High Lipid Induction in Microalgae for Biodiesel Production
Abstract
:1. Introduction
2. Lipids in Microalgae
3. Methods of Lipid Induction
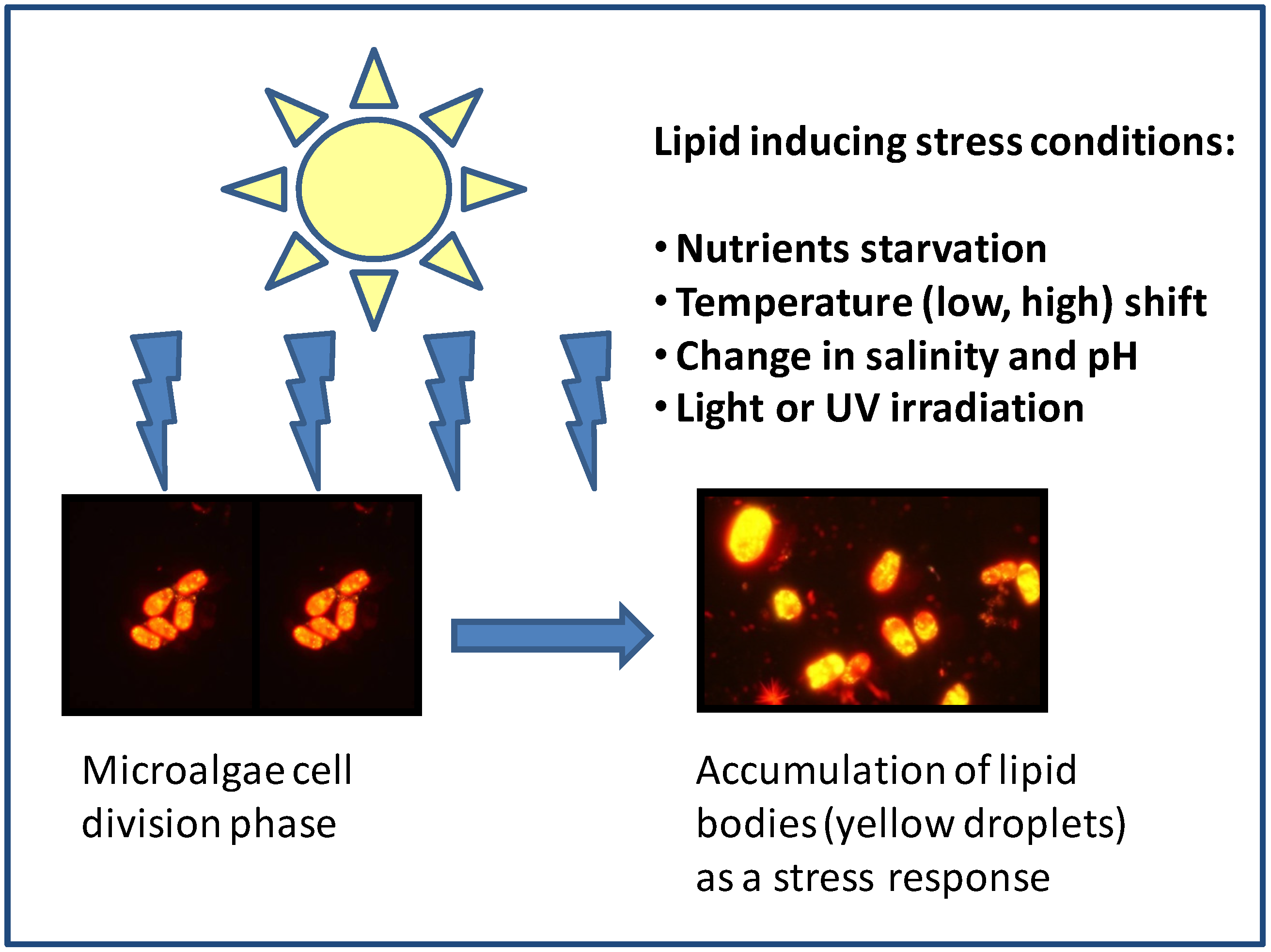
3.1. Nutrient Starvation
| Microalgae species or strain | Nutrient stress | Changes in lipid profile after induction | Reference |
|---|---|---|---|
| Chlamydomonas reinhardtii, Scenedesmus subspicatus | Nitrogen limitation | Increase in total lipids (lipid: amide ratio) | [42] |
| Nannochloropsis oculata | Nitrogen limitation | Total lipid increased by 15.31% | [43] |
| Chlorella vulgaris | Nitrogen limitation | Total lipid increased by 16.41% | [43] |
| Chlorella vulgaris | Nitrogen limitation | Lipid productivity of 78 mg/L d | [24] |
| Chlorella sp. | Nitrogen limitation | Lipid productivity of 53.96 ± 0.63 mg/L d | [25] |
| Phaeodactylum tricornutum | Nitrogen limitation | TAG levels increased from 69 to 75% | [29] |
| Dunaliella tertiolecta | Nitrogen limitation | Five times increase in lipid fluorescence | [44] |
| Chlorella vulgaris | Nitrogen medium | Lipids increased by 40% | [31] |
| Chlorella vulgaris | Nitrogen limitation | Increase in TAG | [35] |
| Chlorella sp. | Nutrient-deprived conditions (nitrogen, phosphate-potassium, iron, and all three combined) | Total lipid production of 49.16 ± 1.36 mg/L d | [25] |
| Chlorella sp. | Urea limitation | Total lipid productivity of 0.124 g/ L d | [26] |
| Neochloris oleoabundans | Ammonium nitrate | Lipid productivity of 0.133 g /L d | [45] |
| Scenedesmus sp., Coelastrella sp. | Combined effect of Ph and N-limitation | Increase in TAG | [46] |
| Phaeodactylum tricornutum , Chaetoceros sp., Isochrysis galbana | Phosphorus limitation | Increase in total lipids with higher relative content of 16:0 and 18:1 | [37] |
| Monodus subterraneus | Phosphorus limitation | Increase in TAG | [15] |
| Scenedesmus sp | Nitrogen and phosphorus starvation | Lipids increased 30% and 53%, respectively | [30] |
| Chlorella sp. | Silicon deficiency | - | [33] |
| Chlorella kessleri | Phosphorus limitation | Increase in unsaturated FAs | [38] |
| Chlamydomonas reinhardtii | Sulphur limitation | PG was increased by 2-fold | [17] |
| Chlamydomonas reinhardtii | Sulphur limition | Increase in TAG | [41] |
| Cyclotella cryptica | Silicon starvation | Increased in total lipids from 27.6% to 54.1% | [47] |
3.2. Temperature Stress
| Microalgae species or strain | Stressing agent | Lipid profile change after induction | Reference |
|---|---|---|---|
| Chaetoceros sp. | Grown at 25 °C | Total lipid increased by 16.8% | [49] |
| Rhodomonas sp., Cryptomonas sp., Isochrysis sp. | Range of 27 °C to 30 °C | Lipid production increased by 15.5, 12.7, and 21.7% respectively | [49] |
| Nannochloropsis oculata | Increase from 20 °C to 25 °C | Lipid production increased by 14.92% | [43] |
| Isochrysis galbana | Increase from 15 ° C to 30 ° C | Increase in neutral lipids | [59] |
| Chlorella ellipsoidea | Lowering temperature | Unsaturated FA was increased by 2-fold | [62] |
| Nannochloropsis salina | Increase in temperature | Increase in total lipids | [57] |
| Dunaliella salina | Shift from 30 °C to 12 °C | Increase in unsaturated lipids | [12] |
| Ochromonas danica | Increase from 15 °C to 30 °C | Increase in total lipids | [55] |
| Selenastrum capricornutum | Temperature from 25 °C to 10 °C | Increase in oleate fatty acid | [58] |
| Isochrysis galbana | Grown at 30 °C | Increase in total lipids | [59] |
| Phaeodactylum tricornutum | Shifted from 25 °C to 10 °C for 12 h | Highest yields of PUFA and EPA | [60] |
| Pavlova lutheri | Grown at 15 °C | Increased relative amount of EPA | [63] |
