Stannous Tungstate Semiconductor for Photocatalytic Degradation and Photoelectrochemical Water Splitting: A Review
Abstract
1. Introduction
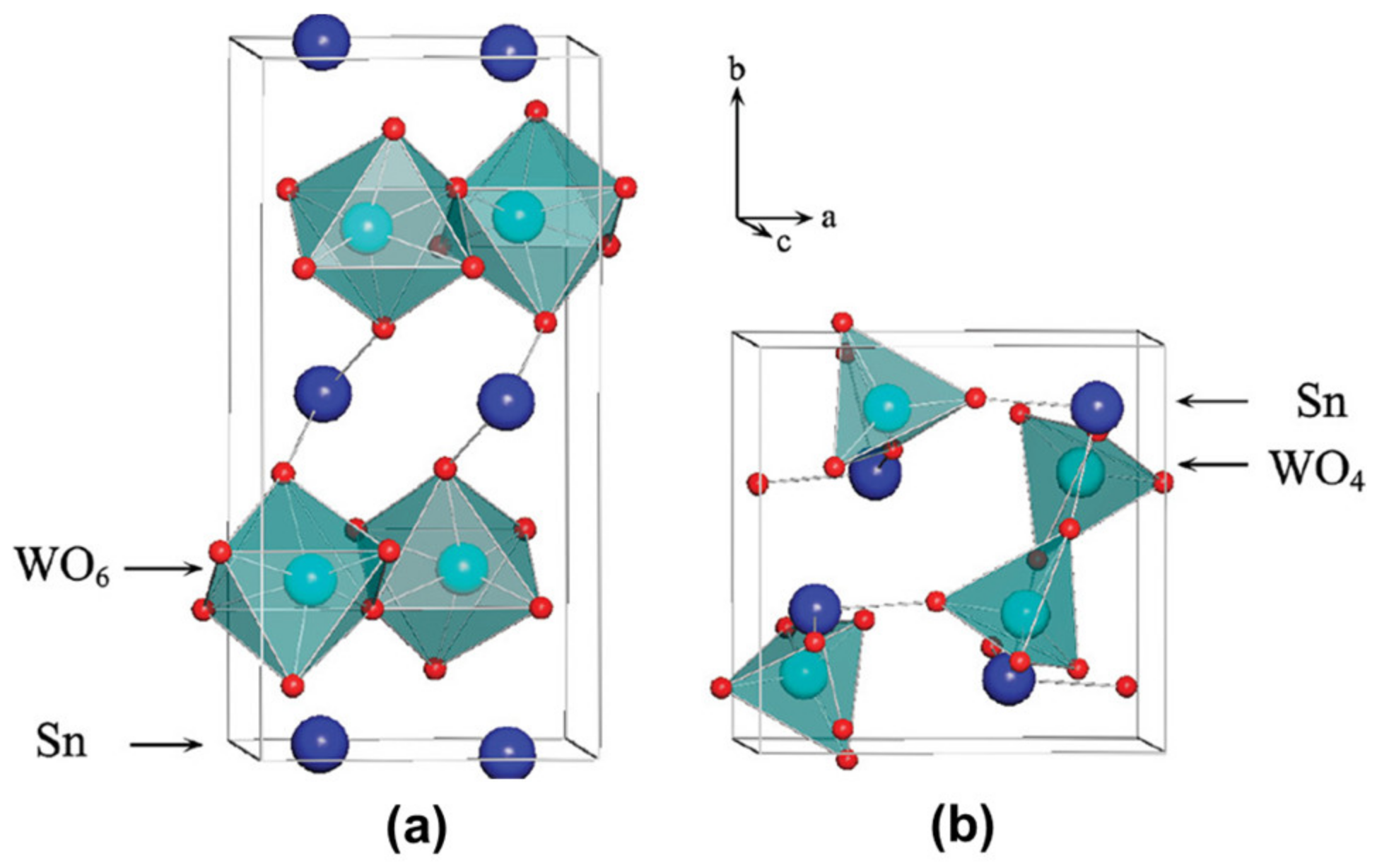
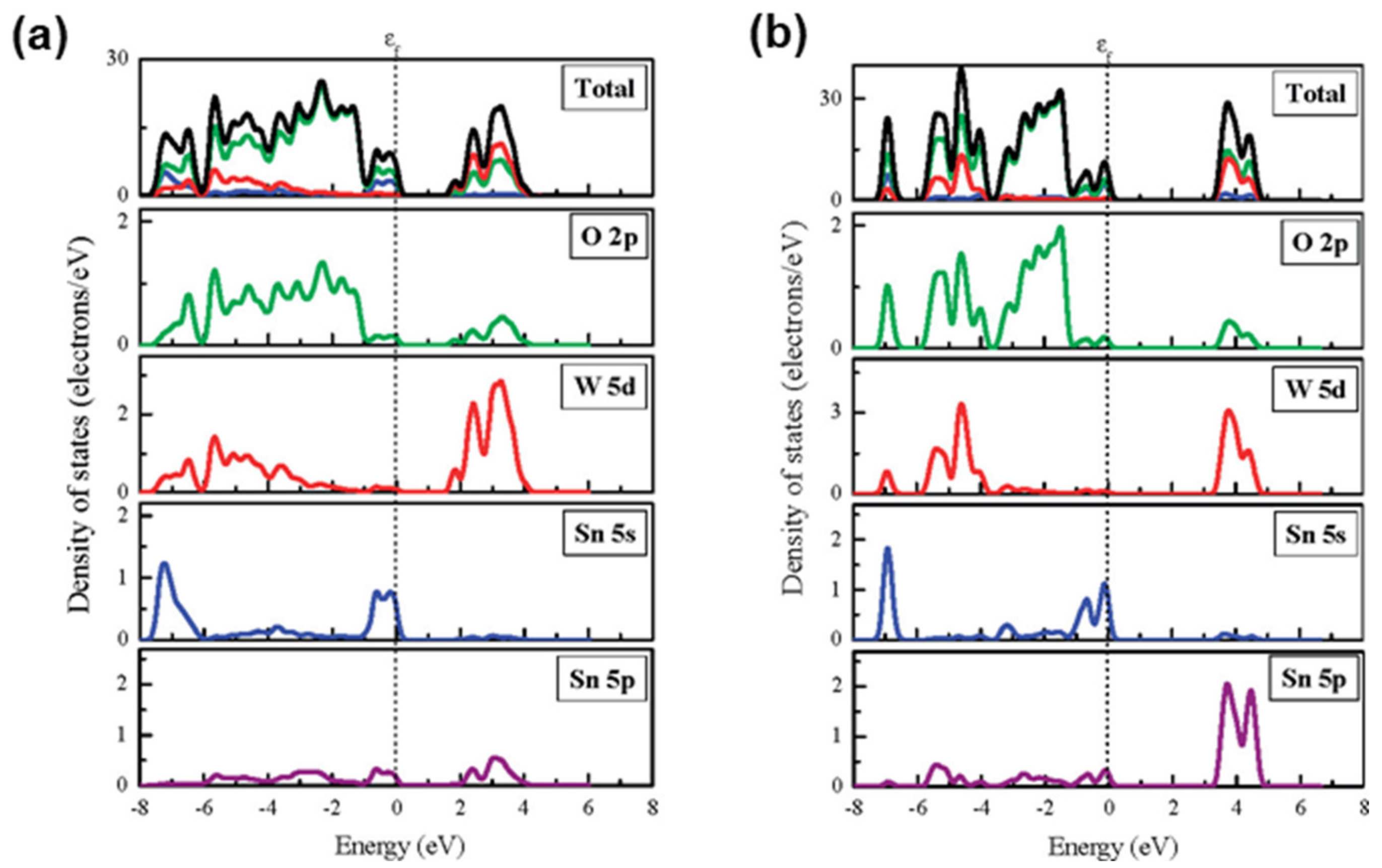
2. Structure, Properties, and Synthesis of SnWO4
3. Research on Modification of SnWO4
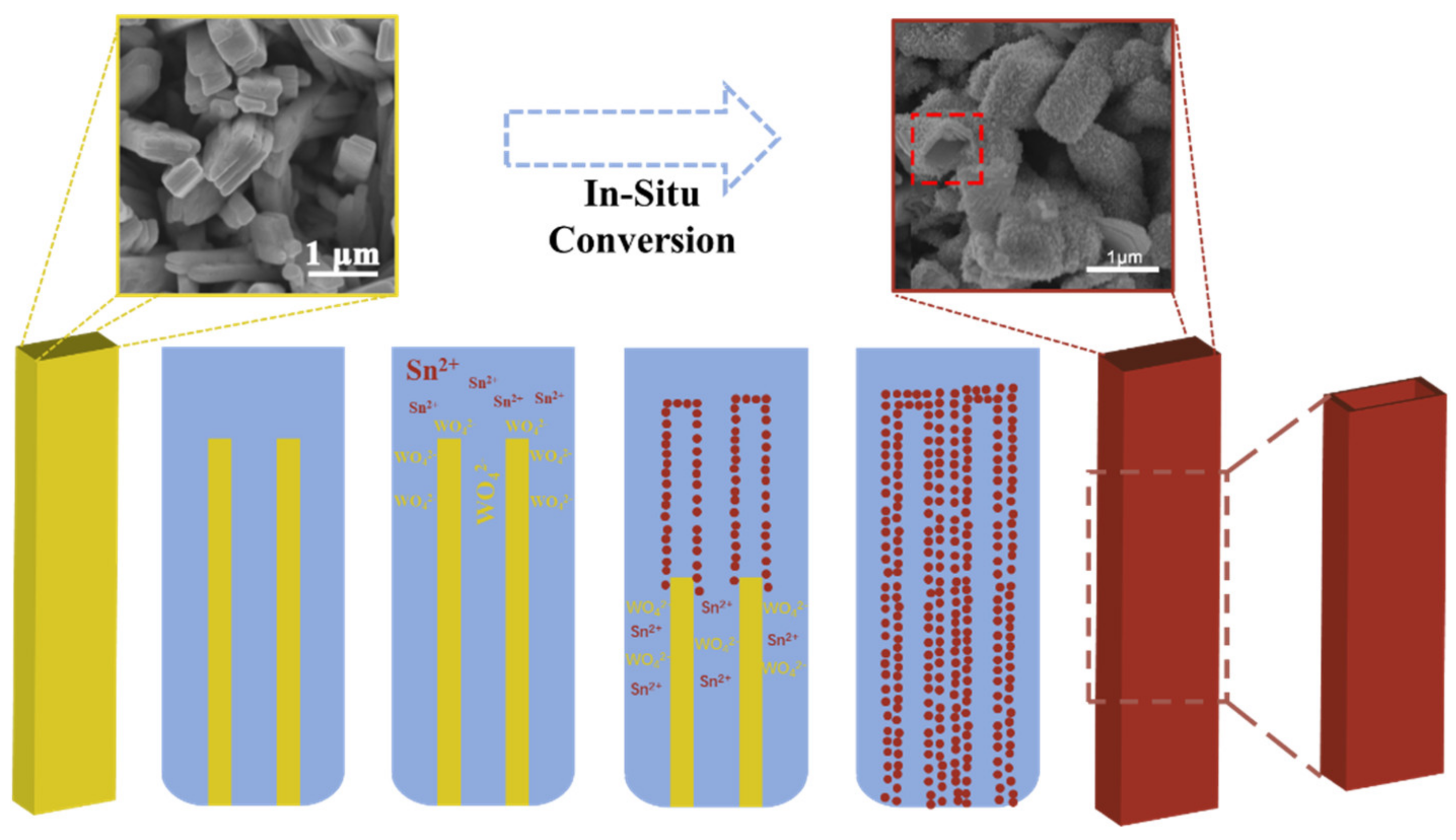
3.1. Morphological Modification
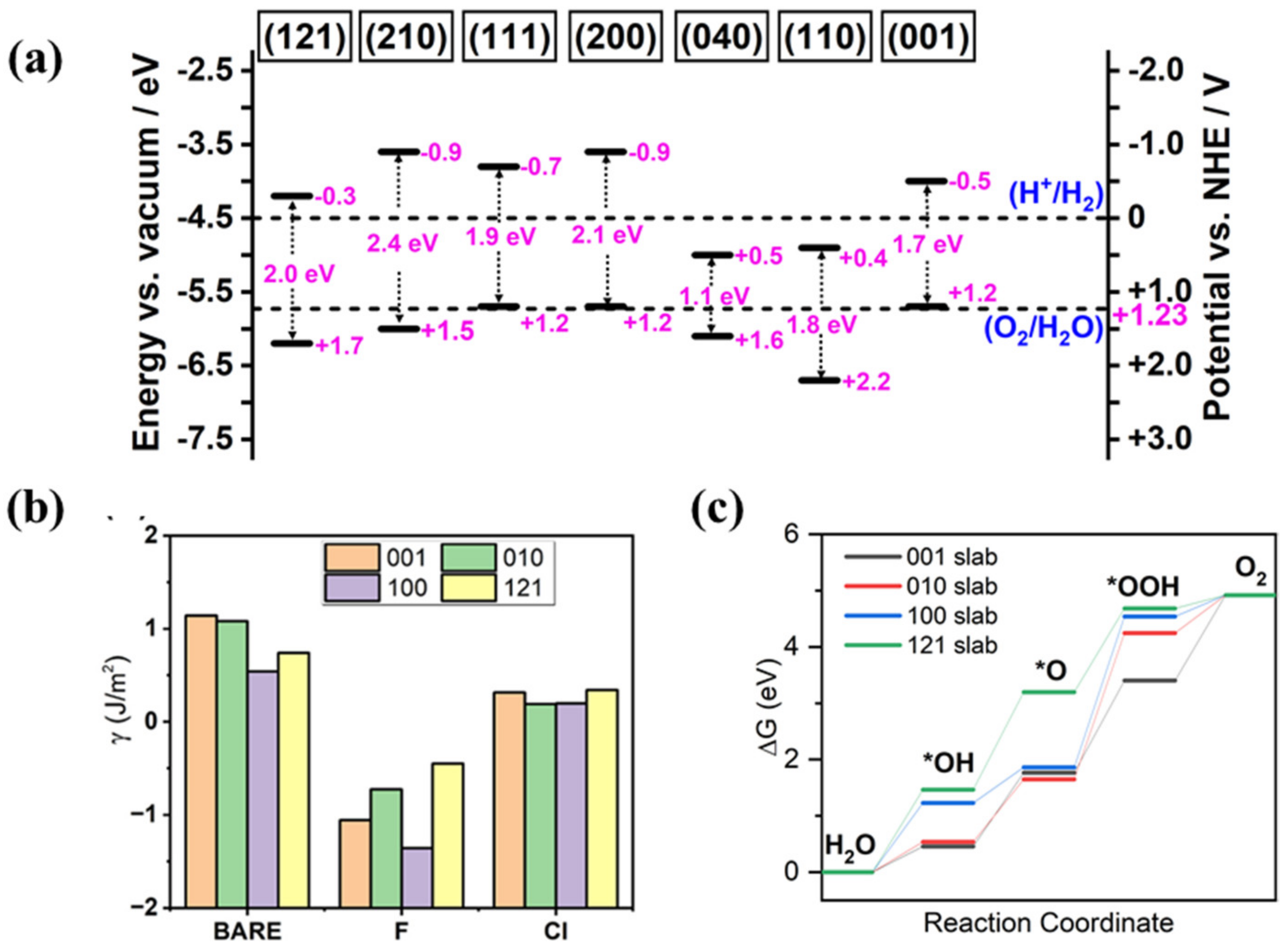
3.2. Crystal Facet Engineering
3.3. Doping Modification
3.4. Multicomponent Composite
3.5. Other Techniques
4. Conclusions and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Zhao, Y.; Wang, S.; Li, C.; Li, R. Recent advances and perspectives for solar-driven water splitting using particulate photocatalysts. Chem. Soc. Rev. 2022, 51, 3561–3608. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.-S.; Kwak, C.H.; Kim, D.W.; Lee, S.; Hong, K.S. Photophysical, Photoelectrochemical, and Photocatalytic Properties of Novel SnWO4 Oxide Semiconductors with Narrow Band Gaps. J. Phys. Chem. C 2009, 113, 10647–10653. [Google Scholar] [CrossRef]
- Kölbach, M.; Pereira, I.J.; Harbauer, K.; Plate, P.; Höflich, K.; Berglund, S.P.; Friedrich, D.; van de Krol, R.; Abdi, F.F. Revealing the performance limiting factors in α-SnWO4 photoanodes for solar water splitting. Chem. Mater. 2018, 30, 8322–8331. [Google Scholar] [CrossRef]
- Jeitschko, W.; Sleight, A. Stannous tungstate: Properties, crystal structure and relationship to ferroelectric SbTaO4 type compounds. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1974, 30, 2088–2094. [Google Scholar] [CrossRef]
- Jeitschko, W.; Sleight, A.W. Synthesis, properties and crystal structure of β-SnWO4. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1972, 28, 3174–3178. [Google Scholar] [CrossRef]
- Ke, J.; Younis, M.A.; Kong, Y.; Zhou, H.; Liu, J.; Lei, L.; Hou, Y. Nanostructured Ternary Metal Tungstate-Based Photocatalysts for Environmental Purification and Solar Water Splitting: A Review. Nano-Micro Lett. 2018, 10, 69. [Google Scholar] [CrossRef]
- Gomes, E.O.; Gouveia, A.F.; Gracia, L.; Lobato, A.; Recio, J.M.; Andres, J. A Chemical-Pressure-Induced Phase Transition Controlled by Lone Electron Pair Activity. J. Phys. Chem. Lett. 2022, 13, 9883–9888. [Google Scholar] [CrossRef]
- Schnell, P.; Kolbach, M.; Schleuning, M.; Obata, K.; Irani, R.; Ahmet, I.Y.; Harb, M.; Starr, D.E.; van de Krol, R.; Abdi, F.F. Interfacial Oxide Formation Limits the Photovoltage of α-SnWO4/NiOx Photoanodes Prepared by Pulsed Laser Deposition. Adv. Energy Mater. 2021, 11, 2003183. [Google Scholar] [CrossRef]
