Biofuels Production through Biomass Pyrolysis —A Technological Review
Abstract
:1. Introduction
2. Pyrolysis Principles
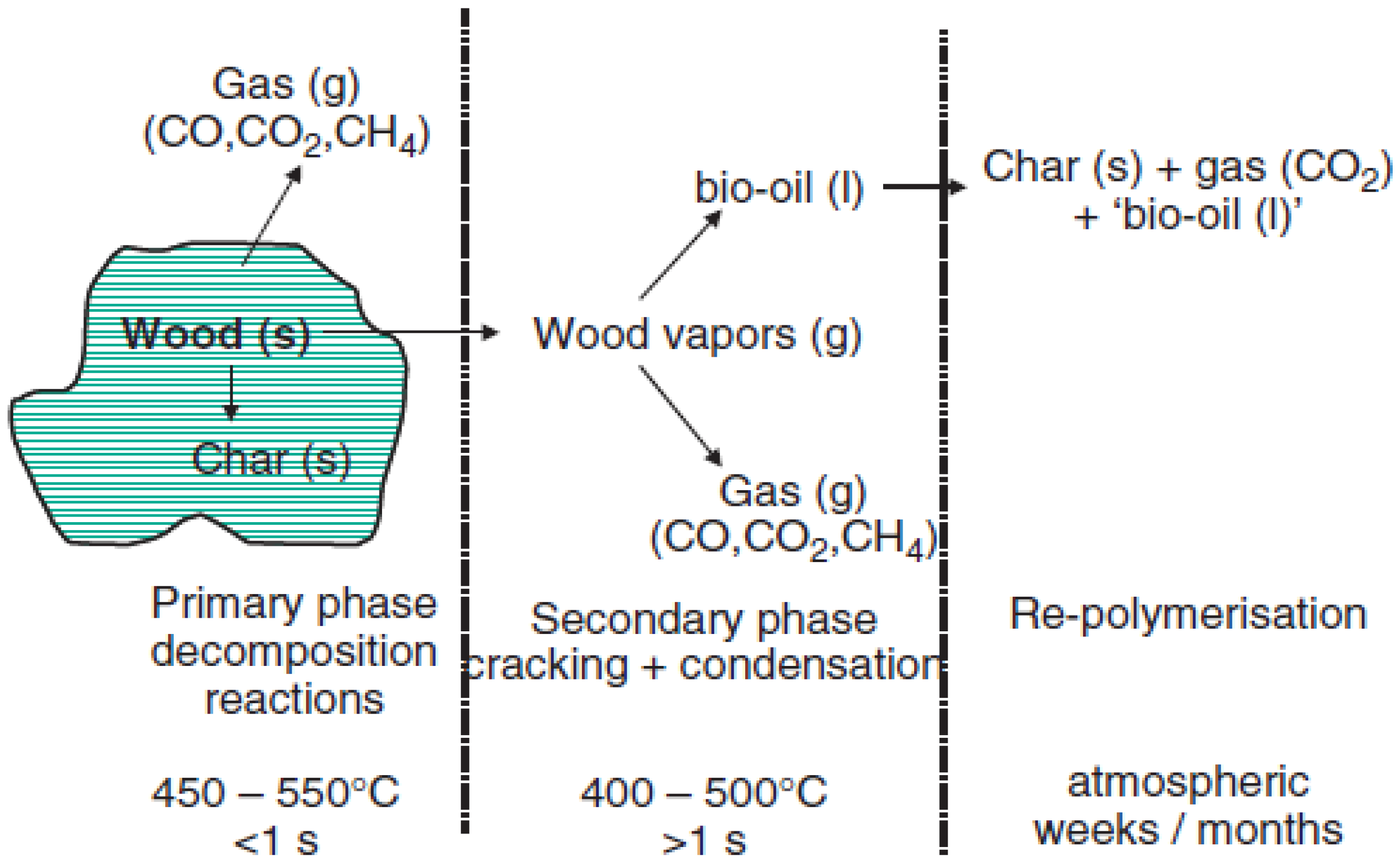
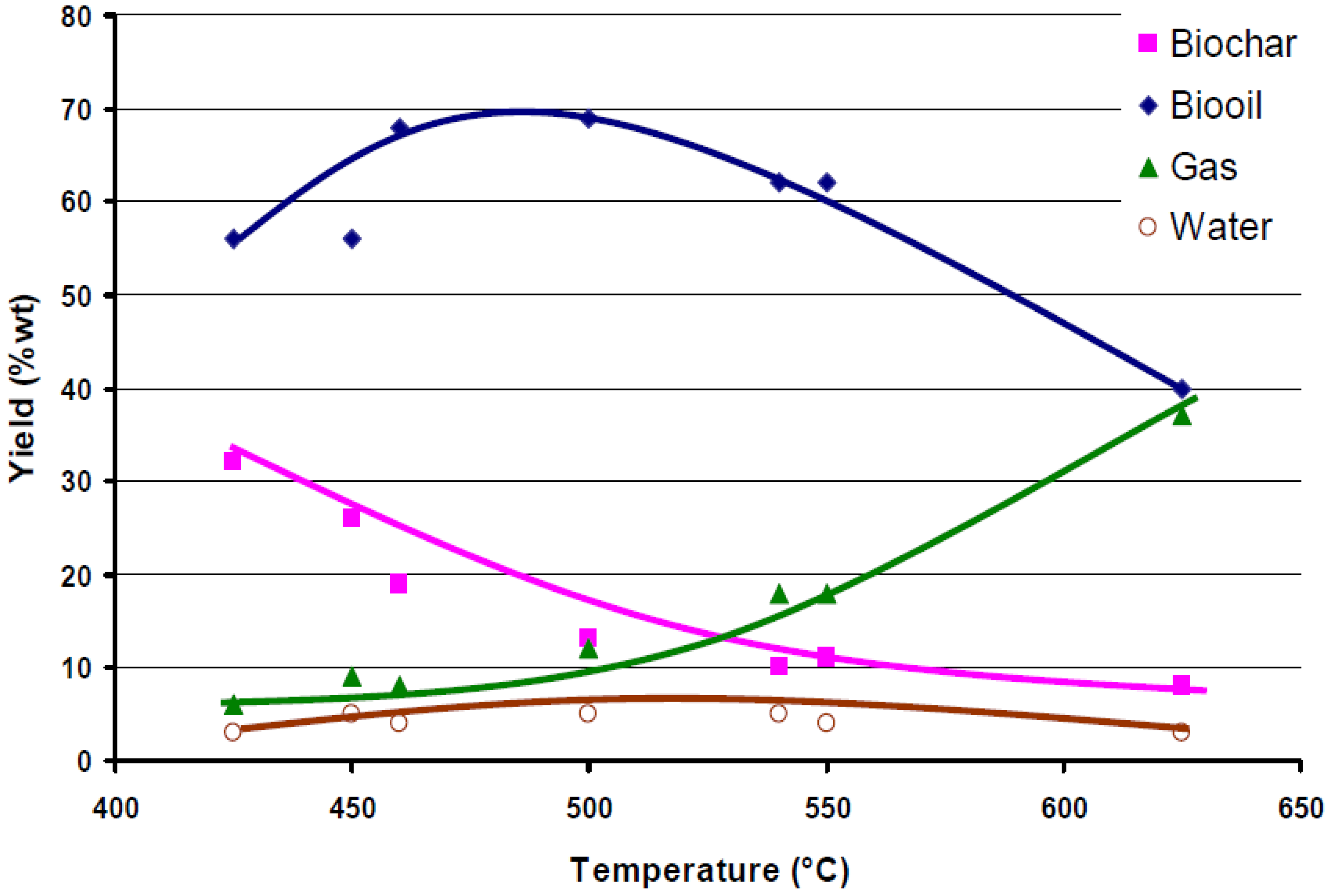
3. Pyrolysis Classification
| Pyrolysis Process | Solid Residence Time (s) | Heating Rate (K/s) | Particle Size (mm) | Temp. (K) | Product Yield (%) | ||
|---|---|---|---|---|---|---|---|
| Oil | Char | Gas | |||||
| Slow | 450–550 | 0.1–1 | 5–50 | 550–950 | 30 | 35 | 35 |
| Fast | 0.5–10 | 10–200 | <1 | 850–1250 | 50 | 20 | 30 |
| Flash | <0.5 | >1000 | <0.2 | 1050–1300 | 75 | 12 | 13 |
3.1 Slow Pyrolysis
3.2 Fast Pyrolysis
- Renewable fuel for boiler, engine, turbine, power generation and industrial processes;
- Low cost and neutral CO2 balance;
- Utilisation of second generation bio-oil feed stocks and waste materials (forest residue, municipal and industrial waste, etc.);
- Storability and transportability of liquid fuels;
- High energy density compared to atmospheric biomass gasification fuel gases;
- Possibility for separating minerals on the site of liquid fuel production to be recycled to the soil as a nutrient;
- Secondary conversion to motor-fuels, additives or special chemicals;
- Primary separation of the sugar and lignin fractions in biomass with subsequent further upgrading.
3.3 Flash Pyrolysis
4. Biomass Feedstock
4.1. Composition of Biomass
| Feedstock | Lignin (%) | Cellulose (%) | Hemicellulose (%) |
|---|---|---|---|
| Wood | 25–30 | 35–50 | 20–30 |
| Wheat straw | 15–20 | 33–40 | 20–25 |
| Switch grass | 5–20 | 30–50 | 10–40 |
| Sugarcane bagasse | 23–32 | 19–24 | 32–48 |
| Miscanthus | 17 | 24 | 44 |
| Corn stover | 16–21 | 28 | 35 |
| Hazelnut shell | 42.9 | 28.8 | 30.4 |
| Olive husk | 48.4 | 24 | 23.6 |
| Corncob | 15 | 50.5 | 31 |
| Tea waste | 40 | 30.20 | 19.9 |
| Walnut shell | 52.3 | 25.6 | 22.7 |
| Almond shell | 20.4 | 50.7 | 28.9 |
| Sunflower shell | 17 | 48.4 | 34.6 |
| Nut Shell | 30–40 | 25–30 | 25–30 |
| Paper | 0–15 | 85–99 | 0 |
| Rice straw | 18 | 32.1 | 24 |
| Stored refuse | 20 | 60 | 20 |
| Leaves | 0 | 15–20 | 80–85 |
| Cotton seed hairs | 0 | 80–95 | 5–20 |
| Industrial waste paper | 5–10 | 60–70 | 10–20 |
| Barley straw | 14–15 | 31–34 | 24–29 |
| Oat straw | 16–19 | 31–37 | 24–29 |
| Bamboo | 21–31 | 26–43 | 15–26 |
| Rye straw | 16–19 | 33–35 | 27–30 |
| Coastal Bermuda grass | 6.4 | 25 | 35.7 |
| Jute fibre | 21–26 | 45–53 | 18–21 |
| Banana waste | 14 | 13.2 | 14.8 |
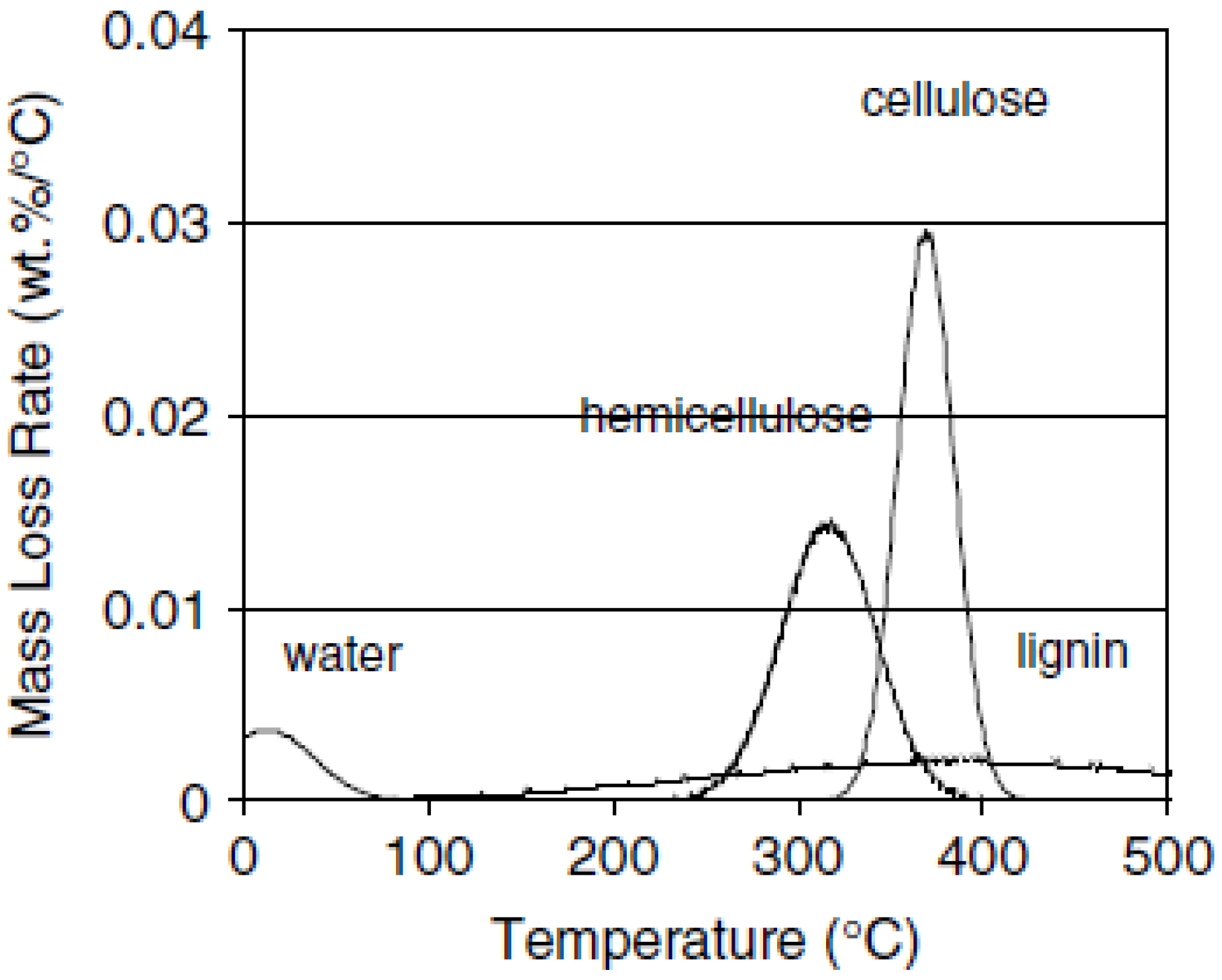
4.2 Physical and Chemical Characteristics of Biomass
| Feedstock | Density (Kg/m3) | Moisture Content (%) | Ash Content (%) | Volatile Matter (%) | Fixed Carbon (%) |
|---|---|---|---|---|---|
| Wood | 1186 | 20 | 0.4–1 | 82 | 17 |
| Bituminous coal | 11 | 8–11 | 35 | 45 | |
| Hybrid polar | 150 | 45 | 0.5–2 | - | - |
| Switchgrass | 108 | 13–15 | 4.5–5.8 | - | - |
| Miscanthus | 70–100 | 11.5 | 1.5–4.5 | 66.8 | 15.9 |
| Sugarcane baggage | 1198 | 3.2–5.5 | - | - | |
| Barley strew | 210 | 30 | 6 | 46 | 18 |
| Wheat straw | 1233 | 16 | 4 | 59 | 21 |
| Danish pine | 8 | 1.6 | 71.6 | 19 | |
| Rice straw | 200 | 6 | 4.3 | 79 | 10.7 |
| Fire wood | - | 7.74 | 1.98 | 80.86 | 17.16 |
| Grateloupia filicina | - | 4.93 | 22.37 | 55.93 | 17.01 |
