Tuning Solid-State Reaction Pathways Using Molecular Sulfur Precursors to Synthesize FeS Anodes of Li-Ion Batteries for Boosted Electrochemical Performance
Abstract
1. Introduction
2. Experimental
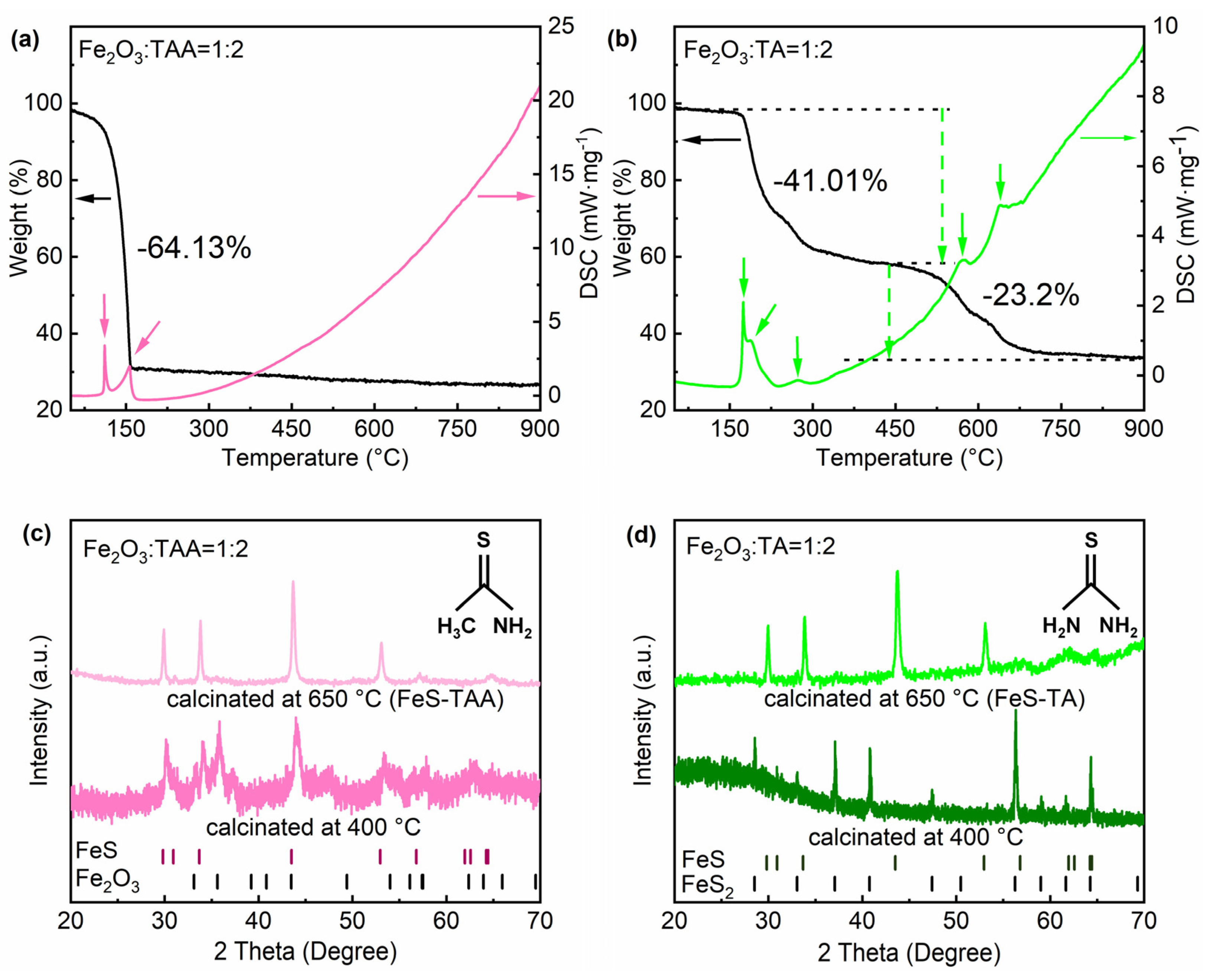
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Foroozan, T.; Sharifi-Asl, S.; Shahbazian-Yassar, R. Mechanistic Understanding of Li Dendrites Growth by In-Situ/Operando Imaging Techniques. J. Power Sources 2020, 461, 228135. [Google Scholar] [CrossRef]
- Gao, X.; Zhou, Y.; Han, D.; Zhou, J.; Zhou, D.; Tang, W.; Goodenough, J.B. Thermodynamic Understanding of Li-Dendrite Formation. Joule 2020, 4, 1864–1879. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, Y.; Xu, H.; Wang, L.; Lu, X.; He, X. Li4Ti5O12 Spinel Anode: Fundamentals and Advances in Rechargeable Batteries. InfoMat 2021, 4, e12228. [Google Scholar] [CrossRef]
- Yan, S.; Luo, S.; Liu, H.; Yang, L.; Wang, Q.; Zhang, Y.; Liu, X. In-Situ Partial Reduction-Sulfurized Fe3O4@FeS Based on Pickling Iron Red as a Versatile Electrode for High-Performance Lithium Ion Batteries and Supercapacitor Devices. Surf. Coat. Technol. 2022, 429, 127980. [Google Scholar] [CrossRef]
- Chen, L.; Han, L.; Liu, X.; Li, Y.; Wei, M. General Synthesis of Sulfonate-based Metal-organic Framework Derived Composite of MxSy@N/S-Doped Carbon for High-performance Lithium/Sodium Ion Batteries. Chem. Eur. J. 2021, 27, 2104–2111. [Google Scholar] [CrossRef]
- Vissers, D.R. Lithium-Aluminum/Iron Sulfide Batteries. Mater. Adv. Batter. 1980, 2, 47–90. [Google Scholar]
- Henriksen, G.L.; Vissers, D.R. Lithium-Aluminum/Iron Sulfide Batteries. J. Power Sources 1994, 51, 115–128. [Google Scholar] [CrossRef]
- Kim, B.-C.; Takada, K.; Ohta, N.; Seino, Y.; Zhang, L.; Wada, H.; Sasaki, T. All Solid State Li-ion Secondary Battery with FeS Anode. Solid State Ion. 2005, 176, 2383–2387. [Google Scholar] [CrossRef]
- Xu, Q.-T.; Li, J.-C.; Xue, H.-G.; Guo, S.-P. Binary Iron Sulfides as Anode Materials for Rechargeable Batteries: Crystal Structures, Syntheses, and Electrochemical Performance. J. Power Sources 2018, 379, 41–52. [Google Scholar] [CrossRef]
- Ramakrishnan, P.; Baek, S.-H.; Park, Y.; Kim, J.H. Nitrogen and Sulfur Co-Doped Metal Monochalcogen Encapsulated Honeycomb Like Carbon Nanostructure as a High Performance Lithium-Ion Battery Anode Material. Carbon 2017, 115, 249–260. [Google Scholar] [CrossRef]
- Wang, M.; Xue, H.; Guo, S. In Situ Hydrothermal Synthesis of rGO-Wrapped Fe1−xS Particles for Lithium Storage. J. Mater. Res. 2019, 34, 3186–3194. [Google Scholar] [CrossRef]
- Liu, Z.; Hu, F.; Xiang, J.; Yue, C.; Lee, D.; Song, T. A Nano-Micro Hybrid Structure Composed of Fe7S8 Nanoparticles Embedded in Nitrogen-Doped Porous Carbon Framework for High-Performance Lithium/Sodium-Ion Batteries. Part. Part. Syst. Charact. 2018, 35, 1800163. [Google Scholar] [CrossRef]