| Spirulina platensis, Chlorella vulgaris, Botryococcus braunii | Increase in temperature | Saturated FAs increased | [56] |
3.3. Salinity-Induced Lipid Production
3.4. The Effect of pH and Heavy Metals Stress
| Microalgae sp | Salinity change | Lipid profile change after induction | Reference |
|---|---|---|---|
| Dunaliella salina | Transferred from 29 to 205 g/L NaCl | Increased concentration of C18 FA | [66] |
| Dunaliella tertiolecta | Transferred from 29 g/L to 58 g/L NaCl | Increase in lipid content and TAG | [67] |
| Dunaliella sp. | Increased salinity from 23 to 234 g/L NaCl | Increase in total FA and monounsaturated FA | [68] |
| Nitzschia laevis | NaCl concentration increased from 10 to 20 g/L | Increase in unsaturated FA | [69] |
| Crythecodinium. cohnii ATCC 30556 | At 9 g/L NaCl | Increase in total FA content and DHA | [70] |
| Schizochytrium limacinum | Salinity at 9–36 g/L at temperature range of 16–30 °C | Saturated FA C15:0 and C17:0 was greatly increased | [71] |
| Unidentified Chlamydomonas sp. | Low pH | Increase in saturated FAs | [73] |
| Chlorella sp. | alkaline pH | Increase in TAG | [72] |
| Euglenia gracilis | Cadmium, copper, zinc | Increase in total lipids | [74] |
| Chlorella vulgaris | Fe3+ | Increase in total lipids to 56.6% of biomass | [34] |
3.5. Light Irradiation Stress
3.6. UV Irradiance for Lipid Induction
| Microalgae sp | Irradiation type | Lipid profile change after induction | Reference |
|---|---|---|---|
| Tichocarpus crinitus | Low light intensity | Increased TAG | [78] |
| Pavlova lutheri | High light intensities | Increased total lipid content | [81] |
| Thalassiosira pseudonana | Continuous or light/dark cycled strong light at exponential growth | Increased PUFA | [77] |
| Thalassiosira pseudonana | Continuous or light/dark cycled strong light at stationary phase | Increased TAG | [77] |
| Unidentified diatoms | Low light (2 µmol photons m−2 s−1) | 50% more MGDG | [82] |
| Selenastrum capricornutum | Dark treatment | Increase in linoleate FA | [58] |
| Prorocentrum minimum | Dark treatment | Marginal increase in phospholipids | [58] |
| Isochrysis galbana | Shorter light period | Increase of PUFA | [83] |
| Nannochloropsis oculata | UV-A | Increase of PUFA, structural lipids | [91] |
| P. antarctica | Low UV-B | Increase in PUFA, structural lipids | [70] |
| C. simplex | High UV-B | Increase in total lipids | [70] |
| Tetraselmis sp. | UV-B radiation | Increase in saturated and monounsaturated FA | [93] |
| Phaeodactylum tricornutum | UV radiation | Increased EPA and PUFA | [98] |
| Chaetoceros muelleri | UV-A | Increased monounsaturated FA | [98] |
| Nannochloropsis sp. | UV-A | Increase in saturated FA to PUFA ratio | [96] |
4. Genetic Engineering of Microalgae to Increase Lipid Production
5. Conclusions and Future Directions
References
- Schenk, P.M.; Thomas-Hall, S.R.; Stephens, E.; Marx, U.; Mussgnug, J.; Posten, C.; Kruse, O.; Hankamer, B. Second generation biofuels: High-efficiency microalgae for biodiesel production. BioEnergy Res. 2008, 1, 20–43. [Google Scholar]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar]
- Chisti, Y. Biodiesel from microalgae beats bioethanol. Trends Biotech. 2008, 26, 126–131. [Google Scholar]
- Christenson, L.; Sims, R. Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnol. Adv. 2011, 29, 686–702. [Google Scholar]
- Stephens, E.; Ross, I.L.; Mussgnug, J.H.; Wagner, L.D.; Borowitzka, M.A.; Posten, C.; Kruse, O.; Hankamer, B. Future prospects of microalgal biofuel production systems. Trends Plant Sci. 2010, 15, 554–564. [Google Scholar]
