A Review on Research on Replacement of CH4 in Natural Gas Hydrates by Use of CO2
Abstract
:1. Introduction
2. Feasibility Study of Replacement Reaction
2.1. The Thermodynamic Feasibility of Replacement
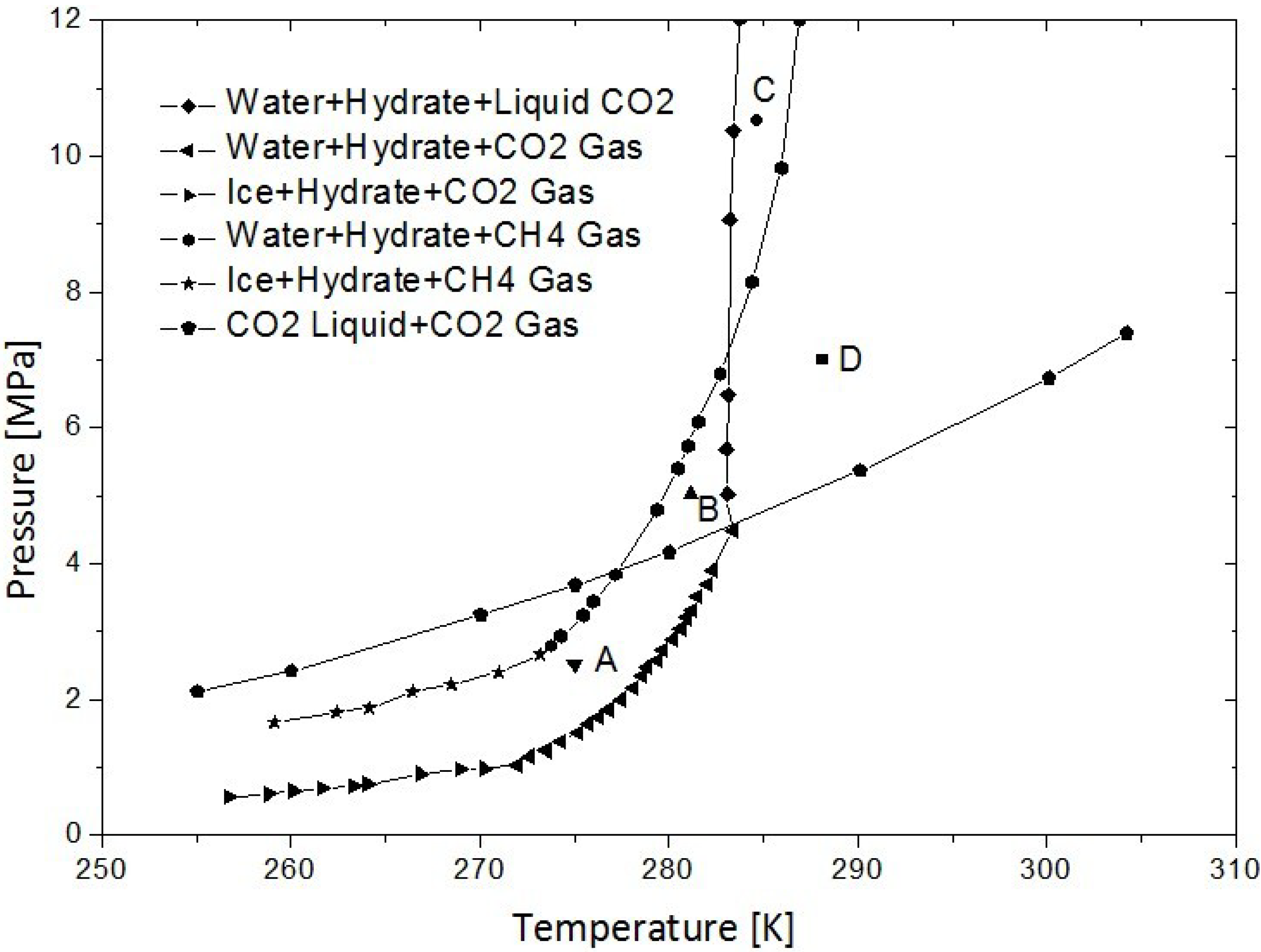
2.2. The Kinetic Feasibility of Replacement
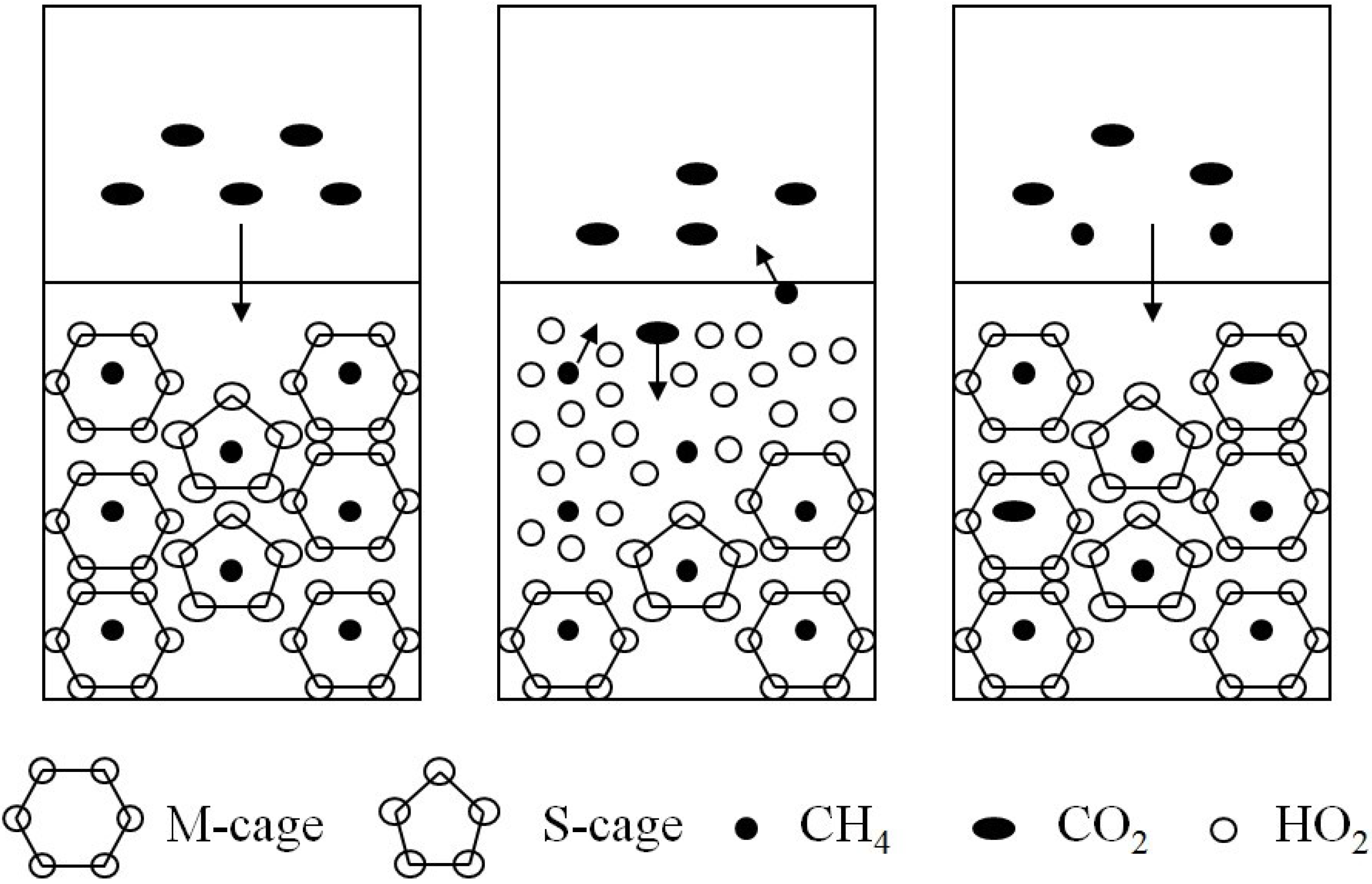
3. Experiment Study of Replacement Reaction
3.1. Replacement of CH4 Hydrate by Use of Gaseous CO2
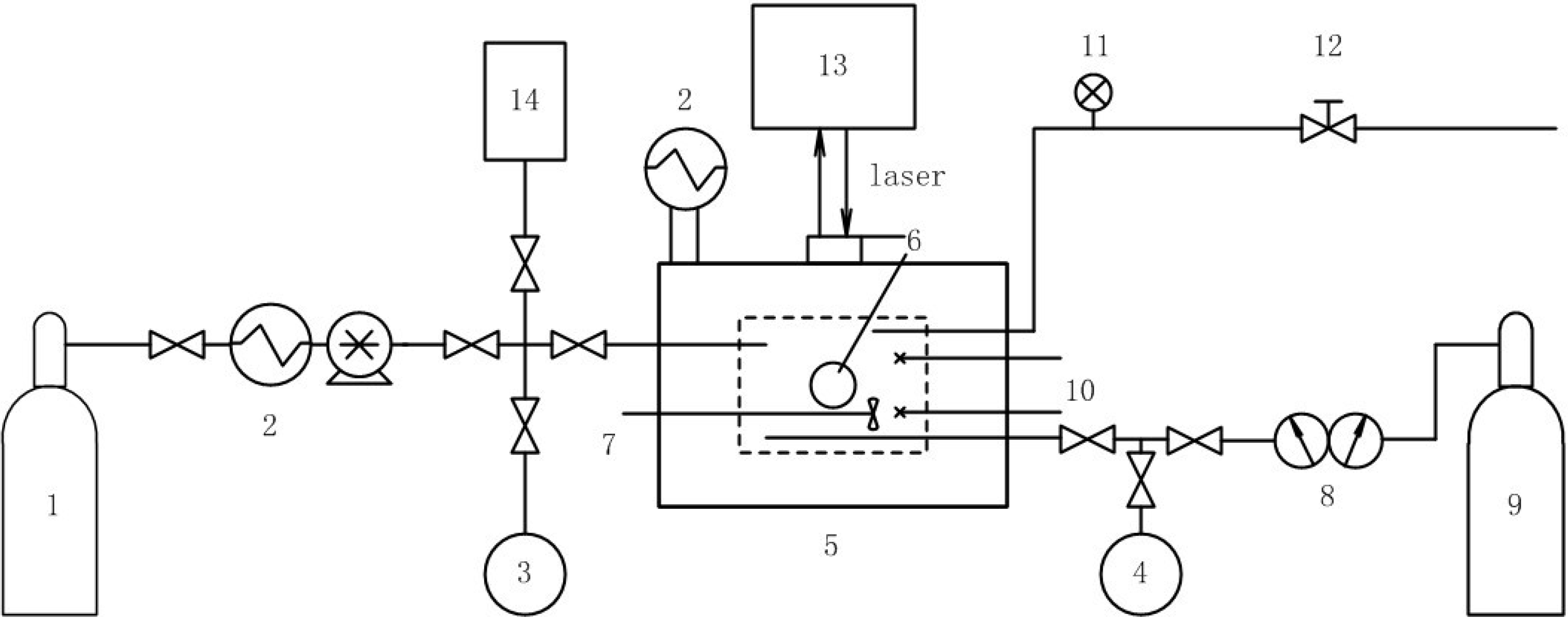
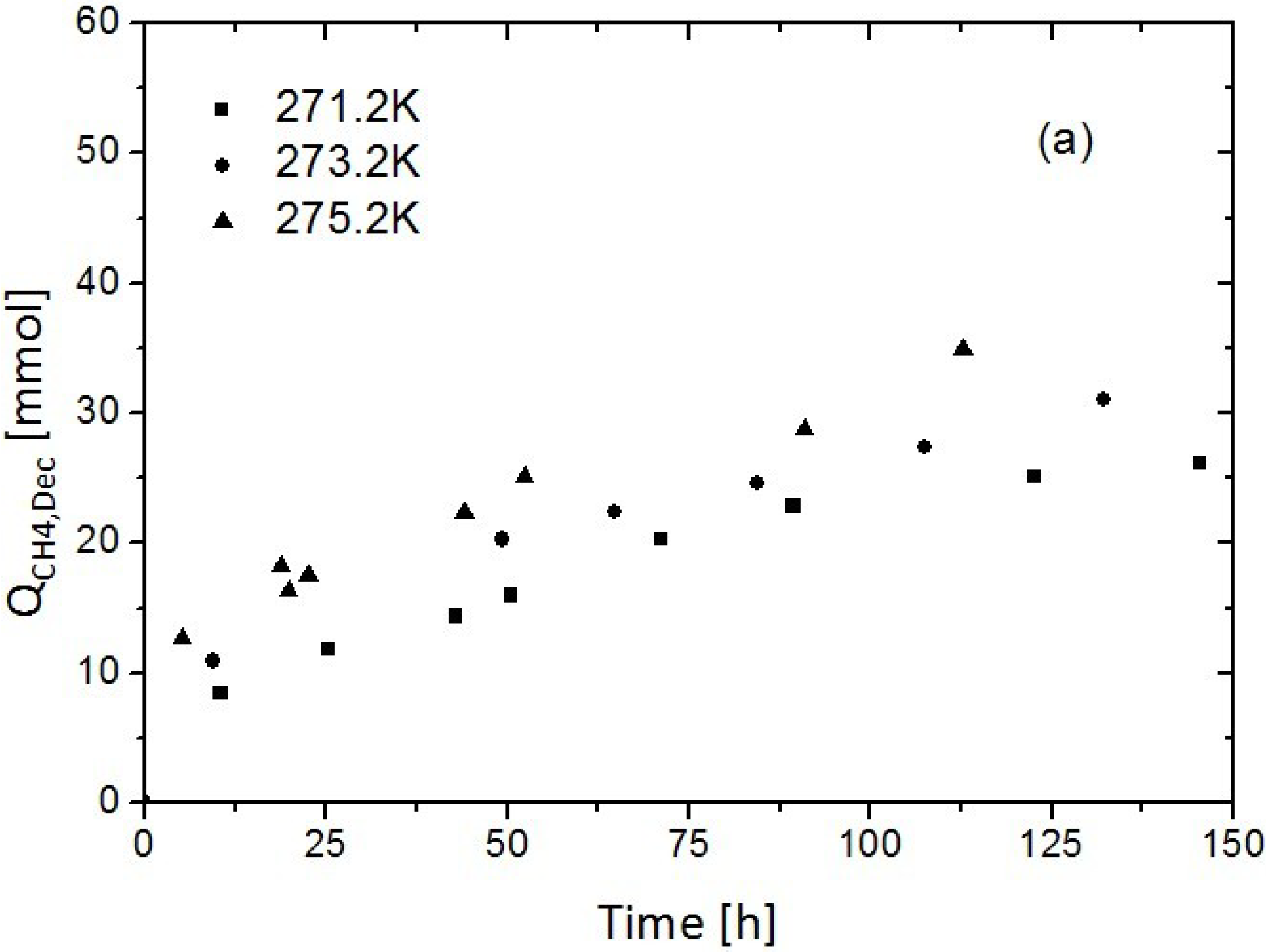
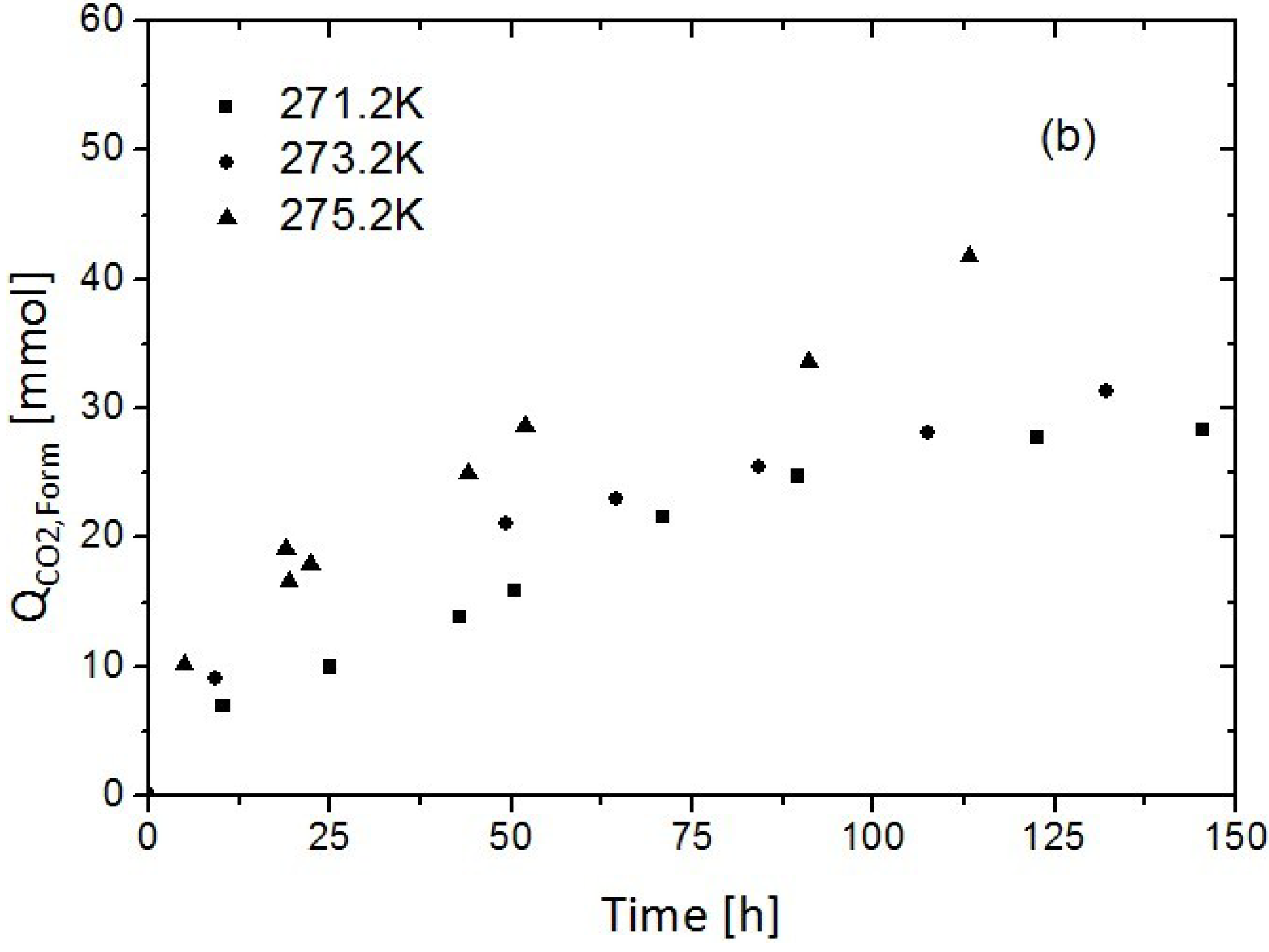
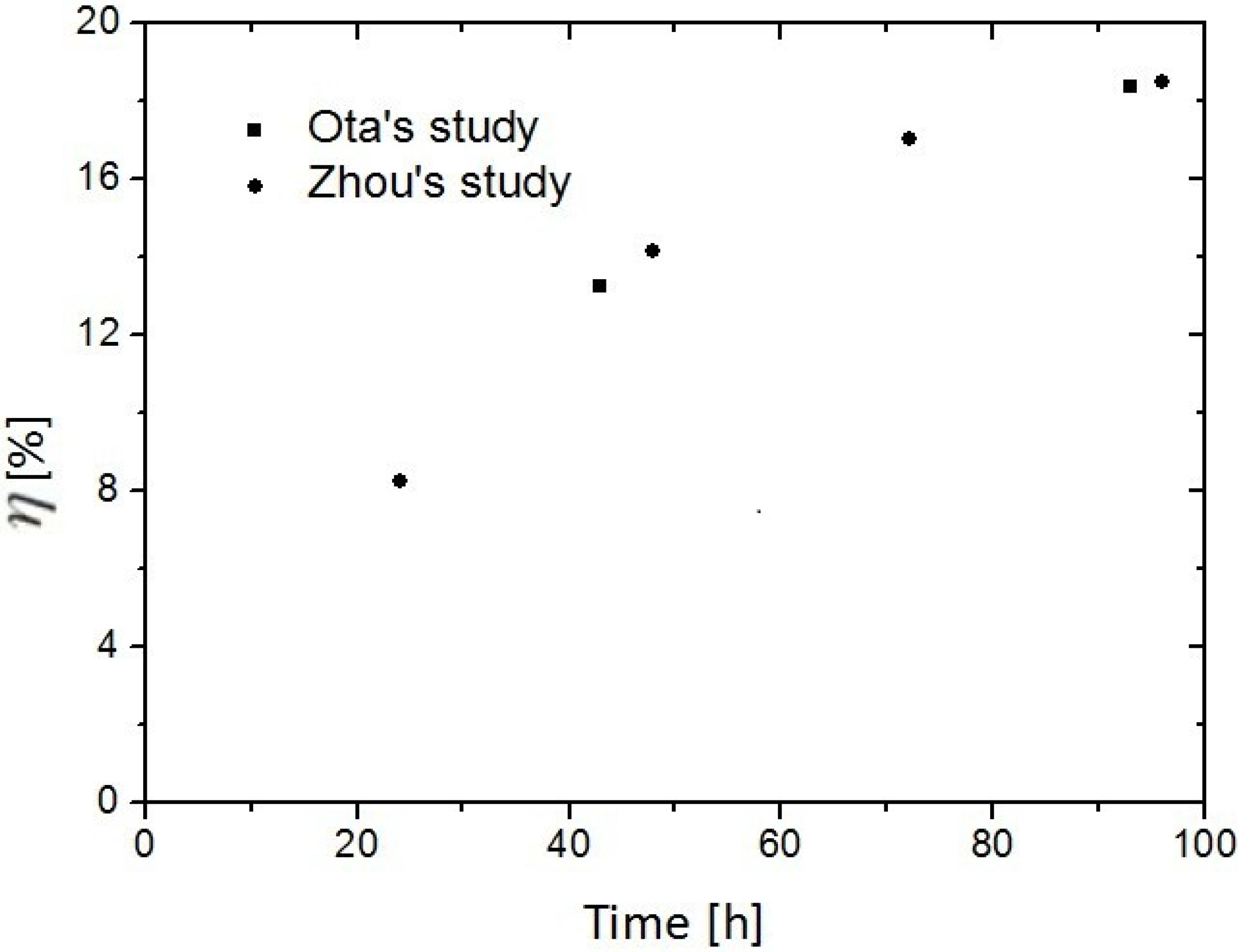
3.2. Replacement of CH4 Hydrate by Use of Liquid CO2
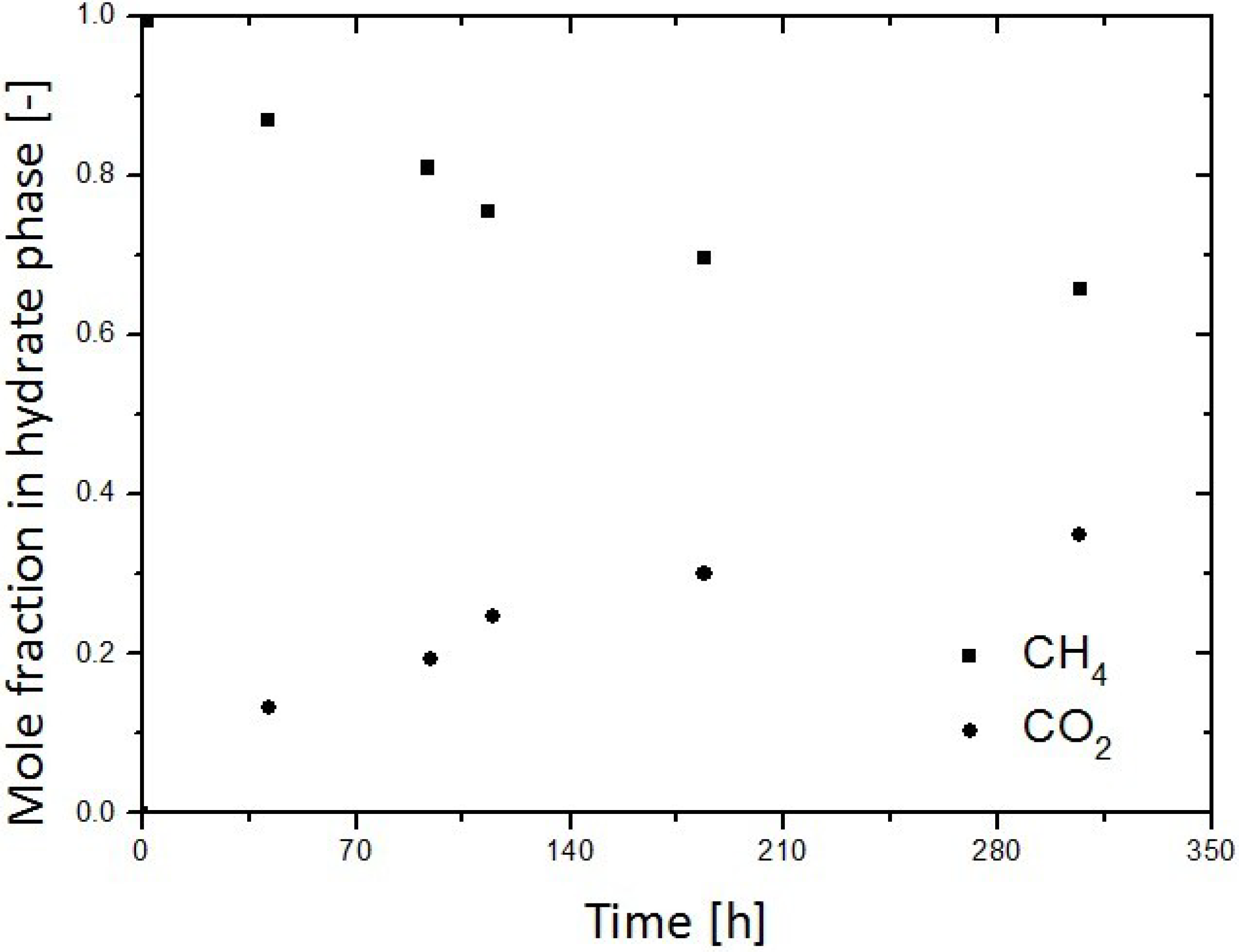
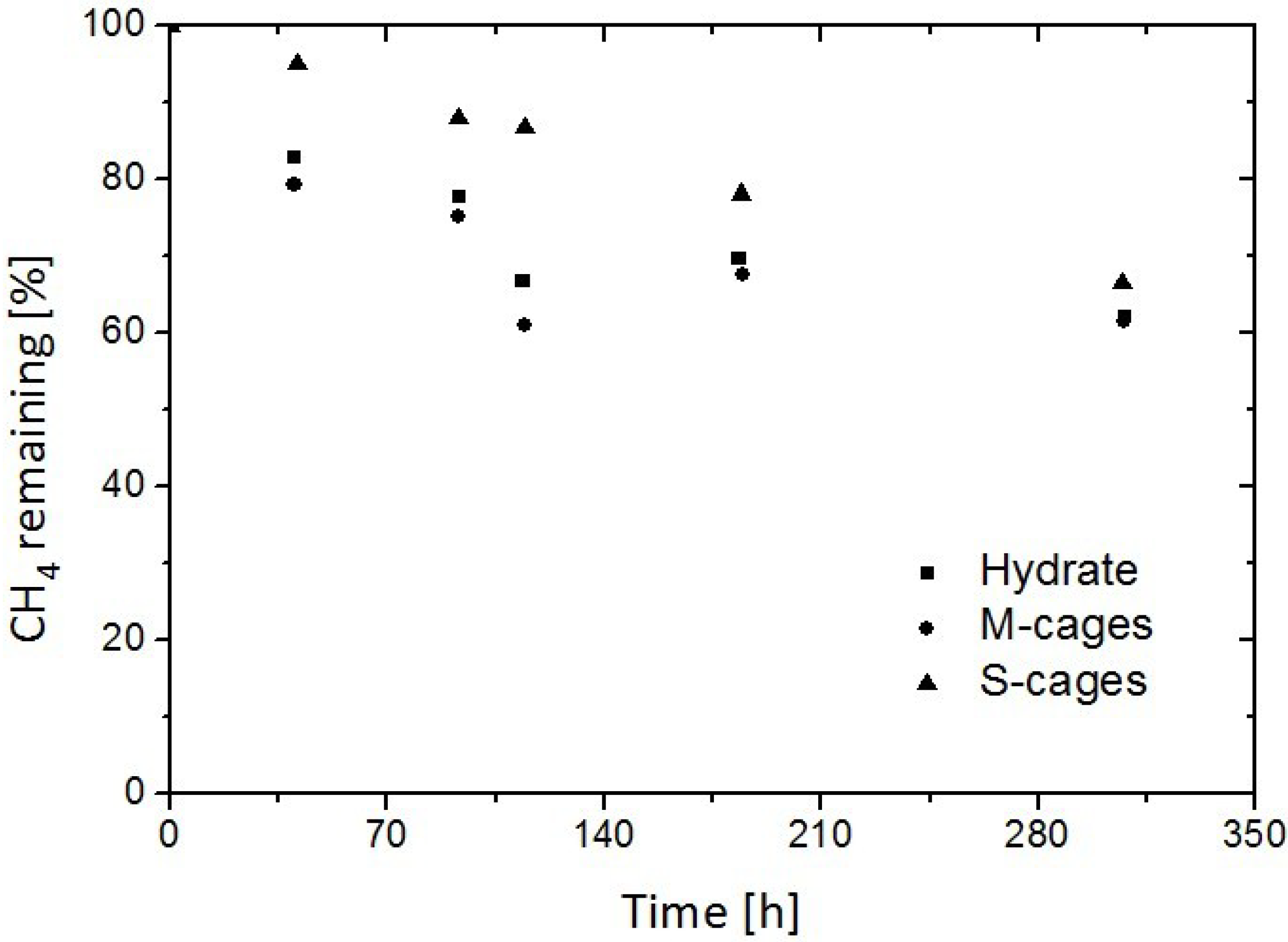
3.3. Replacement of CH4 Hydrate by Use of CO2 Emulsion
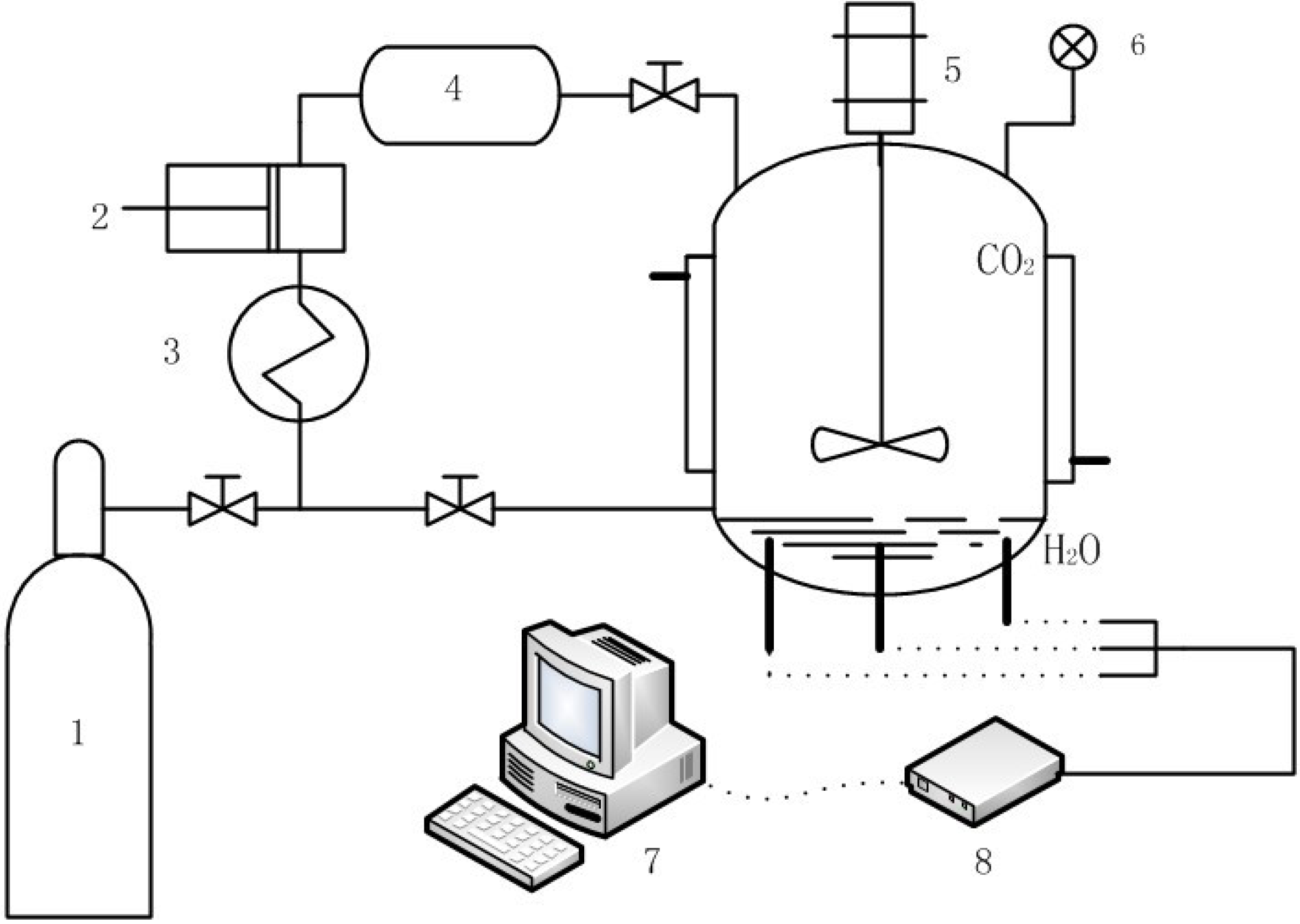
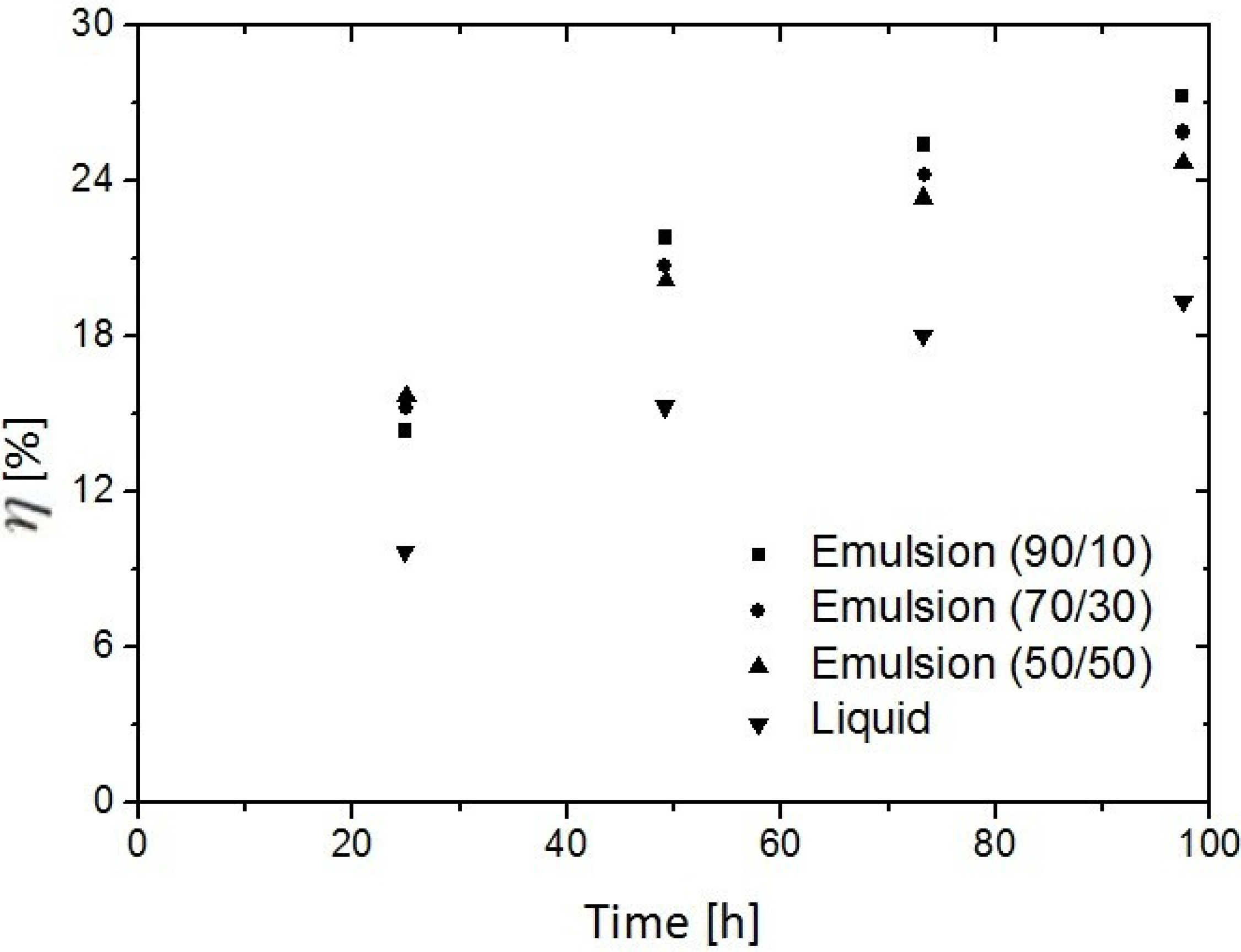
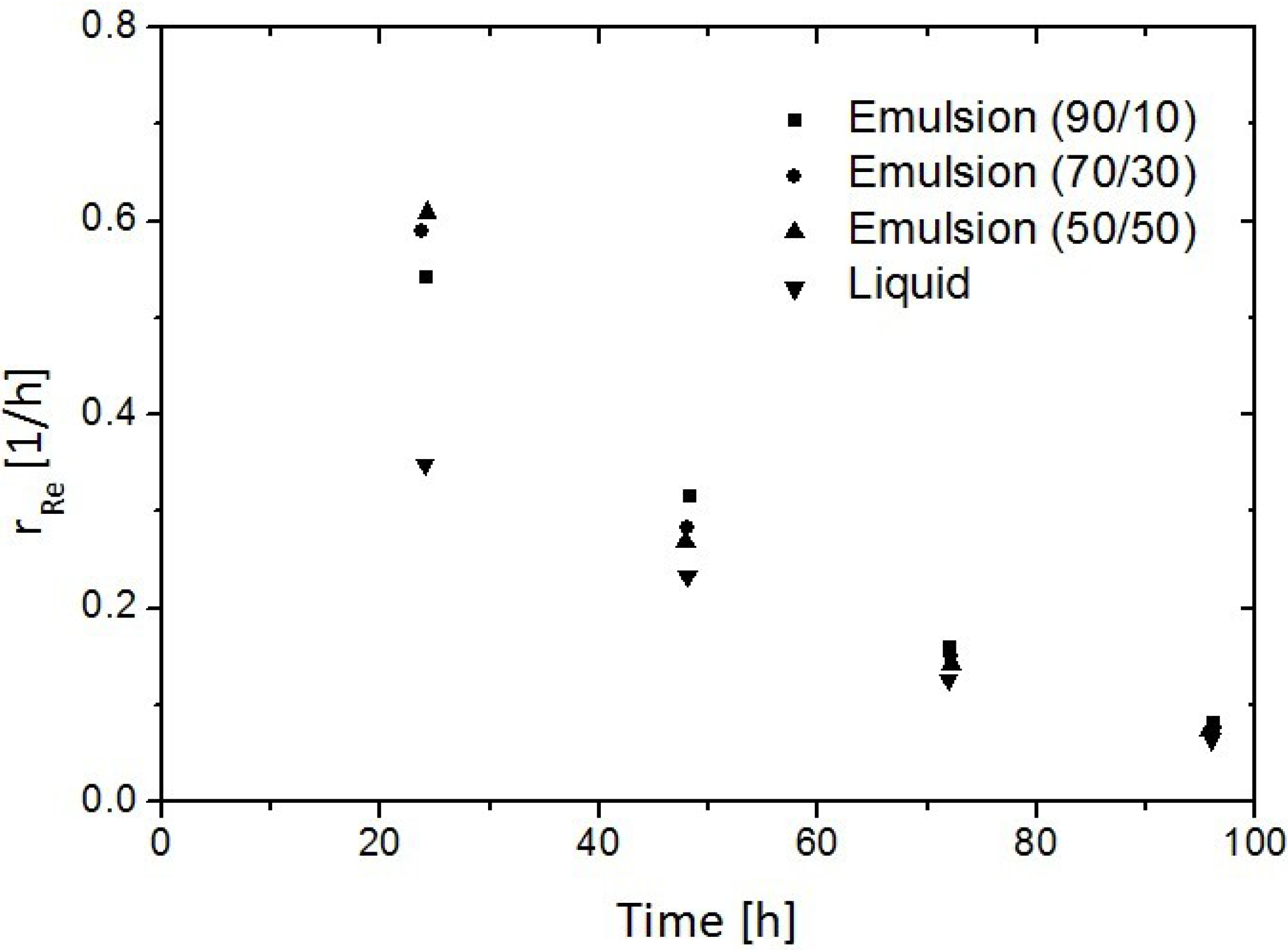
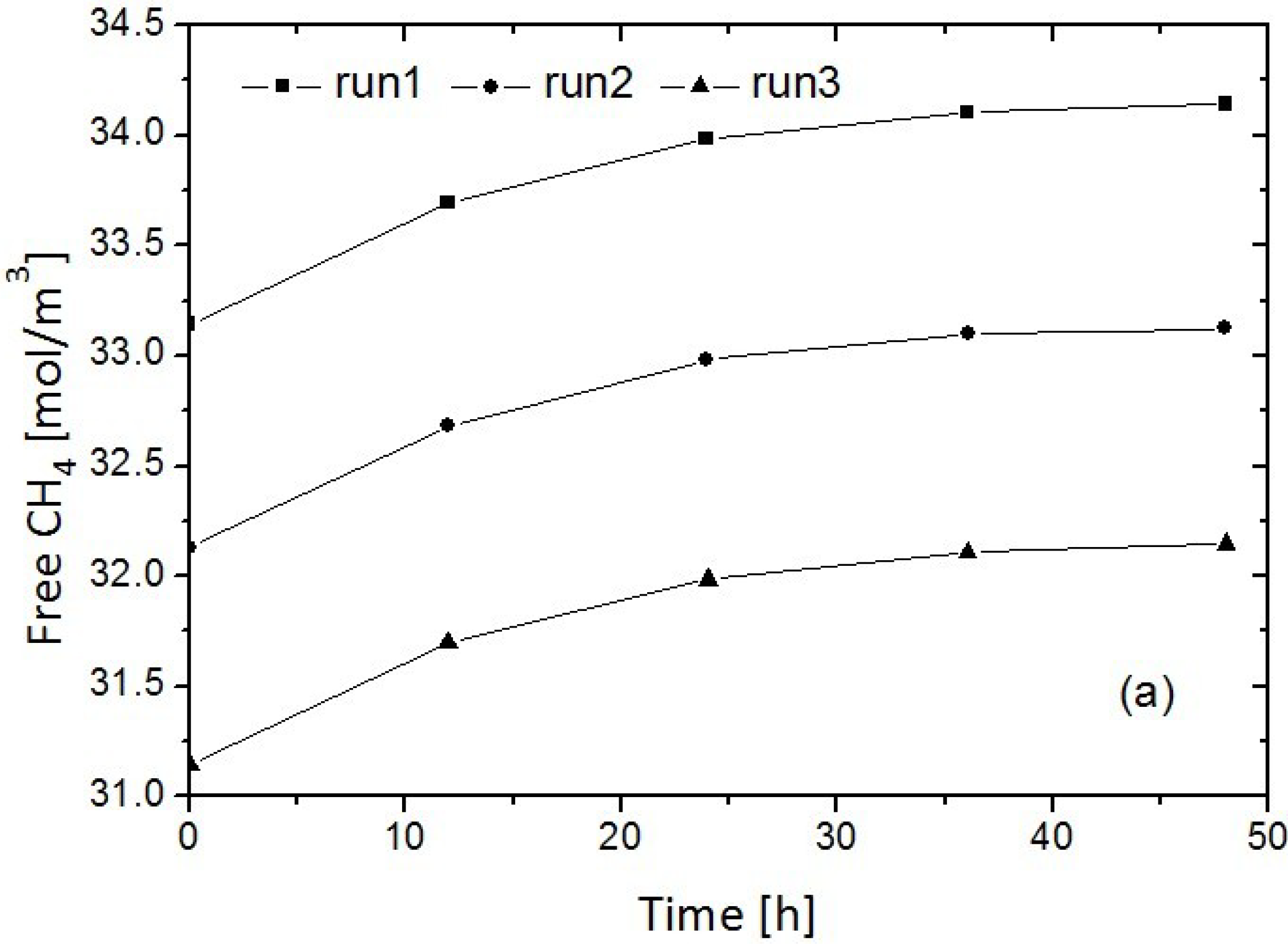

4. Advances in Simulation Research on Replacement
5. Research on Factors Influencing the Replacement Reaction
6. Conclusions
Acknowledgment
Nomenclature
| t | time, s |
| XCH4/XCO2 | ratio of CH4 and CO2 in the vapor phase |
