Reference Test Method for Calculating the Thermal Effect of Coal Spontaneous Combustion
Abstract
:1. Introduction
2. Theory
3. Materials and Methods
3.1. Thermal Properties of Test Sample
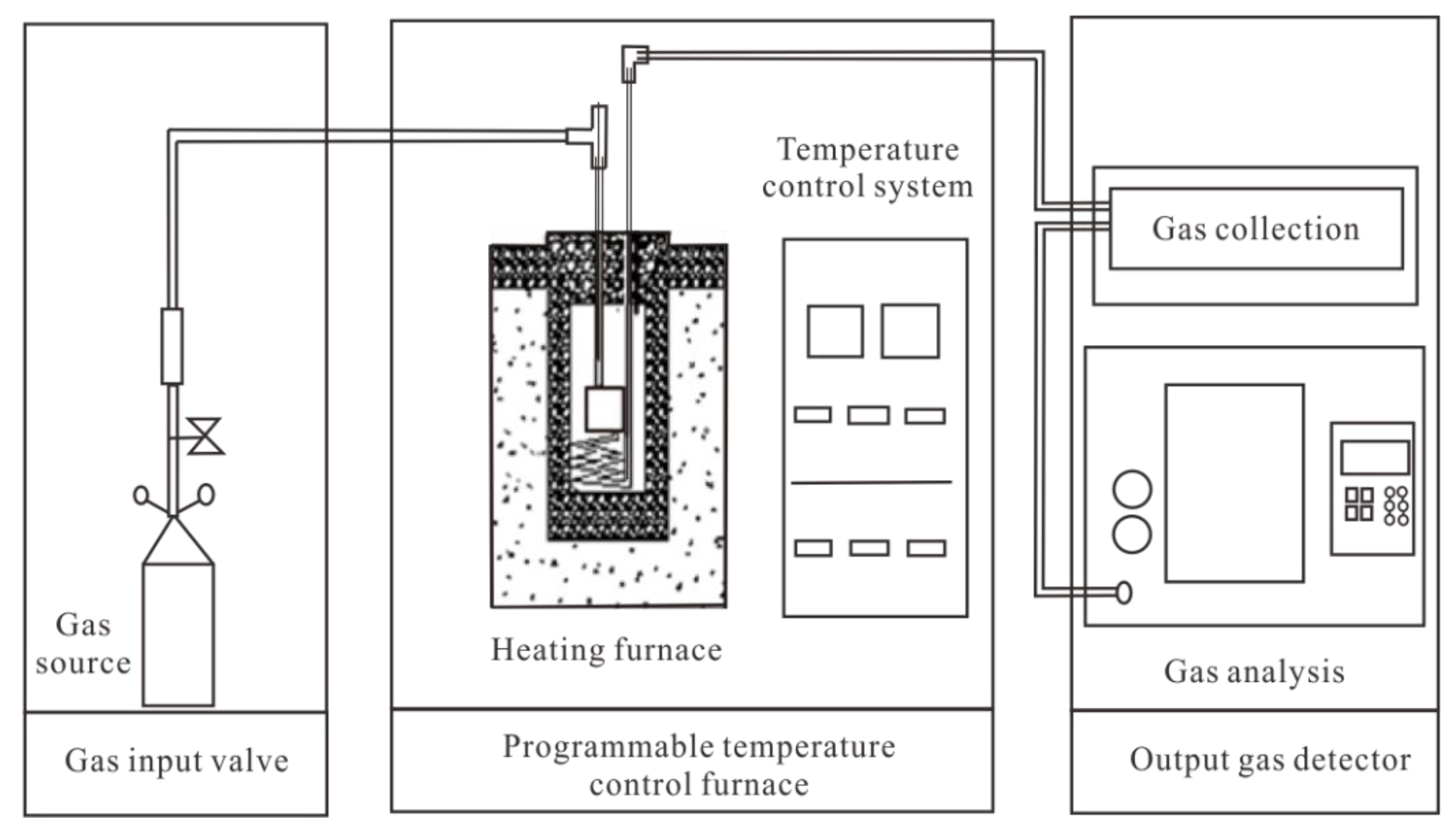
3.2. Experimental System
3.3. Thermophysical Parameters of Reference Sample
3.4. Calculation of Thermal Parameters
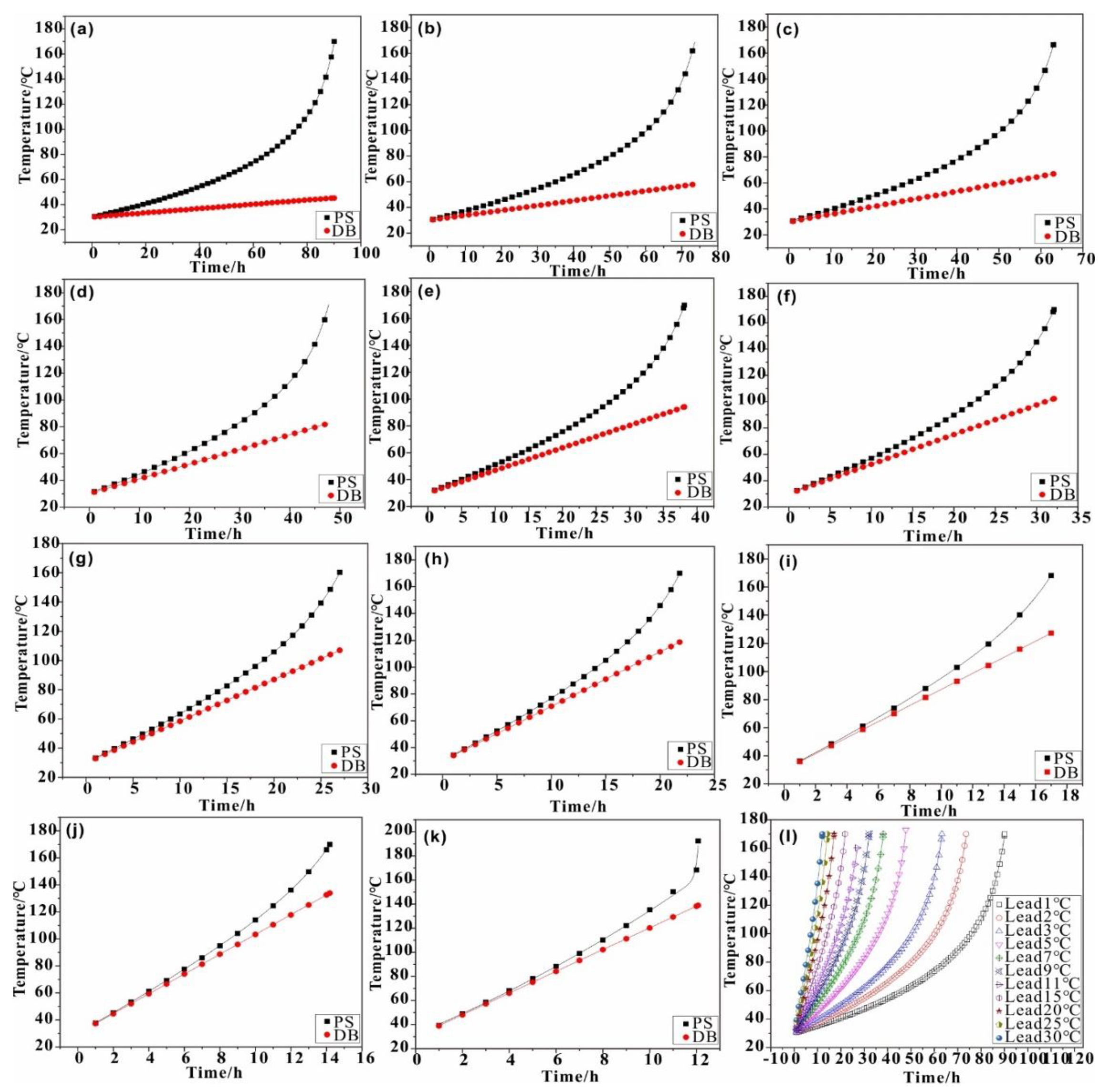
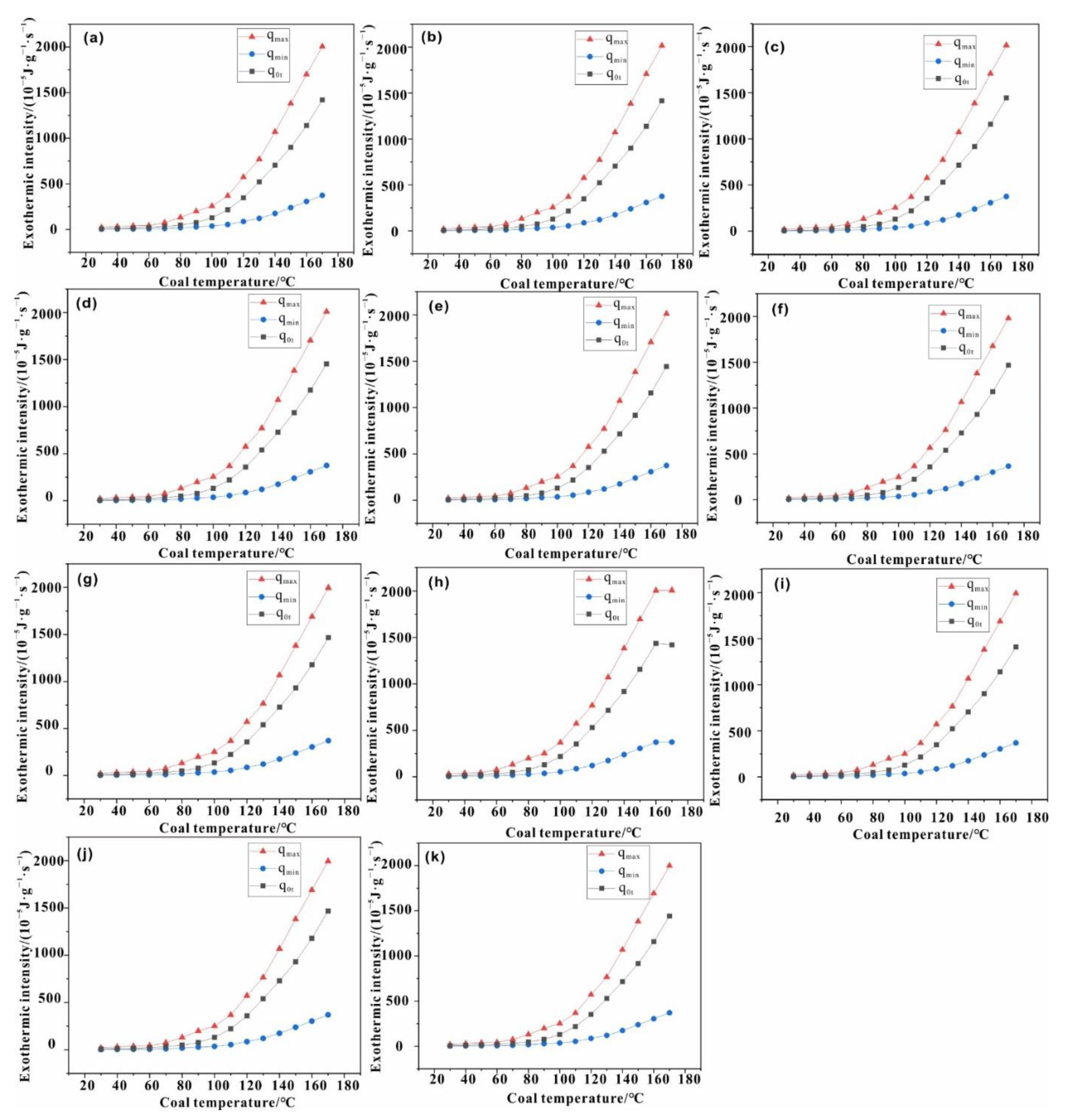
4. Calculation
4.1. Thermal Effect Calculation
4.2. Thermal Effect Verification
5. Conclusions
- Through theoretical analysis, a mathematical model for the thermal effect of CSC was constructed;
- To test the model, a purpose-built experimental system was constructed and data from a physically similar reference material were collected and compared with those of coal;
- The thermal effect of the coal depends primarily on the coal temperature and is less affected by the ambient temperature;
- The thermal effect of the coal increases with the increase in its temperature; the intensity of heat released measured during the experiment is within the constraints imposed by chemical bond energy estimation, lending support to the mathematical model.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| Qc | Maintaining the total heat of the coal sample (J/g) |
| Qm | Total heat of self-oxidation of coal sample (J/g) |
| Qk | Compensation heat of coal sample by experimental system (J/g) |
| Qd | Conduction heat of coal sample tank to coal sample (J/g) |
| Qf | The radiant heat of the coal sample tank to the coal sample (J/g) |
| Qs | Convective heat dissipation of air current to coal sample (J/g) |
| Qt | Other heat during coal spontaneous combustion (J/g) |
| Q | Total heat of coal sample (J/g) |
| C | Specific heat capacity of coal sample (J/g∙°C) |
| M | The mass of the coal sample (g) |
| ΔT | Temperature difference (°C) |
| Tf | Final temperature of coal sample (°C) |