- Bruton, T.; Lyons, H.; Lerat, Y.; Stanley, M.; Rasmussen, M.B. A Review of the Potential of Marine Algae as a Source of Biofuel in Ireland; Technical Report; Sustainable Energy Ireland: Dublin, Ireland, 2009. [Google Scholar]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar]
- McGinn, P.; Dickinson, K.; Bhatti, S.; Frigon, J.-C.; Guiot, S.; O’Leary, S. Integration of microalgae cultivation with industrial waste remediation for biofuel and bioenergy production: Opportunities and limitations. Photosynth. Res. 2011, 109, 231–247. [Google Scholar]
- Fukuda, H.; Kondo, A.; Noda, H. Biodiesel fuel production by transesterification of oils. J. Biosci. Bioeng. 2001, 92, 405–416. [Google Scholar]
- Chen, Y.F. Production of Biodiesel from Algal Biomass: Current Perspectives and Future; Academic Press: Waltham, MA, USA, 2011; p. 399. [Google Scholar]
- Gurr, M.I.; Harwood, J.L.; Frayn, K.N. Lipid Biochemistry: An Introduction, 5th ed.; Blackwell: Oxford, UK, 2002; p. 320. [Google Scholar]
- Thompson, G.A. Lipids and membrane function in green algae. Biochim. Biophys. Acta 1996, 1302, 17–45. [Google Scholar]
- Bigogno, C.; Khozin-Goldberg, I.; Cohen, Z. Accumulation of arachidonic acid-rich triacylglycerols in the microalga Parietochloris incisa (trebuxiophyceae, chlorophyta). Phytochemistry 2002, 60, 135–143. [Google Scholar]
- Alonso, D.L.; Belarbi, E.-H.; Rodríguez-Ruiz, J.; Segura, C.I.; Giménez, A. Acyl lipids of three microalgae. Phytochemistry 1998, 47, 1473–1481. [Google Scholar]
- Khozin-Goldberg, I.; Cohen, Z. The effect of phosphate starvation on the lipid and fatty acid composition of the fresh water eustigmatophyte Monodus subterraneus. Phytochemistry 2006, 67, 696–701. [Google Scholar]
- Makewicz, A.; Gribi, C.; Eichenberger, W. Lipids of Ectocarpus fasciculatus (phaeophyceae). Incorporation of [l-14C]oleate and the role of TAG and MGDG in lipid metabolism. Plant Cell Physiol. 1997, 38, 952–962. [Google Scholar]
- Sato, N.; Hagio, M.; Wada, H.; Tsuzuki, A.M. Environmental effects on acidic lipids of thylakoid membranes. Biochem. Soc. Trans. 2000, 28, 912–914. [Google Scholar]
- Guschina, I.A.; Harwood, J.L. Lipids and lipid metabolism in eukaryotic algae. Prog. Lipid Res. 2006, 45, 160–186. [Google Scholar]
- Schuhmann, H.; Lim, D.K.Y.; Schenk, P.M. Perspectives on metabolic engineering for increased lipid contents in microalgae. Biofuels 2011, 3, 71–86. [Google Scholar]
- Miao, X.; Wu, Q. Biodiesel production from heterotrophic microalgal oil. Bioresour. Technol. 2006, 97, 841–846. [Google Scholar]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar]
- Lynn, S.G.; Kilham, S.S.; Kreeger, D.A.; Interlandi, S.J. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (bacillariophyceae). J. Phycol. 2000, 36, 510–522. [Google Scholar]
- Arisz, S.A.; van Himbergen, J.A.J.; Musgrave, A.; van den Ende, H.; Munnik, T. Polar glycerolipids of Chlamydomonas moewusii. Phytochemistry 2000, 53, 265–270. [Google Scholar]
- Yeh, K.L.; Chang, J.S. Nitrogen starvation strategies and photobioreactor design for enhancing lipid production of a newly isolated microalga Chlorella vulgaris esp-31: Implications for biofuels. Biotechnol. J. 2011, 6, 1358–1366. [Google Scholar]
- Praveenkumar, R.; Shameera, K.; Mahalakshmi, G.; Akbarsha, M.A.; Thajuddin, N. Influence of nutrient deprivations on lipid accumulation in a dominant indigenous microalga Chlorella sp., bum11008: Evaluation for biodiesel production. Biomass Bioenerg. 2012, 37, 60–66. [Google Scholar]
- Hsieh, C.-H.; Wu, W.-T. Cultivation of microalgae for oil production with a cultivation strategy of urea limitation. Bioresour. Technol. 2009, 100, 3921–3926. [Google Scholar]