| (XCH4/XCO2)0 | initial ratio of CH4 and CO2 in the vapor phase |
| α | fitting parameter related to the a condensation rate of CH4 molecules from the vapor phase |
| nCH4.H | remaining amount of CH4 in the hydrate phase, mol |
| nCO2.H | amount of CO2 in the hydrate phase, mol |
| f | fugacity, MPa |
| kDec | overall rate constant of the decomposition, mol/s·m·MPa |
| kDec.R | reaction rate constant of decomposition, mol/s·m·MPa |
| kDec.D | decomposition rate constant of mass transfer in the hydrate phase, mol/s·m·MPa |
| kForm | overall rate constant of the formation, mol/s·m·MPa |
| kForm.R | reaction rate constant of formation, mol/s·m·MPa |
| kForm.D | formation rate constant of mass transfer in the hydrate phase, mol/s·m·MPa |
| A | surface area between the gas and the hydrate phase, m2 |
| H | hydrate phase |
| G | gas phase |
References
- Fan, S.S. Storage and Transportation Technologies of Natural Gas Hydrate, 1st ed.; Chemical Industry Press: Beijing, China, 2005; pp. 1–2. [Google Scholar]
- Chen, G.J.; Sun, C.Y.; Ma, Q.L. Science and Technology of Gas Hydrate, 1st ed.; Chemical Industry Press: Beijing, China, 2007; pp. 1–4. [Google Scholar]
