Abstract
An analysis of the literature data indicates a wide front of research and development in the field of the use of methane–hydrogen mixtures as a promising environmentally friendly low-carbon fuel. The conclusion of most works shows that the use of methane–hydrogen mixtures in internal combustion engines improves their performance and emission characteristics. The most important aspect is the concentration of hydrogen in the fuel mixture, which affects the combustion process of the fuel and determines the optimal operating conditions of the engine. When using methane–hydrogen mixtures with low hydrogen content, the safety measures and risks are similar to those that exist when working with natural gas. Serious logistical problems are associated with the difficulties of using the existing gas distribution infrastructure for transporting methane–hydrogen mixtures. It is possible that, despite the need for huge investments, it will be necessary to create a new infrastructure for the production, storage and transportation of hydrogen and its mixtures with natural gas. Further research is needed on the compatibility of pipeline materials with hydrogen and methane–hydrogen mixtures, safety conditions for the operation of equipment operating with hydrogen or methane–hydrogen mixtures, as well as the economic and environmental feasibility of using these energy carriers.
1. Introduction
Increasing the fuel efficiency and reducing pollutant emissions, including carbon dioxide emissions, have become the leading trends in modern energy. The impossibility of a global solution for the problem of reducing the carbon footprint of the energy sector through the transition of industrial energy to renewable energy sources is already obvious [1]. An analysis of various methods for separating CO2 from flue gases [2], in which its content is less than 10%, and their subsequent utilization [3] or disposal demonstrated the need for additional energy costs. These costs are commensurate with the initially generated energy [4], that is, a minimum doubling of the consumed resources and the cost of energy for the consumer is required. Therefore, an approach to solving the problem of CO2 emissions by the global energy industry based on the replacement of hydrocarbon energy carriers with hydrogen is gaining popularity. Such a transition is actually proclaimed the goal of the Paris Agreement [5].
The trend towards low-carbon energy has contributed to the popularization of the concept of “hydrogen energy” and has intensified research on the use of hydrogen as a fuel [6]. According to forecasts, annual investments in hydrogen technologies in the period 2018–2022 will amount to EUR 1.9 billion per year, and by 2050 the world consumption of hydrogen could reach 370 million tons per year [7]. In the long term, after adopting the energy of thermonuclear fusion, hydrogen, the oxidation of which produces only water, can become a universal environmentally friendly energy carrier.
There are several industrial technologies for producing hydrogen, each with its own advantages and disadvantages, but the cost of hydrogen production remains high [8,9]. Leading industrial countries—USA, EU countries, Great Britain, Japan, China, South Korea, Australia—are developing a national strategy and programs for the development of hydrogen energy. In the United States, the budget for hydrogen projects is USD 1.7 billion over five years, with several times more funds coming from private companies. The funds for the hydrogen theme in the EU comprise EUR 2 billion and in Japan USD 4 billion for 20 years. Nevertheless, the economic feasibility of such a transition at the present time and even its fundamental technological feasibility raise many problems [10,11,12,13]. However, the development of hydrogen energy is necessary, since by the end of the current century or somewhat later, a global transition to thermonuclear energy is inevitable. Then, with an abundance of cheap and affordable thermonuclear energy, hydrogen can become a natural basic energy carrier. Therefore, already now it is necessary to prepare appropriate technological solutions for the transportation of hydrogen and its use in various types of power plants.
The main difference between hydrogen as a fuel and traditional hydrocarbon fuels is the significantly higher burning rate, which under standard conditions is at least six times higher than for methane and approximately eight times for temperatures and pressures typical of internal combustion engine (ICE) operating conditions. Hydrogen has much wider concentration limits of flame propagation compared to hydrocarbons. Therefore, along with the use of hydrogen directly as a fuel, its use as an additive to traditional fuels, primarily to compressed natural gas (CNG), is widely discussed.
Addition of hydrogen to methane increases the velocity of flame propagation, reducing the duration of fuel combustion. This leads to a sharper increase in pressure in the ICE, high rates of heat release and a shorter time interval between fuel ignition and the peak of heat release compared to methane. At the same time, the presence of methane in the methane–hydrogen mixture reduces the risks of abnormal combustion, such as flashback and detonation in the engine, which are characteristic of hydrogen combustion [14]. The use of mixed fuels based on hydrogen and natural gas can significantly increase the thermal efficiency of combustion and reduce greenhouse gas emissions in the ICE [15,16]. The purpose of this work is to analyze the literature data on the most promising and diverse areas of the use of hydrogen and hydrogen-containing mixtures for energy generation, in transport and in the domestic sector, as well as related logistical problems.
2. Energy Characteristics of Hydrogen and Methane–Hydrogen Mixtures
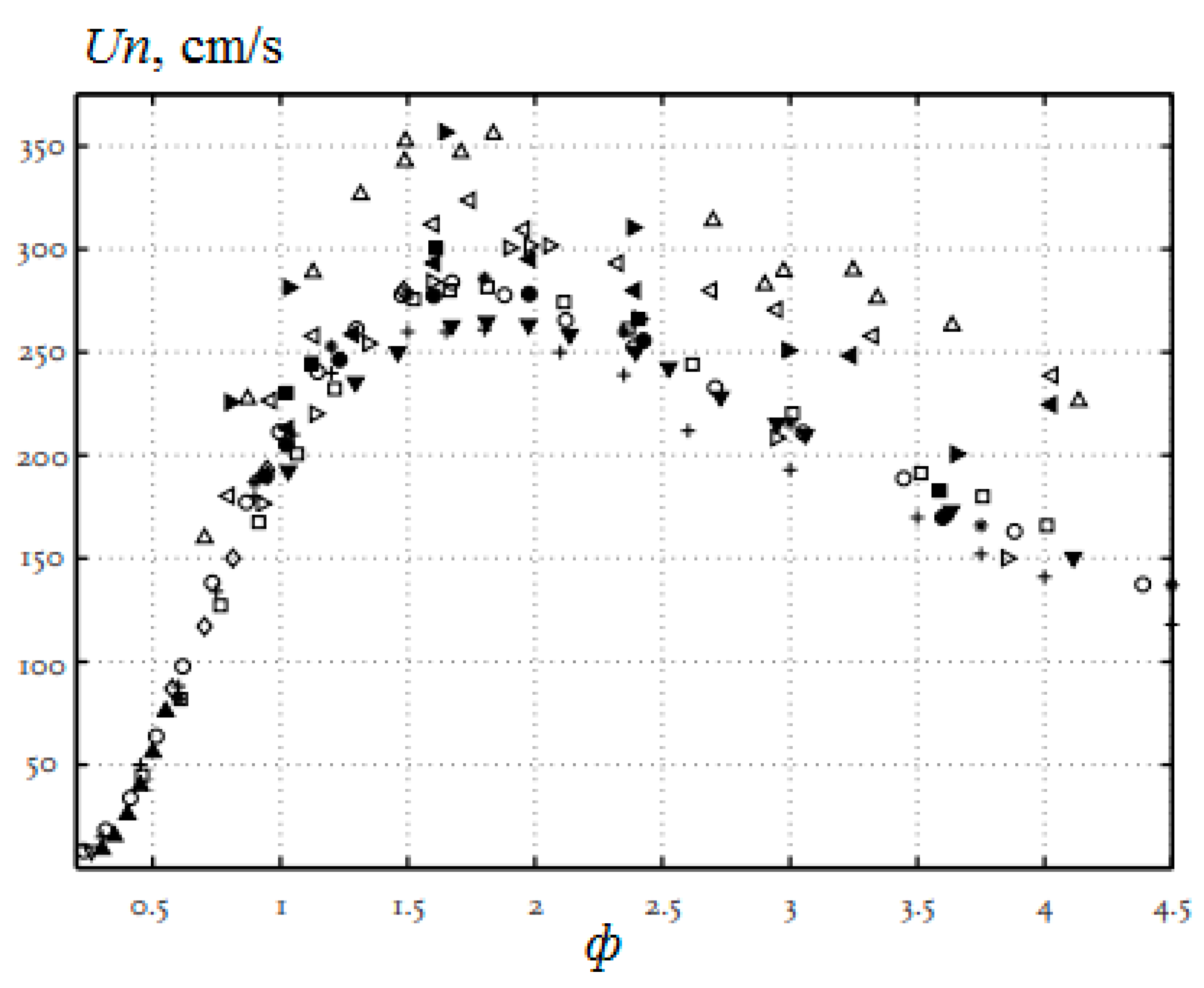
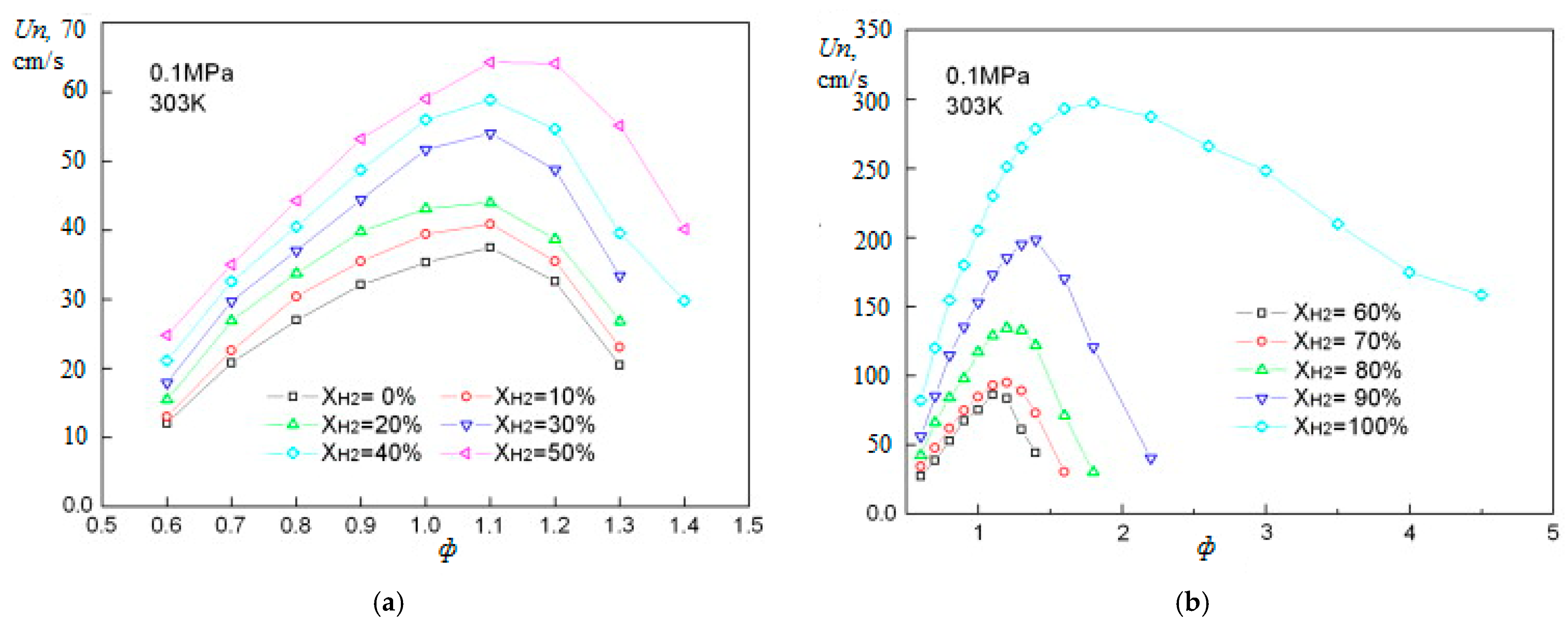
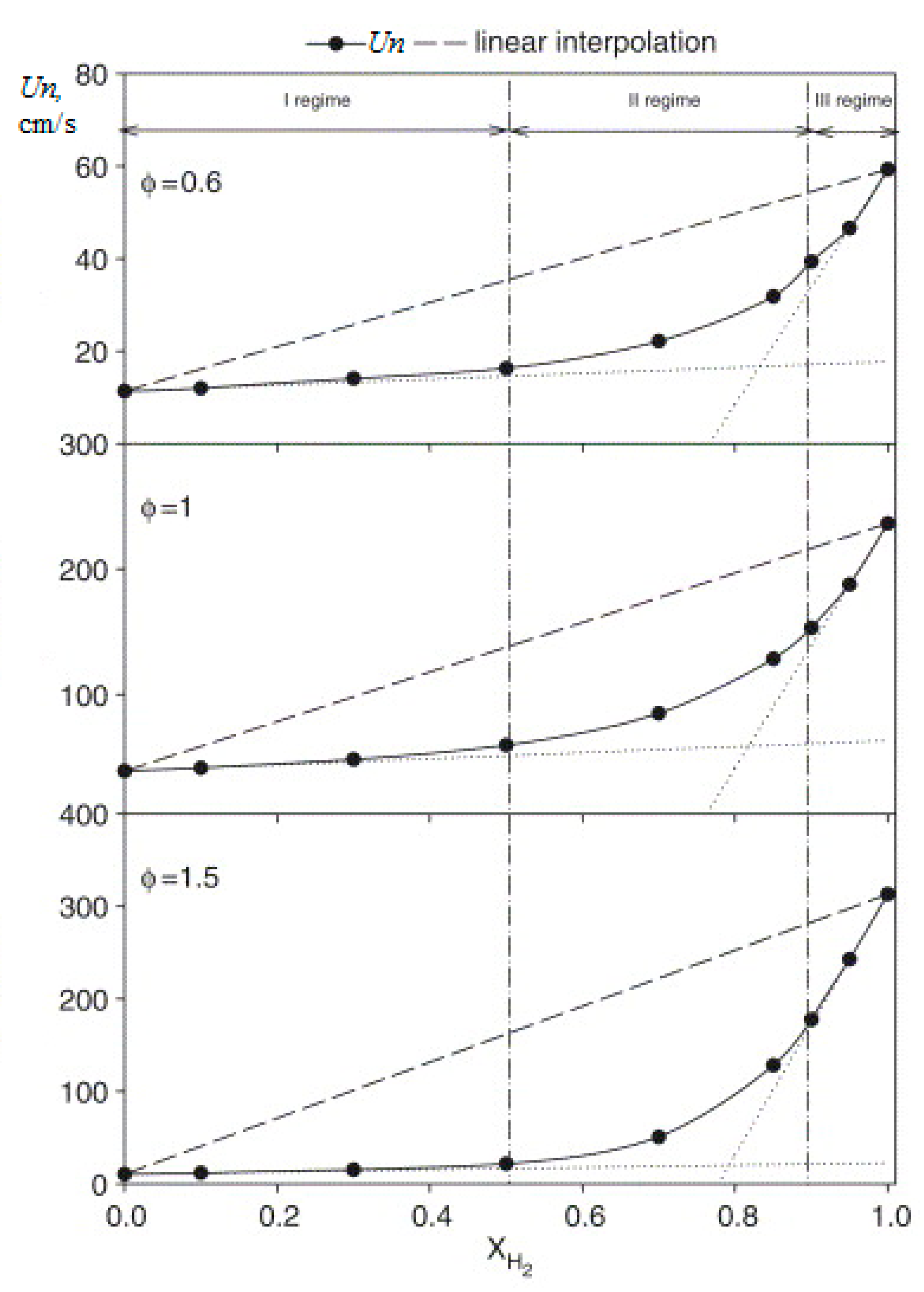
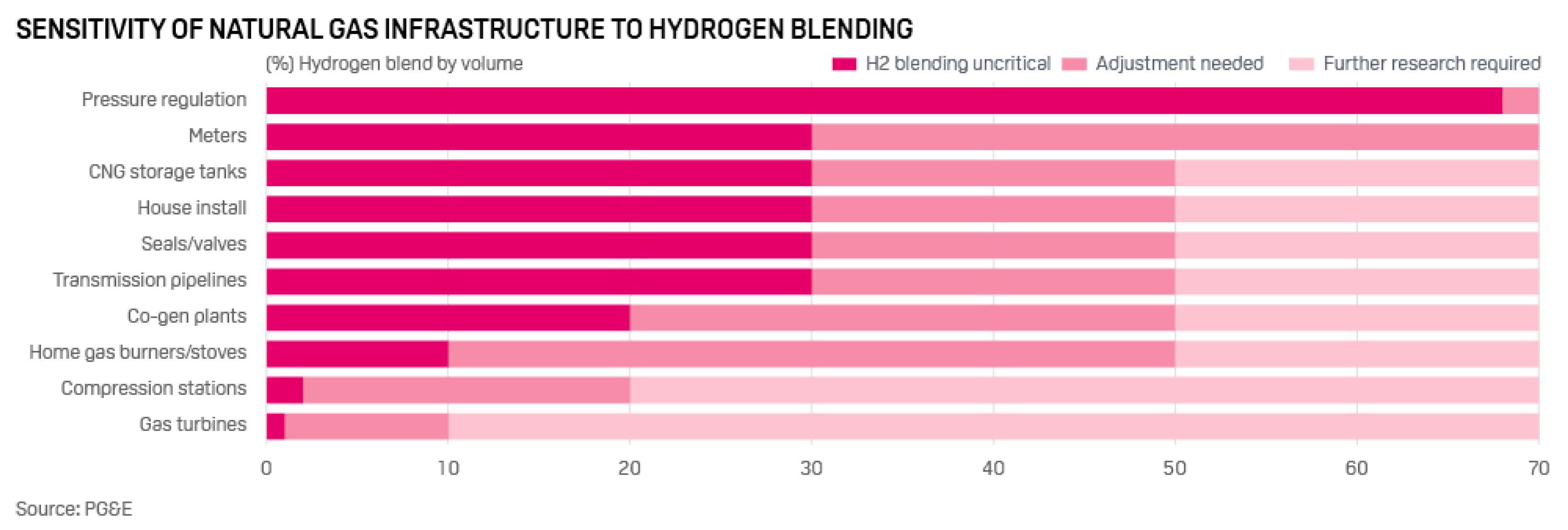
The main parameters characterizing the combustion of fuels include the normal combustion rate Un, the concentration limits of flame propagation, and the ignition delay. The normal combustion rate of the laminar flame determines the stability of the combustion process, and the conditions under which blow off is possible, or vice versa, its reverse flashback towards the flow. A large number of studies have been devoted to the study of the normal combustion rate of hydrogen–air mixtures. The dependence Un of hydrogen–air mixtures under normal conditions on the equivalence ratio (φ) is shown in Figure 1. The scatter of experimental results for the maximum value Un of hydrogen–air mixtures is quite high, the values obtained lie in the range from 260 to 350 cm/s [17]. These values are seven to nine times higher than the corresponding value for stoichiometric methane–air mixtures under normal conditions (Un = 36 ± 1 cm/s), which determines the main differences in the combustion processes of hydrogen and methane. The difference in the combustion rate of methane and hydrogen is associated with a difference in the concentration and kinetics of the formation of H, O, and OH radicals, which determine the course of the process [18].
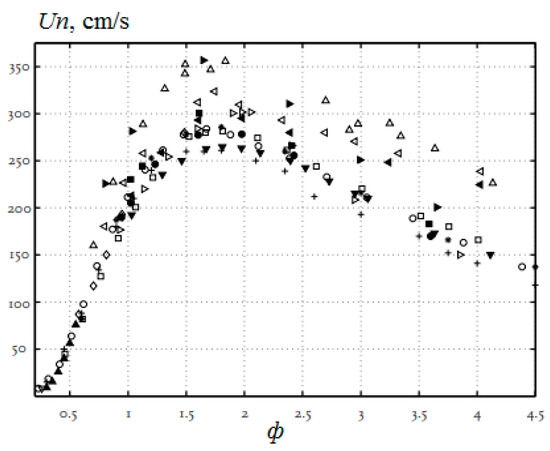
Figure 1.
