Bio-Oil Production from Multi-Waste Biomass Co-Pyrolysis Using Analytical Py–GC/MS
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials Procurement and Characterization
2.2. Thermal Degradation Using Thermogravimetric Analyses (TGA)
2.3. Py-GC-MS Setup for Bio-Oil Production and Analysis
3. Results and Discussion
3.1. Proximate and Elemental Analyses of Biomass Feeds
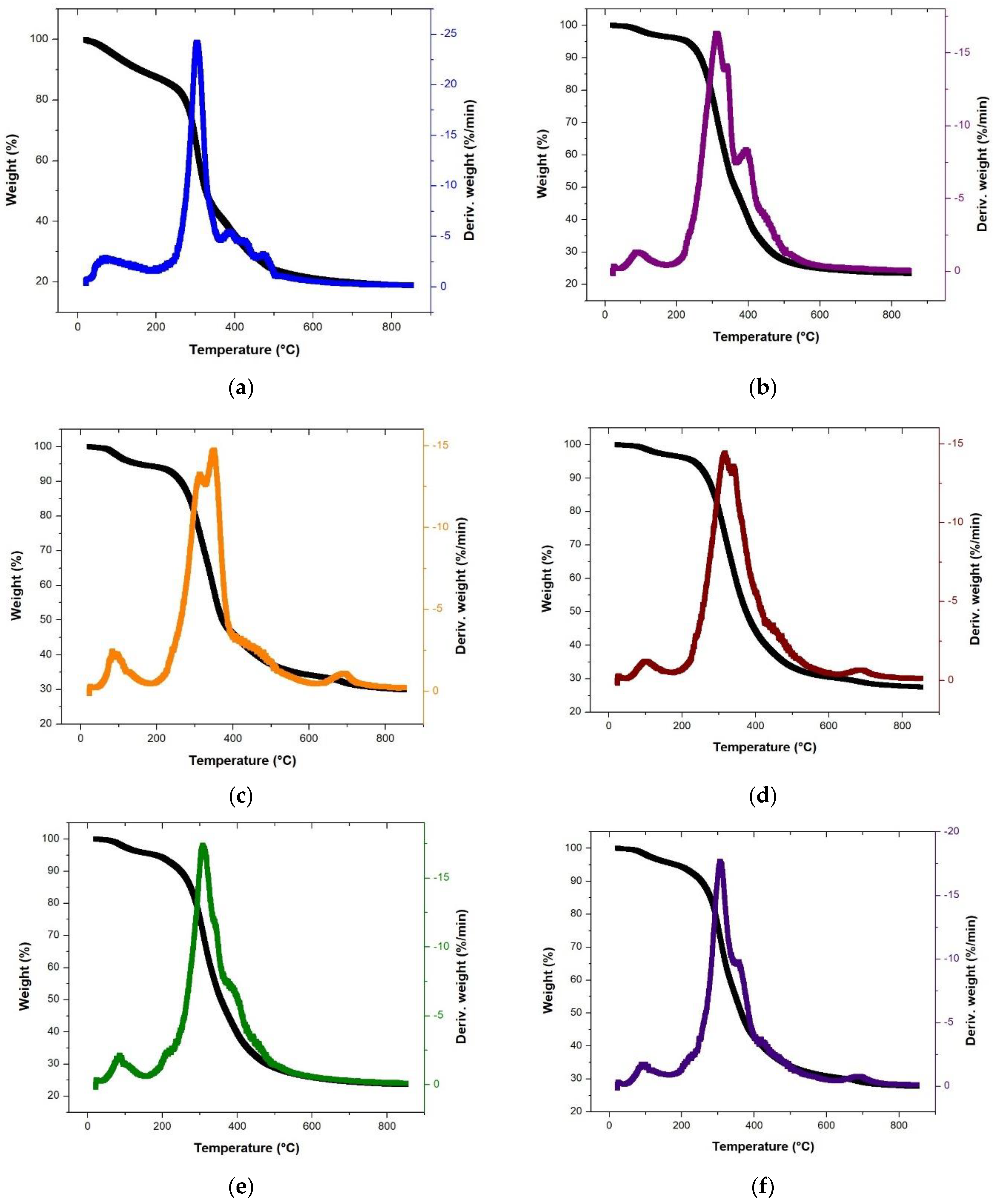
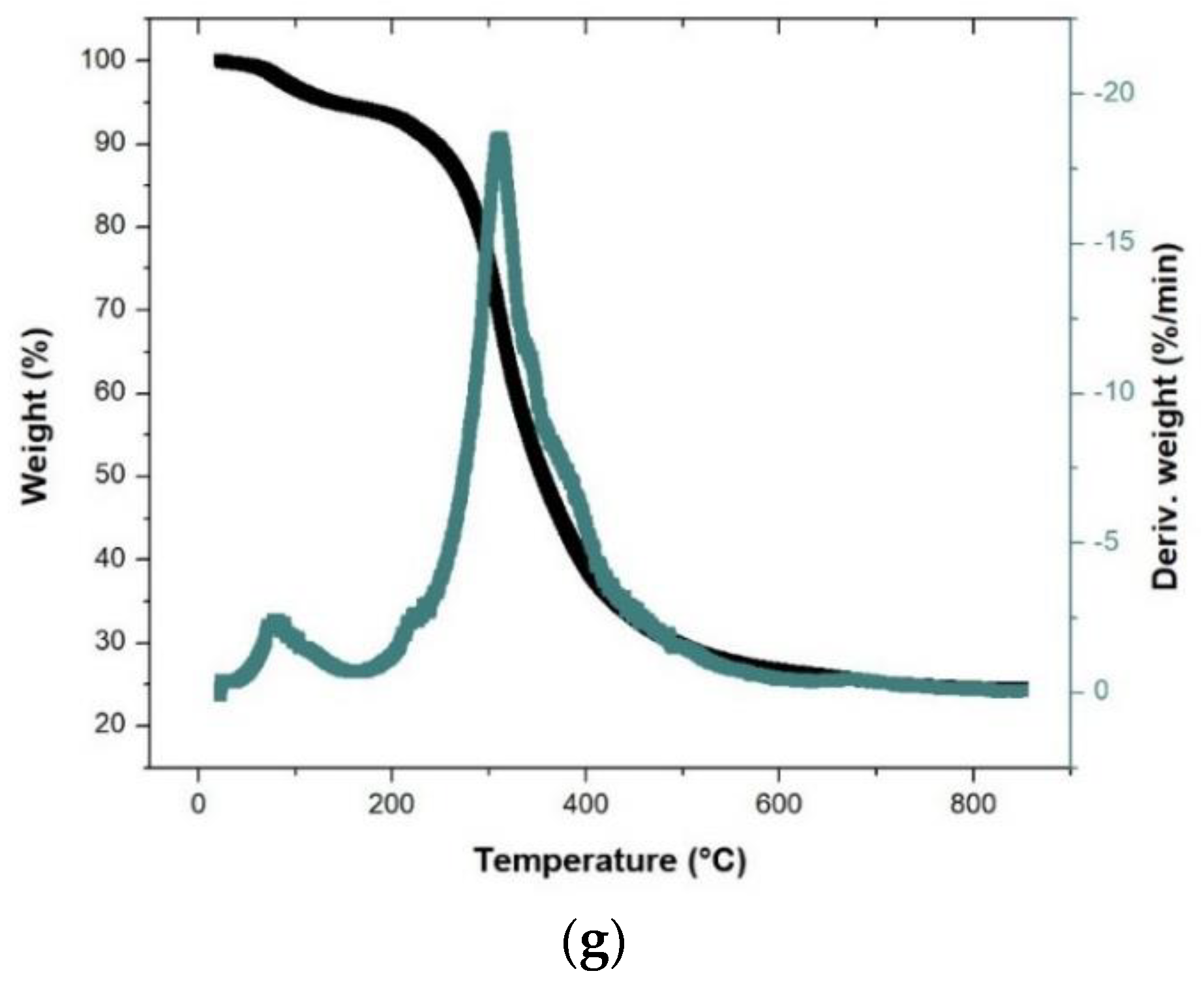
3.2. Thermal Degradation Behavior during Pyrolysis of Single, Binary, and Ternary Samples
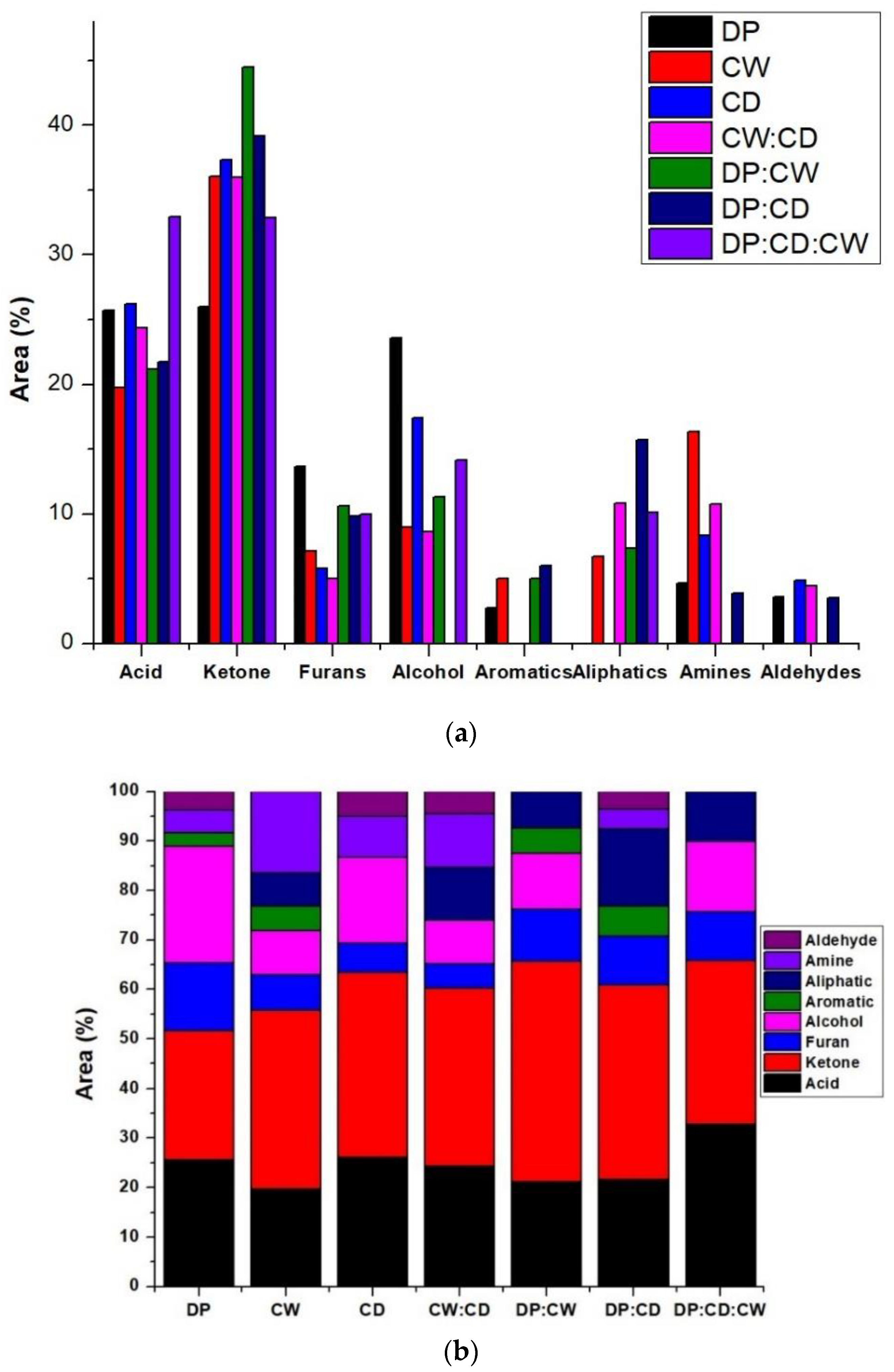
3.3. Bio-Oil Composition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hansen, S.; Mirkouei, A.; Diaz, L.A. A Comprehensive State-of-Technology Review for Upgrading Bio-Oil to Renewable or Blended Hydrocarbon Fuels. Renew. Sustain. Energy Rev. 2020, 118, 109548. [Google Scholar] [CrossRef]
- Haghighat, M.; Majidian, N.; Hallajisani, A.; Samipourgiri, M. Production of Bio-Oil from Sewage Sludge: A Review on the Thermal and Catalytic Conversion by Pyrolysis. Sustain. Energy Technol. Assess. 2020, 42, 100870. [Google Scholar] [CrossRef]
- Gupta, S.; Mondal, P.; Borugadda, V.B.; Dalai, A.K. Advances in Upgradation of Pyrolysis Bio-Oil and Biochar towards Improvement in Bio-Refinery Economics: A Comprehensive Review. Environ. Technol. Innov. 2021, 21, 101276. [Google Scholar] [CrossRef]
- Hu, X.; Gholizadeh, M. Progress of the Applications of Bio-Oil. Renew. Sustain. Energy Rev. 2020, 134, 110124. [Google Scholar] [CrossRef]
- Kumar, V.; Nanda, M. Biomass Pyrolysis-Current Status and Future Directions. Energy Sources Part A Recover. Util. Environ. Eff. 2016, 38, 2914–2921. [Google Scholar] [CrossRef]