- Luo, S.S.; Liu, H.J.; Sun, Y. Advancement on natural gas production from hydrate in deep-sea sediments with CO2. China Resour. Compr. Util. 2008, 26, 19–23. [Google Scholar]
- Milkov, A.V.; Sassen, R. Economic geology of offshore gas hydrate accumulations and provinces. Mar. Pet. Geol. 2002, 19, 1–11. [Google Scholar] [CrossRef]
- Tsimpanogiannis, I.N.; Lichtner, P.C. Parametric study of methane hydrate dissociation in oceanic sediments driven by thermal stimulation. J. Pet. Sci. Eng. 2007, 56, 165–175. [Google Scholar] [CrossRef]
- Ji, C.; Ahmadi, G.; Smith, D.H. Natural gas production from hydrate decomposition by depressurization. Chem. Eng. Sci. 2001, 56, 5801–5814. [Google Scholar] [CrossRef]
- Gayet, P.; Dicharry, C.; Marion, G.; Graciaa, A.; Lachaise, J.; Nesterov, A. Experimental determination of methane hydrate dissociation curve up to 55 MPa by using a small amount of surfactant as hydrate promoter. Chem. Eng. Sci. 2005, 60, 5751–5758. [Google Scholar] [CrossRef]
- Zhou, X.T.; Fan, S.S.; Liang, D.Q. Advancement in research on replacement of CH4 from hydrate with CO2. Chem. Ind. Eng. Process 2006, 25, 524–527. [Google Scholar]