- Tang, Q.; Jiang, Q.; Wu, T.; Wu, T.; Ding, Z.; Wu, J.; Yu, H.; Huang, K. Binary Iron Sulfide as a Low-Cost and High-Performance Anode for Lithium-/Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2020, 12, 52888–52898. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, B.; Wang, G.; Liu, X.; Wang, H. A Scalable Approach to Fabricate Metal Sulfides/Graphene/Carbon Nanotubes Composites with Superior Electrochemical Performances for Lithium and Sodium Ion Batteries. Electrochim. Acta 2017, 258, 764–772. [Google Scholar] [CrossRef]
- Zhang, X.; Li, J.; Li, J.; Han, L.; Lu, T.; Zhang, X.; Zhu, G.; Pan, L. 3D TiO2@Nitrogen-Doped Carbon/Fe7S8 Composite Derived from Polypyrrole-Encapsulated Alkalized MXene as Anode Material for High-Performance Lithium-Ion Batteries. Chem. Eng. J. 2020, 385, 123394. [Google Scholar] [CrossRef]
- Cho, J.S.; Park, J.; Kang, Y.C. Porous FeS Nanofibers with Numerous Nanovoids Obtained by Kirkendall Diffusion Effect for use as Anode Materials for Sodium-Ion Batteries. Nano Res. 2017, 10, 897–907. [Google Scholar] [CrossRef]
- Guo, S.; Li, J.; Ma, Z.; Chi, Y.; Xue, H. A Facile Method to Prepare FeS/Porous Carbon Composite as Advanced Anode Material for Lithium-Ion Batteries. J. Mater. Sci. 2016, 52, 2345–2355. [Google Scholar] [CrossRef]
- Chin, P.P.; Ding, J.; Yi, J.B.; Liu, B.H. Synthesis of FeS2 and FeS Nanoparticles by High-Energy Mechanical Milling and Mechanochemical Processing. J. Alloy. Compd. 2005, 390, 255–260. [Google Scholar] [CrossRef]
- Soori, M.; Zarezadeh, K.; Sheibani, S.; Rashchi, F. Mechano-Chemical Processing and Characterization of Nano-Structured FeS Powder. Adv. Powder Technol. 2016, 27, 557–563. [Google Scholar] [CrossRef]
- Ma, R.; Yin, L.; Li, L.; Zhang, S.; Wen, T.; Zhang, C.; Wang, X.; Chen, Z.; Hayat, T.; Wang, X. Comparative Investigation of Fe2O3 and Fe1–xS Nanostructures for Uranium Decontamination. ACS Appl. Nano Mater. 2018, 1, 5543–5552. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, L.; Wang, J.; Liang, J.; Xi, L. Facile Method for Adjustable Preparation of Nano-Fe7S8 Supported by Carbon as the Anode for Enhanced Lithium/Sodium Storage Properties in Li/Na-Ion Batteries. Electrochim. Acta 2019, 322, 134763. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, T.; Liang, J.; Zhu, Y.; Lin, N.; Qian, Y. A Potential Pyrrhotite (Fe7S8) Anode Material for Lithium Storage. RSC Adv. 2015, 5, 14828–14831. [Google Scholar] [CrossRef]
- Ho, V.; An, H.; Hong, M.; Lee, S.; Kim, J.; Park, M.B.; Mun, J. A Low Temperature Self-Assembled ZrO2 Layer as a Surface Modification for High Energy Density Ni-Rich Cathode Materials in a Lithium-Ion Battery. Energy Technol. 2021, 9, 2000800. [Google Scholar] [CrossRef]
