Suitability and Energy Sustainability of Atmospheric Water Generation Technology for Green Hydrogen Production
Abstract
:1. Introduction
2. Methodology
- A focus on the water quality required for electrolysis and a literature research regarding the AWG water quality findings, in order to better understand the main issues related to the intended use. After that, experimental tests, on specific parameters, were carried out on water that was derived from a real integrated AWG machine, similar to that used by the authors in [21], in the analytical chemistry labs at the University of Pavia.
- A case study analysis was performed concerning an integration between the AWG machine and a system composed of a hydrolyser and a photovoltaic field, serving it. This section was carried out using a part of the methodology proposed, by the authors, in [21]; in particular, the following points were developed:
- ○
- Data collection about the required water quality and quantity, not only for the electrolyzer use, but also to maintain clean photovoltaic panels.
- ○
- Weather conditions data collection, in terms of temperatures and relative humidity of the installation site, taking into account the best sampling frequency that is suitable for the considered climate, in compliance with the findings reported by the authors in [22].
- ○
- Integrated AWG machine behavior analyses, carried out by means of the simulation tool described, by the authors, in [23].
- ○
- Covered needs evaluation, in particular, considering the water used for panel cleaning, applying the DIrt method [24], and the water required for the electrolysis process.
- ○
- Energy sustainability evaluation, intended as the possibility to cover the energy requirements of the system by means of an energy source that does not inflict long-term damage to the environment and can be replenished within the human lifetime [25]. In particular, in this case, it was analyzed as the possibility to cover the energy requirements by means of a renewable source.
3. Water Requirements for Electrolysis
4. Water Extraction from the Air: A Brief Summary of the Current Techniques
- Fog nets: systems composed of a metallic mesh causing the fog or mist droplets coalescence. Various geometries and materials have been studied in order to enhance their yield. Such a solution can only be effective in those places where there are fogs [42].
- Radiative panels: surfaces exposed to the external environment, which exploit the natural temperature gradients and the radiation to the heaven vault to obtain the dew condensation. In order to enhance the panel performances, different bioinspired coating have been recently studied, including, in particular, solutions that alternate between hydrophilic and hydrophobic surfaces. They require large areas to collect meaningful water quantities and particular climates [18].