- Maslin, M.; Mikkelsen, N.; Vilela, C.; Haq, B. Sea-level and gas hydrate-controlled catastrophic sediment failures of the Amazon Fan. Geology 1998, 26, 1107–1110. [Google Scholar] [CrossRef]
- Zhao, X.M. A new advance of gas hydrate research. Mar. Geol. Quat. Geol. 1999, 19, 39–46. [Google Scholar]
- Xu, W.S.; Yu, X.H.; Liu, N.N.; Liu, W.L. The development perspective and environmental problems of natural gas hydrate. Nat. Gas Geosci. 2005, 16, 680–683. [Google Scholar]
- Cui, Z.D.; Liu, D.A.; Zeng, R.S.; Tian, T. Geological Sequestration of CO2 and China’s Sustainable Development. China Popul. Resour. Environ. 2010, 20, 9–13. [Google Scholar]
- Kuang, G.S. Global warming and carbon dioxide mitigate. Modern Chem. Ind. 2007, 27, 1–12. [Google Scholar]
- Ebinuma, T. Method for dumping and disposing of carbon dioxide gas and apparatus therefore. U.S. Patent 5,261,490, 16 November 1993. [Google Scholar]
- Ohgaki, K.; Takano, K.; Sangawa, H.; Matsubara, T.; Nakano, S. Methane exploitation by carbon dioxide from gas hydrates-phase equilibria for CO2-CH4 mixed hydrate system. J. Chem. Eng. Jpn. 1996, 29, 478–483. [Google Scholar] [CrossRef]