| T0 | Initial temperature of coal sample (°C) |
| f(t) | Coal sample temperature as a function of time |
| q0t | Heat release intensity of coal sample (105 J/(s∙cm3)) |
| t | Time (s) |
| qmin | Minimum heat release intensity of coal sample (105 J/(s∙cm3)) |
| qmax | Maximum heat release intensity of coal sample (105 J/(s∙cm3)) |
| f1(T) | Specific heat capacity fitting formula |
| T | Temperature (°C) |
| f2(T) | Thermal conductivity fitting formula |
| Cc | Apparent heat capacity (J/g∙°C) |
| N | Void ratio |
| λc | Apparent thermal conductivity (J/(cm·s·°C) |
| VT | PS self-heating rate (°C/s) |
References
- Xiao, Y.; Lü, H.F.; Yi, X.; Deng, J.; Shu, C.M. Treating bituminous coal with ionic liquids to inhibit coal spontaneous combustion. J. Therm. Anal. Calorim. 2019, 135, 2711–2721. [Google Scholar] [CrossRef]
- Liu, Y.; Wen, H.; Guo, J.; Jin, Y.F.; Wei, G.M.; Yang, Z.W. Coal spontaneous combustion and N2 suppression in triple goafs: A numerical simulation and experimental study. Fuel 2020, 271, 117625. [Google Scholar] [CrossRef]
- Querol, X.; Zhuang, X.; FonO, T.; Izquierdo, M.; Alastuey, A.; Castro, I.; Drooge, B.L.; Moreno, T.; Grimalt, J.O.; Elvira, J.; et al. Influence of soil cover on reducing the environmental impact of spontaneous coal combustion in coal waste gobs: A review and new experimental data. Int. J. Coal Geol. 2011, 85, 2–22. [Google Scholar] [CrossRef]
- Onifade, M.; Genc, B. A review of research on spontaneous combustion of coal. J. Min. Sci. Technol. Engl. Ed. 2020, 30, 303–311. [Google Scholar] [CrossRef]
- Li, J.L.; Lu, W.; Cao, Y.J.Z.; Kong, B.; Zhang, Q.S. Method of pre-oxidation treatment for spontaneous combustion inhibition and its application. Process Saf. Environ. Prot. 2019, 131, 169–177. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Hu, S.S.; Jiang, S.G.; He, X.J.; Shao, H.; Wang, K.; Fan, D.Q.; Li, W.R. Experimental study on prevention and control of coal spontaneous combustion with heat control inhibitor. J. Loss Prev. Process Ind. 2018, 56, 272–277. [Google Scholar] [CrossRef]
