Supercritical Water Gasification of Coconut Shell Impregnated with a Nickel Nanocatalyst: Box–Behnken Design and Process Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Material and Chemicals
2.2. Identification of the pHZPC Value
2.3. Biomass Sample Preparation
2.4. Investigation of Biomass Characteristics
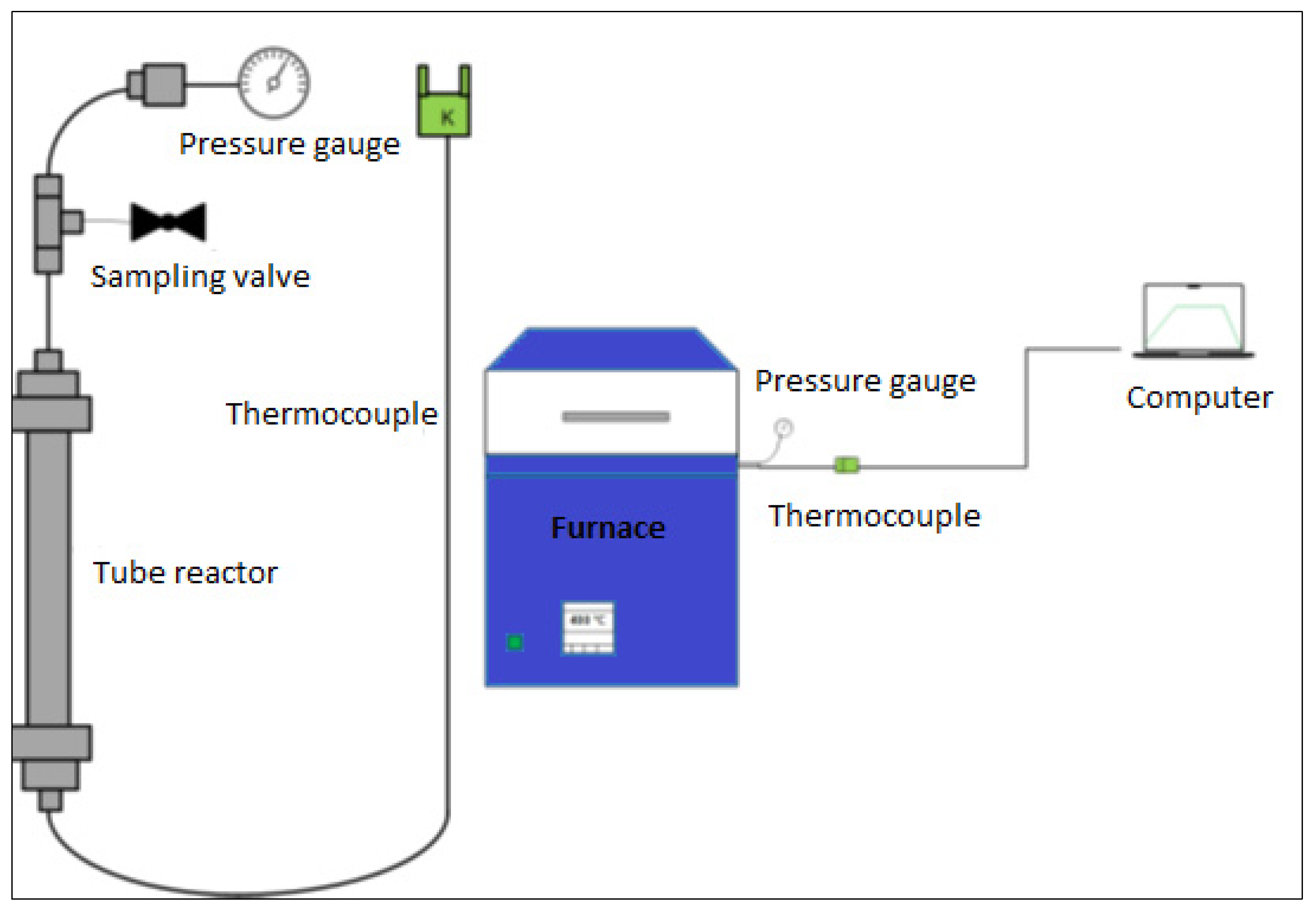
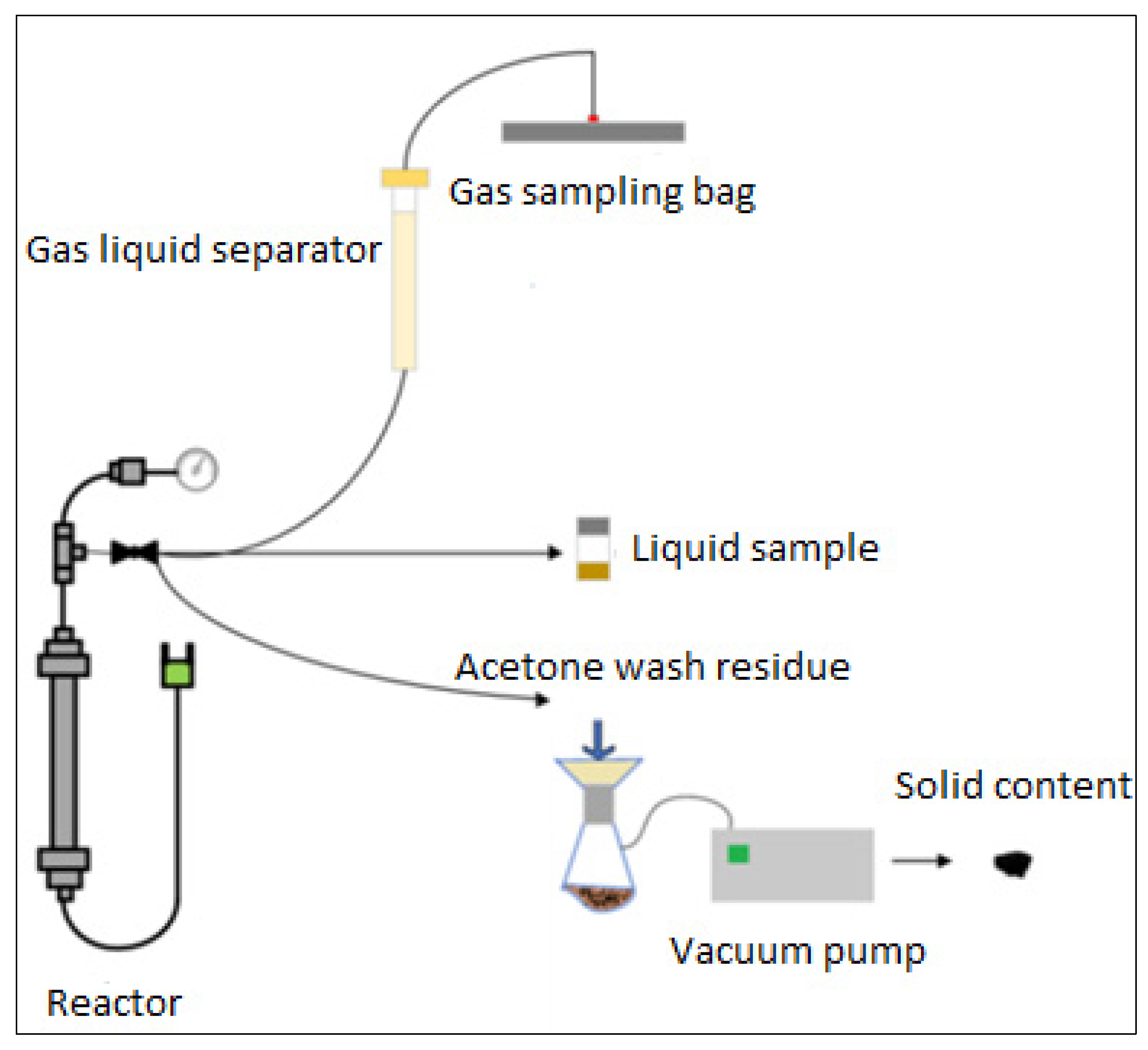
2.5. Supercritical Water Gasification Experiments
2.5.1. Reactor Assembly
2.5.2. Experimental Design
2.6. Performance and Gas Analysis
3. Results and Discussion
3.1. Optimization of pH
3.2. Characterization of Biomass
3.2.1. Proximate and Ultimate Analysis
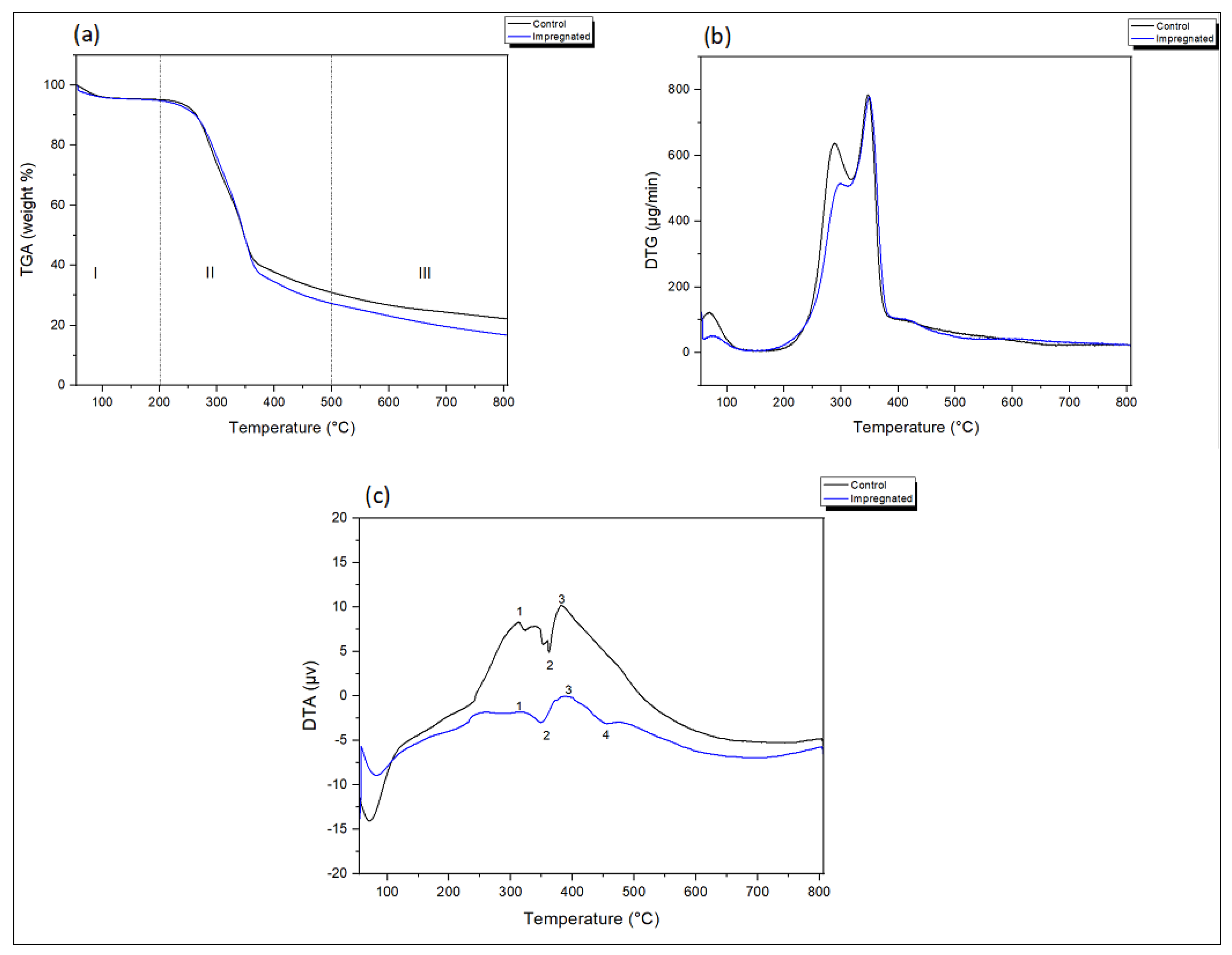
3.2.2. TGA/DTG Thermogravimetric/Derivate/Differential Thermogravimetric Analysis
3.2.3. SEM-EDX-Scanning Electron Microscopy-Energy Dispersive X-ray Spectroscopy Analysis
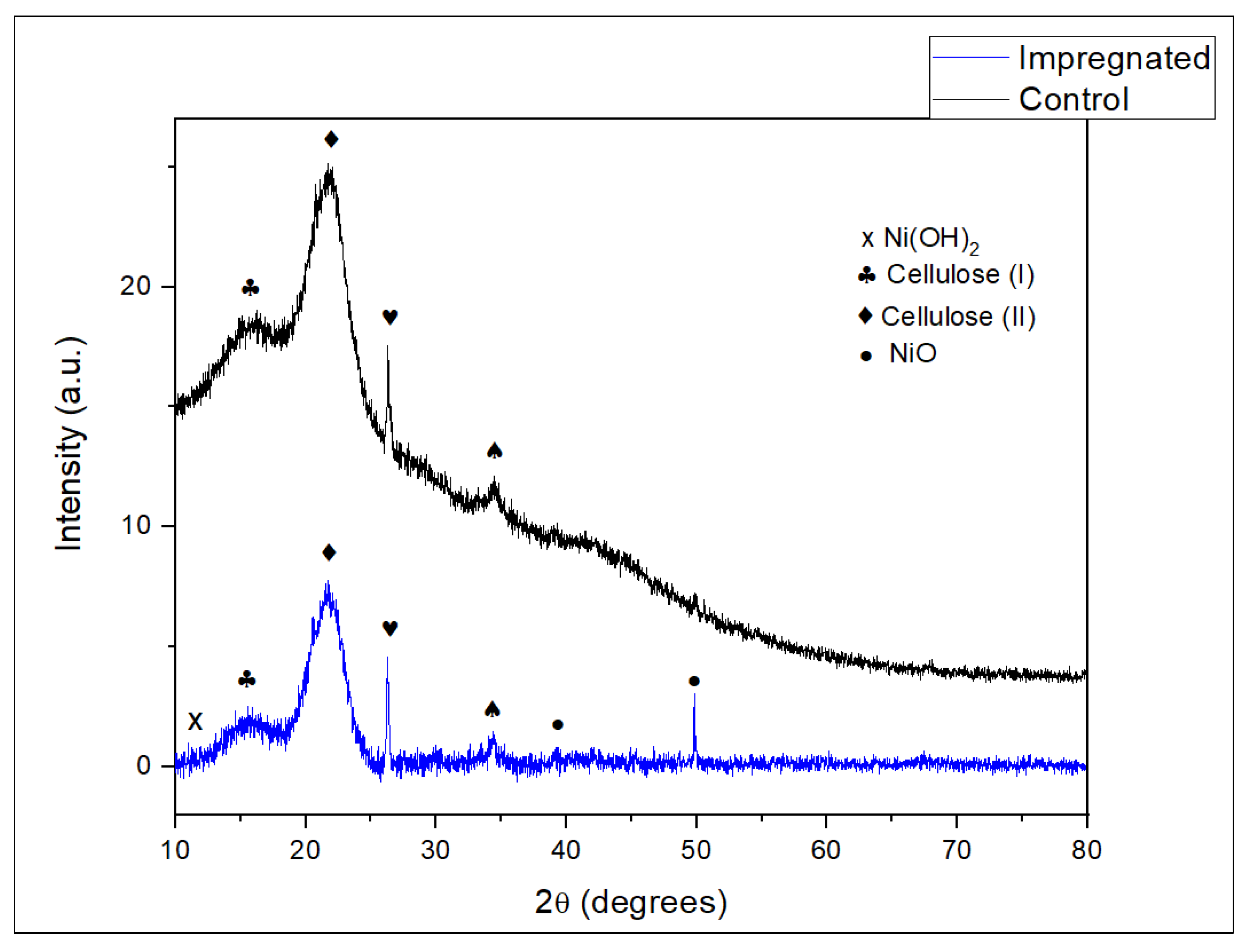
3.2.4. XRD-X-ray Diffraction Analysis
3.2.5. FTIR–Fourier Transform Infrared Spectroscopy
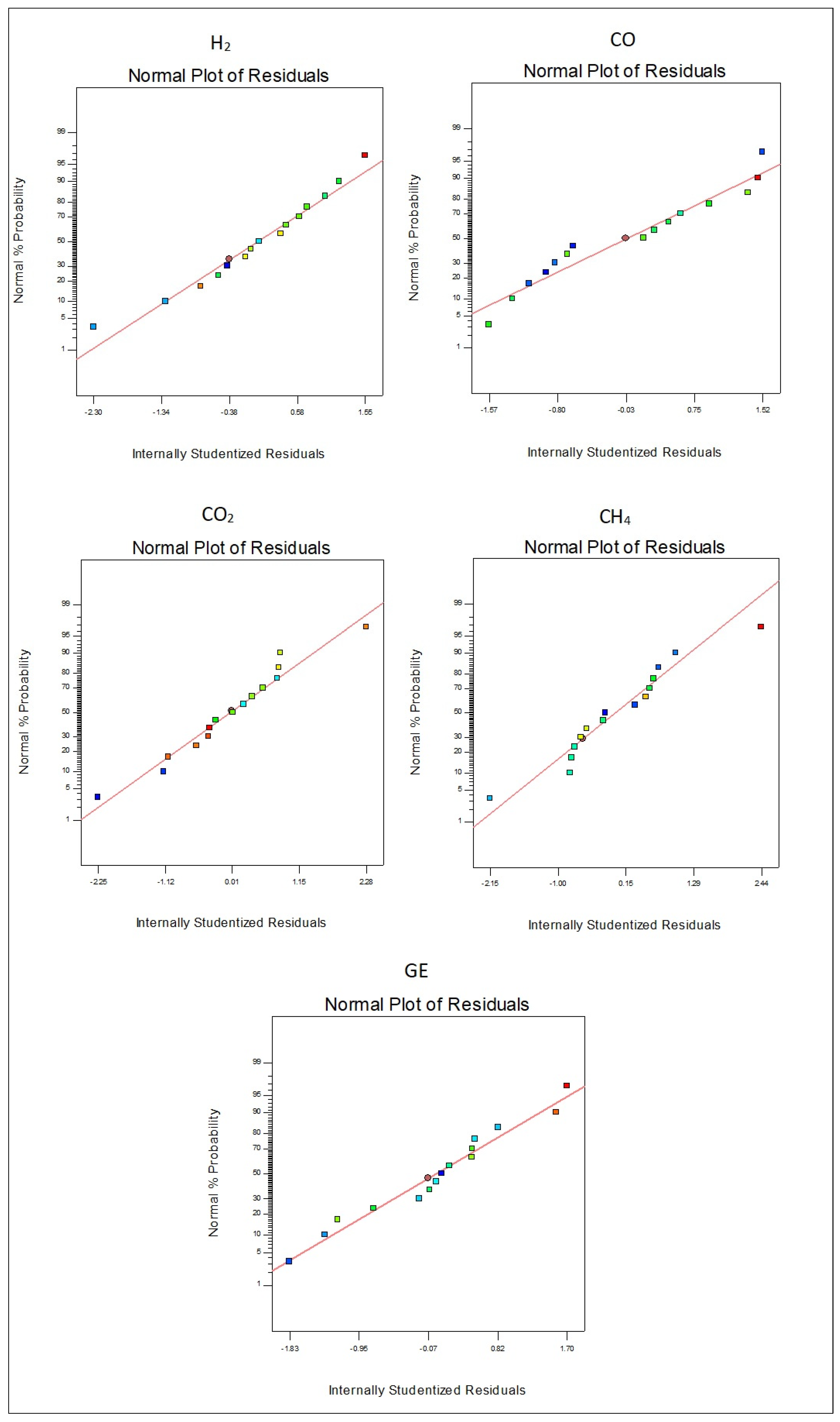
3.3. RSM Experimental Design
3.4. The Effect of Variables on Gasification Products and Performance
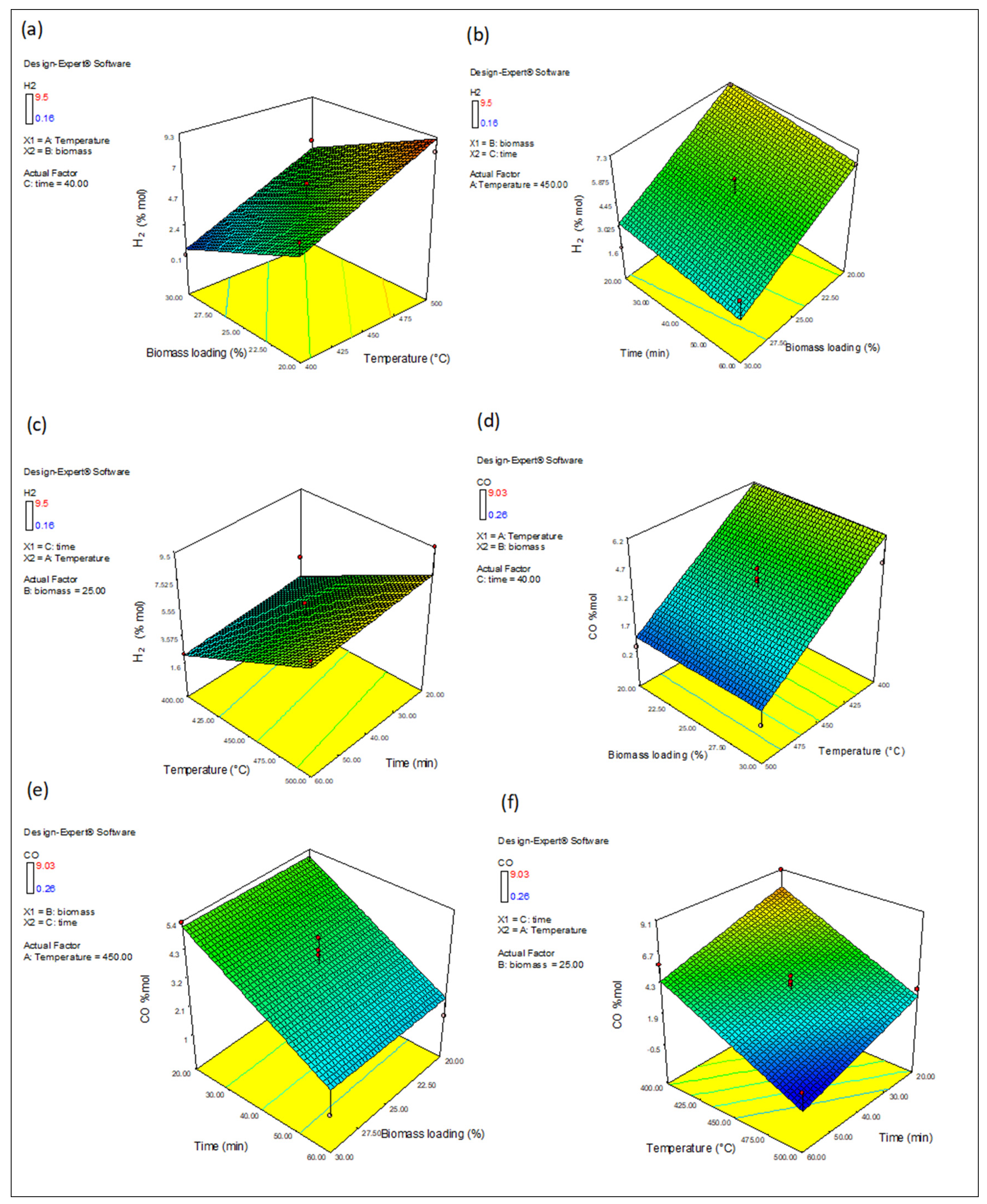
3.4.1. The Effect of Variables on H2 and CO Production
3.4.2. The Effect of Variables on CO2 and CH4 Production
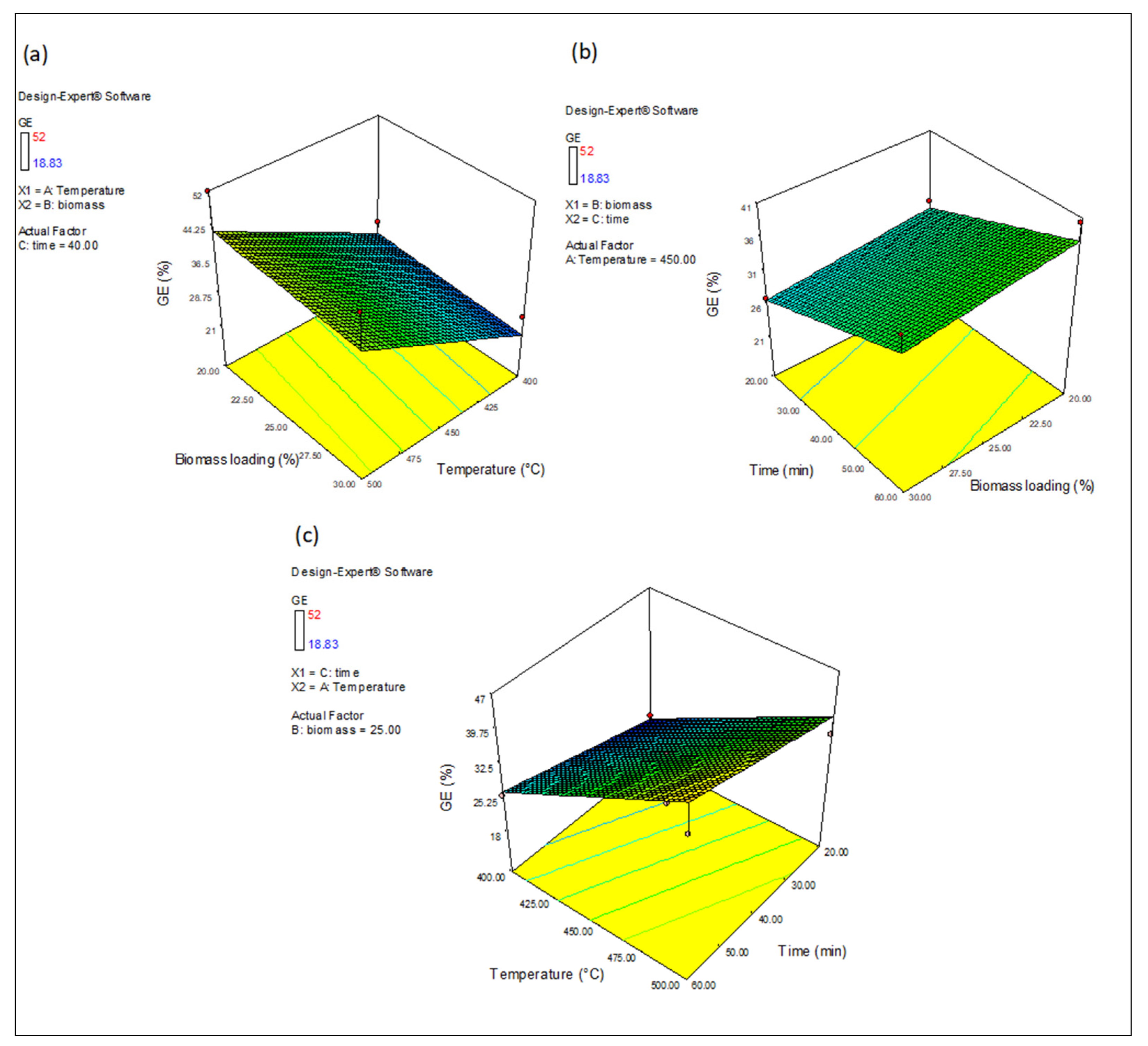
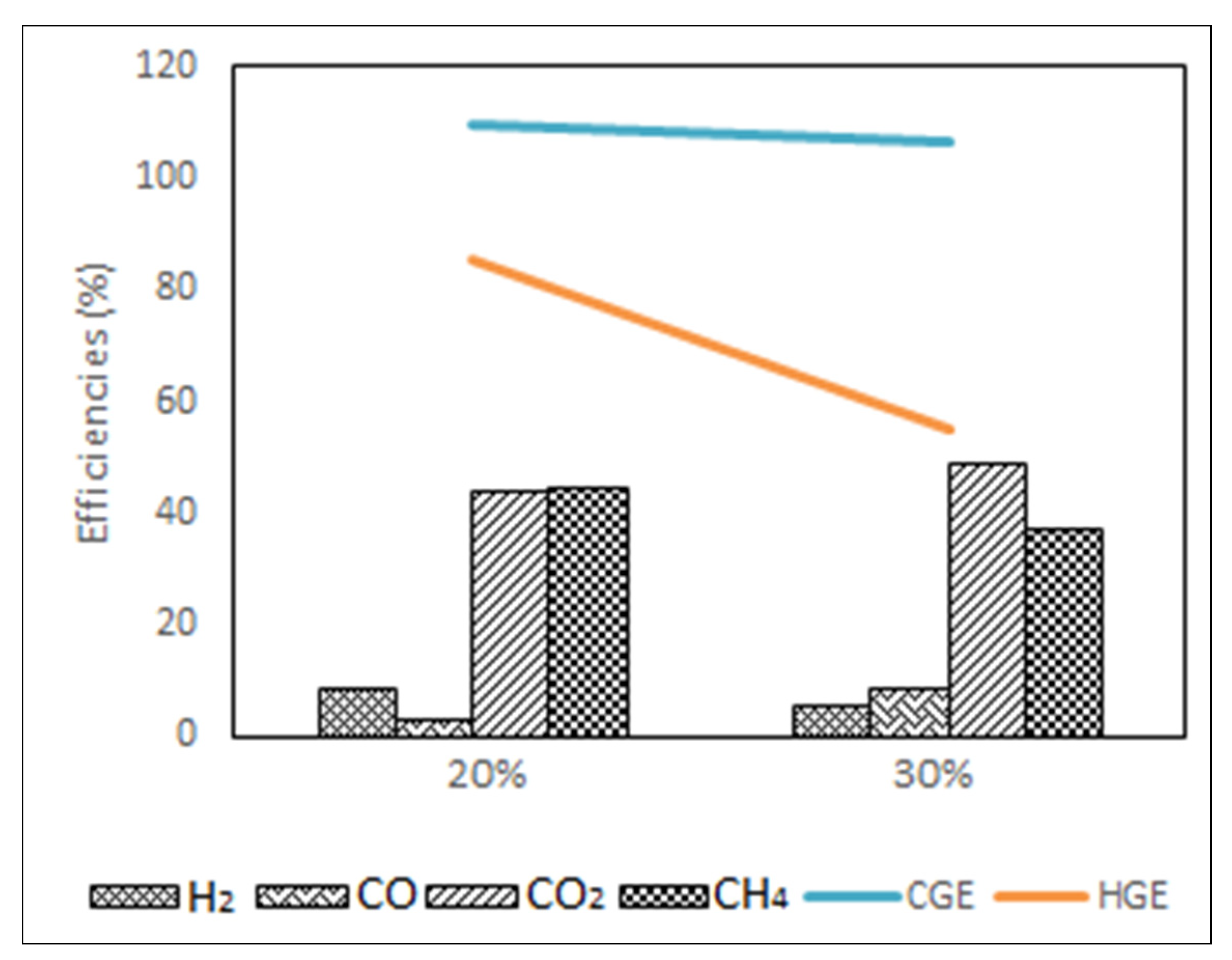
3.4.3. The Effect of Variables on GE, CGE and HGE
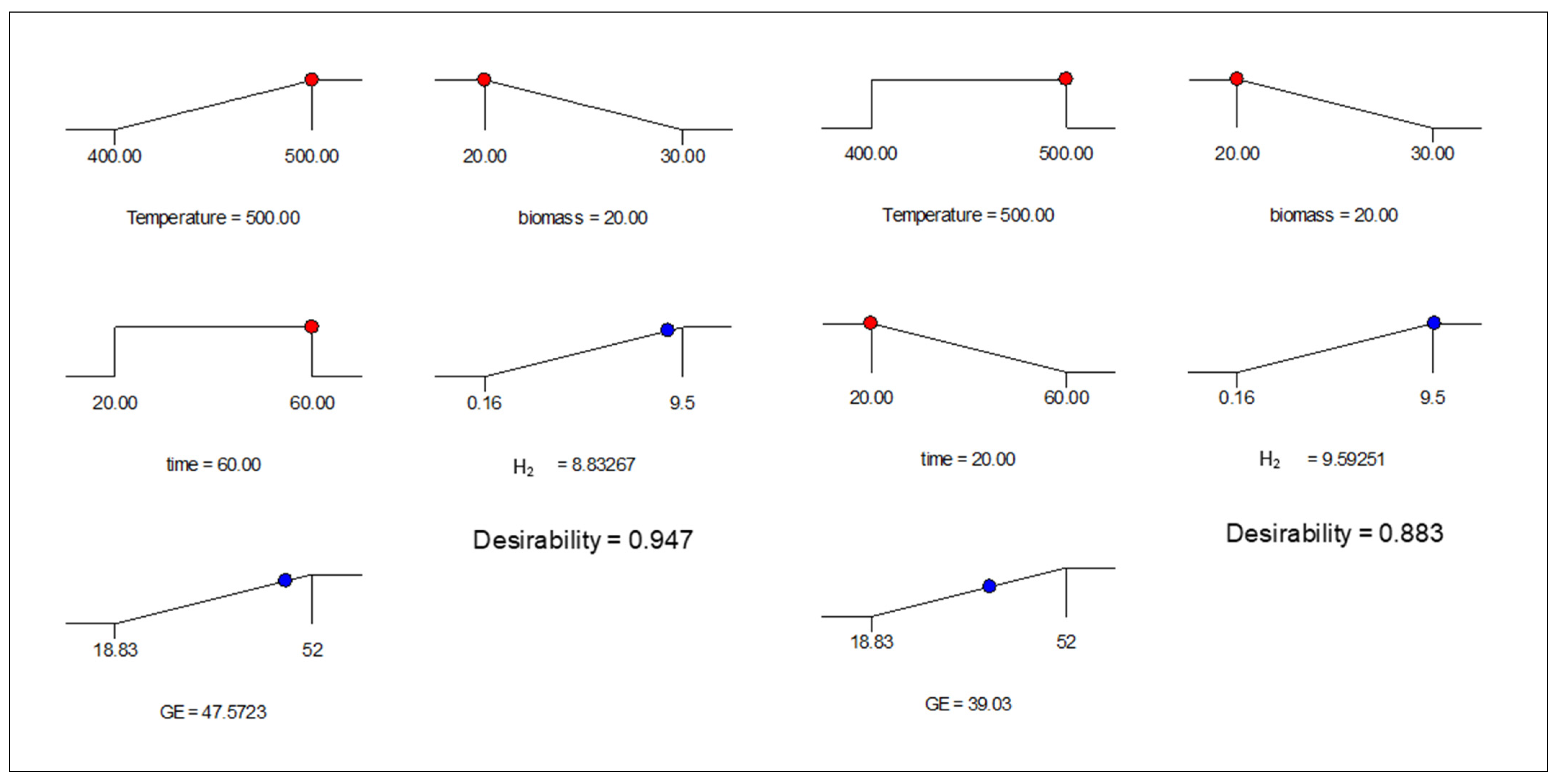
3.5. Optimization of the Model
3.6. Effect of Nickel Nanocatalyst on Supercritical Water Gasification
3.6.1. Effect of Nickel Nanocatalyst on the Gas Composition
Impact of the Process Variables on the Catalytic SCWG
Temperature
Biomass Loading
Reaction Time
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, R.K.; Sulaiman, S.A.; Yusup, S.; Dol, S.S.; Inayat, M.; Umar, H.A. Exploring the potential of coconut shell biomass for charcoal production. Ain Shams Eng. J. 2021, 13, 101499. [Google Scholar] [CrossRef]
- Azeta, O.; Ayeni, A.O.; Agboola, O.; Elehinafe, F.B. A review on the sustainable energy generation from the pyrolysis of coconut biomass. Sci. Afr. 2021, 13, e00909. [Google Scholar] [CrossRef]
- Caballero, D.R.C.; Barraza, J.; Gunasekaran, S.; Roa-Espinosa, A.; Colina-Márquez, J.; Machuca-Martínez, F.; Hernández-Ramírez, A.; Vazquez-Rodriguez, S. Experimental data on the production and characterization of biochars derived from coconut-shell wastes obtained from the Colombian Pacific Coast at low temperature pyrolysis. Data Brief 2020, 28, 104855. [Google Scholar] [CrossRef]
