The Oxygenic Photogranules—Current Progress on the Technology and Perspectives in Wastewater Treatment: A Review
Abstract
1. Introduction
2. Types of Biogranules
| Methanogenic Granules (MGs) | Hydrogenic Granules (HGs) | Anammox Granules | |
|---|---|---|---|
| Date of discovery | 1976 by Gatze Lettinga’s group | - | 1990s in a wastewater pilot plant at Delft University of Technology |
| Start-up period | 2–8 months | A few months; the time of formation of granules may be shorter when acclimatised seed sludge will be acid incubated for 24 h after lowering the pH from 5.5 to 2.0 in an anaerobic CSTR; in this condition, the granules may be formed rapidly within 3–5 days. | 14–800 days |
| Size /diameter, mm | 0.14–5.0 | 0.4–3.5 | 1.75–4.0 Rare > 6.0 mm; not recommended because when granule size larger than 2.2 m may decrease nitrogen removal and cause granule flotation |
| Color | black | changed from black to white (or creamy), | carmine |
| Structure and microbiome | multi-layered structure The inner layer mainly consists of acetoclastic methanogens such as Methanosaeta sp., while the middle layer consists of hydrogenotrophic methanogens; in turn the outer layer consists of Hydrogen producing bacteria, hydrogenotrophic methanogens, Sulfate reducing bacteria | non-layered structure mainly: Clostridium sp., while Klebsiella and Enterobacter were also detected | Two- -layered structure consists outer aerobic layer containing ammonia oxidising bacteria (AOB) and an anoxic core of anammox micro-organisms (AMX bacteria) include members of the Proteobacteria, Chlorobi, Bacteroidetes, and Chloroflexi phyla; most commonly were isolated: Candidatus Brocadia Candidatus, Kuenenia, Candidatus Jettenia |
| settling velocity, m/h | 18–50 | Up to 75 | 35–160 |
| the porosity | 0.64–0.90 | high | - |
| seeding source for biogranules formation | Flocculated sludge, rare: MGs, inoculum requires pretreatment | Anammox granules, Anaerobic granules, MGs, Activated Sludge, Nitrifying and anammox sludge, Inactive methanogenic granules | |
| Potential role at WWTPs | decomposition of complex organic substances to methane mainly in the upflow anaerobic sludge blanket (UASB) and expanded granular sludge blanket (EGSB) | anaerobic hydrogen production from organic wastes; production of a new biofuel, namely the hythane gas (consists of 10%–30% v/v of hydrogen and 70%–90% v/v of methane) | For biological nitrogen removal (BNR), mainly for treatment of ammonium-rich wastewater (up to 2.5 kg N-NH4+/(m3·d) |
3. Cultivation Methods, Structure, and Formation of OPGs
3.1. Cultivation Methods
3.2. Structure and Formation
- An initial contact between bacteria which is achieved by physical movement and further adhesion, which is the requirement for building the stable biofilm structure;
- Influence of the attractive physical and chemical forces that further maintain the contact between cells;
- Aggregation of cells as well as maturation (microbial forces);
- Influence of hydrodynamic shear force that shapes the structure of the final granule.