- Song, Z.Y.; Huang, X.Y.; Jiang, J.C.; Pan, X.H. A laboratory approach to CO2 and CO emission factors from underground coal fires. Int. J. Coal Geol. 2020, 219, 103382. [Google Scholar] [CrossRef]
- Deng, J.; Li, B.; Xiao, Y.; Ma, L.; Wang, C.P.; Lai, W.B.; Shu, C.M. Combustion properties of coal gangue using thermogravimetry-Fourier transform infrared spectroscopy. Appl. Therm. Eng. 2017, 116, 244–252. [Google Scholar] [CrossRef]
- Wang, L.C. Study on Critical Transition Characteristics of Coal Oxidation and Spontaneous Combustion; University of Science and Technology of China: Hefei, China, 2021. [Google Scholar]
- Liu, Z.J.; Xu, Y.L.; Wen, X.L.; Lv, Z.G.; Wu, J.D.; Li, M.J.; Wang, L.Y. Thermal Properties and Key Groups Evolution of Low-Temperature Oxidation for Bituminous Coal under Lean-Oxygen Environment. ACS Omega 2021, 6, 15115–15125. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Zhang, Y.T.; Li, Y.Q.; Shi, X.Q.; Xia, S.W.; Guo, Q. Determination and dynamic variations on correlation mechanism between key groups and thermal effect of coal spontaneous combustion. Fuel 2022, 310, 122454. [Google Scholar] [CrossRef]
- Shen, J.; Zhu, H.Q.; Zhang, Z. Experiment analysis of relationship between oxygen concentration and coal oxidation characteristics. Metall. Min. Ind. 2015, 7, 617–622. [Google Scholar]
- Zhang, Y.L.; Wang, J.F.; Xue, S.; Wu, Y.; Li, Z.F.; Chang, L.P. Evaluation of the susceptibility of coal to spontaneous combustion by a tg profile subtraction method. Korean J. Chem. Eng. 2016, 33, 862–872. [Google Scholar] [CrossRef]
- Xu, J.C.; Xue, H.L.; Wen, H.; Li, L. Analysis on influential factors of thermal effect in coal oxidation. China Saf. Sci. J. 2001, 11, 34–39. [Google Scholar]