- Harb, M.; Ziani, A.; Takanabe, K. Critical difference between optoelectronic properties of α- and β-SnWO4 semiconductors: A DFT/HSE06 and experimental investigation. Phys. Status Solidi B 2016, 253, 1115–1119. [Google Scholar] [CrossRef]
- Kuzmin, A.; Anspoks, A.; Kalinko, A.; Timoshenko, J.; Kalendarev, R. Extended x-ray absorption fine structure spectroscopy and first-principles study of SnWO4. Phys. Scr. 2014, 89, 044005. [Google Scholar] [CrossRef]
- Thangavel, S.; Venugopal, G.; Kim, S.-J. Enhanced photocatalytic efficacy of organic dyes using β-tin tungstate–reduced graphene oxide nanocomposites. Mater. Chem. Phys. 2014, 145, 108–115. [Google Scholar] [CrossRef]
- Alexei, K.; Andris, A.; Aleksandr, K.; Janis, T.; Robert, K.; Lucie, N.; François, B.; Tetsuo, I.; Pascale, R. Pressure-induced insulator-to-metal transition in α-SnWO4. J. Phys. Conf. Ser. 2016, 712, 012122. [Google Scholar]
- Kuzmin, A.; Zubkins, M.; Kalendarev, R. Preparation and Characterization of Tin Tungstate Thin Films. Ferroelectrics 2015, 484, 49–54. [Google Scholar] [CrossRef]
- Ziani, A.; Harb, M.; Noureldine, D.; Takanabe, K. UV-Vis optoelectronic properties of α-SnWO4: A comparative experimental and density functional theory based study. APL Mater. 2015, 3, 096101. [Google Scholar] [CrossRef]
- Stoltzfus, M.W.; Woodward, P.M.; Seshadri, R.; Klepeis, J.-H.; Bursten, B. Structure and Bonding in SnWO4, PbWO4, and BiVO4: Lone Pairs vs. Inert Pairs. Inorg. Chem. 2007, 46, 3839–3850. [Google Scholar] [CrossRef]
- Wojcik, J.; Calvayrac, F.; Goutenoire, F.; Mhadhbi, N.; Corbel, G.; Lacorre, P.; Bulou, A. Lattice Dynamics of β-SnWO4: Experimental and Ab Initio Calculations. J. Phys. Chem. C 2013, 117, 5301–5313. [Google Scholar] [CrossRef]
- Pavithra, N.S.; Patil, S.B.; Kumar, S.R.K.; Alharthi, F.A.; Nagaraju, G. Facile synthesis of nanocrystalline β-SnWO4: As a photocatalyst, biosensor and anode for Li-ion battery. SN Appl. Sci. 2019, 1, 1123. [Google Scholar] [CrossRef]
- Ungelenk, J.; Feldmann, C. Synthesis of faceted β-SnWO4 microcrystals with enhanced visible-light photocatalytic properties. Chem. Commun. 2012, 48, 7838–7840. [Google Scholar] [CrossRef]
- Huang, J.; Liu, H.; Zhong, J.; Li, J. Enhanced simulated sunlight-driven photocatalytic performance of SnWO4 prepared in the presence of cetyltrimethylammonium bromide. Environ. Prog. Sustain. Energy 2020, 39, e13314. [Google Scholar] [CrossRef]
- Alharthi, F.A.; Shashank, M.; Shashikanth, J.; Viswantha, R.; Alghamdi, A.A.; Algethami, J.; Alsaiari, M.A.; Jalalah, M.S.; Ganganagappa, N. Hydrothermal synthesis of α-SnWO4: Application to lithium-ion battery and photocatalytic activity. Ceram. Int. 2021, 47, 10242–10249. [Google Scholar] [CrossRef]
