Determination of 12 Combustion Products, Flame Temperature and Laminar Burning Velocity of Saudi LPG Using Numerical Methods Coded in a MATLAB Application
Abstract
:1. Introduction
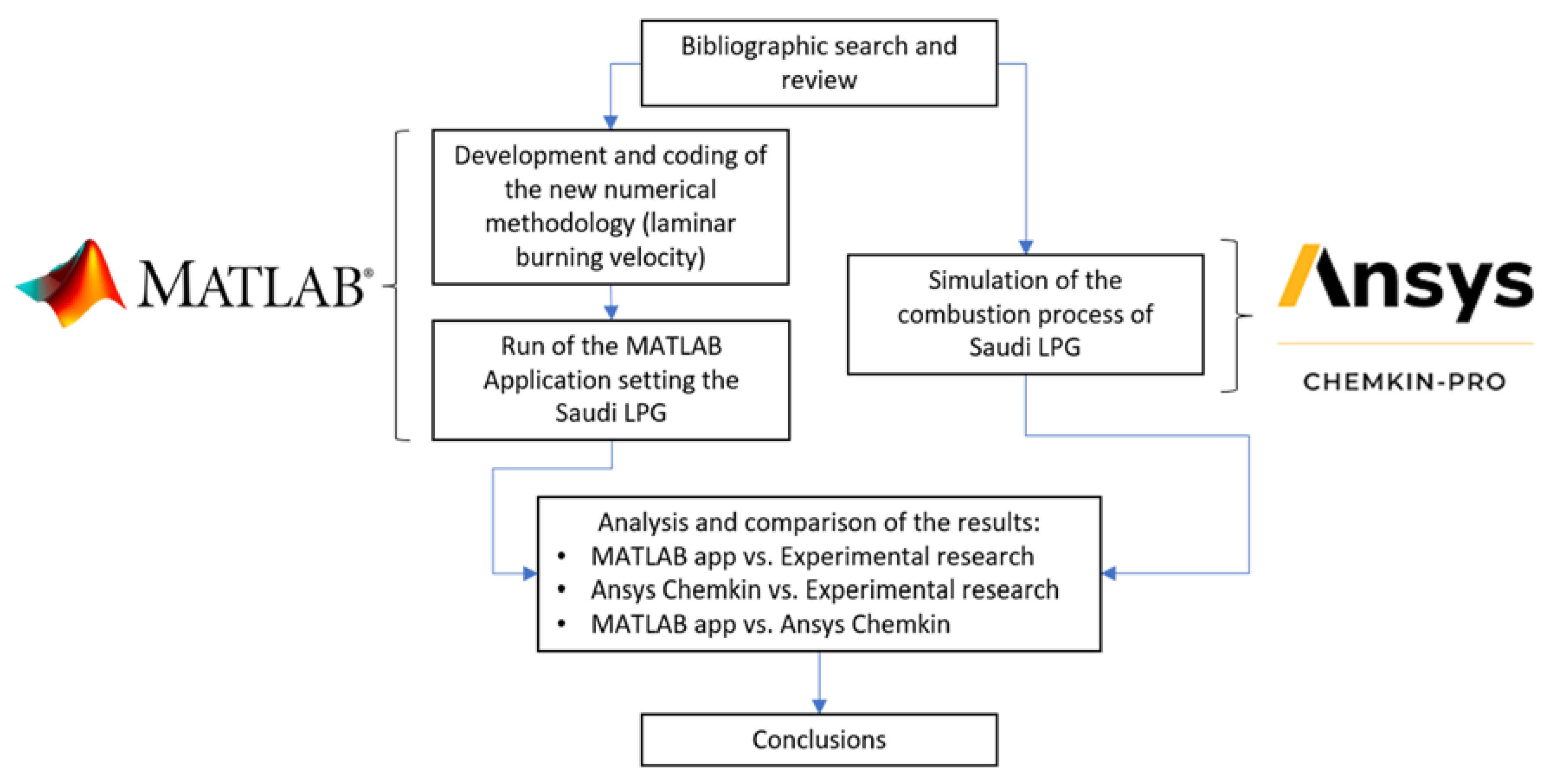
2. Materials and Methods
2.1. Materials and Procedure
2.2. Numerical Methodology for the Determination of Combustion Products and Flame Temperature
X1H + X2O + X3N + X4H2 + X5OH + X6CO + X7NO + X8O2 + X9H2O + X10CO2 + X11N2 + X12Ar