- Hoang, A.T.; Ong, H.C.; Fattah, I.M.R.; Chong, C.T.; Cheng, C.K.; Sakthivel, R.; Ok, Y.S. Progress on the Lignocellulosic Biomass Pyrolysis for Biofuel Production toward Environmental Sustainability. Fuel Process. Technol. 2021, 223, 106997. [Google Scholar] [CrossRef]
- Ahorsu, R.; Medina, F.; Constantí, M. Significance and Challenges of Biomass as a Suitable Feedstock for Bioenergy and Biochemical Production: A Review. Energies 2018, 11, 3366. [Google Scholar] [CrossRef]
- Dai, L.; Zhou, N.; Li, H.; Deng, W.; Cheng, Y.; Wang, Y.; Liu, Y.; Cobb, K.; Lei, H.; Chen, P.; et al. Recent Advances in Improving Lignocellulosic Biomass-Based Bio-Oil Production. J. Anal. Appl. Pyrolysis 2020, 149, 104845. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, S.; Uzoejinwa, B.B.; Zheng, A.; Wang, Q.; Huang, J.; Abomohra, A.E.-F. A State-of-the-Art Review on Dual Purpose Seaweeds Utilization for Wastewater Treatment and Crude Bio-Oil Production. Energy Convers. Manag. 2020, 222, 113253. [Google Scholar] [CrossRef]
- Grams, J. Chromatographic Analysis of Bio-Oil Formed in Fast Pyrolysis of Lignocellulosic Biomass. Rev. Anal. Chem. 2020, 39, 65–77. [Google Scholar] [CrossRef]
- Li, G.; Ji, F.; Bai, X.; Zhou, Y.; Dong, R.; Huang, Z. Comparative Study on Thermal Cracking Characteristics and Bio-Oil Production from Different Microalgae (Chlorella pyrenoidosa and Schizochytrium limacinum) Biomass by Py-GC/MS. Int. J. Agric. Biol. Eng. 2019, 12, 208–213. [Google Scholar] [CrossRef]
- Chen, D.; Wang, Y.; Liu, Y.; Cen, K.; Cao, X.; Ma, Z.; Li, Y. Comparative Study on the Pyrolysis Behaviors of Rice Straw under Different Washing Pretreatments of Water, Acid Solution, and Aqueous Phase Bio-Oil by Using TG-FTIR and Py-GC/MS. Fuel 2019, 252, 1–9. [Google Scholar] [CrossRef]