- Sarkar, J.K.; Wang, Q. Different Pyrolysis Process Conditions of South Asian Waste Coconut Shell and Characterization of Gas, Bio-Char, and Bio-Oil. Energies 2020, 13, 1970. [Google Scholar] [CrossRef]
- Akolgo, G.A.; Awafo, E.A.; Essandoh, E.O.; Owusu, P.A.; Uba, F.; Adu-Poku, K.A. Assessment of the potential of charred briquettes of sawdust, rice and coconut husks: Using water boiling and user acceptability tests. Sci. Afr. 2021, 12, e00789. [Google Scholar] [CrossRef]
- Inayat, M.; Sulaiman, S.A.; Kurnia, J.C. Catalytic co-gasification of coconut shells and oil palm fronds blends in the presence of cement, dolomite, and limestone: Parametric optimization via Box Behnken Design. J. Energy Inst. 2019, 92, 871–882. [Google Scholar] [CrossRef]
- Menon, S.D.; Sampath, K.; Kaarthik, S.S. Feasibility studies of coconut shells biomass for downdraft gasification. Mater. Today Proc. 2021, 44, 3133–3137. [Google Scholar] [CrossRef]
- Yahaya, A.Z.; Somalu, M.R.; Muchtar, A.; Sulaiman, S.A.; Daud, W.R.W. Effect of particle size and temperature on gasification performance of coconut and palm kernel shells in downdraft fixed-bed reactor. Energy 2019, 175, 931–940. [Google Scholar] [CrossRef]
- Nanda, S.; Isen, J.; Dalai, A.K.; Kozinski, J.A. Gasification of fruit wastes and agro-food residues in supercritical water. Energy Convers. Manag. 2016, 110, 296–306. [Google Scholar] [CrossRef]
- Wang, C.; Jin, H. Development of a Partitioning Kinetic Model of Biomass Gasification in Supercritical Water with a Fluidized Bed Reactor. Ind. Eng. Chem. Res. 2022, 61, 10058–10068. [Google Scholar] [CrossRef]
- Wang, C.; Li, L.; Shi, J.; Jin, H. Biochar production by coconut shell gasification in supercritical water and evolution of its porous structure. J. Anal. Appl. Pyrolysis 2021, 156, 105151. [Google Scholar] [CrossRef]
- Ciuta, S.; Tsiamis, D.; Castaldi, M.J. Gasification of Waste Materials: Technologies for Generating Energy, Gas, and Chemicals from Municipal Solid Waste, Biomass, Nonrecycled Plastics, Sludges, and Wet Solid Wastes; Academic Press: Cambridge, MA, USA, 2017. [Google Scholar]
- Ibrahim, A.; Akilli, H. Supercritical water gasification of wastewater sludge for hydrogen production. Int. J. Hydrog. Energy 2019, 44, 10328–10349. [Google Scholar] [CrossRef]