- Sorption-based AHW: systems composed of one or more layers of desiccants which adsorb the vapor from the air, releasing it, by means of solar heating, in a closed air volume to enhance the dew point. After that, through exploiting the natural outside temperature gradients, part of the vapor condensates. Such a kind of system produces typical yields of few liters per day [43]. An interesting development in this field is represented by moisture-adsorbent gels, which demonstrated the ability to directly release liquid water, with a slight heating at low temperature (40 °C) [44].
- Compressor-based AWGs: these machines cool down a flow of air under its dew point, obtaining vapor condensation, by means of a thermodynamic reverse cycle, employing a refrigerant worked using a compressor. Such a kind of approach is the most diffused among the commercially available machines, as it provides water productions ranging from tens to thousands liters per day [36].
- Thermo-electric cooler (TEC)-based AWGs: systems that operate the cooling, on a flux of air, driving it under its dew point, by means of Peltier cells. The advantage, in comparison to the previous technique, is that the refrigerant and the compressor are avoided, thus the only moving parts are the fans. The disadvantage is that they can only work under a very narrow range of temperature and relative humidity, and their cooling efficiency is far less in comparison to that of a compressor reverse cycle [45].
- Hybrid sorption-based AWGs: machines that employ desiccants inside the compressor-based AWGs. These systems are able to enhance the water content in the air by means of desiccants and use the concept of the compressor-based machines to obtain absorb–desorb cycles. In current times, such solutions are still undergoing an experimental stage [46].
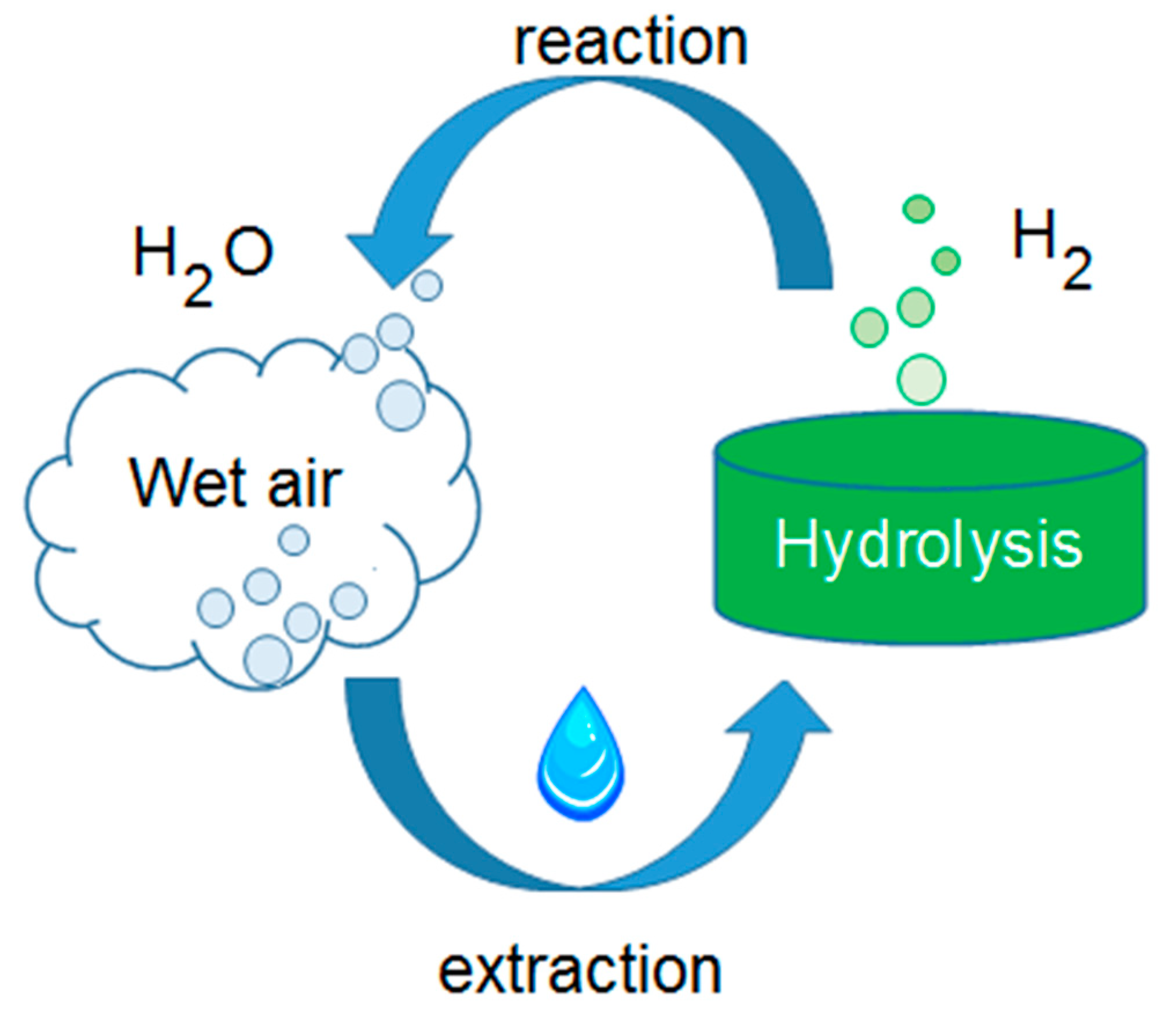
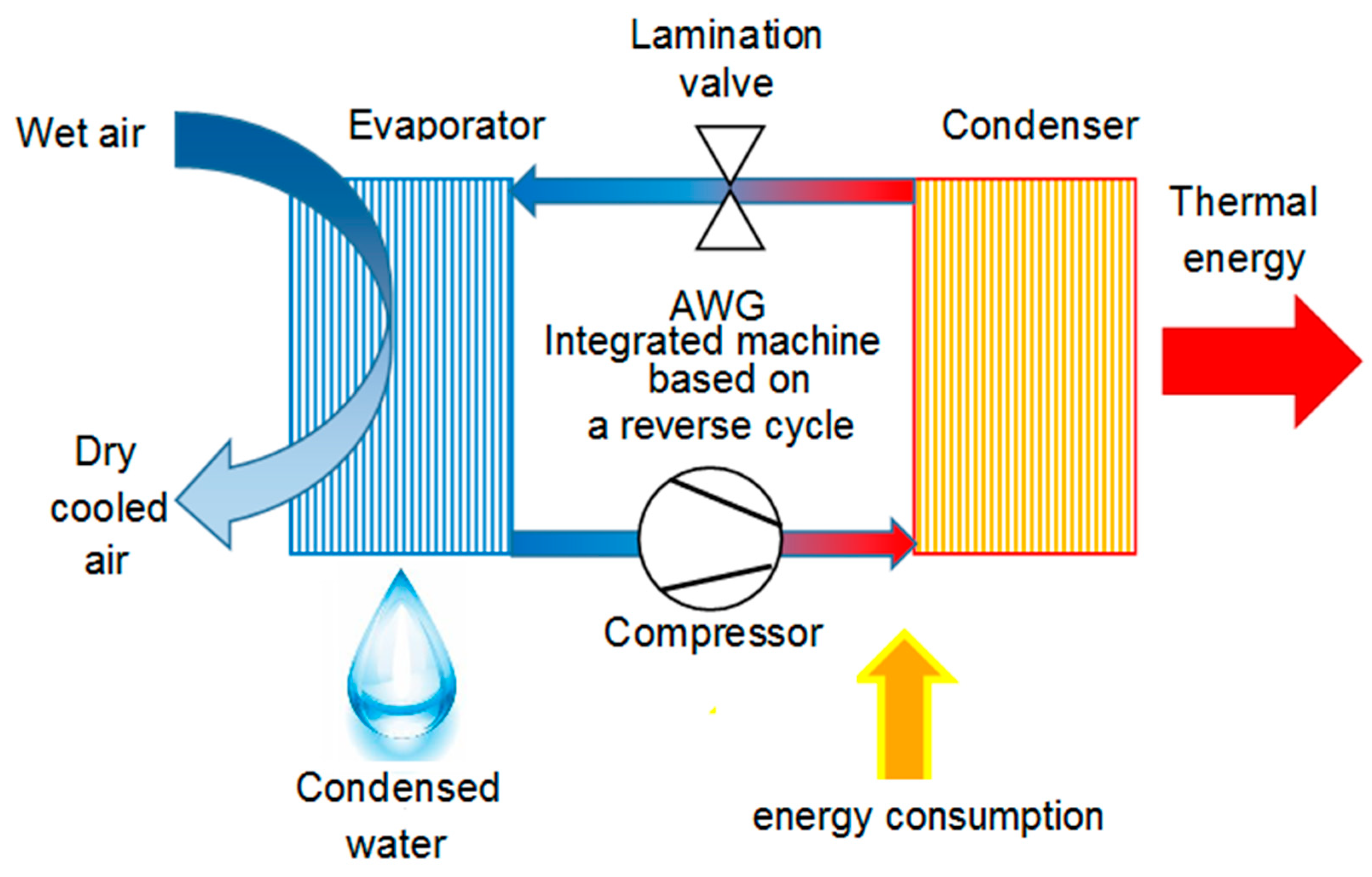
- Integrated AWGs: advanced compressor-based AWGs that are studied to permit the exploitation of all the useful effects of the thermodynamic reverse cycle [47]. Such an approach can be the mean to bypass the energy and economic issues related to water harvesting from the air and obtain sustainable solutions [48]. Using a compressor-based technique, there are two by-products of this water extraction procedure: a flux of cooled and dry air and a heat flux, at low temperatures, coming from the cooling process of the said air flow. The first one is suitable as the primary air into an air-conditioned area, such as that described, by the authors, in [23]. The second one is suitable for domestic water heating, similar to the example reported, by the authors, in [21]. A scheme of an integrated machine working cycle is reported in Figure 2. The use of these by-products inside a building can help the existing heating and cooling plants, providing energy savings. The lower the existing plants efficiency, the higher the energy and the economic savings will be.