Dependence of the normal flame velocity Un of hydrogen–air mixtures on the equivalence ratio under normal conditions according to the results of various studies, which are represented by various symbols (Adapted from ref. [17]).
Data on the combustion rate of methane–hydrogen–air mixtures are scarce, and for high pressures they are practically absent. In the general case, in addition to temperature T, pressure P, and equivalence ratio φ, the normal flame velocity of methane–hydrogen mixtures will depend on the concentration of hydrogen nH2 and other gas components nX. The normal flame velocity is a function of the following parameters:
Un = f (T, P, φ, nH2, nX)
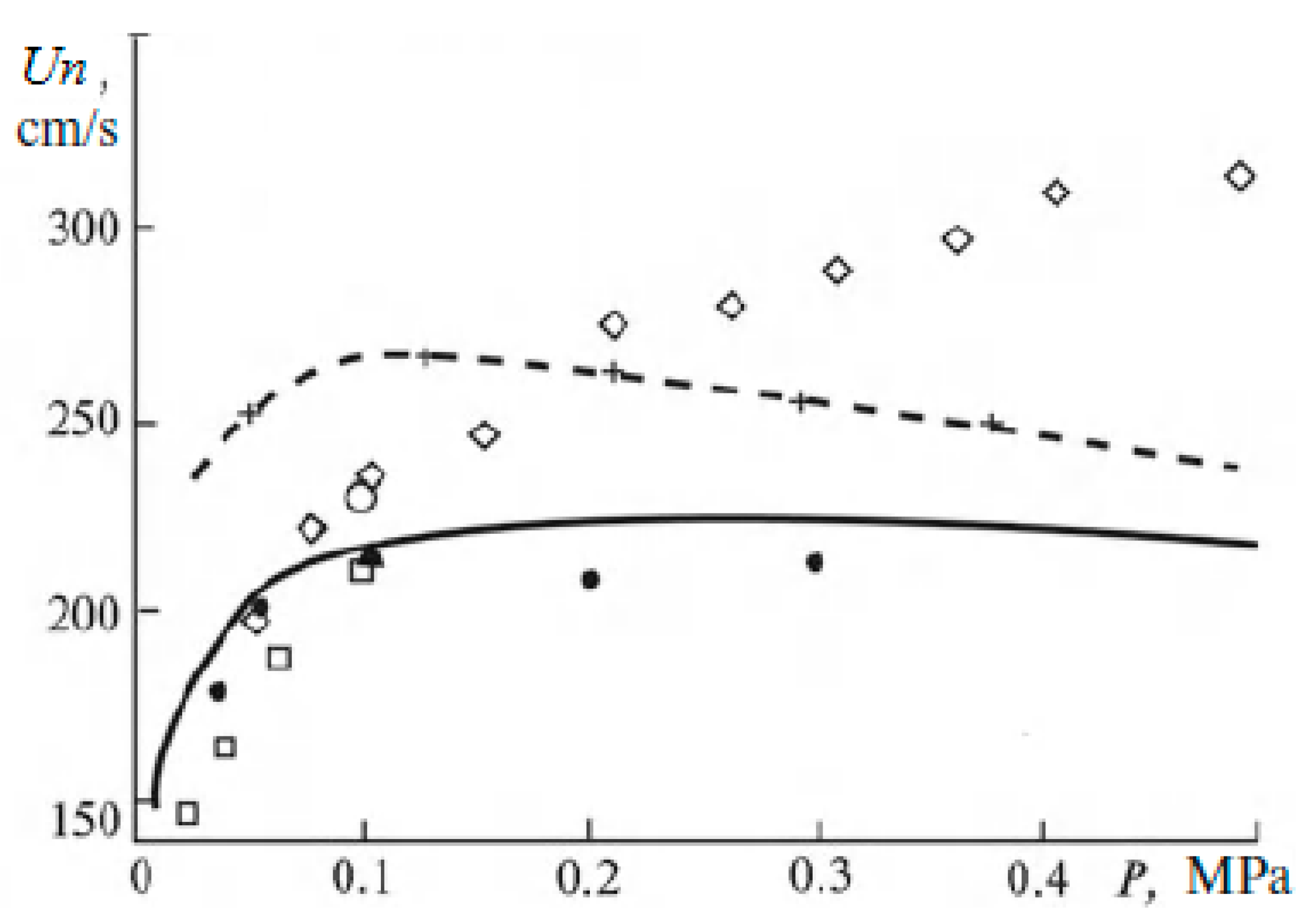
As for the influence of pressure on the normal flame velocity in mixtures containing hydrogen, then, as noted in [19], it is not always possible to determine it correctly in experiments. There are conflicting statements in the literature about the nature of the dependence of the normal flame velocity. When using different methods, sometimes even different signs of the baric index are obtained. This leads to large errors when extrapolating measured values to an unexplored pressure range. In the literature, at one time there was a rule that in mixtures with a flame velocity of more than 100 cm/s at atmospheric pressure, the combustion rate increases with pressure, and in mixtures with a flame velocity of 50 cm/s or less, the combustion rate decreases with pressure. However, it is stated in [19] that experimental material has now been accumulated that refutes this empirical rule for mixtures containing hydrogen. Experimental and calculated data are also given there, showing that with increasing pressure, the normal flame velocity does not decrease and even, under certain conditions, increases (Figure 2 and Figure 3).
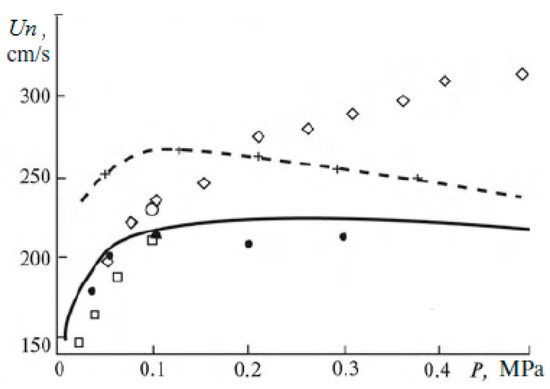
Figure 2.
Flame velocity versus pressure in a stoichiometric mixture of hydrogen and air at T0 = 298 K. Dots are data from various experiments; lines are results of calculations (Adapted with permission from ref. [19]. Copyright 2012, Springer).
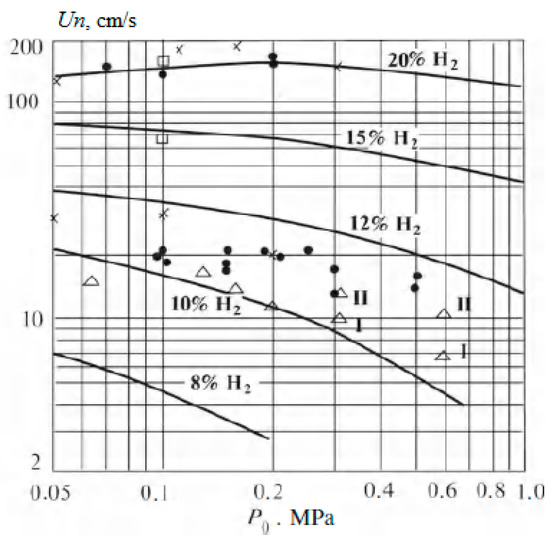
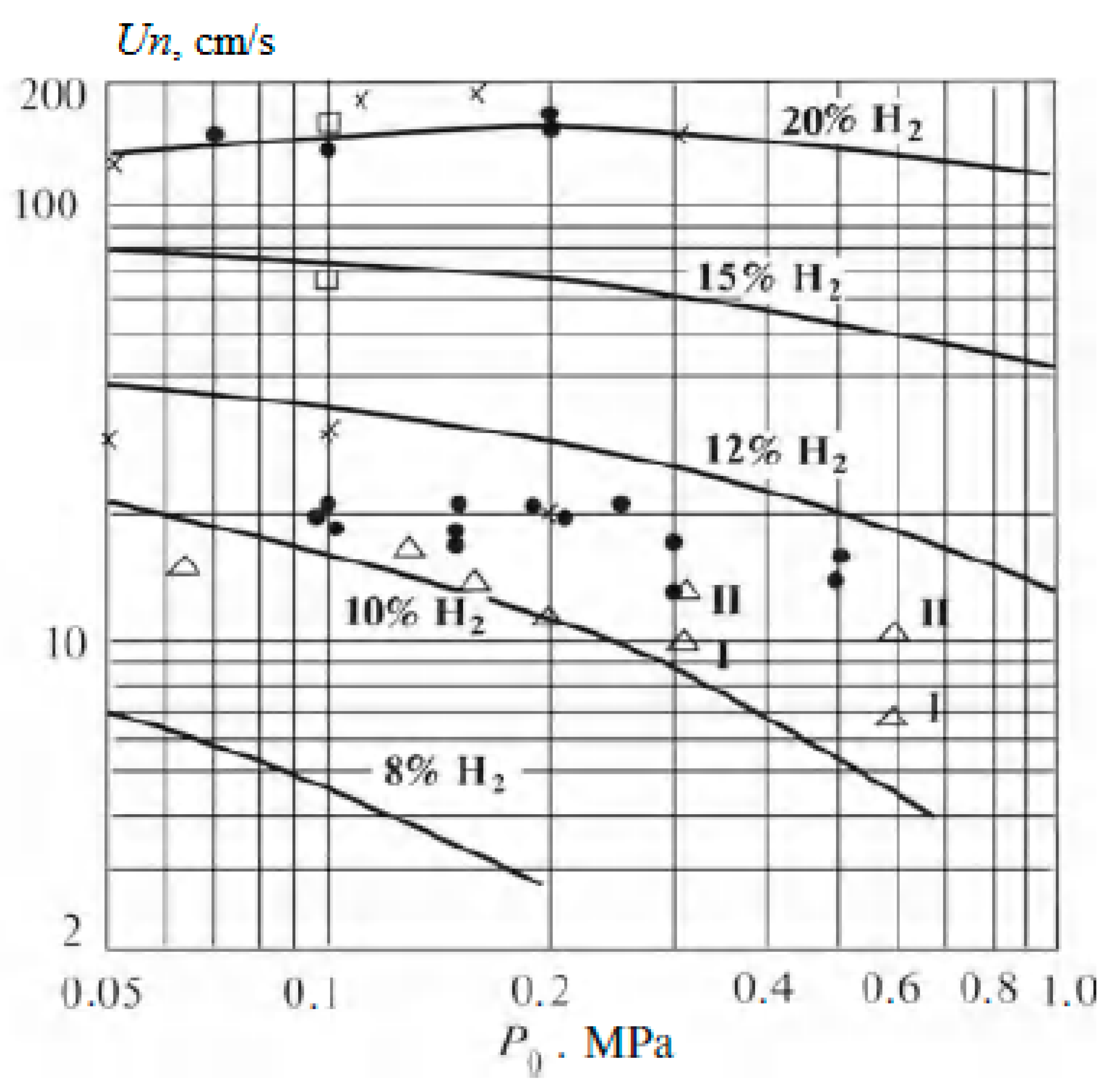
Figure 3.
Dependence of flame velocity on pressure in lean hydrogen–air mixtures. Dots are data from various experiments; lines are results of calculations (Adapted with permission from ref. [19]. Copyright 2012, Springer).
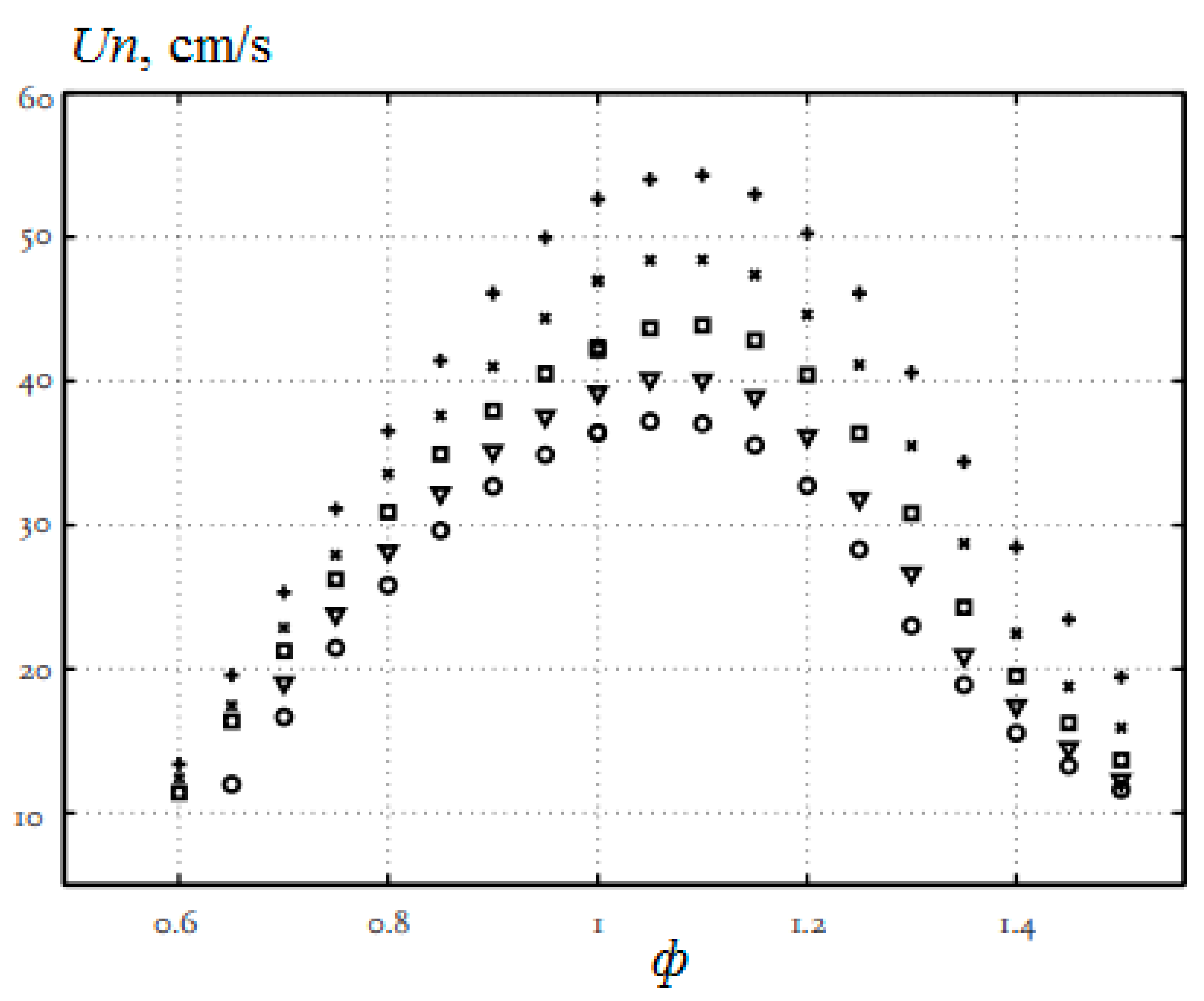
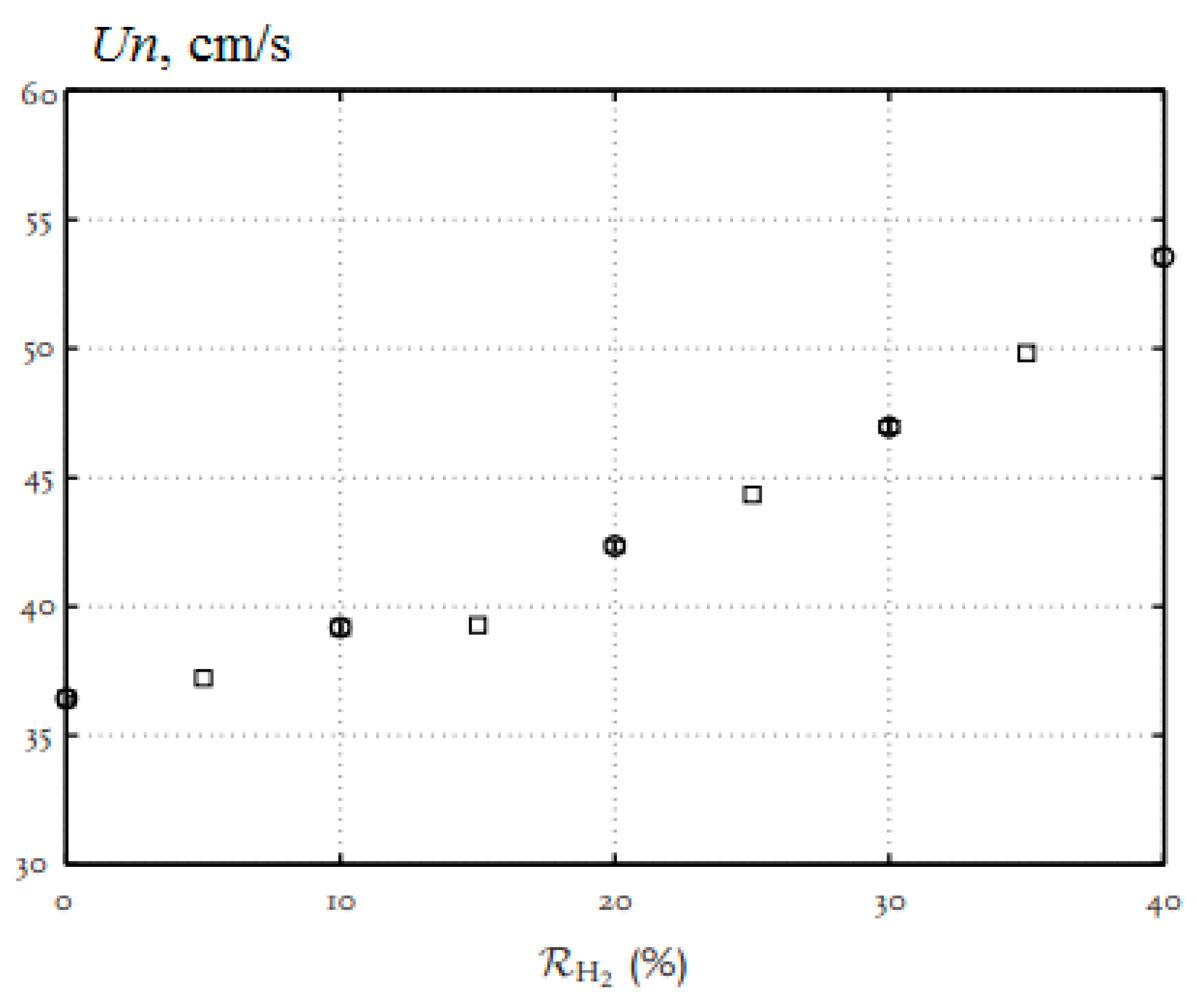
The addition of hydrogen to the methane–air mixture leads to an increase in the combustion rate, a reduction in the width of the flame and an extension of the ignition limits, especially in the region of rich mixtures. In [17], the combustion of methane–air mixtures was studied under normal conditions and the equivalence ratio from 0.6 to 1.5 with additions of up to 40% hydrogen (Figure 4). The laminar flame velocity was determined by the heat flux method. All mixtures show the same behavior, and it is clearly seen that the addition of hydrogen increases the normal flame velocity. For stoichiometric mixtures, the normal flame velocity increases with 36.4 cm/s (H2 = 0) to 39.2 cm/s (H2 = 10%), 42.3 cm/s (H2 = 20%), 47.0 cm/s (H2 = 30%) and 53.5 cm/s (H2 = 40%).