4. Factors That Influence on Formation of OPGs
4.1. TSS and Inoculum Concentration
4.2. Presence of Cyanobacteria of Oscillatoria spp.
4.3. N-NH4+ Concentration
4.4. Hydrodynamic Shear Force/Mixing Conditions
4.5. Temperature
4.6. Light Intensity
4.7. Condition of Cultivation
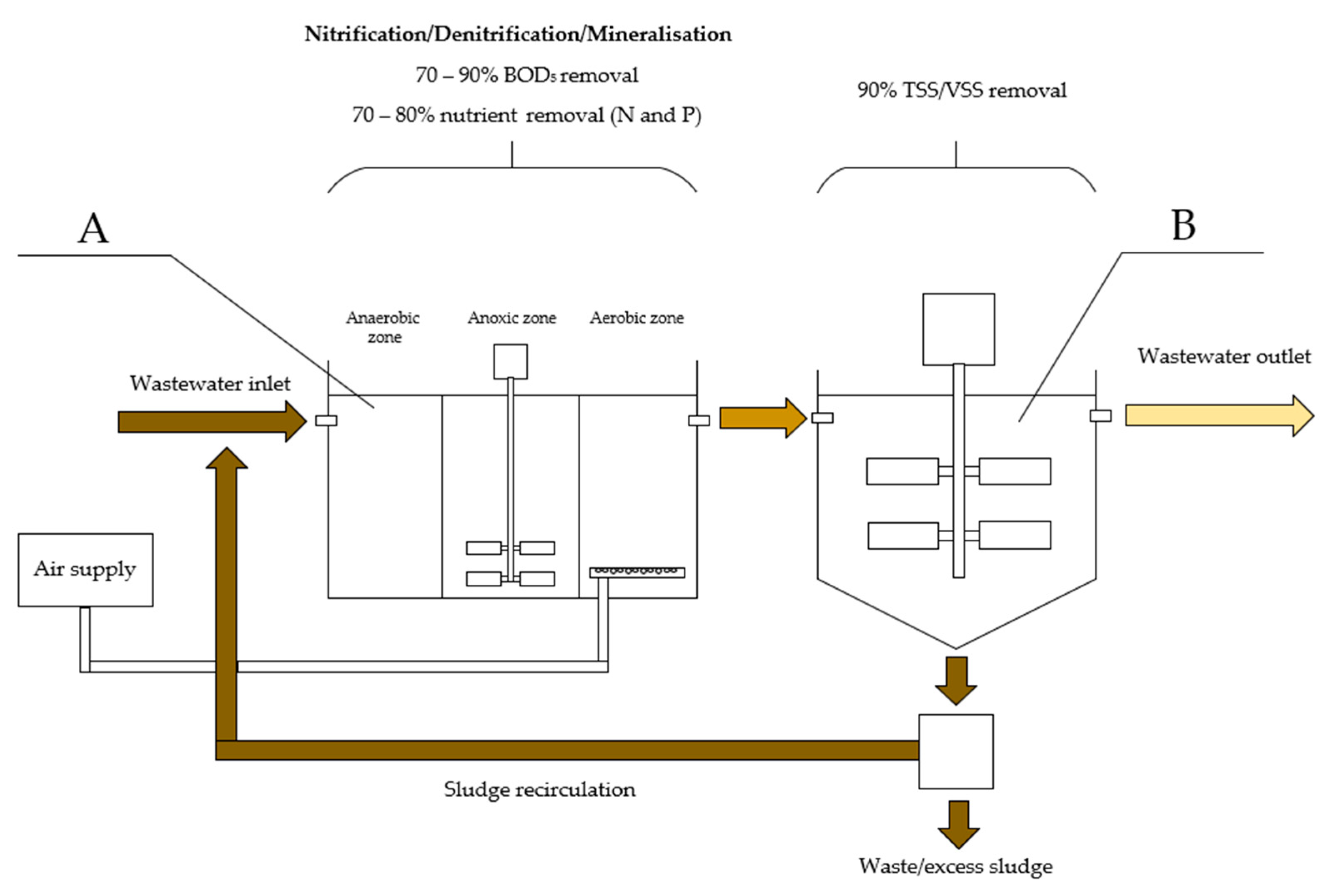
5. Application of OPGs for Wastewater Treatment
6. Conclusions and Potential Future Development Directions
- Excellent settling velocity which allows easy separation of biomass from treated water;
- Better COD and nutrient removal efficiencies compared to CAS;
- In situ oxygen production coupled with denitrification, so mechanical aeration, which characterises CAS is not required;
- Generation of autotrophically rich biomass that may be used as a source of renewable energy.
- Scale-up, most research to date has been conducted at very small laboratory scale using synthetic wastewater under ideal or well controlled conditions;
- Impact of long-term process application on effluent quality;
- Adaptation of the OPGs production cycle to the natural diurnal cycle, weather conditions explore issues relating to symbiosis between bacteria and algae, particularly in the context of energy storage in the form of lipid, poly-P and glycogen;
- Developing new bioreactors and solutions for the cultivation of OPGs in order to ensure the transmission and penetration of light appropriate to their growth, also, include the stability of OPGs;
- The potential of OPGs for the removal of emerging contaminants;
- The reuse of the produced biomass of OPGs, including practical work on its conversion into biofuels and its thermal disposal, as well as the possibility of recovering energy from them through anaerobic digestion. The high methane potential of OPGs, estimated to be up to 20% higher than that of AS, argues in favor of directing research in this direction; to the best of the authors’ knowledge, research in this direction has not yet been carried out;
- Research into technologies for converting biogranules into value-added products.
Author Contributions
Funding
Conflicts of Interest
References
- Gitau, J.G. Scaling up the Oxygenic Photogranule (opg) Wastewater Treatment Process. 2020, Doctoral Dissertations. 1918. Available online: https://scholarworks.umass.edu/dissertations_2/1918 (accessed on 29 November 2022). [CrossRef]
- Nancharaiah, Y.V.; Sarvajith, M.; Mohan, T.V.K. Aerobic granular sludge: The future of wastewater treatment. Curr. Sci. 2019, 117, 395–404. [Google Scholar] [CrossRef]
- Brockmann, D.; Gérand, Y.; Park, C.; Milferstedt, K.; Hélias, A.; Hamelin, J. Wastewater treatment using oxygenic photogranule-based process has lower environmental impact than conventional activated sludge process. Bioresour. Technol. 2021, 319, 124204. [Google Scholar] [CrossRef] [PubMed]
- Downes, S. The Success, Morphology, and Performance of Oxygenic Photogranules Under Light-Induced Stress Conditions. 2019, Environmental & Water Resources Engineering Masters Projects. Available online: https://scholarworks.umass.edu/cee_ewre/96 (accessed on 29 November 2022).