- Barros, M.M.P.; Almeida, K.C.; Silva, S.A.; Botelho, G. Synthesis and characterization of α-SnWO4 powders obtained by microwave-assisted hydrothermal method. Cerâmica 2022, 68, 236–241. [Google Scholar] [CrossRef]
- Kölbach, M.; Hempel, H.; Harbauer, K.; Schleuning, M.; Petsiuk, A.; Höflich, K.; Deinhart, V.; Friedrich, D.; Eichberger, R.; Abdi, F.F.; et al. Grain Boundaries Limit the Charge Carrier Transport in Pulsed Laser Deposited α-SnWO4 Thin Film Photoabsorbers. ACS Appl. Energy Mater. 2020, 3, 4320–4330. [Google Scholar] [CrossRef]
- Bozheyev, F.; Akinoglu, E.M.; Wu, L.; Lu, H.; Nemkayeva, R.; Xue, Y.; Jin, M.; Giersig, M. Band gap optimization of tin tungstate thin films for solar water oxidation. Int. J. Hydrogen Energy 2020, 45, 8676–8685. [Google Scholar] [CrossRef]
- Gottesman, R.; Peracchi, I.; Gerke, J.L.; Irani, R.; Abdi, F.F.; van de Krol, R. Shining a Hot Light on Emerging Photoabsorber Materials: The Power of Rapid Radiative Heating in Developing Oxide Thin-Film Photoelectrodes. ACS Energy Lett. 2022, 7, 514–522. [Google Scholar] [CrossRef]
- Zhu, S.; Liu, D.; Li, J.; Kuang, Y. Chemical Vapor Deposition of Crystalized Nanoscale α-SnWO4 Thin Films and Their Photoelectrocatalytic Properties. ACS Appl. Energy Mater. 2022, 5, 14372–14380. [Google Scholar] [CrossRef]
- Pyper, K.J.; Evans, T.C.; Bartlett, B.M. Synthesis of α-SnWO4 thin-film electrodes by hydrothermal conversion from crystalline WO3. Chin. Chem. Lett. 2015, 26, 474–478. [Google Scholar] [CrossRef]
- Qiu, W.; Zhang, Y.; He, G.; Chen, L.; Wang, K.; Wang, Q.; Li, W.; Liu, Y.; Li, J. Two-Dimensional Long-Plate SnWO4 Photoanode Exposed Active Facets for Enhanced Solar Water Splitting. ACS Appl. Energy Mater. 2022, 5, 11732–11739. [Google Scholar] [CrossRef]
- Liu, Y.; Qiu, W.; He, G.; Wang, K.; Wang, Y.; Chen, L.; Wu, Q.; Li, W.; Li, J. Nail-like α-SnWO4 Array Film with Increased Reactive Facets for Photoelectrochemical Water Splitting. J. Phys. Chem. C 2022, 126, 15596–15605. [Google Scholar] [CrossRef]
- Mirsadeghi, S.; Zandavar, H.; Tooski, H.F.; Rahimi, M.; Sohouli, E.; Rahimi-Nasrabadi, M.; Ganjali, M.R.; Pourmortazavi, S.M. Rapid photodegradation and detection of zolpidem over β-SnWO4 and α-SnWO4 nanoparticles: Optimization and mechanism. Environ. Sci. Pollut. Res. 2020, 28, 5430–5442. [Google Scholar] [CrossRef]
- Zhu, Z.; Sarker, P.; Zhao, C.; Zhou, L.; Grimm, R.L.; Huda, M.N.; Rao, P.M. Photoelectrochemical Properties and Behavior of α-SnWO4 Photoanodes Synthesized by Hydrothermal Conversion of WO3 Films. ACS Appl. Mater. Interfaces 2017, 9, 1459–1470. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; You, X.; Wang, X.; Yu, C.; Xu, L.; Wang, C.; Song, Y.; Zhang, F. Nest-like α-SnWO4 nanostructures assembled by nanowires: Facile synthesis and their superior photocatalytic performance. J. Alloys Compd. 2019, 802, 502–510. [Google Scholar] [CrossRef]
