Rapid Biogas Production by Compact Multi-Layer Membrane Bioreactor: Efficiency of Synthetic Polymeric Membranes
Abstract
:1. Introduction
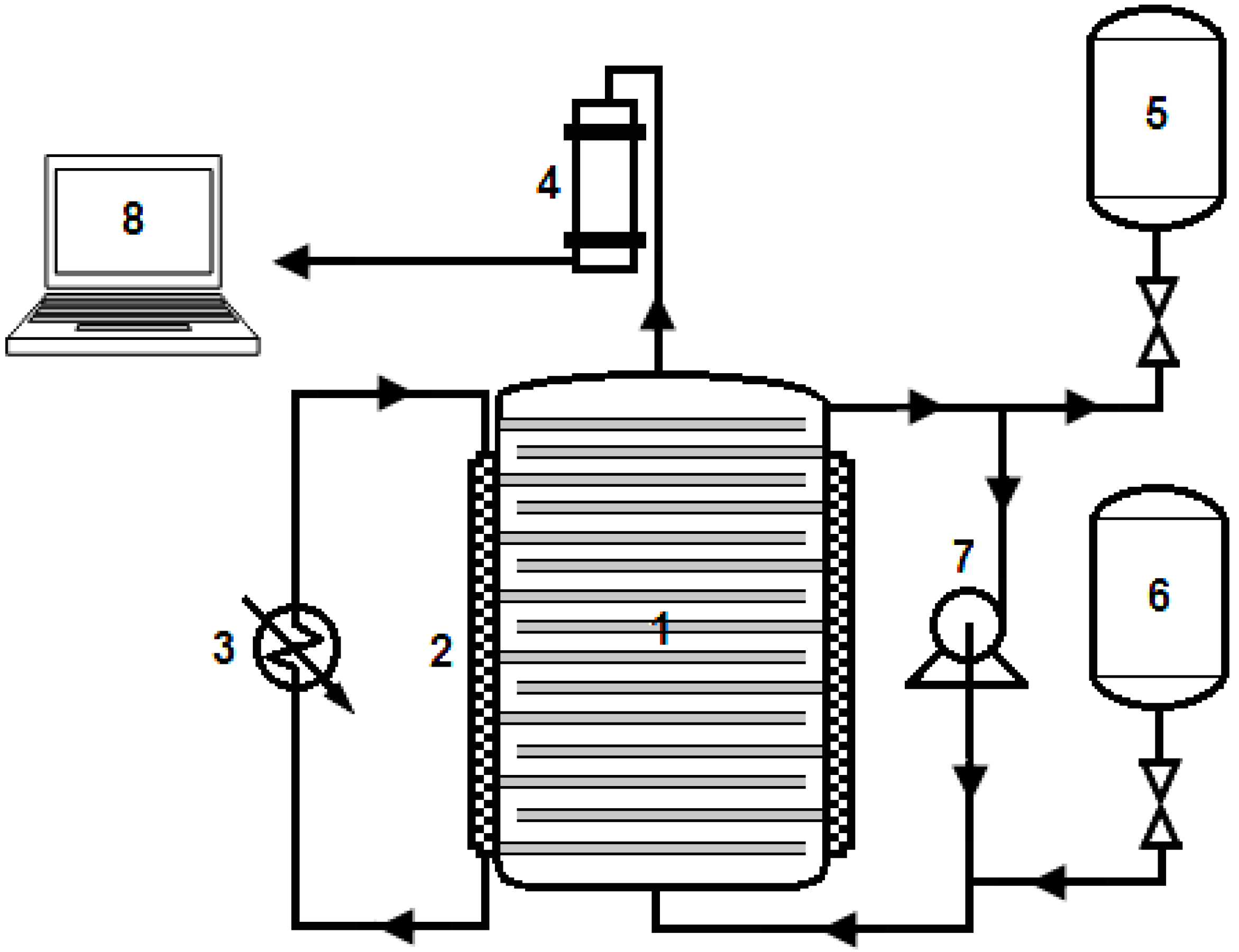
2. Results and Discussion
2.1. Biogas Production Efficacy of the Developed Compact Multi-Layer Membrane Bioreactor (MMBR)
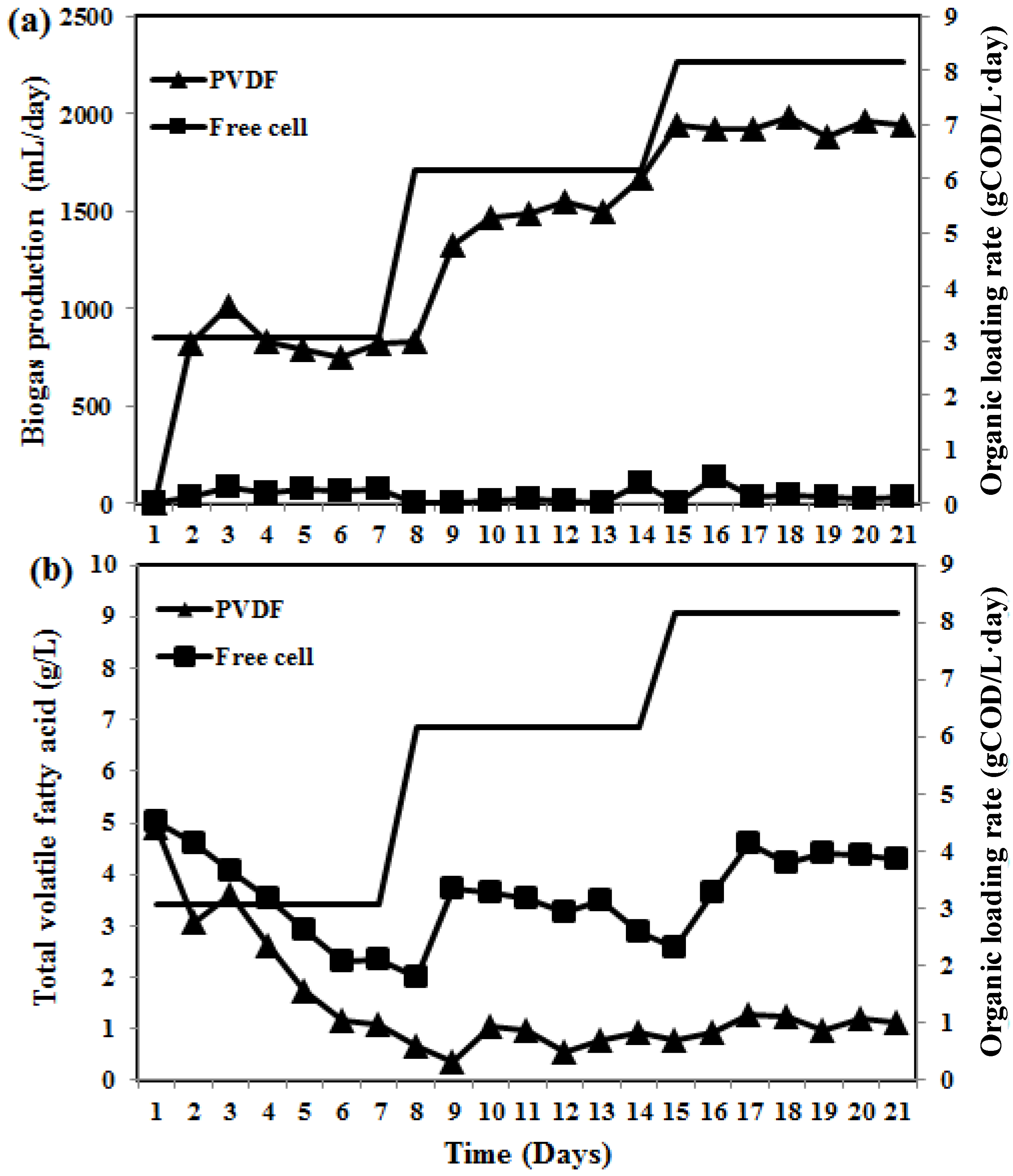
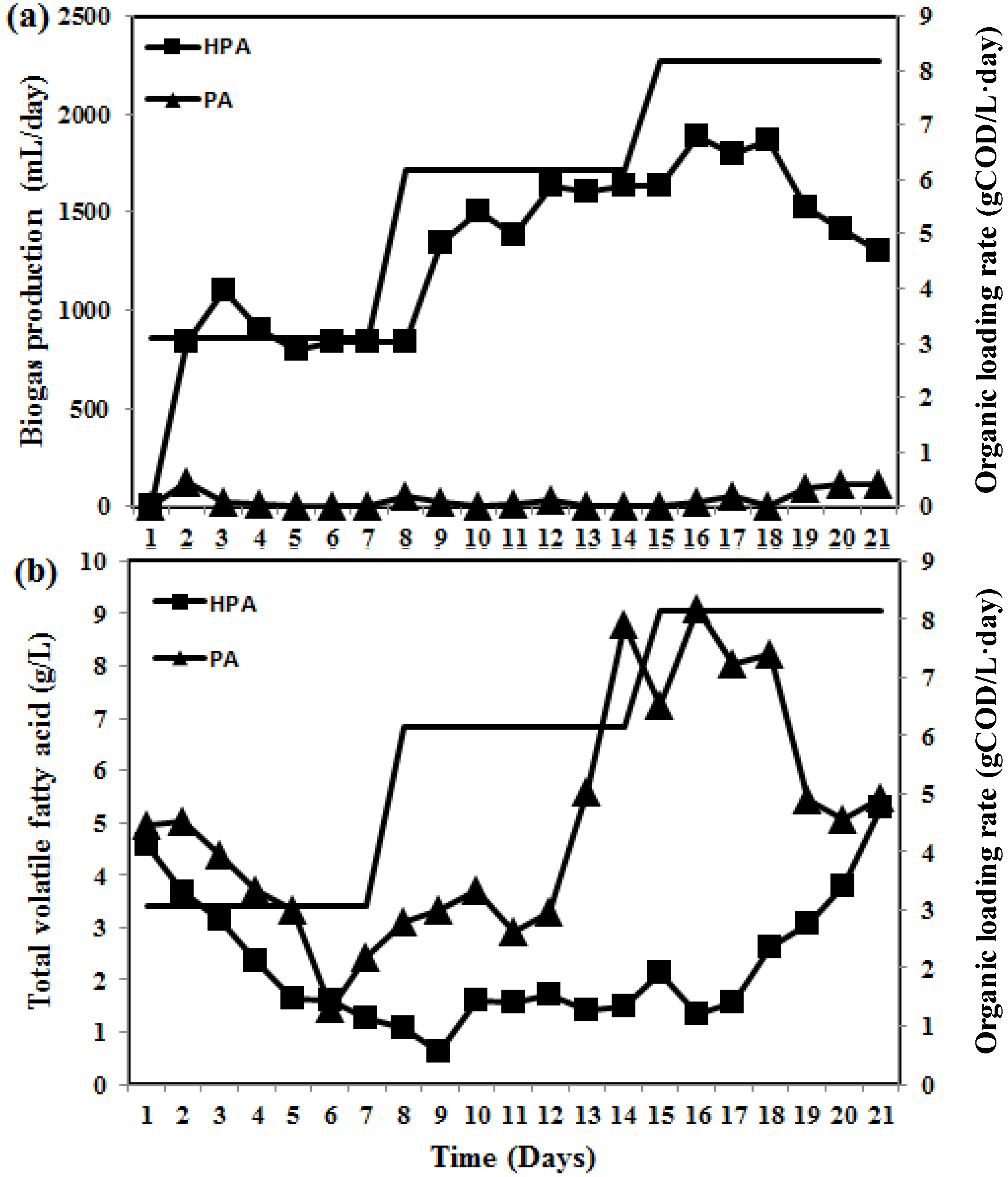
2.2. Characteristics of the Developed Synthetic Membranes and Their Efficacy for Enhancing Biogas Productivity
| Membrane | Pore size (nm) | Bubble point at 20 °C (psi) | Flux at 1.0 bar and 20 °C (L/m2·h) | Water uptake at 20 °C (g/g) | Porosity (%) | Thickness (mm) |
|---|---|---|---|---|---|---|
| PVDF | 110 | 155 | 13.36 | 0.093 | 10.551 | 0.125 |
| HPA | 70 | 173 | 1.94 | 0.101 | 7.616 | 0.13 |
| PA | 280 | 43 | 0.389 | 0.051 | 9.642 | 0.11 |


3. Experimental
3.1. Anaerobic Culture and Medium
3.2. Synthetic Membranes Preparation
3.3. Semi-Continuous Anaerobic Digestion Process
| Organic loading rate (OLR) (g COD/L·day) | Working volume of reactor (mL) | Synthetic medium strength (g COD/L) | Flow rate of medium (mL/day) |
|---|---|---|---|
| 3.08 | 800 | 9.33 | 264 |
| 6.16 | 800 | 9.33 | 528 |
| 8.16 | 800 | 9.33 | 700 |
3.4. Analytical Methods
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Deublein, D.; Steinhauser, A. Biogas from Waste and Renewable Resources; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 49–50. [Google Scholar]
- Gerardi, M.H. The Microbiology of Anaerobic Digesters; John Wiley & Sons Inc: Hoboken, NJ, USA, 2003. [Google Scholar]