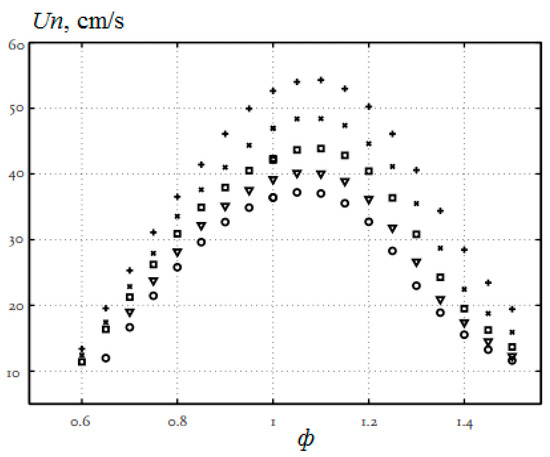
Figure 4.
Dependence of the normal flame velocity Un of hydrogen–methane–air mixtures on the equivalence ratio φ under normal conditions. The hydrogen concentration: o—0%; ▽—10%; □—20%; x—30%; +—40% (Adapted from ref. [17]).
Thus, an increase in the hydrogen concentration in the range up to 40% leads to an almost linear increase in the normal flame velocity (Figure 5).
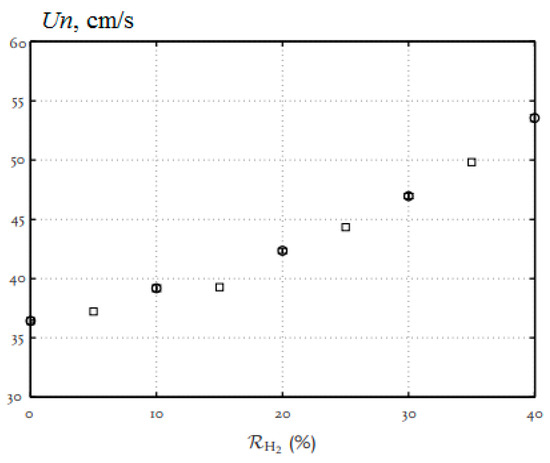
Figure 5.
Dependence of the normal flame velocity Un of hydrogen–methane–air mixtures on the hydrogen concentration under normal conditions (Adapted from ref. [17]).
In [20], the dependence of the laminar flame velocity of methane–hydrogen–air mixtures on the equivalence ratio φ was obtained by studying a stationary flame in a spherical bomb for atmospheric pressure and the initial temperature of the mixture T0 = 360 K at a hydrogen concentration of 10, 20 and 50% (Figure 6).
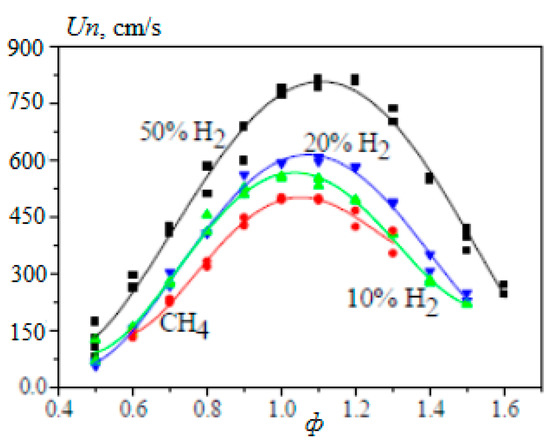
Figure 6.
Dependence of the normal flame velocity Un of hydrogen–methane–air mixtures on the equivalence ratio φ under atmosphere pressure and T0 = 360 K (Adapted with permission from ref. [20], 2007, prof. Prof Alexey Burluka).
The dependence of the laminar flame velocity for methane–hydrogen–air mixtures under normal conditions on φ in the range of hydrogen concentrations from 0 to 100% was measured in [18] (Figure 7).
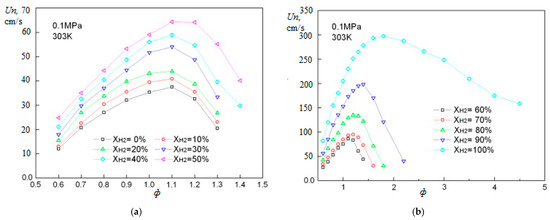
Figure 7.
Dependence of the normal flame velocity Un for hydrogen–methane–air mixtures on the equivalence ratio φ under normal conditions for various hydrogen concentration [18]: (a) hydrogen concentrations 0–50% vol., (b) hydrogen concentrations 60–100% vol. (Adapted with permission from ref. [18], 2009, Elsevier.)
Figure 4, Figure 6 and Figure 7 clearly show not only an increase in the normal flame velocity, but also a shift in the maximum velocity towards richer mixtures as the hydrogen content increases. With the addition of 50% H2, the maximum was at φ = 1.15, which corresponds to a significantly leaner mixture than that at which the maximum combustion rate is achieved in hydrogen (φ =1.8). In addition, the limits of combustion are expanding. Thus, under the experimental conditions for methane, ignition of mixtures with φ > 1.2 was not observed and with the addition of only 10% H2, the combustion limit of rich mixtures expanded to φ = 1.4, and with the addition of 50% H2 to φ = 1.6 [20]. Less significant changes were observed for lean mixtures. The limit decreased from φ = 0.6 for methane to φ = 0.5 for the mixture with the addition of 10% H2 and then did not change with the increasing hydrogen concentration.
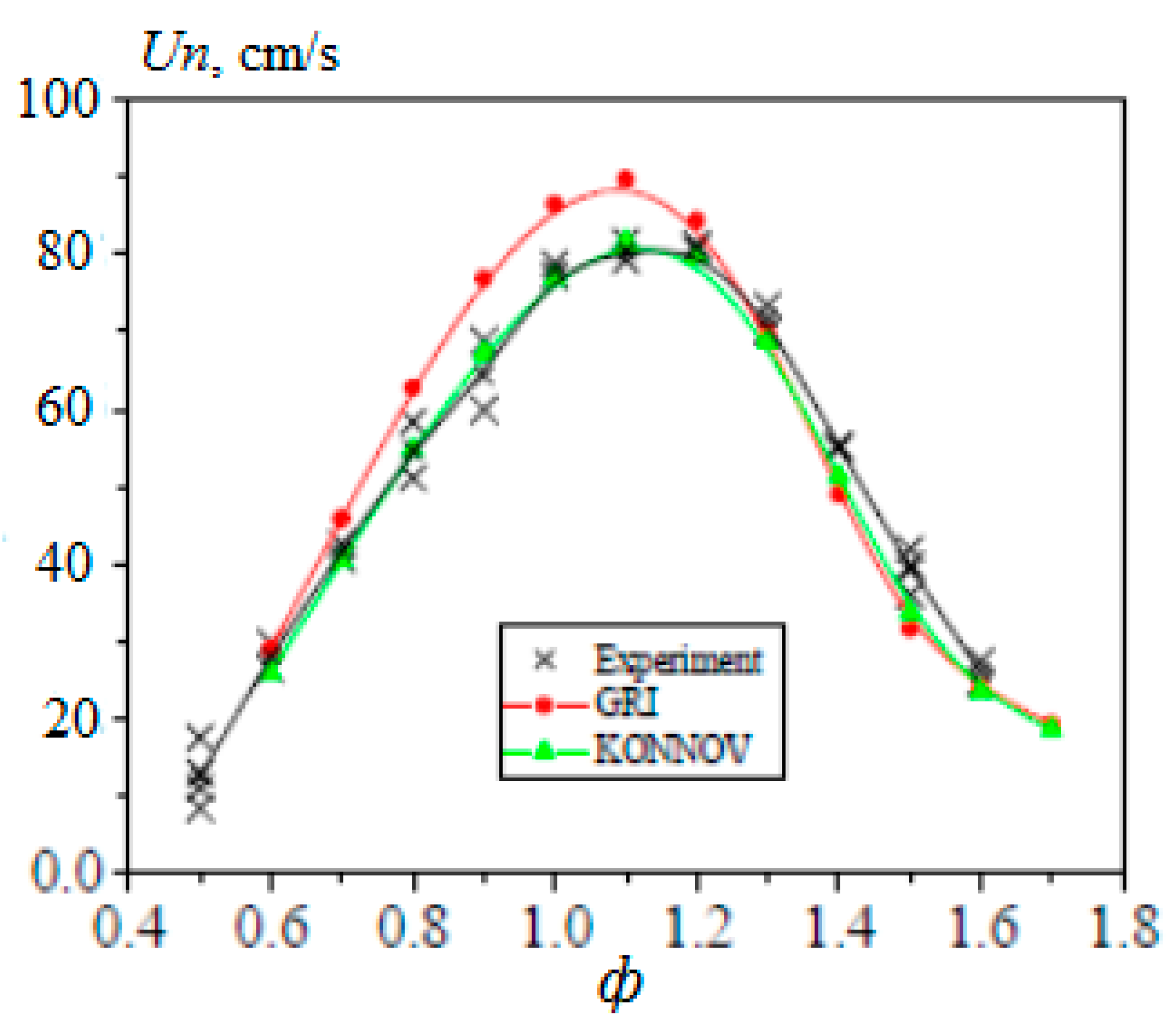
In [20], the experimentally obtained values of the normal velocity of the laminar flame of methane–hydrogen–air mixtures were compared with the values obtained by calculation. The simulation was carried out using two well-known kinetic mechanisms, GRI-Mech 3.0 [21] and Konnov [22], which showed good agreement with experimental results, especially for the Konnov mechanism (Figure 8).
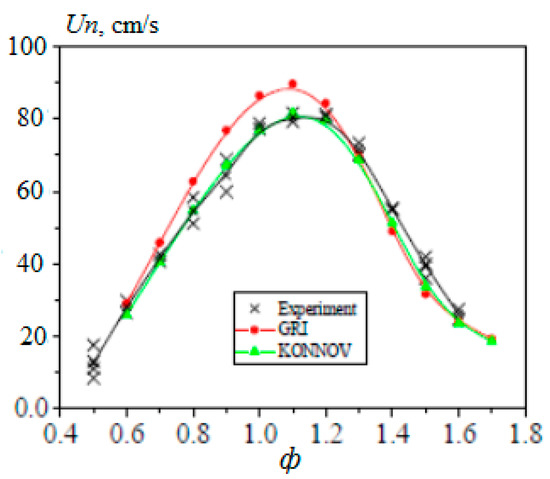
Figure 8.
Comparison of the experimental (x) and calculated (•—GRI-Mech; ▲—Konnov) dependences for the normal flame velocity of the methane–hydrogen–air mixture (50% H2) on the equivalence ratio at atmospheric pressure and T0 = 360 K (Adapted with permission from ref. [20], 2007, Prof Alexey Burluka).
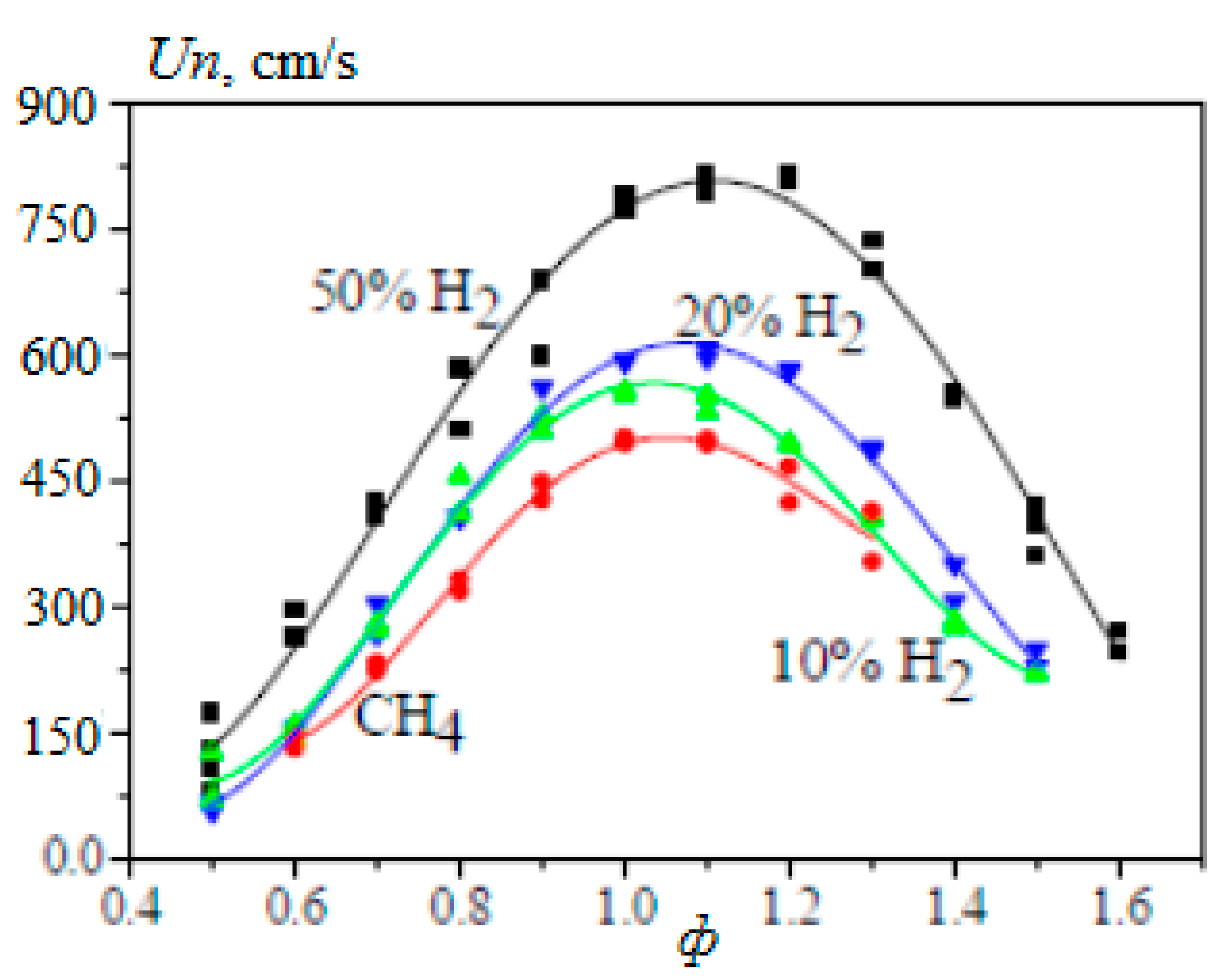
Calculation of the laminar velocity of the flame of methane–hydrogen–air mixtures under normal conditions, carried out in [23] on the basis of the detailed GRI-Mech mechanism, showed that it is always lower than that obtained by simple averaging of the values for methane and hydrogen, taking into account their molar fraction. For methane–hydrogen–air mixtures, depending on the hydrogen content, three areas can be distinguished: (1) with a predominance of methane (XH2 < 0.5); (2) transitional (0.5 < XH2 < 0.9); and region (3) in which methane inhibits the combustion of hydrogen (0.9 < XH2). In regions (1) and (3), the laminar combustion rate increases linearly with an increase in the hydrogen content. In region (1), the increase in the rate of laminar combustion of methane with the addition of hydrogen is insignificant, especially for lean mixtures. In region (3), the replacement of hydrogen by methane significantly reduces the combustion rate (Figure 9). These effects are more significant for rich mixtures. All these phenomena are associated with the concentration of H radicals and their role in branching and chain termination reactions.
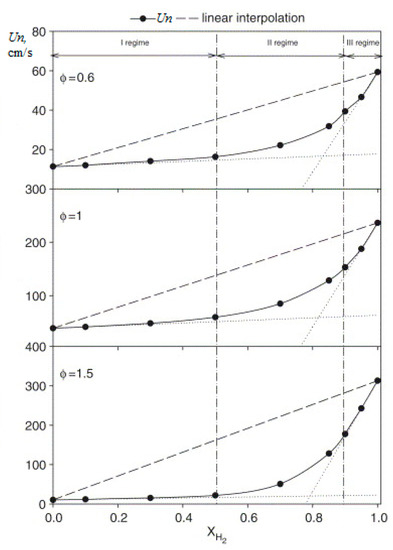
Figure 9.
Dependence of the normal flame velocity Un of methane–hydrogen–air mixtures under normal conditions and various values of φ on the hydrogen concentration (---linear interpolation) (Adapted with permission from ref. [23], Copyright 2007, Elsevier.).
These conclusions are confirmed by the results of work [18]. The only difference is that the boundary of the region of methane dominance in the combustion mechanism is expanded to a hydrogen concentration of 60%. The region of hydrogen concentrations from 60% to 80% is considered transitional, and the region of hydrogen concentrations above 80% is considered the region in which methane addition inhibits the hydrogen combustion. The authors of this work also attribute the high combustion rate at a high hydrogen concentration to a high concentration of H, O and OH radicals. However, as shown in [24], when the methane–air mixture is diluted with nitrogen, the absolute values of the increase in the combustion rate with the addition of hydrogen decrease.
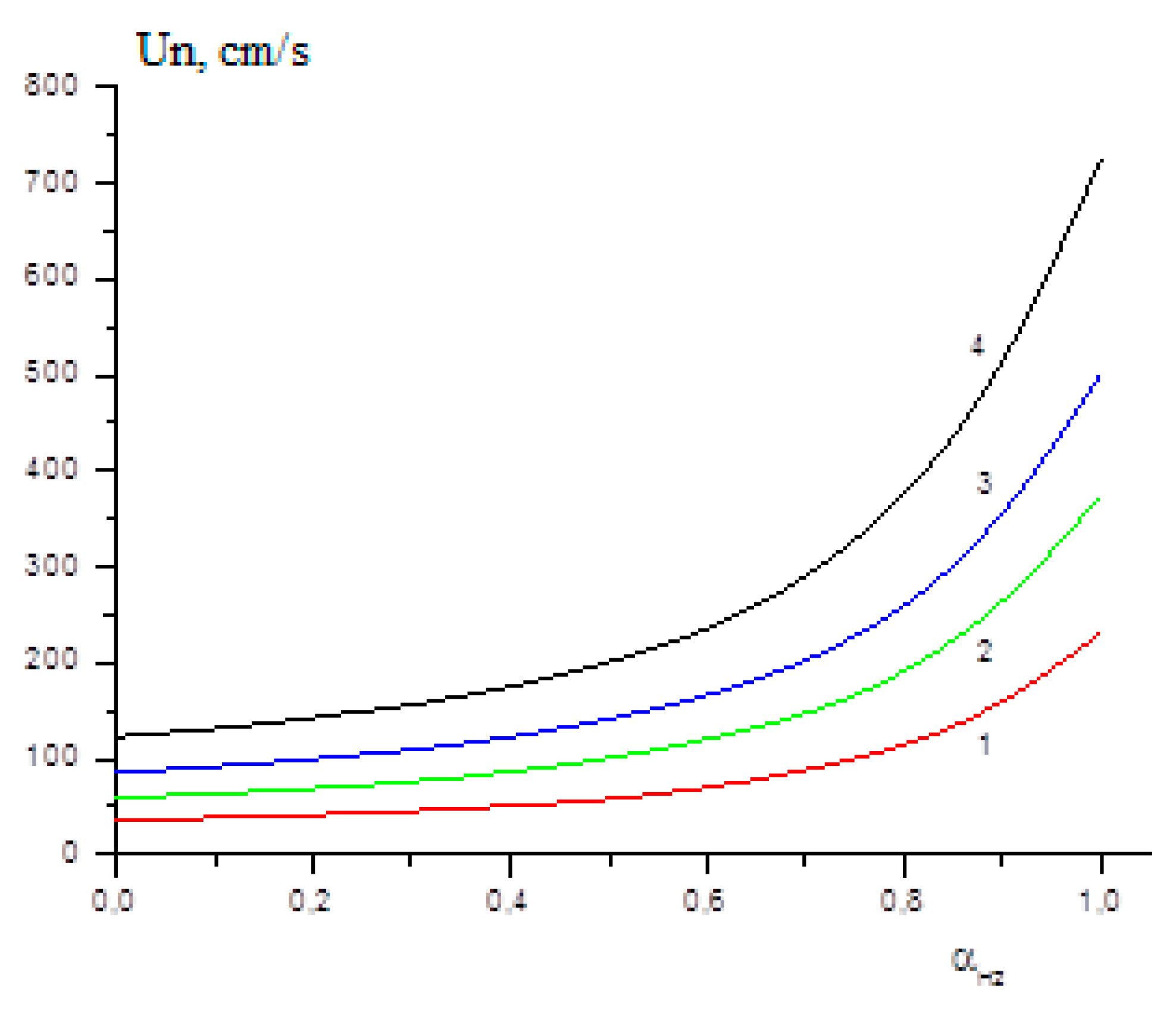
The influence of the initial temperature on the laminar flame velocity of methane–hydrogen mixtures was studied in [25]. Figure 10 shows the calculated dependences of the laminar flame velocity on the hydrogen content in the mixture at initial temperatures 300 K, 400 K, 500 K and 600 K. Figure 10 clearly demonstrates that, in full accordance with the previously obtained experimental data [26], the addition of hydrogen to the methane–air mixture at first has little effect on the laminar flame velocity, but as the concentration of hydrogen increases, a rapid increase of Un begins. The higher the temperature of the initial mixture, the earlier the sharp rise in Un begins.