- Lee, C.S.; Conradie, A.V.; Lester, E. Review of supercritical water gasification with lignocellulosic real biomass as the feedstocks: Process parameters, biomass composition, catalyst development, reactor design and its challenges. Chem. Eng. J. 2021, 415, 128837. [Google Scholar] [CrossRef]
- Azadi, P.; Farnood, R. Review of heterogeneous catalysts for sub- and supercritical water gasification of biomass and wastes. Int. J. Hydrog. Energy 2011, 36, 9529–9541. [Google Scholar] [CrossRef]
- Richardson, Y.; Blin, J.; Volle, G.; Motuzas, J.; Julbe, A. In situ generation of Ni metal nanoparticles as catalyst for H2-rich syngas production from biomass gasification. Appl. Catal. A Gen. 2010, 382, 220–230. [Google Scholar] [CrossRef]
- Huang, J.; Zhu, C.; Lian, X.; Feng, H.; Sun, J.; Wang, L.; Jin, H. Catalytic supercritical water gasification of glucose with in-situ generated nickel nanoparticles for hydrogen production. Int. J. Hydrog. Energy 2019, 44, 21020–21029. [Google Scholar] [CrossRef]
- Kumar, A.; Reddy, S.N. In Situ Sub-and Supercritical Water Gasification of Nano-Nickel (Ni2+) Impregnated Biomass for H2 Production. Ind. Eng. Chem. Res. 2019, 58, 4780–4793. [Google Scholar] [CrossRef]
- Kumar, A.; Reddy, S.N. Subcritical and supercritical water in-situ gasification of metal (Ni/Ru/Fe) impregnated banana pseudo-stem for hydrogen rich fuel gas mixture. Int. J. Hydrog. Energy 2019, 45, 18348–18362. [Google Scholar] [CrossRef]
- Nanda, S.; Reddy, S.N.; Dalai, A.K.; Kozinski, J.A. Subcritical and supercritical water gasification of lignocellulosic biomass impregnated with nickel nanocatalyst for hydrogen production. Int. J. Hydrog. Energy 2016, 41, 4907–4921. [Google Scholar] [CrossRef]
- Bakari, R.; Kivevele, T.; Huang, X.; Jande, Y.A.C. Sub- and Supercritical Water Gasification of Rice Husk: Parametric Optimization Using the I-Optimality Criterion. ACS Omega 2021, 6, 12480–12499. [Google Scholar] [CrossRef]
- Chen, J.; Fan, Y.; Zhao, X.; Jiaqiang, E.; Xu, W.; Zhang, F.; Liao, G.; Leng, E.; Liu, S. Experimental investigation on gasification characteristic of food waste using supercritical water for combustible gas production: Exploring the way to complete gasification. Fuel 2020, 263, 116735. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef]
- Okolie, J.A. Supercritical Water Gasification of Lignocellulosic Biomass Materials for Hydrogen Production. Ph.D. Thesis, University of Saskatchewan, Saskatoon, SK, Canada, 2021. [Google Scholar]