5. Water from Air Quality, and Some Hints and Comments about the Current Findings
- When the water source is the external air, previously mechanically filtered, the main chemical pollution, even under heavily air polluted environments, seems to only be composed of nitrites and ammonia [50]. Ammonia levels frequently exceed the limit of 0.5 mg/L and tend to increase when the relative humidity decreases [51]. Mechanical filtration can be found in active systems, while, normally, it is absent in the passive ones as it requires energy;
- Passive systems, that do not comprise any air filtration nor anti-intrusion protections, are affected by macroscopic pollutants and pollution vectors, such as dust particles, animals, insects, and so on [52], and the gathered water is more polluted in comparison to that collected by means of active systems (that are equipped with filters and protections) [50];
- The materials employed in the condenser surface and into the water collection systems can be the means of a further pollution, as some of the composing elements can be released into the water, as underlined by the authors of [53]. In this perspective, desiccants, liquid or solid, must also be deeply analyzed. In order to avoid any compounds being released into water, it is advisable to employ materials certified as “food-degree” quality;
- Viruses, bacteria, molds, and fungi pollution can affect the collected water; such an issue can be almost negligible at the beginning of the AWG use, but it can become very challenging with time [54];
- If indoor air, of inhabited buildings, is used for water extraction, it is possible to find more pollution in the condensate, due to the pollutants released by humans [53]. The biological contamination hazard could also increase for the same reason. If water harvesting is carried out in greenhouses, it may possible to find more salts [53];
- On the contrary, water obtained from external air is generally poor in mineral content, therefore showing a low conductivity [50]; such a residual conductivity is maybe due to the presence of ammonia and nitrites. Such a characteristic make such a kind of water particularly suitable for photovoltaic panel washing [24] and for various other industrial uses. In addition, a potential suitability for hydrogen generation is mentioned by the authors of [43];
6. Integrated Machine Water Quality Analysis
- An inlet air section equipped with a double filtration layer, comprising pre-filtration and pocket filtration;
- An open screw compressor, with a cooling power of 100 kW and a heating power of 120 kW;
- Evaporative and heat recovery coils, built with materials that have a food-degree quality, and where vapor condensation occurs;