- Hu, Q. PSA abstracts. J. Phycol. 2006, 42, 1–48. [Google Scholar]
- Rodolfi, L.; Chini Zittelli, G.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar]
- Alonso, D.L.; Belarbi, E.-H.; Fernández-Sevilla, J.M.; Rodríguez-Ruiz, J.; Grima, E.M. Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry 2000, 54, 461–471. [Google Scholar]
- Xin, L.; Hong-ying, H.; Ke, G.; Ying-xue, S. Effects of different nitrogen and phosphorus concentrations on the growth, nutrient uptake, and lipid accumulation of a freshwater microalga Scenedesmus sp. Bioresour. Technol. 2010, 101, 5494–5500. [Google Scholar]
- Illman, A.M.; Scragg, A.H.; Shales, S.W. Increase in Chlorella strains calorific values when grown in low nitrogen medium. Enzyme Microb. Technol. 2000, 27, 631–635. [Google Scholar]
- Lv, J.-M.; Cheng, L.-H.; Xu, X.-H.; Zhang, L.; Chen, H.-L. Enhanced lipid production of Chlorella vulgaris by adjustment of cultivation conditions. Bioresour. Technol. 2010, 101, 6797–6804. [Google Scholar]
- Griffiths, M.; Harrison, S. Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J. Appl. Phycol. 2009, 21, 493–507. [Google Scholar]
- Liu, Z.-Y.; Wang, G.-C.; Zhou, B.-C. Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresour. Technol. 2008, 99, 4717–4722. [Google Scholar]
- Widjaja, A.; Chien, C.-C.; Ju, Y.-H. Study of increasing lipid production from fresh water microalgae Chlorella vulgaris. J. Taiwan Inst. Chem. Eng. 2009, 40, 13–20. [Google Scholar]
- Solovchenko, A.; Khozin-Goldberg, I.; Didi-Cohen, S.; Cohen, Z.; Merzlyak, M. Effects of light and nitrogen starvation on the content and composition of carotenoids of the green microalga Parietochloris incisa. Russ. J. Plant Physiol. 2008, 55, 455–462. [Google Scholar]
- Reitan, K.I.; Rainuzzo, J.R.; Olsen, Y. Effect of nutrient limitation on fatty acid and lipid content of marine microalgae. J. Phycol. 1994, 30, 972–979. [Google Scholar]
- El-Sheek, M.M.; Rady, A.A. Effect of phosphorus starvation on growth, photosynthesis and some metabolic processes in the unicellular green alga Chlorella kessleri. Phyton 1995, 35, 139–151. [Google Scholar]
- Härtel, H.; Dörmann, P.; Benning, C. DGD1-independent biosynthesis of extraplastidic galactolipids after phosphate deprivation in Arabidopsis. Proc. Nat. Acad. Sci. USA 2000, 97, 10649–10654. [Google Scholar]
- Andersson, M.X.; Stridh, M.H.; Larsson, K.E.; Liljenberg, C.; Sandelius, A.S. Phosphate-deficient oat replaces a major portion of the plasma membrane phospholipids with the galactolipid digalactosyldiacylglycerol. FEBS Lett. 2003, 537, 128–132. [Google Scholar]
- Matthew, T.; Zhou, W.; Rupprecht, J.; Lim, L.; Thomas-Hall, S.R.; Doebbe, A.; Kruse, O.; Hankamer, B.; Marx, U.C.; Smith, S.M.; et al. The metabolome of Chlamydomonas reinhardtii following induction of anaerobic H2 production by sulfur depletion. J. Biol. Chem. 2009, 284, 23415–23425. [Google Scholar]
- Dean, A.P.; Sigee, D.C.; Estrada, B.; Pittman, J.K. Using FTIR spectroscopy for rapid determination of lipid accumulation in response to nitrogen limitation in freshwater microalgae. Bioresour. Technol. 2010, 101, 4499–4507. [Google Scholar]
- Converti, A.; Casazza, A.A.; Ortiz, E.Y.; Perego, P.; Del Borghi, M. Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem. Eng. Process. 2009, 48, 1146–1151. [Google Scholar]
- Chen, M.; Tang, H.; Ma, H.; Holland, T.C.; Ng, K.Y.S.; Salley, S.O. Effect of nutrients on growth and lipid accumulation in the green algae Dunaliella tertiolecta. Bioresour. Technol. 2011, 102, 1649–1655. [Google Scholar]