- Khanal, M.; Rai, D.; Khanal, R.; Bhattarai, A. Determination of point zero charge (PZC) of homemade charcoals of shorea robusta (Sakhuwa) and pinus roxburghii (Salla). Int. J. Eng. Res. Technol. (IJERT) 2020, 9, 2278-0181. [Google Scholar]
- Borges, A.; Onwudili, J.; Andrade, H.; Alves, C.; Ingram, A.; de Melo, S.V.; Torres, E. Catalytic supercritical water gasification of eucalyptus wood chips in a batch reactor. Fuel 2019, 255, 115804. [Google Scholar] [CrossRef]
- Cao, W.; Guo, L.; Yan, X.; Zhang, D.; Yao, X. Assessment of sugarcane bagasse gasification in supercritical water for hydrogen production. Int. J. Hydrog. Energy 2018, 43, 13711–13719. [Google Scholar] [CrossRef]
- Louw, J. Supercritical Water Gasification of Wood-Related Products: A Thermodynamic and Experimental Study. Ph.D. Thesis, Stellenbosch University, Stellenbosch, South Africa, 2016. [Google Scholar]
- Kleinübing, S.J.; Vieira, R.S.; Beppu, M.M.; Guibal, E.; Da Silva, M.G.C. Characterization and evaluation of copper and nickel biosorption on acidic algae Sargassum Filipendula. Mater. Res. 2010, 13, 541–550. [Google Scholar] [CrossRef]
- Kuncoro, E.P.; Isnadina, D.R.M.; Darmokoesoemo, H.; Fauziah, O.R.; Kusuma, H.S. Characterization, kinetic, and isotherm data for adsorption of Pb2+ from aqueous solution by adsorbent from mixture of bagasse-bentonite. Data Brief 2018, 16, 622–629. [Google Scholar] [CrossRef]
- da Silva, J.C.G.; Alves, J.L.F.; de Araujo Galdino, W.V.; de Sena, R.F.; Andersen, S.L.F. Pyrolysis kinetics and physicochemical characteristics of skin, husk, and shell from green coconut wastes. Energy Ecol. Environ. 2019, 4, 125–132. [Google Scholar] [CrossRef]
- García, R.; Pizarro, C.; Lavín, A.G.; Bueno, J.L. Characterization of Spanish biomass wastes for energy use. Bioresour. Technol. 2012, 103, 249–258. [Google Scholar] [CrossRef]
- Sturion, J.A.; Pereira, J.C.D.; Chemin, M. Qualidade da madeira de Eucalyptus viminalis para fins energéticos em função do espaçamento e idade de corte. Bol. De Pesqui. Florestal. 1998, 16, 55–59. [Google Scholar]
- Said, M.; Cassayre, L.; Dirion, J.-L.; Joulia, X.; Nzihou, A. Effect of Nickel Impregnation on Wood Gasification Mechanism. Waste Biomass-Valoriz. 2017, 8, 2843–2852. [Google Scholar] [CrossRef]
- Singha, R.K.; Yadav, A.; Agrawal, A.; Shukla, A.; Adak, S.; Sasaki, T.; Bal, R. Synthesis of highly coke resistant Ni nanoparticles supported MgO/ZnO catalyst for reforming of methane with carbon dioxide. Appl. Catal. B Environ. 2016, 191, 165–178. [Google Scholar] [CrossRef]