- Zhu, G.; Que, W.; Zhang, J.; Zhong, P. Photocatalytic activity of SnWO4 and SnW3O9 nanostructures prepared by a surfactant-assisted hydrothermal process. Mater. Sci. Eng. B 2011, 176, 1448–1455. [Google Scholar] [CrossRef]
- Liu, X.; Liang, B.; Yang, J.; Li, W. Solvent effect on morphological evolution and photocatalytic property of α-SnWO4. J. Taiwan Inst. Chem. Eng. 2019, 95, 575–582. [Google Scholar] [CrossRef]
- Wang, Q.L.; Li, H.B.; Jiang, H.Y.; Ding, S.T.; Song, Z.W.; Shi, J.S. Effect of solvent on α-SnWO4 photocatalyst for degradation of methyl orange under visible light irradiation. Adv. Perform. Mater. 2015, 30, 288–293. [Google Scholar] [CrossRef]
- Raj, A.T.; Thangavel, S.; Rose, A.; Jipsa, C.V.; Jose, M.; Nallamuthu, G.; Kim, S.-J.; Venugopal, G. Influence of Morphology and Common Oxidants on the Photocatalytic Property of β-SnWO4 Nanoparticles. J. Nanosci. Nanotechnol. 2016, 16, 2541–2547. [Google Scholar] [CrossRef]
- Zhu, Z.; Tian, H.; Zhang, M.; Liang, B.; Li, W. Preparation of α-SnWO4 hierarchical spheres by Bi3+-doping and their enhanced photocatalytic activity under visible light. Ceram. Int. 2016, 42, 14743–14748. [Google Scholar] [CrossRef]
- Liu, X.; Liang, B.; Zhang, M.; Long, Y.; Li, W. Enhanced photocatalytic properties of α-SnWO4 nanosheets modified by Ag nanoparticles. J. Colloid Interface Sci. 2017, 490, 46–52. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, S.; Zhao, G.; Li, C.; Liu, L.; Jiao, F. Fabrication of SnWO4/ZnFe-layered double hydroxide composites with enhanced photocatalytic degradation of methyl orange. J. Mater. Sci.-Mater. Electron. 2020, 31, 12269–12281. [Google Scholar] [CrossRef]
- Liu, X.; Liang, B.; Li, W. In situ decoration of SnS quantum dots on the α-SnWO4 nanosheets for superior visible-light photocatalytic performance. Appl. Surf. Sci. 2020, 531, 147379. [Google Scholar] [CrossRef]
- Wang, J.; Yan, H.; Long, Y.; Li, W. Enhanced photocatalytic property of α-SnWO4 nanoplates by h-BN decorating. J. Mater. Sci. Mater. Electron. 2021, 32, 21858–21868. [Google Scholar] [CrossRef]
- Yao, S.; Zhang, M.; Di, J.; Wang, Z.; Long, Y.; Li, W. Preparation of α-SnWO4/SnO2 heterostructure with enhanced visible-light-driven photocatalytic activity. Appl. Surf. Sci. 2015, 357, 1528–1535. [Google Scholar] [CrossRef]