- Rahman, M.M.; Sarker, M.; Chai, M.; Li, C.; Liu, R.; Cai, J. Potentiality of Combined Catalyst for High Quality Bio-Oil Production from Catalytic Pyrolysis of Pinewood Using an Analytical Py-GC/MS and Fixed Bed Reactor. J. Energy Inst. 2020, 93, 1737–1746. [Google Scholar] [CrossRef]
- Edmunds, C.W.; Molina, E.A.R.; André, N.; Hamilton, C.; Park, S.; Fasina, O.; Adhikari, S.; Kelley, S.S.; Tumuluru, J.S.; Rials, T.G.; et al. Blended Feedstocks for Thermochemical Conversion: Biomass Characterization and Bio-Oil Production from Switchgrass-Pine Residues Blends. Front. Energy Res. 2018, 6, 79. [Google Scholar] [CrossRef]
- Primaz, C.T.; Schena, T.; Lazzari, E.; Caramão, E.B.; Jacques, R.A. Influence of the Temperature in the Yield and Composition of the Bio-Oil from the Pyrolysis of Spent Coffee Grounds: Characterization by Comprehensive Two Dimensional Gas Chromatography. Fuel 2018, 232, 572–580. [Google Scholar] [CrossRef]
- Niazmand, R. Date Wastes and By-Products: Chemistry, Processing, and Utilization. In Handbook of Fruit Wastes and By-Products; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Abingdon, UK, 2022; p. 18. ISBN 9781003164463. [Google Scholar]
- Tallou, A.; Haouas, A.; Jamli, M.Y.; Khadija, A.; Soumia, A.; Faissal, A. Review on Cow Manure as Renewable Energy. In Smart Village Technology; Patnaik, S., Siddhartha, S., Mahmoud, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; pp. 341–352. ISBN 978-3-030-37793-9. [Google Scholar]
- Alherbawi, M.; McKay, G.; Mackey, H.R.; Al-Ansari, T. Multi-Biomass Refinery Siting: A GIS Geospatial Optimisation Approach. Chem. Eng. Trans. 2022, 92, 73–78. [Google Scholar] [CrossRef]