- Behl, M.; Jain, P.K. Catalytic Activation of a Solid Oxide in Electronic Contact with Gold Nanoparticles. Angew. Chem. Int. Ed. 2014, 54, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Khera, N.; Jeevanandam, P. CuCo2S4 Nanoparticles Synthesized via a Thermal Decomposition Approach: Evaluation of their Potential as Peroxidase Mimics. Nanoscale 2024, 16, 18108–18118. [Google Scholar] [CrossRef] [PubMed]
- Stradella, L.; Argentero, M. A Study of the Thermal Decomposition of Urea of Related Compounds and Thiourea using DSC and TG-EGA. Thermochimica Acta 1993, 219, 315–323. [Google Scholar] [CrossRef]
- Otto, K.; Bombicz, P.; Madarász, J.; Oja Acik, I.; Krunks, M.; Pokol, G. Structure and Evolved Gas Analyses (TG/DTA-MS and TG-FTIR) of Mer-Trichlorotris(Thiourea)-Indium(III), a Precursor for Indium Sulfide Thin Films. J. Therm. Anal. Calorim. 2011, 105, 83–91. [Google Scholar] [CrossRef]
- Zhang, C.; Niu, M. Study on Mechanism of Isomerization between Ammonium Thiocyanate and Thiourea. J. Mol. Struct. 2016, 1125, 643–648. [Google Scholar] [CrossRef]
- Arnikar, H.J.; Kapadi, A.H.; Naik, D.G.; Chandwadkar, J.G. Radiation-Induced Isomerization of Thiourea into Ammonium Thiocyanate. J. Radioanal. Nucl. Chem. 1994, 185, 227–230. [Google Scholar] [CrossRef]
- Wang, Z.D.; Yoshida, M.; George, B. Theoretical Study on the Thermal Decomposition of Thiourea. Comput. Theor. Chem. 2013, 1017, 91–98. [Google Scholar] [CrossRef]
- Zhang, X.; Jones, G.O.; Hedrick, J.L.; Waymouth, R.M. Fast and Selective Ring-Opening Polymerizations by Alkoxides and Thioureas. Nat. Chem. 2016, 8, 1047–1053. [Google Scholar] [CrossRef]
- Xu, X.; Pacey, P.D. An Induction Period in the Pyrolysis of Acetylene. Phys. Chem. Chem. Phys. 2001, 3, 2836–2844. [Google Scholar] [CrossRef]
- Ruiz-Bermejo, M.; de la Fuente, J.L.; Pérez-Fernández, C.; Mateo-Martí, E. A Comprehensive Review of HCN-Derived Polymers. Processes 2021, 9, 597. [Google Scholar] [CrossRef]
- Díaz-Ortiz, A.; de la Hoz, A.; Moreno, A.; Sánchez-Migallón, A.; Valiente, G. Synthesis of 1,3,5-Triazines in Solvent-Free Conditions Catalysed by Silica-Supported Lewis Acids. Green Chem. 2002, 4, 339–343. [Google Scholar] [CrossRef]
- Hoch, M.; Johnston, H.L. Formation, Stability and Crystal Structure of the Solid Aluminum Suboxides Al2O and AlO1. J. Am. Chem. Soc. 1954, 76, 250–2561. [Google Scholar] [CrossRef]
- Tuinstra, F.; Koenig, J.L. Raman Spectrum of Graphite. J. Chem. Phys. 1970, 53, 1126–1130. [Google Scholar] [CrossRef]
- Wrogemann, J.M.; Fromm, O.; Deckwirth, F.; Beltrop, K.; Heckmann, A.; Winter, M.; Placke, T. Impact of Degree of Graphitization, Surface Properties and Particle Size Distribution on Electrochemical Performance of Carbon Anodes for Potassium-Ion Batteries. Batter. Supercaps 2022, 5, e202200045. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman Spectra of Disordered and Amorphous Carbon. Phys. Rev. B 2020, 61, 14095–14107. [Google Scholar] [CrossRef]