- Yang, Y.L.; Li, Z.H.; Si, L.L.; Hou, S.S.; Li, Z.W.; Li, J.H. Study on test method of heat release intensity and thermophysical parameters of loose coal. Fuel 2018, 229, 34–43. [Google Scholar] [CrossRef]
- Liang, Y.T. Experimental study on the free diffusing characteristics of oxygen in different granularity loose coal. J. China Coal Soc. 2003, 28, 470–472. [Google Scholar]
- Luo, H.Z.; Liang, Y.T. Current status and perspective of forecast and prediction techniques for spontaneous combustion of coal. China Saf. Sci. J. 2003, 13, 76–78. [Google Scholar]
- Zhou, L.M.; Luo, T.A.; Huang, Q.W. Co-pyrolysis characteristics and kinetics of coal and plastic blends. Energy Convers. Manag. 2008, 50, 705–710. [Google Scholar] [CrossRef]
- Sharma, S.; Ghoshal, A.K. Study of kinetics of co-pyrolysis of coal and waste LDPE blends under argon atmosphere. Fuel 2010, 89, 3943–3951. [Google Scholar] [CrossRef]
- Antonio, J.L.p.; Tromp Peter, J.J.; Moulijn, J.A. Quantitative heat effects associated with pyrolysis of coals, ranging from anthracite to lignite. Fuel 1989, 68, 999–1005. [Google Scholar] [CrossRef]
- Tromp, P.J.J.; Kapteijn, F.; Moulijn, J.A. Characterization of coal pyrolysis by means of differential scanning calorimetry. 1. Quantitative heat effects in an inert atmosphere. Fuel Process. Technol. 1987, 15, 45–57. [Google Scholar] [CrossRef]
- Ma, Q.L.; Han, L.J.; Huang, G.Q. Quantitative characterization of rape straw combustion and gas emissions using tg/dsc-ftir-ms technology. Trans. Chin. Soc. Agric. Mach. 2014, 45, 196–201. [Google Scholar]
- Wang, T.; Li, C.; Zhou, B.X.; Zhang, Y.; Zhang, M.; Yang, H.R.; Wang, Z.Q. Experimental investigation of thermal effect in coal pyrolysis process. Fuel Process. Technol. 2020, 200, 106269. [Google Scholar] [CrossRef]
- Li, L.; Chen, J.C.; Jiang, D.Y.; Chen, J.; Fan, J.Y.; Chu, T.X. Experimental study of the impact of ash on spontaneous combustion characteristics of coal. Chongqing Univ. 2017, 40, 85–92. [Google Scholar]
