Provision of Demand-Side Flexibility through the Integration of Power-to-Gas Technologies in an Electric Steel Mill
Abstract
:1. Introduction
1.1. EAF Steel Production Process and Oxygen Demand
1.2. Technology Overview
1.2.1. Demand-Side Management in EAF Steel Mills
1.2.2. Oxygen Production
1.2.3. Power to Gas
Electrolysis
Methanation
1.3. Research Need and Outline of the Article
2. Materials and Methods
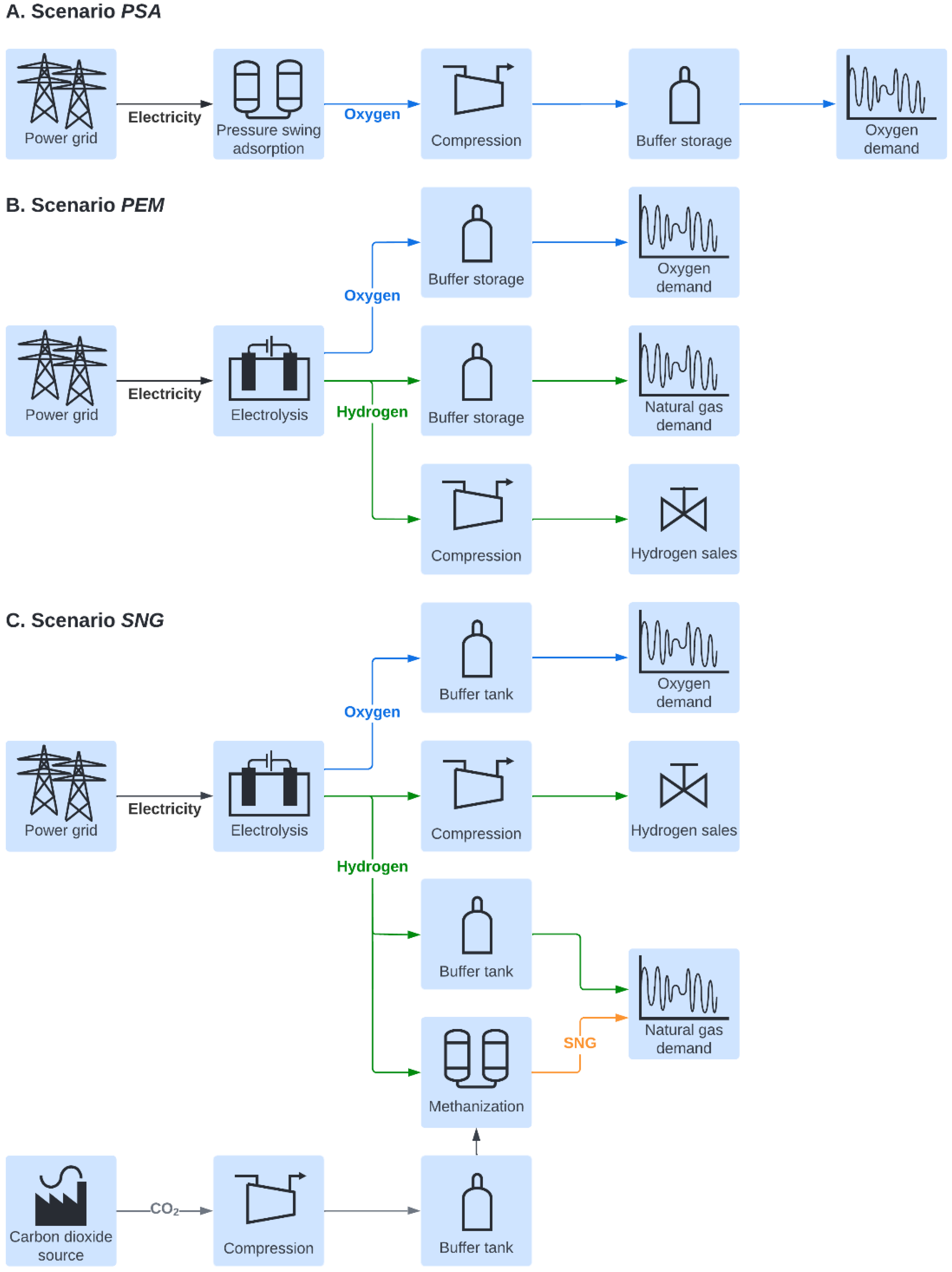
2.1. Considered Scenarios
- The scenario PSA involves the on-site production of oxygen by a pressure swing adsorption unit. Electricity and natural gas are drawn from the grid at wholesale prices. The consumption of natural gas results in additional costs for the purchase of the related emission certificates. The implementation of the PSA into the steel mill enables the omission of oxygen purchase; however, there are additional capital costs for the installation and costs for covering the electricity demand of the oxygen production plant. The resulting levelized cost of oxygen (LCOO) serves as a benchmark for evaluating the alternative system layouts.