- Centrifugal evaporator fans, which are speed controlled and able to process a variable airflow;
- An outlet air section suitable for external air duct connection, for the cool and dry air recovery and use;
- Two condenser coils with alternative uses: a plate coil, suitable for the heat recovery, and a fin and tube coil, for the heat dissipation into the environment, in case the domestic water heating is not required;
- An expansion valve, ruled using a proportional integrative derivative (PID) controller, located between the coolant liquid receiver tank and the evaporator;
- A stainless steel water storage connected using a pump to the stainless steel water collection basin. Such a storage component is equipped with a faucet that allows users to completely drain the storage and to collect water samples before entering the water treatment unit;
- A 500 L capacity stainless steel water storage tank;
- A water treatment system that can be customized in function of the required final water use. For the purpose of the current study, water was directly taken before the treatment, in order to understand whether it was already suitable for the scope, or which treatments were required to achieve the required quality.
7. Case Study
- A total of 10 kg of water is needed to produce 1 kg of hydrogen;
- The hydrogen will only be produced from May to September; in such a period, the hydrolyser requires 1520 L/day;
- An annual amount of 226,000 L of water is required to produce the intended hydrogen quantity;
- The water should be deionized. In particular, the concentrations of aluminum, iron, manganese, nickel, chromium, and zinc should be under 10 μg/L, and the allowed maximum conductivity is 5 μS/cm;
- The photovoltaic field requires about 6400 L of demineralized water to be entirely cleaned, taking into account the value of 3.2 L/kW found by the authors of [56]. Under the hypothesis of “clean plant”, the field is expected to provide 2713 MWh/year of electricity, with an average daily production of 7.43 MWh.
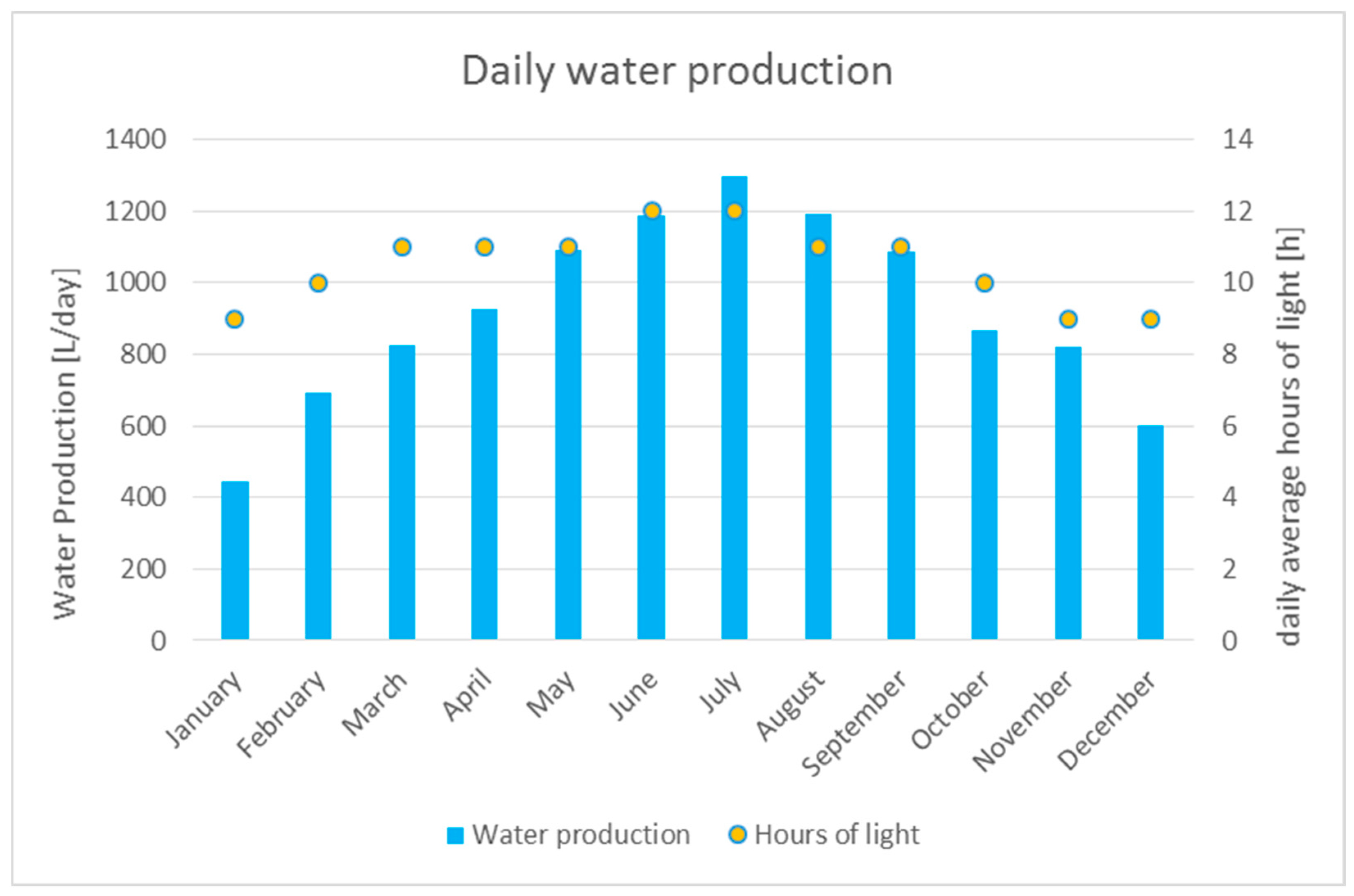
7.1. Weather and Data Set Choice
7.2. Water Treatment
7.3. Photovoltaic Field Production and Integrated Machine Behaviour
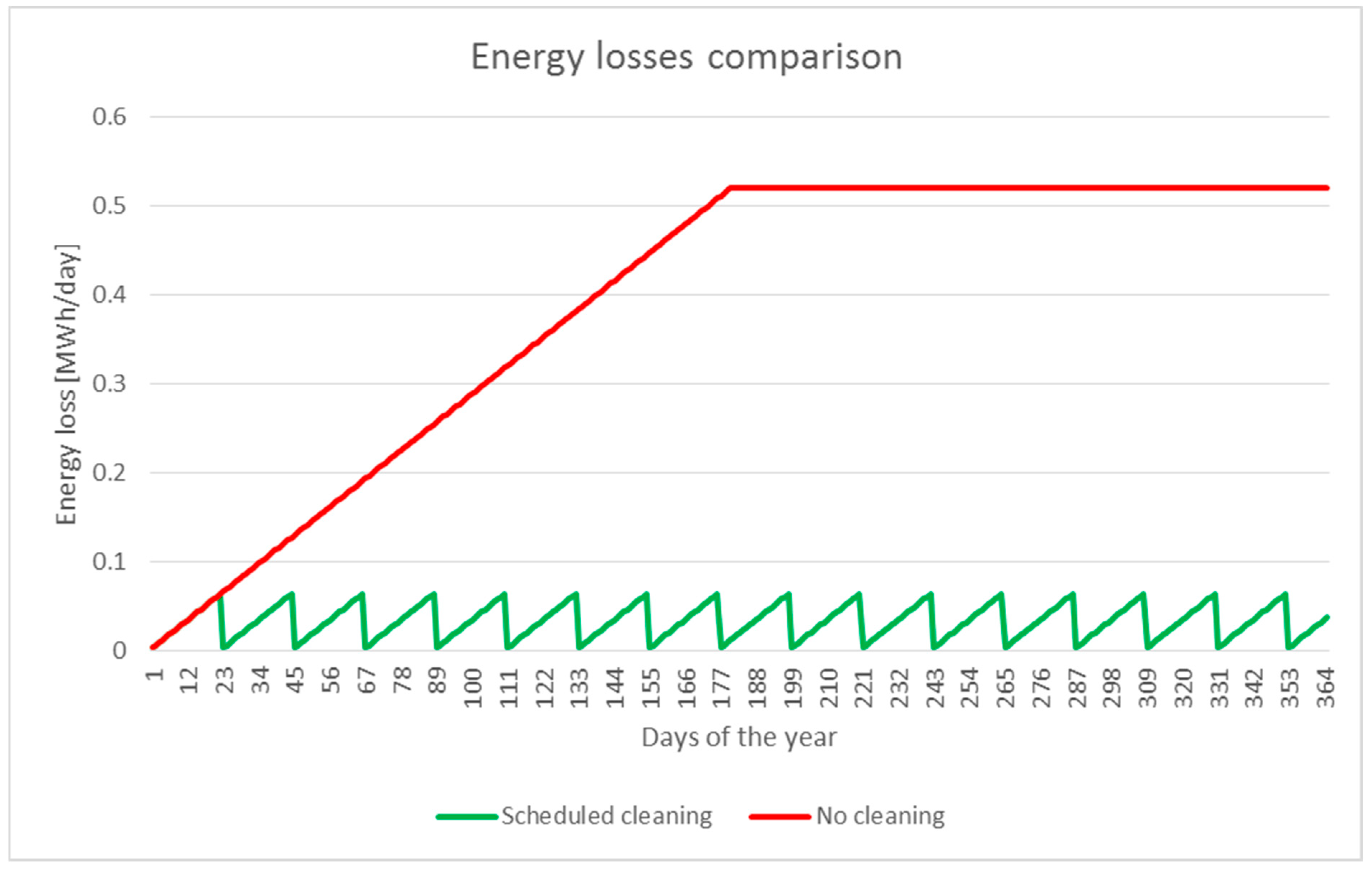
7.4. Photovoltaic Panel Washing and Energy Recovery
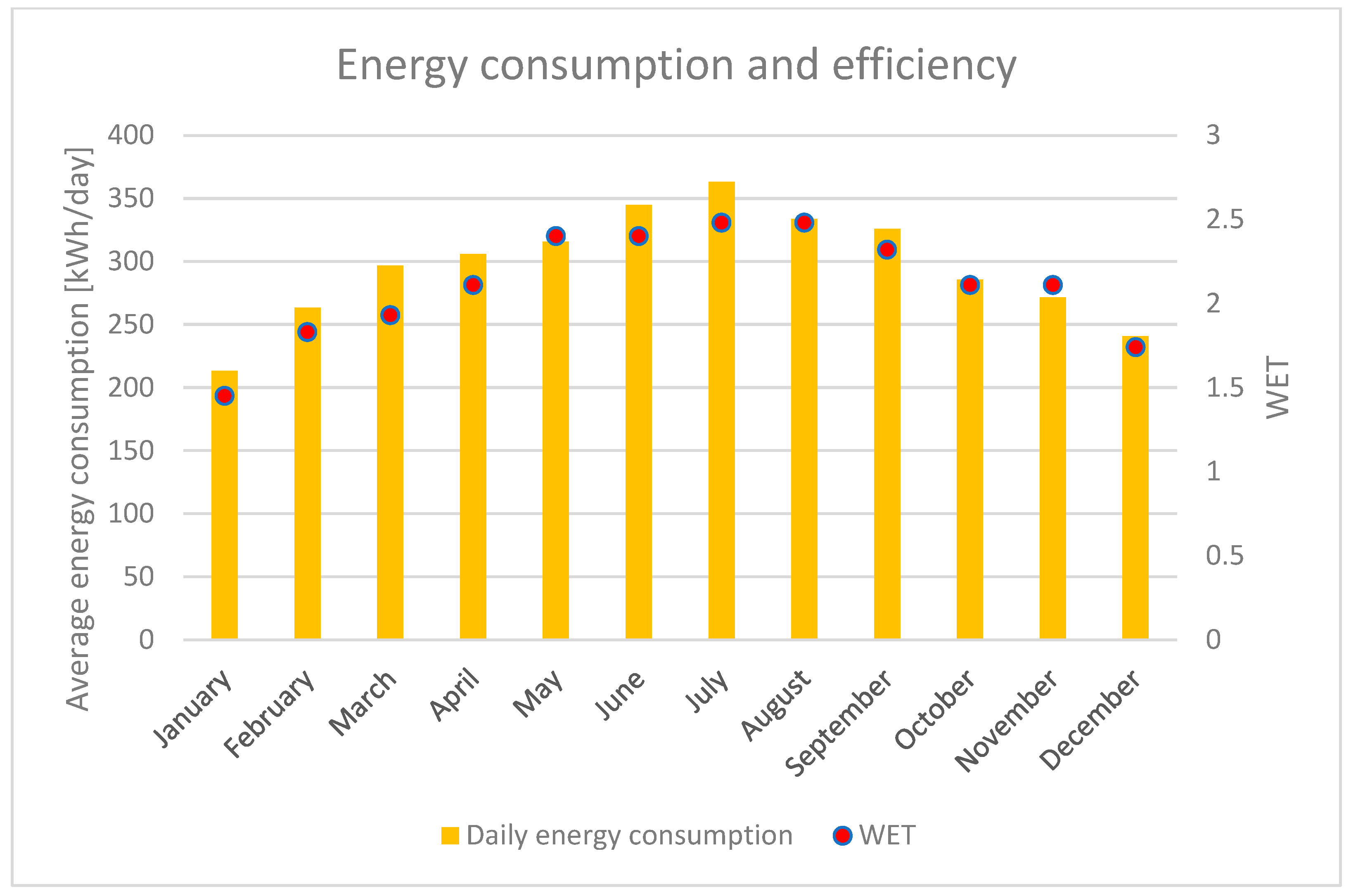
7.5. Results Analysis
8. Future Developments
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| Acronyms | |
| AWG | Air-to-water generator |
| AWH | Air water harvesting |
| CDI | Combined drought indicator |
| COP | Coefficient of performance |
| EER | Energy efficiency ratio |
| EU | European Union |
| FH2R | Hydrogen energy research field |
| HVAC | Heating ventilation air conditioning |
| PID | Proportional integrative derivative |
| PLC | Programmable logic controller |