2.3. Determination of the Laminar Burning Velocity
2.4. Development of the MATLAB Application
2.4.1. First Part: New_Code.m
2.4.2. Second Part: Fractions_Derivatives.m
2.4.3. Third Part: Developing the MATLAB Application in the MATLAB App Designer Interface
2.5. Composition of Fuel and Mixtures to Be Tested
2.6. Simulation in Ansys Chemkin
3. Results and Discussion
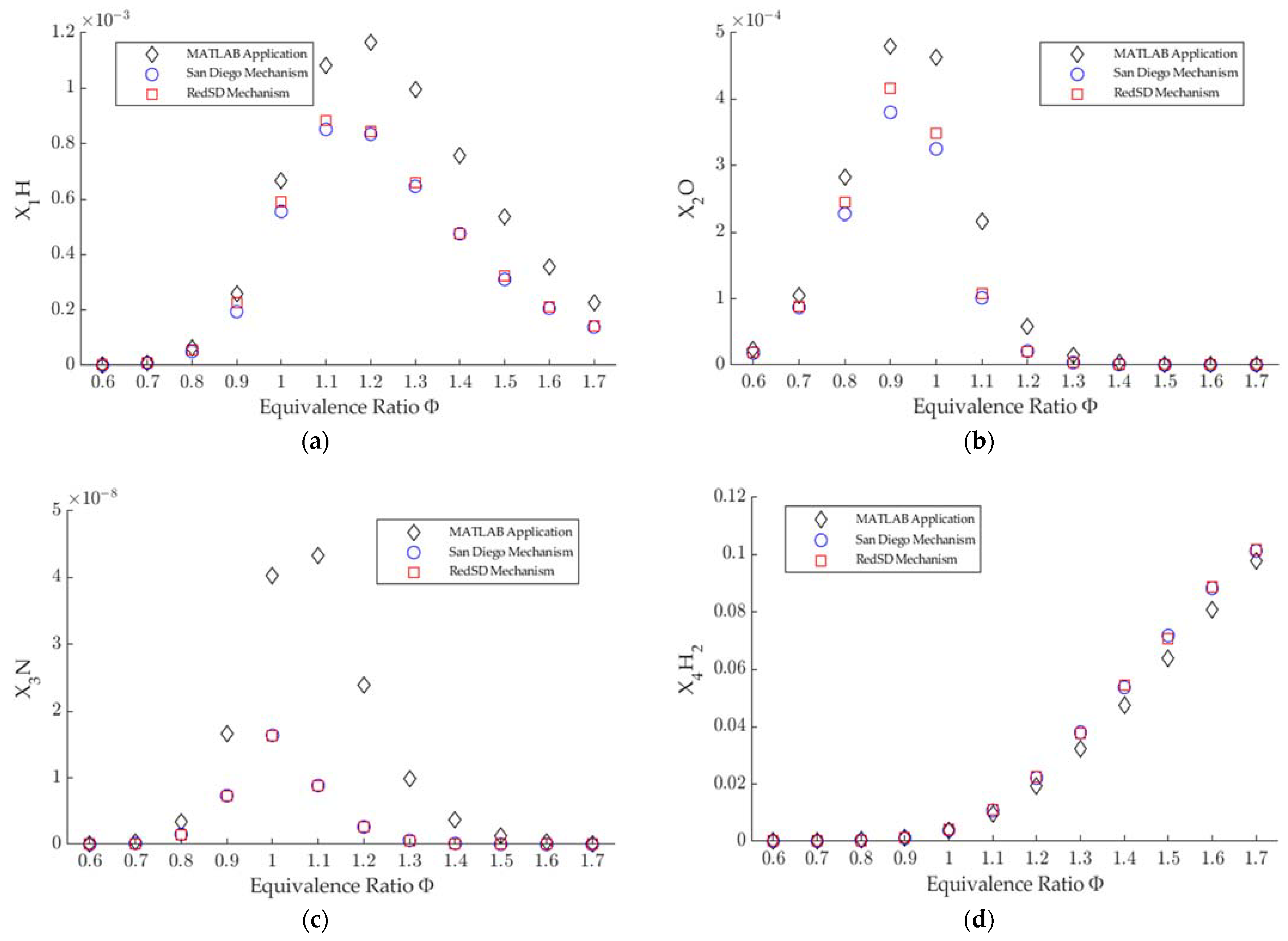
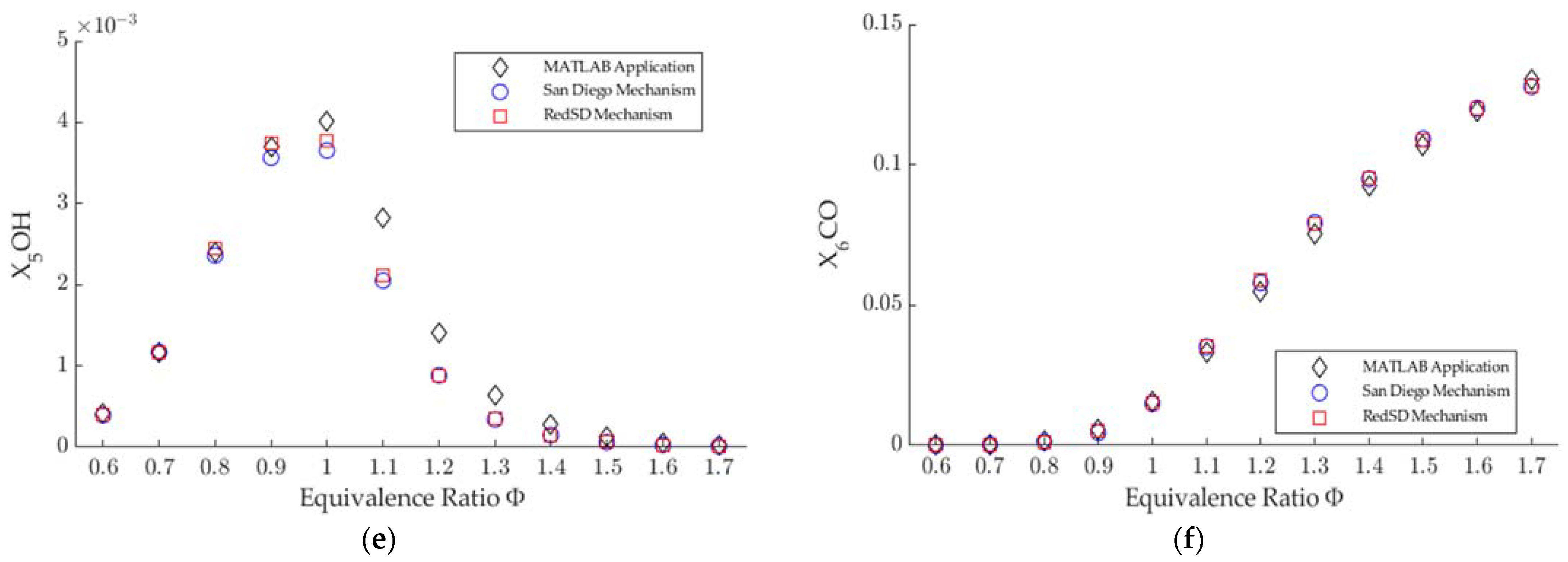
3.1. Combustion Products
3.2. Flame Temperature