- Siatou, A.; Manali, A.; Gikas, P. Energy consumption and internal distribution in activated sludge wastewater treatment plants of Greece. Water 2020, 12, 1204. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y. Resource recovery from municipal wastewater: A critical paradigm shift in the post era of activated sludge. Bioresour. Technol. 2022, 363, 127932. [Google Scholar] [CrossRef]
- de Bruin, L.M.M.; de Kreuk, M.K.; van der Roest, H.F.R.; Uijterlinde, C.; van Loosdrecht, M.C.M. Aerobic granular sludge technology: An alternative to activated sludge? Water Sci. Technol. 2004, 49, 1–7. [Google Scholar] [CrossRef]
- Joel, C.; Kiprop, E.; Mwamburi, L. Effect of Seasonal Variation on Performance of Conventional Wastewater Treatment System. J. Appl. Environ. Microbiol. 2017, 5, 1–7. [Google Scholar] [CrossRef]
- Muhammed, A.; Poduval, A.N.; Oonnikrishnan, P.; Narayanan, P.K.; Yaduraj, K. The oxygenic photogranule for wastewater treatment process. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1114, 012090. [Google Scholar] [CrossRef]
- Yu, R.; Yang, Y.; Zhou, Z.; Li, X.; Liu, C.; Wang, N.; Liu, Y. Attribution of photocatalysis of fluorescent natural organic matter fractions to the alleviation of ceramic membrane ultrafiltration fouling. Sep. Purif. Technol. 2023, 307, 122603. [Google Scholar] [CrossRef]
- Ren, L.; Kong, X.; Su, J.; Zhao, D.; Dong, W.; Liu, C.; Liu, C.; Luo, L.; Yan, B. Oriented conversion of agricultural bio-waste to value-added products-A schematic review towards key nutrient circulation. Bioresour. Technol. 2022, 346, 126578. [Google Scholar] [CrossRef]
- Li, H.; Yu, J.; Gong, Y.; Lin, N.; Yang, Q.; Zhang, X.; Wang, Y. Perovskite catalysts with different dimensionalities for environmental and energy applications: A review. Sep. Purif. Technol. 2023, 307, 122716. [Google Scholar] [CrossRef]
- Wang, Y.; Gong, Y.; Lin, N.; Yu, L.; Du, B.; Zhang, X. Enhanced removal of Cr(VI) from aqueous solution by stabilized nanoscale zero valent iron and copper bimetal intercalated montmorillonite. J. Colloid Interface Sci. 2022, 606, 941–952. [Google Scholar] [CrossRef]
- Ren, X.; Shanb Ghazani, M.; Zhu, H.; Ao, W.; Zhang, H.; Moreside, E.; Zhu, J.; Yang, P.; Zhong, N.; Bi, X. Challenges and opportunities in microwave-assisted catalytic pyrolysis of biomass: A review. Appl. Energy 2022, 315, 118970. [Google Scholar] [CrossRef]
- Wu, Y.; Kong, L.-H.; Ge, W.-T.; Zhang, W.-J.; Dong, Z.-Y.; Guo, X.-J.; Yan, X.; Chen, Y.; Lang, W.-Z. A porous V/SiO2 sphere composite for the selective oxidation of benzyl alcohol to benzaldehyde in aqueous phase through peroxymonosulfate activation. J. Catal. 2022, 413, 668–680. [Google Scholar] [CrossRef]
- Rout, P.R.; Zhang, T.C.; Bhunia, P.; Surampalli, R.Y. Treatment technologies for emerging contaminants in wastewater treatment plants: A review. Sci. Total Environ. 2021, 753, 141990. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, J.; Liu, H.; Guo, X.; Zhang, X.; Yao, X.; Cao, Z.; Zhang, T. A review of the removal of microplastics in global wastewater treatment plants: Characteristics and mechanisms. Environ. Int. 2021, 146, 106277. [Google Scholar] [CrossRef] [PubMed]
