Transesterification of Algae Oil and Little Amount of Waste Cooking Oil Blend at Low Temperature in the Presence of NaOH
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Method
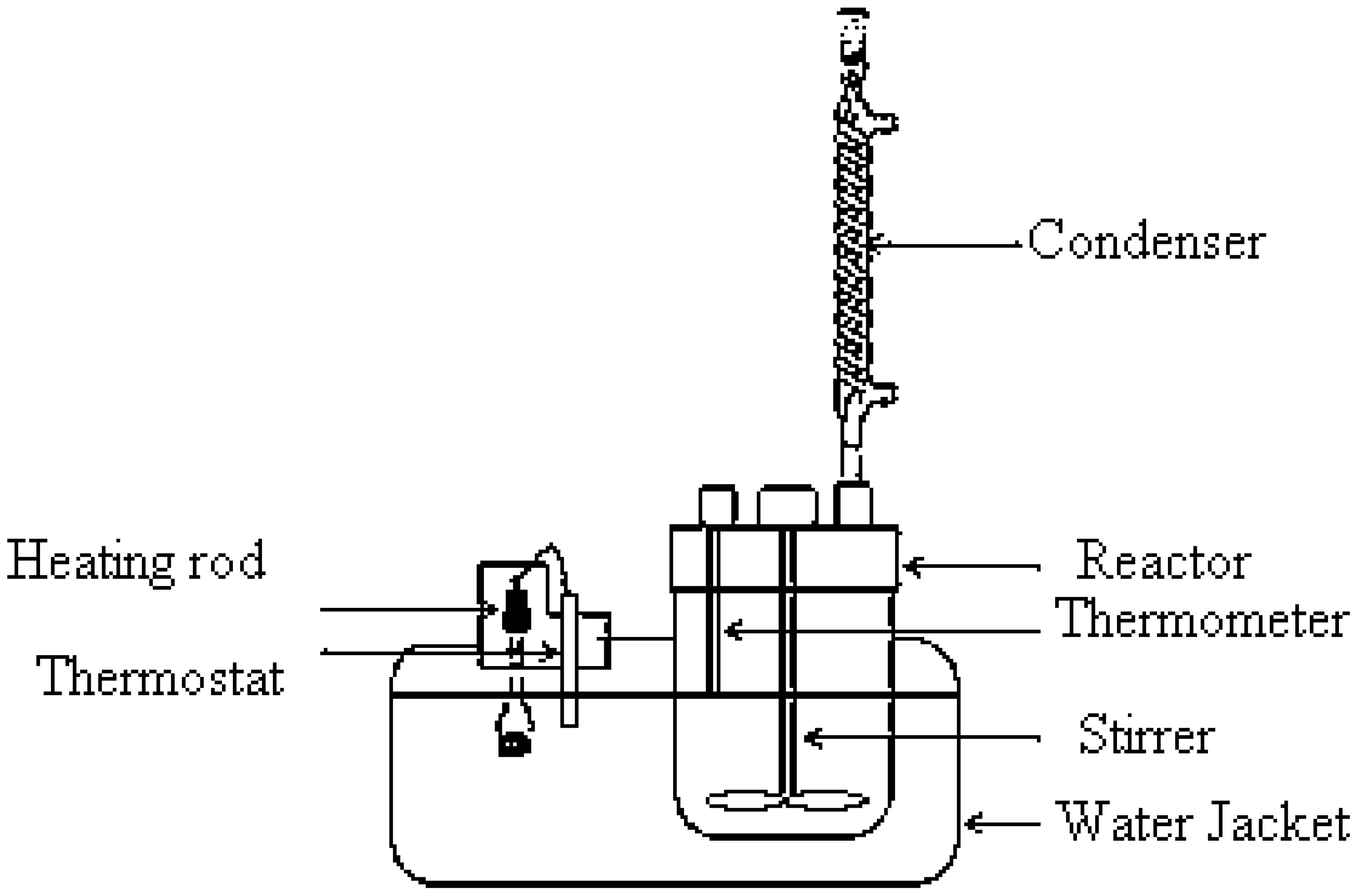
2.3. Experimental Procedure
Design of Experiments
2.4. Transesterification
2.5. Statistical Analysis
3. Results and Discussions
3.1. Transesterification Process
3.2. ANOVA
3.3. Effects of Process Variables on Yield
3.4. Effect of Reaction Parameters on Yield %
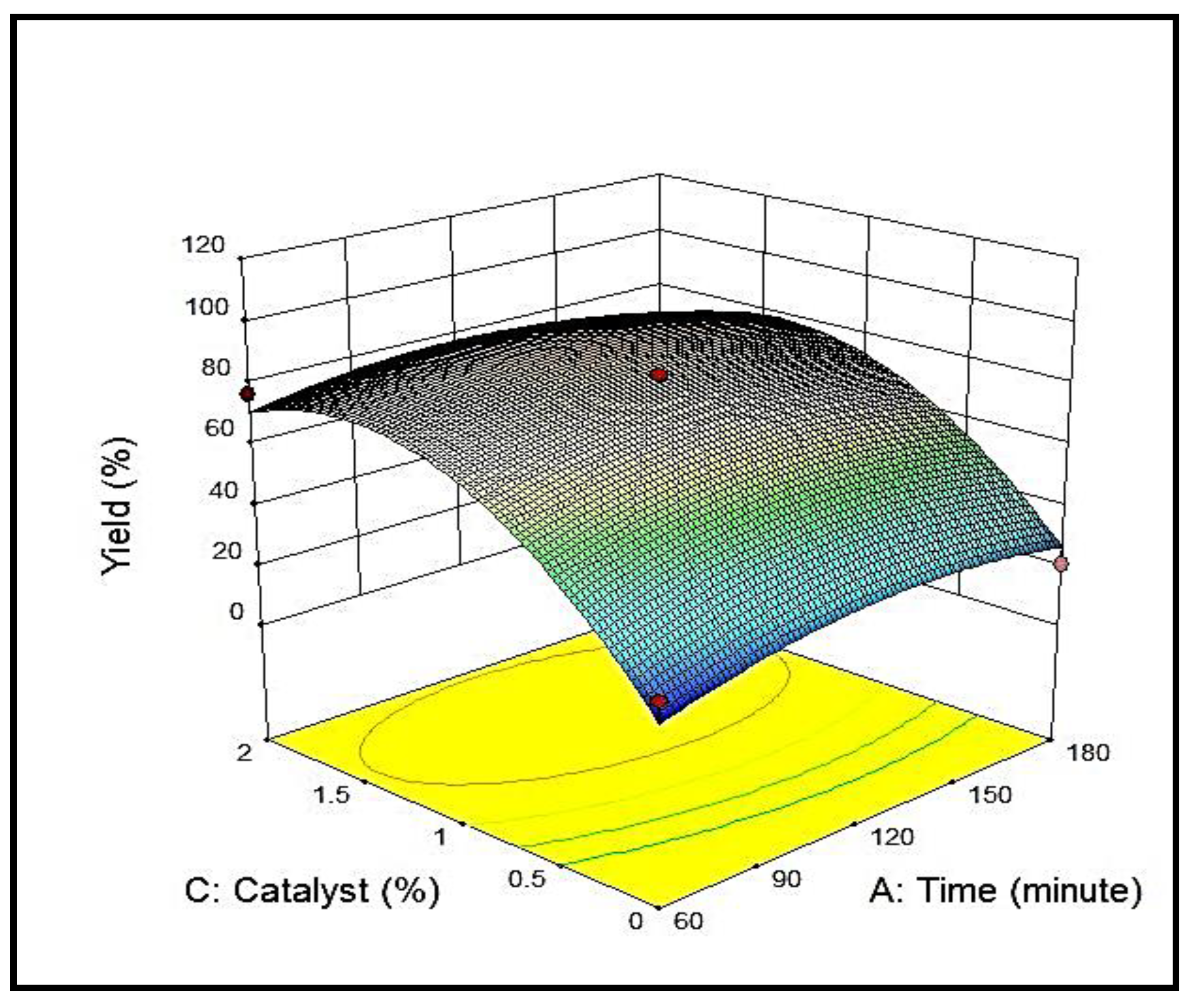
3.4.1. Effect of Reaction Time on Yield %
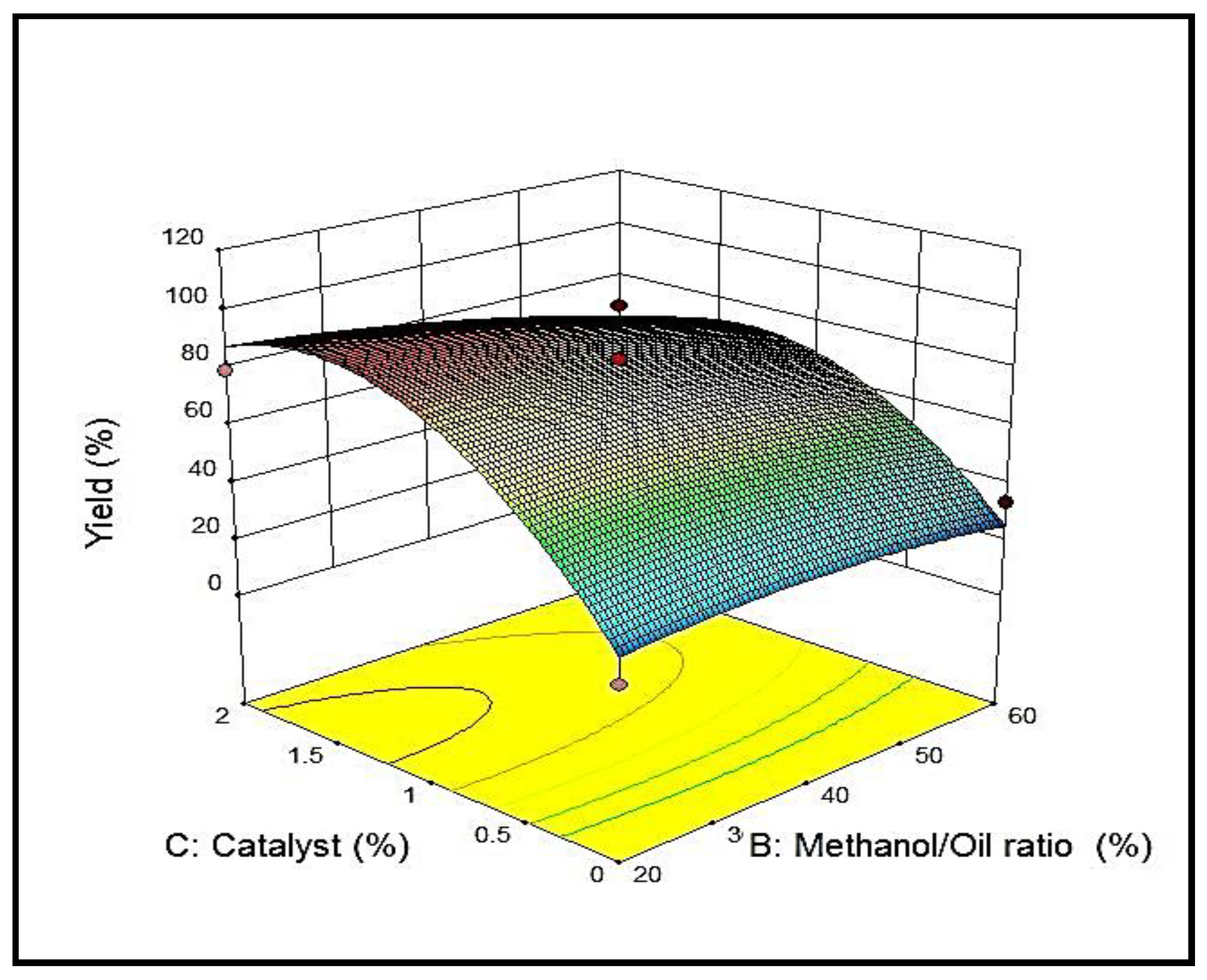
3.4.2. Effect of Methanol/Oil Ratio on Yield %
3.4.3. Effect of Catalyst Concentration on Yield %
4. Optimization of Response Yield
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Saini, M.; Sharma, A.; Singh, V.P.; Dwivedi, G.; Jain, S. Solar Thermal Receivers—A Review. Adv. Mater. Manuf. Energy 2022, 2, 310–325. [Google Scholar] [CrossRef]
- Singh, V.P.; Jain, S.; Karn, A.; Kumar, A.; Dwivedi, G.; Meena, C.S.; Dutt, N.; Ghosh, A. Recent Developments and Advancements in Solar Air Heaters: A Detailed Review. Sustainability 2022, 14, 2149. [Google Scholar] [CrossRef]
- Lo, S.L.Y.; How, B.S.; Leong, W.D.; Teng, S.Y.; Rhamdhani, M.A.; Sunarso, J. Techno-economic analysis for biomass supply chain: A state-of-the-art review. Renew. Sustain. Energy Rev. 2021, 135, 110164. [Google Scholar] [CrossRef]
- Verma, D.; Fortunati, E.; Jain, S.; Zhang, X. Biomass, Biopolymer-Based Materials, and Bioenergy: Construction, Biomedical, and Other Industrial Applications, 1st ed.; Elsevier Science: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Singh, V.P.; Jain, S.; Karn, A.; Dwivedi, G.; Alam, T.; Kumar, A. Experimental Assessment of Variation in Open Area Ratio on Thermohydraulic Performance of Parallel Flow Solar Air Heater. Arab. J. Sci. Eng. 2022, 41, 1–17. [Google Scholar] [CrossRef]