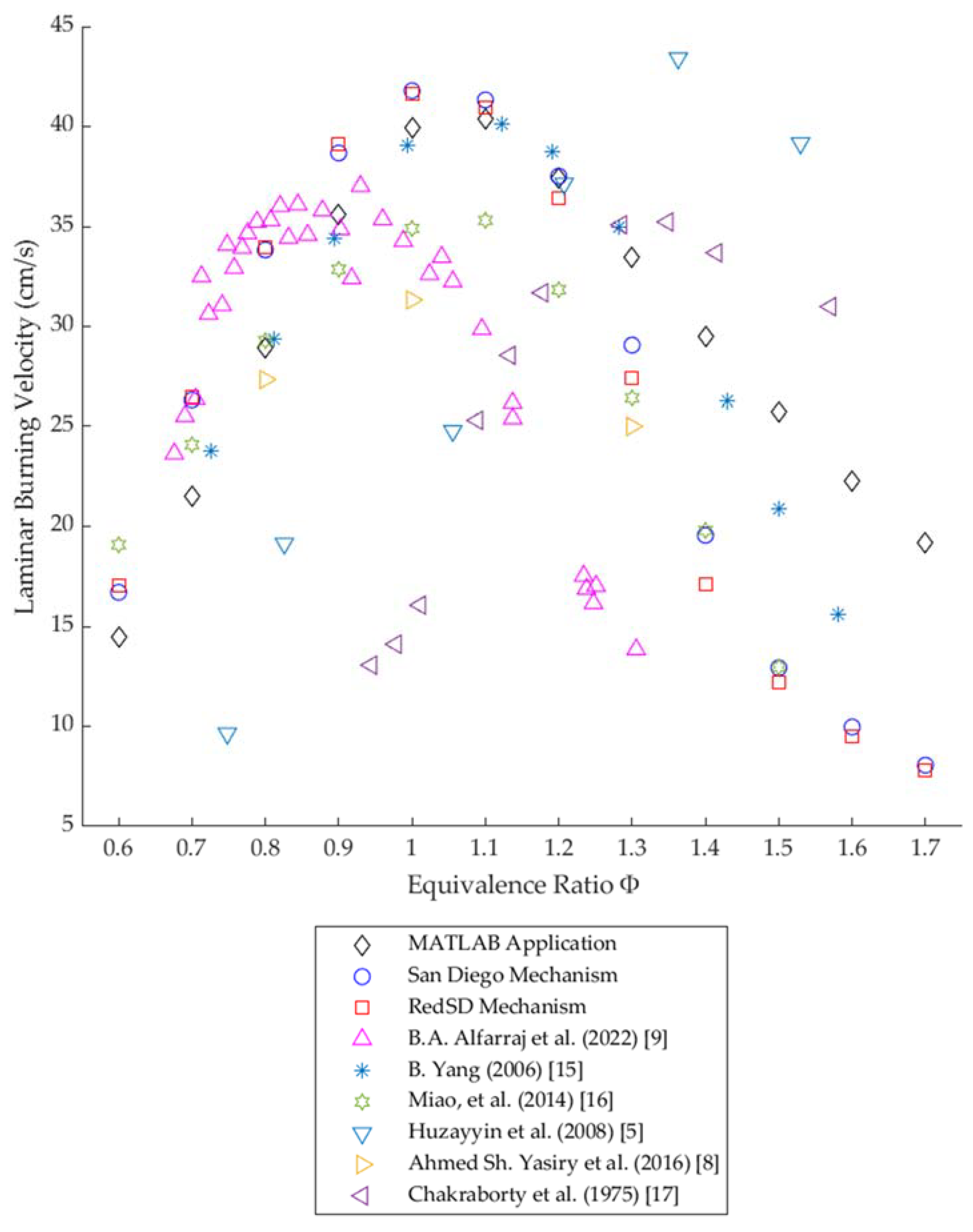
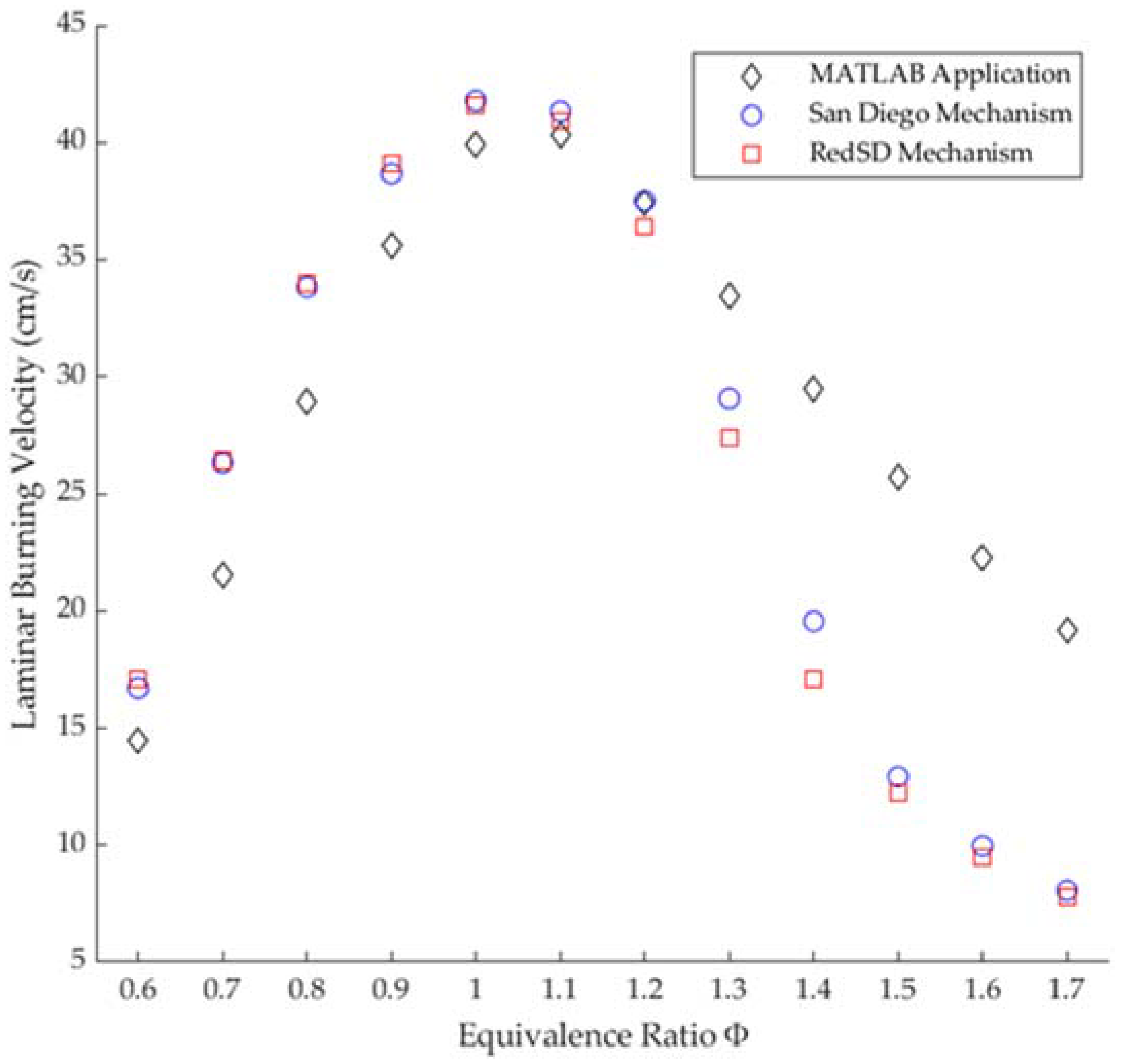
3.3. Laminar Burning Velocity
4. Conclusions
- For the laminar burning velocity results, the numerical method agrees with the experimental results for ratios (0.6–1.2) used by other authors and the simulation carried out in Ansys Chemkin, while, for the highest studied equivalence ratios (1.3–1.7) the laminar burning velocity results show a greater difference between all the resources.