| Birch | 125 | 18.9 | 0.004 | - | 20 |
| Pine | 124 | 17 | 0.03 | - | 16 |
| Polar | 120 | 16.8 | 0.007 | - | - |
| Feedstock | Carbon (%) | Hydrogen (%) | Oxygen (%) | Nitrogen (%) | Ash (%) |
|---|---|---|---|---|---|
| Wood | 51.6 | 6.3 | 41.5 | 0.1 | 1 |
| Cypress | 55 | 6.5 | 38.1 | - | 0.4 |
| Olive baggage | 66.9 | 9.2 | 21.9 | 2 | - |
| Wheat straw | 48.5 | 5.5 | 3.9 | 0.3 | 4 |
| Barley straw | 45.7 | 6.1 | 38.3 | 0.4 | 6 |
| Bituminous coal | 73.1 | 5.5 | 8.7 | 1.4 | 9 |
| Scots | 56.4 | 6.3 | - | 0.1 | 0.09 |
| Birch | 44 | 6.9 | 49 | 0.1 | .004 |
| Pine | 45.7 | 7 | 47 | 0.1 | 0.03 |
| Polar | 48.1 | 5.30 | 46.10 | 0.14 | 0.007 |
| Willow | 47.78 | 5.90 | 46.10 | 0.31 | 1.30 |
| Switchgrass | 44.77 | 5.79 | 49.13 | 0.31 | 4.30 |
| Reed canary grass | 45.36 | 5.81 | 48.49 | 0.34 | 5.10 |
| Dactylis lomarata | 42.96 | 5.70 | 49.44 | 1.90 | 7.50 |
| Festuca arundinacea | 42.22 | 5.64 | 50.65 | 1.50 | 7.30 |
| Lolium perenne | 43.12 | 5.80 | 49.80 | 1.28 | 6.20 |
5. Pyrolysis Reactor
5.1 Fixed Bed Reactor
5.2 Fluidized-Bed Reactor
5.2.1 Bubbling Fluidized-Bed Reactors
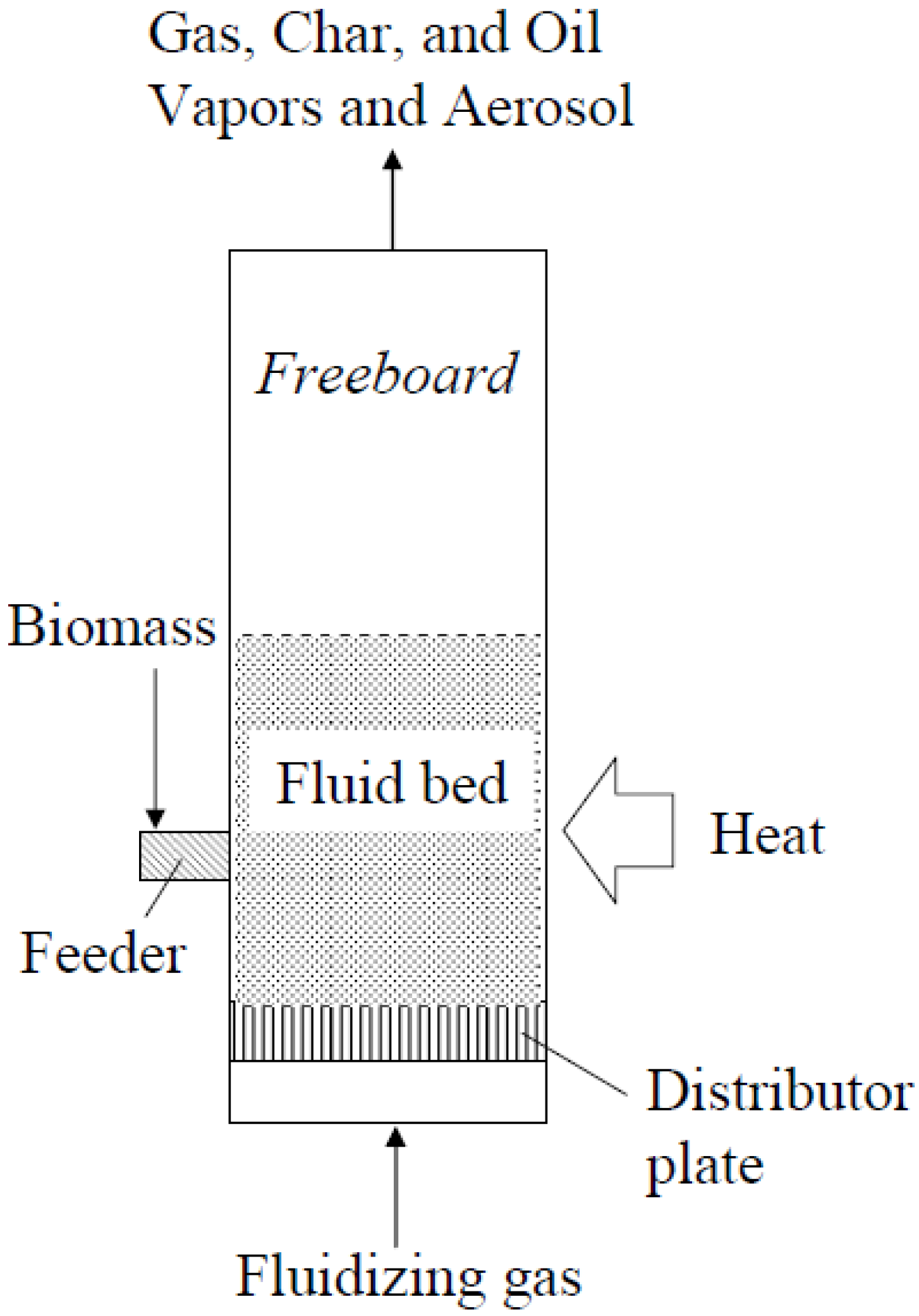
5.2.2 Circulating Fluidized-Bed Reactors
5.3 Ablative Reactor
5.3.1 Vortex Reactor
5.3.2 Rotating Disk Reactor
5.4 Vacuum Pyrolysis Reactor
5.5 Rotating Cone Reactor
5.6 PyRos Reactor
5.7 Auger Reactor
5.8 Plasma Reactor
5.9 Microwave Reactor
5.10 Solar Reactor
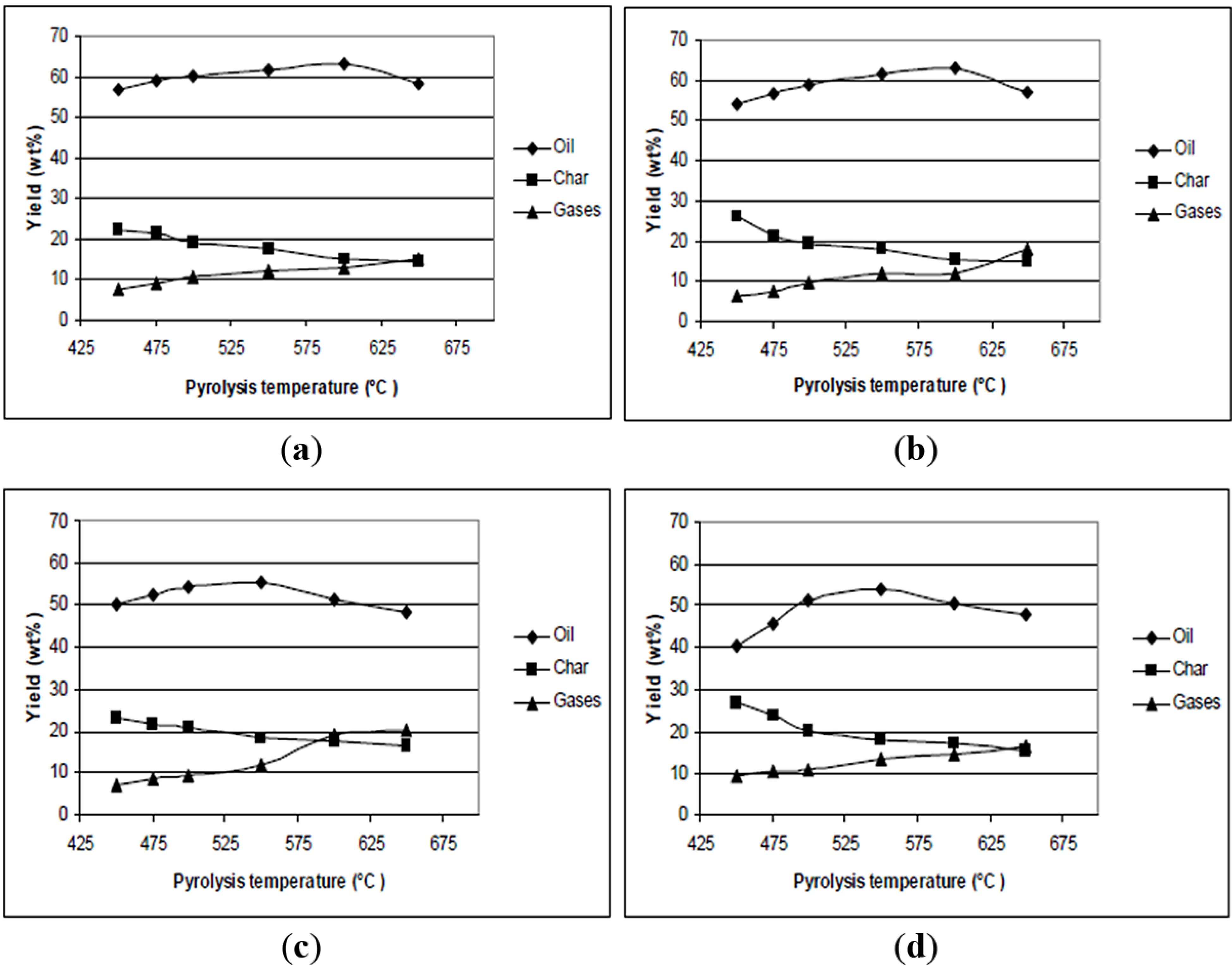
6. Pyrolysis Process Description
6.1 Feed Preparation
6.2 Biomass Heating
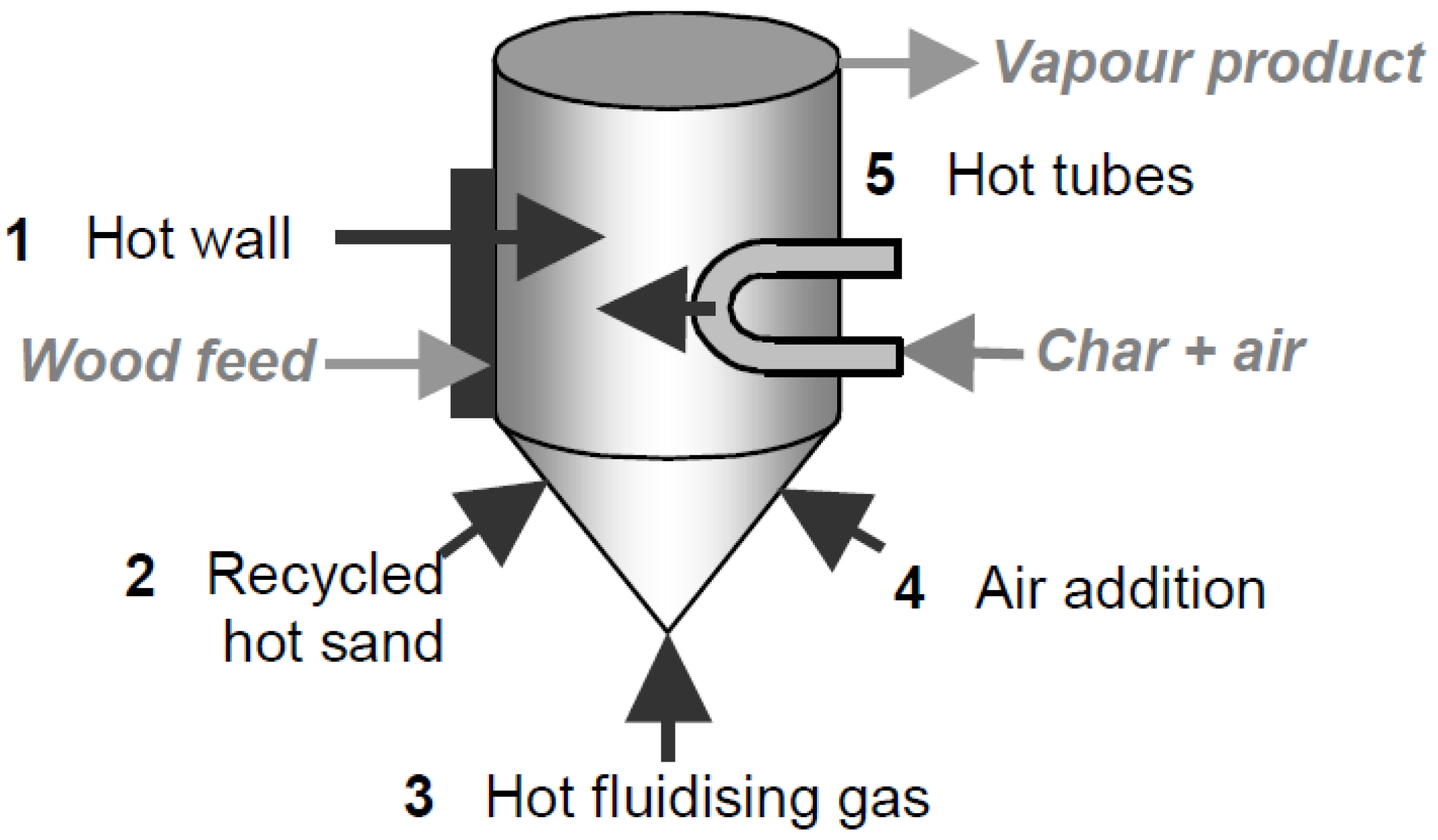
| Heating Method | Reactor Type |
|---|---|
| Heated recycle gas | Bubbling fluidized bed |
| Wall and sand heating | Circulating fluidized bed |
| Gasification of char to heat sand | Rotating cone |
| Direct contract with hot surface | Vacuum |
| Wall heating | Ablative |
| Fire tube | Auger |
| Radio-frequency | Plasma |
| Electromagnetic | Microwave reactor |
| Solar | Fluidized bed/Quartz |
6.3 Char Separation
6.4 Liquids Collection
7. Pyrolysis Products
| Condition | Processes | Products | |||
|---|---|---|---|---|---|
| Below 350 °C | Free radical formation, water elimination and depolymerization | Formation of carbonyl and carboxyl, evolution of CO and CO2, and mainly a charred residue | |||
| Between 350 °C and 450 °C | Breaking of glycosidic linkages of polysaccharide by substitution | Mixture of levoglucosan, anhydrides and oligosaccharides in the form of a tar fraction | |||
| Above 450 °C | Dehydration, rearrangement and fission of sugar units | Formation of carbonyl compounds such as acetaldehyde, glyoxal and acrolein | |||
| Above 500 °C | A mixture of all above processes | A mixture of all above products | |||
| Condensation | Unsaturated products condense and cleave to the char | A highly reactive char residue containing trapped free radicals |
7.1 Pyrolysis Bio-Oil
- CO2 balance is clearly positive in biomass fuel;
- Possibility of utilisation in small-scale power generation systems as well as use in large power stations;
- Storability and transportability of liquid fuel;
- High-energy density compared to biomass gasification fuel;
- Potential of using pyrolysis liquid in existing power plants.