- Ambaye, T.G.; Vaccari, M.; Bonilla-Petriciolet, A.; Prasad, S.; van Hullebusch, E.D.; Rtimi, S. Emerging technologies for biofuel production: A critical review on recent progress, challenges and perspectives. J. Environ. Manag. 2021, 290, 112627. [Google Scholar] [CrossRef]
- Jain, S.; Sharma, M. Engine performance and emission analysis using oxidatively stabilized Jatropha curcas biodiesel. Fuel 2013, 106, 152–156. [Google Scholar] [CrossRef]
- Singh, D.; Sharma, D.; Soni, S.; Inda, C.S.; Sharma, S.; Sharma, P.K.; Jhalani, A. A comprehensive review of biodiesel production from waste cooking oil and its use as fuel in compression ignition engines: 3rd generation cleaner feedstock. J. Clean. Prod. 2021, 307, 127299. [Google Scholar] [CrossRef]
- Agarwal, A.K.; Gupta, J.G.; Dhar, A. Potential and challenges for large-scale application of biodiesel in automotive sector. Prog. Energy Combust. Sci. 2017, 61, 113–149. [Google Scholar] [CrossRef]
- Abomohra, A.E.-F.; Elsayed, M.; Esakkimuthu, S.; El-Sheekh, M.; Hanelt, D. Potential of fat, oil and grease (FOG) for biodiesel production: A critical review on the recent progress and future perspectives. Prog. Energy Combust. Sci. 2020, 81, 100868. [Google Scholar] [CrossRef]
- Garg, A.; Dwivedi, G.; Baredar, P.; Jain, S. Current Status, Synthesis, and Characterization of Biodiesel. In Biodiesel Technology and Applications; Wiley: Hoboken, NJ, USA, 2021. [Google Scholar]
- Jain, S.; Sharma, M.P. Oxidation, Thermal, and Storage Stability Studies of Jatropha Curcas Biodiesel. ISRN Renew. Energy 2012, 2012, 1–15. [Google Scholar] [CrossRef]