- Wang, J.B.; Guo, X.Q.; Chen, G.J.; Li, Z.Z.; Yang, L.Y. Experimental research on methane recovery from natural gas hydrate by carbon dioxide replacement. J. Chem. Eng. Chin. Univ. 2007, 21, 715–719. [Google Scholar]
- Ota, M.; Abe, Y.; Watanabe, M.; Smith, R.L.; Inomata, H. Methane recovery from methane hydrate using pressurized CO2. Fluid Phase Equilib. 2005, 228, 553–559. [Google Scholar] [CrossRef]
- Li, Z.Z.; Guo, X.Q.; Wang, J.B.; Yang, L.Y. Experiment studies on CH4 recovery from hydrate using CO2 in different systems. Natu. Gas Ind. 2008, 28, 129–132. [Google Scholar]
- Li, Z.Z.; Guo, X.Q.; Chen, G.J.; Wang, J.B.; Yang, L.Y.; Wang, T. Experimental and kinetic studies on methane replacement from methane hydrate formed in SDS system by using pressurized CO2. J. Chem. Ind. Eng. (China) 2007, 58, 1197–1203. [Google Scholar]
- Sloan, E.D.; Koh, C.A. Estimation Techniques for Phase Equilibria of Natural Gas Hydrates. In Clathrate Hydrates of Natural Gases, 3rd ed.; Heinemann, H., Speight, J.C., Eds.; Taylor & Francis Group, LLC: New York, NY, USA, 2008; Chapter 6; pp. 320–523. [Google Scholar]