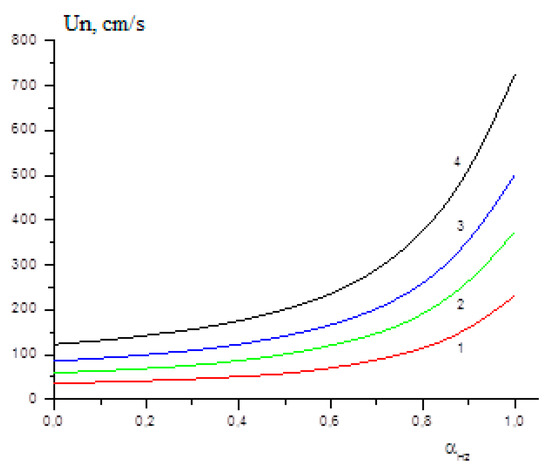
Figure 10.
Calculated dependence of the laminar flame velocity on the hydrogen content αH2 at initial temperatures T0 (K): 300 (1), 400 (2), 500 (3), 600 (4) (Adapted with permission from ref. [25], Copyright 2019, TORUS PRESS.).
The experimental data available in the literature on the combustion rate of methane–hydrogen mixtures, including the dependence of the combustion rate on pressure and temperature, were analyzed in the work [27]. Correlation expressions were proposed for mixtures with an H2 content of up to 50% for pressures up to 7.5 bars and initial temperatures up to 573 K.
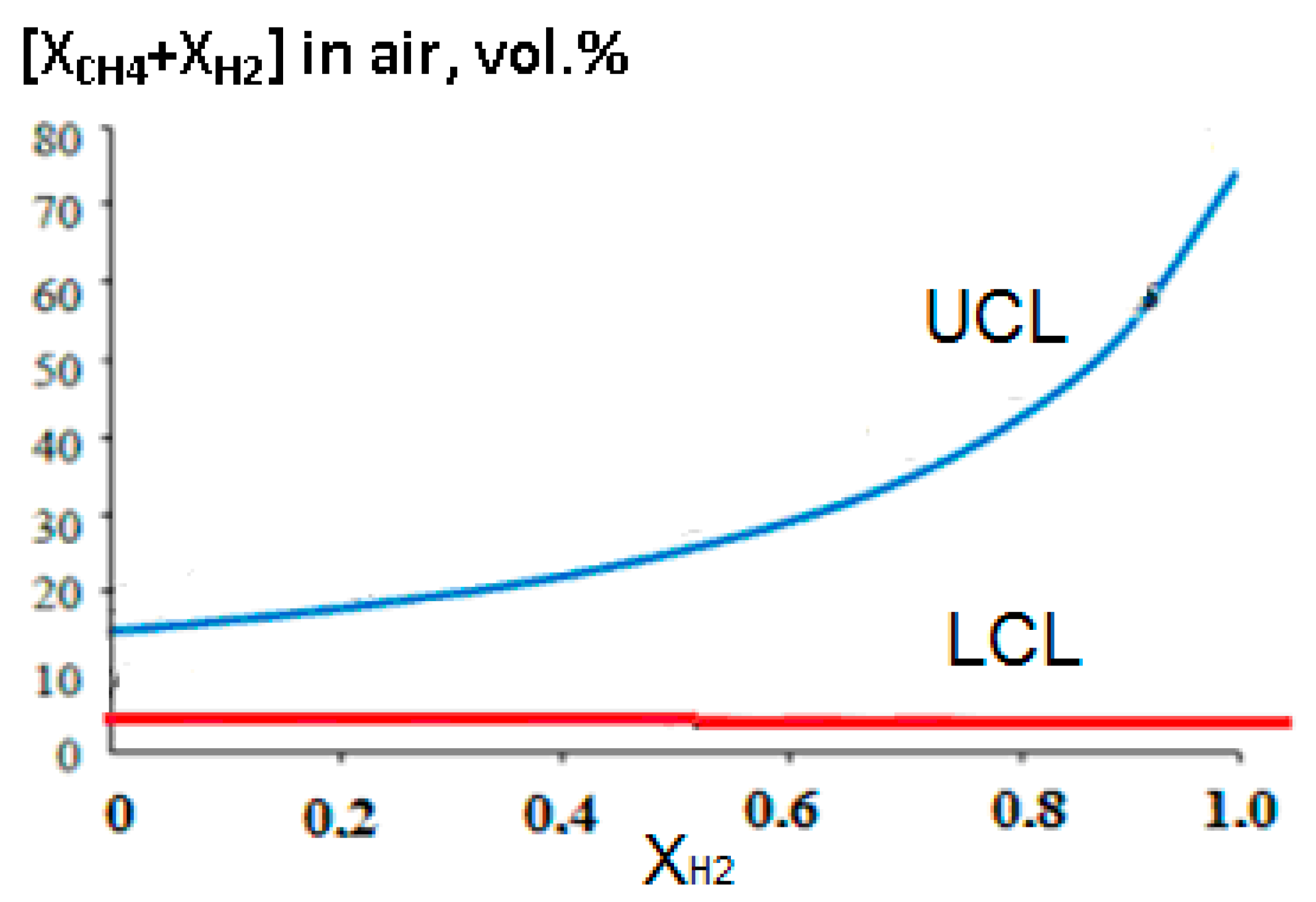
An increase in the concentration of hydrogen in the methane–hydrogen mixture leads to a significant expansion of the concentration limits of flame propagation, primarily due to an increase in the upper concentration limit (Figure 11).
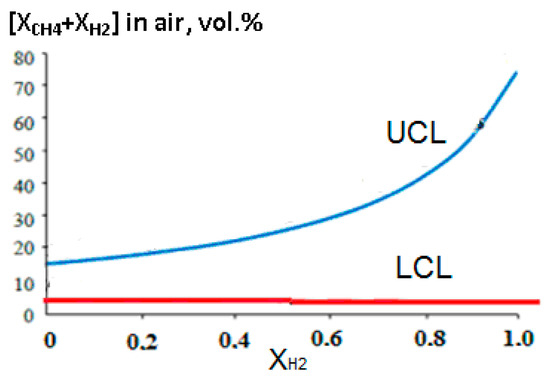
Figure 11.
Dependence of the upper (UCL) and lower (LCL) concentration limits of flame propagation in air on the volume fraction of hydrogen in the methane–hydrogen mixture.
Figure 10 and Figure 11 show that a hydrogen concentration of up to 40% vol. still has little effect on the combustion rate and the concentration limits of the flame propagation of methane–hydrogen mixtures. This allows such mixtures to be used on the same equipment and under the same safety regulations that have long been well established for working with natural gas.
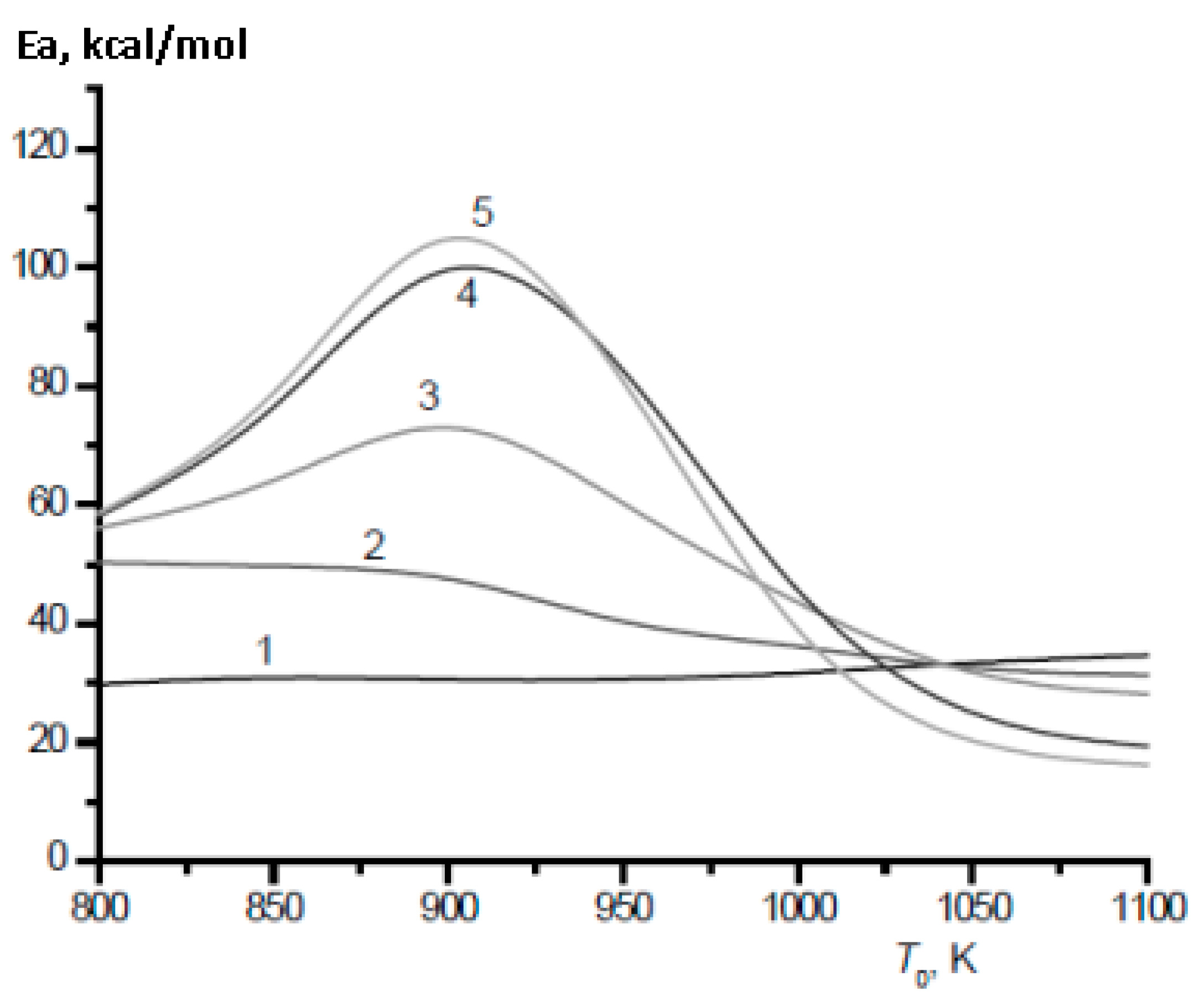
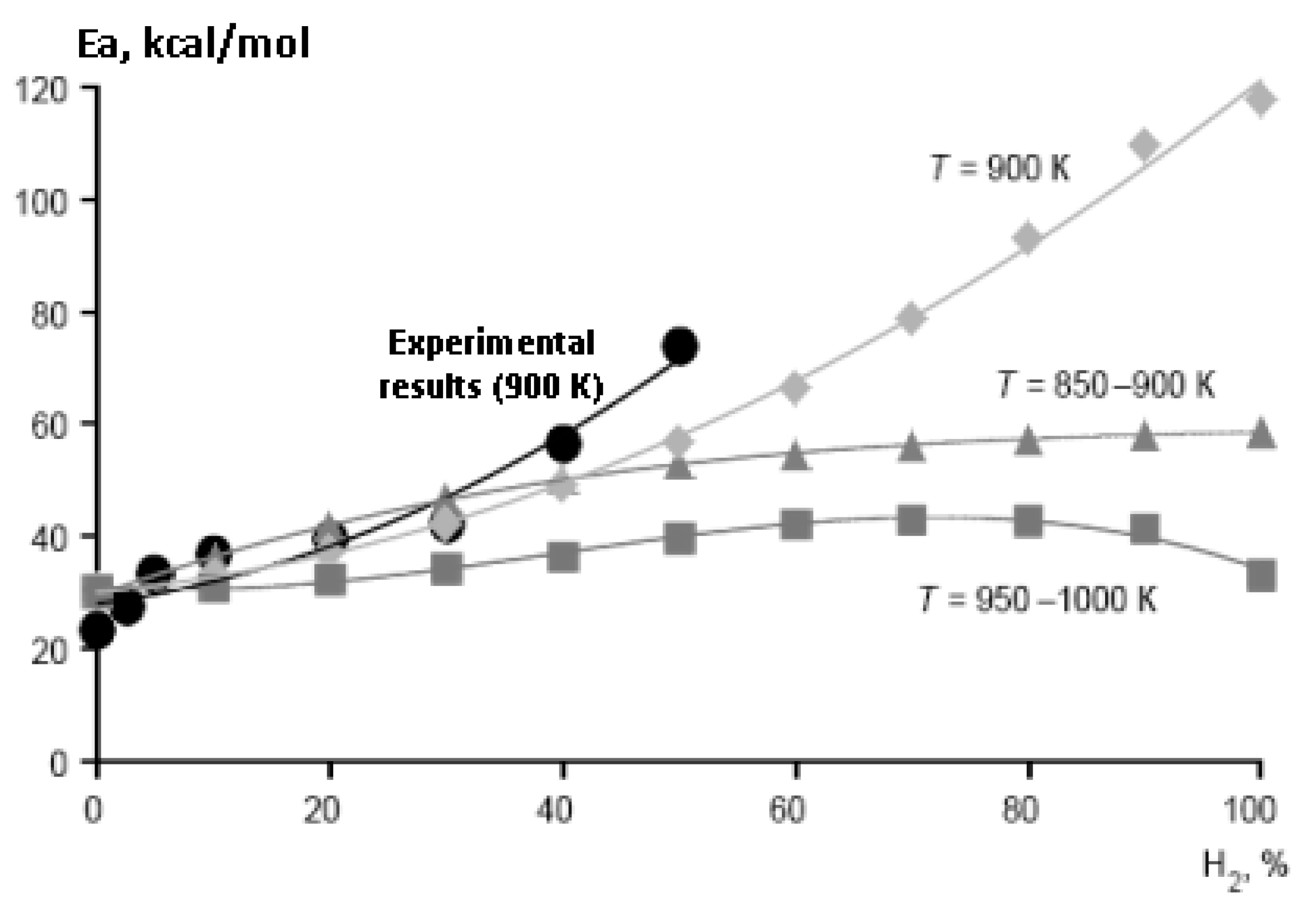
The most important characteristic of fuel mixtures is their ignition delay time. Unlike heavier alkanes, even a small addition of which to methane leads to a sharp decrease in its ignition delay time [28], the effect of hydrogen addition on the ignition of methane is more complex [29,30]. In the temperature range of 800–1000 K, which is characteristic of ignition processes in engines [31], there are serious changes in the mechanism of oxidation of both methane and hydrogen. This leads to a sharp change in their temperature dependence, in particular, the effective activation energy for the ignition of hydrogen and its mixtures with methane (Figure 12 and Figure 13).
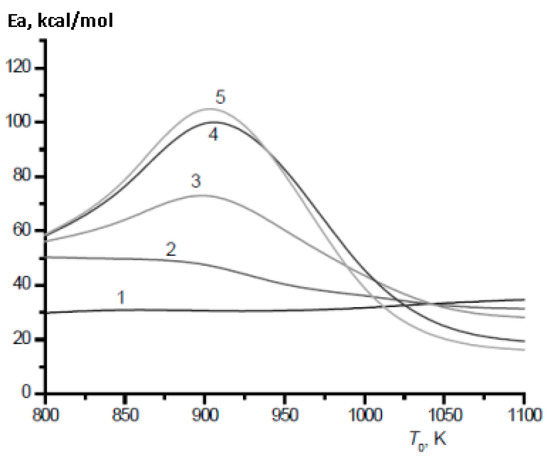
Figure 12.
Calculated temperature dependence of the effective activation energy of the ignition delay time for stoichiometric mixtures CH4–H2–air (Ea, kcal/mol) on the initial temperature T0 at various values of the H2 concentration (%): 0 (1), 40 (2), 70 (3), 90 (4), 100 (5). P0 = 1 bar (Adapted with permission from ref. [30], Copyright 2018, OBRACADEMNAUKA.).
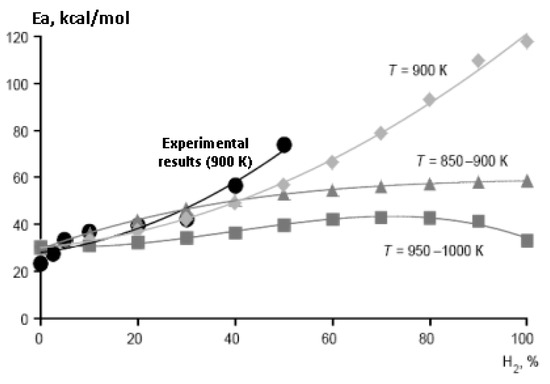
Figure 13.
Dependence of the effective activation energy (Ea, kcal/mol) of the ignition delay time in air of stoichiometric methane–hydrogen mixtures on the concentration of hydrogen in them at P0 = 1 bar, (●) experimental results (T0 = 900 K), calculation at T0 (K) 850–900 (▲), 900 (♦) and 950–1000 (■) (Adapted with permission from ref. [29], Copyright 2020, TORUS PRESS).
The observed complex phenomena require a more thorough analysis of the processes associated with the use of methane–hydrogen mixtures as a motor fuel. Comparative fuel characteristics of hydrogen, HCNG5 fuel mixture (5% hydrogen in methane), compressed methane and gasoline are presented in Table 1.

Table 1.