- Huelgas, A.; Funamizu, N. Flat-plate submerged membrane bioreactor for the treatment of higher-load graywater. Desalination 2010, 250, 162–166. [Google Scholar] [CrossRef]
- Das, D. Advances in biohydrogen production processes: An approach towards commercialization. Int. J. Hydrog. Energy 2009, 34, 7349–7357. [Google Scholar] [CrossRef]
- Gassanova, L.G.; Netrusov, A.I.; Teplyakov, V.V.; Modigell, M. Fuel gases from organic wastes using membrane bioreactors. Desalination 2006, 198, 56–66. [Google Scholar] [CrossRef]
- Kumar, N.; Das, D. Continuous hydrogen production by immobilized Enterobacter cloacae IIT-BT 08 using lignocellulosic materials as solid matrices. Enzym. Microb. Technol. 2001, 29, 280–287. [Google Scholar] [CrossRef]
- Show, K.-Y.; Lee, D.-J.; Chang, J.-S. Bioreactor and process design for biohydrogen production. Bioresour. Technol. 2011, 102, 8524–8533. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-Y.; Li, Y.-Y.; Noike, T. Influence of solids retention time on continuous H2 production using membrane bioreactor. Int. J. Hydrog. Energy 2010, 35, 52–60. [Google Scholar] [CrossRef]
- Lee, D.-Y.; Li, Y.-Y.; Noike, T. Continuous H2 production by anaerobic mixed microflora in membrane bioreactor. Bioresour. Technol. 2009, 100, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Jeong, E.; Kim, H.-W.; Nam, J.-Y.; Shin, H.-S. Enhancement of bioenergy production and effluent quality by integrating optimized acidification with submerged anaerobic membrane bioreactor. Bioresour. Technol. 2010, 101, S7–S12. [Google Scholar] [CrossRef] [PubMed]