- Cao, K.; Wang, D.M. Experimental study on heat release characteristics of coal spontaneous combustion under oxygen depletion condition. China Saf. Sci. J. 2015, 25, 127–132. [Google Scholar]
- Hu, X.C. Micro Physicochemical Properties and Macro Thermal Effect of Coal Influenced by Methane-Containing Environment during Oxidation; China University of Mining and Technology: Xuzhou, China, 2017. [Google Scholar]
- Li, Q.W. Fundamental Study on the Thermokinetics Process of Coal Oxidation under Oxygen-Depleted Atmosphere; Xi’an University of Science and Technology: Xi’an, China, 2018. [Google Scholar]
- Pan, L.S.; Yang, Y.G. Study on activation energy of coal low temperature oxidation based on calorimetry analysis. Coal Eng. 2013, 45, 102–105. [Google Scholar]
- Fan, N.; Zhong, K.Q.; Su, L. Thermal kinetics characteristics of coal oxidation spontaneous combustion under different oxygen concentrations. Shaanxi Coal 2022, 41, 12–17. [Google Scholar]
- Chen, L.Z.; Bai, C.W.; Qi, X.Y.; Huang, J.; Ji, Y.C.; Liu, Y.W. Study on exothermic characteristics of oxidation process of Baijiao anthracite Based on Microcalorimetry Technology. Coal Technol. 2018, 37, 305–307. [Google Scholar]
- Kuchta, J.M.; Rowe, V.R.; Burgess, D.S. Spontaneous Combustion Susceptibility of U.S. Coals; U.S. Department of the Interior, Bureau of Mines: Pittsburgh, PA, USA, 1980; Volume 8474, pp. 1–34. [Google Scholar]
- Wang, Y.M.; Shi, G.Q.; Guo, Z.X. Heat transfer and thermodynamic processes in coal-bearing strata under the spontaneous combustion condition. Numer. Heat Transf. Part A Appl. 2017, 71, 1–16. [Google Scholar] [CrossRef]
- Chen, X.K.; Li, H.T.; Wang, Q.H.; Zhang, H.W. Development and application of experimental device for studying oxygen absorption by high temperature loose coal. China Saf. Sci. J. 2015, 25, 29–34. [Google Scholar]