- Partyka, M.L.; Bond, R.F. Wastewater reuse for irrigation of produce: A review of research, regulations, and risks. Sci. Total Environ. 2022, 828, 154385. [Google Scholar] [CrossRef]
- Preisner, M.; Neverova-Dziopak, E.; Kowalewski, Z. An Analytical Review of Different Approaches to Wastewater Discharge Standards with Particular Emphasis on Nutrients. Environ. Manag. 2020, 66, 694–708. [Google Scholar] [CrossRef]
- Franca, R.D.G.; Pinheiro, H.M.; van Loosdrecht, M.C.M.; Lourenço, N.D. Stability of aerobic granules during long-term bioreactor operation. Biotechnol. Adv. 2018, 36, 228–246. [Google Scholar] [CrossRef]
- Mainardis, M.; Buttazzoni, M.; Goi, D. Up-flow anaerobic sludge blanket (Uasb) technology for energy recovery: A review on state-of-the-art and recent technological advances. Bioengineering 2020, 7, 43. [Google Scholar] [CrossRef]
- Cho, S.; Kambey, C.; Nguyen, V.K. Performance of Anammox Processes for Wastewater Treatment: A Critical Review on Effects of Operational Conditions and Environmental Stresses. Water 2020, 12, 20. [Google Scholar] [CrossRef]
- Milferstedt, K.; Hamelin, J.; Park, C.; Jung, J.; Hwang, Y.; Cho, S.K.; Jung, K.W.; Kim, D.H. Biogranules applied in environmental engineering. Int. J. Hydrog. Energy 2017, 42, 27801–27811. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, S.; Yang, X.; Lei, Z.; Shimizu, K.; Zhang, Z.; Lee, D.J.; Adachi, Y. Stability and performance of algal-bacterial granular sludge in shaking photo-sequencing batch reactors with special focus on phosphorus accumulation. Bioresour. Technol. 2019, 280, 497–501. [Google Scholar] [CrossRef]
- Abouhend, A.S.; Milferstedt, K.; Hamelin, J.; Ansari, A.A.; Butler, C.; Carbajal-González, B.I.; Park, C. Growth Progression of Oxygenic Photogranules and Its Impact on Bioactivity for Aeration-Free Wastewater Treatment. Environ. Sci. Technol. 2019, 54, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Stauch-White, K.; Srinivasan, V.N.; Camilla Kuo-Dahab, W.; Park, C.; Butler, C.S. The role of inorganic nitrogen in successful formation of granular biofilms for wastewater treatment that support cyanobacteria and bacteria. AMB Express 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Abouhend, A.S.; McNair, A.; Kuo-Dahab, W.C.; Watt, C.; Butler, C.S.; Milferstedt, K.; Hamelin, J.; Seo, J.; Gikonyo, G.J.; El-Moselhy, K.M.; et al. The Oxygenic Photogranule Process for Aeration-Free Wastewater Treatment. Environ. Sci. Technol. 2018, 52, 3503–3511. [Google Scholar] [CrossRef] [PubMed]
- Lettinga, G.; van Velsen, A.F.M.; Hobma, S.W.; de Zeeuw, W.; Klapwijk, A. Use of the upflow sludge blanket (USB) reactor concept for biological wastewater treatment, especially for anaerobic treatment. Biotechnol. Bioeng. 1980, 22, 699–734. [Google Scholar] [CrossRef]
- Pronk, M.; Abbas, B.; Al-zuhairy, S.H.K.; Kraan, R.; Kleerebezem, R.; van Loosdrecht, M.C.M. Effect and behaviour of different substrates in relation to the formation of aerobic granular sludge. Appl. Microbiol. Biotechnol. 2015, 99, 5257–5268. [Google Scholar] [CrossRef]
- Ali, M.; Chai, L.-Y.; Tang, C.-J.; Zheng, P.; Min, X.; Yang, Z.-H.; Xiong, L.; Song, Y. The Increasing Interest of ANAMMOX Research in China: Bacteria, Process Development, and Application. Biomed Res. Int. 2013, 2013, 134914. [Google Scholar] [CrossRef]