- In scenario PEM, the required oxygen for the steel production process is generated as a by-product in a PEM electrolysis plant. Thus, the purchase of oxygen is obsolete in this case as well. Revenues from the sales of hydrogen aim to offset the increased expenses for investment and energy. If economically viable, a part of natural gas purchased from the grid is substituted by the produced hydrogen. Thus, we assume that the installed burners allow for the combustion of mixtures of natural gas and hydrogen up to 100% H2. In this case, the internal hydrogen utilization saves energy costs and direct carbon dioxide emissions.
- In scenario SNG, a share of the hydrogen from the electrolysis unit is withdrawn in order to methanize the CO2 produced from a mill-internal carbon capture (CCU) plant. This not only ensures sufficient oxygen supply but also reduces the amount of natural gas obtained from the grid and creates a sink for carbon dioxide. However, the limited availability of captured CO2 results in a surplus of hydrogen. In this scenario, we restrict the admixture of hydrogen into the natural gas infrastructure in the EAF steel mill to 20%vol of the current natural gas load. Analogous to the scenario PEM, excess hydrogen is injected into the gas grid and, therefore, represents a source of revenue.
2.2. Optimization Model
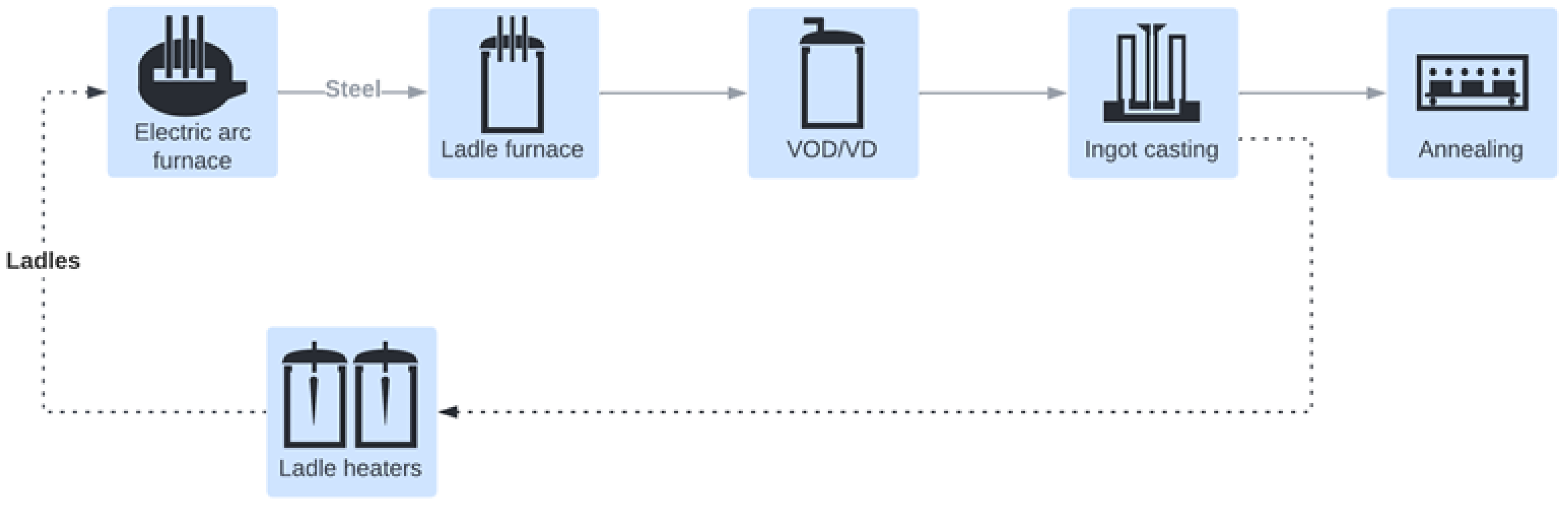
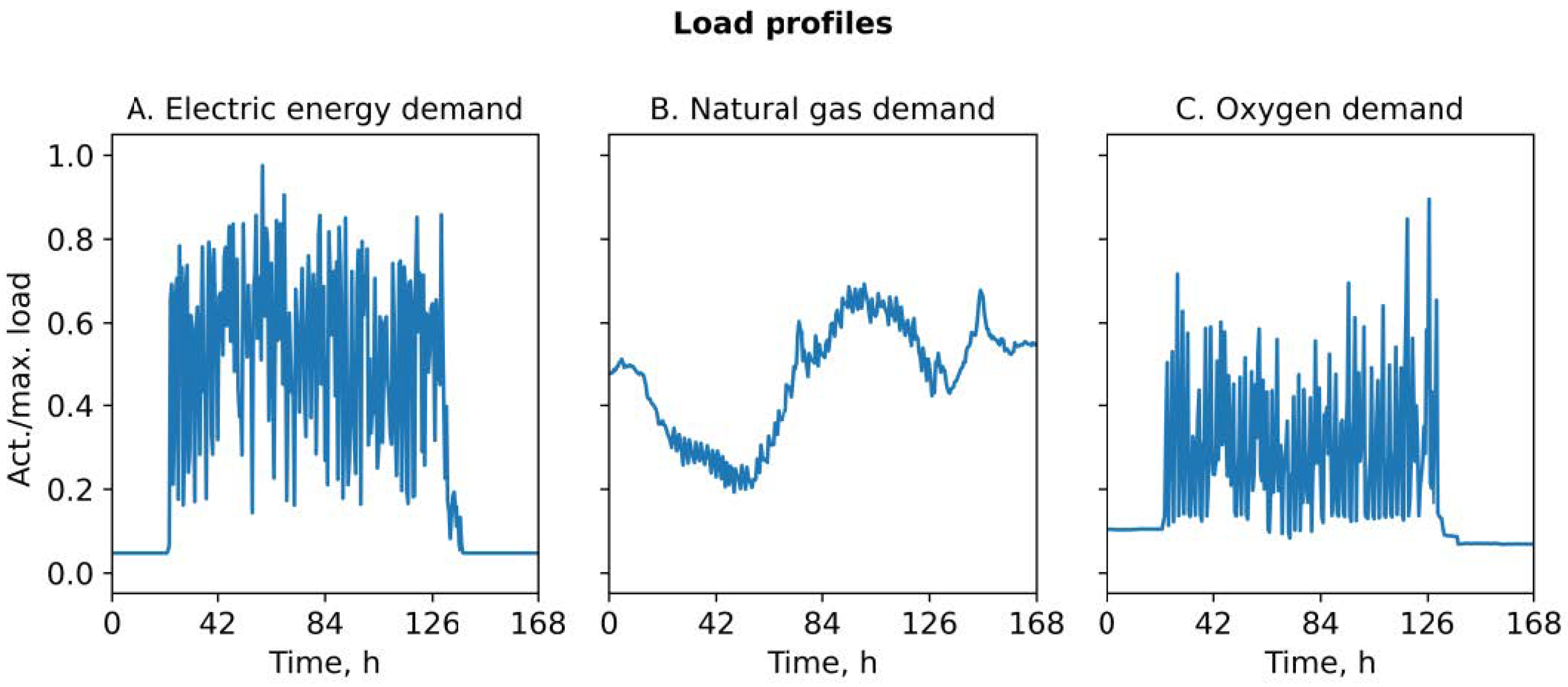
2.3. Load Profiles
- Ladle heaters;
- Annealing furnaces;
- Other consumers.