- Singh, V.P.; Jain, S.; Karn, A.; Dwivedi, G.; Kumar, A.; Mishra, S.; Sharma, N.K.; Bajaj, M.; Zawbaa, H.M.; Kamel, S. Heat transfer and friction factor correlations development for double pass solar air heater artificially roughened with perforated multi-V ribs. Case Stud. Therm. Eng. 2022, 39, 102461. [Google Scholar] [CrossRef]
- Goyal, P.; Sharma, M.P.; Jain, S. Optimization of transesterification of Jatropha curcas Oil to Biodiesel using Response Surface Methodology and its Adulteration with Kerosene. J. Mater. Environ. Sci. 2013, 4, 277–284, 2013. [Google Scholar]
- Goh, B.H.H.; Chong, C.T.; Ge, Y.; Ong, H.C.; Ng, J.-H.; Tian, B.; Ashokkumar, V.; Lim, S.; Seljak, T.; Józsa, V. Progress in utilisation of waste cooking oil for sustainable biodiesel and biojet fuel production. Energy Convers. Manag. 2020, 223, 113296. [Google Scholar] [CrossRef]
- Borges, M.; Díaz, L. Recent developments on heterogeneous catalysts for biodiesel production by oil esterification and transesterification reactions: A review. Renew. Sustain. Energy Rev. 2012, 16, 2839–2849. [Google Scholar] [CrossRef]
- Narula, V.; Thakur, A.; Uniyal, A.; Kalra, S.; Jain, S. Process parameter optimization of low temperature transesterification of algae-Jatropha Curcas oil blend. Energy 2017, 119, 983–988. [Google Scholar] [CrossRef]
- Mohadesi, M.; Aghel, B.; Maleki, M.; Ansari, A. The use of KOH/Clinoptilolite catalyst in pilot of microreactor for biodiesel production from waste cooking oil. Fuel 2020, 263, 116659. [Google Scholar] [CrossRef]
- Hsiao, M.-C.; Kuo, J.-Y.; Hsieh, S.-A.; Hsieh, P.-H.; Hou, S.-S. Optimized conversion of waste cooking oil to biodiesel using modified calcium oxide as catalyst via a microwave heating system. Fuel 2020, 266, 117114. [Google Scholar] [CrossRef]