| TEC | Thermo-electric cooler |
| WET | Water energy transformation |
| WHO | World Health Organization |
| UV | Ultra violet |
| Symbols | |
| DIr | Constant, which summarizes all deposition phenomena [1/h] |
| en | Energy [kWh] or [kJ] |
| ηloss | Efficiency loss due to soiling [-] |
| m | Water mass [kg] |
| Qc | Condensation energy [kJ] |
| R.H. | Relative humidity [%] |
| T | Dry bulb temperature [°C] |
| t | Time period passed after the cleaning (day) |
References
- United Nations Climate Change. Maintaining a Clear Intention to Keep 1.5 °C within Reach. Available online: https://unfccc.int/maintaining-a-clear-intention-to-keep-15degc-within-reach (accessed on 10 March 2023).
- European Commission. 2050 Long-Term Strategy. Available online: https://climate.ec.europa.eu/eu-action/climate-strategies-targets/2050-long-term-strategy_en (accessed on 10 March 2023).
- Kovač, A.; Paranos, M.; Marciuš, D. Hydrogen in energy transition: A review. Int. J. Hydrogen Energy 2021, 46, 10016–10035. [Google Scholar] [CrossRef]
- Green Hydrogen Definition. Available online: https://www.sciencedirect.com/topics/engineering/green-hydrogen (accessed on 22 June 2021).
- The Future of Hydrogen. IEA, Paris. 2019. Available online: https://www.iea.org/reports/the-future-of-hydrogen (accessed on 17 March 2023).
- European Commission. Hydrogen European Strategy. Available online: https://energy.ec.europa.eu/topics/energy-systems-integration/hydrogen_en#eu-hydrogen-strategy (accessed on 10 March 2023).
- European Commission. EU Strategy on Energy System Integration. Available online: https://energy.ec.europa.eu/topics/energy-systems-integration/eu-strategy-energy-system-integration_en (accessed on 10 March 2023).
- Bairrão, D.; Soares, J.; Almeida, J.; Franco, J.F.; Vale, Z. Green Hydrogen and Energy Transition: Current State and Prospects in Portugal. Energies 2023, 16, 551. [Google Scholar] [CrossRef]
- Schill, W.-P. Electricity Storage and the Renewable Energy Transition. Joule 2020, 4, 2059–2064. [Google Scholar] [CrossRef]
- Taseska, T.; Yu, W.; Wilsey, M.K.; Cox, C.P.; Meng, Z.; Ngarnim, S.S.; Müller, A.M. Analysis of the Scale of Global Human Needs and Opportunities for Sustainable Catalytic Technologies. Top. Catal. 2023, 66, 338–374. [Google Scholar] [CrossRef]
- Lamsal, D.; Sreeram, V.; Mishra, Y.; Kumar, D. Output power smoothing control approaches for wind and photovoltaic generation systems: A review. Renew. Sustain. Energy Rev. 2019, 113, 109245. [Google Scholar] [CrossRef]
- The World’s Largest-Class Hydrogen Production, Fukushima Hydrogen Energy Research Field (FH2R) Now is Completed at Namie Town in Fukushima. Available online: https://www.global.toshiba/ww/news/energy/2020/03/news-20200307-01.html (accessed on 1 May 2023).
