reactor blocks were used along with user defined FORTRAN and Excel code. The model was validated with experimental results. The study found very similar performance between simulation and experimental results, with a maximum variation of 3%. The validated model was used to study the effect of air-fuel and steam-fuel ratio on syngas composition. The model will be useful to predict the various operating parameters of a pilot scale SW gasification plant, such as temperature, pressure, air-fuel ratio and steam-fuel ratio. Therefore, the model can assist researchers, professionals and industries to identify optimized conditions for SW gasification.1. Introduction
Solar, wind, tides, geothermal and hydroelectric are very popular renewable energy sources. However, there is another significant source of energy that is created by our everyday activities across the world known as biomass. Biomass plays a vital role in the production of fuel or electricity. The potential of renewable energy for urban and rural development, liquid fuel replacement and greenhouse gas reduction are current concerns all over the world. Stucley et al. [1] examined the use of biomass to generate electricity and produce liquid transport fuels in Australia. The main focus of their study was on biomass from forestry, particularly new forestry that may also provide other environmental benefits in Australia's dry land regions.
According to renewable energy perception, biomass can be defined as [1]: “Recent organic matter originally derived from plants as a result of the photosynthetic conversion process, or from animals, and which is destined to be utilized as a store of chemical energy to provide heat, electricity, or transport fuels.”
Biomass resources include wood from plantation forests, residues from agricultural or forest production, and organic waste by-products from industry, domesticated animals, and human activities [1,2]. According to biomass definition, solid waste (SW) is a proven natural resource for renewable energy. Energy can be recovered from SW through various technologies, such as:
- -
Combustion, which is a rapid chemical reaction of two or more substances, is commonly called burning. In practical combustion systems the chemical reactions of the major chemical species, carbon (C) and hydrogen (H2) in the fuel and oxygen (O2) in the air, are fast at the prevailing high temperatures (approximately, greater than 900 °C) because the reaction rates increases exponentially with temperature.
- -
Pyrolysis and gasification, where the fuel is heated with little or no O2 to produce “syngas” which can be used to generate energy or as a feedstock for producing methane (CH4), chemicals, biofuels or H2.
Amongst a number of conversion routes of waste to energy technology, gasification plays a vital role. Gasification uses partial oxidation, in contrast to combustion which uses excess air. It produces a combustible gas which is a mixture of carbon monoxide (CO), H2 and CH4. Biomass gasification plants are in the early commercial stage of development. When used to produce electricity, there are considerable gains associated with scale of plant. At 1 MW electrical (MWe) an updraft gasifier has an efficiency of 10% to 20%, while a 10 MWe fluid bed gasifier has an efficiency of 25% to 35%, and a 100 MWe entrained flow or pressurized circulating fluid bed gasifier has an efficiency of 40% to 50% [2].
A number of studies have been performed on the gasification process. Doherty et al. [3] developed a simulation model of a circulating fluidized bed (CFB) gasifier using Advanced System for Process ENgineering (Aspen) Plus. They calibrated the model against experimental data and investigated the effects of varying equivalence ratio (ER), temperature, level of air preheating, biomass moisture and steam injection on product gas composition, gas heating value, and cold gas efficiency. Nikoo and Mahinpey [4] developed a fluidised bed gasification model for biomass addressing both hydrodynamic parameters and reaction kinetic in their model. Kumar et al. [5] studied on simulation of corn stover and distillers grains gasification where they developed to simulate the performance of a lab-scale gasifier and predict the flowrate and composition of product from given biomass composition and gasifier operating conditions using Aspen Plus software. They applied mass balance, energy balance, and minimization of Gibbs free energy during the gasification to determine the product gas composition. Abdelouahed et al. [6] presented a detailed modeling of biomass gasification in dual fluidized bed (DFB) reactors under Aspen plus. In their model, the DFB was divided into three modules according to the main chemical phenomena: biomass pyrolysis, secondary reactions and char combustion. Most recently, Mavukwana et al. [7] developed a simulation model of sugarcane bagasse gasification, and Francois et al. [8] reported process modeling of a wood gasification combined heat and power plant using Aspen Plus.
This paper presents a detailed investigation of SW gasification, both experimentally and numerically. Experimental investigations have been done in a pilot-scale gasification plant. A numerical simulation model has been developed using the Aspen Plus software package. The model has been validated with the experimental data. The model will be very useful for the professionals, researchers and industry people involved in waste to energy technologies.