- Padmasiri, S.I.; Zhang, J.; Fitch, M.; Norddahl, B.; Morgenroth, E.; Raskin, L. Methanogenic population dynamics and performance of an anaerobic membrane bioreactor (AnMBR) treating swine manure under high shear conditions. Water Res. 2007, 41, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.R. Membrane Technology and Applications; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2000. [Google Scholar]
- Judd, S.; Judd, C. The MBR Book—Priciples and Applications of Membrabe Bioreactors for Water and Waste Water Treatment, 2nd, ed.; Elsevier Ltd.: Burlington, MA, USA, 2011; p. 519. [Google Scholar]
- Chen, H.C. Up-flow attached-bed bioreactor for continuous ethanol fermentation. Biotechnol. Prog. 1991, 7, 311–314. [Google Scholar] [CrossRef]
- Joshi, S.; Yamazaki, H. Film fermentor for ethanol production by yeast immobilized on cotton cloth. Biotechnol. Lett. 1984, 6, 797–802. [Google Scholar] [CrossRef]
- Shiratori, H.; Ikeno, H.; Ayame, S.; Kataoka, N.; Miya, A.; Hosono, K.; Beppu, T.; Ueda, K. Isolation and characterization of a new Clostridium sp. that performs effective cellulosic waste digestion in a thermophilic methanogenic bioreactor. Appl. Environ. Microbiol. 2006, 72, 3702–3709. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.-D.; Eberiel, D.T.; McCarthy, S.P.; Gross, R.A. Cellulose acetate biodegradability upon exposure to simulated aerobic composting and anaerobic bioreactor environments. J. Environ. Polymer Degrad. 1993, 1, 143–153. [Google Scholar] [CrossRef]
- Youngsukkasem, S.; Rakshit, K.S.; Taherzadeh, M.J. Biogas production by encapsulated methane producing bacteria. Bioresources 2012, 7, 56–65. [Google Scholar]