- Giwa, A.S.; Xu, H.; Chang, F.; Zhang, X.; Ali, N.; Yuan, J.; Wang, K. Pyrolysis Coupled Anaerobic Digestion Process for Food Waste and Recalcitrant Residues: Fundamentals, Challenges, and Considerations. Energy Sci. Eng. 2019, 7, 2250–2264. [Google Scholar] [CrossRef]
- Castellano-Hinojosa, A.; Armato, C.; Pozo, C.; González-Martínez, A.; González-López, J. New Concepts in Anaerobic Digestion Processes: Recent Advances and Biological Aspects. Appl. Microbiol. Biotechnol. 2018, 102, 5065–5076. [Google Scholar] [CrossRef]
- Tayibi, S.; Monlau, F.; Bargaz, A.; Jimenez, R.; Barakat, A. Synergy of Anaerobic Digestion and Pyrolysis Processes for Sustainable Waste Management: A Critical Review and Future Perspectives. Renew. Sustain. Energy Rev. 2021, 152, 111603. [Google Scholar] [CrossRef]
- Font-Palma, C. Methods for the Treatment of Cattle Manure—A Review. C 2019, 5, 27. [Google Scholar] [CrossRef]
- Ma, Z.; Xie, J.; Gao, N.; Quan, C. Pyrolysis Behaviors of Oilfield Sludge Based on Py-GC/MS and DAEM Kinetics Analysis. J. Energy Inst. 2019, 92, 1053–1063. [Google Scholar] [CrossRef]
- Mumbach, G.D.; Alves, J.L.F.; da Silva, J.C.G.; Di Domenico, M.; Arias, S.; Pacheco, J.G.A.; Marangoni, C.; Machado, R.A.F.; Bolzan, A. Prospecting Pecan Nutshell Pyrolysis as a Source of Bioenergy and Bio-Based Chemicals Using Multicomponent Kinetic Modeling, Thermodynamic Parameters Estimation, and Py-GC/MS Analysis. Renew. Sustain. Energy Rev. 2022, 153, 111753. [Google Scholar] [CrossRef]
- Kaewpengkrow, P.R.; Atong, D.; Sricharoenchaikul, V. Bio-Fuel Production from Catalytic Fast Pyrolysis of Jatropha Wastes Using Pyroprobe GC/MS and Drop Tube Pyrolyzer. J. Anal. Appl. Pyrolysis 2022, 165, 105574. [Google Scholar] [CrossRef]