- Uchida, T.; Ikeda, I.Y.; TaKeya, S.; Kamata, Y.; Ohmura, R.; Nagao, J.; Zatsepina, O.Y.; Buffett, B.A. Kinetics and stability of CH4-CO2 mixed gas hydrates during formation and long-term storage. ChemPhysChem 2005, 6, 646–654. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.; Llamedo, M.; Tohidi, B.; Burgass, R.W. Experimental measurement of methane and carbon dioxide clathrate hydrate equilibria in mesoporous silica. J. Phys. Chem. B 2003, 107, 3507–3514. [Google Scholar] [CrossRef]
- Geng, C.Y.; Wen, H.; Zhou, H. Molecular simulation of the potential of methane reoccupation during the replacement of methane hydrate by CO2. J. Phys. Chem. A 2009, 113, 5463–5469. [Google Scholar] [CrossRef] [PubMed]
- Yezdimer, E.M.; Cummings, P.T.; Chialvo, A.A. Determination of the gibbs free energy of gas replacement in SI clathrate hydrates by molecular simulation. J. Phys. Chem. A 2002, 106, 7982–7987. [Google Scholar] [CrossRef]
- Uchida, T.; Takeya, S.; Ebinuma, T. Replacing Methane with CO2 in Clathrate Hydrate: Observation Using Raman Spectroscopy. In Proceedings of the 5th International Conference on Greenhouse Gas Control Technologies, Cairns, Australia, 13–16 September 2000.