- The numerical method used in the MATLAB application agrees with the simulation in Ansys Chemkin for the Saudi LPG combustion products, except for N and NO, for the whole range of equivalence ratios.
- The Saudi LPG maximum laminar burning velocity determined by the modified Bunsen burner method [9] was 35 ± 0.91 while that determined by the MATLAB application was 40.3 cm/s, having a difference of 5.35 ± 0.91 and an overestimate of 15.2% in favor of the MATLAB application.
- The Saudi LPG maximum laminar burning velocity determined by the MATLAB application was 40.3 cm/s, corresponding to a ratio of 1.1, with an underestimate of 2.3% with respect to the simulated values in Ansys Chemkin using the San Diego mechanism and an underestimate of 1.4% using the RedSD mechanism.
- The maximum adiabatic flame temperature of the Saudi LPG determined using the MATLAB application was 2326.2 K, corresponding to a ratio of 1.1, with an overestimate of 2.6% and 2.5% with respect to the simulated values in Ansys Chemkin using the San Diego and RedSD mechanisms, respectively.
- The MATLAB application, compared with previous experimental studies, presents the same behavioral results as those obtained by Miao et al. [16], B. Yang [15] and B.A. Alfarraj et al. (2022) [9] for lean mixture conditions. Meanwhile, for stoichiometric and fuel-rich conditions, it presents the same plot shape as that of B. Yang (2006) [15].
- The new code in the MATLAB application determined the experimental results more accurately, for equivalence ratios from 0.7 to 1.4, compared to Ansys Chemkin, taking B. Yang (2006) [15] as the experimental result reference.
- The MATLAB application has been developed for additional fuels such as methane, propane and natural gas and has the possibility of adding extra fuels, diluents and tools to improve the analysis of the results. It could be also applied to further studies using different kinds of mixtures.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| Φ | is the equivalence ratio; |
| n | is the carbon coefficient value of the fuel; |
| m | is the hydrogen coefficient value of the fuel; |
| l | is the oxygen coefficient value of the fuel; |
| k | is the nitrogen coefficient value of the fuel; |
| X1 | is the molar fraction of hydrogen (H) in the products; |
| X2 | is the molar fraction of oxygen (O) in the products; |
| X3 | is the molar fraction of nitrogen (N) in the products; |
| X4 | is the molar fraction of hydrogen (H2) in the products; |
| X5 | is the molar fraction of hydroxide (OH) in the products; |
| X6 | is the molar fraction of carbon monoxide (CO) in the products; |
| X7 | is the molar fraction of nitric oxide (NO) in the products; |
| X8 | is the molar fraction of oxygen (O2) in the products; |
| X9 | is the molar fraction of dihydrogen oxide (H2O) in the products; |
| X10 | is the molar fraction of carbon dioxide (CO2) in the products; |
| X11 | is the molar fraction of nitrogen (N2) in the products; |
| X12 | is the molar fraction of argon (Ar) in the products; |
| X13 | is the number of moles from fuel that give 1 mol of product; |
| Ki | is the partial pressure equilibrium constant of a chemical reaction; |
| p | is the pressure; |
| fj | is the equation system with 4 variables (X4, X6, X8 and X11) |
| Xi* | is the molar fraction real solution value used in Taylor series; |
| Xi(1) | is the molar fraction approximate value to the real one used in Taylor series; |
| ∆Xi | is the difference between the molar fraction real value and the approximate one; |
| ∂fj/∂Xi | is the equation system derivative with respect to molar fraction (X4, X6, X8 and X11); |
| Xi(2) | is the improved molar fraction value after the first iteration; |
| hr | is the reactant enthalpy; |
| h | is the product enthalpy; |
| po | is the initial pressure; |
| Fo | is the initial equivalence ratio; |
| To | is the initial temperature; |
| T | is the adiabatic flame temperature; |
| Tn | is the first assumed flame temperature (n = 1) or a current temperature iteration (n > 1); |
| Tn + 1 | is the improved temperature after an iteration using the Newton–Raphson method; |
| (∂h/∂T)n | is the enthalpy derivative with respect to temperature at n iterations; |
| M | is the molar mass of the mixture; |
| dhi/dT | is the specific heat at constant pressure of an element (i); |
| Cpi | is the specific heat at constant pressure of an element (i); |
| dXi/dT | is the partial derivative of a molar fraction with respect to temperature; |
| ∂M/∂T | is the molar mass of the mixture with respect to temperature; |
| SL | is the laminar burning velocity; |
| Ea | is the activation energy; |
| Ru | is the universal gas constant. |
References
- Khudhair, O.; Shahad, H.A.K. A Review of Laminar Burning Velocity and Flame Speed of Gases and Liquid Fuels. Int. J. Curr. Eng. Technol. 2017, 7, 183–197. [Google Scholar]
- Kuo, K.K. Principles of Combustion, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005; pp. 437–457. [Google Scholar]
- Samim, S.; Sadeq, A.M.; Ahmed, S.F. Measurements of Laminar Flame Speeds of Gas-to-Liquid-Diesel Fuel Blends. J. Energy Resour. Technol. 2016, 138, 052213. [Google Scholar] [CrossRef]
- Lee, K.; Ryu, J. An experimental study of the flame propagation and combustion characteristics of LPG fuel. Fuel 2005, 84, 1116–1127. [Google Scholar] [CrossRef]
- Huzayyin, A.S.; Moneib, H.A.; Shehatta, M.S.; Attia, A.M.A. Laminar burning velocity and explosion index of LPG-air and pro-pane-air mixtures. Fuel 2008, 87, 39–57. [Google Scholar] [CrossRef]
- Tripathi, A.; Chandra, H.; Agrawal, M. Effect of mixture constituents on the laminar burning velocity of LPG-Co2-air mixtures. J. Eng. Appl. Sci. (Asian Res. Publ. Netw.) 2010, 5, 16–21. [Google Scholar]
- Razus, D.; Brinzea, V.; Mitu, M.; Oancea, D. Burning Velocity of Liquefied Petroleum Gas (LPG)−Air Mixtures in the Presence of Exhaust Gas. Energy Fuels 2010, 24, 1487–1494. [Google Scholar] [CrossRef]
- Yasiry, A.S.; Shahad, H.A. An Experimental Study for Investigating the Laminar Flame Speed and Burning Velocity for LPG. Int. J. Therm. Technol. 2016, 6, 7–12. [Google Scholar]
- Alfarraj, B.A.; Al-Harbi, A.A.; Binjuwair, S.A.; Alkhedhair, A. The Characterization of Liquefied Petroleum Gas (LPG) Using a Modified Bunsen Burner. J. Combust. 2022, 2022, 1–9. [Google Scholar] [CrossRef]
- Chemical-Kinetic Mechanisms for Combustion Applications. San Diego Mechanism Web Page. Available online: https://web.eng.ucsd.edu/mae/groups/combustion/mechanism.html (accessed on 30 November 2022).