- Iqbal, T.; Lu, Q.; Dong, C.Q.; Zhou, M.X.; Arain, Z.; Ali, Z.; Khan, I.; Hussain, Z.; Abbas, A. A Study of Product Distribution under Fast Pyrolysis of Wheat Stalk While Producing Bio-Oil. In Proceedings of the 2018 International Conference on Computing, Mathematics and Engineering Technologies (iCoMET), Sukkur, Pakistan, 3–4 March 2018; pp. 1–6. [Google Scholar] [CrossRef]
- Huber, G.W.; Iborra, S.; Corma, A. Synthesis of Transportation Fuels from Biomass: Chemistry, Catalysts, and Engineering. Chem. Rev. 2006, 106, 4044–4098. [Google Scholar] [CrossRef] [PubMed]
- Bensidhom, G.; Arabiourrutia, M.; Ben Hassen Trabelsi, A.; Cortazar, M.; Ceylan, S.; Olazar, M. Fast Pyrolysis of Date Palm Biomass Using Py-GCMS. J. Energy Inst. 2021, 99, 229–239. [Google Scholar] [CrossRef]
- He, S.; Cao, C.; Wang, J.; Yang, J.; Cheng, Z.; Yan, B.; Pan, Y.; Chen, G. Pyrolysis Study on Cattle Manure: From Conventional Analytical Method to Online Study of Pyrolysis Photoionization Time-of-Flight Mass Spectrometry. J. Anal. Appl. Pyrolysis 2020, 151, 104916. [Google Scholar] [CrossRef]
- Krause, M.C.; Moitinho, A.C.; Ferreira, L.F.R.; de Souza, R.L.; Krause, L.C.; Caramão, E.B. Production and Characterization of the Bio-Oil Obtained by the Fast Pyrolysis of Spent Coffee Grounds of the Soluble Coffee Industry. J. Braz. Chem. Soc. 2019, 30, 1608–1615. [Google Scholar] [CrossRef]
- Zhang, J.; Zou, H.; Liu, J.; Evrendilek, F.; Xie, W.; He, Y.; Buyukada, M. Comparative (Co-)Pyrolytic Performances and by-Products of Textile Dyeing Sludge and Cattle Manure: Deeper Insights from Py-GC/MS, TG-FTIR, 2D-COS and PCA Analyses. J. Hazard. Mater. 2021, 401, 123276. [Google Scholar] [CrossRef]