2. Experimental Investigations
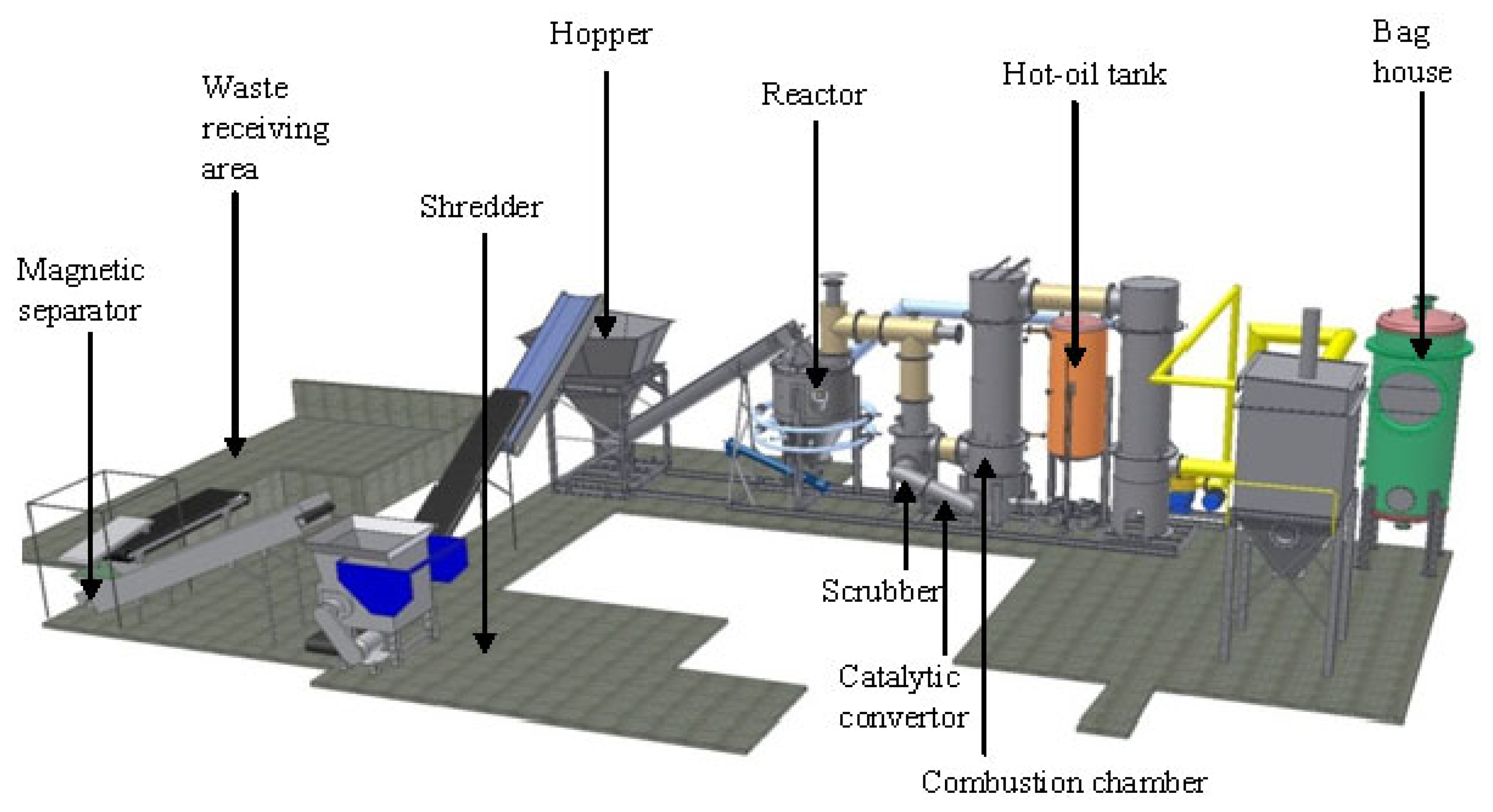
The experimental study was performed using a pilot-scale gasification plant which is used for energy recovery from SW. The layout of the gasification plant is shown in Figure 1. The gasification plant is composed of four modules: a waste pre-processing unit, the gasification/oxidation chambers, the energy recovery section and finally the flue gas cleaning section. In the pre-processing module the waste is sorted, grinded, shredded, stored and dried with the purpose of obtaining a gasification-friendly feed material, free of metals, glass and plastic bottles. The main components of the plant are: waste receiving area, magnetic separator, shredder, hopper, reactor, hot-oil tank and bag house (Figure 1).
The gasification process involves a number of steps. A schematic diagram of the gasification process is shown in Figure 2. Wood was used as SW feedstock. Feedstocks are collected from neighboring city or regional councils. Collected wastes are prepared for gasification. Feedstock preparation includes feedstock sorting, preparation and shredding. In this step, all unexpected materials have been removed. The feedstock is manually hand sorted to ensure that recycling was maximized and the shredder is protected from rocks, glass and metallic items. This ensures that no dangerous items enter the reactor vessel. All feed are shredded before being placed into the hopper and subsequent screw conveyor. As ideal sorting is complex, reactors have scrubbing systems to manage any slippage through the sorting process. The scrubbing systems are not a substitute for lack of sorting. A slippage rate of 2% is considered normal.
To increase gasification performance, prepared feedstocks need to be dried to reduce moisture through the drier. Gasification process is placed in reaction vessel in the presence of air and steam which produce raw syngas. Syngas cleaning is done to remove the pollution from produced raw syngas. Burning is done in an internal combustion engine depending on its requirement.
The reaction vessel of the plant is used to convert waste to a small amount of char and ash along with a large amount of syngas (called gasification). In the reaction vessel:
- -
Waste's surface moisture is dried, along with all of the inherent moisture.
- -
Waste is further heated.
- -
Waste generates volatiles (oils, tars and CH4 which appear as yellow smoke).
- -
Partial combustion (yellow smoke) generates heat and evaporates plastics (if present).
- -
Char is gasified by reacting with O2, water or steam (H2O) and carbon dioxide (CO2) to form H2 and CO.