The fuel characteristics of hydrogen, HCNG5 fuel mixture (5% hydrogen in methane), compressed natural gas (CNG) and gasoline (Reprinted from ref. [32]).
It is also necessary to take into account that H2 enrichment of CH4/air premixed flames affects the flow field in both quantitative and qualitative terms and causes a transition from wrinkled to breakthrough regime of combustion. This transition is accompanied by faster increase in flame surface area and improves the burning quality [33,34].
3. The Practice of Using Hydrogen in Power Plants and Transport
Hydrogen, as well as methane–hydrogen mixtures (MHM), can be used directly as a fuel for power plants, providing high efficiency and good environmental performance. It is believed that mixtures of compressed natural gas with hydrogen will allow, with a minimum increase in capital investments and a minimum of additional equipment, to eliminate a number of disadvantages inherent in traditional gasoline and diesel engines [35]. The main difference between gaseous fuels and liquid fuels is that the former mix better with air, resulting in more complete combustion of the mixtures with significantly less carbon deposits and emissions of toxic products of incomplete combustion. According to available data, the use of methane–hydrogen fuel contributes to the reduction of toxic emissions, greenhouse gas emissions (by 8–15%) and reduced fuel consumption.
In the early 1980s, the use of a mixture of hydrogen with natural gas at ratios of 100:0, 80:20, 50:50 and 0:100 as a fuel for automobile engines was tested by the AVL company [36]. Subsequently, in order to improve the combustion characteristics and reduce the toxicity of engine exhaust gases, fuel mixtures of hydrogen and natural gas of various compositions were studied. In many countries, methane–hydrogen mixtures are successfully used in vehicles with an internal combustion engine. Tests of the possibility of using MHM with a hydrogen content of up to 20% were also carried out in housing and communal services, including such domestic appliances as cooking burners [37,38,39].
Methane–hydrogen fuel, used mainly in the automotive sector instead of methane, is a gas mixture consisting of 10–30% vol. hydrogen and 70–90% vol. methane, and was given its own name, Hythane. The presence of hydrogen improves the efficiency of fuel combustion in the engine and reduces CO2 and NOx emissions. The second direction of the Hythane application is the addition to the network of natural gas in small volumes (typically H2 concentration < 10%). Currently, Hythane is mainly produced from fossil sources, but the use of alternative renewable sources can further reduce greenhouse gas emissions.
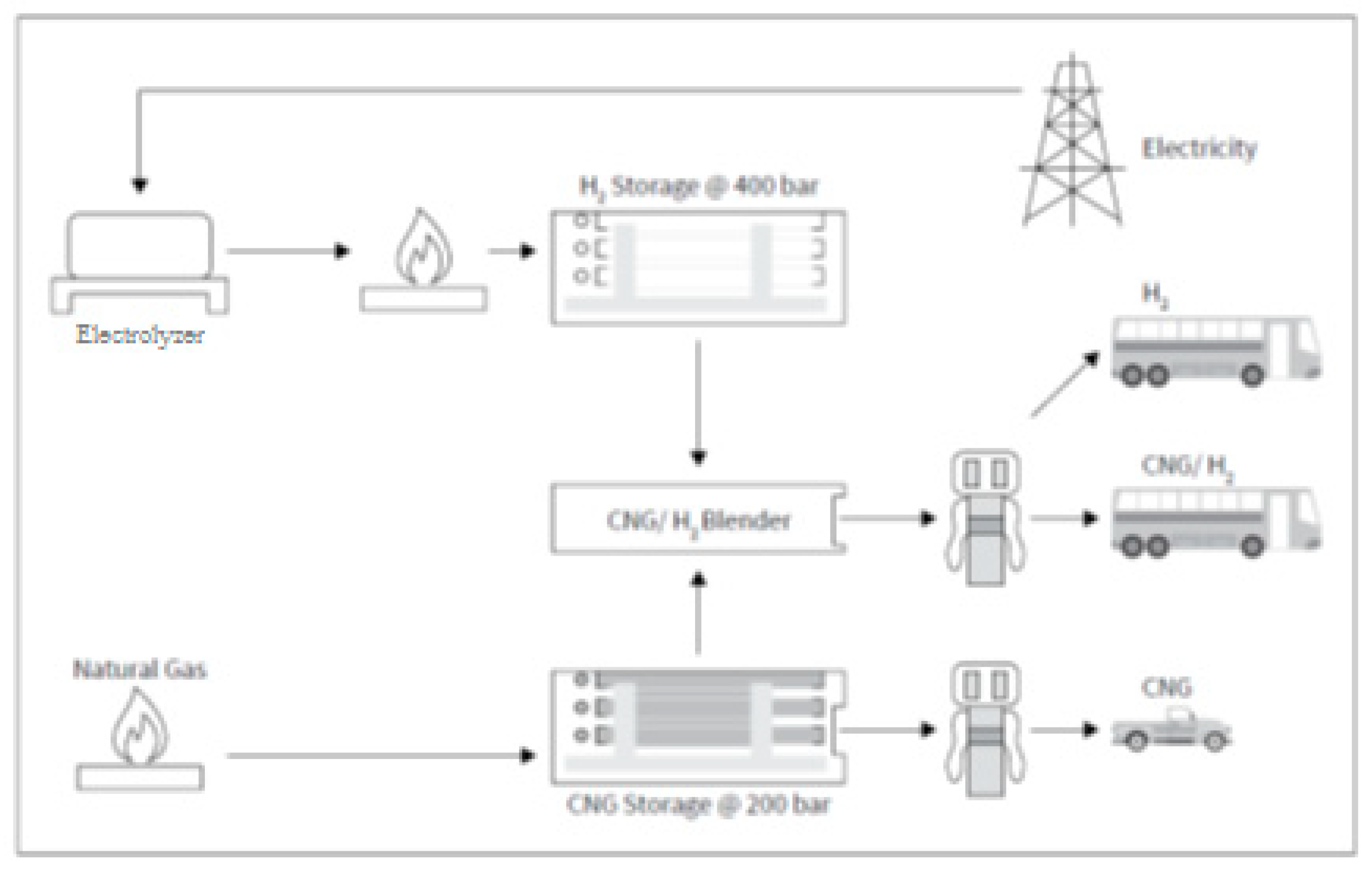
Figure 14 shows an integrated model of a filling station, where the fuel is compressed natural gas (CNG) and a compressed methane–hydrogen mixture (HCNG). These combo stations require the following main components: NG source, H2 source (electrolyzer), CNG/H2 mixer, CNG storage tanks, and CNG and HCNG dispensers [32].
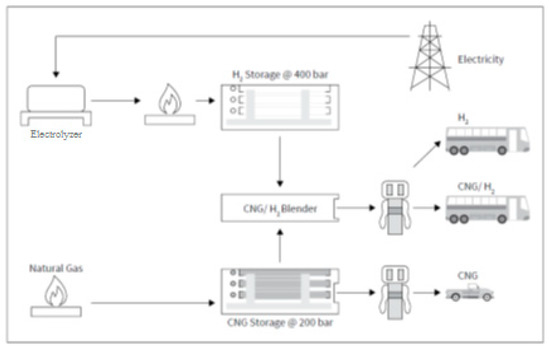
Figure 14.
Scheme of an integrated filling station for compressed natural gas and methane–hydrogen mixtures (Reprinted from ref. [32]).
The possibility of using methane–hydrogen mixtures in internal combustion engines with compression ignition was studied in [40,41,42,43], and in internal combustion engines with spark ignition in [44,45,46]. Already in an article dated by the end of the last century [47], it was experimentally confirmed that the performance of a single-cylinder internal combustion engine with spark ignition, running on methane, can be significantly improved by transition to a mixture of hydrogen and methane.
The use of methane–hydrogen mixtures in internal combustion engines at quite moderate financial costs can significantly reduce CO2 emissions. On the example of a prototype internal combustion engine adapted to work with such mixtures, a technical assessment was given of the benefits of the simultaneous use of several types of fuel. The storage and distribution methods and combustion characteristics of these mixtures were also assessed, taking into account technical conditions, safety issues, regulations and infrastructure features [48]. Information is provided on international standards and regulatory documents for internal combustion engines running on gas fuel, as well as on the advantages of using multicomponent fuels in them.
Investigation of methane–hydrogen mixtures with hydrogen content of 5, 10 and 15% vol. have been completed in [49]. Comparison of the performance characteristics of internal combustion engines on such fuel mixtures showed that:
- -
- Adding 15% hydrogen to CNG engines on lean fuel increases ignition time to 1.46 ms compared to 1.20 ms for natural gas;
- -
- Methane–hydrogen–air mixtures have a significantly increased braking efficiency (adding only 5–7% hydrogen is enough) and power output (an increase of 5–10% compared to natural gas), i.e., hydrogen additives are especially effective in lean mixtures;
- -
- When testing methane–hydrogen mixtures, a strong decrease in CO emission was observed (with the addition of 15% hydrogen by volume, emission was not observed at all, and for an engine with a speed of 3500 rpm when operating on natural gas, CO emission was 0.12–0.40%), i.e., mixed hydrogen fuel is more environmentally friendly.
Work was carried out in which the combustion performance of two methane–hydrogen mixtures (5 and 20% by volume of methane in hydrogen, which corresponds to 30 and 65% by weight of methane) was compared with the operation of an engine running on hydrogen [50]. The study confirmed that for an engine calibrated for hydrogen, the transition to operation on methane–hydrogen mixtures requires only small adjustments to the ignition delay time of the mixture.
After numerous studies, a composite gas fuel called Hythane, which is usually a mixture of 20% hydrogen and 80% natural gas, has been established in world practice as a typical methane–hydrogen fuel [51]. It is believed that, in principle, any methane–hydrogen mixtures containing 8–30% hydrogen can reduce emissions of nitrogen oxides (NOx), greenhouse gases and CO2 without major changes to the existing natural gas infrastructure. With a specific fuel mixture for many types of vehicles running on compressed natural gas, no modification is required for transition to the Hythane fuel mixture.
Among the advantages of using Hythane compared to natural gas, in addition to a significant reduction in NOx emissions when burning lean mixtures, there is a significant increase in the velocity of flame propagation. The Hythane has lower ignition energy, since for hydrogen it is an order of magnitude less than for methane. The presence of hydrogen also accelerates the combustion process and increases the efficiency of catalytic exhaust gas treatment at low temperatures.
4. Emission and Performance Characteristics of Methane–Hydrogen Mixtures
The issues of reducing the emission of environmental pollutants from the exhaust gases of internal combustion engines through the use of fuels with hydrogen additives are analyzed in the review [52]. It is noted that at present the most effective way to use hydrogen additives in internal combustion engines are dual-fuel systems. Low-temperature combustion in such engines makes it possible to use all types of modern fuels, and their thermal efficiency exceeds that of classic engines, which makes it possible to further reduce CO2 emissions. Further research should be aimed at reducing the emission of other greenhouse gases such as N2O and CH4.
The total emission of CO2 in the entire technology for the production and use of MHM will depend on the method of hydrogen production. For example, if steam reforming of methane, which currently produces more than 70% of the world’s hydrogen, is not combined with carbon capture and storage technologies, then the total CO2 emissions will still be very high.
Because hydrogen enrichment promotes faster and more complete combustion of methane, it reduces exhaust emissions of methane and incomplete combustion products. Internal combustion engines running on HCNG, unlike fuel cells with a hydrogen proton exchange membrane, do not pollute the air with carbon monoxide [51].
The operation of ICE on HCNG is similar to their operation on natural gas, which allows the use of already existing infrastructure. In fact, ICE for HCNG operation is modified versions of current CNG engine with the potential to switch from one type of fuel to another. The process of obtaining a mixture of HCNG from available natural gas and hydrogen at the point of use can be carried out on standard equipment. The HCNG and Hythane have lower emissions than natural gas and diesel fuel. Therefore, they can be used in the same types of engines that use CNG, and they can also be considered as competitive technology that ensures the transition from petroleum to hydrogen fuel.
The results of using mixtures of methane and hydrogen of various compositions, both rich in natural gas (80% CH4:20% H2) and hydrogen (20% CH4:80% H2), showed [53] that their use in ICE makes it possible to overcome a number of disadvantages observed during the separate combustion of their constituent gases. These problems are the low flame propagation velocity inherent in methane combustion, or the high probability of flashback and detonation inherent in hydrogen combustion. In addition, when operating on methane–hydrogen mixtures under all engine conditions, a decrease in the emission of particulate matter with exhaust gases is observed compared to diesel fuel. However, the emission of CO and unburned hydrocarbons when using methane–hydrogen mixtures in comparison with diesel fuel turned out to be higher. With an increase in the hydrogen content in the methane–hydrogen mixture, the emission of NOx increased.
The effect of hydrogen on the performance and emission characteristics of an SI engine operating on a 100:0, 95:5, 90:10, and 80:20 methane–hydrogen mixture was studied in [54]. The experiments were carried out on a modified 3.9 L Isuzu engine with a compression ratio of 12.5 at 1500, 2000 and 2500 rpm. The brake power of the engine, the brake thermal efficiency, the composition of combustion products and emission parameters were determined at 5, 10, 15 and 20 degrees of crank angle before top dead center ignition timings. The results showed that the maximum engine power is achieved using fuel containing 5% hydrogen in natural gas. The ignition delay time with an increase in the concentration of hydrogen in the mixture and the engine speed decreased. The pressure in the cylinder and the rate of heat release approached the maximum values with an increase in hydrogen content in the mixture and a decrease in engine speed. Emissions of unburned hydrocarbons and CO turned out to be below the values determined by the Euro-5 and Euro-6 standards.
The higher efficiency of using Hythane (80% natural gas + 20% hydrogen) in a carbureted engine compared to gasoline in terms of reducing harmful emissions and increasing its power is shown in [55]. In [56], three mixtures with a hydrogen concentration in natural gas of 5%, 10% and 15% (HCNG5, HCNG10 and HCNG15, respectively) were studied in a single-cylinder ICE. It has been established that an increase in the hydrogen content in natural gas increases the thermal efficiency of the engine. The most efficient mixture was HCNG10, which can be used without any modifications to the engine. It has been shown that the emissions of NOx and unburned hydrocarbons when the engine is running on a methane–hydrogen mixture are higher than when running on natural gas. At the same time, studies with six-cylinder heavy duty engines running on natural gas and a mixture with 25% vol. hydrogen content showed a slight extension of the limit of admissible depletion of the mixture when switching to the specified fuel mixture. At the same time, changes in the emissions and knocking margins were also small [57].
The results of experiments with Hythane in a prototype single-cylinder supercharged engine with various ignition sources (spark and laser) showed that when the initial pressure is increased from 5 to 10 bars, ignition occurs faster, while the duration of fuel burning increases. Increasing the hydrogen content in the mixture reduces the ignition time and combustion duration of fuel by 10% for both pressures tested. A mixture containing 30% hydrogen was characterized by rapid ignition [58].
Review [59] analyzes studies of the effect of hydrogen additions to natural gas on combustion processes and emissions, as well as on expanding the combustion limits of lean fuels and improving their combustion conditions. Despite a decrease in engine power and an increase in heat transfer to the cylinder walls, the expansion of the combustion limits of lean mixtures of natural gas due to the addition of hydrogen makes it possible to increase combustion efficiency and reduce the emission of NOx. A study on emissions and carbureted engine performance (braking power, thermal braking efficiency) is reported when running on Hythane (80% natural gas + 20% hydrogen), gasoline–methanol and gasoline–ethanol fuels. The results obtained were compared with the data of engine operation on gasoline, as a result of which the effectiveness of the use of methane–hydrogen mixtures was confirmed [60].
There is also great interest in mixtures of hydrogen with biogas, which usually consists of 55–65% methane and 30–40% CO2. However, the presence of CO2 generally adversely affects the combustion process, reducing the laminar flame velocity and narrowing the ignition limits [61,62,63,64].
When modeling the combustion of mixtures of methane with hydrogen or biogas in a controlled auto-ignition engine, the authors of [65] used the radical chain mechanism GRI-Mech 3.0. The simulation of the operating conditions of a single-cylinder ICE was carried out using the ANSYS Chemkin 17.0 code. The kinetics of chemical processes, the efficiency of the engine and the composition of emissions were studied. It was shown that when using methane–hydrogen mixtures, it is possible to operate at a lower equivalence ratio of 0.38 compared to that required for operation on methane (0.4). The addition of 10% H2 to methane reduces the required initial temperature from 468 K to 448 K, and the addition of 20% H2 reduces it to 443 K. The simulation results are consistent with the results of studies on engines with spark ignition.
The possibility of using methane–hydrogen mixtures for operation under low-temperature regimes of the flameless combustion, which makes it possible to significantly reduce NOx emissions, was studied in [66]. To optimize the design of a semi-industrial combustion chamber with an increase in the concentration of H2, the operation of a recuperative flameless burner with a power of 20 kW on methane–hydrogen mixtures was simulated. The influence of fuel dilution with CO2 and H2O additives was also studied, which showed that such dilution slows down the process of mixing the fuel and oxidizer, thereby facilitating the transition to flameless combustion conditions, which makes it possible to increase the hydrogen content in the mixture. If hydrogen concentrations in the mixture exceed 50%, then it becomes very difficult to work in flameless combustion conditions. However, due to the selection of the optimal combination of the length of the fuel injection nozzle and the corresponding equivalence ratio, it is possible to pass to the conditions of flameless combustion for fuel mixtures of the widest composition.
Mention should also be made of maritime transport, which is a key component of the world economy and accounts for 80–90% of international trade. Given the growing interest in ocean environmental issues, there is a growing interest in exploring the possibility of using marine engines on hydrogen/natural gas/diesel mixtures [67,68]. To analyze the effect of hydrogen concentration in these mixtures on the characteristics of their combustion and the emission of exhaust gases from marine vessels equipped with such engines, modeling and experimental testing were carried out [69]. As in all other cases, with an increase in the hydrogen content in the mixture, the velocity of laminar flame propagation increases, and the ignition delay time decreases. With a high hydrogen content in fuel mixtures, the pressure and temperature in the marine engine cylinder increase, while the combustion temperature region expands. Therefore, such a tri-fuel engine (H2 + CH4 + diesel) has a faster response and high power compared to a dual-fuel engine (CH4 + diesel). With an increase in the hydrogen content in the mixture, the emission of nitrogen oxides increases, while the emission of CO and CO2 decreases. The possibility of reducing pollutant emissions into the atmosphere makes the practical use of ships with three-fuel engines attractive in the shipping industry.
Of particular interest is the use of methane–hydrogen mixtures to solve problems associated with the operation of diesel ICE. Despite the fact that the performance of modern diesel fuels has improved markedly in recent years, the operation of many vehicles on diesel fuel is still associated with serious disadvantages. They are high emission of harmful substances into the atmosphere, low fuel efficiency, high fuel consumption, low level of their frost resistance, etc. Therefore, in order to reduce the impact of these problems, many countries have decided to completely abandon diesel engines by 2030, or at least ban their use in large cities. These issues are discussed in detail in the review [70]. The main task of optimizing the operation of diesel engines is the need to simultaneously combine low emissions of harmful gases with efficient engine operation [71]. The use of MHM in diesel engines is an effective method of increasing the fuel combustion rate. The main task is the correct selection of the ratio H2: natural gas [72,73]. The model of operation of a single-cylinder diesel engine with a power of 3.5 kW on a combined fuel consisting of a mixture of Hythane10 (as additional fuel) and diesel fuel (as the main fuel) showed the possibility of increasing the thermal efficiency of the engine by 11% and reducing NOx formation by 13% [74]. However, compared to diesel fuel, the operation of a diesel engine on Hythane10 was accompanied by a significant increase in emissions of unburned hydrocarbons (by 63%).