- Li, Y.; Horsman, M.; Wang, B.; Wu, N.; Lan, C. Effects of nitrogen sources on cell growth and lipid accumulation of green alga Neochloris oleoabundans. Appl. Microbiol. Biotechnol. 2008, 81, 629–636. [Google Scholar]
- Gardner, R.; Peters, P.; Peyton, B.; Cooksey, K.E. Medium ph and nitrate concentration effects on accumulation of triacylglycerol in two members of the chlorophyta. J. Appl. Phycol. 2011, 23, 1005–1016. [Google Scholar]
- Roessler, P.G. Effects of silicon deficiency on lipid composition and metabolism in the diatom Cyclotella cryptica. J. Phycol. 1988, 24, 394–400. [Google Scholar]
- Morgan-Kiss, R.M.; Priscu, J.C.; Pocock, T.; Gudynaite-Savitch, L.; Huner, N.P.A. Adaptation and acclimation of photosynthetic microorganisms to permanently cold environments. Microbiol. Mol. Biol. Rev. 2006, 70, 222–252. [Google Scholar]
- Renaud, S.M.; Thinh, L.-V.; Lambrinidis, G.; Parry, D.L. Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 2002, 211, 195–214. [Google Scholar]
- Lynch, D.V.; Thompson, T.G., Jr. Low temperature-induced alterations in the chloroplast and microsomal membranes of Dunaliella salina. Plant Physiol. 1982, 69, 1369–1375. [Google Scholar]
- Murata, N.; Troughton, J.H.; Fork, D.C. Relationships between the transition of the physical phase of membrane lipids and photosynthetic parameters in Anacystis nidulans and lettuce and spinach chloroplasts. Plant Physiol. 1975, 56, 508–517. [Google Scholar]
- Sato, N.; Murata, N. Temperature shift-induced responses in lipids in the blue-green alga, Anabaena variabilis: The central role of diacylmonogalactosylglycerol in thermo-adaptation. BBA-Lipid Lipid Metab. 1980, 619, 353–366. [Google Scholar]
- Somerville, C. Direct tests of the role of membrane lipid composition in low-temperature-induced photoinhibition and chilling sensitivity in plants and cyanobacteria. Proc. Nat. Acad. Sci. USA 1995, 92, 6215–6218. [Google Scholar]
- Harwood, J.L.; Jones, A.L. Lipid Metabolism in Algae. In Advances in Botanical Research; Callow, J.A., Ed.; Academic Press: Waltham, MA, USA, 1989; Volume 16, pp. 1–53. [Google Scholar]
- Aaronson, S. Effect of incubation temperature on the macromolecular and lipid content of the phytoflagellate Ochromonas danica. J. Phycol. 1973, 9, 111–113. [Google Scholar]
- Sushchik, N.N.; Kalacheva, G.S.; Zhila, N.O.; Gladyshev, M.I.; Volova, T.G. A temperature dependence of the intra- and extracellular fatty-acid composition of green algae and cyanobacterium. Russ. J. Plant Physiol. 2003, 50, 374–380. [Google Scholar]
- Boussiba, S.; Vonshak, A.; Cohen, Z.; Avissar, Y.; Richmond, A. Lipid and biomass production by the halotolerant microalga Nannochloropsis salina. Biomass 1987, 12, 37–47. [Google Scholar]
- McLarnon-Riches, C.J.; Rolph, C.E.; Greenway, D.L.A.; Robinson, P.K. Effects of environmental factors and metals on Selenastrum capricornutum lipids. Phytochemistry 1998, 49, 1241–1247. [Google Scholar]
- Zhu, C.; Lee, Y.; Chao, T. Effects of temperature and growth phase on lipid and biochemical composition of Isochrysis galbana tk1. J. Appl. Phycol. 1997, 9, 451–457. [Google Scholar]
- Jiang, H.; Gao, K. Effects of lowering temperature during culture on the production of polyunsaturated fatty acids in the marine diatom Phaeodactylum tricornutum (bacillariophyceae). J. Phycol. 2004, 40, 651–654. [Google Scholar]
- Murata, N. Molecular species composition of phosphatidylglycerols from chilling-sensitive and chilling-resistant plants. Plant Cell Physiol. 1983, 24, 81–86. [Google Scholar]
- Joh, T.; Yoshida, T.; Yoshimoto, M.; Miyamoto, T.; Hatano, S. Composition and positional distribution of fatty acids in polar lipids from Chlorella ellipsoidea differing in chilling susceptibility and frost hardiness. Physiol. Plant. 1993, 89, 285–290. [Google Scholar]