- Huang, R.K.; Ge, H.; Lin, X.J.; Guo, Y.L.; Yuan, R.S.; Fu, X.Z.; Li, Z.H. Facile one-pot preparation of α-SnWO4/reduced graphene oxide (RGO) nanocomposite with improved visible light photocatalytic activity and anode performance for Li-ion batteries. RSC Adv. 2013, 3, 1235–1242. [Google Scholar] [CrossRef]
- Su, Y.G.; Hou, L.C.; Du, C.F.; Peng, L.M.; Guan, K.; Wang, X.J. Rapid synthesis of Zn2+ doped SnWO4 nanowires with the aim of exploring doping effects on highly enhanced visible photocatalytic activities. RSC Adv. 2012, 2, 6266–6273. [Google Scholar] [CrossRef]
- Warmuth, L.; Feldmann, C. β-SnWO4 with Morphology-Controlled Synthesis and Facet-Depending Photocatalysis. ACS Omega 2019, 4, 13400–13407. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Lin, Y.-G.; Hsu, L.-C.; Tarasov, A.; Chen, P.-T.; Hayashi, M.; Ungelenk, J.; Hsu, Y.-K.; Feldmann, C. β-SnWO4 Photocatalyst with Controlled Morphological Transition of Cubes to Spikecubes. ACS Catal. 2016, 6, 2357–2367. [Google Scholar] [CrossRef]
- Liang, Q.; Jin, J.; Liu, C.; Xu, S.; Yao, C.; Chen, Z.; Li, Z. Hydrothermal fabrication of α-SnWO4/g-C3N4 heterostructure with enhanced visible-light photocatalytic activity. J. Mater. Sci. Mater. Electron. 2017, 28, 11279–11283. [Google Scholar] [CrossRef]
- Zhuang, H.; Xu, W.; Lin, L.; Huang, M.; Xu, M.; Chen, S.; Cai, Z. Construction of one dimensional ZnWO4@SnWO4 core-shell heterostructure for boosted photocatalytic performance. J. Mater. Sci. Technol. 2019, 35, 2312–2318. [Google Scholar] [CrossRef]
- Chowdhury, A.P.; Shambharkar, B.H. Fabrication and characterization of BiOBr-SnWO4 heterojunction nanocomposites with boosted photodegradation capability. Chem. Eng. J. Adv. 2020, 4, 100040. [Google Scholar] [CrossRef]
- Alharthi, F.A.; Alsaiari, M.A.; Jalalah, M.S.; Shashank, M.; Shashikanth; Alghamdi, A.A.; Algethami, J.S.; Ganganagappa, N. Combustion synthesis of β-SnWO4-rGO: Anode material for Li-ion battery and photocatalytic dye degradation. Ceram. Int. 2021, 47, 10291–10300. [Google Scholar] [CrossRef]
- Elviera; Yulizar, Y.; Apriandanu, D.O.B.; Surya, R.M. Fabrication of novel SnWO4/ZnO using Muntingia calabura L. leaf extract with enhanced photocatalytic methylene blue degradation under visible light irradiation. Ceram. Int. 2022, 48, 3564–3577. [Google Scholar] [CrossRef]
- Ungelenk, J.; Feldmann, C. Nanoscale β-Sn1-nWO4·nα-Sn-A highly efficient photocatalyst for daylight-driven degradation of organic dyes and its real “green” synthesis. Appl. Catal. B-Environ. 2011, 102, 515–520. [Google Scholar] [CrossRef]
- Alharthi, F.A.; AlFawaz, A.; Ahmad, N. Photocatalytic degradation of anionic dye using well-crystalline SnWO4 polyoxometalate. Phys. Scr. 2022, 97, 085813. [Google Scholar] [CrossRef]