The experimental reaction vessel had a nominal volume of 2.5 m3, a waste and char capacity of approximately 1.75 m3 and a freeboard capacity of 0.75 m3. The freeboard is the space above the fluidised bed where the gas slows down after leaving the bed and most of the solids fall back into the bed. The waste is fluidised in the reactor by a gaseous mixture of waste gas [burnt syngas—nitrogen (N2), CO2 and H2O] and fresh air (N2, O2) [9].
The wastes are heated and dried in the drier which uses hot oil at 220 °C. The SWs are heated in the range of 700–1100 °C in a reduced O2 environment in the reactor. In the reactor, raw syngas is produced which is a mixture of CO, CO2, H2, H2O, N2 and CH4. The raw syngas is contaminated with tar hydrogen sulphide (H2S), ammonia (NH3) and trace hydrochloric acid (HCl). The fine SW leaves the reactor and settles on the top of the scrubber and forms a network of fine C char. The tar gets trapped on the char network and on burnt dolomite at the top of the scrubber and is subsequently broken down into C, CO, and H2 and H2O. The H2S and NH3 are converted to sulphur dioxide (SO2), nitric oxide (NO) and H2O in the scrubber. The dolomite mops up the acid gases like SO2, NO, NO2 and HCl. O2 is added at the scrubber to maintain the scrubber temperature at about 600 °C [9].
The hot syngas is indirectly cooled using heat transfer oil. The heated oil is used for drying the SW. The hot syngas is further indirectly cooled using fluidising gas. The heated fluidizing gas is used in the gasifier. The warm syngas is further cooled and compressed using liquid ring compressors.
This is a final scrub for acid gases and also removes about 80% of the water out of the syngas. The clean syngas is a mixture of CO, CO2, H2, N2 and H2O. The compounds of syngas found at different stages are shown in Table 1. Energy content of SW was distributed to, approximately, 65% for syngas production, 23% for char production, 6% for hot oil and remaining was lost as heat. This implies that the energy conversion efficiency for syngas production using gasification is approximately 65%.
3. Model Development
A simulation model was developed and validated with experimental results. Amongst the whole experimental study, the model was developed for the main part of the gasification process (as identified by the red rectangle in Figure 2).
3.1. Model Assumptions
Because of the influence of hydrodynamic parameters on SW gasification in a fluidized bed, both the hydrodynamic and kinetic reactions were treated simultaneously. A number of assumptions were incorporated into the Aspen Plus fluidized bed gasifier modeling [4,10–13]:
- -
The gasification process is steady state and isothermal (uniform bed temperature).
- -
SW de-volatilisation is instantaneous in comparison to char gasification.
- -
Volatile products mainly consist of CO, H2, CO2, CH4 and H2O.
- -
Gases are uniformly distributed within the emulsion phase.
- -
Char comprises C and ash.
- -
Char gasification starts in the bed and is completed in the freeboard. According to Lee et al. [14], combustion and gasification take place in the main bed region and pyrolysis in the freeboard region.
- -
Based on the shrinking core model, particles are considered spherical and of uniform size and the average diameter remains constant during gasification.
- -
The simulation is performed using power-law kinetics.
3.2. Reaction Kinetics
The overall gasification process starts with pyrolysis and continues with combustion and steam gasification considering the following reactions:
Combustion reactions [4,14–16]:
Steam-gasification reactions [4,17]:
In the reaction shown in Equation (1), α is a mechanism factor [18] that differs in the range of 0.5–1 when CO2 and CO is carried away from the char particle during combustion of char. α is a function of temperature and average diameter of char particles. In the reaction shown in Equation (5), the term (2 − β)/β represents the fraction of the steam consumed by the reaction shown in Equation (2) and 2(β − 1)/β represents the fraction of steam consumed by the reaction shown in Equation (4). Matsui et al. [15] experimentally determined β to be in the range of 1.1–1.5 at 750–900 °C. The values of α and β for fluidized bed model of SW gasification were considered as 0.9 and 1.4, respectively, as determined by Nikoo and Mahinpey [4].
According to Lee et al. [14], the reaction rate equations defined for the mentioned reactions are as follows:
Walker et al. [19] and Dutta and Wen [20] considered parameter n to be equal to 1.0 in Equations (7) and (8). For the steam-gasification reaction, Kasaoka et al. [21] and Chin et al. [22] reported different numbers for n, but it is actually 1.0 in the steam partial pressure range of 0.25–0.8 atm. Kinetic parameters can be found in Table 2 [4]. All the symbols for the reaction kinetics have been defined in the nomenclature.
3.3. Hydrodynamic Assumptions
The following assumptions were considered in simulating the hydrodynamics [4,23]:
- -
Fluidized bed reactor comprises with two regions: bed and freeboard.
- -
The fluidisation condition in the bed is maintained in the bubbling regime.
- -
With the increasing height, the solids volume fraction decreases, similar to the grouping of bubbles in with solid particles returning to the bed.
- -
With increasing height, the volumetric flow rate of gas increases corresponding to the production of gaseous products.
- -
Solid particles mixing, such as consisting of ash, char particles, and bed material, are considered perfect.
- -
With constant hydrodynamic parameters, the reactor is divided into a finite number of equal elements.