- Yang, D.; Yadav, D.; Jeon, I.; Seo, J.; Jeong, S.-Y.; Cho, C.R. Enhanced High-Rate Capability and Long Cycle Stability of FeS@NCG Nanofibers for Sodium-Ion Battery Anodes. ACS Appl. Mater. Interfaces 2022, 14, 44303–44316. [Google Scholar] [CrossRef]
- Hansson, E.B.; Odziemkowski, M.S.; Gillham, R.W. Formation of Poorly Crystalline Iron Monosulfides: Surface Redox Reactions on High Purity Iron, Spectroelectrochemical Studies. Corros. Sci. 2006, 48, 3767–3783. [Google Scholar] [CrossRef]
- Cooney, R.P.J.; Hall, J.R. Raman Spectra of Mercury(II) Nitrate in Aqueous Solution and as the Crystalline Hydrate. Aust. J. Chem. 1969, 22, 2117–2123. [Google Scholar] [CrossRef]
- Almanqur, L.; Vitorica-yrezabal, I.; Whitehead, G.; Lewis, D.J.; O’Brien, P. Synthesis of Nanostructured Powders and Thin Films of Iron Sulfide from Molecular Precursors. RSC Adv. 2018, 8, 29096–29103. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Zhang, Y.; Wang, N.; Zou, G.; Zhang, Q.; Gan, S.; He, L.; Bai, Z. Pod-Like Fe7S8@N−C Nanowires for High Performance Sodium Ion Batteries. Batter. Supercaps 2021, 5, e202100203. [Google Scholar] [CrossRef]
- Fan, H.; Qin, B.; Wang, Z.; Li, H.; Guo, J.; Wu, X.; Zhang, J. Pseudocapacitive Sodium Storage of Fe1−xS@N-Doped Carbon for Low-Temperature Operation. Sci. China Mater. 2019, 63, 505–515. [Google Scholar] [CrossRef]
- Xie, Z.; Zhang, L.; Li, L.; Deng, Q.; Jiang, G.; Wang, J.; Cao, B.; Wang, Y. A Ternary FeS2/Fe7S8@Nitrogen-Sulfur Co-Doping Reduced Graphene Oxide Hybrid Towards Superior-Performance Lithium Storage. Prog. Nat. Sci. Mater. Int. 2021, 31, 207–214. [Google Scholar] [CrossRef]
- Yang, Y.; Li, Z.; Xu, S.; Tang, Y.; Lee, C.-S.; Zhang, W. Hierarchically Nanostructured ZnCo2O4 Particles in 3D Graphene Networks for High-Rate and Long-Life Lithium Ion Batteries. Mater. Today Energy 2019, 12, 46–52. [Google Scholar] [CrossRef]
- Ding, X.; Du, C.; Li, J.; Huang, X. FeS2 Microspheres Wrapped by N-Doped rGO from an Fe-Based Ionic Liquid Precursor for Rechargeable Lithium Ion Batteries. Sustain. Energy Fuels 2019, 3, 701–708. [Google Scholar] [CrossRef]
- Du, Y.; Wu, S.; Huang, M.; Tian, X. Reduced Graphene Oxide-Wrapped Pyrite as Anode Materials for Li-Ion Batteries with Enhanced Long-term Performance under Harsh Operational Environments. Chem. Eng. J. 2017, 326, 257–264. [Google Scholar] [CrossRef]
- Xu, X.; Ma, Q.; Zhang, Z.; Peng, H.; Liu, J.; Mao, C.; Li, G. Pomegranate-Like Mesoporous Microspheres Assembled by N-doped Carbon Coated Fe1−xS Nanocrystals for High-Performance Lithium Storage. J. Alloys Compd. 2019, 797, 952–960. [Google Scholar] [CrossRef]