- Wei, Q.; Xiong, S.; Li, W.; Jin, C.; Chen, Y.; Hou, L.; Wu, Z.; Pan, Z.; He, Q.; Wang, Y.; et al. Double Z-scheme system of α-SnWO4/UiO-66(NH2)/g-C3N4 ternary heterojunction with enhanced photocatalytic performance for ibuprofen degradation and H2 evolution. J. Alloys Compd. 2021, 885, 160984. [Google Scholar] [CrossRef]
- Liu, D.; Chen, X.; Qiao, Y.; Zhou, Y.; Kuang, Y. Awakening the Photoelectrochemical Activity of α-SnWO4 Photoanodes with extraordinary Crystallinity Induced by Reductive Annealing. Adv. Energy Sustain. Res. 2021, 3, 2100146. [Google Scholar] [CrossRef]
- He, G.; Li, W.; Qiu, W.; Xu, C.; Wang, K.; Chen, L.; Wang, Y.; Liu, Y.; Li, J. Constructing a Two-Dimensional SnWO4 Nanosheet Array Film for Enhanced Photoelectrochemical Performance. ACS Appl. Energy Mater. 2022, 5, 11883–11891. [Google Scholar] [CrossRef]
- Wu, H.; Tan, H.L.; Toe, C.Y.; Scott, J.; Wang, L.; Amal, R.; Ng, Y.H. Photocatalytic and Photoelectrochemical Systems: Similarities and Differences. Adv. Mater. 2019, 32, 1904717. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wygant, B.R.; Kawashima, K.; Mabayoje, O.; Hong, T.E.; Lee, S.-G.; Lin, J.; Kim, J.-H.; Yubuta, K.; Li, W.; et al. Facet effect on the photoelectrochemical performance of a WO3/BiVO4 heterojunction photoanode. Appl. Catal. B 2019, 245, 227–239. [Google Scholar] [CrossRef]
- Chen, L.; Li, W.; Qiu, W.; He, G.; Wang, K.; Liu, Y.; Wu, Q.; Li, J. Oriented CuWO4 Films for Improved Photoelectrochemical Water Splitting. ACS Appl. Energy Mater. 2022, 14, 47737–47746. [Google Scholar] [CrossRef]
- Liu, C.; Chen, L.; Su, X.; Chen, S.; Zhang, J.; Yang, H.; Pei, Y. Activating a TiO2/BiVO4 Film for Photoelectrochemical Water Splitting by Constructing a Heterojunction Interface with a Uniform Crystal Plane Orientation. ACS Appl. Energy Mater. 2022, 14, 2316–2325. [Google Scholar] [CrossRef]
- Harb, M.; Cavallo, L.; Basset, J.-M. Remarkable Influence of α-SnWO4 Exposed Facets on Their Photocatalytic Performance for H2 and O2 Evolution Reactions. J. Phys. Chem. C 2020, 124, 18684–18689. [Google Scholar] [CrossRef]
- Wang, W.; Wu, Y.; Chen, D.-L.; Liu, H.; Xu, M.; Liu, X.; Xin, L. The surface reconstruction induced enhancement of the oxygen evolution reaction on α-SnWO4 (010) based on a density functional theory study. Phys. Chem. Chem. Phys. 2022, 24, 19382–19392. [Google Scholar] [CrossRef] [PubMed]
- Azofra, L.M.; Cavallo, L.; Basset, J.-M.; Harb, M. Need for Rationally Designed SnWO4 Photo(electro)catalysts to Overcome the Performance Limitations for O2 and H2 Evolution Reactions. J. Phys. Chem. C 2021, 125, 8488–8496. [Google Scholar] [CrossRef]
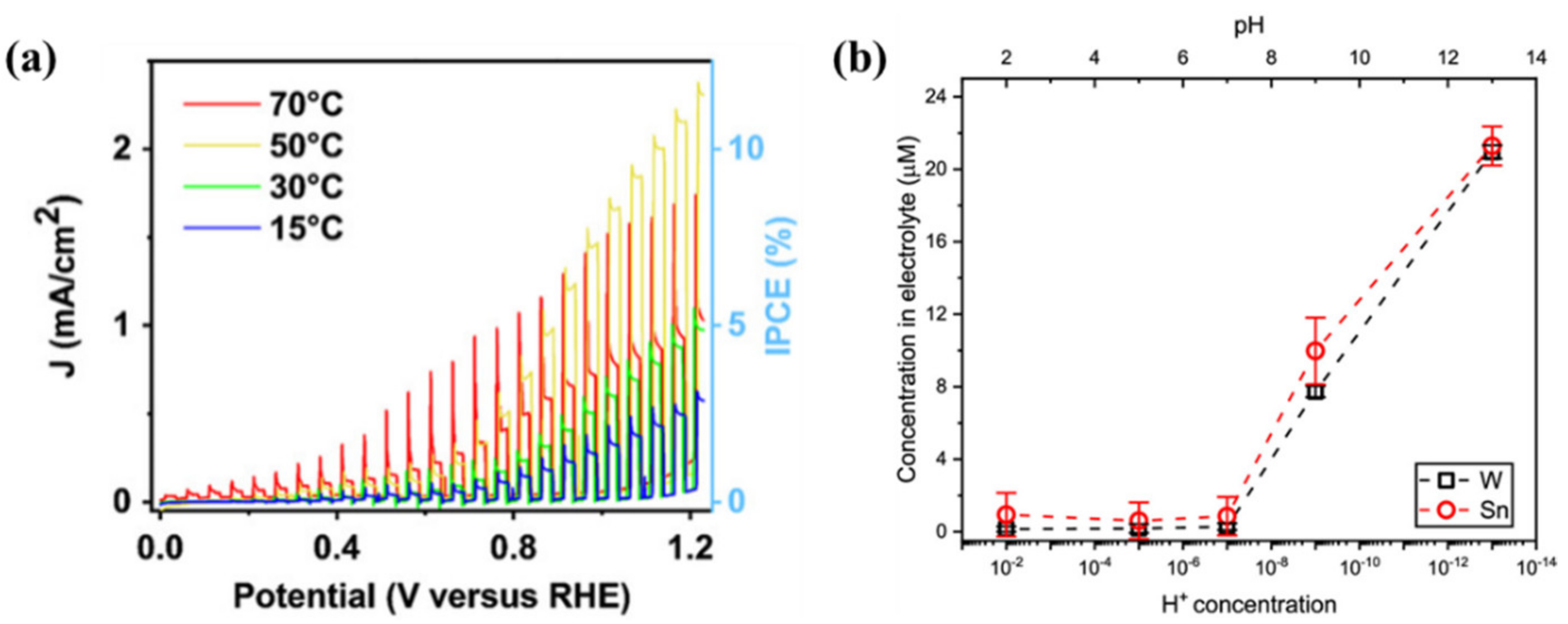