- Aghel, B.; Mohadesi, M.; Ansari, A.; Maleki, M. Pilot-scale production of biodiesel from waste cooking oil using kettle limescale as a heterogeneous catalyst. Renew. Energy 2019, 142, 207–214. [Google Scholar] [CrossRef]
- Degfie, T.A.; Mamo, T.T.; Mekonnen, Y.S. Optimized Biodiesel Production from Waste Cooking Oil (WCO) using Calcium Oxide (CaO) Nano-catalyst. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef]
- Komintarachat, C.; Chuepeng, S. Catalytic enhancement of calcium oxide from green mussel shell by potassium chloride impregnation for waste cooking oil-based biodiesel production. Bioresour. Technol. Rep. 2020, 12, 100589. [Google Scholar] [CrossRef]
- Mardhiah, H.H.; Ong, H.C.; Masjuki, H.; Lim, S.; Lee, H. A review on latest developments and future prospects of heterogeneous catalyst in biodiesel production from non-edible oils. Renew. Sustain. Energy Rev. 2017, 67, 1225–1236. [Google Scholar] [CrossRef]
- Singh, B.; Jain, S.; Gangil, B. Effectiveness of Homogeneous and Heterogeneous Catalyst on Biodiesel Yield: A Review. Adv. Clean Energy Technol. 2021, 375–385. [Google Scholar] [CrossRef]
- Mohadesi, M.; Aghel, B.; Maleki, M.; Ansari, A. Production of biodiesel from waste cooking oil using a homogeneous catalyst: Study of semi-industrial pilot of microreactor. Renew. Energy 2019, 136, 677–682. [Google Scholar] [CrossRef]
- Shadidi, B.; Najafi, G.; Zolfigol, M.A. A Review of the Existing Potentials in Biodiesel Production in Iran. Sustainability 2022, 14, 3284. [Google Scholar] [CrossRef]