| Properties | Oil Characteristics | Reasons | |||
|---|---|---|---|---|---|
| Appearance | Dark red-brown to dark green | Micro-carbon and chemical composition in oil | |||
| Odor | Distinctive odor—an acrid smoky smell | Lower molecular weight aldehydes and acids | |||
| Density | Very high compared to fossil fuel Pyrolysis bio-oil: 1.2 kg/liter Fossil oil: 0.85 kg/liter | High moisture and heavy molecule contamination | |||
| Viscosity | Can vary from as low as 25 centistokes (cSt) to as high as 1000 cSt | Wide range of feedstock, water content and the amount of light ends collected | |||
| Heating value | Significantly lower than fossil oil | High oxygen content | |||
| Aging | Viscosity increase, volatility decrease, phase separation and deposition of gum occur with time | Complex structure and high pH value | |||
| Miscibility | Miscible with polar solvent but totally immiscible with petroleum fuel | Polar in nature |
| Moisture Content (wt%) | pH | Viscosity mm2/s | HHV (MJ/kg) | LHV (MJ/kg) | |
|---|---|---|---|---|---|
| Diesel | - | 1 | 2.39 | 44.7 | 42.5 |
| Wood | 15–30 | 2.5 | 40–100 | 16 | 19 |
| Willow | 17.4 | 2.68 | 53.2 | 18.4 | 16.8 |
| Straw | 47.4 | 3.45 | 17.2 | 13.6 | 11.6 |
| Sweet grass | 24.7 | 2.87 | 34.2 | 16.4 | 14.8 |
| Dactylis glomerata | 43.05 | 3.12 | 9.2 | 17.6 | 15.7 |
| Festuca arundiance | 34.1 | 3.18 | 10.9 | 16.7 | 14.5 |
| Lonium perenne | 48 | 3.16 | 6.5 | 15.8 | 13.7 |
| Reed canary grass | 23.2 | 3.01 | 31.6 | 17.1 | 15.3 |
| Rice husk | 27.2 | 2.8 | 128 | - | 16.4 |
| Corn stalk | 26.9 | 2.3 | 140 | - | 16.8 |
| Cotton talk | 26.7 | 2.6 | 156 | - | 17.2 |
| Sawdust | 24 | 2.1 | 140 | - | 17.4 |
| Palm shells | 10 | 2.7 | 14.6 | - | 20.6 |
| C (wt%) | H (wt%) | N (wt%) | S (wt%) | O (wt%) | |
|---|---|---|---|---|---|
| Diesel | 86 | 11.1 | 1 | 0.80 | 0 |
| Wood | 55–58 | 5.5–7 | 0–0.2 | 0 | 35–40 |
| Willow | 43.17 | 7.15 | 0.10 | 0.10 | 49.49 |
| Straw | 28.2 | 8.78 | 0.10 | 0.10 | 62.83 |
| Sweet grass | 38.3 | 7.42 | 0.10 | 0.10 | 54.08 |
| Dactylis glomerata | 36.75 | 8.82 | 1.88 | 0.10 | 52.46 |
| Festuca arundiance | 32.05 | 9.76 | 1.41 | 0.10 | 56.69 |
| Lonium perenne | 30.64 | 9.63 | 0.77 | 0.10 | 58.86 |
| Reed canary grass | 38.42 | 7.89 | 0.10 | 0.10 | 53.49 |
| Sesame stalk | 61.69 | 7.96 | 29.37 | 0.98 | - |
| Rapeseed | 74.04 | 10.29 | 11.70 | 3.97 | - |
| Miscanthus | 48.1 | 5.4 | 42.2 | 0.5 | .01 |
| Cherry stone | 46.9 | 7.9 | 45.1 | 0.1 | - |
| Sunflower oil cake | 66.5 | 9.2 | 4.5 | 19.8 | - |
| Sugar cane bagasse | 46.27 | 6.55 | 46.9 | - | 0.1–0.15 |
| Jute stick | 47.18 | 8.36 | 44.1 | - | - |
| Rice straw | 41.4 | 5 | 39.9 | 0.7 | 0.1 |
| Palm shells | 53.5 | 6.7 | 0.44 | 0.02 | 39.37 |
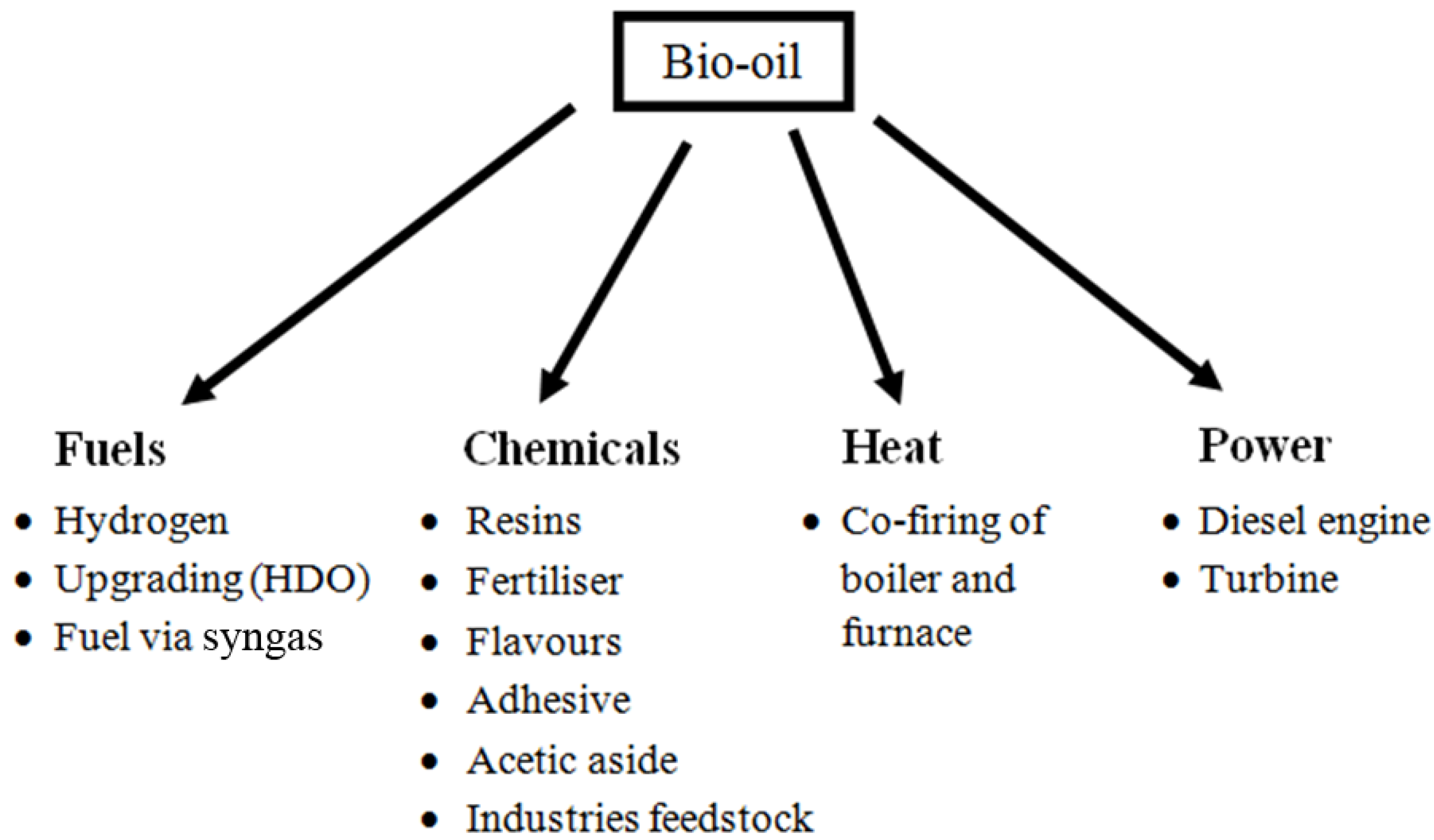
7.1.1 Furnaces and Boilers
- Pyrolysis bio-oils have significantly different combustion characteristics compared to fossil fuels;
- Bio-oils with high viscosity and high solids and water content exhibit worse combustion performances in boilers;
- Different pyrolysis bio-oils differ in combustion behaviour and exhaust gas emissions;
- The flame from bio-oil combustion is longer compared to that of standard fossil oil;
- Harmful gas emissions from pyrolysis bio-oil in boiler applications are lower than from burning heavy fuel oils except for particulate levels;
- Some modifications of the burners and boilers are required for proper utilization of pyrolysis bio-oil in heat and power generation.
7.1.2 Diesel Engines
7.1.3 Gas Turbines
7.1.4 Chemicals
7.2 Bio-Char From Pyrolysis
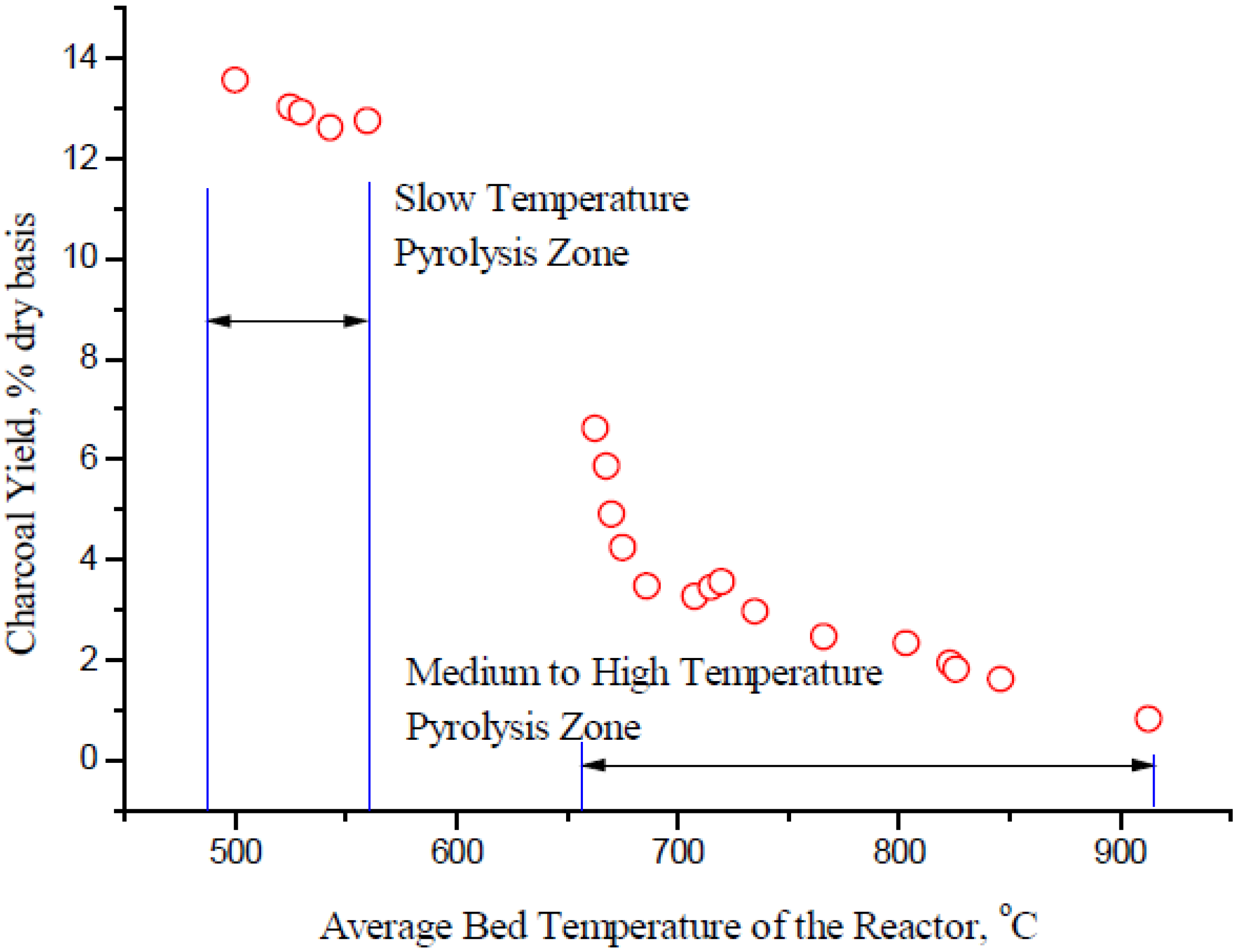
7.3 Syngas
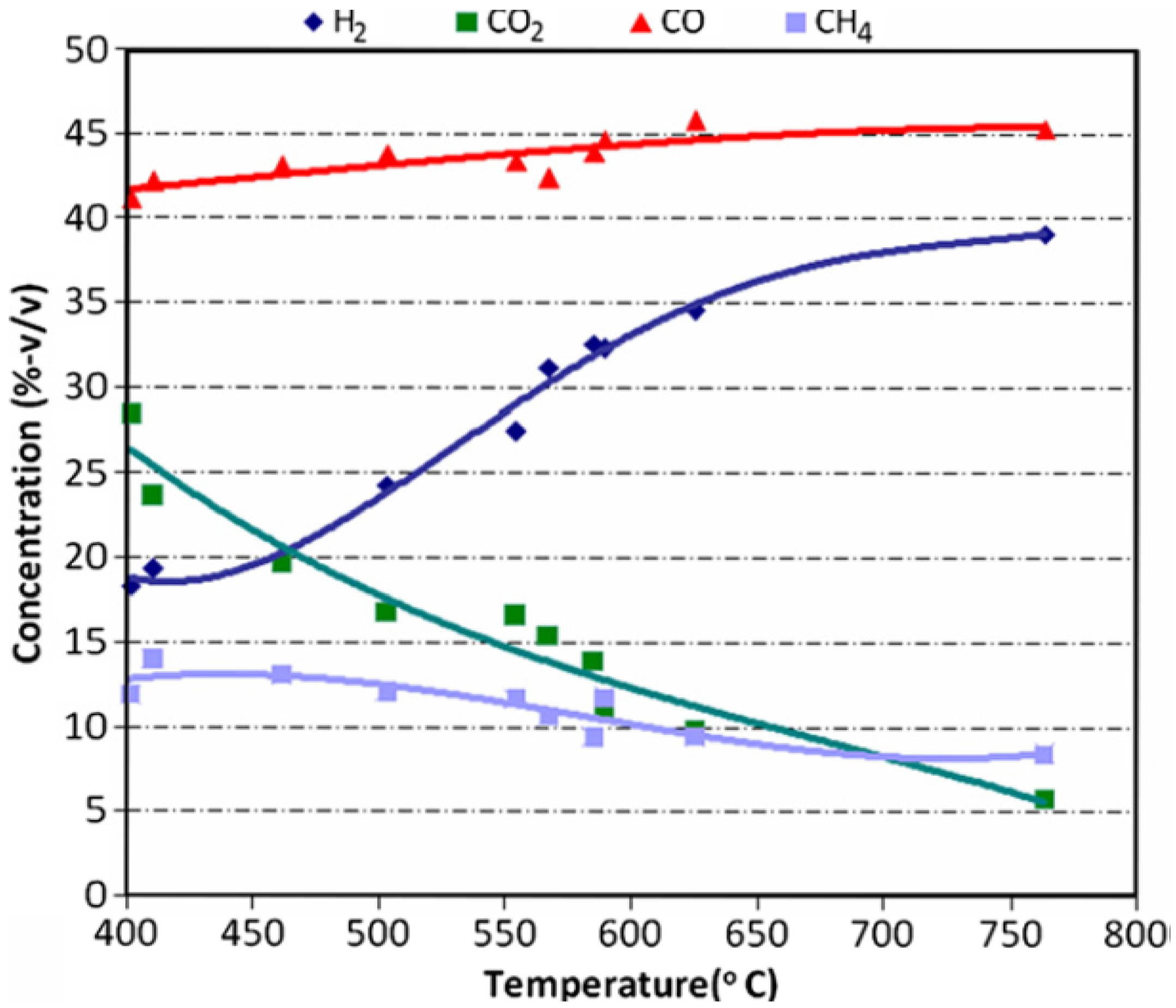
8. Catalysts in Pyrolysis
9. Energy Consumption in Pyrolysis
10. Pyrolysis Economics
| Items | Percentages |
|---|---|
| Biomass harvesting or feedstock | 23%–30% |
| Maintenance | 17%–24% |
| Utilites | 22%–25% |
| Labor | 12%–19% |
| Grinding | 7%–9% |
| Transportation | 5%–7% |
| Plant capacity | 0.3, 100 and 1000 kg/h rice husk | ||
| Plant life | 10 years | ||
| Annual operating time | 3120 h @ 10 h/day | ||
| Maintenance labour | 1% of FCI | ||
| Maintenance materials | 3% of FCI | ||
| Overheads | 2% of FCI | ||
| Insurance | 2% of FCI | ||
| Other fixed operating costs | 1% of FCI | ||
| Interest rate | 10% | ||
| Feedstock cost | 20 US$/ton | ||
| Labour hire rate | 1 US$/h | ||
| Number of labours | 1, 5 and 20 respectively for the three different plant capacity mentioned above | ||
| Nitrogen gas price | 5 US$ per cylinder of 6.3 m3 | ||
| Electricity price | 0.04 US$/kWh | ||
| Catalyst cost | 100 US$/kg | ||
| Catalytic life | 1 year |
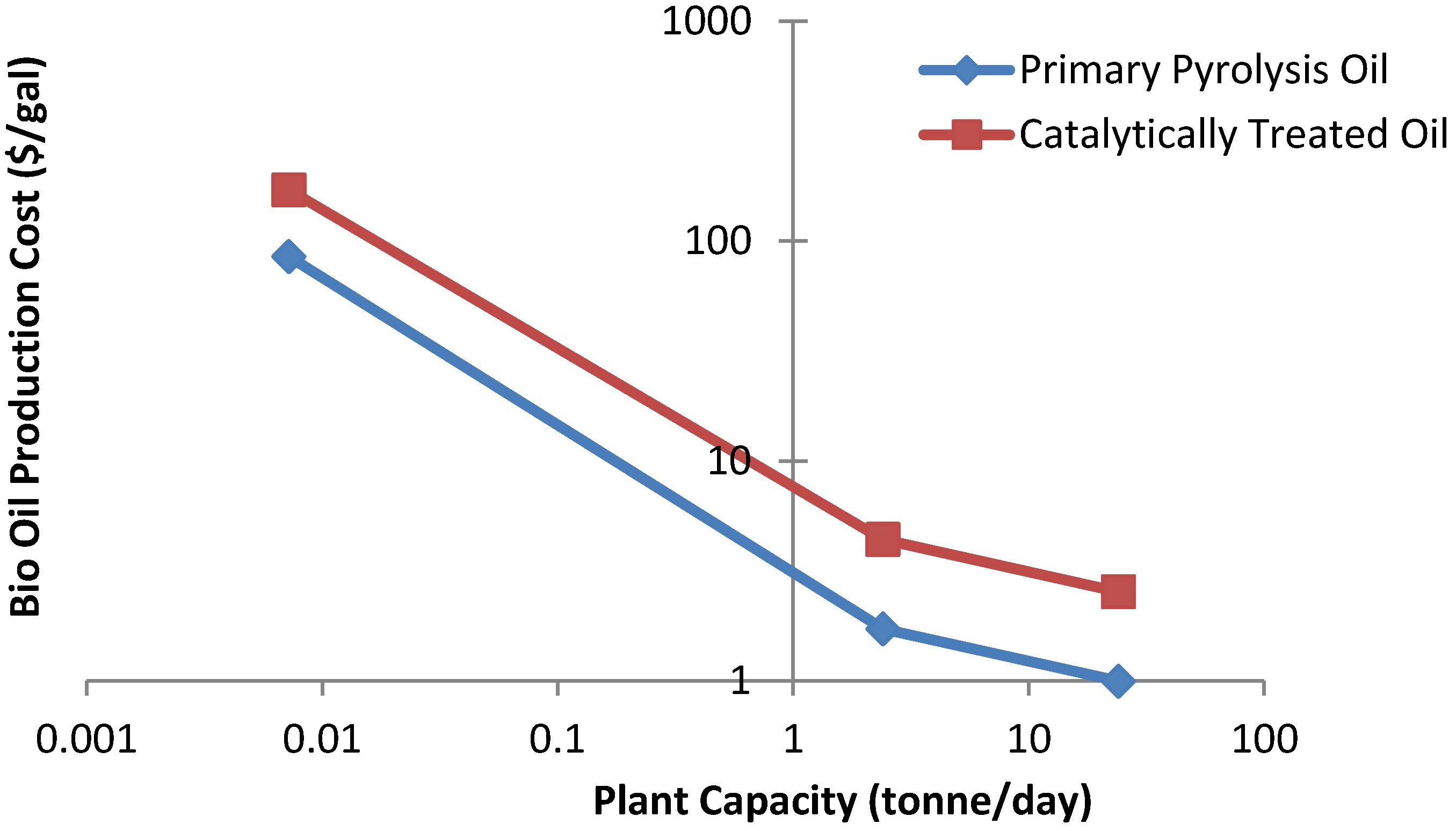
| Plant Size (tonne/day) | Feedstock | Capital Investment (million $) | Annual Operating Costs (M $) | Feed Costs ($/tonne) | Production Costs ($/gal) | References | Type of pyrolysis process |
|---|---|---|---|---|---|---|---|
| 2000 | Corn Stover | 200 | 12.3 | 83 | 0.26 | [182] | Fast pyrolysis |
| 1650 | Wood pellet | 180 | 12 | - | 0.24 | [181] | Fast pyrolysis |
| 1000 | Dry wood | 68 | 10.6 | 44 | 0.41 | [184] | Fast pyrolysis |
| 1000 | Wet wood | 72 | 11.3 | 30 | 0.60 | [185] | Flash pyrolysis |
| 1000 | Peat | 76 | 10.2 | 20 | 0.61 | [96] | Fast pyrolysis |
| 1000 | Staw | 82 | 10.2 | 42.5 | 0.64 | [96] | Fast pyrolysis |
| 900 | Wet wood | 46 | 9.9 | 34 | 0.50 | [183] | Fast pyrolysis |
| 550 | Dry wood | 48.2 | 9.6 | 45 | 0.71 | [65] | Fast pyrolysis |
| 400 | Wet wood | 14.3 | 8.80 | 36 | 1.02 | [116] | Fast pyrolysis |
| 250 | Dry wood | 14 | 8.92 | 44 | 0.55 | [186] | Fast pyrolysis |