- Yang, G.; Qi, Y.X.; Zhang, H.; Tang, C.W. Advancement in experimental research on replacement of CH4 from hydrate with CO2. Cryo. Supercond. 2010, 38, 70–75. [Google Scholar]
- Fan, Y.; Liu, D.P.; Xie, Y.M.; Zhong, D.L.; Xiao, Y. Feasibility Analysis on Replacement of CH4 from Hydrate Sediment by CO2. Nat. Gas Geosci. 2007, 18, 317–320. [Google Scholar]
- Ota, M.; Morohashi, K.; Abe, Y.; Watanabe, M.; Smith, R.L.; Inomata, H. Replacement of CH4 in the Hydrate by Use of Liquid CO2. Energy Convers. Manag. 2005, 46, 1680–1691. [Google Scholar] [CrossRef]
- Hirohama, S.; Shimoyama, Y.; Wakabayashi, A.; Tatsuta, S.; Nishida, N. Conversion of CH4-hydrate to CO2-hydrate in liquid CO2. J. Chem. Eng. Jpn. 1996, 29, 1014–1020. [Google Scholar] [CrossRef]
- van der Waals, J.H.; Platteeuw, J.C. Clathrate solutions. Adv. Chem. Phys. 1959, 2, 1–57. [Google Scholar]
- Soave, G. Equilibrium constants from a modified Redlich-Kwong equation of state. Chem. Eng. Sci. 1972, 27, 1197–1203. [Google Scholar] [CrossRef]
- Nagayev, V.B.; Gritsenko, A.I.; Murin, V.I. CO2 Hydrates and CO2 Sequestration. In Proceedings of the All Union Conference on Calorimetry and Chemical Thermodynamics, Ivonovo, Russia, 25–27 September 1979.