- Kumaran, S.M.; Shanmugasundaram, D.; Narayanaswamy, K.; Raghavan, V. Reduced mechanism for flames of propane, n-butane, and their mixtures for application to burners: Development and validation. Int. J. Chem. Kinet. 2021, 53, 731–750. [Google Scholar] [CrossRef]
- Olikara, C.; Borman, G. A Computer Program for Calculating Properties of Equilibrium Combustion Products with Some Applications to I.C. Engines. SAE Tech. Pap. 1975, 23. [Google Scholar] [CrossRef]
- Chase, M.W. JANAF Thermochemical Tables, 3rd ed.; American Chemical Society and American Institute of Physics: New York, NY, USA, 1986; pp. 1–1856. [Google Scholar]
- Markatou, P.; Pfefferle, L.D.; Smooke, M.D. A computational study of methane-air combustion over heated catalytic and non-catalytic surfaces. Combust. Flames 1993, 93, 185–201. [Google Scholar] [CrossRef]
- Yang, B. Laminar Burning Velocity of Liquefied Petroleum Gas Mixtures. Doctoral Thesis, Loughborough University, Loughborough, UK, 2005. [Google Scholar]
- Miao, J.; Leung, C.W.; Huang, Z.; Cheung, C.S.; Yu, H.; Xie, Y. Laminar burning velocities, Markstein lengths, and flame thickness of liquefied petroleum gas with hydrogen enrichment. Int. J. Hydrogen Energy 2014, 39, 13020–13030. [Google Scholar] [CrossRef]
- Chakraborty, S.K.; Mukhopadhyay, B.N.; Chanda, B.C. Effect of inhibitors on flammability range of flames produced from LPG/air mixtures. Fuel 1975, 54, 10–16. [Google Scholar] [CrossRef]
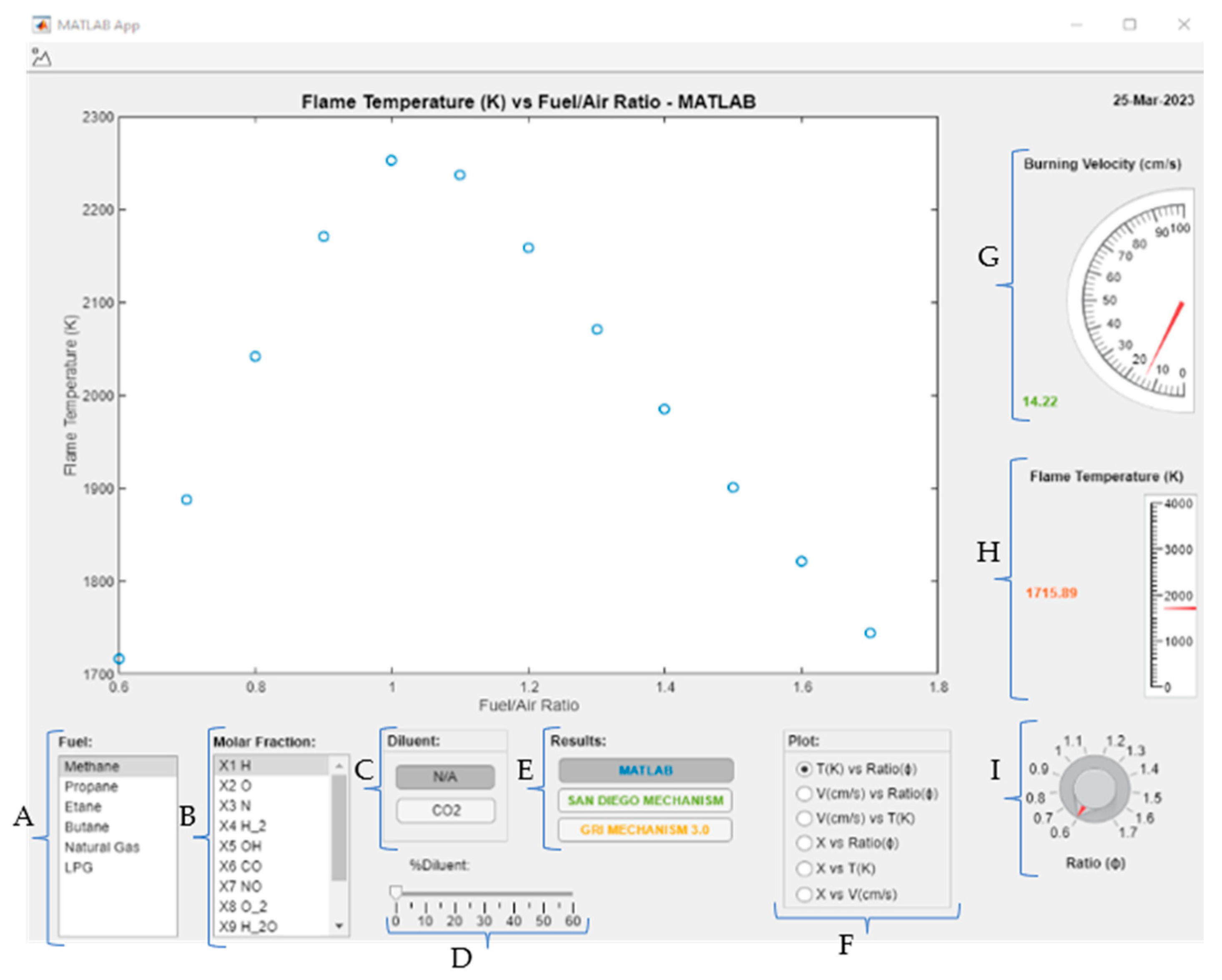





| Position in Figure 2 | Name of Tool | Description | Type of Selection |
|---|---|---|---|
| A | Fuel list | List of fuels available for calculus in the application | Unique |
| B | Molar fraction list | List of molar fractions available for plotting results | Unique |
| C | Diluent button list | List of diluents available to use in the calculus | Unique |
| D | Percentage bar of diluent | Percentage of diluent by volume to be considered in fuel | Unique |
| E | Results button list | Results from resources available to be shown in the plot | Multi |
| F | Plot button list | Type of plot to be shown on the screen | Unique |
| G | Equivalence ratio knob | Knob that shows the value of laminar burning velocity and flame temperature in their respective gauges | Unique |
| H | Laminar burning velocity gauge | Laminar burning velocity value at the knob equivalence ratio value selected | NA |
| I | Flame temperature gauge | Flame temperature value at the knob equivalence ratio value selected | NA |
| Equivalence Ratio (Φ) | X1H | X2O | X3N | X4H2 | X5OH | X6CO |
|---|---|---|---|---|---|---|
| 0.6 | 9.2644 × 10−7 | 2.3773 × 10−5 | 1.6175 × 10−11 | 1.2640 × 10−5 | 0.00041207 | 3.5819 × 10−5 |
| 0.7 | 1.0249 × 10−5 | 0.00010408 | 3.4726 × 10−10 | 8.5230 × 10−5 | 0.0011626 | 0.00028795 |
| 0.8 | 6.5024 × 10−5 | 0.00028288 | 3.4003 × 10−9 | 0.00039475 | 0.00240522 | 0.0015017 |
| 0.9 | 0.00025914 | 0.00048031 | 1.6695 × 10−8 | 0.00138214 | 0.00369457 | 0.00556398 |
| 1 | 0.00066624 | 0.00046349 | 4.0295 × 10−8 | 0.00393763 | 0.00401577 | 0.0154476 |
| 1.1 | 0.00108218 | 0.00021577 | 4.3271 × 10−8 | 0.00962653 | 0.00281794 | 0.0331991 |
| 1.2 | 0.00116468 | 5.8248 × 10−5 | 2.3838 × 10−8 | 0.0194133 | 0.00141014 | 0.0549746 |
| 1.3 | 0.00099712 | 1.3753 × 10−5 | 9.9015 × 10−9 | 0.0323431 | 0.00063771 | 0.075101 |
| 1.4 | 0.00075851 | 3.2136 × 10−6 | 3.6705 × 10−9 | 0.047353 | 0.00028167 | 0.0922573 |
| 1.5 | 0.00053636 | 7.5281 × 10−7 | 1.2757 × 10−9 | 0.0636388 | 0.00012302 | 0.106798 |