- Tomeczek, J.; Palugniok, H.; Ochman, J. Modelling of deposits formation on heating tubes in pulverized coal boilers. Fuel 2004, 83, 213–221. [Google Scholar] [CrossRef]
- Zhumagulov, M.G. Experimental study of thermophysical properties of Shubarkol coal. Chem. Technol. Fuels Oils 2013, 49, 100–107. [Google Scholar] [CrossRef]
- Beck, M.P.; Yuan, Y.H.; Warrier, P.; Teja, A.S. The effect of particle size on the thermal conductivity of alumina nanofluids. J. Nanoparticle Res. 2008, 11, 1129–1136. [Google Scholar] [CrossRef]
- Wang, W.; Lu, C.F.; Li, Y.X.; Li, Q.T. An investigation on thermal conductivity of fly ash concrete after elevated temperature exposure. Constr. Build. Mater. 2017, 148, 148–154. [Google Scholar] [CrossRef]
- Benazzouk, A.; Douzane, O.; Mezreb, K.; Laidoudi, B.; Quéneudec, M. Thermal conductivity of cement composites containing rubber waste particles: Experimental study and modelling. Constr. Build. Mater. 2008, 22, 573–579. [Google Scholar] [CrossRef]
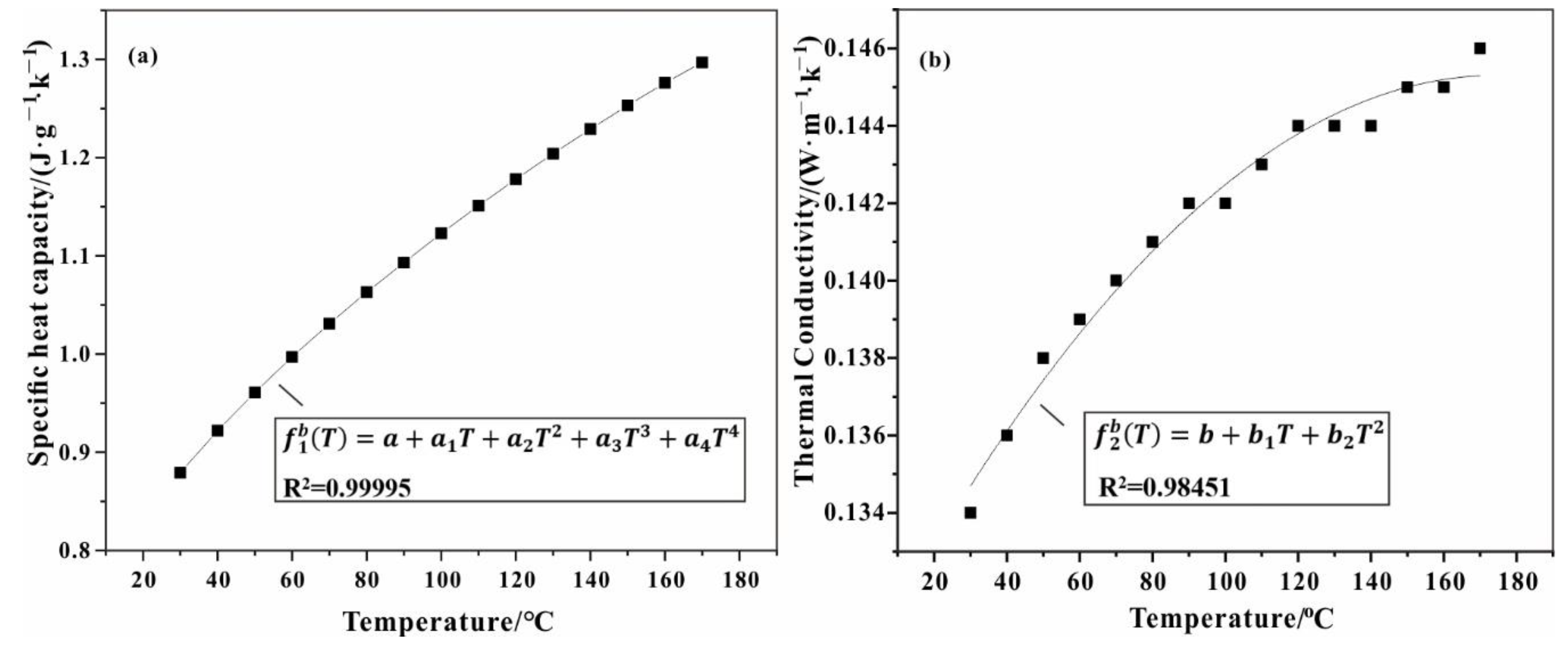
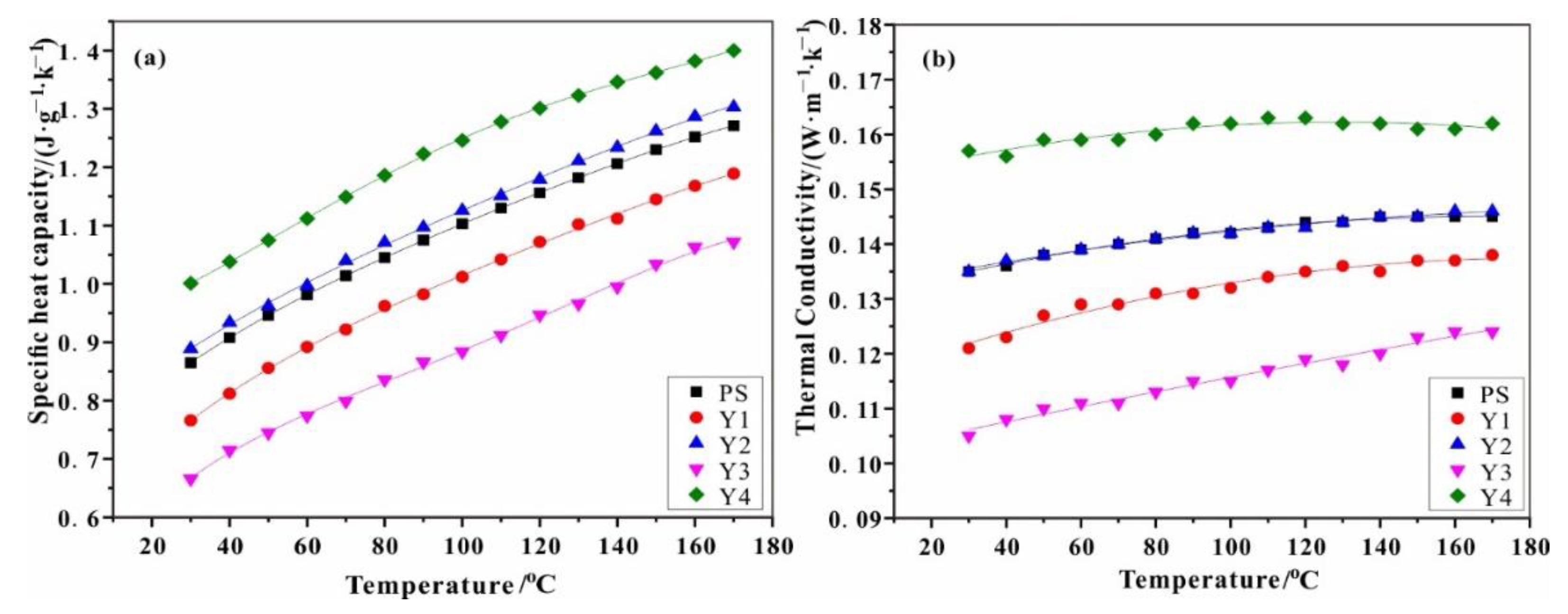
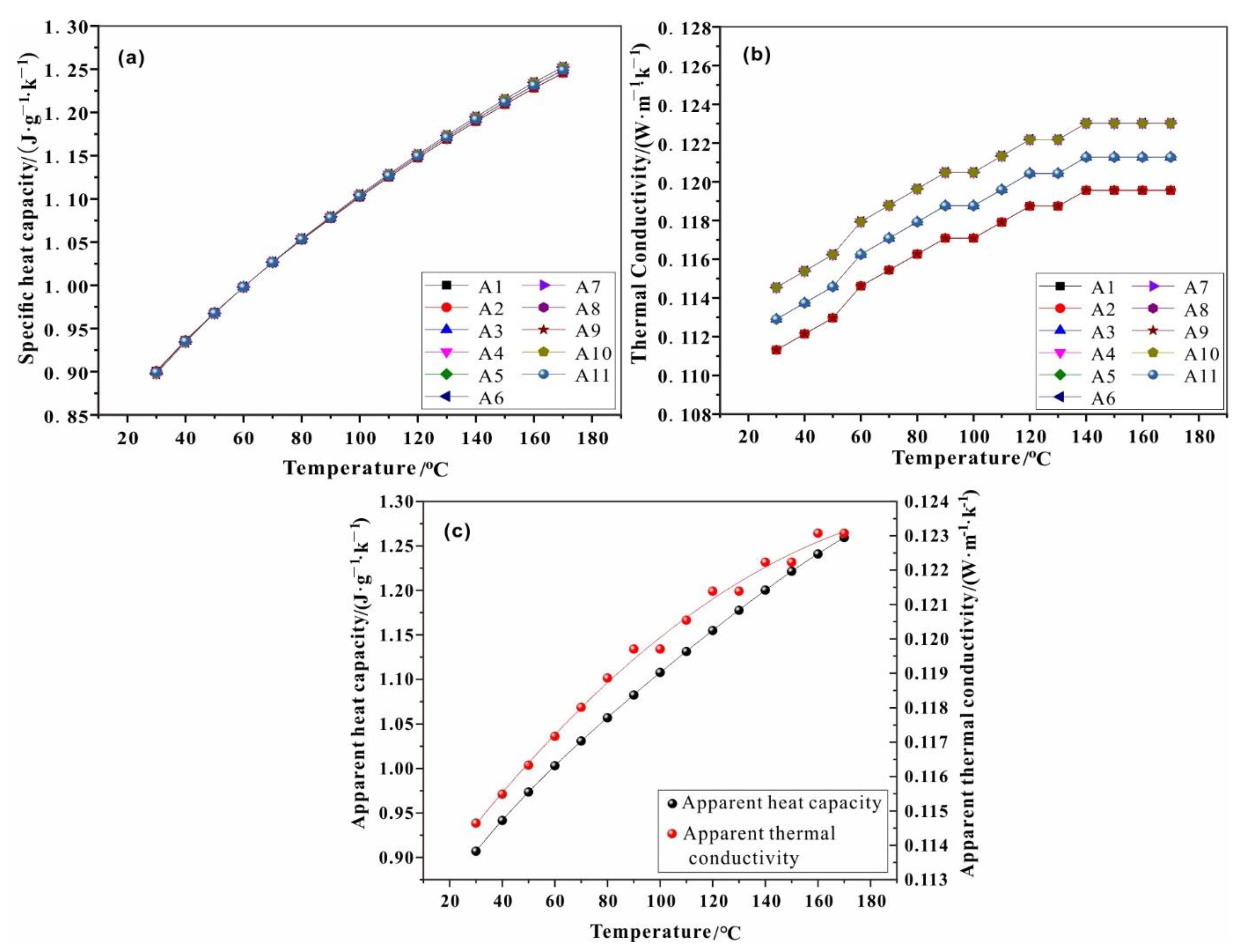
| a | a1 | a2 | a3 | a4 | b1 | b2 | b3 |
|---|---|---|---|---|---|---|---|
| 0.7209 | 0.0062 | −3.5902 × 10−5 | 1.7916 × 10−7 | −3.8416 × 10−10 | 0.1298 | 1.7720 × 10−4 | −5.0743 × 10−5 |
| Reference Sample | White Corundum | Fly Ash | Cement |
|---|---|---|---|
| Y1 | 1 | 8 | 1 |
| Y2 | 1 | 7 | 2 |
| Y3 | 2 | 7 | 1 |
| Y4 | 1.5 | 7 | 1.5 |
| Leading Temperature | Weight (g) | Volume (mm3) | Bulk Density | Porosity | Block Volume | Coal Sample Height (mm) | Residual Height (mm) |
|---|---|---|---|---|---|---|---|
| 1 °C | 160 | 139 | 1.148 | 0.1755 | 114.94 | 71 | 4 |
| 2 °C | 160 | 139 | 1.148 | 0.1755 | 114.94 | 71 | 4 |
| 3 °C | 160 | 137 | 1.164 | 0.1637 | 114.94 | 70 | 5 |
| 5 °C | 160 | 135 | 1.181 | 0.1516 | 114.94 | 69 | 6 |