- Hann, M. Factors Impacting the Cultivation, Structure, and Oxygen Profiles of Oxygenic Photogranules for Aeration-Free Wastewater Treatment. 2018, Environmental & Water Resources Engineering Masters Projects. Available online: https://scholarworks.umass.edu/cee_ewre/90 (accessed on 29 November 2022). [CrossRef]
- Sun, H.; Yu, P.; Li, Q.; Ren, H.; Liu, B.; Ye, L.; Zhang, X.-X. Transformation of anaerobic granules into aerobic granules and the succession of bacterial community. Appl. Microbiol. Biotechnol. 2017, 101, 7703–7713. [Google Scholar] [CrossRef]
- Sarma, S.J.; Tay, J.-H. Carbon, nitrogen and phosphorus removal mechanisms of aerobic granules. Crit. Rev. Biotechnol. 2018, 38, 1077–1088. [Google Scholar] [CrossRef]
- Trego, A.C.; Mills, S.; Collins, G. Granular biofilms: Function, application, and new trends as model microbial communities. Crit. Rev. Environ. Sci. Technol. 2021, 51, 1702–1725. [Google Scholar] [CrossRef]
- Fang, H.H.P.; Liu, H.; Zhang, T. Characterization of a hydrogen-producing granular sludge. Biotechnol. Bioeng. 2002, 78, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Show, K.Y.; Yan, Y.; Yao, H.; Guo, H.; Li, T.; Show, D.Y.; Chang, J.S.; Lee, D.J. Anaerobic granulation: A review of granulation hypotheses, bioreactor designs and emerging green applications. Bioresour. Technol. 2020, 300, 122751. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.; Xie, J.; Kabore, A.; Wendinso, J.; Chang, Y.; Xie, J.; Guo, M.; Chen, C. Research advances in anammox granular sludge: A review. Crit. Rev. Environ. Sci. Technol. 2022, 52, 631–674. [Google Scholar] [CrossRef]
- Driessen, W.; Hendrickx, T. Two decades of experience with the granular sludge-based anammox® process treating municipal and industrial effluents. Processes 2021, 9, 1207. [Google Scholar] [CrossRef]
- Wang, W.; Wang, J.; Wang, H.; Ma, J.; Wu, M.; Wang, Y. Anammox Granule Enlargement by Heterogenous Granule Self-assembly. Water Res. 2020, 187, 116454. [Google Scholar] [CrossRef]
- Park, J.H.; Chandrasekhar, K.; Jeon, B.H.; Jang, M.; Liu, Y.; Kim, S.H. State-of-the-art technologies for continuous high-rate biohydrogen production. Bioresour. Technol. 2021, 320, 124304. [Google Scholar] [CrossRef]
- Milferstedt, K.; Kuo-Dahab, W.C.; Butler, C.S.; Hamelin, J.; Abouhend, A.S.; Stauch-White, K.; McNair, A.; Watt, C.; Carbajal-González, B.I.; Dolan, S. The importance of filamentous cyanobacteria in the development of oxygenic photogranules. Sci. Rep. 2017, 7, 1–15. [Google Scholar]
- Ansari, A.A.; Ansari, A.A.; Khoja, A.H.; Gikonyo, G.J.; Abouhend, A.S.; Park, C. The fate and dynamics of iron during the transformation of activated sludge into oxygenic photogranules (OPGs) under hydrodynamic batch conditions for environmental applications. J. Environ. Chem. Eng. 2022, 10, 108190. [Google Scholar] [CrossRef]
- Kuo-Dahab, W.C.; Stauch-White, K.; Butler, C.S.; Gikonyo, G.J.; Carbajal-González, B.; Ivanova, A.; Dolan, S.; Park, C. Investigation of the Fate and Dynamics of Extracellular Polymeric Substances (EPS) during Sludge-Based Photogranulation under Hydrostatic Conditions. Environ. Sci. Technol. 2018, 52, 10462–10471. [Google Scholar] [CrossRef]