| 1.6 | 0.00035653 | 1.7192 × 10−7 | 4.1393 × 10−10 | 0.0806081 | 5.2459 × 10−5 | 0.119274 |
| 1.7 | 0.00022589 | 3.8207 × 10−8 | 1.2688 × 10−10 | 0.097815 | 2.1858 × 10−5 | 0.13018 |
| Equivalence Ratio (Φ) | X7NO | X8O2 | X9H2O | X10CO2 | X11N2 | X12AR |
|---|---|---|---|---|---|---|
| 0.6 | 0.00232901 | 0.0786287 | 0.0935573 | 0.0729014 | 0.743231 | 0.00886708 |
| 0.7 | 0.00353978 | 0.0575011 | 0.107843 | 0.084112 | 0.73656 | 0.00879482 |
| 0.8 | 0.00436595 | 0.0373029 | 0.121284 | 0.094098 | 0.729583 | 0.00871664 |
| 0.9 | 0.00425856 | 0.0196666 | 0.133438 | 0.100834 | 0.721799 | 0.00862329 |
| 1 | 0.00300759 | 0.0071197 | 0.143566 | 0.101098 | 0.712176 | 0.0085012 |
| 1.1 | 0.0013496 | 0.00141652 | 0.150092 | 0.0925431 | 0.699319 | 0.00833818 |
| 1.2 | 0.00044049 | 0.00019234 | 0.151587 | 0.0790268 | 0.683587 | 0.00814537 |
| 1.3 | 0.00013844 | 2.6966 × 10−5 | 0.148982 | 0.0665658 | 0.667245 | 0.0079489 |
| 1.4 | 4.4765 × 10−5 | 4.1781 × 10−6 | 0.143582 | 0.056652 | 0.651305 | 0.00775848 |
| 1.5 | 1.4838 × 10−5 | 6.9691 × 10−7 | 0.136333 | 0.0489914 | 0.635987 | 0.00757583 |
| 1.6 | 4.9101 × 10−6 | 1.1887 × 10−7 | 0.127913 | 0.0430682 | 0.621321 | 0.00740107 |
| 1.7 | 1.6125 × 10−6 | 2.0403 × 10−8 | 0.118823 | 0.0384122 | 0.607287 | 0.00723388 |
| Equivalence Ratio (Φ) | X1H | X2O | X3N | X4H2 | X5OH | X6CO |
|---|---|---|---|---|---|---|
| 0.6 | 6.6093 × 10−7 | 1.8192 × 10−5 | 3.5006 × 10−12 | 1.0017 × 10−5 | 0.00039345 | 2.7661 × 10−5 |
| 0.7 | 8.3166 × 10−6 | 8.693 × 10−5 | 1.3135 × 10−10 | 7.4804 × 10−5 | 0.00117059 | 0.00024538 |
| 0.8 | 4.9654 × 10−5 | 0.00022725 | 1.4973 × 10−9 | 0.00033309 | 0.00236175 | 0.00123292 |
| 0.9 | 0.00019441 | 0.00038015 | 7.3197 × 10−9 | 0.00114976 | 0.00356367 | 0.00454945 |
| 1 | 0.00055523 | 0.00032513 | 1.6402 × 10−8 | 0.00388401 | 0.00365408 | 0.0149326 |
| 1.1 | 0.00085203 | 0.00010131 | 8.8505 × 10−9 | 0.0107692 | 0.00205185 | 0.0351553 |
| 1.2 | 0.00083450 | 2.1024 × 10−5 | 2.6195 × 10−9 | 0.0221748 | 0.00088483 | 0.058047 |
| 1.3 | 0.00064603 | 3.7060 × 10−6 | 5.9878 × 10−10 | 0.0380662 | 0.00034043 | 0.0792958 |
| 1.4 | 0.00047560 | 8.6550 × 10−7 | 1.3136 × 10−10 | 0.0536471 | 0.00014997 | 0.0948882 |
| 1.5 | 0.00031061 | 1.7192 × 10−7 | 1.7254 × 10−11 | 0.0717481 | 6.0068 × 10−5 | 0.109207 |
| 1.6 | 0.00020575 | 4.1360 × 10−8 | 1.9497 × 10−12 | 0.0882793 | 2.6349 × 10−5 | 0.120062 |
| 1.7 | 0.00013802 | 1.1794 × 10−8 | 2.8318 × 10−13 | 0.101292 | 1.2937 × 10−5 | 0.12784 |
| Equivalence Ratio (Φ) | X7NO | X8O2 | X9H2O | X10CO2 | X11N2 | X12AR |
|---|---|---|---|---|---|---|
| 0.6 | 1.2659 × 10−6 | 0.0778787 | 0.0957267 | 0.0734329 | 0.0743441 | 0.00906312 |
| 0.7 | 1.0758 × 10−5 | 0.0564053 | 0.111239 | 0.0849199 | 0.73685 | 0.00898358 |
| 0.8 | 7.4972 × 10−5 | 0.0364389 | 0.124853 | 0.0951511 | 0.730366 | 0.00890481 |
| 0.9 | 0.00028322 | 0.0190562 | 0.136546 | 0.102239 | 0.723214 | 0.00881881 |
| 1 | 0.00036887 | 0.00517981 | 0.147765 | 0.102416 | 0.712228 | 0.00868567 |
| 1.1 | 8.0115 × 10−5 | 0.00060559 | 0.15336 | 0.0911168 | 0.697399 | 0.00850336 |
| 1.2 | 1.8270 × 10−5 | 6.1474 × 10−5 | 0.153406 | 0.076219 | 0.680035 | 0.0082915 |
| 1.3 | 3.9921 × 10−6 | 6.2188 × 10−6 | 0.149864 | 0.0628266 | 0.660878 | 0.00805869 |
| 1.4 | 7.1151 × 10−7 | 9.9641 × 10−7 | 0.143506 | 0.0540122 | 0.645431 | 0.00786988 |
| 1.5 | 8.0017 × 10−8 | 1.4057 × 10−7 | 0.135334 | 0.0466537 | 0.628933 | 0.00766906 |