- Tatsuzawa, H.; Takizawa, E. Changes in lipid and fatty acid composition of Pavlova lutheri. Phytochemistry 1995, 40, 397–400. [Google Scholar]
- Fork, D.C.; Murata, N.; Sato, N. Effect of growth temperature on the lipid and fatty acid composition, and the dependence on temperature of light-induced redox reactions of cytochrome f and of light energy redistribution in the thermophilic blue-green alga Synechococcus lividus. Plant Physiol. 1979, 63, 524–530. [Google Scholar]
- Patterson, G. Effect of culture temperature on fatty acid composition of Chlorella sorokiniana. Lipids 1970, 5, 597–600. [Google Scholar]
- Azachi, M.; Sadka, A.; Fisher, M.; Goldshlag, P.; Gokhman, I.; Zamir, A. Salt induction of fatty acid elongase and membrane lipid modifications in the extreme halotolerant alga Dunaliella salina. Plant Physiol. 2002, 129, 1320–1329. [Google Scholar]
- Takagi, M.; Yoshida, T. Effect of salt concentration on intracellular accumulation of lipids and triacylglyceride in marine microalgae Dunaliella cells. J. Biosci. Bioeng. 2006, 101, 223–226. [Google Scholar]
- Xu, X.-Q.; Beardall, J. Effect of salinity on fatty acid composition of a green microalga from an antarctic hypersaline lake. Phytochemistry 1997, 45, 655–658. [Google Scholar]
- Chen, G.-Q.; Jiang, Y.; Chen, F. Salt-induced alterations in lipid composition of diatom Nitzschia laevis (bacillariophyceae) under heterotrophic culture condition1. J. Phycol. 2008, 44, 1309–1314. [Google Scholar]
- Jiang, Y.; Chen, F. Effects of salinity on cell growth and docosahexaenoic acid content of the heterotrophic marine microalga Crypthecodinium cohnii. J. Ind. Microbiol. Biotechnol. 1999, 23, 508–513. [Google Scholar]
- Zhu, L.Y.; Zhang, X.C.; Ji, L.; Song, X.J.; Kuang, C.H. Changes of lipid content and fatty acid composition of Schizochytrium limacinum in response to different temperatures and salinities. Process Biochem. 2007, 42, 210–214. [Google Scholar]
- Guckert, J.B.; Cooksey, K.E. Triglyceride accumulation and fatty acid profile changes in Chlorella (chlorophyta) during high ph-induced cell cycle inhibition. J. Phycol. 1990, 26, 72–79. [Google Scholar]
- Tatsuzawa, H.; Takizawa, E.; Wada, M.; Yamamoto, Y. Fatty acid and lipid composition of the acidophilic green alga Chlamydomonas sp. J. Phycol. 1996, 32, 598–601. [Google Scholar]
- Einicker-Lamas, M.; Mezian, G.A.; Fernandes, T.B.; Silva, F.L.S.; Guerra, F.; Miranda, K.; Attias, M.; Oliveira, M.M. Euglena gracilis as a model for the study of Cu2+ and Zn2+ toxicity and accumulation in eukaryotic cells. Environ. Pollut. 2002, 120, 779–786. [Google Scholar]
- Richardson, K.; Beardall, J.; Raven, J.A. Adaptation of unicellular algae to irradiance: An analysis of strategies. New Phytol. 1983, 93, 157–191. [Google Scholar]
- Harwood, J.L. Membrane Lipids in Algae. In Lipids in Photosynthesis: Structure, Fuction and Genetics; Siegenthaler, P.-A., Murata, N., Eds.; Kluwer Academic Publishers: Kluwer, The Netherlands, 1998; pp. 53–64. [Google Scholar]
- Brown, M.R.; Dunstan, G.A.; Norwood, S.J.; Miller, K.A. Effects of harvest stage and light on the biochemical composition of the diatom Thalassiosira pseudonana. J. Phycol. 1996, 32, 64–73. [Google Scholar]
- Khotimchenko, S.V.; Yakovleva, I.M. Lipid composition of the red alga Tichocarpus crinitus exposed to different levels of photon irradiance. Phytochemistry 2005, 66, 73–79. [Google Scholar]
- Napolitano, G.E. The relationship of lipids with light and chlorophyll measurements in freshwater algae and periphyton. J. Phycol. 1994, 30, 943–950. [Google Scholar]