| 200 | Wet wood | 8.8 | 4.84 | 36 | 1.11 | [116] | Fast pyrolysis |
| 100 | Wet wood | 6.6 | 2.84 | 36 | 1.48 | [116] | Fast pyrolysis |
| 24 | Rice husk | 3.89 | 0.170 | 22 | 0.82 | [180] | Fast pyrolysis |
| 2.4 | Rice husk | 0.97 | 0.034 | 22 | 1.73 | [180] | Fast pyrolysis |
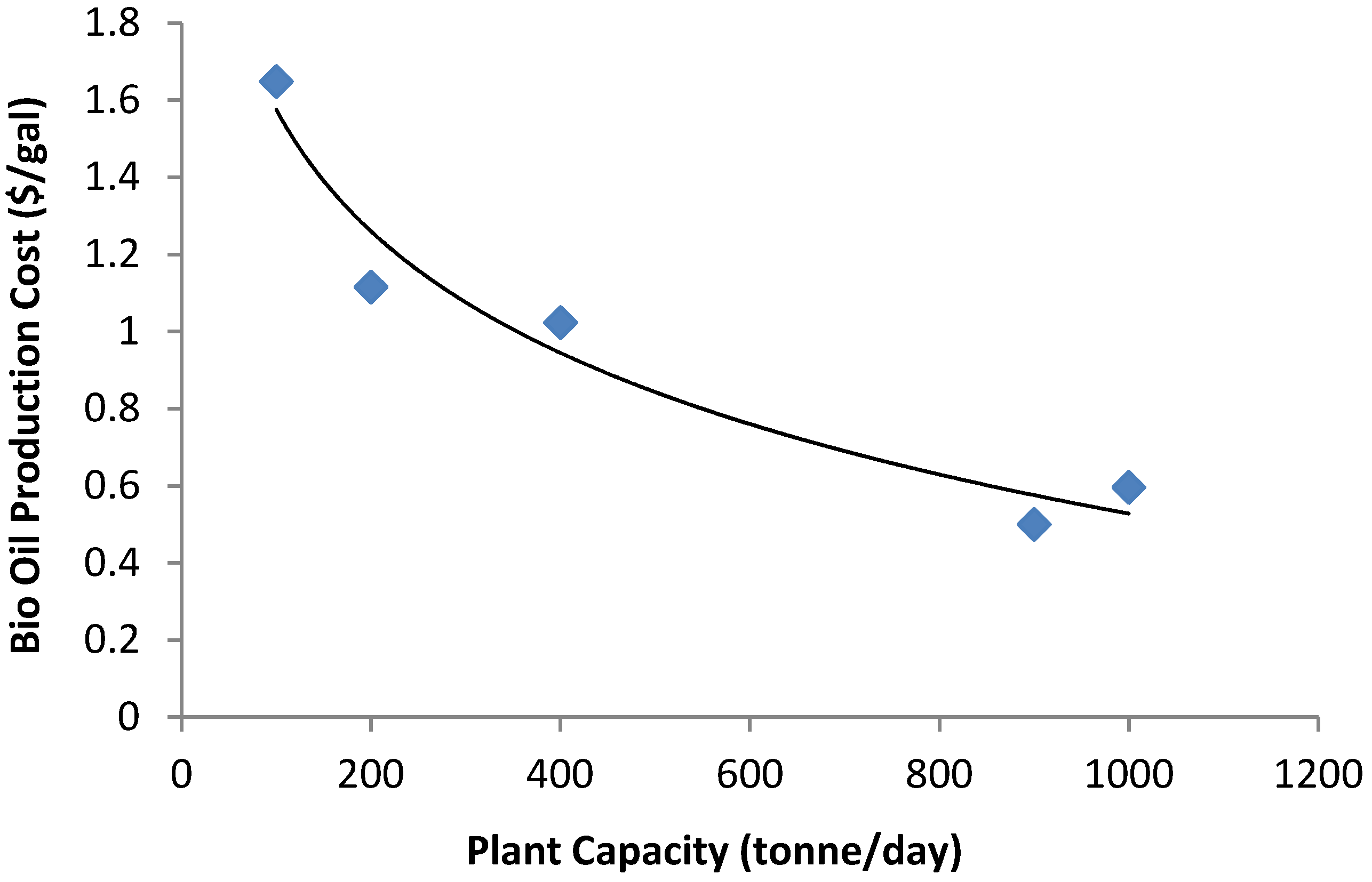
11. Discussion on Current Status of Pyrolysis Technology
| Reactor Technology | Organisation/Location | Capacity (kg/h) | Desired Product |
|---|---|---|---|
| Fixed bed | Bio-alternative, USA | 2000 | Char |
| Bubbling fluidised bed | THEE | 500 | Gas |
| DynaMotive, Canada | 400 | Oil | |
| BEST Energy, Australia | 300 | Oil | |
| Wellman, UK | 250 | Oil | |
| Union Fenosa, Spain | 200 | Oil | |
| Zhejiang University, China | 20 | Oil | |
| RTI, Canada | 20 | Oil | |
| Waterloo university | 3 | Oil | |
| Zhejiang University, China | 3 | Oil | |
| Circulating fluidised bed | Red Arrow, WI; Ensyn | 1700 | Chemicals |
| Red Arrow, WI; Ensyn | 1500 | Chemicals | |
| Ensyn Engineering | 30 | Oil | |
| VTT, Finland, Ensyn | 20 | Oil | |
| Rotating cone | BTG, Netherlands | 200 | Oil |
| University Twente | 10 | Oil | |
| Vacuum | Pyrovac, Canada | 350 | Oil |
| Laval University | 30 | Oil | |
| Ablative | PYTEC, Gernamy | 250 | Oil |
| BBC, Canada | 10-15 | Char | |
| PYTEC, Germany | 15 | Oil | |
| Vortex | Solar energy research Ins. | 30 | Oil |
| Other type | Fortum, Finland | 350 | Oil |
| University Zaragoza | 100 | Gas | |
| Georgia Tech. Research Ins. | 50 | Oil |
| Reactor type | Advantages | Disadvantages | Bio-Oil Yield |
|---|---|---|---|
| Fixed bed | Simple design Reliable Biomass size independent | High carbon conservation Long solid residence time Low ash carry over Difficult to remove char | 35%–50% |
| Bubbling fluidised bed | Simple design Easy operation Good temperature control Suitable for large scale | Small particle sizes are needed | 70%–75% |
| Circulating fluidised bed | Well-understood technology Good thermal control Large particle sizes can be used | Unlikely to be suitable for large scale Complex hydrodynamics Char is finer | 70%–75% |
| Rotating cone | Centrifugal force moves heated sand and biomass No carrier gas required Less wear | Complex process Small particle sizes needed Not proven yet for large scale | 65% |
| Vacuum | Produces clean oil Can process larger particles of 3–5 cm No carrier gas required Lower temperature required Easier liquid product condensation | Slow process Solid residence time is too high Require large scale equipment Poor heat and mass transfer rate Generates more water | 35%–50% |
| Ablative | Inert gas is not required Large particle sizes can be processed System is more intensive Moderate temperature required | Reactor is costly Low reaction rate Low reaction rate | 70% |
| Auger | Compact No carrier gas required Lower process temperature | Moving parts in hot zone Heat transfer in large scale is not suitable | 30%–50% |
| PyRos | Compact and low cos tHigh heat transfer Short gas residence time | Complex design Solids in the oil Alkali dissolved in the oil High temperature required | 70%–75% |
| Plasma | High energy density High heat transfer High temperature Very good control | High electrical power consumption High operating costs Small particle sizes required | 30%–40% |
| Microwave | Efficient heat transfer Exponential control Compact High heating rate Large size biomass can be processed Uniform temperature distribution High temperature | High electrical power consumption High operating costs | 60%–70% |
| Solar | Use renewable energy High heating rate High temperature | High costs Weather dependant | 40%–60% |
| Product | Pyrolysis Type | Reactor | Heating Method | Temp. (°C) | Biomass | |
|---|---|---|---|---|---|---|
| Bio-char | Slow | Fixed bed | Furnace or kilns | <300 | Walnut shell, olive husk, hazelnut shell | |
| Bio-oil | Large scale | Fast | Bubbling fluidised bed | Heated recycle gas | 450–550 | Agriculture residue, wood chip, fruit shell |
| Medium scale | Fast | Circulating fluidised bed | Wall and sand heating | 450–550 | Forest residue, municipal waste, dry wood, waste tyres | |
| Small scale | Flash | PyRos | PyRos heating | 450–550 | Grass, husk, wood dust | |
| Syngas | Slow/Fast | Microwave | Electromagnetic | >800 | Rice husk, wood dust | |
12. Computational Fluid Dynamics Modeling of Pyrolysis Process
13. Future Challenges
- Understand the trade-off between the size of the pyrolysis oil plant and feedstock and transportation costs to a centralized upgrading facility;
- Understand the limitations of the processes and where improvements can be made;
- Improvement of the reliability of pyrolysis reactors and processes;
- Identify norms and quality standards of pyrolysis bio-oil for producers and users;
- Undertake in-depth life cycle energy and economic analyses of integrated pyrolysis plants;
- Encourage implementation processes and applications of pyrolysis products;
- Document environmental health and safety issues in handling, transport and usages;
- Improve the quality and consistency of bio-oil;
- Development of catalysts for bio-oil upgrading;
- Determine detailed characteristics of upgraded oil and other products;
- Development of more efficient technologies for the production of chemicals and biofuels from pyrolysis oils;
- Development of deoxygenated catalysts for pyrolysis processes to remove the oxygen containing compounds in the oil to improve the oil properties.