- Electric arc furnace;
- Vacuum oxygen decarburization units;
- Ladle heaters;
- Other consumers.
2.4. Cost Model
2.4.1. Equipment Cost
| Cost Component | Unit | 2020 | 2030 | 2050 | Reference |
|---|---|---|---|---|---|
| VPSA unit | |||||
| CAPEX | EUR/kWel | 3000 | 3000 | 3000 | calculated from [21] |
| OPEX | %CAPEX | 2 | 2 | 2 | own assumption |
| Lifetime | a | 10 | 10 | 10 | [21] |
| PEM electrolysis unit | |||||
| CAPEX | EUR/kWel | 1000 | 750 | 500 | [39,46] |
| OPEX | %CAPEX | 2.75 | 2.75 | 2.75 | [35] |
| Lifetime | a | 20 | 20 | 20 | [35] |
| Methanation unit | |||||
| CAPEX | EUR/kWSNG | 450 | 450 | 450 | [35] |
| OPEX | %CAPEX | 3 | 3 | 3 | [35] |
| Lifetime | a | 20 | 20 | 20 | [35] |
| Buffer storage | |||||
| CAPEX | EUR/m3 | 50 | 50 | 50 | [35,45] |
| OPEX | %CAPEX | 1 | 1 | 1 | [35] |
| Lifetime | a | 20 | 20 | 20 | [35] |
| O2 compressor | |||||
| CAPEX | EUR/kWel | 1100 | 1100 | 1100 | calculated from [47] |
| Lifetime | a | 10 | 10 | 10 | own assumption |
| H2 compressor | |||||
| CAPEX | EUR/kWel | 700 | 700 | 700 | calculated from [47] |
| Lifetime | a | 10 | 10 | 10 | own assumption |
| CO2 compressor | |||||
| CAPEX | EUR/kWel | 1480 | 1480 | 1480 | calculated from [47] |
| Lifetime | a | 10 | 10 | 10 | own assumption |
2.4.2. Energy Costs