- Orcutt, D.; Patterson, G. Effect of light intensity upon lipid composition Nitzschia closterium (cylindrotheca fusiformis). Lipids 1974, 9, 1000–1003. [Google Scholar]
- Carvalho, A.P.; Malcata, F.X. Optimization of ω-3 fatty acid production by microalgae: Crossover effects of CO2 and light intensity under batch and continuous cultivation modes. Mar. Biotech. 2005, 7, 381–388. [Google Scholar]
- Mock, T.; Kroon, B.M.A. Photosynthetic energy conversion under extreme conditions—ii: The significance of lipids under light limited growth in antarctic sea ice diatoms. Phytochemistry 2002, 61, 53–60. [Google Scholar]
- Bandarra, N.M.; Pereira, P.A.; Batista, I.; Vilela, M.H. Fatty acids, sterols and α-tocopherol in isochrysis galbana. J. Food Lipids 2003, 10, 25–34. [Google Scholar]
- Fábregas, J.; Maseda, A.; Domínguez, A.; Otero, A. The cell composition of Nannochloropsis sp. changes under different irradiances in semicontinuous culture. World J. Microbiol. Biotechnol. 2004, 20, 31–35. [Google Scholar]
- Holzinger, A.; Lütz, C. Algae and UV irradiation: Effects on ultrastructure and related metabolic functions. Micron 2006, 37, 190–207. [Google Scholar]
- Xue, L.; Zhang, Y.; Zhang, T.; An, L.; Wang, X. Effects of enhanced ultraviolet-B radiation on algae and cyanobacteria. Crit. Rev. Microbiol. 2005, 31, 79–89. [Google Scholar]
- He, Y.-Y.; Häder, D.-P. UV-B-induced formation of reactive oxygen species and oxidative damage of the cyanobacterium Anabaena sp.: Protective effects of ascorbic acid and n-acetyl-cysteine. J. Photochem. Photobiol. B-Biol. 2002, 66, 115–124. [Google Scholar]
- Bhandari, R.; Sharma, P. Photosynthetic and biochemical characterization of pigments and UV-absorbing compounds in Phormidium tenue due to UV-B radiation. J. Appl. Phycol. 2011, 23, 283–292. [Google Scholar]
- Wiley, P.S. Photosynthetic and Oxidative Stress in the Green Alga Dunaliella tertiolecta: The Effects of UV-B and UV-A Radiation. Ph.D. Thesis, University of New Hampshire, Durham, NH, USA, 2009. [Google Scholar]
- Fouqueray, M.; Mouget, J.-L.; Morant-Manceau, A.; Tremblin, G. Dynamics of short-term acclimation to UV radiation in marine diatoms. J. Photochem. Photobiol. B-Biol. 2007, 89, 1–8. [Google Scholar]
- Srinivas, R.; Ochs, C. Effect of UV-A irradiance on lipid accumulation in Nannochloropsis oculata. Photochem. Photobiol. 2012, 88, 684–689. [Google Scholar]
- Skerratt, J.H.; Davidson, A.D.; Nichols, P.D.; McMeekin, T.A. Effect of UV-B on lipid content of three antarctic marine phytoplankton. Phytochemistry 1998, 49, 999–1007. [Google Scholar]
- Goes, J.I.; Handa, N.; Taguchi, S.; Hama, T.; Saito, H. Impact of uv radiation on the production patterns and composition of dissolved free and combined amino acids in marine phytoplankton. J. Plankton Res. 1995, 17, 1337–1362. [Google Scholar]
- Liang, Y.; Beardall, J.; Heraud, P. Effect of uv radiation on growth, chlorophyll fluorescence and fatty acid composition of Phaeodactylum tricornutum and Chaetoceros muelleri (bacillariophyceae). Phycologia 2006, 45, 605–615. [Google Scholar]
- Guihéneuf, F.; Fouqueray, M.; Mimouni, V.; Ulmann, L.; Jacquette, B.; Tremblin, G. Effect of UV stress on the fatty acid and lipid class composition in two marine microalgae Pavlova lutheri (pavlovophyceae) and Odontella aurita (bacillariophyceae). J. Appl. Phycol. 2010, 22, 629–638. [Google Scholar]
- Forján, E.; Garbayo, I.; Henriques, M.; Rocha, J.; Vega, J.; Vílchez, C. UV-A mediated modulation of photosynthetic efficiency, xanthophyll cycle and fatty acid production of Nannochloropsis. Mar. Biotechnol. 2011, 13, 366–375. [Google Scholar]