- World Health Organization. Drinking Water. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/drinking-water (accessed on 22 March 2023).
- Toreti, A.; Bavera, D.; Acosta Navarro, J.; De Jager, A.; Di Ciollo, C.; Maetens, W.; Magni, D.; Masante, D.; Mazzeschi, M.; Spinoni, J.; et al. Drought in Europe: August 2022: GDO Analytical Report; Publications Office of the European Union: Luxembourg, 2022. [CrossRef]
- Toreti, A.; Bavera, D.; Acosta Navarro, J.; Arias-Muñoz, C.; Avanzi, F.; Marinho Ferreira Barbosa, P.; De Jager, A.; Di Ciollo, C.; Ferraris, L.; Fioravanti, G.; et al. Drought in Europe March 2023; Publications Office of the European Union: Luxembourg, 2023. [CrossRef]
- Karimidastenaei, Z.; Avellán, T.; Sadegh, M.; Kløve, B.; Haghighi, A.T. Unconventional water resources: Global opportunities and challenges. Sci. Total Environ. 2022, 827, 154429. [Google Scholar] [CrossRef]
- Woods, P.; Bustamante, H.; Aguey-Zinsou, K.-F. The hydrogen economy—Where is the water? Energy Nexus 2022, 7, 100123. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, R.; Li, Y. Diversifying Water Sources with Atmospheric Water Harvesting to Enhance Water Supply Resilience. Sustainability 2022, 14, 7783. [Google Scholar] [CrossRef]
- Kabir, M.M.; Akter, M.M.; Huang, Z.; Tijing, L.; Shon, H.K. Hydrogen production from water industries for a circular economy. Desalination 2023, 554, 116448. [Google Scholar] [CrossRef]
- Thimmappa, R.; Gautam, M.; Bhat, Z.M.; Thodika, A.R.A.; Devendrachari, M.C.; Mukhopadhyay, S.; Dargily, N.C.; Thotiyl, M.O. An atmospheric water electrolyzer for decentralized green hydrogen production. Cell Rep. Phys. Sci. 2021, 2, 100627. [Google Scholar] [CrossRef]
- Cattani, L.; Magrini, A.; Cattani, P. Water Extraction from Air by Refrigeration—Experimental Results from an Integrated System Application. Appl. Sci. 2018, 8, 2262. [Google Scholar] [CrossRef]
- Cattani, L.; Magrini, A.; Leoni, V. Energy Performance of Water Generators from Gaseous Mixtures by Condensation: Climatic Datasets Choice. Energies 2022, 15, 7581. [Google Scholar] [CrossRef]
- Cattani, L.; Cattani, P.; Magrini, A. Air to Water Generator Integrated System Real Application: A Study Case in a Worker Village in United Arab Emirates. Appl. Sci. 2023, 13, 3094. [Google Scholar] [CrossRef]
- Cattani, L.; Cattani, P.; Magrini, A. Photovoltaic Cleaning Optimization: A Simplified Theoretical Approach for Air to Water Generator (AWG) System Employment. Energies 2021, 14, 4271. [Google Scholar] [CrossRef]
- Hollaway, L. Sustainable energy production: Key material requirements. In Woodhead Publishing Series in Civil and Structural Engineering, Advanced Fiber-Reinforced Polymer (FRP) Composites for Structural Applications, 2nd ed.; Bai, J., Ed.; Woodhead Publishing: Soston, UK, 2013; pp. 675–701. ISBN 9780128203460. [Google Scholar] [CrossRef]
- Lagioia, G.; Spinelli, M.P.; Amicarelli, V. Blue and green hydrogen energy to meet European Union decarbonisation objectives. An overview of perspectives and the current state of affairs. Int. J. Hydrogen Energy 2023, 48, 1304–1322. [Google Scholar] [CrossRef]
- Chi, J.; Yu, H. Water electrolysis based on renewable energy for hydrogen production. Chin. J. Catal. 2018, 39, 390–394. [Google Scholar] [CrossRef]
- Vadivel, D.; Sturini, M.; Speltini, A.; Dondi, D. Tungsten Catalysts for Visible Light Driven Ofloxacin Photocatalytic Degradation and Hydrogen Production. Catalysts 2022, 12, 310. [Google Scholar] [CrossRef]
- Gong, M.; Wang, D.-Y.; Chen, C.-C.; Hwang, B.-J.; Dai, H. A mini review on nickel-based electrocatalysts for alkaline hydrogen evolution reaction. Nano Res. 2016, 9, 28–46. [Google Scholar] [CrossRef]
- Khan, M.A.; Al-Attas, T.; Roy, S.; Rahman, M.M.; Ghaffour, N.; Thangadurai, V.; Larter, S.; Hu, J.; Ajayan, P.M.; Kibria, G. Seawater electrolysis for hydrogen production: A solution looking for a problem? Energy Environ. Sci. 2021, 14, 4831–4839. [Google Scholar] [CrossRef]
- Rosca, V.; Duca, M.; de Groot, M.T.; Koper, M.T.M. Nitrogen Cycle Electrocatalysis. Chem. Rev. 2009, 109, 2209–2244. [Google Scholar] [CrossRef]
- Shi, X.; Liao, X.; Li, Y. Quantification of fresh water consumption and scarcity footprints of hydrogen from water electrolysis: A methodology framework. Renew. Energy 2020, 154, 786–796. [Google Scholar] [CrossRef]
- Gleick, P.H. Water Resources. In Encyclopedia of Climate and Weather; Schneider, S.H., Ed.; Oxford University Press: New York, NY, USA, 2011; Volume 2, pp. 817–823. ISBN 978-019-531-386-4. [Google Scholar]
- Graham, S.; Parkinson, C.; Chahine, M. The Water Cycle. Available online: https://earthobservatory.nasa.gov/features/Water (accessed on 7 April 2023).
- Gimeno, L.; Eiras-Barca, J.; Durán-Quesada, A.M.; Dominguez, F.; van der Ent, R.; Sodemann, H.; Sánchez-Murillo, R.; Nieto, R.; Kirchner, J.W. The residence time of water vapour in the atmosphere. Nat. Rev. Earth Environ. 2021, 2, 558–569. [Google Scholar] [CrossRef]
- Atmospheric Research. Atmospheric Water Generator or Water-from-Air Machine Suppliers Links. Available online: http://www.atmoswater.com/manufacturers-and-suppliers-of-atmospheric-water-generators--water-from-air-machines.html (accessed on 6 April 2023).