- Csákberényi-Malasics, D.; Rodriguez-Blanco, J.D.; Kis, V.K.; Rečnik, A.; Benning, L.G.; Pósfai, M. Structural Properties and Transformations of Precipitated FeS. Chem. Geol. 2012, 294, 249–258. [Google Scholar] [CrossRef]
- Wang, H.; Salveson, I. A Review on the Mineral Chemistry of the Non-Stoichiometric Iron Sulphide, Fe1−xS (0 ≤ x ≤ 0.125): Polymorphs, Phase Relations and Transitions, Electronic and Magnetic Structures. Phase Transit. 2005, 78, 547–567. [Google Scholar] [CrossRef]
- Zhu, C.; Wen, Y.; van Aken, P.A.; Maier, J.; Yu, Y. High Lithium Storage Performance of FeS Nanodots in Porous Graphitic Carbon Nanowires. Adv. Funct. Mater. 2015, 25, 2335–2342. [Google Scholar] [CrossRef]
- Guo, D.; Qian, X.; Jin, L.; Yao, S.; Shen, X.; Li, T.; Qin, S. Graphene Oxide Coated Nanosheet-Like γ-MnS@KB-S Fabricated by Spray Drying for High Energy Density Li-S Batteries. Int. J. Energy Res. 2020, 44, 11274–11287. [Google Scholar] [CrossRef]
- Duan, M.; Meng, Y.; Zhang, H.; Zhao, G.; Hu, J.; Ren, G.; Zhu, F. Facile Synthesis of Graphene-Like Carbon-Coated Ni3S2 Nanoparticles Self-Assembled on 3D Dendritic Nanostructure as High-Performance Anode Materials of Sodium-Ion Batteries. Ionics 2020, 26, 4511–4522. [Google Scholar] [CrossRef]
- Xie, J.; Carrasco, J.; Li, R.; Shen, H.; Chen, Q.; Yang, M. Novel 3D Flower-Like Micro/Nano-Structure FeS/N-Doped-C Composites as Advanced Cathodes with High Lithium Storage Performances. J. Power Sources 2019, 431, 226–231. [Google Scholar] [CrossRef]
- Zhang, Q.; Liao, J.; Liao, M.; Dai, J.; Ge, H.; Duan, T.; Yao, W. One-Dimensional Fe7S8@C Nanorods as Anode Materials for High-Rate and Long-Life Lithium-Ion Batteries. Appl. Surf. Sci. 2019, 473, 799–806. [Google Scholar] [CrossRef]
- Zhao, J.; Hu, Z.; Sun, D.; Jia, H.; Liu, X. MOF-Derived FeS/C Nanosheets for High Performance Lithium Ion Batteries. Nanomaterials 2019, 9, 492. [Google Scholar] [CrossRef]
- Ye, Z.; Zhang, W.; Liu, G.; Liu, L. Nitrogen-Sulfur Co-Doped FeS/C Nanofibers for High-Performance Lithium/Potassium Storage. J. Solid State Electrochem. 2023, 27, 1373–1381. [Google Scholar] [CrossRef]
- Vishwanathan, S.; Mohanta, M.K.; Jena, P.; Matte, H.S.S.R. Experimental and Theoretical Insights on Interface Engineered FeS/rGO as Anode for Fast-Charging Lithium- and Sodium-Ion Batteries. Small 2025, 21, 2410482. [Google Scholar] [CrossRef]
- Rudy, A.; Mironenko, A.; Naumov, V.; Novozhilova, A.; Skundin, A.; Fedorov, I. Determination of Diffusion Coefficients of Lithium in Solid Electrolyte LiPON. Batteries 2021, 7, 21. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods Fundamentals and Applications; Library of Congress Cataloging in Publication Data; John Wiley and Sons: Hoboken, NJ, USA, 2001. [Google Scholar]