- Hidayat, S.; Abu Bakar, M.S.; Yang, Y.; Phusunti, N.; Bridgwater, A.V. Characterisation and Py-GC/MS Analysis of Imperata Cylindrica as Potential Biomass for Bio-Oil Production in Brunei Darussalam. J. Anal. Appl. Pyrolysis 2018, 134, 510–519. [Google Scholar] [CrossRef]
- Liu, C.; Duan, X.; Chen, Q.; Chao, C.; Lu, Z.; Lai, Q.; Megharaj, M. Investigations on Pyrolysis of Microalgae Diplosphaera Sp. MM1 by TG-FTIR and Py-GC/MS: Products and Kinetics. Bioresour. Technol. 2019, 294, 122126. [Google Scholar] [CrossRef]
| DP | CW | CD | CW:CD | DP:CW | DP:CD | DP:CD:CW | |
|---|---|---|---|---|---|---|---|
| Proximate analysis (% w/w) * | |||||||
| Moisture | 8.15 | 8.62 | 5.24 | 6.06 | 7.14 | 5.84 | 5.97 |
| Volatiles | 69.89 | 75.71 | 57.62 | 69.36 | 73.36 | 66.31 | 69.00 |
| Ash | 0.280 | 1.86 | 19.16 | 7.71 | 1.35 | 9.16 | 4.69 |
| Fixed carbon ** | 21.68 | 13.81 | 17.98 | 16.88 | 18.15 | 18.69 | 20.34 |
| Elemental analysis (% w/w) *** | |||||||
| Carbon | 45.95 | 44.48 | 34.22 | 38.17 | 44.10 | 38.99 | 40.65 |
| Hydrogen | 6.20 | 5.75 | 4.50 | 5.33 | 5.12 | 5.12 | 5.32 |
| Oxygen ** | 43.30 | 46.14 | 39.73 | 46.80 | 46.33 | 43.54 | 46.61 |
| Sulphur | - | - | - | - | - | - | - |
| Nitrogen | 4.27 | 1.77 | 2.39 | 1.99 | 3.10 | 3.19 | 2.73 |
| DP | CW | CD | CW:CD | DP:CW | DP:CD | DP:CD:CW | |
|---|---|---|---|---|---|---|---|
| P1Ti | 200.9 | 175.0 | 182.7 | 168.1 | 161.6 | 167.3 | 165.5 |
| P1TF | 517.4 | 619.9 | 618.9 | 604.6 | 611.7 | 575.2 | 607.3 |
| PmaxT1 | 304.6 | 309.3 | 348.9 | 315.1 | 307.5 | 306.8 | 309.4 |
| Wmax DTG1 | 24.2 | 16.3 | 14.8 | 14.4 | 17.4 | 17.7 | 18.5 |