- -
As the fluidised bed is considered one-dimensional, any variations in conditions are considered to occur only in the axial direction.
3.4. Bed Hydrodynamics
According to Kunii and Levenspiel [24], the minimum fluidisation velocity of fine particles can be determined by:
To determine the volume fraction occupied by bubbles in a fluidized bed, the following correlations developed by Babu et al. [25] are used:
Here, u is the superficial velocity which is not a constant parameter due to the gas production resulting from heterogeneous and homogeneous reactions. The importance of considering varying superficial gas velocity in obtaining results with higher precision in simulation is demonstrated by Yan et al. [26]. According to Kunii and Levenspiel [24], the bed void fraction is calculated using the following equation:
3.5. Freeboard Hydrodynamics
The volume fraction of solid at various levels z in the freeboard falls off exponentially from the value at the bed surface as calculated by the following equation [27]:
Here, a is a constant. Kunii and Levenspiel [24] present a graph that correlates the constant a with superficial gas velocity and particle size. The graph is useful in the following range: u ≤ 1.25 m/s:
3.6. Process Simulator: Aspen Plus
A number of processes modeling software package are used to develop computational model of fluidized bed gasification process and to perform simulation and validation of the model. Generally, researchers and professionals uses, Aspen Plus, Computational Fluid Dynamics (CFD, composed of GAMBIT and FLUENT), ChemCAD and MatLab software packages to develop and optimize the gasification model. Mhilu [28] conducted a study on modeling performance of high-temperature biomass gasification process using MatLab. In this study, the derived model equations were computed using the MAPLE process simulation code in MatLab. Sofialidis and Faltsi [29] studied on simulation of biomass gasificationin fludixed beds using CFD approach. Although CFD is a powerful software, its programs have high computational requirements. On the other hand, Aspen Plus, a familiar, proven and acceptable processes modeling software, used in the fields of SW and coal gasification, oil industry and others. It is a powerful process modeling tool which offers to include customize user models to embed with built in Aspen reactor blocks. Since it contains a large property database for conventional compounds and convergence algorithms for solving minimization problems, Aspen Plus is selected to develop a gasification model. Many researchers have used Aspen Plus to develop gasification and downstream models for SW, coal and other biomass [3–6]. The SW gasification model can be separated in two ways according to Lu et al. [30]:
- -
Kinetic model: is capable of simulating the reaction conditions at different times and sites which is suitable for reactor amplification design and operation parameters optimisation.
- -
Equilibrium model: predicts only end reaction product distribution but no idea is provided about instantaneous product distribution along with geometric dimensions.
In this study, both reaction kinetics parameters and bed hydrodynamics aspects are considered to develop the model. The development of a fluidized bed gasification model through Aspen Plus involves the following steps:
- -
steam class specification;
- -
property method selection;
- -
system component specification (from databank) and identifying conventional and non-conventional components;
- -
defining the process flowsheet (using unit operation blocks and connecting material and energy streams);
- -
specifying feed streams (flow rate, composition and thermodynamic condition), and
- -
specifying unit operation blocks (thermodynamic condition, chemical reactions, etc.).
Aspen Plus lacks a built-in library of models with which to develop a customized fluidized bed gasification model. However, Aspen Plus provides the facility for the user to input their own models using FORTRAN/Excel codes and reactions nested within the input file.
3.7. Model Description
The fluidized bed gasification model is comprised of a number of Aspen reactor blocks. In order to show the overall gasification process, there are different phases considered in the Aspen Plus simulation, these being drying, pyrolysis (decomposition), volatile reactions, char gasification, and gas-solid separation. The process flowchart and an Aspen Plus simulation flowsheet of biomass gasification are shown in Figures 3 and 4, respectively.
Feed (SW), dry-feed and ash are specified as a non-conventional component in Aspen Plus and defined in the simulation model by using the ultimate and proximate analysis. The characteristics of feed are shown in Table 3.
The input parameters of the corresponding gasifier operating conditions were similar to experimental measurements, are given in Table 4. Methods and processes used for developing the Aspen Plus model is briefly described below.
3.7.1. Physical Property Method
The IDEAL property method was set for this simulation in which ideal behaviors are assumed, such as systems at vacuum pressures and isomeric systems at low pressures. In the vapor phase, small deviations from the ideal gas law are allowed. These deviations occur at low pressures (either below atmospheric pressure, or at pressures not exceeding 2 bar) and very high temperatures. Ideal behavior in the liquid phase is exhibited by molecules with either very small interactions or interactions that cancel each other out. The IDEAL property method is generally used for systems with and without non-condensable components. In this method, permanent gases can be dissolved in the liquid [31].
The steam class was set as MIXED, NC and PSD (MIXNCPSD). MIXNCPSD represents that, nonconventional solids are present, with a particle size distribution. The NC properties: Enthalpy and Density model was selected as HCOALGEN and DCOALIGT, respectively, for both feed, dry-feed and ash which are non-conventional components. HCOALGEN is the general coal/SW model for computing enthalpy in the Aspen Physical Property System which includes a number of different correlations for: heat of combustion, heat of formation and heat capacity. The density model, DCOALIGT, gives the true (skeletal or solid-phase) density of coal/SW on a dry basis using ultimate and sulfur analyses.