- Verma, P.; Sharma, M. Review of process parameters for biodiesel production from different feedstocks. Renew. Sustain. Energy Rev. 2016, 62, 1063–1071. [Google Scholar] [CrossRef]
- Abdullah, S.H.Y.S.; Hanapi, N.H.M.; Azid, A.; Umar, R.; Juahir, H.; Khatoon, H.; Endut, A. A review of biomass-derived heterogeneous catalyst for a sustainable biodiesel production. Renew. Sustain. Energy Rev. 2017, 70, 1040–1051. [Google Scholar] [CrossRef]
- Jain, S.; Sharma, M.; Rajvanshi, S. Acid base catalyzed transesterification kinetics of waste cooking oil. Fuel Process. Technol. 2011, 92, 32–38. [Google Scholar] [CrossRef]
- Garg, A.; Jain, S. Process parameter optimization of biodiesel production from algal oil by response surface methodology and artificial neural networks. Fuel 2020, 277, 118254. [Google Scholar] [CrossRef]
- Zhao, S.; Niu, S.; Yu, H.; Ning, Y.; Zhang, X.; Li, X.; Zhang, Y.; Lu, C.; Han, K. Experimental investigation on biodiesel production through transesterification promoted by the La-dolomite catalyst. Fuel 2019, 257, 116092. [Google Scholar] [CrossRef]
- Supraja, K.V.; Behera, B.; Paramasivan, B. Optimization of process variables on two-step microwave-assisted transesterification of waste cooking oil. Environ. Sci. Pollut. Res. 2020, 27, 27244–27255. [Google Scholar] [CrossRef]
- Izida, T.; Silva, J.; Andrade, L.; Simionatto, E.; Scharf, D.; Lima, S. Modeling transesterification reaction kinetics using fluorescence spectroscopy to interpret biodiesel production. Chem. Eng. Sci. 2020, 211, 115292. [Google Scholar] [CrossRef]
- Behera, B.; Selvam S, M.; Dey, B.; Balasubramanian, P. Algal biodiesel production with engineered biochar as a heterogeneous solid acid catalyst. Bioresour. Technol. 2020, 310, 123392. [Google Scholar] [CrossRef] [PubMed]