| WT1 | 22.0 | 24.7 | 33.9 | 30.4 | 25.6 | 31.3 | 26.6 |
| Total weight loss | 81.3 | 76.7 | 70.4 | 72.6 | 76.6 | 72.5 | 73.8 |
| Residual weight % | 18.8 | 23.3 | 29.9 | 27.5 | 23.5 | 27.7 | 24.5 |
| Area (%) | Compound | Formula | Compound Family | DP | CW | CD | CW:CD | DP:CW | DP:CD | DP:CD:CW |
|---|---|---|---|---|---|---|---|---|---|---|
| 26.19–17.19 | Acetic acid | C2H4O2 | Acid | √ | √ | √ | √ | √ | √ | √ |
| 19.10–11.52 | 2-Propanone, 1-hydroxy- | C3H6O2 | Ketone | √ | √ | √ | √ | √ | √ | √ |
| 8.48–6.52 | Benzyl methyl ketone | C9H10O | Ketone | √ | √ | √ | √ | √ | √ | √ |
| 5.01–2.306 | Furan, 2,5-dimethyl- | C6H8O | Furan | √ | √ | √ | √ | √ | √ | |
| 8.68–5.78 | Furfural | Furan | √ | √ | √ | √ | √ | |||
| 11.35–8.63 | 3-Furanmethanol | C5H6O2 | Alcohol | √ | √ | √ | √ | √ | ||
| 8.30–4.32 | 1,2-Cyclopentanedione | C5H6O2 | Ketone | √ | √ | √ | √ | √ | √ | |
| 10.80–7.36 | 1-Pentyne, 4-methyl- | C6H10 | Aliphatic | √ | √ | √ | √ | |||
| 3.18–2.71 | 2-Butanone, 1-(acetyloxy)- | C6H10O3 | Ketone | √ | √ | √ | ||||
| 5.03–2.53 | 1H-Imidazole, 4,5-dihydro-2-methyl- | C12H20N2O12 | N-containing aromatic | √ | √ | √ | ||||
| 8.08–7.48 | 1-Propen-2-ol, acetate | C5H8O2 | Alcohol | √ | √ | |||||
| 9.95–4.45 | 3-Hexen-1-ol, acetate, (Z)- | C8H14O2 | Alcohol | √ | √ | |||||
| 9.29 | Hydroxylamine, O-(3-methylbutyl)- | C5H13NO | Amine | √ | √ | |||||
| 4.69 | Pyrrolidine, N-(4-methyl-3-pentenyl) | C10H19N | Amine | √ | ||||||
| 3.82 | 2-Propenoic acid, 4-methylpentyl ester | C9H16O2 | Acid | √ | ||||||
| 3.59 | 2-Furancarboxaldehyde, 5-methyl- | C6H6O2 | Furan | √ | ||||||
| 2.73 | o-Xylene | C8H10 | Aromatic | √ | √ | |||||
| 2.58 | Citronellic acid | C10H18O2 | Acid | √ | ||||||