3.7.2. Model Sequence
A number of Aspen Plus blocks were used to complete the overall gasification process. The main processes were simulated by three reactors in Aspen Plus: RYield, RGibbs and RCSTR. Additionally, a MIXER and a number of SEPERATOR blocks were incorporated in the simulation model to complete the entire process. The whole gasification consists of four processes, namely drying, decomposition, volatile reaction and char gasification and combustion.
3.7.3. Drying
Reducing moisture from feed plays important role to improve gasifier performance. An Aspen Plus block, RYield (block ID: DRYER in Figure 4) was used to simulate the drying process of SW. The SW is fed into the block, and the water bound in SW is vaporized in this block. The yield of gaseous water is determined by the water content in the proximate analysis of SW. The moisture content of the SW is 25%, therefore, the mass yield of gaseous water is set as 25%, based on the assumption that the physically bound water is vaporized completely in this process. The mass yield of dried SW is correspondingly equal to 100% − 25% = 75%. After the drying process, the gaseous water and dried SW flow into a gas and solid separator, SEP0. The separated gaseous water is drained out from the process and the separated dried SW goes on to the next block for the decomposition of dried feed.
3.7.4. Decomposition
RYield (block ID: DCOMP in Figure 4), an Aspen Plus yield reactor, was used to decompose the feed in the simulation. RYield is used when reaction stoichiometry and reaction kinetics are unknown or unimportant but the component yield distribution is known. In this step, SW is converted into its constituent components H2, O2, C, sulphur (S), N2 and ash by specifying the yield distribution according to the feed's ultimate analysis.
3.7.5. Volatile Reactions
An Aspen Plus reactor, RGibbs (block ID: GASIFY in Figure 4), uses Gibbs free energy minimization with phase splitting to calculate equilibrium. This reactor does not require specifying the reaction stoichiometry, but reactor temperature and pressure is known from experiment. RGibbs is capable of calculating the chemical equilibrium between any number of conventional solid components and the fluid phases [32].
In this study, RGibbs was used for volatile combustion. SW mainly consists of C, O2, H2, N2, chlorine (Cl), S, moisture and ash. Here, C will partly compose the gas phase to take part in de-volatilization and the remaining part of C comprises the solid phase (char) and consequently results in char gasification. A separator was used before RGibbs reactor to separate the volatile materials and solids from the decomposed components. RGibbs reactor performs the volatile reactions of separated volatile materials.
3.7.6. Char Gasification
RCSTR, one of the vital reactors in Aspen Plus, rigorously models continuously stirred tank reactors. This reactor is capable of modelling one, two or three phase reactors. RCSTR assumes perfect mixing in the reactor, that is, the reactor contents have the same properties and composition as the outlet stream. This reactor handles kinetic and equilibrium reactions as well as reactions involving solids. The user can provide the reaction kinetics through the built-in Reactions models or through a user-defined FORTRAN/Excel subroutine [32].
In this study, the reactor, RCSTR (block ID: BED and FREEBRD in Figure 4) was used to perform char gasification using reaction kinetics as discussed previously. The reaction kinetics were integrated by written FORTRAN and Excel code via the CALCULATOR block. The reactor was divided into bed and freeboard regions using hydrodynamic parameters where each region is simulated by one RCSTR reactor. Using FORTRAN code, each RCSTR is divided into a series of CSTR reactors with equal volume. Bed and Freeboard reactors require matching a number of variables, some specified at the top and others at the bottom of the gasifier. This feature causes the solution process to be usually complicated and time-consuming. Therefore, from the viewpoint of directly using the built-in algorithm in Aspen Plus and then simplifying the problem, a number of RCSTR reactors in series are used to model the char gasification and combustion processes. The RCSTR reactor has the characteristic that all phases have the same temperature, which means the temperatures of solid and gas phases in the char gasification and combustion processes are equal in the model. The kinetic and hydrodynamic parameters such as fractional pressure, superficial velocity and voidage of O2 and steam remain constant in these reactors.
3.7.7. Solid Separation
A splitter block, CYCLONE, was used in this model to separate C solid from the gas mixture provided by the split fraction of MIXED and NCPSD.
4. Results and Discussions
4.1. Model Validation
This section presents a comparison of simulation results with experimentally measured data using a pilot scale gasifier shown in Figure 1. The operating conditions of the gasifier model were similar to the experimental measurement as shown in Table 4. The composition of raw syngas, such as H2, CO, CO2, CH4, N2 and H2O, was measured experimentally and is compared in Table 5 with the simulation results. It can be clearly seen from Table 5 that there is only about 3% variation between measured and simulated results. Therefore, it is fair to say that the developed model can be used for further analysis with acceptable accuracy. The validated model was used to study the effect of air-fuel ratio and steam-fuel ratio on syngas composition as discussed below.
4.2. Simulation Model Analysis
4.2.1. Effect of Air-Fuel Ratio
The effect of air-fuel ratio on product gas composition was examined. Simulation results for syngas composition (in percent) versus air-fuel ratios in the range of 0.5 to 1.0 are shown in Figure 5. The production of both H2 and CO decreases with the increasing amount of air, while the volume of the inert gas N2 in the syngas increases. The composition of CO2 decreases with very small deviation and CH4 remains almost the same with increasing air-fuel ratio. Due to the high concentration of N2 in air, N2 concentration increases in syngas composition with increasing air supply.