- Tashtoush, B.; Alshoubaki, A. Atmospheric water harvesting: A review of techniques, performance, renewable energy solutions, and feasibility. Energy 2023, 280, 128186. [Google Scholar] [CrossRef]
- Raveesh, G.; Goyal, R.; Tyagi, S. Advances in atmospheric water generation technologies. Energy Convers. Manag. 2021, 239, 114226. [Google Scholar] [CrossRef]
- Ahrestani, Z.; Sadeghzadeh, S.; Emrooz, H.B.M. An overview of atmospheric water harvesting methods, the inevitable path of the future in water supply. RSC Adv. R. Soc. Chem. 2023, 13, 10273–10307. [Google Scholar] [CrossRef] [PubMed]
- Thavalengal, M.S.; Jamil, M.A.; Mehroz, M.; Bin Xu, B.; Yaqoob, H.; Sultan, M.; Imtiaz, N.; Shahzad, M.W. Progress and Prospects of Air Water Harvesting System for Remote Areas: A Comprehensive Review. Energies 2023, 16, 2686. [Google Scholar] [CrossRef]
- Nikkhah, H.; Azmi, W.M.B.W.; Nikkhah, A.; Najafi, A.M.; Babaei, M.M.; Fen, C.S.; Nouri, A.; Mohammad, A.W.; Lun, A.W.; Yong, N.L.; et al. A comprehensive review on atmospheric water harvesting technologies: From thermodynamic concepts to mechanism and process development. J. Water Process Eng. 2023, 53, 103728. [Google Scholar] [CrossRef]
- Tu, Y.; Wang, R.; Zhang, Y.; Wang, J. Progress and Expectation of Atmospheric Water Harvesting. Joule 2018, 2, 1452–1475. [Google Scholar] [CrossRef]
- Ansari, E.; Ferber, N.L.; Milošević, T.; Barron, J.; Karanikolos, G.N.; AlMarzooqi, F.; Dumée, L.F.; Calvet, N. Atmospheric water generation in arid regions—A perspective on deployment challenges for the Middle East. J. Water Process Eng. 2022, 49, 103163. [Google Scholar] [CrossRef]
- Zhao, F.; Zhou, X.; Liu, Y.; Shi, Y.; Dai, Y.; Yu, G. Super Moisture-Absorbent Gels for All-Weather Atmospheric Water Harvesting. Adv. Mater. 2019, 31, e1806446. [Google Scholar] [CrossRef]
- Asham, A.D.; Abdelaal, W.G.A.; Mamdouh, W.M. A Novel Ambiently Adaptive Atmospheric Water Generator Using Maximum Production Tracking Algorithm. Teh. Vjesn. Tech. Gaz. 2022, 29, 797–805. [Google Scholar] [CrossRef]
- Shemelin, V.; Pokorny, N.; Novotny, J. Experimental investigation of silica gel and zeolite coated fin-tube heat exchangers under arid climatic conditions. Energy Rep. 2022, 8, 331–341. [Google Scholar] [CrossRef]
- Cattani, L.; Cattani, P.; Magrini, A. Air to Water Generator Integrated Systems: The Proposal of a Global Evaluation Index—GEI Formulation and Application Examples. Energies 2021, 14, 8528. [Google Scholar] [CrossRef]
- Moghimi, F.; Ghoddusi, H.; Asiabanpour, B.; Behroozikhah, M. Is atmospheric water generation an economically viable solution? Clean Technol. Environ. Policy 2021, 23, 1045–1062. [Google Scholar] [CrossRef]
- Mandal, C.S.; Agarwal, M.; Reddy, V.; Kudapa, V.K. Water from air—A sustainable source of water. Mater. Today Proc. 2021, 46, 3352–3357. [Google Scholar] [CrossRef]
- Kaplan, A.; Ronen-Eliraz, G.; Ratner, S.; Aviv, Y.; Wolanov, Y.; Avisar, D. Impact of industrial air pollution on the quality of atmospheric water production. Environ. Pollut. 2023, 325, 121447. [Google Scholar] [CrossRef]
- Inbar, O.; Chudnovsky, A.; Ohneiser, K.; Ansmann, A.; Ratner, S.; Sirota, R.; Aviv, Y.; Avisar, D. Air-water interactions: The signature of meteorological and air-quality parameters on the chemical characteristics of water produced from the atmosphere. Sci. Total. Environ. 2021, 790, 147940. [Google Scholar] [CrossRef]
- Algarni, S. Assessment of fog collection as a sustainable water resource in the southwest of the Kingdom of Saudi Arabia. Water Environ. J. 2018, 32, 301–309. [Google Scholar] [CrossRef]
- Jurga, A.; Pacak, A.; Pandelidis, D.; Kaźmierczak, B. Condensate as a water source in terrestrial and extra-terrestrial conditions. Water Resour. Ind. 2023, 29, 100196. [Google Scholar] [CrossRef]
- Jahne, M.; Pfaller, S.; Garland, J.; Impellitteri, C. Evaluation of Atmospheric Water Generation Technology: Microbial Water Quality; U.S. Environmental Protection Agency: Washington, DC, USA, 2018; EPA/600/R-18/379.