- Liu, Y.; Chen, L.; Zhu, X.; Qiu, H.; Wang, K.; Li, W.; Cao, S.; Zhang, T.; Cai, Y.; Wu, Q.; et al. Effects of operating temperature on photoelectrochemical performance of CuWO4 film photoanode. J. Electroanal. Chem. 2022, 924, 116859. [Google Scholar] [CrossRef]
- Schnell, P.; Dela Cruz, J.M.C.M.; Kölbach, M.; van de Krol, R.; Abdi, F.F. pH-Dependent Stability of α-SnWO4 Photoelectrodes. Chem. Mater. 2022, 34, 1590–1598. [Google Scholar] [CrossRef]

| Type of Catalyst | Organic Pollutant | Light | Degradation Efficiency | Ref. |
|---|---|---|---|---|
| Nest-like α-SnWO4 | methyl orange (MO) | 300 W Xe arc lamp > 420 nm | 96.1% (60 min) | [32] |
| Flower-like α-SnWO4 | MO | 300 W tungsten-halogen lamp > 420 nm | 95% (80 min) | [33] |
| α-SnWO4 nanostrips + small nanosheets | MO | 500 W Xe lamp | 90.4% (30 min) | [34] |
| α-SnWO4 synthesized with different solvents | MO | 300 W Xe lamp > 420 nm | 98% (80 min) | [35] |
| leaf-like β-SnWO4 | MO | 250 W tungsten-halogen lamp | 60% (2 h) | [36] |
| Bi3+-doped α-SnWO4 | MO | 500 W Xe lamp > 420 nm | 95% (2 h) | [37] |
| α-SnWO4/Ag-NPs | MO | 500 W Xe lamp > 420 nm | 97% (70 min) | [38] |
| α-SnWO4/ZnFe-LDH | MO | 500 W Xe lamp | 95.1% (40 min) | [39] |
| SnS/α-SnWO4 | MO | 500 W Xe lamp | 95.6% (90 min) | [40] |
| CTAB-α-SnWO4 | MO | 500 W Xe lamp | 58.7% (30 min) | [20] |
| h-BN/α-SnWO4 | MO | 300 W Xe lamp > 420 nm | 94.7% (90 min) | [41] |
| α-SnWO4/SnO2 | MO | 500 W Xe lamp > 420 nm | 97% (40 min) | [42] |
| β-SnWO4-GO | MO | tungsten-halogen lamp > 420 nm | 90% (25 min) | [12] |
| α-SnWO4-GO | MO | 300 W tungsten-halogen lamp > 420 nm | 41.2% (6 h) | [43] |
| Zn2+ doped α-SnWO4 nanowires | MO | 300 W Hg lamp > 420 nm | ~100% (90 min) | [44] |
| β-SnWO4 with different morphology | rhodamine B (RhB) | AM 1.5 G solar light (100 mW cm−2) | ~22% (2 h) | [45] |
| Cube β-SnWO4 | RhB | 8 W UV-lamp with a monowavelength of 366 nm | TOF:1.14 | [46] |
| Spike-cube β-SnWO4 | RhB | 8 W UV-lamp with a monowavelength of 366 nm | TOF:2.77 | [46] |
| SnS/α-SnWO4 | RhB | 500 W Xe lamp | 97.62% (2 h) | [40] |
| α-SnWO4/g-C3N4 | RhB | 500 W Xe arc lamp > 420 nm | 91% (80 min) | [47] |
| 1D ZnWO4@β-SnWO4 | RhB | 300 W Xe lamp > 420 nm | ~100% (2 h) | [48] |
| β-SnWO4-GO | RhB | under visible light | 91% (25 min) | [12] |
| α-SnWO4 | RhB | 100 W tungsten-halogen lamp > 420 nm | ~82% (4 h) | [3] |
| β-SnWO4 | RhB | 100 W tungsten-halogen lamp > 420 nm | ~97% (4 h) | [3] |
| BiOBr/α-SnWO4 | RhB | sunlight | 97.9% (1 h) | [49] |
| spike-cube β-SnWO4 | methylene blue (MB) | 8 W UV-lamp with a monowavelength of 366 nm | TOF:2.65 | [43] |
| β-SnWO4-rGO nanocomposite | MB | tungsten-halogen lamp > 420 nm | 94% (2 h) | [50] |