| 7 °C | 160 | 137 | 1.164 | 0.1637 | 114.94 | 70 | 5 |
| 9 °C | 160 | 135 | 1.181 | 0.1516 | 114.94 | 69 | 6 |
| 11 °C | 160 | 135 | 1.181 | 0.1516 | 114.94 | 69 | 6 |
| 15 °C | 160 | 137 | 1.164 | 0.1637 | 114.94 | 70 | 5 |
| 20 °C | 160 | 139 | 1.148 | 0.1755 | 114.94 | 71 | 4 |
| 25 °C | 160 | 135 | 1.181 | 0.1516 | 114.94 | 69 | 6 |
| 30 °C | 160 | 137 | 1.164 | 0.1637 | 114.94 | 70 | 5 |
| DB | 160 | 137 | 1.164 | 0.1571 | 115.86 | 70 | 5 |
| Leading Temperature/°C | PS | DB | ||||||
|---|---|---|---|---|---|---|---|---|
| y0 | x0 | A1 | t1 | A2 | t2 | a | b | |
| 1 | 8.404 | 42.099 | 0.01 | 5.664 | 47.511 | 55.917 | 30.026 | 0.17 |
| 2 | −1.679 | 31.145 | 0.013 | 5.219 | 57.365 | 54.449 | 30.023 | 0.000038 |
| 3 | −10.632 | 25.376 | 0.016 | 4.792 | 66.821 | 52.767 | 30.016 | 0.0000585 |
| 5 | −27.208 | 16.313 | 0.006 | 3.631 | 82.17 | 46.807 | 30.034 | 0.00011 |
| 7 | −63.794 | 10.241 | 0.019 | 3.714 | 115.135 | 51.23 | 30.028 | 0.000168 |
| 9 | −94.199 | 4.032 | 0.009 | 3.427 | 134.679 | 51.887 | 30.017 | 2.25 |
| 11 | −144.933 | −4.021 | 0.003 | 3.467 | 163.523 | 58.03 | 30.029 | 2.85 |
| 15 | −185.04 | −4.646 | 0.001 | 2.684 | 196.846 | 51.54 | 30.021 | 4.072 |
| 20 | −292.803 | −8.579 | 4.704 | 2.376 | 276.923 | 55.567 | 30.031 | 5.72 |
| 25 | −382.815 | −9.604 | 1.152 | 1.975 | 347.541 | 55.628 | 29.998 | 7.323 |
| 30 | −168.739 | 7.466 | 3.312 | 0.112 | 272.058 | 24.176 | 30.004 | 9.024 |
| Temperature/°C | Leading the Temperature Difference/°C | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 5 | 7 | 9 | 11 | 15 | 20 | 25 | 30 | |
| 30 | 6.4 | 6.4 | 6.6 | 6.5 | 6.4 | 6.5 | 6.5 | 6.4 | 6.4 | 6.5 | 6.4 |
| 40 | 10.2 | 10.2 | 9.9 | 10.9 | 10.6 | 10.5 | 10.2 | 10.3 | 10.5 | 10.6 | 10.2 |
| 50 | 14.8 | 14.5 | 14.3 | 14.7 | 14.7 | 15.0 | 14.7 | 14.4 | 14.4 | 14.9 | 14.6 |
| 60 | 22.1 | 21.8 | 22.1 | 22.4 | 22.1 | 22.6 | 22.4 | 22.1 | 21.8 | 22.5 | 22.2 |