- Youngsukkasem, S.; Akinbomi, J.; Rakshit, S.K.; Taherzadeh, M.J. Biogas production by encased bacteria in synthetic membranes: Protective effects in toxic media and high loading rates. Environ. Technol. 2013, 34, 1–8. [Google Scholar] [CrossRef] [PubMed]
- García, J.M.; García, F.C.; Serna, F.; de la Peña, J.L. High-performance aromatic polyamides. Prog. Polymer Sci. 2010, 35, 623–686. [Google Scholar] [CrossRef]
- Gabelich, C.J.; Frankin, J.C.; Gerringer, F.W.; Ishida, K.P.; Suffet, I.H. Enhanced oxidation of polyamide membranes using monochloramine and ferrous iron. J. Membr. Sci. 2005, 258, 64–70. [Google Scholar] [CrossRef]
- Espeso, J.; Lozano, A.E.; de la Campa, J.G.; de Abajo, J. Effect of substituents on the permeation properties of polyamide membranes. J. Membr. Sci. 2006, 280, 659–665. [Google Scholar] [CrossRef]
- Jin, Y.; Su, Z. Effects of polymerization conditions on hydrophilic groups in aromatic polyamide thin films. J. Membr. Sci. 2009, 330, 175–179. [Google Scholar] [CrossRef]
- Louie, J.S.; Pinnau, I.; Reinhard, M. Effects of surface coating process conditions on the water permeation and salt rejection properties of composite polyamide reverse osmosis membranes. J. Membr. Sci. 2011, 367, 249–255. [Google Scholar] [CrossRef]
- Mitrouli, S.T.; Karabelas, A.J.; Isaias, N.P.; Al Rammah, A.S. Application of hydrophilic macromolecules on thin film composite polyamide membranes for performance restoration. Desalination 2011, 278, 105–116. [Google Scholar] [CrossRef]