Air-fuel ratio not only represents the O2 quantity introduced into the reactor, but also affects the gasification temperature under the condition of auto thermal operation. Higher air-fuel ratios can cause syngas quality to degrade because of an increased oxidation reaction. Alternatively, higher air-fuel ratios mean a higher gasification temperature which can accelerate the gasification and improve the product quality to a certain extent. The oxidation reaction for CO production is:
The oxidation reaction for CO2 production is:
Based on the oxidation reactions, Equations (17) and (18), CO production consumes more C for the same amount of O2.
4.2.2. Effect of Steam-Fuel Ratio
The effect of steam-fuel ratio in the range of 4–10 on syngas composition is shown in Figure 6. The concentration of CO and H2 exhibits a trend that slowly increases when the steam to fuel ratio increases. This can be explained by more steam reforming reactions of CO and H2 taking place because of increased steam quantity. The change in CH4 and CO2 concentration is very small with increasing steam-fuel ratio. The C-steam reaction is highly temperature sensitive, as an increase in low steam flows cause the efficiency to increase slightly in the experiment, but it then drops for additional rises in the steam flow amount. The concentration of N2 composition shows a significant decrease (from 20% to 13%) with increasing steam-fuel ratio as there is only a fixed amount of air supplied with increasing steam-fuel ratio.
4.2.3. Effect of Gasifier Temperature
The effect of gasifier temperature on produced syngas composition is shown in Figure 7. The temperature considered varies from 600 °C to 1000 °C. The concentration of syngas components (i.e., CO, H2, CO2, CH4 and N2) varying with a small range with increasing gasifier temperature. There are about 28%, 20%, 15%, 11% and 2% of CO, H2, N2, CO2 and CH4 produced, respectively. The overall gasification process, along with C conversion, is improved by increases in gasification temperature. The rate of increase in C conversion efficiency becomes slow at higher gasification temperatures.
In lower gasifier temperature, SW produces more unburned hydrocarbon and tar, which decreases H2 production. In Figure 7, the higher amount of H2 favors the backward reaction and causes prediction of lower CO2 production in simulation related to the reaction in Equation (6). The trend of syngas composition in simulation is following the same trend as literature [4].
5. Conclusions
Experimental and numerical investigations of atmospheric fluidized bed gasification have been done for SW. The experimental investigation was performed using a pilot-scale gasifier plant. An Aspen Plus simulation model was developed based on the experimental setup and findings. To develop the simulation model, several Aspen Plus reactor blocks were used with a number of separators and a mixer block. Reaction kinetics and hydrodynamic equations were incorporated through FORTRAN and Excel code. The simulation model was validated with experimental results of a pilot scale SW gasification plant. A very good agreement was found between simulation and experimental results, with a maximum variation of 3%. The effects of air-fuel and steam-fuel ratio on syngas composition were simulated using the validated model. The model will assist researchers, professionals and industry people to identify the optimized conditions for SW gasification.
Acknowledgments
The experimental investigation was supported by The Corky's Group: Corkys Carbon and Combustion.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Stucley, C.; Schuck, S.; Sims, R.; Larsen, P.; Turvey, N.; Marino, B. Biomass Energy Production in Australia, revised ed.; Rural Industries Research and Development Corporation: Canberra, Australia, 2004. [Google Scholar]
- Dopita, M.; Williamson, R. Australia's Renewable Energy Future; Australian Academy of Science: Canberra, Australia, 2010. [Google Scholar]
- Doherty, W.; Reynolds, A.; Kennedy, D. The effect of air preheating in a biomass CFB gasifier using ASPEN Plus simulation. Biomass Bioenergy 2009, 33, 1158–1167. [Google Scholar]
- Nikoo, M.B.; Mahinpey, N. Simulation of biomass gasification in fluidized bed reactor using ASPEN PLUS. Biomass Bioenergy 2008, 32, 1245–1254. [Google Scholar]
- Kumar, A.; Noureddini, H.; Demirel, Y.; Jones, D.D.; Hanna, M.A. Simulation of corn stover and distillers grains gasification with Aspen Plus. Trans. ASABE 2009, 52, 1989–1995. [Google Scholar]
- Abdelouahed, L.; Authier, O.; Mauviel, G.; Corriou, J.P.; Verdier, G.; Dufour, A. Detailed modeling of biomass gasification in dual fluidized bed reactors under Aspen Plus. Energy Fuels 2012, 26, 3840–3855. [Google Scholar]
- Mavukwana, A.; Jalama, K.; Ntuli, F.; Harding, K. Simulation of Sugarcane Bagasse Gasification using Aspen Plus. Presented at the International Conference on Chemical and Environmental Engineering (ICCEE), Johannesburg, South Africa, 15–16 April 2013.
- Francois, J.; Abdelouahed, L.; Mauviel, G.; Patisson, F.; Mirgaux, O.; Rogaume, C.; Rogaume, Y.; Feidt, M.; Dufour, A. Detailed process modeling of a wood gasification combined heat and power plant. Biomass Bioenergy 2013, 51, 68–82. [Google Scholar]
- Gasifier Operator Manual; The Corky's Group: Mayfield, Australia, 2010.