- Devi, T.G.; Kannan, M.P. X-ray Diffraction (XRD) Studies on the Chemical States of Some Metal Species in Cellulosic Chars and the Ellingham Diagrams. Energy Fuels 2007, 21, 596–601. [Google Scholar] [CrossRef]
- Koltypin, Y.; Fernandez, A.; Rojas, T.C.; Campora, J.; Palma, P.; Prozorov, R.; Gedanken, A. Encapsulation of nickel nanoparticles in carbon obtained by the sonochemical decomposition of Ni(C8H12)2. Chem. Mater. 1999, 11, 1331–1335. [Google Scholar] [CrossRef]
- Rout, T.; Pradhan, D.; Singh, R.; Kumari, N. Exhaustive study of products obtained from coconut shell pyrolysis. J. Environ. Chem. Eng. 2016, 4, 3696–3705. [Google Scholar] [CrossRef]
- Sarki, J.; Hassan, S.; Aigbodion, V.; Oghenevweta, J. Potential of using coconut shell particle fillers in eco-composite materials. J. Alloys Compd. 2011, 509, 2381–2385. [Google Scholar] [CrossRef]
- Salavati-Niasari, M.; Mir, N.; Davar, F. A novel precursor in preparation and characterization of nickel oxide nanoparticles via thermal decomposition approach. J. Alloys Compd. 2010, 493, 163–168. [Google Scholar] [CrossRef]
- Ingham, B.; Toney, M.F. X-ray diffraction for characterizing metallic films. In Metallic Films for Electronic, Optical and Magnetic Applications: Structure, Processing and Properties; Elsevier: Amsterdam, The Netherlands, 2014; pp. 3–38. [Google Scholar]
- Saleh, T.A. Polymer Hybrid Materials and Nanocomposites: Fundamentals and Applications; William Andrew: New York, NY, USA, 2021. [Google Scholar]
- Saleh, T.A.; Gupta, V.K. Nanomaterial and Polymer Membranes: Synthesis, Characterization, and Applications; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Chan, F.L.; Tanksale, A. Review of recent developments in Ni-based catalysts for biomass gasification. Renew. Sustain. Energy Rev. 2014, 38, 428–438. [Google Scholar] [CrossRef]
- Lazim, Z.M.; Hadibarata, T.; Puteh, M.H.; Yusop, Z. Adsorption Characteristics of Bisphenol A onto Low-Cost Modified Phyto-Waste Material in Aqueous Solution. Water Air Soil Pollut. 2015, 226, 34. [Google Scholar] [CrossRef]
- Liyanage, C.D.; Pieris, M. A Physico-Chemical Analysis of Coconut Shell Powder. Procedia Chem. 2015, 16, 222–228. [Google Scholar] [CrossRef]
- Orooji, Y.; Ghanbari, M.; Amiri, O.; Salavati-Niasari, M. Facile fabrication of silver iodide/graphitic carbon nitride nanocomposites by notable photo-catalytic performance through sunlight and antimicrobial activity. J. Hazard. Mater. 2020, 389, 122079. [Google Scholar] [CrossRef] [PubMed]