- Liu, H.; Cao, Q.; Fu, L.J.; Li, C.; Wu, Y.P.; Wu, H.Q. Doping effects of zinc on LiFePO4 cathode material for lithium ion batteries. Electrochem. Commun. 2006, 8, 1553–1557. [Google Scholar] [CrossRef]
- Ma, L.; Hou, B.; Shang, N.; Zhang, S.; Wang, C.; Zong, L.; Song, J.; Wang, J.; Zhao, X. The precise synthesis of twin-born Fe3O4/FeS/carbon nanosheets for high-rate lithium-ion batteries. Mater. Chem. Front. 2021, 5, 4579–4588. [Google Scholar] [CrossRef]
- Mohanty, D.; Chang, M.-J.; Hung, I.M. The Effect of Different Amounts of Conductive Carbon Material on the Electrochemical Performance of the LiFePO4 Cathode in Li-Ion Batteries. Batteries 2023, 9, 515. [Google Scholar] [CrossRef]
- Xu, Y.; Li, W.; Zhang, F.; Zhang, X.; Zhang, W.; Lee, C.-S.; Tang, Y. In Situ Incorporation of FeS Nanoparticles/Carbon Nanosheets Composite with an Interconnected Porous Structure as a High-Performance Anode for Lithium Ion Batteries. J. Mater. Chem. A 2016, 4, 3697–3703. [Google Scholar] [CrossRef]






| Element | FeS-TAA | FeS-TA | ||
|---|---|---|---|---|
| at.% | sigma | at.% | sigma | |
| C | 40.0 | 0.7 | 39.7 | 0.5 |
| N | 0 | 0 | 19.8 | 0.4 |
| O | 4.2 | 0.2 | 3.7 | 0.1 |
| S | 28.6 | 0.2 | 19.1 | 0.1 |
| Fe | 27.2 | 0.2 | 17.6 | 0.1 |
| Sample | Rs | Rct | CPE1-T | CPE1-P | W1 |
|---|---|---|---|---|---|
| FeS-TAA | 4.372 | 404 | 2.4232 × 10−5 | 0.77363 | 8.591 |
| FeS-TA | 1.583 | 265.7 | 2.3502 × 10−5 | 0.78514 | 3.249 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Xu, M.; Tian, J.; Cao, S.; Cao, B.; Bai, Y.; Hu, X.; Yang, C.; Zhang, J.; Dong, C. Tuning Solid-State Reaction Pathways Using Molecular Sulfur Precursors to Synthesize FeS Anodes of Li-Ion Batteries for Boosted Electrochemical Performance. Energies 2025, 18, 4623. https://doi.org/10.3390/en18174623
Zhang X, Xu M, Tian J, Cao S, Cao B, Bai Y, Hu X, Yang C, Zhang J, Dong C. Tuning Solid-State Reaction Pathways Using Molecular Sulfur Precursors to Synthesize FeS Anodes of Li-Ion Batteries for Boosted Electrochemical Performance. Energies. 2025; 18(17):4623. https://doi.org/10.3390/en18174623
Chicago/Turabian StyleZhang, Xiaoyang, Mingxia Xu, Jialiang Tian, Shaorong Cao, Botao Cao, Yongmei Bai, Xiaojie Hu, Cunfeng Yang, Jiliang Zhang, and Chuang Dong. 2025. "Tuning Solid-State Reaction Pathways Using Molecular Sulfur Precursors to Synthesize FeS Anodes of Li-Ion Batteries for Boosted Electrochemical Performance" Energies 18, no. 17: 4623. https://doi.org/10.3390/en18174623
APA StyleZhang, X., Xu, M., Tian, J., Cao, S., Cao, B., Bai, Y., Hu, X., Yang, C., Zhang, J., & Dong, C. (2025). Tuning Solid-State Reaction Pathways Using Molecular Sulfur Precursors to Synthesize FeS Anodes of Li-Ion Batteries for Boosted Electrochemical Performance. Energies, 18(17), 4623. https://doi.org/10.3390/en18174623