- Rezania, S.; Oryani, B.; Park, J.; Hashemi, B.; Yadav, K.K.; Kwon, E.E.; Hur, J.; Cho, J. Review on transesterification of non-edible sources for biodiesel production with a focus on economic aspects, fuel properties and by-product applications. Energy Convers. Manag. 2019, 201, 112155. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Gurav, R.; Choi, T.-R.; Kim, H.J.; Yang, S.-Y.; Song, H.-S.; Park, J.Y.; Park, Y.-L.; Han, Y.-H.; Choi, Y.-K.; et al. Conversion of waste cooking oil into biodiesel using heterogenous catalyst derived from cork biochar. Bioresour. Technol. 2020, 302, 122872. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.A.; Sakthinathan, G.; Vignesh, R.; Banu, J.R.; Al-Muhtaseb, A.H. Optimized transesterification reaction for efficient biodiesel production using Indian oil sardine fish as feedstock. Fuel 2019, 253, 921–929. [Google Scholar] [CrossRef]


| S.NO. | Properties | Waste Cooking Oil | Algae Oil | Blend of Waste Cooking Oil and Algae-Oil |
|---|---|---|---|---|
| 1. | Flash point (°C) | 256.00 | 102.00 | 105.85 |
| 2. | FFA (%) | 20.00 | 0.50 | 1.00 |
| 3. | Density (kg/m3) | 0.919 | 0.86 | 0.8614 |
| 4. | Viscosity (cst) @40 °C | 39.4 | 21.00 | 21.46 |
| Parameters | Symbol | Unit | Levels | ||
|---|---|---|---|---|---|
| −1 | 0 | 1 | |||
| Reaction time (Rt) | A | Minutes | 60 | 120 | 180 |
| Methanol/oil ratio | B | % | 20 | 40 | 60 |
| Catalyst-concentration (CC) | C | % | 00 | 01 | 02 |
| Run No. | Time (in Minutes) | Methanol/Oil Ratio (%) | Catalyst-Concentration (CC) (%) |
|---|---|---|---|
| A | B | C | |
| 1 | 180 | 40 | 0 |
| 2 | 60 | 60 | 1 |
| 3 | 120 | 60 | 2 |
| 4 | 60 | 20 | 1 |
| 5 | 120 | 40 | 1 |
| 6 | 120 | 40 | 1 |
| 7 | 180 | 60 | 1 |
| 8 | 120 | 20 | 2 |