- Okolie, J.; Rana, R.; Nanda, S.; Dalai, A.K.; Kozinski, J.A. Supercritical water gasification of biomass: A state-of-the-art review of process parameters, reaction mechanisms and catalysis. Sustain. Energy Fuels 2019, 3, 578–598. [Google Scholar] [CrossRef]
- Amrullah, A.; Matsumura, Y. Supercritical water gasification of sewage sludge in continuous reactor. Bioresour. Technol. 2018, 249, 276–283. [Google Scholar] [CrossRef]
- Samiee-Zafarghandi, R.; Karimi-Sabet, J.; Abdoli, M.A.; Karbassi, A. Supercritical water gasification of microalga Chlorella PTCC 6010 for hydrogen production: Box-Behnken optimization and evaluating catalytic effect of MnO2/SiO2 and NiO/SiO2. Renew. Energy 2018, 126, 189–201. [Google Scholar] [CrossRef]
- Elif, D.; Nezihe, A. Hydrogen production by supercritical water gasification of fruit pulp in the presence of Ru/C. Int. J. Hydrog. Energy 2016, 41, 8073–8083. [Google Scholar] [CrossRef]
- Demirel, E.; Erkey, C.; Ayas, N. Supercritical Water Gasification of Fruit Pulp for Hydrogen Production: Effect of Reaction Parameters. J. Supercrit. Fluids 2021, 177, 105329. [Google Scholar] [CrossRef]
- Wang, C.; Li, L.; Chen, Y.; Ge, Z.; Jin, H. Supercritical water gasification of wheat straw: Composition of reaction products and kinetic study. Energy 2021, 227, 120449. [Google Scholar] [CrossRef]
- Baig, M.; Leeke, G.; Hammond, P.; Santos, R. Modelling the extraction of soil contaminants with supercritical carbon dioxide. Environ. Pollut. 2011, 159, 1802–1809. [Google Scholar] [CrossRef]
- Nanda, S.; Gong, M.; Hunter, H.N.; Dalai, A.K.; Gökalp, I.; Kozinski, J.A. An assessment of pinecone gasification in subcritical, near-critical and supercritical water. Fuel Process. Technol. 2017, 168, 84–96. [Google Scholar] [CrossRef]
- Su, H.; Kanchanatip, E.; Wang, D.; Zhang, H.; Antoni; Mubeen, I.; Huang, Z.; Yan, M. Catalytic gasification of food waste in supercritical water over La promoted Ni/Al2O3 catalysts for enhancing H2 production. Int. J. Hydrog. Energy 2020, 45, 553–564. [Google Scholar] [CrossRef]
- Williams, P.T.; Onwudili, J. Composition of Products from the Supercritical Water Gasification of Glucose: A Model Biomass Compound. Ind. Eng. Chem. Res. 2005, 44, 8739–8749. [Google Scholar] [CrossRef]
- Borges, A.C.P. Gaseificação de Cavaco de Eucalipto em Água Supercrítica na Presença do Catalisador NiFe2O4. 2021. Available online: https://repositorio.ufba.br/handle/ri/32553 (accessed on 1 April 2023).