- Sadaka, S.S.; Ghaly, A.E.; Sabbah, M.A. Two phase biomass air-steam gasification model for fluidized bed reactors: Part I—Model development. Biomass Bioenergy 2002, 22, 439–462. [Google Scholar]
- Lv, P.M.; Xiong, Z.H.; Chang, J.; Wu, C.Z.; Chen, Y.; Zhu, J.X. An experimental study on biomass air–steam gasification in a fluidized bed. Bioresour. Technol 2004, 95, 95–101. [Google Scholar]
- Buekens, A.G.; Schoeters, J.G. Modelling of Biomass Gasification. In Fundamentals of Thermochemical Biomass Conversion; Overend, R.P., Milne, T.A., Mudge, K.L., Eds.; Elsevier Applied Science Publishers: London, UK, 1985; pp. 619–689. [Google Scholar]
- Ergudenler, A. Gasification of Wheat Straw in a Dual-Distributor Type Fluidized Bed Reactor. In Ph.D. Thesis; Technical University of Nova: Scotia, Halifax, NS, Canada, 1993. [Google Scholar]
- Lee, J.M.; Kim, Y.J.; Lee, W.J.; Kim, S.D. Coal-gasification kinetics derived from pyrolysis in a fluidized-bed reactor. Energy 1998, 23, 475–488. [Google Scholar]
- Liu, G.-S.; Niksa, S. Coal conversion submodels for design applications at elevated pressures. Part II. Char gasification. Prog. Energy Combust. Sci 2004, 30, 679–717. [Google Scholar]
- Montagnaro, F.; Salatino, P. Analysis of char-slag interaction and near-wall particle segregation in entrained-flow gasification of coal. Combust. Flame 2010, 157, 874–883. [Google Scholar]
- Matsui, I.; Kunii, D.; Furusawa, T. Study of fluidized bed steam gasification of char by thermogravimetrically obtained kinetics. J. Chem. Eng. Jpn 1985, 18, 105–113. [Google Scholar]
- Rajan, R.R.; Wen, C.Y. A comprehensive model for fluidized bed coal combustors. AIChE J 1980, 26, 642–655. [Google Scholar]
- Walker, P.L.J.; Rusinko, F.J.; Austin, L.G. Gas reactions of carbon. Adv. Catal 1959, 11, 133–221. [Google Scholar]
- Dutta, S.; Wen, C.Y. Reactivity of coal and char 2. In oxygen-nitrogen atmosphere. Ind. Eng. Chem. Process Des. Dev 1977, 16, 31–36. [Google Scholar]
- Kasaoka, S.; Skata, Y.; Tong, C. Kinetic evaluation of the reactivity of various coal chars for gasification with carbon dioxide in comparison with steam. Ind. Eng. Chem 1985, 25, 160–175. [Google Scholar]
- Chin, G.; Kimura, S.; Tone, S.; Otake, T. Gasification of coal char with steam. Part 2. Pore structure and reactivity. Ind. Eng. Chem 1983, 23, 113–120. [Google Scholar]
- Athar, M. Simulation of Coal Gasification in Circulating Fludized Bed (CFB) Reactor. Master's Thesis, University of Engineering & Technology, Lahore, Pakistan, 2009. [Google Scholar]
- Kunii, D.; Levenspiel, O. Fluidization Engineering, 2nd ed.; Butterworth-Heinemann: Newton, MA, USA, 1991. [Google Scholar]
- Babu, S.P.; Shah, B.; Talwalker, A. Fluidization correlations for coal gasification materials-minimum fluidization velocity and bed expansion ratio. AIChE Symp. Ser 1978, 74, 176–186. [Google Scholar]
- Yan, H.M.; Heidenreichayb, C.; Zhanga, D.K. Mathematical modelling of a bubbling fluidized-bed coal gasifier and the significance of “net flow”. Fuel 1998, 77, 1067–1079. [Google Scholar]
- Lewis, W.K.; Gilliland, E.R.; Lang, P.M. Entrainment from fluidized beds. Chem. Eng. Prog. Symp. Ser 1962, 58, 65–78. [Google Scholar]
- Mhilu, C.F. Modeling performance of high-temperature biomass gasification process. ISRN Chem. Eng. 2012, 2012. [Google Scholar] [CrossRef]
- Sofialidis, D.; Faltsi, O. Simulation of biomass gasification in fluidized beds using computational fluid dynamics approach. Therm. Sci 2001, 5, 95–105. [Google Scholar]
- Lü, P.; Kong, X.; Wu, C.; Yuan, Z.; Ma, L.; Chang, J. Modeling and simulation of biomass air-steam gasification in a fluidized bed. Front. Chem. Eng. China 2008, 2, 209–213. [Google Scholar]
- Aspen Plus, Ed.; Aspen Physical Property System V7.2. Available online: http://www.aspentech.com (accessed 2 August 2013).
- Aspen Plus. Aspen Plus User Models V7.3. Available online: http://support.aspentech.com/Public/Documents/Engineering/Aspen%20Plus/V7.3/AspenPlusUserModelsV7_3-Ref.pdf (accessed 2 August 2013).