- Trebuch, L.M.; Oyserman, B.O.; Janssen, M.; Wijffels, R.H.; Vet, L.E.M.; Fernandes, T.V. Impact of hydraulic retention time on community assembly and function of photogranules for wastewater treatment. Water Res. 2020, 173, 115506. [Google Scholar] [CrossRef]
- Gikonyo, J.G.; Ansari, A.A.; Abouhend, A.S.; Tobiason, J.E.; Park, C. Hydrodynamic granulation of oxygenic photogranules. Environ. Sci. Water Res. Technol. 2021, 7, 427–440. [Google Scholar] [CrossRef]
- Sun, Y.; Chang, H.; Zhang, C.; Xie, Y.; Ho, S.H. Emerging biological wastewater treatment using microalgal-bacterial granules: A review. Bioresour. Technol. 2022, 351, 127089. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.J.; Kim, T.-H. Applicability and trends of anaerobic granular sludge treatment processes. Biomass Bioenergy 2014, 60, 189–202. [Google Scholar] [CrossRef]
- Gikonyo, J.G.; Keyser, A.; Tobiason, J.; Jeong, J.; Park, C. In Vivo Evaluation of Oxygenic Photogranules’ Photosynthetic Capacity by Pulse Amplitude Modulation and Phototrophic–Irradiance Curves. ACS ES&T Eng. 2021, 1, 551–561. [Google Scholar] [CrossRef]
- Jiang, Q.; Chen, H.; Fu, Z.; Fu, X.; Wang, J.; Liang, Y.; Yin, H.; Yang, J.; Jiang, J.; Yang, X.; et al. Current Progress, Challenges and Perspectives in the Microalgal-Bacterial Aerobic Granular Sludge Process: A Review. Int. J. Environ. Res. Public Health 2022, 19, 13950. [Google Scholar] [CrossRef]
- Zhang, X.; Lei, Z.; Liu, Y. Microalgal-bacterial granular sludge for municipal wastewater treatment: From concept to practice. Bioresour. Technol. 2022, 354, 127201. [Google Scholar] [CrossRef] [PubMed]
- Quijano, G.; Arcila, J.S.; Buitrón, G. Microalgal-bacterial aggregates: Applications and perspectives for wastewater treatment. Biotechnol. Adv. 2017, 35, 772–781. [Google Scholar] [CrossRef]
- Park, C.; Takeuchi, N. Unmasking photogranulation in decreasing glacial albedo and net autotrophic wastewater treatment. Environ. Microbiol. 2021, 23, 6391–6404. [Google Scholar] [CrossRef]
- Ansari, A.A.; Abouhend, A.S.; Park, C. Effects of seeding density on photogranulation and the start-up of the oxygenic photogranule process for aeration-free wastewater treatment. Algal Res. 2019, 40, 101495. [Google Scholar] [CrossRef]
- Mcnair, A.M. Pilot Reactor Operation of the Oxygenic Photogranule ( OPG ) Wastewater Treatment Process. 2017, Environmental & Water Resources Engineering Masters Projects. Available online: https://scholarworks.umass.edu/cee_ewre/83 (accessed on 29 November 2022). [CrossRef]
- Ji, B.; Zhu, L.; Wang, S.; Liu, Y. Temperature-effect on the performance of non-aerated microalgal-bacterial granular sludge process in municipal wastewater treatment. J. Environ. Manag. 2021, 282, 111955. [Google Scholar] [CrossRef] [PubMed]
- Massé, D.I.; Rajagopal, R.; Singh, G. Technical and operational feasibility of psychrophilic anaerobic digestion biotechnology for processing ammonia-rich waste. Appl. Energy 2014, 120, 49–55. [Google Scholar] [CrossRef]
- Fan, S.; Zhu, L.; Ji, B. Deciphering the effect of light intensity on microalgal-bacterial granular sludge process for non-aerated municipal wastewater treatment. Algal Res. 2021, 58, 102437. [Google Scholar] [CrossRef]