- Grigoriev, S.; Fateev, V.; Bessarabov, D.; Millet, P. Current status, research trends, and challenges in water electrolysis science and technology. Int. J. Hydrogen Energy 2020, 45, 26036–26058. [Google Scholar] [CrossRef]
- Jones, R.K.; Baras, A.; Al Saeeri, A.; Al Qahtani, A.; Al Amoudi, A.O.; Al Shaya, Y.; Alodan, M.; Al-Hsaien, S.A. Optimized Cleaning Cost and Schedule Based on Observed Soiling Conditions for Photovoltaic Plants in Central Saudi Arabia. IEEE J. Photovolt. 2016, 6, 730–738. [Google Scholar] [CrossRef]
- Gaspar, P.D. Handbook of Research on Advances and Applications in Refrigeration Systems and Technologies; Gaspar, P.D., Dihno da Silva, P., Eds.; IGI Global: Hershey, PA, USA, 2015; ISBN 978-146-668-398-3. [Google Scholar]
- Cattani, L.; Magrini, A.; Cattani, P. Water Extraction from Air: A Proposal for a New Indicator to Compare Air Water Generators Efficiency. Energies 2021, 14, 224. [Google Scholar] [CrossRef]
- Appels, R.; Lefevre, B.; Herteleer, B.; Goverde, H.; Beerten, A.; Paesen, R.; De Medts, K.; Driesen, J.; Poortmans, J. Effect of soiling on photovoltaic modules. Sol. Energy 2013, 96, 283–291. [Google Scholar] [CrossRef]
- Schill, C.; Brachmann, S.; Koehl, M. Impact of soiling on IV-curves and efficiency of PV-modules. Sol. Energy 2015, 112, 259–262. [Google Scholar] [CrossRef]
- Paudyal, B.R.; Shakya, S.R. Dust accumulation effects on efficiency of solar PV modules for off grid purpose: A case study of Kathmandu. Sol. Energy 2016, 135, 103–110. [Google Scholar] [CrossRef]
- Commercial Cleaning Robot Vendor Site. Available online: http://www.washpanel.com/index.php (accessed on 20 June 2023).
| Characteristics | Low-Temperature Electrolysis | High-Temperature Electrolysis | |||
|---|---|---|---|---|---|
| Charge carrier | OH− | OH− | H+ | H+ | O2− |
| Temperature range (°C) | 20–80 | 20–200 | 20–200 | 500–1000 | 500–1000 |
| Electrolyte type | Liquid | Polymer | Polymer | Ceramic | Ceramic |
| Efficiency (%) | 60–70 | 65–82 | >99 | >99 | |
| Availability | Commercial | Laboratory scale | Commercial | Laboratory scale | Demonstration |
| Metal Ion | Detected Values | |
|---|---|---|
| Al | <10 | μg/L |
| Fe | <5 | μg/L |
| Mn | <5 | μg/L |
| Ni | <5 | μg/L |
| Cr | <5 | μg/L |
| Zn | <5 | μg/L |
| Weather Condition | January | February | March | April | May | June | July | August | September | October | November | December | Yearly Average |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T (°C) | 18.6 | 19.6 | 21.2 | 23.2 | 26.2 | 29.0 | 29.7 | 29.4 | 28.5 | 25.7 | 23.9 | 19.8 | 24.6 |
| RH (%) | 71 | 77 | 77 | 79 | 82 | 83 | 79 | 80 | 75 | 74 | 76 | 74 | 77 |
| Hour of the Day | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Photovoltaic Power Yield [kW} | |||||||||||||
| January | 0.0 | 0.1 | 90.2 | 286.0 | 508.1 | 692.0 | 818.4 | 875.7 | 794.8 | 736.7 | 149.4 | 0.0 | 0.0 |
| February | 0.0 | 6.1 | 140.0 | 375.1 | 607.5 | 824.0 | 958.0 | 1035.4 | 1008.4 | 935.7 | 792.6 | 65.7 | 0.0 |
| March | 0.0 | 33.2 | 199.4 | 442.2 | 690.8 | 886.8 | 1027.1 | 1083.0 | 1036.1 | 973.2 | 763.9 | 102.0 | 0.0 |
| April | 2.7 | 91.1 | 294.4 | 520.9 | 745.4 | 909.7 | 1046.3 | 1016.4 | 1028.8 | 936.2 | 723.3 | 111.7 | 0.0 |
| May | 14.5 | 138.4 | 346.3 | 577.0 | 785.3 | 989.2 | 1085.1 | 1153.8 | 1055.9 | 956.5 | 728.1 | 116.7 | 0.0 |
| June | 20.4 | 160.7 | 403.3 | 679.5 | 921.2 | 1099.6 | 1214.1 | 1251.1 | 1210.1 | 1110.7 | 903.4 | 407.0 | 35.6 |
| July | 12.2 | 139.4 | 379.4 | 646.3 | 895.0 | 1091.1 | 1237.9 | 1239.8 | 1202.5 | 1050.1 | 843.7 | 607.0 | 41.6 |
| August | 3.5 | 106.8 | 332.0 | 598.9 | 823.3 | 1029.2 | 1125.1 | 1163.6 | 1109.5 | 980.8 | 769.6 | 135.2 | 0.0 |
| September | 0.3 | 93.9 | 329.0 | 623.3 | 886.5 | 1093.7 | 1242.8 | 1224.7 | 1167.0 | 1031.0 | 848.0 | 84.1 | 0.0 |
| October | 0.0 | 58.8 | 271.0 | 549.6 | 818.2 | 988.7 | 1080.2 | 1096.6 | 1025.1 | 963.4 | 434.7 | 0.0 | 0.0 |
| November | 0.0 | 24.3 | 180.1 | 409.7 | 643.8 | 809.9 | 915.1 | 924.1 | 871.1 | 777.5 | 90.7 | 0.0 | 0.0 |
| December | 0.0 | 1.0 | 67.5 | 198.5 | 345.4 | 449.7 | 537.9 | 524.4 | 443.1 | 428.5 | 75.2 | 0.0 | 0.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cattani, L.; Cattani, P.; Magrini, A.; Figoni, R.; Dondi, D.; Vadivel, D. Suitability and Energy Sustainability of Atmospheric Water Generation Technology for Green Hydrogen Production. Energies 2023, 16, 6440. https://doi.org/10.3390/en16186440
Cattani L, Cattani P, Magrini A, Figoni R, Dondi D, Vadivel D. Suitability and Energy Sustainability of Atmospheric Water Generation Technology for Green Hydrogen Production. Energies. 2023; 16(18):6440. https://doi.org/10.3390/en16186440
Chicago/Turabian StyleCattani, Lucia, Paolo Cattani, Anna Magrini, Roberto Figoni, Daniele Dondi, and Dhanalakshmi Vadivel. 2023. "Suitability and Energy Sustainability of Atmospheric Water Generation Technology for Green Hydrogen Production" Energies 16, no. 18: 6440. https://doi.org/10.3390/en16186440
APA StyleCattani, L., Cattani, P., Magrini, A., Figoni, R., Dondi, D., & Vadivel, D. (2023). Suitability and Energy Sustainability of Atmospheric Water Generation Technology for Green Hydrogen Production. Energies, 16(18), 6440. https://doi.org/10.3390/en16186440