- Shin, D.H.; Kim, N.; Lee, Y.T. Modification to the polyamide TFC RO membranes for improvement of chlorine-resistance. J. Membr. Sci. 2011, 376, 302–311. [Google Scholar] [CrossRef]
- Isci, A.; Demirer, G.N. Biogas production potential from cotton wastes. Renew. Energy 2007, 32, 750–757. [Google Scholar] [CrossRef]
- Lhommeau, C.; Toillon, S.; Pith, T.; Kessler, L.; Jesser, C.; Pinget, M. Polyamide 4,6 membranes for the encapsulation of Langerhans islets: Preparation, physico-chemical properties and biocompatibility studies. J. Mater. Sci. Mater. Med. 1997, 8, 163–174. [Google Scholar] [CrossRef] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Youngsukkasem, S.; Barghi, H.; Rakshit, S.K.; Taherzadeh, M.J. Rapid Biogas Production by Compact Multi-Layer Membrane Bioreactor: Efficiency of Synthetic Polymeric Membranes. Energies 2013, 6, 6211-6224. https://doi.org/10.3390/en6126211
Youngsukkasem S, Barghi H, Rakshit SK, Taherzadeh MJ. Rapid Biogas Production by Compact Multi-Layer Membrane Bioreactor: Efficiency of Synthetic Polymeric Membranes. Energies. 2013; 6(12):6211-6224. https://doi.org/10.3390/en6126211
Chicago/Turabian StyleYoungsukkasem, Supansa, Hamidreza Barghi, Sudip K. Rakshit, and Mohammad J. Taherzadeh. 2013. "Rapid Biogas Production by Compact Multi-Layer Membrane Bioreactor: Efficiency of Synthetic Polymeric Membranes" Energies 6, no. 12: 6211-6224. https://doi.org/10.3390/en6126211
APA StyleYoungsukkasem, S., Barghi, H., Rakshit, S. K., & Taherzadeh, M. J. (2013). Rapid Biogas Production by Compact Multi-Layer Membrane Bioreactor: Efficiency of Synthetic Polymeric Membranes. Energies, 6(12), 6211-6224. https://doi.org/10.3390/en6126211