- Ji, B.; Zhang, M.; Gu, J.; Ma, Y.; Liu, Y. A self-sustaining synergetic microalgal-bacterial granular sludge process towards energy-efficient and environmentally sustainable municipal wastewater treatment. Water Res. 2020, 179, 115884. [Google Scholar] [CrossRef]
- Kant Bhatia, S.; Ahuja, V.; Chandel, N.; Mehariya, S.; Kumar, P.; Vinayak, V.; Saratale, G.D.; Raj, T.; Kim, S.H.; Yang, Y.H. An overview on microalgal-bacterial granular consortia for resource recovery and wastewater treatment. Bioresour. Technol. 2022, 351, 127028. [Google Scholar] [CrossRef] [PubMed]
- Ji, B. Towards environment-sustainable wastewater treatment and reclamation by the non-aerated microalgal-bacterial granular sludge process: Recent advances and future directions. Sci. Total Environ. 2022, 806, 150707. [Google Scholar] [CrossRef] [PubMed]
- Bland, E.; Angenent, L.T. Pigment-targeted light wavelength and intensity promotes efficient photoautotrophic growth of Cyanobacteria. Bioresour. Technol. 2016, 216, 579–586. [Google Scholar] [CrossRef]
- Kang, D.; Kim, K.; Jang, Y.; Moon, H.; Ju, D.; Jahng, D. Nutrient removal and community structure of wastewater-borne algal-bacterial consortia grown in raw wastewater with various wavelengths of light. Int. Biodeterior. Biodegrad. 2018, 126, 10–20. [Google Scholar] [CrossRef]
- Kilbane, J.J. Shining a Light on Wastewater Treatment with Microalgae. Arab. J. Sci. Eng. 2022, 47, 45–56. [Google Scholar] [CrossRef]
- Gikonyo, J.G.; Ansari, A.; Park, C.; Tobiason, J. Physical characterization of oxygenic photogranules. Biochem. Eng. J. 2022, 186, 108592. [Google Scholar] [CrossRef]
- Shankar, R.; Kumar, S.; Prasad, A.K.; Khare, P.; Varma, A.K.; Yadav, V.K. Chapter 9-Biological Wastewater Treatment Plants (WWTPs) for Industrial Wastewater; Shah, M., Rodriguez-Couto, S.B.T.-M.E., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 193–216. ISBN 978-0-12-822503-5. [Google Scholar]
- Stott, R. 31-Fate and Behaviour of Parasites in Wastewater Treatment Systems; Mara, D., Horan, N.B.T.-H., Eds.; Academic Press: London, UK, 2003; pp. 491–521. ISBN 978-0-12-470100-7. [Google Scholar]
- Acién Fernández, F.G.; Gómez-Serrano, C.; Fernández-Sevilla, J.M. Recovery of Nutrients From Wastewaters Using Microalgae. Front. Sustain. Food Syst. 2018, 2, 1–13. [Google Scholar] [CrossRef]
- Yang, J.; Shi, W.; Fang, F.; Guo, J.; Lu, L.; Xiao, Y.; Jiang, X. Exploring the feasibility of sewage treatment by algal–bacterial consortia. Crit. Rev. Biotechnol. 2020, 40, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; You, H.; Li, Z.; Xie, B.; Li, W.; Qi, S.; Wang, R.; Chen, H.; Ma, B.; Ding, Y.; et al. Formation mechanism and metabolic pathways of photogranule in batch and continuous-flow mode under high salinity stress. Chem. Eng. J. 2023, 452, 139306. [Google Scholar] [CrossRef]
- dos Santos Neto, A.G.; Barragán-Trinidad, M.; Florêncio, L.; Buitrón, G. Strategy for the formation of microalgae-bacteria aggregates in high-rate algal ponds. Environ. Technol. 2021, 1–14. [Google Scholar] [CrossRef]
- Foladori, P.; Petrini, S.; Andreottola, G. Evolution of real municipal wastewater treatment in photobioreactors and microalgae-bacteria consortia using real-time parameters. Chem. Eng. J. 2018, 345, 507–516. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, L.; Ji, B.; Hou, H.; Ma, Y. Microalgal-bacterial granular sludge process in non-aerated municipal wastewater treatment under natural day-night conditions: Performance and microbial community. Water 2021, 13, 1479. [Google Scholar] [CrossRef]