| 1.6 | 2.5797 × 10−8 | 2.5968 × 10−8 | 0.127036 | 0.0415919 | 0.614249 | 0.00749027 |
| 1.7 | 1.1576 × 10−8 | 6.0938 × 10−9 | 0.119289 | 0.0384194 | 0.602665 | 0.00734881 |
| Equivalence Ratio (Φ) | X1H | X2O | X3N | X4H2 | X5OH | X6CO |
|---|---|---|---|---|---|---|
| 0.6 | 6.9492 × 10−7 | 1.8833 × 10−5 | 3.5006 × 10−12 | 1.0326 × 10−5 | 0.00039894 | 2.8759 × 10−5 |
| 0.7 | 8.4643 × 10−6 | 8.8159 × 10−5 | 1.3135 × 10−10 | 7.5337 × 10−5 | 0.00117376 | 0.00024771 |
| 0.8 | 5.5236 × 10−5 | 0.00024429 | 1.4973 × 10−9 | 0.00035698 | 0.00244458 | 0.00132638 |
| 0.9 | 0.00022744 | 0.00041572 | 7.3197 × 10−9 | 0.0012917 | 0.00374339 | 0.00510691 |
| 1 | 0.00059085 | 0.00034823 | 1.6402 × 10−8 | 0.00399172 | 0.00377842 | 0.015321 |
| 1.1 | 0.00088504 | 0.00010743 | 8.8505 × 10−9 | 0.0108747 | 0.00211538 | 0.0354616 |
| 1.2 | 0.00084480 | 2.0644 × 10−5 | 2.6195 × 10−9 | 0.0226464 | 0.00087630 | 0.0588178 |
| 1.3 | 0.00066097 | 3.9565 × 10−6 | 5.9878 × 10−10 | 0.0377389 | 0.00035226 | 0.0789434 |
| 1.4 | 0.00047605 | 8.4477 × 10−7 | 1.3136 × 10−10 | 0.0542954 | 0.00014795 | 0.0952242 |
| 1.5 | 0.00032336 | 1.9393 × 10−7 | 1.7254 × 10−11 | 0.0706793 | 6.4263 × 10−5 | 0.108435 |
| 1.6 | 0.00021240 | 4.3565 × 10−8 | 1.9497 × 10−12 | 0.0887154 | 2.7351 × 10−5 | 0.120091 |
| 1.7 | 0.00014340 | 1.2649 × 10−8 | 2.8318 × 10−13 | 0.101579 | 1.3416 × 10−5 | 0.127924 |
| Equivalence Ratio (Φ) | X7NO | X8O2 | X9H2O | X10CO2 | X11N2 | X12AR |
|---|---|---|---|---|---|---|
| 0.6 | 1.2659 × 10−6 | 0.0778787 | 0.0957267 | 0.0734329 | 0.0743441 | 0.00906312 |
| 0.7 | 1.0758 × 10−5 | 0.0564053 | 0.111239 | 0.0849199 | 0.73685 | 0.00898358 |
| 0.8 | 7.4972 × 10−5 | 0.0364389 | 0.124853 | 0.0951511 | 0.730366 | 0.00890481 |
| 0.9 | 0.00028322 | 0.0190562 | 0.136546 | 0.102239 | 0.723214 | 0.00881881 |
| 1 | 0.00036887 | 0.00517981 | 0.147765 | 0.102416 | 0.712228 | 0.00868567 |
| 1.1 | 8.0115 × 10−5 | 0.00060559 | 0.15336 | 0.0911168 | 0.697399 | 0.00850336 |
| 1.2 | 1.8270 × 10−5 | 6.1474 × 10−5 | 0.153406 | 0.076219 | 0.680035 | 0.0082915 |
| 1.3 | 3.9921 × 10−6 | 6.2188 × 10−6 | 0.149864 | 0.0628266 | 0.660878 | 0.00805869 |
| 1.4 | 7.1151 × 10−7 | 9.9641 × 10−7 | 0.143506 | 0.0540122 | 0.645431 | 0.00786988 |
| 1.5 | 8.0017 × 10−8 | 1.4057 × 10−7 | 0.135334 | 0.0466537 | 0.628933 | 0.00766906 |
| 1.6 | 2.5797 × 10−8 | 2.5968 × 10−8 | 0.127036 | 0.0415919 | 0.614249 | 0.00749027 |
| 1.7 | 1.1576 × 10−8 | 6.0938 × 10−9 | 0.119289 | 0.0384194 | 0.602665 | 0.00734881 |
| Equivalence Ratio (Φ) | MATLAB Application T (K) | San Diego Mechanism T (K) | RedSD Mechanism T (K) |
|---|---|---|---|
| 0.6 | 1763.1 | 1723.7 | 1721.8 |
| 0.7 | 1945.7 | 1914.3 | 1911.5 |
| 0.8 | 2108.1 | 2076.8 | 2075.6 |
| 0.9 | 2238.6 | 2202.8 | 2206.0 |
| 1 | 2318.5 | 2283.9 | 2283.3 |
| 1.1 | 2326.2 | 2267.3 | 2268.9 |
| 1.2 | 2272.7 | 2198.0 | 2196.9 |
| 1.3 | 2197.8 | 2109.6 | 2111.1 |
| 1.4 | 2118.8 | 2033.4 | 2031.4 |
| 1.5 | 2040.1 | 1956.1 | 1961.1 |
| 1.6 | 1963.1 | 1889.2 | 1889.0 |
| 1.7 | 1888.0 | 1831.0 | 1831.7 |
| Equivalence Ratio (Φ) | MATLAB Application—San Diego Mechanism | MATLAB Application—RedSD Mechanism | ||
|---|---|---|---|---|
| ∆T (K) | % | ∆T (K) | % | |
| 0.6 | 39.4 | 2.2 | 41.2 | 2.4 |
| 0.7 | 31.3 | 1.6 | 34.1 | 1.7 |
| 0.8 | 31.2 | 1.5 | 32.4 | 1.5 |
| 0.9 | 35.8 | 1.6 | 32.6 | 1.4 |
| 1 | 34.5 | 1.5 | 35.1 | 1.5 |
| 1.1 | 58.5 | 2.6 | 57.3 | 2.5 |
| 1.2 | 74.7 | 3.4 | 75.8 | 3.4 |
| 1.3 | 88.1 | 4.1 | 86.7 | 4.1 |