14. Conclusions and Recommendations
- It is a research challenge to optimize the process by maximizing product quality and quantity while paying proper attention to minimizing costs and environmental concerns;
- For fast pyrolysis, a wide range of different reactor configurations have been investigated. The most important reactor types are bubbling fluidized bed, circulating fluidized bed, rotating cone, ablative and entrained flow reactors. Among those, fluidizing bed reactors are the most popular types because of ease of operation, high stability under pyrolysis conditions and high oil yields;
- Bio-oil production through pyrolysis is still an immature technology and is not commercially feasible yet. Pyrolysis bio-oil needs to overcome many technical, economic and social barriers to compete with tradition fossil fuels. Effective and rapid char separation techniques need to be developed so as to reduce solids contamination in bio-oil;
- Along with pyrolysis technology, proper biomass selection is also a critical issue to achieve high bio-oil yields. Biomass with high cellulose content could be chosen, as bio-oils are mainly derived from it. In addition, biomass with low water content is desirable to reduce drying costs and improve oil quality;
- Modifications of engine, turbine and boiler combustion systems are required for proper utilization of pyrolysis bio-oil considering the effect of physical and chemical properties, gaseous and particle emissions, combustion efficiency, slag and carbon deposition and component material corrosion;
- Numerous catalysts have been developed and tested to in pyrolysis process. Most of those were used only at small scale to improve gas production for research purposes having their own limitations. However sustainable industrial scale catalysts for biomass pyrolysis are yet to be identified;
- Although a lot of studies have been conducted on pyrolysis economy but most of those were limited in small or pilot scale production. Detail economical assessment of industrial scale pyrolysis plant may be required to establish this technology as a competitor with conventional energy and other alternative energy sources;
- Further study is recommended to address the issues mentioned above as well as the challenges outlined in Section 12.
References
- Howarth, N.A.A.; Foxall, A. The Veil of Kyoto and the politics of greenhouse gas mitigation in Australia. Political Geogr. 2010, 29, 167–176. [Google Scholar] [CrossRef]
- Ramage, J.; Scurlock, J. Biomass. In Renewable Energy-Power for a Sustainable Future; Boyle, G., Ed.; Oxford University Press: Oxford, UK, 1996. [Google Scholar]
- Zhang, O.; Chang, J.; Wang, T.; Xu, Y. Review of biomass pyrolysis oil properties and upgrading research. Energy Convers. Manag. 2007, 48, 87–92. [Google Scholar] [CrossRef]
- Tsai, W.T.; Lee, M.K.; Chang, Y.M. Fast pyrolysis of rice husk: Product yields and compositions. Bioresour. Technol. 2007, 98, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Bridgwater, A.V. Biomass fast pyrolysis. Therm. Sci. 2004, 8, 21–49. [Google Scholar] [CrossRef]
- Downie, A. BEST Pyrolysis Technology: A Solution for the Greenhouse Challenge. BEST Energies, Australia. ThermalNet Newsl. 2007, 5, 5. [Google Scholar]
- Demirbas, A. Pyrolysis of ground beech wood in irregular heating rate conditions. J. Appl. Pyroyisis 2005, 73, 39–43. [Google Scholar] [CrossRef]
- Asadullah, M.; Rahman, M.A.; Ali, M.M.; Motin, M.A.; Sultan, M.B.; Alam, M.R. Production of bio-oil from fixed bed pyrolysis of bagasse. Fuel 2007, 86, 2514–2520. [Google Scholar] [CrossRef]
- Demiral, I.; Sensoz, S. The effects of different catalysts on the pyrolysis of industrial wastes (olive and hazelnut bagasse). Bioresour. Technol. 2008, 99, 8002–8007. [Google Scholar]
- Mohan, D.; Pittman, C.U.; Bricka, M.; Smith, F.; Yancey, B.; Mohammad, J. Sorption of arsenic, cadmium, and lead by chars produced from fast pyrolysis of wood and bark during bio-oil production. J. Colloid Interf. Sci. 2007, 310, 57–73. [Google Scholar] [CrossRef]
- Aho, A.; Kumar, N.; Eranen, K.; Salmi, T.; Hupa, M.; Murzin, D.Y. Catalytic pyrolysis of woody biomass in a fluidized bed reactor: influence of the zeolite structure. Fuel 2008, 87, 2493–2501. [Google Scholar] [CrossRef]
- Karaosmanoglu, F.; Tetik, E. Fuel properties of pyrolytic oil of the straw and stalk of rape plant. Renew. Energy 1999, 16, 1090–1093. [Google Scholar] [CrossRef]
- Jensen, P.A.; Sander, B.; Dam-Johansen, K. Pretreatment of straw for power production by pyrolysis and char wash. Biomass Bioenergy 2001, 20, 431–446. [Google Scholar] [CrossRef]
- Putun, E.; Uzun, B.B.; Putun, A.E. Fixed-bed catalytic pyrolysis of cotton-seed cake: Effects of pyrolysis temperature, natural zeolite content and sweeping gas flow rate. Bioresour. Technol. 2006, 97, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Antal, M.J., Jr.; Grönli, M. The art, science, and technology of charcoal production. Ind. Eng. Chem. Res. 2003, 42, 1619–1640. [Google Scholar] [CrossRef]
- Mohan, D.; Pittman, C.U.; Steele, P.H. Pyrolysis of wood/biomass for bio-oil: A critical review. Energy Fuels 2006, 20, 848–889. [Google Scholar]
- Demirbas, A. Partly chemical analysis of liquid fraction of flash pyrolysis products from biomass in the presence of sodium carbonate. Energy Convers. Manag. 2002, 43, 1801–1809. [Google Scholar] [CrossRef]
- Demirbas, A. The influence of temperature on the yields of compounds existing in bio-oils obtained from biomass samples via pyrolysis. Fuel Proc. Technol. 2007, 88, 591–597. [Google Scholar] [CrossRef]
- Demirbas, A. Combustion characteristics of different biomass fuels. Prog. Energy Combus. Sci. 2004, 30, 219–230. [Google Scholar] [CrossRef]
- Muradov, N.Z.; Veziroglu, T.N. “Green” path from fossil-based to hydrogen economy: An overview of carbon-neutral technologies. Int. J. Hydrog. Energy 2008, 33, 6804–6839. [Google Scholar] [CrossRef]
- Frassoldati, A.; Migliavacca, G.; Crippa, T.; Velata, F.; Faravelli, T.; Ranzi, E. Detailed Kinetic Modeling of Thermal Degradation of Biomasses. Proceeding of the 29th Meeting on Combustion, Napoli, Italia, September 2006; Available online: ci.irc.na.cnr.it/download/proc%2006/documenti/Papers/09-02-frassoldati-039.pdf (accessed on 20 November 2012).
- Pei-dong, Z.; Guomei, J.; Gang, W. Contribution to emission reduction of CO2 and SO2 by household biogas construction in rural China. Renew. Sustain. Energy Rev. 2007, 11, 1903–1912. [Google Scholar] [CrossRef]
- Thornley, P.; Upham, P.; Huang, Y.; Rezvani, S.; Brammer, J.; Rogers, J. Integrated assessment of bioelectricity technology options. Energy Policy 2009, 37, 890–903. [Google Scholar] [CrossRef]
- Somerville, C. Energy from Biomass; Workshop Presentration for the Inter Academy Council Study Report. In Lighting the Way: Towards Sustainable Energy Future; IAC: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Grønli, M.G.; Varhegyi, G.; Blassi, C.D. Thermogravimetric analysis and devolatilization kinetics of wood. Ind. Eng. Chem. Res. 2002, 41, 4201–4208. [Google Scholar] [CrossRef]
- Fisher, T.; Hajaligol, M.; Waymack, B.; Kellogg, D. Pyrolysis behaviour and kinetics of biomass derived materials. J. Appl. Pyrolysis 2002, 62, 331–349. [Google Scholar] [CrossRef]
- Lanzetta, M.; Blasi, D.C. Pyrolysis kinetics of wheat and corn straw. J. Anal. Appl. Pyrolysis. 1998, 44, 181–192. [Google Scholar] [CrossRef]
- Sinha, S.; Jhalani, A.; Ravi, M.R.; Ray, A. J. Solar Energy Soc. Ind. 2000, 10, 41–62.