In [75], a methane–hydrogen mixture consisting of 90% methane and 10% hydrogen (Hythane10) was added to liquid diesel fuel in amounts of 0%, 25%, and 50%. The results obtained showed that smoke emission decreased by 95.44%. Although these additives did not prevent the increase in unburnt hydrocarbons, CO and NOx emissions, they did increase cylinder pressure and heat generation rate.
To study the operating and technical and economic characteristics of fuel mixtures and optimize their composition, mathematical modeling methods are widely used. In particular, mathematical models are created based on the combustion mechanism of methane–hydrogen mixtures. Most often, the combustion of mixtures with a hydrogen content of less than 15% is modeled using the GRI-Mech 3.0 mechanism [76,77,78].
Despite a large number of experimental and theoretical studies on the use of methane–hydrogen mixtures in ICE, there are still many issues, for example, the correct selection of hydrogen concentration to avoid a rapid increase in pressure. It is also important to select such parameters as the optimal combination of hydrogen additives, excess air ratio, ignition delay time and other parameters. This is especially important for cogeneration ICE with spark ignition, combining the production of thermal and electrical energy. To study this issue, a numerical model of a cogeneration engine equipped with a turbocharger was developed [79]. An algorithm for searching the optimal conditions under which the operation of the engine is characterized by the highest energy production with minimal fuel consumption and minimal environmental pollution is proposed. It has been established that cogeneration systems with additions of up to 15% hydrogen to natural gas will be viable if the engine is operated at lower air/fuel ratios. In this case, it will be possible to avoid a sharp rise in pressure and excessive formation of toxic emissions.
The effect of H2 and CH4 concentrations on the ignition delay time and laminar flame velocity during the combustion of CH4/H2 and multicomponent mixtures based on synthesis gas was studied in [80] using an original kinetic mechanism. The results obtained were compared with experiments and calculations by the GRI Mech 3.0 mechanism. It has been established that the reactivity of mixtures increases only in the case of an increase in the concentration of CH4 and H2. This leads to an increase in the formation of OH radicals and, accordingly, to an increase in the velocity of the laminar flame and a decrease in the flashback of the flame in the fuel mixture. It has been established that at high pressure in the system, methane is consumed slowly, but the presence of hydrogen increases the rate of its consumption. The study of the combustion process of various mixtures of H2/CH4/CO showed that the reactions:
predominate in methane-rich mixtures, while the following reactions play an important role in the combustion of hydrogen-rich mixtures:
CH2O + OH = HCO + H2O
CH4 + HO2 = CH3 + H2O2
CH4 + HO2 = CH3 + H2O2
OH + HO2 = H2O + O2
HO2 + H = OH + OH
HO2 + H = OH + OH
In [81], based on the results of numerical simulation, it was assumed that the presence of hydrogen in methane–hydrogen mixtures increases the depth of methane oxidation, thereby reducing the rate of formation of solid particles (soot) in the exhaust gases of engines.
5. Transportation Logistics of Hydrogen and Methane–Hydrogen Mixtures
The development of hydrogen energy and the hydrogen market is constrained by the lack of economically viable technical solutions for a full-cycle hydrogen economy, including for transporting large volumes of hydrogen over long distances. A serious logistical problem is the lack of an appropriate infrastructure for the storage, transportation and distribution of industrially significant volumes of hydrogen [82], for the creation of which there are still no practically acceptable solutions. Most of the metals from which existing gas pipelines are made are subject to destruction under the influence of hydrogen diffusion in them, especially at high pressures. This imposes much more stringent requirements on the manufacture of pipelines for transporting hydrogen compared to pipelines for transporting natural gas [83] and dramatically increases their cost. Therefore, already existing gas pipelines cannot be used to transport hydrogen, and the cost of special pipelines will be many times higher than the existing ones. As a more practical, economically justified and technologically safe alternative, the possibility of transporting hydrogen through pipelines in the form of methane–hydrogen mixtures (MHM) is discussed. However, the significantly higher energy costs for hydrogen compression compared to natural gas [82] (Figure 15) and the lower volumetric energy content in such mixtures (Figure 16) raise the issue of the economic feasibility of such a hydrogen transportation system [12].
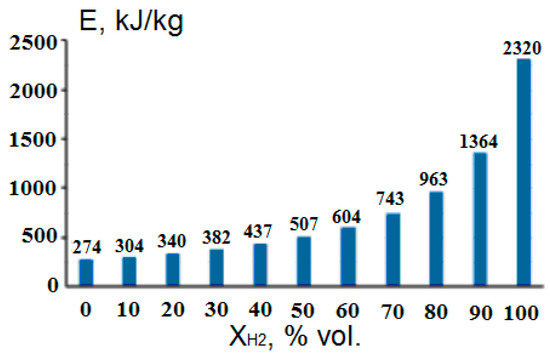
Figure 15.
Dependence of energy costs for compressing a methane–hydrogen mixture with a pressure of 1 MPa on the concentration of hydrogen in it (Adapted from ref. [82]).
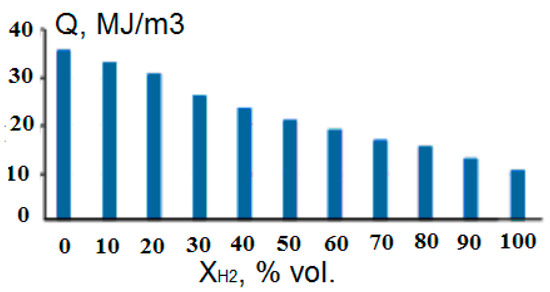
Figure 16.
Dependence of the calorific value of methane–hydrogen mixtures on the concentration of hydrogen (Adapted from ref. [82]).
In parallel with the discussion of the prospects for creating a special hydrogen infrastructure, which will require very large initial investments, research continues on the possibility of less expensive transportation of methane–hydrogen mixtures using existing gas distribution systems and their equipment (compressors, storage tanks, pipelines). A technical and economic analysis of the features of such infrastructure is presented in a number of works [84,85,86,87].
The possibility of adding hydrogen directly to the existing gas distribution system is being considered by the governments of a number of countries. Since the chemical and physical properties of hydrogen and natural gas are very different, the European Commission has funded studies on the possibility of using the existing infrastructure for the transport and distribution of natural gas with hydrogen additives (Project Contract No. SE56/CT/2004/502661). In particular, the influence of the hydrogen content in the hydrogen/natural gas mixture on the value of the minimum ignition energy of the mixture was experimentally studied [88].
It has turned out to be difficult to give recommendations on the maximum allowable concentration of hydrogen in methane–hydrogen mixtures for all parts of the European gas infrastructure. However, since most natural gas transportation systems are applicable to mixtures with hydrogen concentrations up to 10%, the authors of [89] proposed recommendations on the maximum allowable hydrogen concentration in methane–hydrogen mixtures:
- (a)
- 2% hydrogen if the system is connected to a natural gas filling station;
- (b)
- 5% hydrogen if no filling station, no gas turbines and no gas engines with a hydrogen specification less than 5% are connected;
- (c)
- 10% hydrogen if no filling station, no gas turbines and no gas engines with a hydrogen specification less than 10% are connected.
In this case, for hydrogen concentrations of 5% and 10%, the correspondence of Wobbe index and methane number must be taken into account. In any case, the introduction of hydrogen into the piping system must be strictly controlled. The sudden increases in the hydrogen concentration during supply should not exceed 2%/min, so as not to exceed its maximum allowable concentration.
In various countries, the permissible content of hydrogen in mixtures with natural gas ranges from 0.1% (Belgium, New Zealand, Great Britain and the USA) to 10% in Germany and 12% in the Netherlands, which is regulated by the national safety standards of these countries. It is estimated that the introduction of 20% hydrogen into a large European gas transmission system could reduce CO2 emissions by 60 million tons per year (by 7%) [90]. Since hydrogen has a lower volumetric energy content compared to natural gas, additions to the latter 5% vol. hydrogen replaces only 1.6% of natural gas demand [91]. Table 2 presents the characteristics of mixtures of compressed natural gas and methane–hydrogen mixtures with different hydrogen content.

Table 2.
Characteristics of CNG and HCNG mixtures with different hydrogen content (10%, 20% and 30%) (Reprinted from ref. [32]).
Figure 17 shows the sensitivity of elements of the natural gas transportation system and infrastructure to the hydrogen additives. At low values of the volume fraction of hydrogen, only a slight adjustment of the operating conditions is required, which is a very important advantage. At higher hydrogen concentrations, gas distribution systems will already need to be upgraded, which will result in additional costs, for example, those associated with the need to manufacture pipes from special materials. That is, the introduction of hydrogen into the existing natural gas infrastructure is only feasible at low volume concentrations, since low additional capital investments are sufficient for this.
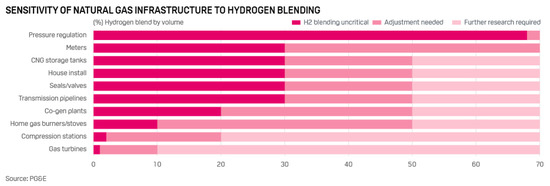
Figure 17.
Sensitivity of natural gas infrastructure to hydrogen blending (Reprinted from ref. [91]).
The influence of the composition of methane–hydrogen mixtures on the materials of pipelines of the existing natural gas infrastructure was experimentally studied. The results obtained were compared with the literature data [92]. At a pressure of 2 bar in an environment of 100% hydrogen, a mixture of 20% hydrogen with methane and 100% methane, materials such as polyethylene, steel, cast iron, copper and brass worked. At the same time, there was no negative effect of a mixture of 20% hydrogen with methane on the characteristics of metal and polymer (PE80 pipe polyethylene) materials, as well as on the places of connecting of pipelines. It was concluded that all tested materials of the existing gas transmission pipeline infrastructure are suitable for transporting mixtures of natural gas and hydrogen in the range of hydrogen concentrations up to 20% and pressures up to 2 bars.
The possibility of using the existing gas distribution infrastructure for refueling vehicles with hydrogen is discussed in the review [93]. Two options are considered: adaptation of the existing infrastructure for the use of hydrogen and the direct introduction of hydrogen into natural gas pipeline networks. Hydrogen can be delivered to filling stations as compressed gas or liquid, as well as through pipelines. As one of the possible ways of supplying hydrogen, the supply of natural gas through pipelines to filling stations is also considered, where hydrogen can be produced from it in small-scale methane steam reformers. This eliminates the need for special transportation of hydrogen to the filling station. At filling stations, the methane–hydrogen mixture can be used directly as a fuel, or hydrogen can be extracted from it for use in fuel cells. The authors believe that the pipeline transportation of methane–hydrogen mixtures with a hydrogen concentration of 5–15% vol. is technically feasible. However, in each specific case, a preliminary assessment of the cost of such a solution is necessary. It should also be taken into account that the optimal ratio of hydrogen and natural gas is limited by many factors, such as the volume of consumption in the market, technical and economic indicators, features of regional networks, etc. The authors come to the conclusion that, most likely, it will be necessary to create a special hydrogen gas distribution infrastructure, consisting of new hydrogen filling stations. The design and construction of such stations will be regulated by international standards. They should include hydrogen storage and cooling systems, compressors and dispensers for refueling vehicles with hydrogen–methane fuel. Japan is already building 100 hydrogen filling stations along highways connecting Japan’s four largest cities. In Germany, it is planned to build 400 by 2025 with the production of hydrogen directly at filling stations based on wind and solar energy [94].
When developing mathematical models for the safe addition of hydrogen to existing gas distribution systems, it is important to take into account many factors: the distance over which the gas is transferred, the gas pressure at the inlet, the rate of hydrogen addition to the system, the gas flow rate, the ambient temperature, the geometric parameters of the pipeline, etc. [95,96]. A three-dimensional statistical spiral model of a mixer for creating methane–hydrogen mixtures has been developed, the study of which has shown that the optimal number of mixing devices used is three [97]. Taking into account the influence of the design of mixers for creating fuel mixtures allowed the authors to study the influence of the process of mixing gases on such indicators as the coefficient of variation and pressure drop in the pipeline.
Hydraulic studies of the transportation of large flows of hydrogen through pipelines at a mass flow rate from 0.3 to 3.0 kg/s (volume flow rates from 12,000 m3/h to 120,000 m3/h (STP), respectively) have been carried out [98]. The gas flow in the pipeline was modeled, as well as the thermodynamic parameters of the transportation of both hydrogen and its mixtures with methane. The analysis showed that the maximum hydrogen content in natural gas should not exceed 15–20%, otherwise the high calorific value of the gas mixture is reduced to the values of the lower limit of 35 MJ/m3 STP, defined by most standards. In the case of using a mixture consisting of 85% methane and 15% hydrogen, the pressure at which gas is delivered to the consumer turned out to be 10% lower than for methane. Modeling has shown the real possibility of efficient transportation of hydrogen over long distances.
Since the presence of hydrogen in a mixture with natural gas affects the dynamics of flow in the pipeline, in the Transitgas project (Switzerland) using the Aspen Hysys Version 9 and Aspen Plus code, the hydraulics of transporting the gas mixtures along a 94 km long pipeline were simulated for natural gas and up to 10% hydrogen [99]. It is shown that for a hydrogen concentration in the mixture up to 2%, an increase in the hydrogen content increases the viscosity of the fluid, and over 2% it reduces viscosity. The presence of hydrogen has a strong effect on the phase state of natural gas and increases pressure losses when the mixture moves through the gas pipeline. The work also investigated the effect of the height of the gas pipeline above the ground when lowering and raising it by 25 m. The results showed that with increasing installation height, pressure losses occur along the entire length of the gas pipeline. The presence of hydrogen in mixtures increased the critical pressure in the pipeline and reduced the critical temperature of the fuel mixture.
Transportation of hydrogen in the form of a methane–hydrogen mixture through the existing gas distribution system of natural gas has the problem of hydrogen separation from the mixture at the points of its delivery to the consumer. The traditional technology for hydrogen separation is pressure swing adsorption. The specialists of the NREL laboratory (National Renewable Energy Laboratory, USA) found that when using this technology, the cost of extracting 80% of hydrogen from a mixture with its content of 10% is 3.3–8.3 USD/kg. To separate hydrogen from pipeline networks at its low concentration, a number of combinations of structural changes in the technological scheme were proposed. They led to a significant increase in the level of hydrogen extraction and its purity, as well as to a decrease in the cost of its separation [100]. The simulation results of 17 different schemes showed that a scheme of two-stage membrane modules arranged in series and combined with a vacuum pump and an electrochemical hydrogen compressor makes it possible to extract hydrogen of high (99.9997%) purity from low-pressure gas networks at a level of 25 kg per day. However, this scheme requires high power consumption due to the need to install an additional mechanical compressor between the two-stage membrane modules and the electrochemical compressor. If the compressor is removed from the process, and the membrane module with a double layer of palladium is used in a single-stage version, being attached to a vacuum pump, then the purity of the hydrogen separation is reduced to 99.92%, while the energy consumption is reduced by 1.53 times. The possibility of obtaining hydrogen of relatively high purity was also shown by a scheme consisting of one membrane module, a vacuum pump and an electrochemical compressor.
If the existing natural gas pipeline infrastructure were to be used for the transportation of hydrogen, one of the issues would be the separation of the hydrogen from the various methane–hydrogen mixtures. Such a separation using proton exchange membrane (PEM) fuel cell technology at temperatures from 293 to 343 K with either platinum or platinum/ruthenium anode electrocatalysts and at atmospheric pressure was described in [101]. The experimental results show that little energy is consumed in producing pure hydrogen and that the methane does not cause significant fouling of the anode electrocatalyst. The data are reported on an electrochemical cell with Gortex-based electrodes capable of efficiently extracting pure hydrogen in a single step from highly dilute hydrogen–methane mixtures containing as little as 5% hydrogen in methane [102]. The hydrogen separation and purification from methane–hydrogen mixtures can also be realized by means of electrochemical membrane technology with Pt-Co/C as the anode electrocatalyst in the temperature range 100–160 °C [103]. High purity hydrogen (>99.9%) was obtained from a low purity feed (20% H2) after separating hydrogen from H2/CH4 mixtures.
Theoretical analysis allowed the authors of [104] to conclude that the use of methane–hydrogen mixtures can lead to a significantly higher, almost twofold, energy storage capacity compared to the separate use of hydrogen or methane.
In addition to pressure swing absorption, a good efficiency of hydrogen extraction from dilute methane–hydrogen mixtures was demonstrated by the use of metal hydrides. Successful experiments were carried out on the separation of lean mixtures of hydrogen–methane with a percentage ratio of 10:90 using the intermetallic compound LaNi4.8Mn0.3Fe0.1 (inlet pressure 9.5 bar) with reversible (adsorption/desorption) separation of 74% hydrogen in one stage [105].
The possibility of transporting hydrogen in liquefied form is also being considered. However, the low temperature of liquid hydrogen, only 20.3 K, requires a large amount of energy to liquefy it and presents the problem of its long-term storage. However, the storage and transportation of hydrogen in a chemically bound form (hydrides, solid and liquid carriers, etc.) does not yet provide the hydrogen capacity necessary for economically justified use [106].
6. Safety Problems of Using of Hydrogen and Methane–Hydrogen Mixtures
The possibility of practical use of methane–hydrogen mixtures is already being successfully implemented in public transport. However, there are still little literature data on the risk analysis and safety of the use of such fuels. The authors of [107] came to the conclusion that the use of Hythane mixtures is safer not only using hydrogen, but also methane. This is due to the fact that methane–hydrogen mixtures combine the positive qualities of hydrogen (high diffusion coefficient) and methane (lower flame propagation velocity and narrower ignition limits). Modeling has shown that the explosive property of the Hythane is not much higher than that of methane, and that for both of these fuels the explosion pressure is significantly lower than that of hydrogen.
Since feasibility studies show that the use of pipelines for pumping large volumes of hydrogen over long distances is currently the most profitable, the requirements for their design and conditions for safe operation were carefully analyzed [82]. Among the key provisions related to the use of methane–hydrogen mixtures analyzed in the NREL report [83] were the safety issues of their use. One of the most important issues is the maximum allowable concentration of hydrogen in methane–hydrogen mixtures, which is explained by the special physicochemical properties of hydrogen, primarily its high flammability. It is known that, in general, the risks associated with dealing with natural gas, accompanied by severe consequences, are less significant compared to other large energy systems. Small, up to 20%, hydrogen additions to natural gas pipelines lead to a slight increase in the risk of ignition. The introduction of more than 50% hydrogen already leads to a significant increase in all risks, including the probability and consequences of their occurrence. Great attention is also paid to the strength of the pipeline material, especially in the case of high hydrogen concentrations in high pressure systems. Greater risks are also associated with the possibility of gas leakage from pipelines. Therefore, when using methane–hydrogen mixtures, additional leak detection devices are required; this increases the cost of equipment and its maintenance. In general, the modification of the safety of natural gas distribution systems when transitioning to methane–hydrogen mixtures leads to an increase in costs by about 10%.
Estimated costs of operating the existing natural gas transportation infrastructure for hydrogen transportation show that such transportation of hydrogen will cost 30–50% more than natural gas [108,109]. At the same time, the use of the existing infrastructure for the transportation of methane–hydrogen mixtures makes it possible to reduce these costs, but it is necessary to take into account possible risks.