- Nanda, S.; Reddy, S.N.; Hunter, H.N.; Vo, D.-V.N.; Kozinski, J.A.; Gökalp, I. Catalytic subcritical and supercritical water gasification as a resource recovery approach from waste tires for hydrogen-rich syngas production. J. Supercrit. Fluids 2019, 154, 104627. [Google Scholar] [CrossRef]
- Adar, E.; Ince, M.; Bilgili, M.S. Supercritical water gasification of sewage sludge by continuous flow tubular reactor: A pilot scale study. Chem. Eng. J. 2020, 391, 123499. [Google Scholar] [CrossRef]
- Wang, C.; Jin, H.; Feng, H.; Wei, W.; Cao, C.; Cao, W. Study on gasification mechanism of biomass waste in supercritical water based on product distribution. Int. J. Hydrog. Energy 2020, 45, 28051–28061. [Google Scholar] [CrossRef]
- Ruya, P.M.; Purwadi, R.; Lim, S.S. Supercritical water gasification of sewage sludge for power generation–thermodynamic study on auto-thermal operation using Aspen Plus. Energy Convers. Manag. 2020, 206, 112458. [Google Scholar] [CrossRef]
- Yildirir, E.; Ballice, L. Supercritical water gasification of wet sludge from biological treatment of textile and leather industrial wastewater. J. Supercrit. Fluids 2019, 146, 100–106. [Google Scholar] [CrossRef]
- Nanda, S.; Reddy, S.N.; Vo, D.-V.N.; Sahoo, B.N.; Kozinski, J.A. Catalytic gasification of wheat straw in hot compressed (subcritical and supercritical) water for hydrogen production. Energy Sci. Eng. 2018, 6, 448–459. [Google Scholar] [CrossRef]
- Basu, P. Biomass Gasification and Pyrolysis: Practical Design and Theory; Academic Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Alam, M.A.; Hamdan, H.Y.; Azeem, M.; Hussain, P.B.; bin Salit, M.S.; Khan, R.; Arif, S.; Ansari, A.H. Modelling and optimisation of hardness behaviour of sintered Al/SiC composites using RSM and ANN: A comparative study. J. Mater. Res. Technol. 2020, 9, 14036–14050. [Google Scholar] [CrossRef]
- Abdpour, S.; Santos, R.M. Recent advances in heterogeneous catalysis for supercritical water oxidation/gasification processes: Insight into catalyst development. Process. Saf. Environ. Prot. 2021, 149, 169–184. [Google Scholar] [CrossRef]
- Zhang, J.; Dasgupta, A.; Chen, Z.; Xu, D.; Savage, P.E.; Guo, Y. Supercritical water gasification of phenol over Ni-Ru bimetallic catalysts. Water Res. 2019, 152, 12–20. [Google Scholar] [CrossRef]
- Resende, F.L.P.; Savage, P.E. Effect of Metals on Supercritical Water Gasification of Cellulose and Lignin. Ind. Eng. Chem. Res. 2010, 49, 2694–2700. [Google Scholar] [CrossRef]
- Pairojpiriyakul, T.; Croiset, E.; Kiatkittipong, K.; Kiatkittipong, W.; Arpornwichanop, A.; Assabumrungrat, S. Catalytic reforming of glycerol in supercritical water with nickel-based catalysts. Int. J. Hydrog. Energy 2014, 39, 14739–14750. [Google Scholar] [CrossRef]








| Coded Values | Temperature (°C) | Residence Time (min) | Biomass Loading (wt%) |
|---|---|---|---|
| −1 | 400 | 20 | 20 |
| 0 | 450 | 40 | 25 |
| 1 | 500 | 60 | 30 |
| Proximate Analysis (wt%) | |
|---|---|
| Moisture | 3.2 |
| Volatile matter | 77.5 |
| Ash | 2.9 |
| Fixed carbon | 19.6 |
| Parameter | Control (wt%) | Impregnated (wt%) |
|---|---|---|
| C | 49.0 | 45.8 |
| H | 6.2 | 5.5 |
| N | 0.3 | 0.8 |
| O | 44.5 | 46.4 |
| S | nd | nd |
| Response Variables (%) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Run | A-Temperature (°C) | B-Residence Time (min) | C-Biomass Loading (wt%) | H2 | CO | CO2 | CH4 | GE |
| 1 | 450 | 20 | 20 | 7.1 | 3.9 | 58.6 | 30.4 | 30.3 |
| 2 | 500 | 60 | 25 | 7.3 | 0.9 | 48.7 | 43.2 | 40.1 |
| 3 | 450 | 60 | 20 | 6.5 | 1.3 | 64.2 | 28.1 | 40.4 |
| 4 | 500 | 40 | 20 | 8.3 | 0.5 | 48.3 | 43.0 | 52.0 |
| 5 | 450 | 40 | 25 | 4.2 | 4.1 | 67.0 | 24.8 | 24.0 |
| 6 | 450 | 40 | 25 | 1.7 | 3.9 | 62.8 | 31.7 | 21.3 |
| 7 | 400 | 20 | 25 | 4.5 | 9.0 | 75.8 | 10.7 | 18.8 |
| 8 | 500 | 20 | 25 | 9.5 | 3.2 | 40.7 | 46.8 | 33.9 |
| 9 | 450 | 40 | 25 | 5.8 | 4.6 | 64.5 | 25.1 | 32.0 |
| 10 | 450 | 20 | 30 | 1.8 | 5.4 | 73.7 | 19.2 | 27.0 |
| 11 | 450 | 60 | 30 | 3.8 | 1.1 | 69.1 | 26.0 | 37.8 |
| 12 | 500 | 40 | 30 | 5.8 | 0.3 | 38.5 | 55.5 | 48.8 |
| 13 | 400 | 40 | 30 | 0.2 | 4.8 | 78.8 | 16.2 | 25.5 |
| 14 | 400 | 40 | 20 | 5.5 | 5.5 | 74.1 | 14.9 | 26.5 |
| 15 | 400 | 60 | 25 | 2.3 | 5.8 | 74.9 | 13.8 | 25.9 |
| Source | H2 | CO | CO2 | CH4 | GE | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SS | F | p | SS | F | p | SS | F | p | SS | F | p | SS | F | p | |
| Model | 74.83 | 11.40 | 0.0011 | 70.88 | 19.65 | 0.0001 | 2074.27 | 23.68 | <0.0001 | 2208.54 | 28.89 | <0.0001 | 919.51 | 7.98 | 0.0042 |
| Significant | Significant | Significant | Significant | Significant | |||||||||||
| A | 42.23 | 19.30 | 0.0011 | 51.56 | 42.89 | <0.0001 | 2037.77 | 69.80 | <0.0001 | 2206.47 | 86.59 | <0.0001 | 760.89 | 19.81 | 0.0010 |
| B | 31.44 | 14.37 | 0.0030 | 0.0084 | 0.007 | 0.9347 | 28.05 | 0.96 | 0.6469 | 0.053 | 0.002 | 0.9645 | 12.68 | 0.33 | 0.5775 |