- Venderbosch, R.H.; Prins, W. Review: Fast pyrolysis technology development. Biofuel 2010, 4, 178–208. [Google Scholar]
- International Energy Agency. Annual Report,2006: IEA Bioenergy: Task 34,Pyrolysis of Biomass; International Energy Agency: Paris, France, 2006. [Google Scholar]
- Balat, M.; Balat, M.; Kirtay, E.; Balat, H. Main routes for the thermo-conversion of biomass into fuels and chemicals. Part 1: Pyrolysis systems. Energy Conv. Manag. 2009, 50, 3147–3157. [Google Scholar] [CrossRef]
- Bridgwater, T. Pyrolysis of Biomass. IEA Bioenergy: Task 34; Bioenergy Research Group, Aston University: Birmingham, UK, 2007. [Google Scholar]
- Bridgwater, A.V.; Czernik, S.; Piskorz, J. An overview of fast pyrolysis. Prog. Thermochem. Biomass Convers. 2001, 2, 977–997. [Google Scholar]
- Demirbas, A.H. Yields and heating values of liquids and chars from spruce trunkbark pyrolysis. Energy Source Part A 2005, 27, 1367–1373. [Google Scholar] [CrossRef]
- Tippayawong, N.; Kinorn, J.; Thavornun, S. Yields and gaseous composition from slow pyrolysis of refuse-derived fuels. Energy Source Part A 2008, 30, 1572–1578. [Google Scholar] [CrossRef]
- Demibas, A.; Arin, G. An overview of biomass pyrolysis. Energy Source Part A 2002, 24, 471–482. [Google Scholar] [CrossRef]
- Brammer, J.G.; Lauer, M.; Bridgwater, A.V. Opportunities for biomass-derived ‘‘bio-oil” in European heat and power markets. Energy Policy 2006, 34, 2871–2880. [Google Scholar] [CrossRef]
- Chiaramonti, D.; Oasmaa, A.; Solantausta, Y. Power generation using fast pyrolysis liquids from biomass. Renew. Sustain. Energy Rev. 2007, 11, 1056–1086. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Fast Pyrolysis of Biomass: A Handbook; CRL Press: Newbury, UK, 2005. [Google Scholar]
- Demirbas, A. Recent advances in biomass conversion technologies. Energy Educ. Sci. Technol. 2000, 6, 77–83. [Google Scholar]
- Aguado, R.; Olazar, M.; Gaisan, B.; Prieto, R.; Bilbao, J. Kinetic study of polyolefin pyrolysis in a conical spouted bed reactor. Ind. Eng. Chem. Res. 2002, 41, 4559–4566. [Google Scholar] [CrossRef]
- Cornelissen, T.; Yperman, Y.; Reggers, G.; Schreurs, S.; Carleer, R. Flash co-pyrolysis of biomass with polylactic acid. Part 1: Influence on bio-oil yield and heating value. Fuel 2008, 87, 1031–1041. [Google Scholar] [CrossRef]
- Vamvuka, D.; Karakas, E.; Kastanaki, E.; Grammelis, P. Pyrolysis characteristics and kinetics of biomass residuals mixtures with lignite. Fuel 2003, 82, 1949–1960. [Google Scholar] [CrossRef]
- Goyal, H.B.; Seal, D.; Saxena, R.C. Bio-fuels from thermochemical conversion of renewable resources: A review. Renew. Sustain. Energy Rev. 2008, 12, 504–517. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Twidell, J. Biomass energy. Renew. Energy World 1998, 3, 38–39. [Google Scholar]
- Fegbemi, L.; Khezami, L.; Capart, R. Pyrolysis products from different biomasses: Application to the thermal cracking of tar. Appl. Energy 2001, 69, 293–306. [Google Scholar] [CrossRef]
- Yaman, S. Pyrolysis of biomass to produce fuels and chemical feedstocks. Energy Convers. Manag. 2004, 45, 651–671. [Google Scholar] [CrossRef]
- Gupta, A.K.; Lilley, D.G. Thermal destruction of wastes and plastics. In Plastics and the Environment; Andrady, A.L., Ed.; John Wiley & Sons Ltd.: Chichester, UK, 2003; pp. 629–696. [Google Scholar]
- Wang, G.; Li, W.; Li, B.; Chen, H. TG study on pyrolysis of biomass and its three components under syngas. Fuel 2008, 87, 552–558. [Google Scholar] [CrossRef]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Liang, D.T.; Zheng, C. Pyrolysis of palm oil wastes for enhanced production of hydrogen rich gases. Fuel Proc. Technol. 2006, 87, 935–942. [Google Scholar] [CrossRef]
- Abbasi, T.; Abbasi, S.A. Biomass energy and the environmental impacts associated with its production and utilization. Renew. Sustain. Energy Rev. 2010, 14, 919–937. [Google Scholar] [CrossRef]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Wang, J.; Wang, G.; Zhang, M.; Chen, M.; Li, D.; Min, F.; Chen, M.; Zhang, S.; Ren, Z.; Yen, Y. A comparative study of thermolysis characteristic and kinetics of seaweeds and fir-wood. Proc. Biochem. 2006, 41, 1883–1886. [Google Scholar] [CrossRef]
- Fahmi, R.; Bridgwater, A.V.; Donnison, I.; Yates, N.; Jones, J.M. The effect of lignin and inorganic species in biomass on pyrolysis oil yields, quality and stability. Fuel 2008, 87, 1230–1240. [Google Scholar] [CrossRef]
- Demirbas, A. Current technologies for the thermo-conversion of biomass into fuels and chemicals. Energy Source Part A 2004, 26, 715–730. [Google Scholar] [CrossRef]
- Demirbas, A. Calculation of higher heating values of biomass fuels. Fuel 1997, 76, 431–434. [Google Scholar] [CrossRef]
- Yang, W.; Ponzio, A.; Lucas, C.; Blasiak, W. Performance analysis of a fixed–bed biomass gasifier using high-temperature air. Fuel Proc. Technol. 2006, 87, 235–245. [Google Scholar] [CrossRef]
- Gaunt, J.L.; Lehmann, J. Energy balance and emissions associated with biochar sequestration and pyrolysis bioenergy production. Environ. Sci. Technol. 2008, 42, 4152–4158. [Google Scholar] [CrossRef] [PubMed]
- Das, L.M.; Gulati, R.; Gupta, P.K. A comparative evaluation of the performance characteristics of a spark ignition engine using hydrogen and compressed natural gas as alternative fuels. Int. J. Hydrog. Energy 2000, 25, 783–793. [Google Scholar] [CrossRef]
- Blanco Lopez, M.C.; Blanco, C.G.; Martinez-Alonso, A.; Toscon, J.M.D. Composition of gases released during olive stones pyrolysis. J. Anal. Appl. Pyrolysis 2002, 65, 313–322. [Google Scholar] [CrossRef]
- Minkova, V.; Razvigorova, M.; Bjornbom, E.; Zanzi, R.; Budinova, T.; Petrov, N. Effect of water vapour and biomass nature on the yield and quality of the pyrolysis products from biomass. Fuel Proc. Technol. 2001, 70, 53–61. [Google Scholar] [CrossRef]
- Arni, S.A.; Bosio, B.; Arato, E. Syngas from sugarcane pyrolysis: An experimental study for fuel cell applications. Renew. Energy 2010, 35, 29–35. [Google Scholar] [CrossRef]
- Friedl, A.; Padouvas, E.; Rotter, H.; Varmuza, K. Prediction of heating values ofbiomass fuel from elemental composition. Anal. Chim. Acta 2005, 544, 191–198. [Google Scholar] [CrossRef]
- Ringer, M.; Putsche, V.; Scahill, J. Large-Scale Pyrolysis Oil Production and Economic Analysis; Technical Report NREL/TP-510–37779; National Renewable Energy Laboratory: Cole Boulevard, CO, USA, 2006. [Google Scholar]
- Wang, X. Biomass Fast Pyrolysis in a Fluidized Bed. Ph.D. Thesis, University of Twente, Enscheda, The Netherlands, 2006. [Google Scholar]
- Wang, X.; Kersten, S.R.A.; Prins, W.; Van Swaaij, W.P.M. Biomass pyrolysis in a fluidized bed reactor. Part 2: Experimental Validation of Model Results. Ind. Eng. Chem. Res. 2005, 44, 8786–8795. [Google Scholar] [CrossRef]
- Chopra, S.; Jain, A. A Review of fixed bed gasification systems for biomass. Agric. Eng. Int. 2007, 5, 1–23. [Google Scholar]
- Altafini, C.R.; Wander, P.R.; Barreto, R.M. Prediction of the working parameters of a wood waste gasifier through an equilibrium model. Energy Convers. Manag. 2003, 44, 2763–2777. [Google Scholar] [CrossRef]
- Leung, D.Y.C.; Yin, X.L.; Wu, C.Z. A review on the development and commercialization of biomass gasification technologies in China. Renew. Sustain. Energy Rev. 2004, 8, 565–580. [Google Scholar] [CrossRef]
- Barker, S.N. Gasification and pyrolysis-routes to competitive electricity production in the UK. Energy Convers. Manag. 1996, 37, 861–866. [Google Scholar] [CrossRef]
- Filippis, P.D.; Borgianni, C.; Paolucci, M.; Pochetti, F. Gasification process of Cuban bagasse in a two-stage reactor. Biomass Bioenergy 2004, 27, 247–252. [Google Scholar] [CrossRef]
- Rao, M.S.; Singha, S.P.; Sodhaa, M.S.; Dubey, A.K.; Shyam, M. Stoichiometric, mass, energy and exergy balance analysis of countercurrent fixed-bed gasification of post-consumer residues. Biomass Bioenergy 2004, 27, 155–171. [Google Scholar] [CrossRef]
- Pathak, B.S. Biomass to power rural development. In Proceedings of National Seminar on Biomass Based Decentralized Power Generation; Pathak, B.S., Ed.; Sardar Patel Renewable Energy Research Institute: Vallabh Vidyanagar, India; pp. 1–6.
- Lv, P.M.; Xiong, Z.H.; Chang, J.; Wu, C.Z.; Chen, Y.; Zhu, J.X. An experimental study on biomass air–steam gasification in a fluidized bed. Bioresour. Technol. 2004, 95, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Sadaka, S.; Boateng, A.A. Pyrolysis and Bio-Oil, Agriculture and Natural Resources. FSA1052. Available online: http://www.uaex.edu/Other_Areas/publications/PDF/FSA-1052.pdf (accessed on 5 August 2010).
- Li, X.T.; Grace, R.; Lim, C.J.; Watkinson, A.P.; Chen, H.P.; Kim, J.R. Biomass gasification in a circulating fluidized bed. Biomass Bioenergy 2004, 26, 171–193. [Google Scholar] [CrossRef]
- Jones, S.B.; Holladay, J.E.; Valkenburg, C.; Stevens, D.J.; Walton, C.W.; Kinchin, C.; Elliott, D.C.; Czernik, S. Production of Gasoline and Diesel from Biomass via Fast Pyrolysis, Hydrotreating and Hydrocracking: A Design Case; Report No. PNNL-18284; U.S. Department of Energy: Springfield, VA, USA, 2009. [Google Scholar]
- FAO. The Research Progress of Biomass Pyrolysis Processes; National Resource Management Environment Department. Available online: www.fao.org/docrep (accessed on 14 June 2010).
- Roy, C.; Blanchette, D.; Korving, L.; Yang, J.; DeCaumia, B. Development of a Novel Vacuum Pyrolysis Reactor with Improved Heat Transfer Potential. In Developments in Thermochemical. Biomass Conversion; Bridgewater, A.V., Boocock, D.G.B., Eds.; Blackie Academic and Professional: London, UK, 1997; pp. 351–367. [Google Scholar]
- Wagenaar, B.M.; Venderbosch, R.H.; Carrasco, J.; Strenziok, R.; Van der Aa, B.J. Rotating Cone Bio-oil Production and Applications. In Progress in Thermochemical. Biomass Conversion; Bridgewater, A.V., Ed.; Blackwell Science: Oxford, UK, 2001; pp. 1268–1280. [Google Scholar]
- Bramer, E.A.; Holthuis, M.R. Clean Liquid Fuel through Flash Pyrolysis. In The Development of the PyRos Process; AFTUR Final Report; University of Twente: Enschede, The Netherlands, 2005. [Google Scholar]
- Tang, L.; Huang, H. Plasma pyrolysis of biomass for production of syngas and carbon adsorbent. Energy Fuels 2005, 19, 1174–1178. [Google Scholar] [CrossRef]
- Chen, G.; Andries, K.; Luo, Z.; Spliethoff, H. Biomass pyrolysis/Gasification for product gas production. The overall investigation of parametric effects. Energy Convers. Manag. 2003, 44, 1873–1884. [Google Scholar]
- Fernández, Y.; Menéndez, J.A. Influence of feed characteristics on the microwave-assisted pyrolysis used to produce syngas from biomass wastes. J. Anal. Appl. Pyrolysis 2011, 91, 316–322. [Google Scholar] [CrossRef]
- Lam, S.S.; Russell, A.D.; Chase, H.A. Microwave pyrolysis, a novel process for recycling waste automotive engine oil. Energy 2010, 35, 2985–2991. [Google Scholar] [CrossRef]
- Domínguez, A.; Fernández, Y.; Fidalgo, B.; Pis, J.J.; Menéndez, J.A. Bio-syngasproduction with low concentration of CO2 and CH4 from microwave-induced pyrolysis of wet and dried sewage sludge. Chemosphere 2008, 70, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, A.; Menéndez, J.A.; Fernández, Y.; Pis, J.J.; Valente Nabais, J.M.; Carrott, P.J.M.; Ribeiro Carrott, M.M.L. Conventional and microwave induced pyrolysis of coffee hulls for the production of a hydrogen rich fuel gas. J. Anal. Appl. Pyrolysis 2007, 79, 128–135. [Google Scholar] [CrossRef]
- Fernández, Y.; Arenillas, M.A.; Díez, P.J.J.; Menéndez, J.A. Pyrolysis of glycerol over activated carbons for syngas production. J. Anal. Appl. Pyrolysis 2009, 84, 145–150. [Google Scholar] [CrossRef]
- Huang, Y.F.; Kuan, W.H.; Lo, S.L.; Lin, C.F. Total recovery of resources and energy from rice straw using microwave-induced pyrolysis. Bioresour. Technol. 2008, 99, 8252–8258. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Song, Z.; Liu, H.; Li, Z.; Li, L.; Ma, C. Microwave pyrolysis of corn stalk bale: A promising method for direct utilization of large-sized biomass and syngas production. J. Anal. Appl. Pyrolysis 2010, 89, 87–94. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, D.; Sun, C. Effect of heavy metals on the sorption of hydrophobic organic compounds to wood charcoal. Environ. Sci. Technol. 2007, 41, 2536–2541. [Google Scholar] [CrossRef] [PubMed]
- Boutin, O.; Ferrer, M.; Lede, J. Flash pyrolysis of cellulose pellets submitted to a concentrated radiation: experiments and modeling. Chem. Eng. Sci. 2002, 57, 15–25. [Google Scholar] [CrossRef]
- Hofmann, L.; Antal, M.J. Numerical simulations of the performance of solar fired flash pyrolysis reactors. Sol. Energy 1984, 33, 427–440. [Google Scholar] [CrossRef]
- Shakya, B.D. Pyrolysis of Waste Plastics to Generate Useful Fuel Containing Hydrogen Using a Solar Thermo-Chemical Process. Ph.D. Thesis, School of Chemical and Biomolecular Engineering, University of Sydney, Sydney, Australia, 2007. [Google Scholar]
- Solantausta, Y.; Oasmaa, A. Fast Pyrolysis of Forestry Residues and Sawdust, Production and Fuel Oil Quality. In Proceedings of International Nordic Bioenergy Conference, Javaskyla, Frinland, September 2003; pp. 1–3.
- Oasmaa, A.; Kuoppala, E. Fast pyrolysis of forestry residue. 3. Storage stability of liquid fuel. Energy Fuel 2003, 17, 1075–1084. [Google Scholar] [CrossRef]
- Czernik, S.; Bridgwater, A.V. Applications of biomass fast pyrolysis oil. Energy Fuel 2004, 18, 590–598. [Google Scholar] [CrossRef]
- Siemons, R.V. Bio-oil production from biomass: The pyrolysis process. Available online: http://www.cleanfuels.nl/Projects%20&%20publications/CFUL%20pyrolysis.pdf (accessed on 20 November 2012).
- Dobele, G.; Urbanovich, I.; Volpert, A.; Kampars, V.; Samulis, E. Fast pyrolysis—Effect of wood drying on the yield and properties of bio-oil. BioResources 2007, 2, 699–706. [Google Scholar]
- Sharma, R.K.; Wooten, J.B.; Baliga, V.L.; Lin, X.; Chan, W.G.; Hajaligol, M.R. Characterization of chars from pyrolysis of lignin. Fuel 2004, 83, 1469–1482. [Google Scholar] [CrossRef]
- Wang, X.; Cents, A.H.G.; Kersten, S.R.A.; Prins, W.; Van Swaaij, W.P.M. A Filter-Assisted Fluid Bed Reactor for Integrated Production and Clean-Up of Bio-Oil. In Proceedings of the 2nd World Conference on Bioenergy, Rome, Italy, 10–14 May 2004.
- Scahill, J.; Diebold, J.P.; Feek, C. Removal of Residual Char Fines from Pyrolysis Vapors by Hot Gas Filtration. In Developments in Thermochemical Biomass Conversion; Blackie Academic & Professional: London, UK, 1997; pp. 253–266. [Google Scholar]
- Bramer, E.A.; Holthis, M.R.; Brem, G. Development of a Cyclonic Reactor with Internal Particle Filter for the Flash Pyrolysis of Biomass; the PyRos Reactor. In Proceedings of the 2nd World Conference on Bioenergy, Rome, Italy, 10–14 May 2004.