The safety of operation of existing gas distribution systems on hydrogen-containing mixtures will strongly depend on the concentration of hydrogen. In addition, compared to methane, these mixtures are lighter, so the volumetric flow rate of the gas flow and gas leakage at the same pressures in the system will be higher. To evaluate the behavior of methane–hydrogen mixtures during their long stay in the pipeline system, experiments were carried out to determine the level of stratification of these mixtures. The experiments were carried out in closed cubic apparatus with a volume of 25 m3 both under natural ventilation conditions and without it [110]. Two fuel mixtures were tested: 10% hydrogen: 90% methane and 30% hydrogen: 70% methane. The results of long-term experiments of these mixtures did not reveal their stratification into their constituent hydrogen and methane, neither under conditions of natural ventilation, nor in its absence.
The effects of enriching natural gas with hydrogen on local flame extinction, combustion instabilities and power output have been widely studied for both stationary and mobile systems, while the issues of explosion safety for hydrogen–methane mixtures are still under investigation. In work [111], experimental tests were performed in a 5 L closed cylindrical vessel for explosions of hydrogen–methane mixtures in stoichiometric air. Different compositions of hydrogen–methane were tested (from pure methane to pure hydrogen) at varying initial pressures (1, 3 and 6 bar). Results have allowed the quantification of the combined effects of both mixture composition (i.e., hydrogen content in the fuel) and initial pressure on maximum pressure, maximum rate of pressure rise and burning velocity. The measured burning velocities were also correlated by means of a Le Chatelier’s Rule-like formula. Good predictions have been obtained (at any initial pressure), except for mixtures with hydrogen molar content in the fuel higher than 50%.
An important problem is also the correct choice of an efficient technology for compressing large volumes of hydrogen and the minimum gas mass flow rate. At low hydrogen concentrations, only minor changes to the operation and maintenance of the existing gas pipeline network are required. Some difficulties may arise at hydrogen concentrations of 5–15% vol. At higher hydrogen concentrations 15–50%, these difficulties will become more noticeable. Mixtures containing more than 50% hydrogen will already cause serious problems with pipeline material, safety and the need for significant modifications to existing equipment. Therefore, at present, the least risks are associated with the use of methane–hydrogen mixtures with a hydrogen content of less than 20% by volume.
Safety, compression and transportation of methane–hydrogen mixtures are studied in the work [112]. The results of a study of the behavior of methane–hydrogen mixtures of the compositions H2:CH4 = 10:90%, 25:75% and 50:50% in pipelines with an internal diameter of 0.15 m and 1.0 m, obtained by mathematical modeling using the Aspen Plus code, are presented. In pipelines with a diameter of 0.15 m, the value of the mass flow rate of the mixture was 1.03 kg/s, 1.49 kg/s and 1.75 kg/s, and with a diameter of 1.0 m—269 kg/s, 383 kg/s and 449.5 kg/s. The various types of compressors were considered: traditional four-section and 16-stage centrifugal. An analysis of energy losses in pipelines made it possible to establish the maximum safe distance for transporting methane–hydrogen mixtures, depending on the internal diameter of the pipeline. It is shown that the area of hazardous zones that arise around pipelines in the case of transportation of methane–hydrogen fuel mixture is narrower than for natural gas. The issues of damage resistance of the pipeline material were also investigated. Taking into account the effect of hydrogen concentration on the combustion of the resulting fuel, it was concluded that for gas appliances for domestic and commercial purposes, the safest and most effective hydrogen concentration is 15% [77].
To assess the risk of overpressure and high flame propagation velocity in MHM, flame propagation in premixed gas mixtures with hydrogen concentrations in methane of 0%, 10% and 20% was experimentally studied at an equivalent ratio of 1 [113]. Dangerous zones of overpressure occurrence in the pipeline were identified. With hydrogen content in the mixture of 20%, the maximum overpressure was 1.266 MPa, and the maximum flame velocity was 168 m/s (the initial pressure of the mixture is 1 bar).
It is important to ensure the accuracy and safety of measuring instruments when using the existing gas infrastructure for transporting methane–hydrogen mixtures, in particular, gas meters that determine the concentration of hydrogen. In addition, when designing measuring equipment, it is necessary to ensure the safe thickness of the instrument case, depending on the type of controlled gas fuel. Special safety requirements must be observed when installing equipment in obviously dangerous areas. The work [114] tested the durability of membrane gas meters for controlling the supply of methane–hydrogen mixtures with different hydrogen concentrations (0%, 5%, 10% and 15% vol.) in natural gas. In the study of mixtures with a hydrogen concentration in the range from 0 to 15% vol., no significant metrological differences were found in the average measurement error of gas meters. However, the accuracy of measurements was strongly influenced by the duration of operation of the gas meter. This, however, did not depend on the concentration of hydrogen in the mixture, but rather on the wear and tear and fragility of the internal parts of the tested devices.
7. Conclusions and Perspectives
An analysis of the literature data indicates a wide front of research and development in the field of the use of methane–hydrogen mixtures as a promising environmentally friendly low-carbon fuel. The influence of the composition of such mixtures on the performance characteristics of various types of engines, the level of emission of CO2, CO, nitrogen oxides and unburnt hydrocarbons, the safety of operation and their durability is studied. The general conclusion of most of these studies is that the use of methane–hydrogen mixtures in internal combustion engines improves the performance of engines operating with a separate supply of natural gas and hydrogen. The most important is the concentration of hydrogen in the fuel mixture, which affects the main characteristics of fuel combustion and determines the optimal operating conditions for engines. The analysis shows that the safety measures and risks when working with methane–hydrogen mixtures with a small content of hydrogen are similar to those that exist when working with natural gas.
Serious logistical problems are associated with the difficulties of using the existing gas distribution infrastructure for transporting methane–hydrogen mixtures. Further research is needed on the compatibility of pipeline materials with hydrogen and methane–hydrogen mixtures and safety conditions for the operation of equipment with hydrogen or methane–hydrogen mixtures. It is possible that, despite the need for huge investments, it will be necessary to create a new infrastructure for the production, storage and transportation of hydrogen and its mixtures with natural gas.
Serious studies are also needed on the economic and environmental feasibility of transition to such energy carriers as hydrogen and methane–hydrogen mixtures in the near future.
Thus, in our opinion, the most promising areas for the development of hydrogen energy in the near future will be the increasingly widespread use of methane–hydrogen mixtures with a hydrogen concentration of up to 20% for energy production and in the domestic sector, as well as the wide appearance of low-tonnage hydrogen production plants for use in urban transport.
Author Contributions
Conceptualization, I.A.M. and V.S.A.; methodology, I.V.S.; formal analysis, I.A.M.; resources, E.A.S. and A.V.A.; data curation, I.A.M.; writing—original draft preparation, I.V.S. and A.V.A.; writing—review and editing, E.A.S. and V.S.A. All authors have read and agreed to the published version of the manuscript.
Funding
The work was carried out within the framework of the Program of Fundamental Scientific Research of the State Academies of Sciences: the theme 0089-2019-0018 (registration number AAAA-A19-119022690098-3) (IPCP RAS) and the theme 0082-2019-0014 (registration number AAAA-A20-120020590084-9) (FRC CP RAS).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Arutyunov, V.S.; Lisichkin, G.V. Energy resources of the 21st century: Problems and forecasts. Can renewable energy sources replace fossil fuels. Russ. Chem. Rev. 2017, 86, 777–804. [Google Scholar] [CrossRef]
- Vasudevan, S.; Farooq, S.; Karimi, I.A.; Saeys, M.; Quah, M.C.G.; Agrawal, R. Energy penalty estimates for CO2 capture: Comparison between fuel types and capture-combustion modes. Energy 2016, 103, 709–714. [Google Scholar] [CrossRef]
- Yu, K.M.K.; Curcic, I.; Gabriel, J.; Tsang, S.C.E. Recent Advances in CO2 Capture and Utilization. ChemSusChem 2008, 1, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Carbon Capture, Use and Storage. European Commission: A Legal Framework for the Safe Geological Storage of Carbon Dioxide. Available online: https://ec.europa.eu/clima/eu-action/carbon-capture-use-and-storage_en (accessed on 20 December 2021).
- The Paris Agreement. 2015. Available online: https://unfccc.int/process-and-meetings/the-paris-agreement/the-paris-agreement (accessed on 29 December 2021).
- Hydrogen Council. Available online: https://hydrogencouncil.com/en/ (accessed on 29 December 2021).
- Hydrogen Scaling Up. A Sustainable Pathway for the Global Energy Transition. Hydrogen Council (November 2017). Available online: https://hydrogencouncil.com/wp-content/uploads/2017/11/Hydrogen-scaling-up-Hydrogen-Council.pdf (accessed on 24 November 2021).
- Makaryan, I.A.; Sedov, I.V. Cost-Effectiveness Assessment of the Scale of Hydrogen Production by Various Methods. Russ. J. Gen. Chem. 2021, 91, 2743–2757. [Google Scholar] [CrossRef]
- Makaryan, I.A.; Sedov, I.V. Catalytic Reactors for Dehydrogenation of Liquid Organic Hydrogen Carriers. Russ. J. Appl. Chem. 2021, 94, 1011–1021. [Google Scholar] [CrossRef]
- Source and Scale Are Biggest Challenges as Hydrogen Interest Grows. S&P Global Platts. Available online: https://www.spglobal.com/en/research-insights/articles/source-and-scale-are-biggest-challenges-as-hydrogen-interest-grows (accessed on 8 December 2021).
- Arutyunov, V.S. On the sources of hydrogen for the global replacement of hydrocarbons. Acad. Lett. 2021, 3692. [Google Scholar] [CrossRef]
- Arutyunov, V.S. Problems and challenges of hydrogen energy. Combust. Plasma Chem. 2021, 19, 245–255. (In Russian) [Google Scholar]
- Arutyunov, V.S. Hydrogen power engineering: Meaning, sources, problems, prospect. Pet. Chem. 2022, in press. [Google Scholar]
- Verhelst, S.; Demuynck, J.; Sierens, R.; Huyskens, P. Impact of variable valve timing on power, emissions and backfire of a bi-fuel hydrogen/gasoline engine. Int. J. Hydrogen Energy 2010, 35, 4399–4408. [Google Scholar] [CrossRef]
- Baird, A.R.; Glover, A.M.; Ehrhart, B.D. Review of Release Behavior of Hydrogen & Natural Gas Blends from Pipelines. Sandia Report 2021. SAND2021-9802. Available online: https://www.energy.sandia.gov/wp-content (accessed on 24 November 2021).
- Singh, A.P.; Pal, A.; Agarwal, A.K. Comparative particulate characteristics of hydrogen, CNG, HCNG, gasoline and diesel fueled engines. Fuel 2016, 185, 491–499. [Google Scholar] [CrossRef]
- Hermanns, R.T.E. Laminar Burning Velocities of Methane-Hydrogen-Air Mixtures. Ph.D. Thesis, Technische Universiteit Eindhoven, Eindhoven, The Netherlands, 1 January 2007. Available online: https://research.tue.nl/en/publications/laminar-burning-velocities-of-methane-hydrogen-air-mixtures (accessed on 21 January 2021). [CrossRef]
- Hu, E.; Huang, Z.; He, J.; Jin, C.; Zheng, J. Experimental and numerical study on laminar burning characteristics of premixed methane–hydrogen–air flames. Int. J. Hydrogen Energy 2009, 34, 4876–4888. [Google Scholar] [CrossRef]
- Gelfand, B.E.; Silnikov, M.V.; Sergey, P.; Medvedev, S.P.; Khomik, S.V. Thermo-Gas Dynamics of Hydrogen Combustion and Explosion; Springer: Berlin/Heidelberg, Germany, 2012; 326p, eBook; ISBN 978-3-642-25352-2. [Google Scholar]
- Burluka, A.A.; Fairweather, M.; Ormsby, M.P.; Sheppard, C.G.W.; Woolley, R. The laminar burning properties of premixed methane-hydrogen flames determined using a novel analysis method. In Proceedings of the Third European Combustion Meeting (ECM 2007), Chania Crete, Greece, 11–13 April 2007; pp. 1–5. [Google Scholar]
- Smith, P.G.; Golden, D.M.; Frenklach, M.; Moriarty, N.W.; Goldenberg, M.; Bowman, N.W.; Hanson, R.K.; Song, S.; Gardiner, W.C., Jr.; Lissianski, V.V.; et al. WHAT’S NEW IN GRI-Mech 3.0. Available online: http://combustion.berkeley.edu/gri-mech/version30/text30.html (accessed on 19 March 2022).
- Konnov, A. Detailed Reaction Mechanism for Small Hydrocarbons Combustion. Release 0.5. 2000. Available online: http://homepages.vub.ac.be/~akonnov/ (accessed on 21 December 2021).
- Di Sarli, V.; Di Benedettob, A. Laminar burning velocity of hydrogen–methane/air premixed flames. Int. J. Hydrogen Energy 2007, 32, 637–646. [Google Scholar] [CrossRef]
- Tatouh, T.; Halter, F.; Mounaim-Rousselle, C. The Effect of Hydrogen Enrichment on CH4-Air Combustion in Strong Dilution. In Proceedings of the 31st Meeting on Combustion, Italian Section of the Combustion Institute, Firenze, Italy, 17–20 June 2008; XII-13. XII-13.rtf-10.1.1.567.3214. Available online: https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.567.3214&rep=rep1&type=pdf (accessed on 15 January 2022).
- Arutyunov, A.V.; Belyaev, A.A.; Inovenkov, I.N.; Arutyunov, V.S. The influence of hydrogen on the burning velocity of methane-air mixtures at elevated temperatures. Combust. Explos. 2019, 12, 4–10. [Google Scholar] [CrossRef]
- Troshin, K.Y.; Borisov, A.A.; Rakhmetov, A.N.; Arutyunov, V.S.; Politenkova, G.G. Burning velocity of methane–hydrogen mixtures at elevated pressures and temperatures. Russ. J. Phys. Chem. B 2013, 7, 290–301. [Google Scholar] [CrossRef]
- Eckart, S.; Pizzuti, L.; Fritsche, C.; Krause, H. Experimental study and proposed power correlation for laminar burning velocity of hydrogendiluted methane with respect to pressure and temperature variation. Int. J. Hydrogen Energy 2022, 47, 6334–6348. [Google Scholar] [CrossRef]
- Troshin, K.Y.; Nikitin, A.V.; Belyaev, A.A.; Arutyunov, A.V.; Kiryushin, A.A.; Arutyunov, V.S. Experimental determination of self-ignition delay of mixtures of methane with light alkanes. Combust. Explos. Shock. Waves 2019, 55, 526–533. [Google Scholar] [CrossRef]
- Troshin, K.Y.; Belyaev, A.A.; Arutyunov, A.V.; Tsarenko, A.A.; Nikitin, A.V.; Arutyunov, V.S. The influence of pressure on the self-ignition delay ofmethane–hydrogen–air mixtures. Combust. Explos. 2020, 13, 18–32. [Google Scholar] [CrossRef]
- Arutyunov, V.S.; Belyaev, A.A.; Troshin, K.Y.; Arutyunov, A.V.; Tsarenko, A.A.; Nikitin, A.V. On the use of methane-hydrogen mixtures in internal combustion engines. Oil Gas Chem. 2018, 3–4, 5–17. [Google Scholar] [CrossRef]
- Westbrook, C.K.; Sjöberg, M.; Cernansky, N.P. A new chemical kinetic method of determining RON and MON values for single component and multicomponent mixtures of engine fuels. Combust. Flame 2018, 195, 50–62. [Google Scholar] [CrossRef]
- Yadav, K.; Anirbid, S. Hydrogen compressed natural gas and liquefied compressed natural gas: Fuels for future. PDPU J. Energy Manag. 2017, 2, 29–33. Available online: https://www.pdpu.ac.in/downloads/3%20Hydrogen-Compressed-Natural-Gas.pdf (accessed on 14 January 2022).