| β-SnWO4/ZnO | MB | 70 W sodium lamps | 82.9% (2 h) | [51] |
| β-SnWO4 truncated rhombic dodecahedrons | MB | 150 W halogen lamp | 100% (20 min) | [19] |
| β-Sn1−nWO4·nα-Sn | MB | halogen bulb (3300 K) | ~90% (1 h) | [52] |
| h-BN/α-SnWO4 | tetracycline (TC) | 300 W Xe lamp > 420 nm | 82.2% (4 h) | [41] |
| SnS/α-SnWO4 | TC | 500 W Xe lamp | 57.0% (3 h) | [40] |
| β-SnWO4-rGO nanocomposite | rose bengal (RB) | tungsten-halogen lamp > 420 nm | 98% (2 h) | [50] |
| β-SnWO4 NPs | RB | 300 W tungsten bulb | 94.6% (2.5 h) | [53] |
| α-SnWO4/UiO-66(NH2)/g-C3N4 | ibuprofen (IPF) | high pressure Xe lamp source simulated sunlight | 95.5% (2 h) | [54] |
| β-Sn1−nWO4·nα-Sn | basic green 4 (BG) | halogen bulb (3300 K) | ~63% (1 h) | [52] |
| BiOBr/α-SnWO4 | BG | sunlight | 95.5% (45 min) | [49] |
| β-Sn1−nWO4·nα-Sn | methyl red (MR) | halogen bulb (3300 K) | ~71% (1 h) | [52] |
| Photoanode | Photocurrent Density (at 1.23 V vs. RHE) | Electrolyte | Ref. |
|---|---|---|---|
| α-SnWO4 nanowires | 0.032 mA cm−2 | 0.1 M KPi buffer (pH ≈ 5) | [27] |
| α-SnWO4 porous nanostructure | 0.080 mA cm−2 | 0.5 M KPi buffer (pH ≈ 7) | [31] |
| α-SnWO4 nanocrystalline particles | ~0.750 mA cm−2 | 0.5 M KPi buffer and 0.5 M Na2SO3 (pH ≈ 7) | [4] |
| α-SnWO4 microcrystalline particles | 0.375 mA cm−2 | 0.5 M Na2SO4 (pH ≈ 7) | [24] |
| α-SnWO4 coral-like morphology | 0.420 mA cm−2 | KOH/H3BO3 buffer and 0.2 M Na2SO3 (pH ≈ 9) | [55] |
| 2D nanosheets α-SnWO4 | 0.411 mA cm−2 | 0.2 M KPi buffer (pH ≈ 7) | [56] |
| Nail-like α-SnWO4 | 0.300 mA cm−2 | 0.2 M KPi buffer (pH ≈ 7) | [29] |
| 2D long-plate α-SnWO4 | 0.790 mA cm−2 | 0.2 M KPi buffer (pH ≈ 7) | [28] |
| NiOx coated RTP-α-SnWO4 | 0.950 mA cm−2 | 0.5 M KPi buffer with 0.5 M Na2SO3 (pH ≈ 7) | [25] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, W.; Liu, Y. Stannous Tungstate Semiconductor for Photocatalytic Degradation and Photoelectrochemical Water Splitting: A Review. Energies 2022, 15, 9194. https://doi.org/10.3390/en15239194
Qiu W, Liu Y. Stannous Tungstate Semiconductor for Photocatalytic Degradation and Photoelectrochemical Water Splitting: A Review. Energies. 2022; 15(23):9194. https://doi.org/10.3390/en15239194
Chicago/Turabian StyleQiu, Weixin, and Yang Liu. 2022. "Stannous Tungstate Semiconductor for Photocatalytic Degradation and Photoelectrochemical Water Splitting: A Review" Energies 15, no. 23: 9194. https://doi.org/10.3390/en15239194
APA StyleQiu, W., & Liu, Y. (2022). Stannous Tungstate Semiconductor for Photocatalytic Degradation and Photoelectrochemical Water Splitting: A Review. Energies, 15(23), 9194. https://doi.org/10.3390/en15239194