- Johnson, W.L.; Yavari, G.H.; Radelin, D.; St. A, G. Apparatus for Separating Fouling Contaminants from Non-Condensable Gases at the End of Pyrolysis/Thermolysis of Biomass Process. US Patent 7,004,999 B2, 2006. [Google Scholar]
- Demirbas, A. Production of gasoline and diesel fuels from bio-materials. Energy Source Part A 2007, 29, 753–760. [Google Scholar] [CrossRef]
- Demirbas, A. Mechanisms of liquefaction and pyrolysis reactions of biomass. Energy Convers. Manag. 2000, 41, 633–646. [Google Scholar] [CrossRef]
- Bridgwater, A.V.; Czernik, S.; Diebold, J. Fast Pyrolysis of Biomass: A Handbook; CRL Press: London, UK, 1999. [Google Scholar]
- Diebold, J. A Review of the Chemical and Physical Mechanisms of the Storage and Stability of Fast Pyrolysis Bio-Oils; NREL 1; Thermalchemie, Inc.: Lakewood, CO, USA, 2000. [Google Scholar]
- Evans, R.; Milne, T. Molecular characterisation of the pyrolysis of biomass 1. Energy Fuel 1987, 1, 123–137. [Google Scholar] [CrossRef]
- Oasmaa, A.; Meier, D. Norms and standards for fast pyrolysis liquids: 1. Round robin test. J. Anal. Appl. Pyrolysis 2005, 73, 323–334. [Google Scholar] [CrossRef]
- Shihadeh, A.; Hochgreb, S. Diesel engine combustion of biomass pyrolysis oils. Energy Fuels 2000, 14, 260–274. [Google Scholar] [CrossRef]
- Abdullah, N. An Assessment of Pyrolysis for Processing Empty Fruit Bunches. Ph.D. Thesis, Aston University, Birmingham, UK, 2005. [Google Scholar]
- Fahmi, R.; Bridgwater, A.; Thain, V.S.; Donnison, I. Prediction of Klason lignin and lignin thermal degradation products by Py-GC/MS in a collection of Lolium. and Festuca. grasses. J. Anal. Appl. Pyrolysis 2007, 80, 16–23. [Google Scholar] [CrossRef]
- Sensoz, S.; Angin, D. Pyrolysis of safflower (Charthamus tinctorius L.) seed press cake in a fixed-bed reactor: Part 2. Structural characterization of pyrolysis bio-oils. Bio-Resour. Technol. 2008, 99, 5498–5504. [Google Scholar] [CrossRef]
- Mullaney, H.; Farag, I.H.; LaClaire, C.L.; Barrett, C.J. Technical, Environmental and Economic Feasibility of Bio-Oil in New Hampshire’s North Country; Final Report; New Hampshire Industrial Research Center (NHIRC): Durham City, NH, USA, 2002. [Google Scholar]
- Freel, B.A.; Graham, R.G.; Huffman, D.R. Commercial Aspects of Rapid Thermal Processing (RTMTM). In Bio-Oil Production and Utilization; CPL Press: Newbery, UK, 1996; pp. 86–95. [Google Scholar]
- Gust, S. Combustion Experiences of Flash Pyrolysis Fuel in Intermediate Size Boilers. In Developments in Thermochemical Biomass Conversion; Bridgwater, A.V., Boocock, D.G., Eds.; Blackie Academic & Professional: London, UK, 1997; pp. 481–488. [Google Scholar]
- Oasmaa, A.; Kytö, M.; Sipilä, K. Pyrolysis Oil Combustion Tests in an Industrial Boiler. In Progress in Thermochemical Biomass Conversion; Blackwell Science: Oxford, UK, 2001; pp. 1468–1481. [Google Scholar]
- Agarwal, A.K. Biofuels (alcohols and biodiesel) applications as fuels for internal combustion engines. Prog. Energy Combust. Sci. 2007, 33, 233–271. [Google Scholar] [CrossRef]
- Labeckas, G.; Slavinskas, S. Performance of direct-injection off-road diesel engine on rapeseed oil. Renew. Energy 2006, 31, 849–863. [Google Scholar] [CrossRef]
- Ramadhas, A.S.; Jayaraj, S.; Muraleedharan, C. Characterization and effect of using rubber seed oil as fuel in the compression ignition engines. Renew. Energy 2005, 30, 795–803. [Google Scholar] [CrossRef]
- Jayed, M.H.; Masjuki, H.H.; Saidur, R.; Kalam, M.A.; Jahirul, M.I. Environmental Aspects and Challenges of Oilseed Produced Biodiesel in Southeast Asia. Renew. Sustain. Energy Rev. 2009, 13, 2452–2462. [Google Scholar] [CrossRef]
- Solantausta, Y.; Beckman, D.; Bridgwater, A.V.; Diebold, J.P.; Ellioit, D.C. Assessment of Liquefaction and Pyrolysis Systems. Biomass Bioenergy 1994, 2, 279–297. [Google Scholar] [CrossRef]
- Chiaramonti, D.; Bonini, M.; Fratini, E.; Tondi, G.; Gartner, K.; Bridgwater, A.V.; Grimm, H.P.; Soldaini, I.; Webster, A.; Baglioni, P. Development of Emulsions from Biomass Pyrolysis Liquid and Diesel and Their Use in Engines. Biomass Bioenergy 2003, 25, 101–111. [Google Scholar] [CrossRef]
- Baglioni, P.; Chiaramonti, D.; Bonini, M.; Soldaini, I.; Tondi, G. Bio-Crude-Oil/Diesel Oil Emulsification: Main Achievements of the Emulsification Process and Preliminary Results of Tests on Diesel Engine. In Progress in Thermochemical Biomass Conversion; Blackwell Science: Oxford, UK, 2001; pp. 1525–1539. [Google Scholar]
- Ormrod, D.; Webster, A. Progress in Utilization of Bio-Oil in Diesel Engines. In PyNe News Letter; Aston University: Birmingham, UK, 2000; p. 15. [Google Scholar]
- Strenziok, R.; Hansen, U.; Künster, H. Combustion of Bio-Oil in a Gas Turbine. In Progress in Thermochemical Biomass Conversion; Blackwell Science: Oxford, UK, 2001; pp. 1452–1458. [Google Scholar]
- Hague, R.A. The Pre-Treatment and Pyrolysing of Biomass for the Production of Liquids for Fuel and Speciality Chemicals. Ph.D Thesis, Aston University, Birmingham, UK, 1998. [Google Scholar]
- Rocha, J.D.; Olivares-Gómez, E.; Mesa-Pérez, J.M.; Cortez, L.A.B.; Seye, O.; Brossard-González, L.E. The demonstration fast pyrolysis plant to biomassconversion in Brazil. In Proceedings of World Renewable Energy Congress VII (WRC 2002); 2002; Cologne, Germany. [Google Scholar]
- González, J.F.; Román, S.; Encinar, J.M.; Martinéz, G. Pyrolysis of various biomass residues and char utilization for the production of activated carbons. J. Anal. Appl. Pyrolysis 2009, 85, 134–141. [Google Scholar] [CrossRef]
- Downie, A.; Crosky, A.; Munroe, P. Physical Properties of Biochar. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009. [Google Scholar]
- Brown, R. Biochar Production Technology. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009. [Google Scholar]
- Cetin, E.; Moghtaderi, B.; Gupta, R.; Wall, T.F. Influence of pyrolysis conditions on the structure and gasification reactivity of biomass chars. Fuel 2004, 83, 2139–2150. [Google Scholar] [CrossRef]
- Lua, A.C.; Yang, T.; Guo, J. Effects of pyrolysis conditions on the properties of activated carbons prepared from pistachio-nut shells. J. Anal. Appl. Pyrolysis 2004, 72, 279–287. [Google Scholar] [CrossRef]
- Dawei, A.; Zhimin, W.; Shuting, Z.; Hongxing, Y. Low-temperature pyrolysis of municipal solid waste: Influence of pyrolysis temperature on the characteristics of solid fuel. Int. J. Energy Res. 2006, 30, 349–357. [Google Scholar] [CrossRef]
- Sohi, S.; Lopez-Capel, E.; Krull, E.; Bol, R. Biochar, Climate Change and Soil: A Review to Guide Future Research; CSIRO Land and Water Science Report; CSIRO: Canberra, Australia, 2009. [Google Scholar]
- Verheijen, F.; Jeffery, S.; Bastos, A.C.; van der Velde, M.; Diafas, I. Biochar Application to Soils: A Critical Scientific Review of Effects on Soil Properties, Processes and Functions; European Commission Report No. EUR 24099 EN; European Communities: Ispara, Italy, 2010. [Google Scholar]
- Bourke, J.; Manley-Harris, M.; Fushimi, C.; Dowaki, K.; Nunoura, T.; Antal, M.J., Jr. Do all carbonised charcoals have the same structure? A model of the chemical structure of carbonized charcoal. Ind. Eng. Chem. Res. 2007, 46, 5954–5967. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Kimber, S.; Morris, S.; Chan, K.Y.; Downie, A.; Rust, J.; Joseph, S.; Cowie, A. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant. Soil 2009, 1–12. [Google Scholar]
- Amonette, J.E.; Jospeh, S. Characteristics of Biochar: Microchemical Properties. In Biochar for Environmental Management Science and Technology; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009. [Google Scholar]
- Gaskin, J.W.; Steiner, C.; Harris, K.; Das, K.C.; Bibens, B. Effect of low-temperature pyrolysis conditions on biochar for agricultural use. Trans. ASABE 2008, 51, 2061–2069. [Google Scholar] [CrossRef]
- Kantarelis, E.; Zabaniotou, A. Valorization of cotton stalks by fast pyrolysis and fixed bed air gasification for syngas production as precursor of second generation biofuels and sustainable agriculture. Bioresour. Technol. 2009, 100, 942–947. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Xiao, B.; Liu, S.; Hu, Z.; Guo, X.; Luo, S. Syngas production from pyrolysis of municipal solid waste (MSW) with dolomite as downstream catalysts. J. Anal. Appl. Pyrolysis 2010, 87, 181–187. [Google Scholar] [CrossRef]
- Wei, L.; Thomasson, J.A.; Bricka, R.M.; Batchelor, W.D.; Columbus, E.P.; Wooten, J.R. Experimental Study of a Downdraft Gratifier; ASABE Meeting Paper No. 066029; American Society of Agricultural and Biological Engineers: St. Joseph, Michigan, 2006. [Google Scholar]
- Ahmed, I.; Gupta, A.K. Syngas yield during pyrolysis and steam gasification of paper. Appl. Energy 2009, 86, 1813–1821. [Google Scholar] [CrossRef]
- Dasappa, S.; Paul, P.J.; Mukunda, H.S.; Rajan, G.N.K.; Sridhar, S.; Sridhar, H.V. Biomass gasification technology—A route to meet energy needs. Curr. Sci. 2004, 87, 908–916. [Google Scholar]
- Couher, C.; Commandre, J.M.; Salvador, S. Failure of the component additivity rule to predict gas yields of biomass in flash pyrolysis at 950 °C. Biomass Bioenergy 2009, 33, 316–326. [Google Scholar] [CrossRef]
- Couher, C.; Commandre, J.M.; Salvador, S. Is it possible to predict gas yields of any biomass after rapid pyrolysis at high temperature from its composition in cellulose, hemicelluloses and lignin? Fuel 2009, 88, 408–417. [Google Scholar] [CrossRef]
- Jahirul, M.I.; Masjuki, H.H.; Saidur, R.; Kalam, M.A.; Jayed, M.H.; Wazed, M.A. Comparative Engine performance and Emission Analysis of CNG and Gasoline in a Retrofitted Car Engine. Appl. Ther. Eng. 2010, 30, 2219–2226. [Google Scholar] [CrossRef]
- Kahraman, E.; Ozcanli, S.C.; Ozerdem, B. An experimental study on performance and emission characteristics of a hydrogen fuelled spark ignition engine. Int. J. Hydrog. Energy 2007, 32, 2066–2072. [Google Scholar] [CrossRef]
- Saidur, R.; Jahirul, M.I.; Moutushi, T.Z.; Imtiaz, H.; Masjuki, H.H. Effect of partial substitution of diesel by natural gas on performance parameters of a four cylinder diesel engine. Proceedings of the Institution of Mechanical Engineers, UK. J. Power Energy Part A 2007, 221, 1–10. [Google Scholar] [CrossRef]
- Shah, A.; Srinivasan, R.; To, S.D.F.; Columbus, E.P. Performance and emissions of a spark-ignited engine driven generator on biomass based syngas. Bioresour. Technol. 2010, 101, 4656–4661. [Google Scholar] [CrossRef] [PubMed]
- Mustafi, N.N.; Miragliab, Y.C.; Rainea, R.R.; Bansala, P.K.; Elder, S.T. Spark-ignition engine performance with “Powergas” fuel (mixture of CO/H2): A comparison with gasoline and natural gas. Fuel 2006, 85, 1605–1612. [Google Scholar] [CrossRef]
- Papagiannakis, R.G.; Rakopoulos, C.D.; Hountalas, D.T.; Giakoumis, E.G. Study of the performance and exhaust emissions of a spark-ignited engine operating on syngas fuel. Int. J. Alter. Propuls. 2007, 2, 190–215. [Google Scholar] [CrossRef]
- McMillian, M.H.; Lawson, S.A. Experimental and modeling study of hydrogen/syngas production and particulate emissions from a natural gas-fueled partial oxidation engine. Int. Hydrog. Energy 2006, 31, 847–860. [Google Scholar] [CrossRef]
- Shudo, T.; Nagano, T.; Kobayashi, M. Combustion characteristics of waste pyrolysis gases in an internal combustion engine. Int. J. Automot. Technol. 2003, 4, 1–8. [Google Scholar]
- Sobyanin, V.; Sadykov, V.; Kirillov, V.; Kuzmin, V.; Kuzin, N.; Vostrikov, Z.; Khristolyubov, A.; Luksho, V.; Afanasiev, A. Syngas as a Fuel for IC and Diesel Engines: Efficiency and Harmful Emissions Cut-off. In Proceedings of International Hydrogen Energy Congress and Exhibition, Istanbul, Turkey, 13–15 July 2005.