- Rueff, R.M.; Sloan, E.D.; Yesavage, V.F. Heat-capacity and heat of dissociation of methane hydrates. AIChE J. 1988, 34, 1468–1476. [Google Scholar] [CrossRef]
- Zhou, X.T.; Fan, S.S.; Liang, D.Q.; Du, J.W. Replacement of methane from quartz sand-bearing hydrate with carbon dioxide-in-water emulsion. Energy Fuels 2008, 22, 1759–1764. [Google Scholar] [CrossRef]
- Xiong, L.J.; Li, X.S.; Zeng, Z.Y.; Li, G.; Chen, Z.Y.; Zhang, Y.; Li, Q.P. Experimental Investigation into Replacement of CH4 in Hydrate in Porous Sediment with Liquid CO2 injection. In Proceedings of the 7th International Conference on Gas Hydrates (ICGH), Edinburgh, UK, 17–21 July 2011.
- Yoon, J.H.; Kawamura, T.; Yamamoto, Y.; Komai, T. Transformation of methane hydrate to carbon dioxide hydrate: in situ Raman spectroscopic observations. J. Phys. Chem. A 2004, 108, 5057–5059. [Google Scholar] [CrossRef]
- McGrail, B.P.; Zhu, T.; Hunter, R.B.; White, M.D.; Patil, S.L.; Kulkarni, A.S. A New Method for Enhanced Production of Gas Hydrate with CO2. In Proceedings of the AAPG Hedberg Conference on Gas Hydrates: Energy Resource Potential and Associated Geologic Hazards, Vancouver, Canada, 12–16 September 2004.
- White, M.; McGrail, P. Designing a Pilot-Scale Experiment for the production of natural gas hydrates and sequestration of CO2 in class 1 hydrate accumulations. Energy Procedia 2009, 1, 3099–3106. [Google Scholar] [CrossRef]
- DhanuKa, V.V.; DicKson, J.L.; Ryoo, W.; Johnston, K.P. High internal phase CO2-in-water emulsions stabilized with a branched nonionic hydrocarbon surfactant. J. Colloid Interface Sci. 2006, 298, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.T.; Fan, S.S.; Liang, D.Q.; Du, J.W. Determination of appropriate condition on replacing methane from hydrate with carbon dioxide. Energy Convers. Manag. 2008, 49, 2124–2129. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, Z.; Li, W.Q.; Li, W.Y.; He, D.W. Research progress in the enhanced replacing methane out of gas hydrate by carbon dioxide emulsion. Nat. Gas Chem. Eng. 2009, 34, 59–63. [Google Scholar]
- Kvamme, B.; Graue, A.; Aspenes, E.; Kuznetsova, T.; Gránásy, L.; Tóth, G.; Pusztai, T.; Tegzeb, G. Kinetics of solid hydrate formation by carbon dioxide Phase field theory of hydrate nucleation and magnetic resonance imaging. Phys. Chem. Chem. Phys. 2004, 6, 2327–2334. [Google Scholar] [CrossRef]
- Kvamme, B.; Graue, A.; Buanes, T.; Kuznetsova, T.; Ersland, G. Storage of CO2 in natural gas hydrate reservoirs and the effect of hydrate as an extra sealing in cold aquifers. Int. J. Greenh. Gas Control 2007, 1, 236–246. [Google Scholar] [CrossRef]
- Baldwin, B.A.; Stevens, J.; Howard, J.J.; Graue, A.; Kvamme, B.; Aspenes, E.; Ersland, G.; Husebø, J.; Zornes, D.R. Using magnetic resonance imaging to monitor CH4 hydrate formation and spontaneous conversion of CH4 to CO2 hydrate in porous media. Magn. Reson. Imaging 2009, 27, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Ersland, G.; Husebø, J.; Graue, A.; Baldwin, B.A.; Howard, J.; Stevens, J. Measuring gas hydrate formation and exchange with CO2 in Bentheim sandstone using MRI tomography. Chem. Eng. J. 2010, 158, 25–31. [Google Scholar] [CrossRef]
- Tegze, G.; Pusztai, T.; Tóth, G.; Gránásy, L.; Svandal, A.; Buanes, T.; Kuznetsova, T.; Kvamme, B. Multiscale approach to CO2 hydrate formation in aqueous solution: Phase field theory and molecular dynamics. Nucleation and growth. J. Chem. Phys. 2006, 124, 234710:1–234710:12. [Google Scholar] [CrossRef]
- Tegze, G.; Gránásy, L.; Kvamme, B. Phase field modeling of CH4 hydrate conversion into CO2 hydrate in the presence of liquid CO2. Phys. Chem. Chem. Phys. 2007, 9, 3014–3111. [Google Scholar] [CrossRef]
- Zhou, W.; Fan, S.S.; Liang, D.Q.; Li, D.L.; Tang, C.P.; Tian, G.L. Influence of pressure to replacement of CH4 in the hydrate by use of CO2. J. Wuhan Univ. Technol. (Transp. Sci. Eng.) 2008, 32, 547–550. [Google Scholar]
- Qi, Y.X.; Zhang, H.; Zhao, S.X.; Ji, L.M. Experimental Research of Thermal Effects on Replacement of Methane Hydrates with Carbon Dioxide. In Proceedings of the 7th International Conference on Gas Hydrates (ICGH), Edinburgh, UK, 17–21 July 2011.
- Ota, M.; Saito, T.; Aida, T.; Watanabe, M.; Sato, Y.; Smith, R.L.; Inomata, H. Macro and microscopic CH4-CO2 replacement in CH4 hydrate under pressurized CO2. AIChE J. 2007, 53, 2715–2721. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, J.; Xu, K.; Song, Y.; Liu, W.; Lam, W.; Liu, Y.; Xue, K.; Zhu, Y.; Yu, X.; Li, Q. A Review on Research on Replacement of CH4 in Natural Gas Hydrates by Use of CO2. Energies 2012, 5, 399-419. https://doi.org/10.3390/en5020399
Zhao J, Xu K, Song Y, Liu W, Lam W, Liu Y, Xue K, Zhu Y, Yu X, Li Q. A Review on Research on Replacement of CH4 in Natural Gas Hydrates by Use of CO2. Energies. 2012; 5(2):399-419. https://doi.org/10.3390/en5020399
Chicago/Turabian StyleZhao, Jiafei, Kun Xu, Yongchen Song, Weiguo Liu, Weihaur Lam, Yu Liu, Kaihua Xue, Yiming Zhu, Xichong Yu, and Qingping Li. 2012. "A Review on Research on Replacement of CH4 in Natural Gas Hydrates by Use of CO2" Energies 5, no. 2: 399-419. https://doi.org/10.3390/en5020399
APA StyleZhao, J., Xu, K., Song, Y., Liu, W., Lam, W., Liu, Y., Xue, K., Zhu, Y., Yu, X., & Li, Q. (2012). A Review on Research on Replacement of CH4 in Natural Gas Hydrates by Use of CO2. Energies, 5(2), 399-419. https://doi.org/10.3390/en5020399