| 9 | 120 | 40 | 1 |
| 10 | 180 | 40 | 2 |
| 11 | 60 | 40 | 2 |
| 12 | 60 | 40 | 0 |
| 13 | 180 | 20 | 1 |
| 14 | 120 | 20 | 0 |
| 15 | 120 | 40 | 1 |
| 16 | 120 | 60 | 0 |
| 17 | 120 | 40 | 1 |
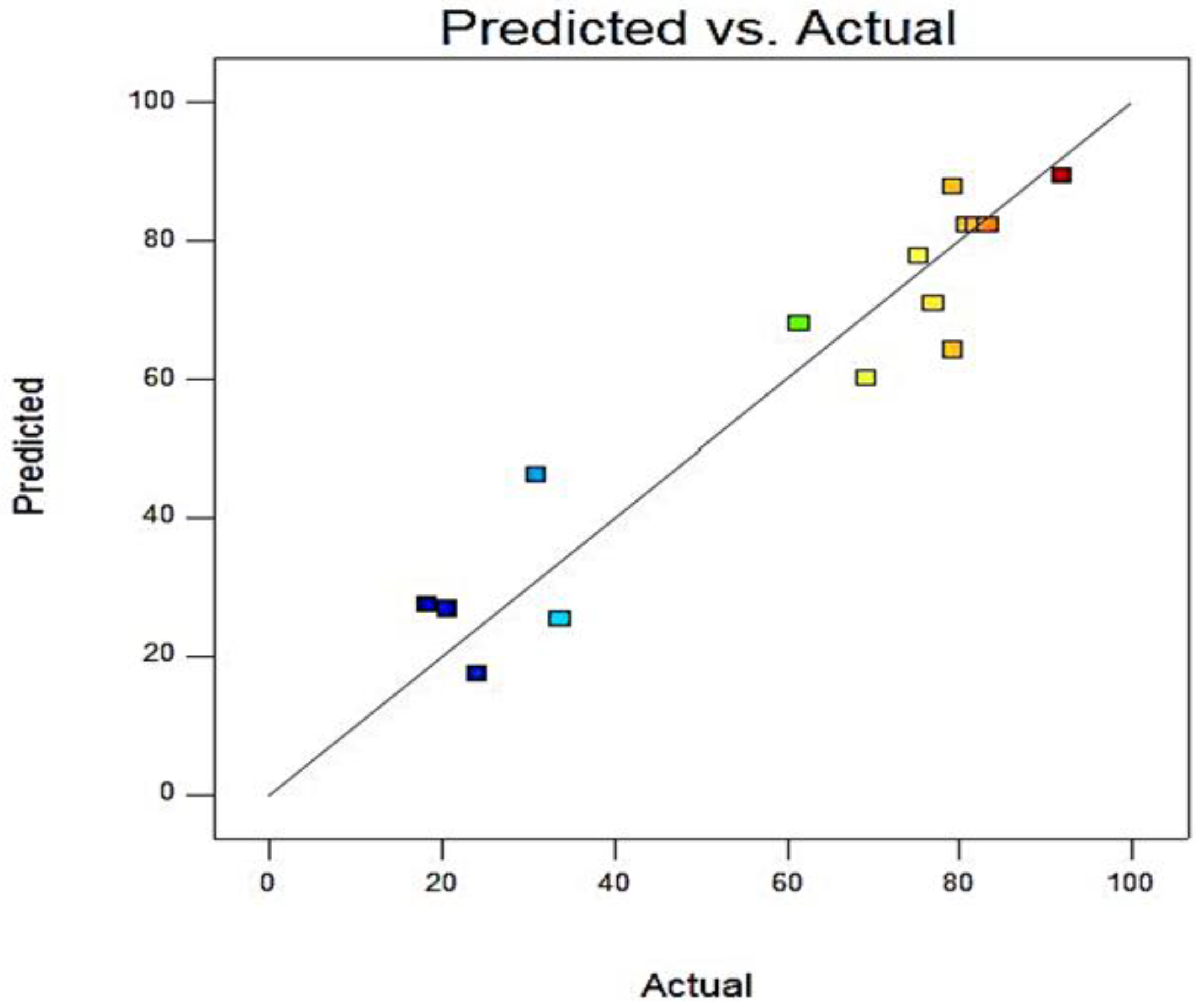
| Run | Time (Minutes) | Methanol/Oil Ratio (%) | Catalyst Concentration (CC) (%) | Blend Biodiesel Yield (%) | Error (%) | |
|---|---|---|---|---|---|---|
| Experimental | Predicted | |||||
| A | B | C | ||||
| 1 | 180 | 40 | 0 | 20.8 | 26.78 | 22.33 |
| 2 | 60 | 60 | 1 | 31.01266 | 46.11 | 32.742 |
| 3 | 120 | 60 | 2 | 69.23077 | 60.14 | −15.116 |
| 4 | 60 | 20 | 1 | 91.90556 | 89.46 | −2.7336 |
| 5 | 120 | 40 | 1 | 82.8125 | 82.36 | −0.5494 |
| 6 | 120 | 40 | 1 | 80.71429 | 82.36 | 1.998 |
| 7 | 180 | 60 | 1 | 75.33333 | 77.81 | 3.1829 |
| 8 | 120 | 20 | 2 | 79.27928 | 87.71 | 9.6120 |
| 9 | 120 | 40 | 1 | 81.95489 | 82.36 | 0.4918 |
| 10 | 180 | 40 | 2 | 61.42857 | 68.10 | 9.7965 |
| 11 | 60 | 40 | 2 | 76.97842 | 71.02 | −8.3897 |
| 12 | 60 | 40 | 0 | 24.06015 | 17.41 | −38.197 |
| 13 | 180 | 20 | 1 | 79.27928 | 64.20 | −23.487 |
| 14 | 120 | 20 | 0 | 18.42105 | 27.54 | 33.1116 |
| 15 | 120 | 40 | 1 | 83.33333 | 82.36 | −1.1817 |
| 16 | 120 | 60 | 0 | 33.77483 | 25.37 | −33.129 |
| 17 | 120 | 40 | 1 | 82.92683 | 82.36 | −0.6882 |
| S.No. | Parameters | Value |
|---|---|---|
| 1. | Model | Significant |
| 2. | Lack of fit | Non-Significant |
| 3. | R2 | 0.9904 |
| 4. | Adjusted R2 | 0.9745 |
| 5. | Predicted R2 | 0.9044 |
| Parameters | Objective | Optimized Value RSM |
|---|---|---|
| Reaction time (Rt) | In-range | 110 |
| Methanol volumetric ratio | Minimized | 21 |
| Catalyst concentration (CC) | In-range | 1.5 |
| Biodiesel-yield | Maximized | 95.15 |