- Ahmad, I.; Gupta, A.K. Pyrolysis and gasification of food waste: Syngas characteristics and char gasification kinetics. Appl. Energy 2010, 87, 101–108. [Google Scholar] [CrossRef]
- He, M.; Hu, Z.; Xiao, B.; Li, J.; Guo, X.; Luo, S.; Yang, F.; Feng, Y.; Yang, G.; Liu, S. Hydrogen-rich gas from catalytic steam gasification of municipal solid waste (MSW): Influence of catalyst and temperature on yield and product composition. Int. J. Hydrog. Energy 2009, 34, 195–203. [Google Scholar] [CrossRef]
- Hao, X.H.; Guo, L.J.; Mao, X.; Zhang, X.M.; Chen, X.J. Hydrogen production from glucose used as a model compound of biomass gasified in supercritical water. Int. J. Hydrog. Energy 2003, 28, 55–64. [Google Scholar] [CrossRef]
- Han, J.; Kim, H. The reduction and control technology of tar during biomass gasification/pyrolysis: An overview. Renew. Sustain. Energy Rev. 2008, 12, 397–416. [Google Scholar] [CrossRef]
- Hu, G.; Xu, S.P.; Li, S.G.; Xiao, C.G.; Liu, S.Q. Steam gasification of apricot stones with olivine and dolomite as downstream catalysts. Fuel Process. Technol. 2006, 87, 375–382. [Google Scholar]
- Xiao, B.; Wang, Y.Y.; Su, Q. The research of treating municipal solid waste by gasification. China Resour. Compr. Utilization 2006, 24, 18–20. [Google Scholar]
- Chaudhari, S.T.; Dalai, A.K.; Bakhshi, N.N. Production of hydrogen and/or syngas (H2 + CO) via steam gasification of biomass-derived chars. Energy Fuels 2003, 17, 1062–1067. [Google Scholar] [CrossRef]
- Gallucci, K.; Stendardo, S.; Foscolo, P.U. CO2 capture by means of dolomite in hydrogen production. Int. J. Hydrog. Energy 2008, 33, 3049–3055. [Google Scholar] [CrossRef]
- Mahishi, M.R.; Goswami, D.Y. An experimental study of hydrogen production by gasification of biomass in the presence of a CO2 sorbent. Int. J. Hydrog. Energy 2007, 32, 2803–2808. [Google Scholar] [CrossRef]
- Wei, L.; Xu, S.P.; Zhang, L.; Liu, C.H.; Zhu, H.; Liu, S.Q. Steam gasification of biomass for hydrogen-rich gas in a free-fall reactor. Int. J. Hydrog. Energy 2007, 32, 24–31. [Google Scholar] [CrossRef]
- Xiaodong, Z. The Mechanism of Tar Cracking by Catalyst and the Gasification of Biomass. Ph.D Thesis, Zhejiang University, Zejiang, China, 2003. [Google Scholar]
- Elbaba, I.F.; Wu, C.; Williams, P.T. Hydrogen production from the pyrolysis-gasification of waste tyres with a nickel/cerium catalyst. Int. J. Hydrog. Energy 2011, 36, 6628–6637. [Google Scholar] [CrossRef]
- Ates, F.M.; Isikdag, M.A. Influence of temperature and alumina catalyst on pyrolysis of corncob. Fuel 2009, 88, 1991–1997. [Google Scholar]
- Tomishinge, K.; Asadullah, M.; Kunimori, K. Syngas production by biomass gasification using Rh/CeO/SiO2 catalysts and fluidized bed reactor. Catal. Today 2004, 89, 389–403. [Google Scholar] [CrossRef]
- Iwaki, H.; Ye, S.; Katagiri, H.; Kitagawa, K. Wastepaper gasification with CO2 or steam using catalysts of molten carbonates. Appl. Catal. A: Gen. 2004, 270, 237–243. [Google Scholar] [CrossRef]
- Demirbas, A. Gaseous products from biomass by pyrolysis and gasification: Effects of catalyst on hydrogen yield. J. Energy Convers. Manag. 2002, 43, 897–909. [Google Scholar] [CrossRef]
- Wang, P.; Tanabe, E.; Ito, K.; Jiab, J.; Morioka, H.; Shishido, T.; Takehira, K. Filamentous carbon prepared by the catalytic pyrolysis of CH4 on Ni/SiO2. Appl. Catal. A: Gen. 2002, 231, 35–44. [Google Scholar] [CrossRef]
- Williams, P.T.; Alexander, J. Catalytic pyrolysis of tyres: Influence of catalyst temperature. Fuel 2002, 81, 2425–2434. [Google Scholar] [CrossRef]
- Watanabe, M.; Inomata, H.; Arai, K. Catalytic hydrogen generation from biomass (glucose and cellulose) with ZrO2 in supercritical water. Biomass Bioenergy 2002, 22, 405–410. [Google Scholar] [CrossRef]
- Stals, M.; Carleer, R.; Reggers, G.; Schreurs, S.; Yperman, J. Flash pyrolysis of heavy metal contaminated hardwoods from phytoremediation: Characterisation of biomass, pyrolysis oil and char/ash fraction. J. Anal. Appl. Pyrolysis 2010, 89, 22–29. [Google Scholar] [CrossRef]
- Thornley, P.; Wright, E. Evaluation of Bio-Energy Projects. In PyNe Final Report to the EC; European Commission: Brussels, Belgium, 2001. [Google Scholar]
- Islam, M.N.; Ani, F.N. Techno-economics of rice husk pyrolysis, conversion with catalytic treatment to produce liquid fuel. Bisour. Technol. 2000, 73, 67–75. [Google Scholar] [CrossRef]
- Polagye, L.B.; Hodgson, K.T.; Malte, P.C. An economic analysis of bio-energy options using thinnings from overstocked forests. Biomass Bioenergy 2007, 31, 105–125. [Google Scholar] [CrossRef]
- Wright, M.M.; Daugaard, D.E.; Satrio, J.A.; Brown, R.C. Techno-economic analysis of biomass fast pyrolys to transportation fuels. Fuel 2010, 89, S2–S10. [Google Scholar] [CrossRef]
- Luo, Z.; Wang, S.; Liao, Y.; Zhou, J.; Gu, Y.; Cen, K. Research on biomass fast pyrolysis for liquid fuel. Biomass Bioenergy 2004, 26, 455–462. [Google Scholar] [CrossRef]
- Gregoire, C.E.; Bain, R.L. Technoeconomic analysis of the production of biocrude from wood. Biomass Bioenergy 1994, 7, 275–283. [Google Scholar] [CrossRef]
- Cottam, M.L.; Bridgwater, A.V. Techno-economic modeling of biomass flash pyrolysis and upgrading systems. Biomass Bioenergy 1994, 7, 267–273. [Google Scholar] [CrossRef]
- Gregoire, C.E. Technoeconomic Analysis of the Production of Biocrude from Wood; NREL/TP-430–5435; National Renewable Energy Laboratory: Golden, CO, USA, 1992. [Google Scholar]
- Thewys, T.; Kuppens, T. Economics of willow pyrolysis after phytoextraction. Int. J. Phytoremediat. 2008, 10, 561–583. [Google Scholar] [CrossRef]
- Voets, T.; Kuppens, T. Economics of electricity and heat production by gasification or flash pyrolysis of short rotation coppice in Flanders (Belgium). Biomass Bioenergy 2011, 35, 1912–1924. [Google Scholar] [CrossRef]
- Bridgwater, A.V.; Toft, A.J. A techno-economic comparison of power production by biomass fast pyrolysis with gasification and combustion. Renew. Sustain. Energy Rev. 2002, 6, 181–248. [Google Scholar] [CrossRef]
- Peacocke, G.V.C.; Bridgwater, A.V. Techno-Economic Assessment of Power Production from the Wellman and btg Fast Pyrolysis Processes. In Science in Thermal and Chemical Biomass Conversion; Bridgwater, A.V., Boocock, D.G., Eds.; CPL Press: Birmingham, UK, 1785. [Google Scholar]
- Rogers, J.G.; Brammer, J.G. Estimation of the production cost of fast pyrolysis bio-oil. Biomass Bioenergy 2012, 36, 208–217. [Google Scholar] [CrossRef]
- Lora, E.S.; Andrade, R.V. Biomass as energy source in Brazil. Renew. Sustain. Energy Rev. 2009, 13, 777–788. [Google Scholar] [CrossRef]
- Demirbas, M.F.; Balat, M.; Balat, H. Potential contribution of biomass to the sustainable energy development. Energy Convers. Manag. 2009, 50, 1746–1760. [Google Scholar] [CrossRef]
- Yu, S.; Tao, J. Simulation-based life cycle assessment of energy efficiency of biomass-based ethanol fuel from different feedstocks in China. Energy 2009, 34, 476–484. [Google Scholar] [CrossRef]
- Saxena, R.C.; Adhikari, D.K.; Goyal, H.B. Biomass-based energy fuel through biochemical routes. Renew. Sustain. Energy Rev. 2009, 13, 167–178. [Google Scholar] [CrossRef]
- Hill, J. Environmental costs and benefits of transportation biofuel production from food- and lignocelluloses-based energy crops. Agron. Sustain. Devel. 2007, 27, 1–12. [Google Scholar] [CrossRef]
- Homepage of DynaMotive Energy Systems Corporation. Available online: http://www.dynamotive.com (accessed on 15 September 2010).
- Homepage of Ensyn Group Inc. Available online: http://www.ensyn.com (accessed on 10 June 2011).
- Homepage of Renewable Oil International. Available online: http://www.renewableoil.com (accessed on 12 October 2011).
- PYTEC. Bio-Fuel Based on Agricultural and Forest Residues—The Alternative Renewable Energy of Our Future; PYTEC: Northampton, UK, 2005. [Google Scholar]
- Meier, D. New Ablative Pyrolyzer in Operation in GERMANY. Available online: http://www.pyne.co.uk/Resources/user/PYNE%20Newsletters/PyNews%2017.pdf (accessed on 20 November 2012).
- Boulard, D.C. Bio-Oil; The New Crude. In Presented at Ensyn RTPTM Bio-Refinary: Bio-oil Briefing session, Concord, NH, USA, 16 August 2002.
- Brown, D.B. Continuous ablative regenerator system. In Proceedings of the 2nd EU-Canada Workshop on Thermal Biomass Processing; Bridgwater, A.V., Hogan, E.N., Eds.; CPL Press: Newbury, UK, 1996; pp. 96–100. [Google Scholar]
- Menendez, J.A.; Dominguez, A.; Inguanzo, M.; Pis, J.J. Microwave pyrolysis of sewage sludge: Analysis of the gas fraction. J. Appl. Pyrolysis 2004, 71, 657–667. [Google Scholar] [CrossRef]
- Zhao, X.; Hunang, H.; Wu, C.; Li, H.; Chen, Y. Biomass pyrolysis in an Argon/Hydrogen plasma reactor. Chem. Eng. Technol. 2001, 24, 197–199. [Google Scholar]
- Scott, D.S.; Majerski, P.; Piskorz, J.; Radlein, D. A second look at fast pyrolysis of biomass—The RTI process. J. Anal. Appl. Pyrolysis 1999, 51, 23–37. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Moghtaderi, B. The state-of-the-art in pyrolysis modelling of lignocellulosic solid fuels. Fire Mater. 2006, 30, 1–34. [Google Scholar] [CrossRef]
- Papadikis, K.; Gu, S.; Bridgwater, A.V. A CFD approach on the effect of particle size on char entrainment in bubbling fluidised bed reactors. Biomass Bioenergy 2010, 34, 21–29. [Google Scholar] [CrossRef]
- Babu, B.V.; Chaurasia, A.S. Dominant design variables in pyrolysis of biomass particles of different geometries in thermally thick regime. Chem. Eng. Sci. 2004, 59, 611–622. [Google Scholar] [CrossRef]
- Papadikis, K.; Gu, S.; Bridgwater, A.V. Eulerian model for the condensation of pyrolysis vapors in a water condenser. Energy Fuels 2011, 25, 1859–1868. [Google Scholar] [CrossRef]
- Babu, B.V.; Chaurasia, A.S. Modeling, simulation and estimation of optimum parameters in pyrolysis of biomass. Energy Convers. Manag. 2003, 44, 2135–2158. [Google Scholar] [CrossRef]
- Tanoue, K.-I.; Hinauchi, T.; Oo, T.; Nishimura, T.; Taniguchi, M.; Sasauchi, K.-I. Modeling of heterogeneous chemical reactions caused in pyrolysis of biomass particles. Adv. Powder Technol. 2007, 18, 825–840. [Google Scholar] [CrossRef]
- Thunman, H.; Leckner, B. Thermal conductivity of wood—Models for different stages of combustion. Biomass Bioenergy 2002, 23, 47–54. [Google Scholar] [CrossRef]
- Boateng, A.A.; Mtui, P.L. CFD modeling of space-time evolution of fast pyrolysis products in a bench-scale fluidized-bed reactor. Appl. Ther. Eng. 2012, 33–34. [Google Scholar]
- Papadikis, K.; Gu, S.; Bridgwater, A.V. 3D simulation of the effects of sphericity on char entrainment in fluidised beds. Fuel Proc. Technol. 2010, 91, 749–758. [Google Scholar] [CrossRef]
- Papadikis, K.; Gu, S.; Bridgwater, A.V.; Gerhauser, H. Application of CFD to model fast pyrolysis of biomass. Fuel Proc. Technol. 2009, 90, 504–512. [Google Scholar] [CrossRef]
- Brown, A.L.; Dayton, D.C.; Nimlos, M.R.; Daily, J.W. Design and characterization of an entrained flow reactor for the study of biomass pyrolysis chemistry at high heating rates. Energy Fuels 2001, 15, 1276–1285. [Google Scholar] [CrossRef]
- Brown, T.R.; Wright, M.M.; Brown, R.C. Estimating profitability of two biochar production scenarios: slow pyrolysis vs. fast pyrolysis. Biofuels Bioprod. Bioref. 2011, 5, 54–68. [Google Scholar] [CrossRef]
- Papadikis, K.; Gu, S.; Bridgwater, A.V. Computational modelling of the impact of particle size to the heat transfer coefficient between biomass particles and a fluidised bed. Fuel Proc. Technol. 2010, 91, 68–79. [Google Scholar] [CrossRef]
- Yuen, R.K.K.; Yeoh, G.H.; Davis, D.V.; Leonardi, E. Modelling the pyrolysis of wet wood-I. Three-dimensional formulation and analysis. Int. J. Heat Mass Transfer 2007, 50, 4371–4386. [Google Scholar] [CrossRef]
- Yuen, R.K.K.; Yeoh, G.H.; Davis, D.V.; Leonardi, E. Modelling the pyrolysis of wet wood-II. Three-dimensional cone calorimeter simulation. Int. J. Heat Mass Transfer 2007, 50, 4387–4399. [Google Scholar] [CrossRef]
- Hoekman, S.K. Biofuels in the U.S.—Challenges and opportunities. Renew. Energy 2009, 34, 14–22. [Google Scholar] [CrossRef]
- Sarath, G.; Mitchell, R.B.; Sattler, S.E.; Funnell, D.; Pedersen, J.F.; Graybosch, R.A.; Vogel, K.P. Opportunities and roadblocks in utilizing forages and small grains for liquid fuels. J. Ind. Microbiol. Biotechnol. 2008, 35, 343–354. [Google Scholar] [CrossRef] [PubMed]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jahirul, M.I.; Rasul, M.G.; Chowdhury, A.A.; Ashwath, N. Biofuels Production through Biomass Pyrolysis —A Technological Review. Energies 2012, 5, 4952-5001. https://doi.org/10.3390/en5124952
Jahirul MI, Rasul MG, Chowdhury AA, Ashwath N. Biofuels Production through Biomass Pyrolysis —A Technological Review. Energies. 2012; 5(12):4952-5001. https://doi.org/10.3390/en5124952
Chicago/Turabian StyleJahirul, Mohammad I., Mohammad G. Rasul, Ashfaque Ahmed Chowdhury, and Nanjappa Ashwath. 2012. "Biofuels Production through Biomass Pyrolysis —A Technological Review" Energies 5, no. 12: 4952-5001. https://doi.org/10.3390/en5124952
APA StyleJahirul, M. I., Rasul, M. G., Chowdhury, A. A., & Ashwath, N. (2012). Biofuels Production through Biomass Pyrolysis —A Technological Review. Energies, 5(12), 4952-5001. https://doi.org/10.3390/en5124952