- Guihéneuf, F.; Mimouni, V.; Ulmann, L.; Tremblin, G. Combined effects of irradiance level and carbon source on fatty acid and lipid class composition in the microalga Pavlova lutheri commonly used in mariculture. J. Exp. Mar. Biol. Ecol. 2009, 369, 136–143. [Google Scholar]
- Liang, Y.; Beardall, J.; Heraud, P. Effects of nitrogen source and uv radiation on the growth, chlorophyll fluorescence and fatty acid composition of Phaeodactylum tricornutum and Chaetoceros muelleri (bacillariophyceae). J. Photochem. Photobiol. B-Biol. 2006, 82, 161–172. [Google Scholar]
- Shrager, J.; Hauser, C.; Chang, C.-W.; Harris, E.H.; Davies, J.; McDermott, J.; Tamse, R.; Zhang, Z.; Grossman, A.R. Chlamydomonas reinhardtii genome project. A guide to the generation and use of the cdna information. Plant Physiol. 2003, 131, 401–408. [Google Scholar]
- Jain, M.; Shrager, J.; Harris, E.H.; Halbrook, R.; Grossman, A.R.; Hauser, C.; Vallon, O. Est assembly supported by a draft genome sequence: An analysis of the Chlamydomonas reinhardtii transcriptome. Nucl. Acid. Res. 2007, 35, 2074–2083. [Google Scholar]
- Molnar, A.; Schwach, F.; Studholme, D.J.; Thuenemann, E.C.; Baulcombe, D.C. Mirnas control gene expression in the single-cell alga Chlamydomonas reinhardtii. Nature 2007, 447, 1126–1129. [Google Scholar]
- Courchesne, N.M.D.; Parisien, A.; Wang, B.; Lan, C.Q. Enhancement of lipid production using biochemical, genetic and transcription factor engineering approaches. J. Biotechnol. 2009, 141, 31–41. [Google Scholar]
- Merchant, S.S.; Kropat, J.; Liu, B.; Shaw, J.; Warakanont, J. TAG, You’re it! Chlamydomonas as a reference organism for understanding algal triacylglycerol accumulation. Curr. Opin. Biotechnol. 2011, 23, 1–12. [Google Scholar]
- Boyle, N.R.; Page, M.D.; Liu, B.; Blaby, I.K.; Casero, D.; Kropat, J.; Cokus, S.; Hong-Hermesdorf, A.; Shaw, J.; Karpowicz, S.J.; et al. Three acyltransferases and a nitrogen responsive regulator are implicated in nitrogen starvation-induced triacylglycerol accumulation in Chlamydomonas. J. Biol. Chem. 2012, 287, 15811–15825. [Google Scholar] [CrossRef]
- Yohn, C.; Mendez, M.; Behnke, C.; Brand, A. Stress-Induced Lipid. Trigger. Patent No. WO/2011/097261, 11 August 2011. [Google Scholar]
- Siaut, M.; Cuine, S.; Cagnon, C.; Fessler, B.; Nguyen, M.; Carrier, P.; Beyly, A.; Beisson, F.; Triantaphylides, C.; Li-Beisson, Y.; et al. Oil accumulation in the model green alga Chlamydomonas reinhardtii: Characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnol. 2011, 11, 7. [Google Scholar]
- Roessler, P.G. Purification and characterization of acetyl-coa carboxylase from the diatom Cyclotella cryptica. Plant Physiol. 1990, 92, 73–78. [Google Scholar]
- Dunahay, T.G.; Jarvis, E.E.; Roessler, P.G. Genetic transformation of the diatoms Cyclotella cryptica and Navicula saprophila. J. Phycol. 1995, 31, 1004–1012. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sharma, K.K.; Schuhmann, H.; Schenk, P.M. High Lipid Induction in Microalgae for Biodiesel Production. Energies 2012, 5, 1532-1553. https://doi.org/10.3390/en5051532
Sharma KK, Schuhmann H, Schenk PM. High Lipid Induction in Microalgae for Biodiesel Production. Energies. 2012; 5(5):1532-1553. https://doi.org/10.3390/en5051532
Chicago/Turabian StyleSharma, Kalpesh K., Holger Schuhmann, and Peer M. Schenk. 2012. "High Lipid Induction in Microalgae for Biodiesel Production" Energies 5, no. 5: 1532-1553. https://doi.org/10.3390/en5051532
APA StyleSharma, K. K., Schuhmann, H., & Schenk, P. M. (2012). High Lipid Induction in Microalgae for Biodiesel Production. Energies, 5(5), 1532-1553. https://doi.org/10.3390/en5051532