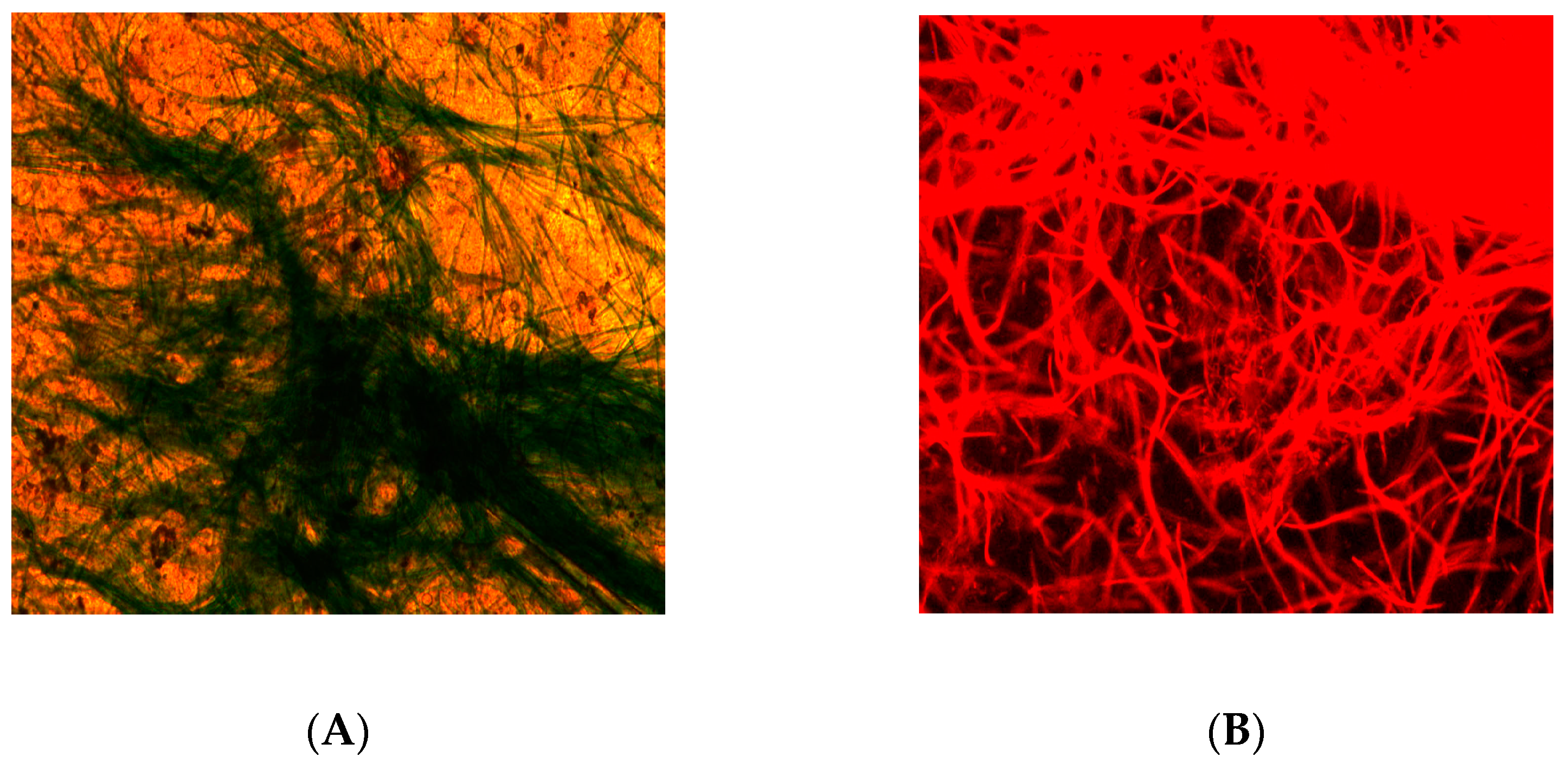
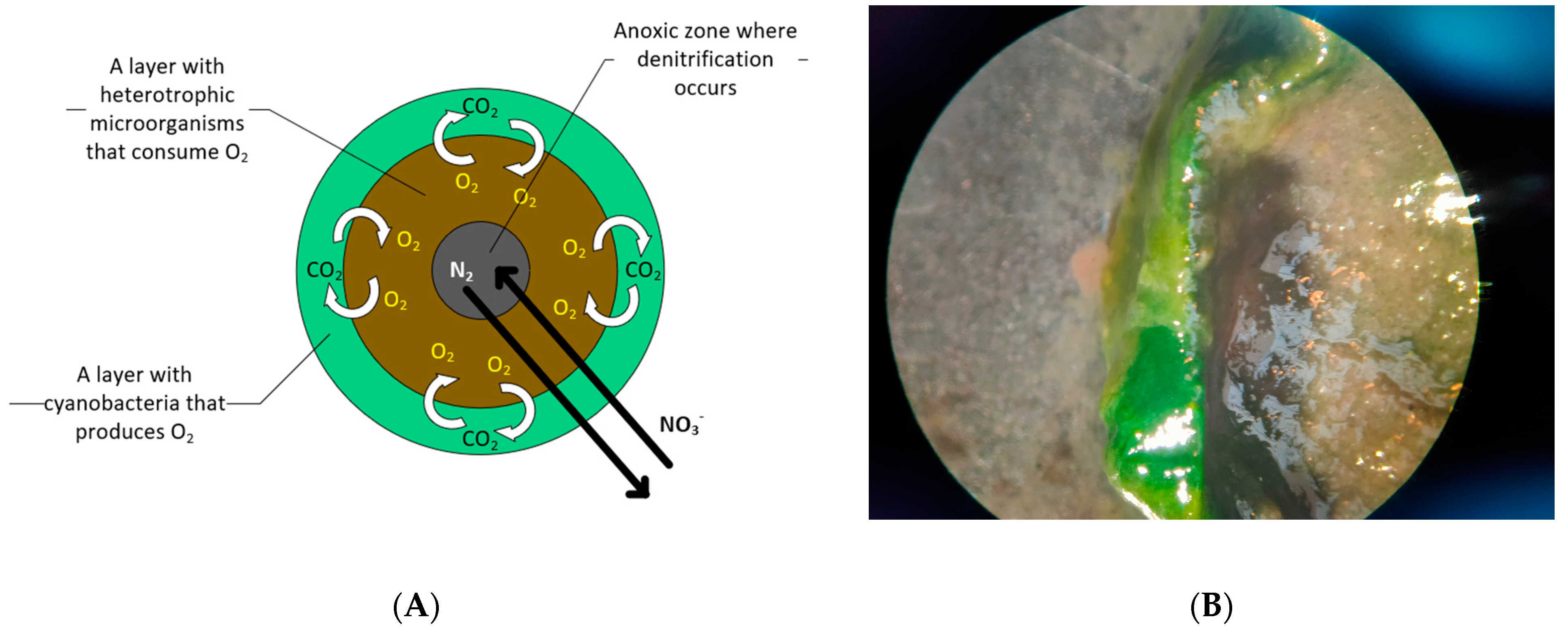

| Light Intensity (mmol/m2·s) | Light/Dark Cycles | Size (mm) | SVI (mL/g) |
|---|---|---|---|
| 2000 | 10 h/14 h | 0.6 | 100 |
| 200 | 24 h/0 h | 1.3 | 24 |
| 200 | 24 h/0 h | 0.5 | 58 |
| 150 | 3.5 h/2.5 h | 0.8 | 61 |
| 150 | 3.5 h/2.5 h | 1.2 | Nd |
| 100 | 3.5 h/2.5 h | 1.8 | 53 |
| 284 | 8 h/16 h | 2.2 | 42 |
| Size | Density (g/mL) | Settling Velocity (m/h) | Sludge Volume Index (mL/g) | Porosity | Water Content (%) | |
|---|---|---|---|---|---|---|
| Aerobic granules | 0.2–16 mm | 1.004–1.065 | 18–130 | Below 80 | 0.68–0.93 | 94–97 |
| OPGs | 0.1–5 mm | Highly variable | 36–360 | Nd | Nd | 78–95 |
| AS flocs | 0.5–1000 μm (mostly < 100 μm) | 1.002–1.006 | 0.6–15 | 100–150 | >0.95 | >99 |
| Type of Wastewater | Reactor Volume, (L) | PAR, (μmol/m2·s) | Stirring Intensity, (rpm) | Time of Operation | COD Removal, (%) | Nutrient Removal (Nitrogen/ Phosphorus), (%) | HRT (d) | Reference |
|---|---|---|---|---|---|---|---|---|
| PE as well as screened raw wastewater | 1.2–3 | 90–150 | 100 | 3 (1) | Nd | Nd | 0.75 | [41] |
| PE | 1.2 | 150 | 100 | 150 (2) | 82–86 | 90–96 (3) 52–57 (4) 21–44 (5) | 0.9, 0.75 | [27] |
| Raw municipal wastewater | Nd | 150 | Nd | Nd | 85 | 71 (6) 75 (5) | 0.5 | [3] |
| Nd | 1 | Nd | Nd | Nd | 59.68 | 87.50 (6) 85.37 (5) | Nd | [9] |
| Synthetic wastewater (modified BG11 medium) | 1.7 | 500 | Nd | 148 (2) | Nd | Nd | 0.33, 0.67, 1, 2 | [44] |
| PE | 1 | 101–115 | 100 | 150 (2) | 41–90 | 85–95 (7) 95–100 (8) | 1, 3 | [4] |
| PE | 2 | 150 | 100 | Nd | 50–98 | 14–65 (4) | 0.75 | [53] |
| PE | 8, 10–30 | 10 (9) | 100 | 53 (2) | 50–76 | 93 | 0.75, 1 | [54] |
| High saline wastewater | 3 (9) | 46 | Nd | Nd | 85.36 ± 2.84 (10) | 93.30 ± 2.07 (3) 77.68 ± 5.81% (5) | 1 | [69] |
| Raw | 60 (HRAP) | Natural sunlight | Nd | Nd | 80 | 80 (3) | Nd | [70] |
| Municipal | 2 (P-SBR) | Artificial light | 200 | Nd | 87 | 68 (6) 16 (5) | 2 | [71] |
| Synthetic municipal | 0.5 | 200 | Nd | 33 (2) | 47.6–59.9 | 56.5–78.1 (3) 61.5–74.25 (5) | 0.5 | [72] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smetana, G.; Grosser, A. The Oxygenic Photogranules—Current Progress on the Technology and Perspectives in Wastewater Treatment: A Review. Energies 2023, 16, 523. https://doi.org/10.3390/en16010523
Smetana G, Grosser A. The Oxygenic Photogranules—Current Progress on the Technology and Perspectives in Wastewater Treatment: A Review. Energies. 2023; 16(1):523. https://doi.org/10.3390/en16010523
Chicago/Turabian StyleSmetana, German, and Anna Grosser. 2023. "The Oxygenic Photogranules—Current Progress on the Technology and Perspectives in Wastewater Treatment: A Review" Energies 16, no. 1: 523. https://doi.org/10.3390/en16010523
APA StyleSmetana, G., & Grosser, A. (2023). The Oxygenic Photogranules—Current Progress on the Technology and Perspectives in Wastewater Treatment: A Review. Energies, 16(1), 523. https://doi.org/10.3390/en16010523