| 70 | 32.4 | 31.8 | 32.7 | 32.8 | 32.5 | 33.1 | 33.0 | 32.5 | 31.9 | 33.0 | 32.5 |
| 80 | 48.1 | 47.7 | 48.4 | 49.1 | 48.6 | 49.3 | 49.1 | 48.4 | 47.8 | 49.3 | 48.5 |
| 90 | 75.2 | 74.5 | 75.9 | 76.8 | 76.0 | 77.1 | 77.0 | 75.8 | 74.7 | 77.1 | 75.9 |
| 100 | 127.8 | 127.6 | 129.7 | 131.3 | 129.7 | 131.8 | 131.7 | 129.5 | 127.5 | 131.8 | 129.7 |
| 110 | 215.3 | 215.6 | 219.1 | 222.2 | 218.9 | 222.4 | 222.6 | 218.9 | 215.4 | 222.4 | 219.0 |
| 120 | 347.4 | 347.0 | 352.9 | 358.0 | 352.8 | 358.6 | 358.5 | 352.5 | 346.9 | 358.6 | 352.8 |
| 130 | 521.9 | 521.9 | 530.4 | 539.6 | 530.4 | 539.2 | 539.2 | 530.6 | 522.1 | 539.2 | 530.4 |
| 140 | 704.5 | 704.2 | 715.9 | 727.5 | 716.0 | 728.0 | 727.9 | 715.7 | 704.1 | 728.0 | 716.0 |
| 150 | 900.9 | 900.6 | 916.1 | 935.3 | 915.9 | 931.5 | 931.5 | 917.8 | 902.6 | 931.5 | 915.9 |
| 160 | 1138.8 | 1138.9 | 1158.0 | 1177.0 | 1158.1 | 1178.1 | 1178.0 | 1157.5 | 1138.3 | 1178.1 | 1158.1 |
| 170 | 1420.1 | 1416.2 | 1444.3 | 1453.1 | 1442.4 | 1467.5 | 1467.4 | 1436.3 | 1411.2 | 1467.5 | 1442.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, W.; Zhang, W.; Ma, S.; Zhang, Z. Reference Test Method for Calculating the Thermal Effect of Coal Spontaneous Combustion. Energies 2022, 15, 7707. https://doi.org/10.3390/en15207707
Liu W, Zhang W, Ma S, Zhang Z. Reference Test Method for Calculating the Thermal Effect of Coal Spontaneous Combustion. Energies. 2022; 15(20):7707. https://doi.org/10.3390/en15207707
Chicago/Turabian StyleLiu, Wenyong, Wenzhao Zhang, Shuai Ma, and Ze Zhang. 2022. "Reference Test Method for Calculating the Thermal Effect of Coal Spontaneous Combustion" Energies 15, no. 20: 7707. https://doi.org/10.3390/en15207707
APA StyleLiu, W., Zhang, W., Ma, S., & Zhang, Z. (2022). Reference Test Method for Calculating the Thermal Effect of Coal Spontaneous Combustion. Energies, 15(20), 7707. https://doi.org/10.3390/en15207707