- Di Sarli, V.; Di Benedetto, A.; Long, E.J.; Hargrave, G.K. Time-Resolved Particle Image Velocimetry of dynamic interactions between hydrogen-enriched methane/air premixed flames and toroidal vortex structures. Int. J. Hydrogen Energy 2012, 37, 16201–16213. [Google Scholar] [CrossRef]
- Garcia-Agreda, A.; Di Sarli, V.; Di Bene-detto, A. Bifurcation analysis of the effect of hydrogen addition on the dynamic behavior of lean premixed pre-vaporized ethanol combustion. Int. J. Hydrogen Energy 2012, 37, 6922–6932. [Google Scholar] [CrossRef]
- Moreno, F.; Muñoz, M.; Magén, O.; Monné, C.; Arroyo, J. Modifications of a spark ignition engine to operate with hydrogen and methane blends. Renew. Energy Power Qual. J. 2010, 1, 421–426. [Google Scholar] [CrossRef]
- Nagalingam, B.; Duebel, F.; Schmillen, K. Performance study using natural gas, hydrogensupplemented natural gas and hydrogen in AVL research engine. Int. J. Hydrogen Energy 1983, 8, 715–720. [Google Scholar] [CrossRef]
- De Vries, H.; Mokhov, A.V.; Levinsky, H.B. The impact of natural gas/hydrogen mixtures on the performance of end-use equipment: Interchangeability analysis for domestic appliances. Appl. Energy 2017, 208, 1007–1019. [Google Scholar] [CrossRef]
- Zhao, Y.; McDonell, V.; Samuelsen, S. Influence of hydrogen addition to pipeline natural gas on the combustion performance of a cooktop burner. Int. J. Hydrogen Energy 2019, 44, 12239–12253. [Google Scholar] [CrossRef]
- Arrieta, C.E.; Amell, A.A. Combustion analysis of an equimolar mixture of methane and syngas in a surface-stabilized combustion burner for household appliances. Fuel 2014, 137, 11–20. [Google Scholar] [CrossRef]
- Imran, S.; Emberson, D.R.; Ihracska, B.; Wen, D.S.; Crookes, R.J.; Korakianitis, T. Effect of pilot fuel quantity and type on performance and emissions of natural gas and hydrogen based combustion in a compression ignition engine. Int. J. Hydrogen Energy 2014, 39, 5163–5175. [Google Scholar] [CrossRef]
- Liu, S.; Li, H.; Liew, C.; Liu, S.; Gatts, T.; Wayne, S.; Shade, B.; Clark, N. An experimental investigation of H2 emissions characteristics of heavy-duty H2-diesel dual fuel engine. Int. J. Hydrogen Energy 2011, 36, 12015–12024. [Google Scholar] [CrossRef]
- Christodoulou, F.; Megaritis, A. Experimental investigation of the effects of separate hydrogen and nitrogen addition on the emissions and combustion of a diesel engine. Int. J. Hydrogen Energy 2013, 38, 10126–10140. [Google Scholar] [CrossRef]
- Niculae, A.L.; Miron, L.; Chiriac, R. On the possibility to simulate the operation of a SI engine using alternative gaseous fuels. Energy Rep. 2020, 6, 167–176. [Google Scholar] [CrossRef]
- Bade Shrestha, S.O.; Karim, G.A. Hydrogen as an additive to methane for spark ignition engine applications. Int. J. Hydrogen Energy 1999, 24, 577–586. [Google Scholar] [CrossRef]
- Dieguez, P.M.; Urroz, J.C.; Marcelino-Sadaba, S.; Perez-Ezcurdia, A.; Benito-Amurrio, M.; Sainz, D. Experimental study of the performance and emission characteristics of an adapted commercial four-cylinder spark ignition engine running on hydrogen-methane mixtures. Appl. Energy 2014, 113, 1068–1076. [Google Scholar] [CrossRef]
- Van Blarigan, P.; Keller, J.O. A hydrogen fuelled internal combustion engine designed for single speed/power operation. Int. J. Hydrogen Energy 1998, 23, 603–609. [Google Scholar] [CrossRef]
- Karim, G.A.; Wierzba, I.; Al-Alousi, Y. Methane-hydrogen mixtures as fuels. Int. J. Hydrogen Energy 1996, 21, 625–631. [Google Scholar] [CrossRef]
- Klell, M.; Eichlseder, H.; Sartory, M. Mixtures of hydrogen and methane in the internal combustion engine—Synergies, potential and regulations. Int. J. Hydrogen Energy 2012, 37, 11531–11540. [Google Scholar] [CrossRef]
- Das, L.M.; Polly, M. Experimental Evaluation of a Hydrogen Added Natural Gas (HANG) Operated SI Engine; SAE Technical Paper; The Automotive Research Association of India: Pune, India, 2005; 2005-26-029. [Google Scholar] [CrossRef]
- Wallner, T.; Ng, H.; Peters, R. The Effects of Blending Hydrogen with Methane on Engine Operation, Efficiency, and Emissions; SAE Technical Paper; SAE International: Warrendale, PA, USA, 2007; 2007-01-0474. [Google Scholar] [CrossRef]
- Anstrom, J.R.; Collier, K. Blended hydrogen–natural gas-fueled internal combustion engines and fueling infrastructure. Compend. Hydrog. Energy 2016, 3, 219–232. [Google Scholar] [CrossRef]
- Karczewski, M.; Chojnowski, J.; Szamrej, G. A Review of Low-CO2 Emission Fuels for a Dual-Fuel RCCI Engine. Energies 2021, 14, 5067. [Google Scholar] [CrossRef]
- Talibi, M.; Ramanarayanan, B.; Ladommatos, N. Influence of combusting methane-hydrogen mixtures on compression–ignition engine exhaust emissions and in-cylinder gas composition. Int. J. Hydrogen Energy 2017, 42, 2381–2396. [Google Scholar] [CrossRef]
- Tangoz, S.; Kahraman, N.; Akansu, S.O. The effect of hydrogen on the performance and emissions of an SI engine having a high compression ratio fuelled by compressed natural gas. Int. J. Hydrogen Energy 2017, 42, 25766–25780. [Google Scholar] [CrossRef]
- Agarwal, J.; Alam, M.; Jaiswal, A.; Yadav, K.; Kumar, N. Comparative Study of Emissions and Performance of Hythane Boosted SI Engine Powered by Gasoline-Methanol Blend and Gasoline-Ethanol Blend//SAE 2016 World Congress and Exhibition; Technical Paper; SAE International: Warrendale, PA, USA, 2016; 8p, 2016-01-1281. [Google Scholar] [CrossRef]
- Yadav, B.K.; Soni, S.L.; Sharma, D.; Sharma, P.K. Performance and emission analysis of constant speed SI engine operated on hythane (HCNG). Int. J. Sci. Technol. 2017, 3, 64–79. [Google Scholar] [CrossRef][Green Version]
- Kaiadi, M.; Tunestal, P.; Johansson, B. How hythane with 25% hydrogen can affect the combustion in a 6-cylinder natural-gas engine. SAE Int. J. Fuels Lubr. 2010, 3, 47–59. [Google Scholar] [CrossRef]
- Agarwal, A.K.; Prasad, R.K. Laser ignition of hydrogen enriched compressed natural gas (HCNG) fueled supercharged engine. In Proceedings of the Laser Ignition Conference 2017, Bucharest, Romania, 20–23 June 2017. OSA Technical Digest (online) (Optical Society of America, 2017), paper LThA3. [Google Scholar] [CrossRef]
- Sofianopoulos, A.; Assanis, D.N.; Mamalis, S. Effects of Hydrogen Addition on Automotive Lean-Burn Natural Gas Engines: Critical Review. J. Energy Eng. 2016, 142, E4015010. [Google Scholar] [CrossRef]
- Hora, T.S.; Agarwal, A.K. Compressed natural gas and hythane for on-road passenger and commercial vehicles. In Prospects of Alternative Transportation Fuels; Springer: Singapore, 2018; pp. 79–106. [Google Scholar] [CrossRef]
- Gómez-Montoya, J.P.; Cacua-Madero, K.P.; Iral-Galeano, L.; Amell-Arrieta, A.A. Effect of biogas enriched with hydrogen on the operation and performance of a diesel-biogas dual engine. CT&F-Cienc. Tecnologia y Futuro 2013, 5, 61–72. [Google Scholar]
- Hinton, N.; Stone, R. Laminar burning velocity measurements of methane and carbon dioxide mixtures (biogas) over wide ranging temperatures and pressures. Fuel 2014, 116, 743–750. [Google Scholar] [CrossRef]
- Yadav, V.K.; Ray, A.; Ravi, M.R. Experimental and computational investigation of the laminar burning velocity of hydrogen-enriched biogas. Fuel 2019, 235, 810–821. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, X. Experimental study on flame stability of biogas/hydrogen combustion. Int. J. Hydrogen Energy 2019, 44, 5607–5614. [Google Scholar] [CrossRef]
- Mariani, A.; Unich, A.; Minale, M. Combustion of Hydrogen Enriched Methane and Biogases Containing Hydrogen in a Controlled Auto-Ignition Engine. Appl. Sci. 2018, 8, 2667. [Google Scholar] [CrossRef]
- Amaduzzi, R.; Ferrarotti, M.; Parente, A. Strategies for Hydrogen-Enriched Methane Flameless Combustion in a Quasi-Industrial Furnace. Front. Energy Res. 2021, 8, 590300. [Google Scholar] [CrossRef]
- Lister, J.; Poulsen, R.T.; Ponte, S. Orchestrating transnational environmental governance in maritime shipping. Glob. Environ. Change 2015, 34, 185–195. [Google Scholar] [CrossRef]
- Balcombe, P.; Brierley, J.; Lewis, C.; Skatvedt, L.; Speiers, J.; Hawkes, A.; Staffell, I. How to decarbonise international shipping: Options for fuels, technologies and policies. Energy Convers. Manag. 2019, 182, 72–88. [Google Scholar] [CrossRef]
- Zhao, R.; Xu, L.; Su, X.; Feng, S.; Li, C.; Tan, Q.; Wang, Z. A numerical and experimental study of marine hydrogen–natural gas–diesel tri–fuel engines. Pol. Marit. Res. 2020, 4, 80–90. [Google Scholar] [CrossRef]
- Arat, H.; Aydin, K.; Baltacioğlu, E.; Yasar, E.; Baltacioglu, M.; Conker, C.; Burgaç, A. A review of hydrogen-enriched compressed natural gas (HCNG)-fuel in diesel engines. J. Macro Trends Energy Sustain. (JMES) 2013, 1, 115–122. [Google Scholar]
- Premkartikkumar, S.R.; Gopidesi, R.K. Review on Effects of Performance, Emission and Combustion Characteristics of Emulsified Fuel in Bifuel Engine. Prog. Ind. Ecol. Int. J. 2018, 12, 59–66. [Google Scholar] [CrossRef][Green Version]
- Premkartikkumar, S.R.; Annamalai, K.; Pradeepkumar, A.R. Impact of ambient air temperature and injection timing on reduction of engine out emissions of DI diesel engine operating under the influence of oxygen enriched hydrogen gas. Int. J. Oil Gas Coal Technol. 2015, 9, 109–127. [Google Scholar] [CrossRef]
- Yon, S.; Sautet, J.C. Flame lift-off height, velocity flow and mixing of hythane in oxy-combustion in a burner with two separated jets. Appl. Therm. Eng. 2012, 32, 83–92. [Google Scholar] [CrossRef]
- Kumma, N.; Gopidesi, R.K.; Rao, T.R.; Kumar, K.M.; Premkartikkumar, S.R. Experimental Investigation on Diesel Engine. Fuelled with Hythane Gas. Int. J. Mech. Eng. Technol. 2019, 10, 571–575. [Google Scholar]
- Sandalc, T.; Işın, Ö.; Galata, S.; Karagöz, Y.; Güler, I. Effect of hythane enrichment on performance, emission and combustion characteristics of an ci engine. Int. J. Hydrogen Energy 2019, 44, 3208–3220. [Google Scholar] [CrossRef]
- Chugh, S.; Posina, V.A.; Sonkar, K.; Srivatsava, U.; Sharma, A.; Acharya, G.K. Modeling & simulation study to assess the effect of CO2 on performance and emissions characteristics of 18% HCNG blend on a light duty SI engine. Int. J. Hydrogen Energy 2016, 41, 6155–6161. [Google Scholar] [CrossRef]
- Wojtowicz, R. An analysis of the effects of hydrogen addition to natural gas on the work of gas appliances. Naft. -Gaz 2019, 8, 465–473. [Google Scholar] [CrossRef]
- Xiang, L.; Jiang, H.; Ren, F.; Chu, H.; Wang, P. Numerical study of the physical and chemical effects of hydrogen addition on laminar premixed combustion characteristics of methane and ethane. Int. J. Hydrogen Energy 2020, 45, 20501–20514. [Google Scholar] [CrossRef]
- Costa, M.; Piazzullo, D.; Dolce, A. Hydrogen addition to natural gas in cogeneration engines: Optimization of performances through numerical modeling. Front. Mech. Eng. 2021, 67, 680193. [Google Scholar] [CrossRef]
- Stylianidis, N.; Azimov, U.; Birkett, M. Investigation of the Effect of Hydrogen and Methane on Combustion of Multicompoment Syngas Mixtures using a Constructed Reduced Chemical Kinetics Mechanism. Energies 2019, 12, 2442. [Google Scholar] [CrossRef]
- Wang, J.; Huang, Z.; Tang, C.; Miao, H.; Wang, X. Numerical study of the effect of hydrogen addition on methane-air mixtures combustion. Int. J. Hydrogen Energy 2009, 34, 1084–1096. [Google Scholar] [CrossRef]
- Litvinenko, V.S.; Tsvetkov, P.S.; Dvoynikov, M.V.; Buslaev, G.V. Barriers to implementation of hydrogen initiatives in the context of global energy sustainable development. J. Min. Inst. 2020, 244, 428–438. [Google Scholar] [CrossRef]
- Hydrogen Pipeline Systems. Doc 121/14. European Industrial Gases Association AISBL. Available online: https://www.eiga.eu/ct_documents/doc121-pdf/ (accessed on 19 March 2022).
- Melaina, M.W.; Sozinova, O.; Penev, M. Blending hydrogen into natural gas pipeline networks: A review of key issues. Durability 2013, 1, 17–34. [Google Scholar] [CrossRef]
- Fan, Y.; Lee, A.; Parker, N.; Scheitrum, D.; Jaffe, A.M.; Dominguez-Faus, R.; Medlock, K. Geospatial, temporal and economic analysis of alternative fuel infrastructure: The case of freight and US natural gas markets. Energy J. 2018, 38, 199–230. [Google Scholar] [CrossRef]
- Schiebahn, S.; Grube, T.; Robinius, M.; Tietze, V.; Kumar, B.; Stolten, D. Power to gas: Technological overview, systems analysis and economic assessment for a case study in Germany. Int. J. Hydrogen Energy 2015, 40, 4285–4294. [Google Scholar] [CrossRef]
- Çeper, B.A. Use of hydrogen-methane blends in internal combustion engines. In Hydrogen Energy—Challenges and Perspectives; Dragica Minic; IntechOpen: London, UK, 2012; Available online: https://www.intechopen.com/chapters/40243 (accessed on 19 March 2022). [CrossRef]
- Hankinson, G.; Mathurkar, H.; Lowesmith, B.J. Ignition Energy and Ignition Probability of Methane-Hydrogen-Air Mixtures. Conference Paper 2009. Available online: http://conference.ing.unipi.it/ichs2009/images/stories/papers/125.pdf (accessed on 19 March 2022).
- Altfeld, K.; Pinchbeck, D. Admissible Hydrogen Concentrations in Natural Gas Systems; DIV Deutscher Industrieverlag GmbH: Munich, Germany, 2013; Reprint: Gas for Energy 03/2013; ISSN 2192-158X. Available online: https://gerg.eu/g21/wp-content/uploads/2019/10/HIPS_Final-Report.pdf (accessed on 19 March 2022).
- Makaryan, I.A.; Sedov, I.V. The State and Development Prospects of the Global Hydrogen Energy Sector. Russ. J. Gen. Chem. 2021, 91, 1912–1928. [Google Scholar] [CrossRef]
- Injecting Hydrogen in Natural Gas Grids Could Provide Steady Demand the Sector Needs to Develop. Electric Power. Natural Gas (19 May 2020). Insight Blog. Available online: https://www.spglobal.com/platts/en/market-insights/blogs/natural-gas/051920-injecting-hydrogen-in-natural-gas-grids-could-provide-steady-demand-the-sector-needs-to-develop (accessed on 21 December 2021).
- Birkitt, K.; Loo-Morrey, M.; Sanchez, C.; O’Sullivan, L. Materials aspects associated with the addition of up to 20 mol% hydrogen into an existing natural gas distribution network. Int. J. of Hydrogen Energy 2021, 46, 12290–12299. [Google Scholar] [CrossRef]
- Jaffe, A.M.; Dominguez-Faus, R.; Ogden, J.M.; Parker, N.C.; Scheitrum, D.; McDonald, Z.; Fan, Y.; Durbin, T.; Karavalakis, G.; Wilcock, J.; et al. The Potential to Build Current Natural Gas Infrastructure to Accommodate the Future Conversion to Near-Zero Transportation Technology; Institute of Transportation Studies, University of California: Davis, CA, USA, 2017; (Research Report UCD-ITS-RR-17-04); Available online: https://its.ucdavis.edu/research/publications/ (accessed on 19 January 2022).
- Could Hydrogen Piggyback on Natural Gas Infrastructure? 17 March 2016. Networks. Available online: https://networks.online/gas/could-hydrogen-piggyback-on-natural-gas-infrastructure/ (accessed on 19 January 2022).
- Ogden, J.; Jaffe, F.M.; Scheitrum, D.; McDonald, Z.; Miller, M. Natural gas as a bridge to hydrogen transportation fuel: Insights from the literature. Energy Policy 2018, 115, 317–329. [Google Scholar] [CrossRef]
- Wahl, J.; Kallo, J. Quantitative valuation of hydrogen blending in European gas grids and its impact on the combustion process of large-bore gas engines. Int. J. Hydrogen Energy 2020, 45, 32534–32546. [Google Scholar] [CrossRef]
- Kong, M.; Feng, S.; Xia, Q.; Chen, C.; Pan, Z.; Gao, Z. Investigation of mixing behavior of hydrogen blended to natural gas in gas network. Sustainability 2021, 13, 4255. [Google Scholar] [CrossRef]
- Kuczynski, S.; Łaciak, M.; Olijnyk, A.; Szurlej, A.; Włodek, T. Thermodynamic and Technical Issues of Hydrogen and Methane-Hydrogen Mixtures Pipeline Transmission. Energies 2019, 12, 569. [Google Scholar] [CrossRef]
- Abd, A.A.; Naji, S.Z.; Thian, T.C.; Othman, M.R. Evaluation of hydrogen concentration effect on the natural gas properties and flow performance. Int. J. of Hydrogen Energy 2021, 46, 1974–1983. [Google Scholar] [CrossRef]
- Nordio, M.; Wassie, S.A.; Van Sint Annaland, M.; Pacheco Tanaka, D.A.; Viviente Sole, J.L.; Gallucci, F. Techno-economic evaluation on a hybrid technology for low hydrogen concentration separation and purification from natural gas grid. Int. J. Hydrogen Energy 2021, 46, 23417–23435. [Google Scholar] [CrossRef]
- Ibeh, B.; Gardner, C.; Ternan, M. Separation of hydrogen from a hydrogen/methane mixture using a PEM fuel cell. Int. J. Hydrogen Energy 2007, 32, 908–914. [Google Scholar] [CrossRef]
- Wagner, K.; Tiwari, P.; Swiegers, G.F.; Wallace, G.G. An electrochemical cell with Gortex-based electrodes capable of extracting pure hydrogen from highly dilute hydrogen–methane mixtures. Energy Environ. Sci. 2018, 11, 172–184. [Google Scholar] [CrossRef]
- Vermaak, L.; Neomagus, H.W.J.P.; Bessarabov, D.G. Hydrogen Separation and Purification from Various Gas Mixtures by Means of Electrochemical Membrane Technology in the Temperature Range 100–160 °C. Membranes 2021, 11, 282. [Google Scholar] [CrossRef]
- Qianqian, X.; Wu, M.; Xiao, C.Z.; Puru, J. Co-mixing hydrogen and methane may double the energy storage capacity. J. Mater. Chem. A 2018, 6, 8916–8922. [Google Scholar] [CrossRef]
- Dunikov, D.O.; Blinov, D.V. Use of methane-hydrogen mixtures for energy accumulation. J. Phys. Conf. Ser. 2019, 1370, 012033. [Google Scholar] [CrossRef]
- Makaryan, I.A.; Sedov, I.V.; Maksimov, A.L. Hydrogen Storage Using Liquid Organic Carriers. Russ. J. Appl. Chem. 2020, 93, 1815–1830. [Google Scholar] [CrossRef]
- Middha, P.; Engel, D.; Hansen, O.R. Can the addition of hydrogen to natural gas reduce the explosion risk? Int. J. Hydrogen Energy 2011, 36, 2628–2636. [Google Scholar] [CrossRef]
- Parfomak, P.W. Pipeline Transportation of Hydrogen: Regulation, Research, and Policy. Pipeline Transportation of Hydrogen: Regulation, Research, and Policy. Congressional Research Service (CRS). CRS Report R46700. 2 March 2021. Available online: https://crsreports.congress.gov/product/pdf/R/R46700 (accessed on 19 March 2022).
- Witkowski, A.; Rusin, A.; Majkut, M.; Stolecka, K. Comprehensive analysis of hydrogen compression and pipeline transportation with thermodynamics and safety aspects. Energy 2017, 141, 2508–2518. [Google Scholar] [CrossRef]
- Marangon, A.; Carcassi, M.N. Hydrogen–methane mixtures: Dispersion and stratification studies. Int. J. Hydrogen Energy 2014, 39, 6160–6168. [Google Scholar] [CrossRef]
- Salzano, E.; Cammarota, F.; Di Benedetto, A.; Di Sarli, V. Explosion behavior of hydrogen–methane/air mixtures. J. Loss Prev. Process Ind. 2012, 25, 443–447. [Google Scholar] [CrossRef]
- Witkowski, A.; Rusin, A.; Majkut, M.; Stolecka, K. Analysis of compression and transport of the methane/hydrogen mixture in existing natural gas pipelines. Int. J. Press. Vessel. Pip. 2018, 166, 24–34. [Google Scholar] [CrossRef]
- Jia, J.; Chen, Y.; Che, G.; Zhu, J.; Wang, F.; Jia, P. Experimental study on the explosion characteristics of hydrogen-methane premixed gas in complex pipe networks. Sci. Rep. 2021, 11, 21204. [Google Scholar] [CrossRef]
- Jaworski, J.; Kułaga, P.; Blacharski, T. Study of the Effect of Addition of Hydrogen to Natural Gas on Diaphragm Gas Meters. Energies 2020, 13, 3006. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).