Nomenclature
| Ar | Archimedes number |
| E | Activation energy (kcal/mol) |
| g | Gravitational acceleration (m/s2) |
| k | Rate constant (s−1 atm−1) |
| MC | Molecular weight of carbon (C) (kg/kmol) |
| P | Pressure (MPa) |
| R | Universal gas constant (kcal/mol K) |
| T | Temperature (K) |
| XCO | C conversion due to combustion |
| XSG | C conversion due to steam gasification |
| YC | Volume fraction of C in solid |
| a | Decay constant of clusters in freeboard (m−1) |
| u | Superficial velocity (m/s) |
| t | Time (s) |
| z | Distance above the surface of the bed (m) |
| dp | Particle diameter (mm) |
| rc | Reaction rate of C (kmol m3 s) |
| umf | Minimum fluidisation velocity (m/s) |
| Greek Symbols | |
|---|---|
| α | Kinetic parameter |
| β | Kinetic parameter |
| εb | Volume fraction of bed occupied by bubble |
| εf | Average voidage of bed |
| εs | Volume fraction of solid in bed |
| εfb | Average voidage of freeboard |
| εmf | Voidge in emulsion at minimum fluidisation |
| ρs | Density of solid (kg m3) |
| ρc | Density of C (kg m3) |
| ρg | Density of gas (kg m3) |
| μ | Viscosity (kg/m s) |






| Compound | Raw syngas (%) | Scrubbed syngas (%) | Dewatered syngas (%) |
|---|---|---|---|
| Hydrogen (H2) | 19.0 | 20.0 | 20.8 |
| Carbon dioxide (CO2) | 11.4 | 12.0 | 12.5 |
| Carbon monoxide (CO) | 28.5 | 30.0 | 31.3 |
| Methane (CH4) | 1.9 | 2.0 | 2.1 |
| Water or steam (H2O) | 5.7 | 6.0 | 2.1 |
| Nitrogen (N2) | 28.5 | 30 | 31.3 |
| Phenol, cresol, tars, hydrogen sulphide (H2S) and ammonia (NH3) | 5 | - | - |
| Process | E/R (K) | k (s−1 atm−1) |
|---|---|---|
| Combustion | 13,523 | 0.046 |
| Gasification with steam | 19,544 | 6,474.7 |
| Moisture content (MC) (%) | 25 | |
| Proximate analysis (mass %) (Dry basis) | Volatile matter (VM) | 82.6 |
| Fixed carbon (FC) | 16.3 | |
| Ash | 1.1 | |
| Ultimate Analysis (mass %) | Carbon (C) | 49.8 |
| H2 | 6.1 | |
| Oxygen (O2) | 33.9 | |
| N2 | 0.2 | |
| Sulphur (S) | 0.1 | |
| Average particle size (mm) | 0.25–0.95 | |
| Char density (kg/m3) | 1400 | |
| Gross specific energy (MJ/kg) | 18.6 | |
| Feed | Flow rate (kg/h) | 4.5 |
| Pressure (MPa) | 0.3 | |
| Temperature (°C) | 25 | |
| Air | Flow rate (kg/h) | 4.5 |
| Pressure (MPa) | 0.3 | |
| Temperature (°C) | 350 | |
| Air/fuel ratio | 1 | |
| Steam | Flow rate (kg/h) | 27 |
| Pressure (MPa) | 0.3 | |
| Temperature (°C) | 200 | |
| Steam/fuel ratio | 6 | |
| Reaction vessel volume | Total (m3) | 2.5 |
| Waste/char capacity (m3) | 1.75 | |
| Freeboard capacity (m3) | 0.75 | |
| Gasifier | Pressure (MPa) | 0.3 |
| Temperature (°C) | 700–1100 | |
| Dryer | Pressure (MPa) | 0.3 |
| Temperature (°C) | 400 | |
| Decomposition | Pressure (MPa) | 0.3 |
| Temperature (°C) | 400 |
| Measurement | H2 | CO | CO2 | CH4 | N2 | H2O | Others |
|---|---|---|---|---|---|---|---|
| Experimental (%) | 19 | 28.5 | 11.4 | 1.9 | 28.5 | 5.7 | 5.0 |
| Model (%) | 17.37 | 25.49 | 10.55 | 2.44 | 27.29 | 9.2 | 7.65 |
| Difference (%) | −1.63 | −3.01 | −0.85 | 0.54 | −1.21 | 3.5 | 2.65 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Begum, S.; Rasul, M.G.; Akbar, D.; Cork, D. An Experimental and Numerical Investigation of Fluidized Bed Gasification of Solid Waste. Energies 2014, 7, 43-61. https://doi.org/10.3390/en7010043
Begum S, Rasul MG, Akbar D, Cork D. An Experimental and Numerical Investigation of Fluidized Bed Gasification of Solid Waste. Energies. 2014; 7(1):43-61. https://doi.org/10.3390/en7010043
Chicago/Turabian StyleBegum, Sharmina, Mohammad G. Rasul, Delwar Akbar, and David Cork. 2014. "An Experimental and Numerical Investigation of Fluidized Bed Gasification of Solid Waste" Energies 7, no. 1: 43-61. https://doi.org/10.3390/en7010043
APA StyleBegum, S., Rasul, M. G., Akbar, D., & Cork, D. (2014). An Experimental and Numerical Investigation of Fluidized Bed Gasification of Solid Waste. Energies, 7(1), 43-61. https://doi.org/10.3390/en7010043