| 1.4 | 85.4 | 4.2 | 87.4 | 4.3 |
| 1.5 | 84.0 | 4.3 | 79.0 | 4.0 |
| 1.6 | 73.8 | 3.9 | 74.0 | 3.9 |
| 1.7 | 57.0 | 3.1 | 56.3 | 3.0 |
| Equivalence Ratio (Φ) | MATLAB Application SL (cm/s) | San Diego Mechanism SL (cm/s) | RedSD Mechanism SL (cm/s) |
|---|---|---|---|
| 0.6 | 14.4 | 16.7 | 17.0 |
| 0.7 | 21.5 | 26.3 | 26.4 |
| 0.8 | 28.9 | 33.8 | 33.9 |
| 0.9 | 35.5 | 38.6 | 39.1 |
| 1 | 39.9 | 41.7 | 41.6 |
| 1.1 | 40.3 | 41.3 | 40.9 |
| 1.2 | 37.4 | 37.5 | 36.4 |
| 1.3 | 33.4 | 29.0 | 27.3 |
| 1.4 | 29.4 | 19.5 | 17.1 |
| 1.5 | 25.7 | 12.9 | 12.2 |
| 1.6 | 22.2 | 9.9 | 9.5 |
| 1.7 | 19.1 | 8.0 | 7.7 |
| Equivalence Ratio (Φ) | MATLAB Application—San Diego Mechanism | MATLAB Application—RedSD Mechanism | ||
|---|---|---|---|---|
| ∆SL (cm/s) | % | ∆SL (cm/s) | % | |
| 0.6 | −2.2 | −13.3 | −2.5 | −15.1 |
| 0.7 | −4.7 | −18.1 | −4.9 | −18.6 |
| 0.8 | −4.8 | −14.4 | −5.0 | −14.8 |
| 0.9 | −3.0 | −7.9 | −3.5 | −9.0 |
| 1 | −1.8 | −4.4 | −1.7 | −4.0 |
| 1.1 | −0.9 | −2.3 | −0.5 | −1.4 |
| 1.2 | −0.1 | −0.2 | 0.9 | 2.6 |
| 1.3 | 4.3 | 15.0 | 6.0 | 22.1 |
| 1.4 | 9.9 | 50.7 | 12.3 | 72.3 |
| 1.5 | 12.8 | 99.0 | 13.5 | 110.6 |
| 1.6 | 12.3 | 123.5 | 12.7 | 134.6 |
| 1.7 | 11.1 | 137.7 | 11.3 | 145.9 |
| Work | Type of Study | Pressure (atm) | Initial Temperature (K) | LPG Composition | |||
|---|---|---|---|---|---|---|---|
| Propane C3H8 | Butane C4H10 | Ethane C2H6 | Pentane C5H12 | ||||
| MATLAB Application—Current Work | Numerical methodology in MATLAB | 1 | 298.15 | 50% | 50% | 0% | 0% |
| San Diego Mechanism—Current Work | Numerical Simulation in Ansys Chemkin | 1 | 298.15 | 50% | 50% | 0% | 0% |
| RedSD Mechanism—Current Work | Numerical Simulation in Ansys Chemkin | 1 | 298.15 | 50% | 50% | 0% | 0% |
| B.A. Alfarraj et al. [9] | Experimental—Modified Bunsen Burner Method | 1 | 298.15 | 50% | 50% | 0% | 0% |
| B. Yang [15] | Experimental—Constant Volume Bomb Method | 1 | 298.15 | 50% | 50% | 0% | 0% |
| Huzayyin et al. [5] | Experimental—Constant Volume Chamber Method | 1 | 294 ± 3 | 26.41% | 73.54% | 0.04% | 0% |
| Ahmed Sh. Yasiry et al. [8] | Experimental—Constant Volume Chamber Method | 1 | 308 | 36.3% | 62.3% | 0.9% | 0.5% |
| Miao et al. [16] | Experimental—Constant Volume Chamber Method | 1 | 298.15 | 30% | 70% | 0% | 0% |
| Chakraborty et al. [17] | Experimental—Flat Flame Burner Method | 1 | 298 | 30.1% | 67.7% | 1.4% | 0% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cisneros, R.F.; Rojas, F.J. Determination of 12 Combustion Products, Flame Temperature and Laminar Burning Velocity of Saudi LPG Using Numerical Methods Coded in a MATLAB Application. Energies 2023, 16, 4688. https://doi.org/10.3390/en16124688
Cisneros RF, Rojas FJ. Determination of 12 Combustion Products, Flame Temperature and Laminar Burning Velocity of Saudi LPG Using Numerical Methods Coded in a MATLAB Application. Energies. 2023; 16(12):4688. https://doi.org/10.3390/en16124688
Chicago/Turabian StyleCisneros, Roberto Franco, and Freddy Jesús Rojas. 2023. "Determination of 12 Combustion Products, Flame Temperature and Laminar Burning Velocity of Saudi LPG Using Numerical Methods Coded in a MATLAB Application" Energies 16, no. 12: 4688. https://doi.org/10.3390/en16124688
APA StyleCisneros, R. F., & Rojas, F. J. (2023). Determination of 12 Combustion Products, Flame Temperature and Laminar Burning Velocity of Saudi LPG Using Numerical Methods Coded in a MATLAB Application. Energies, 16(12), 4688. https://doi.org/10.3390/en16124688