| C | 1.16 | 0.53 | 0.4827 | 19.31 | 16.06 | 0.0021 | 8.45 | 0.29 | 0.6089 | 2.01 | 0.079 | 0.7840 | 145.95 | 3.80 | 0.0772 |
| Lack of fit | 15.28 | 0.39 | 0.8705 | 14.09 | 12.29 | 0.0775 | 312.26 | 7.81 | 0.1186 | 250.27 | 1.84 | 0.3996 | 360.51 | 1.29 | 0.5097 |
| Not significant | Not significant | Not significant | Not significant | Not significant | |||||||||||
| Response Variables (%) | Final Equations in Terms of Coded Factors | R2 | R2adj | R2pred |
|---|---|---|---|---|
| H2 | 4.93 + 2.30A − 1.98B − 0.38C | 0.7566 | 0.6902 | 0.6004 |
| CO | 3.61–2.54A + 0.032B − 1.55C | 0.8428 | 0.7999 | 0.6784 |
| CO2 | 62.62–15.96A + 1.87B + 1.03C | 0.8659 | 0.8294 | 0.7221 |
| CH4 | 28.63 + 16.61A + 0.081B + 0.50C | 0.8874 | 0.8567 | 0.7733 |
| GE | 32.29 + 9.75A − 1.26B + 4.27C | 0.6852 | 0.5994 | 0.4653 |
| Scenario 1 | Scenario 2 | Lower Limit | Upper Limit | |
|---|---|---|---|---|
| Name | Goal | Goal | ||
| A—Temperature (°C) | maximize | in range | 400 | 500 |
| B—Biomass loading (wt%) | minimize | minimize | 20 | 30 |
| C—Time (min) | in range | minimize | 20 | 60 |
| H2 (mol%) | maximize | maximize | 0.2 | 9.5 |
| GE (%) | maximize | maximize | 18.8 | 52.0 |
| Response Variables | ||||||||
|---|---|---|---|---|---|---|---|---|
| Run | A—Temperature (°C) | B—Residence Time (min) | C—Biomass Loading (wt%) | H2 | CO | CO2 | CH4 | GE |
| 1 | 500 | 40 | 20 | 8.6 | 3.0 | 43.7 | 44.8 | 54.8 |
| 2 | 400 | 40 | 20 | 4.5 | 7.1 | 71.8 | 16.5 | 41.4 |
| 3 | 400 | 60 | 25 | 3.3 | 9.7 | 59.9 | 27.0 | 45.4 |
| 4 | 450 | 40 | 25 | 7.8 | 12.6 | 52.8 | 26.9 | 29.4 |
| 5 | 450 | 20 | 30 | 4.6 | 12.4 | 55.8 | 27.2 | 31.5 |
| 6 | 450 | 20 | 20 | 17.2 | 15.7 | 48.1 | 18.9 | 33.7 |
| 7 | 450 | 60 | 30 | 3.9 | 6.0 | 49.1 | 40.9 | 42.8 |
| 8 | 500 | 20 | 25 | 13.0 | 7.7 | 42.8 | 36.7 | 49.3 |
| 9 | 400 | 20 | 25 | 4.6 | 14.5 | 56.1 | 24.8 | 29.4 |
| 10 | 450 | 40 | 25 | 4.2 | 13.0 | 54.8 | 27.9 | 48.0 |
| 11 | 450 | 60 | 20 | 7.6 | 12.0 | 42.0 | 38.4 | 44.5 |
| 12 | 400 | 40 | 30 | 5.6 | 11.9 | 59.5 | 22.9 | 40.5 |
| 13 | 500 | 40 | 30 | 5.8 | 8.7 | 48.6 | 36.9 | 51.2 |
| 14 | 500 | 60 | 25 | 8.0 | 1.02 | 48.4 | 42.6 | 58.6 |
| 15 | 450 | 40 | 25 | 6.9 | 12.5 | 53.2 | 27.4 | 37.4 |
| Variables | Control Sample | Impregnated Sample | ||||||
|---|---|---|---|---|---|---|---|---|
| Biomass Loading (%) | Temperature (°C) | Residence Time (min) | Solid (wt%) | Liquid (wt%) | Gas (wt%) | Solid (wt%) | Liquid (wt%) | Gas (wt%) |
| 20 | 400 | 40 | 12.4 | 61.1 | 26.5 | 11.9 | 46.7 | 41.4 |
| 500 | 40 | 11.9 | 36.1 | 52.0 | 9.2 | 36.0 | 54.7 | |
| 450 | 20 | 10.1 | 59.6 | 30.3 | 9.1 | 57.2 | 33.7 | |
| 450 | 60 | 9.8 | 49.9 | 40.4 | 9.6 | 45.9 | 44.5 | |
| 25 | 450 | 40 | 14.7 | 61.3 | 24.0 | 12.0 | 58.6 | 29.4 |
| 450 | 40 | 12.5 | 66.1 | 21.3 | 11.5 | 40.5 | 48.0 | |
| 450 | 40 | 9.9 | 58.1 | 32.0 | 9.6 | 53.0 | 37.4 | |
| 400 | 20 | 15.8 | 65.4 | 18.8 | 12.5 | 58.1 | 29.4 | |
| 400 | 60 | 12.5 | 61.6 | 25.9 | 7.5 | 47.1 | 45.4 | |
| 500 | 20 | 15.7 | 50.4 | 33.9 | 13.3 | 37.4 | 49.3 | |
| 500 | 60 | 12.3 | 47.6 | 40.1 | 11.1 | 30.3 | 58.6 | |
| 30 | 400 | 40 | 13.7 | 60.8 | 25.5 | 13.3 | 46.2 | 40.5 |
| 500 | 40 | 13.5 | 37.7 | 48.8 | 13.0 | 35.2 | 51.8 | |
| 450 | 20 | 13.9 | 59.1 | 26.9 | 12.6 | 55.9 | 31.5 | |
| 450 | 60 | 14.9 | 47.2 | 37.9 | 12.1 | 45.1 | 42.8 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcelino, M.M.; Leeke, G.A.; Jiang, G.; Onwudili, J.A.; Alves, C.T.; Santana, D.M.d.; Torres, F.A.; Torres, E.A.; Vieira de Melo, S.A.B. Supercritical Water Gasification of Coconut Shell Impregnated with a Nickel Nanocatalyst: Box–Behnken Design and Process Evaluation. Energies 2023, 16, 3563. https://doi.org/10.3390/en16083563
Marcelino MM, Leeke GA, Jiang G, Onwudili JA, Alves CT, Santana DMd, Torres FA, Torres EA, Vieira de Melo SAB. Supercritical Water Gasification of Coconut Shell Impregnated with a Nickel Nanocatalyst: Box–Behnken Design and Process Evaluation. Energies. 2023; 16(8):3563. https://doi.org/10.3390/en16083563
Chicago/Turabian StyleMarcelino, Marcela M., Gary A. Leeke, Guozhan Jiang, Jude A. Onwudili, Carine T. Alves, Delano M. de Santana, Felipe A. Torres, Ednildo A. Torres, and Silvio A. B. Vieira de Melo. 2023. "Supercritical Water Gasification of Coconut Shell Impregnated with a Nickel Nanocatalyst: Box–Behnken Design and Process Evaluation" Energies 16, no. 8: 3563. https://doi.org/10.3390/en16083563
APA StyleMarcelino, M. M., Leeke, G. A., Jiang, G., Onwudili, J. A., Alves, C. T., Santana, D. M. d., Torres, F. A., Torres, E. A., & Vieira de Melo, S. A. B. (2023). Supercritical Water Gasification of Coconut Shell Impregnated with a Nickel Nanocatalyst: Box–Behnken Design and Process Evaluation. Energies, 16(8), 3563. https://doi.org/10.3390/en16083563