| 5.26 | Cyclopentanone, 2-ethyl- | C7H12O | Ketone | √ | ||||||
| 7.08 | Pyridine, 2-(4-nitrobenzylidenamino)- | C13H10N4O3 | Amine | √ | ||||||
| 6.69 | 1,1,6-trimethyl-3-methylene-2-(3,6,9,13-tetrame | C33H56 | Aliphatic | √ | ||||||
| 5.09 | 2-Propanone, 1-(acetyloxy)- | C5H8O3 | Ketone | √ | ||||||
| 8.35 | N-Aminopyrrolidine | C4H10N2 | Amine | √ | ||||||
| 4.90 | 3-Furaldehyde | C5H4O2 | Aldehyde | √ | ||||||
| 4.48 | Octanal | C8H16O | Aldehyde | √ | ||||||
| 4.40 | 3,3,3-Trifluoro-1-piperidin-1-yl-2-trifluoromethyl-propan-1-one | C8H14F3NO | Ketone | √ | ||||||
| 2.39 | Styrene | C8H8 | Aromatic | √ | ||||||
| 2.24 | Bicyclo-hexan-2-one | C6H8O | Ketone | √ | ||||||
| 9.32 | n-Hexadecanoic acid | C16H32O2 | Acid | √ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mariyam, S.; Alherbawi, M.; Rashid, N.; Al-Ansari, T.; McKay, G. Bio-Oil Production from Multi-Waste Biomass Co-Pyrolysis Using Analytical Py–GC/MS. Energies 2022, 15, 7409. https://doi.org/10.3390/en15197409
Mariyam S, Alherbawi M, Rashid N, Al-Ansari T, McKay G. Bio-Oil Production from Multi-Waste Biomass Co-Pyrolysis Using Analytical Py–GC/MS. Energies. 2022; 15(19):7409. https://doi.org/10.3390/en15197409
Chicago/Turabian StyleMariyam, Sabah, Mohammad Alherbawi, Naim Rashid, Tareq Al-Ansari, and Gordon McKay. 2022. "Bio-Oil Production from Multi-Waste Biomass Co-Pyrolysis Using Analytical Py–GC/MS" Energies 15, no. 19: 7409. https://doi.org/10.3390/en15197409
APA StyleMariyam, S., Alherbawi, M., Rashid, N., Al-Ansari, T., & McKay, G. (2022). Bio-Oil Production from Multi-Waste Biomass Co-Pyrolysis Using Analytical Py–GC/MS. Energies, 15(19), 7409. https://doi.org/10.3390/en15197409