| S.No. | Methanol/Oil Volumetric Ratio | Reaction-Temperature (°C) | Catalyst-Loading (wt%) | Reaction-Time (min) | Predicted-Biodiesel Yield (%) | Experimental-Biodiesel Yield (%) | Error (%) |
|---|---|---|---|---|---|---|---|
| 1 | 21 | 50 | 1.5 | 110 | 95.15 | 92 | −3.423 |
| S.NO. | Property (Unit) | IS-15607 | IS-15607 Limits | Biodiesel |
|---|---|---|---|---|
| 1 | Flashpoint (°C) | IS 1448 | - | 105.85 |
| 2 | Viscosity at 40 °C (cSt) | IS 1448 | 6.18 | 4.20 |
| 3 | Water and sediment (vol%) | D: 2709 | Max 0.05 | 0.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jain, S.; Kumar, N.; Singh, V.P.; Mishra, S.; Sharma, N.K.; Bajaj, M.; Khan, T.M.Y. Transesterification of Algae Oil and Little Amount of Waste Cooking Oil Blend at Low Temperature in the Presence of NaOH. Energies 2023, 16, 1293. https://doi.org/10.3390/en16031293
Jain S, Kumar N, Singh VP, Mishra S, Sharma NK, Bajaj M, Khan TMY. Transesterification of Algae Oil and Little Amount of Waste Cooking Oil Blend at Low Temperature in the Presence of NaOH. Energies. 2023; 16(3):1293. https://doi.org/10.3390/en16031293
Chicago/Turabian StyleJain, Siddharth, Nitin Kumar, Varun Pratap Singh, Sachin Mishra, Naveen Kumar Sharma, Mohit Bajaj, and T. M. Yunus Khan. 2023. "Transesterification of Algae Oil and Little Amount of Waste Cooking Oil Blend at Low Temperature in the Presence of NaOH" Energies 16, no. 3: 1293. https://doi.org/10.3390/en16031293
APA StyleJain, S., Kumar, N., Singh, V. P., Mishra, S., Sharma, N. K., Bajaj, M., & Khan, T. M. Y. (2023). Transesterification of Algae Oil and Little Amount of Waste Cooking Oil Blend at Low Temperature in the Presence of NaOH. Energies, 16(3), 1293. https://doi.org/10.3390/en16031293