| Cost Component | Unit | 2020 | 2030 | 2050 | Reference |
|---|---|---|---|---|---|
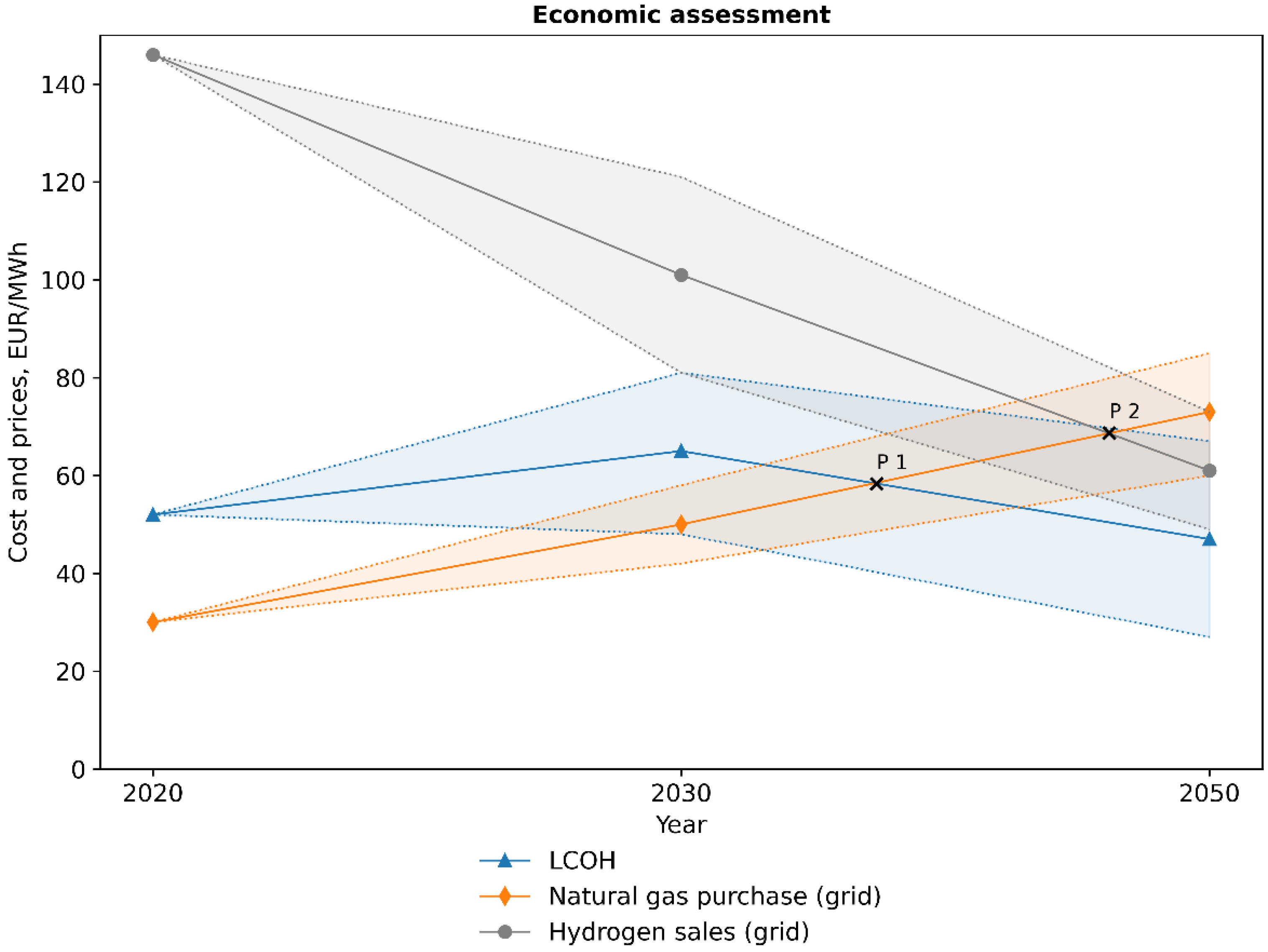
| electric energy (mean) | EUR/MWh | 33 | 62 | 82 | [43,44,48] |
| natural gas | EUR/MWhHHV | 13 | 12 | 11 | [50] |
| hydrogen | EUR/MWhHHV | 146 | 101 | 61 | [53] |
| emission allowances | EUR/t | 24 | 130 | 250 | [50,51,52] |
3. Results and Discussion
3.1. General Results
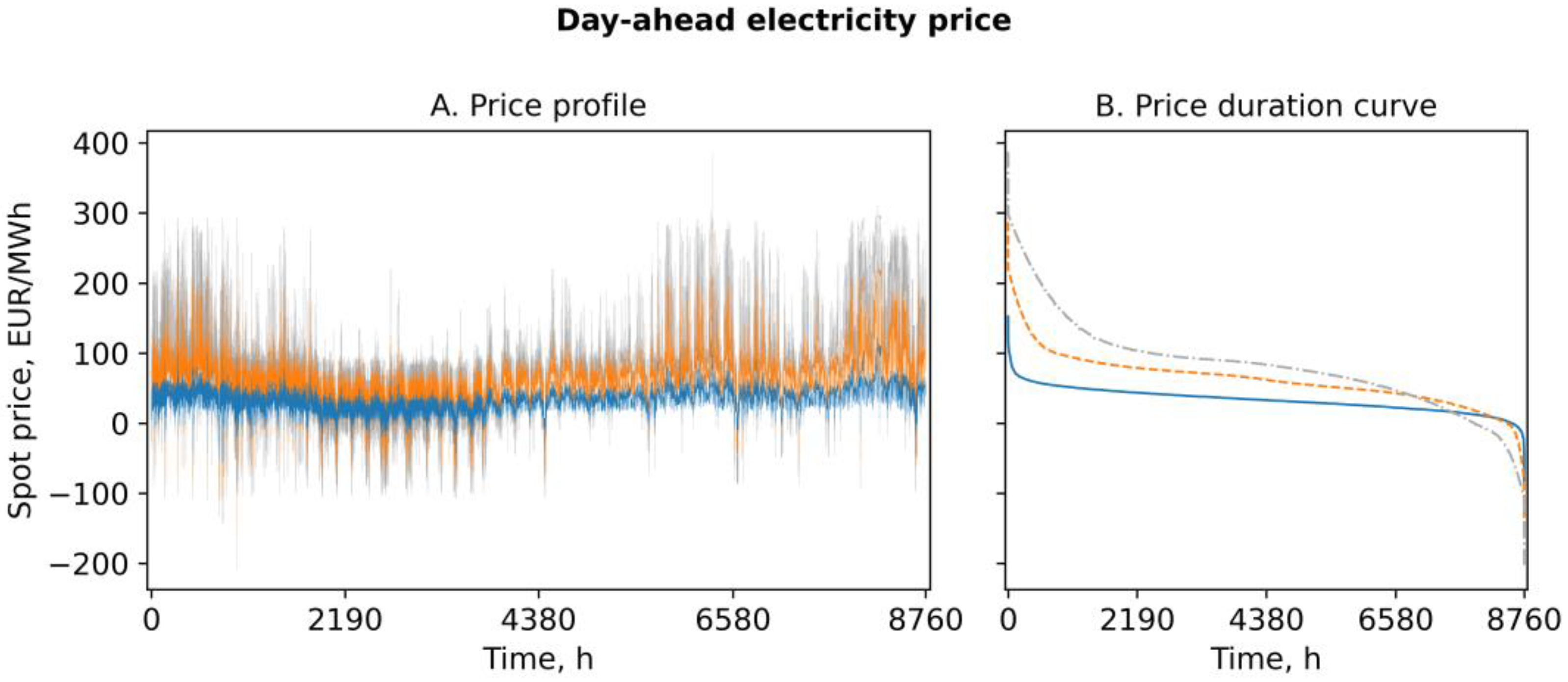
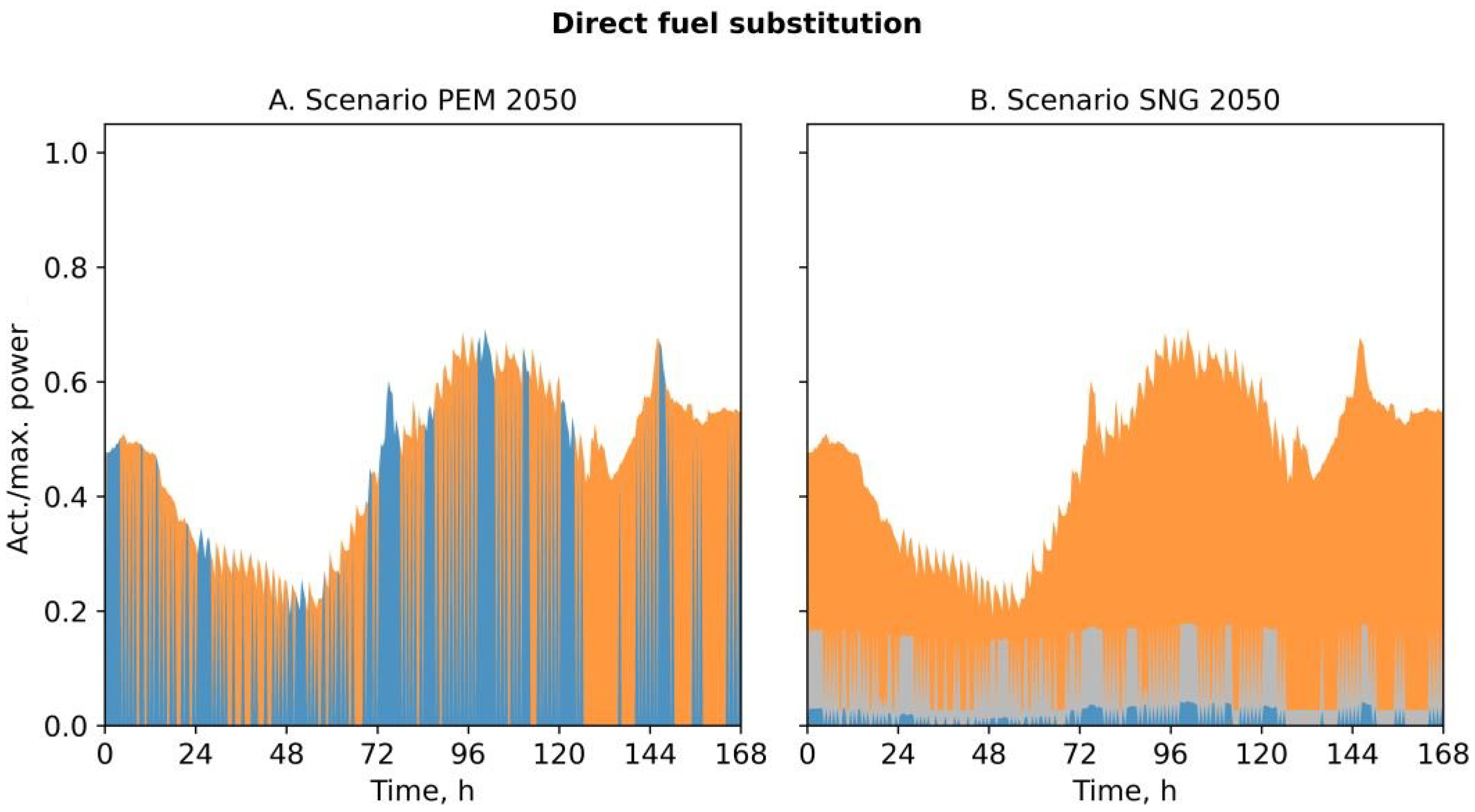
3.2. Demand-Side Management Potential
3.3. Carbon Dioxide Emissions
3.4. Economic Assessment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Material Economics. Industrial Transformation 2050—Pathways to Net-Zero Emissions from EU Heavy Industry. 2019. Available online: https://materialeconomics.com/publications/industrial-transformation-2050 (accessed on 21 October 2021).
- International Energy Agency. Iron and Steel Technology Roadmap, Paris. 2020. Available online: https://www.iea.org/reports/iron-and-steel-technology-roadmap (accessed on 22 October 2021).
- The Boston Consulting Group (BCG), Steel Institute VDEh. Steel’s Contribution to a Low-Carbon Europe 2050: Technical and Economic Analysis of the Sector’s CO2 Abatement Potential. 2013. Available online: https://www.bcg.com/publications/2013/metals-mining-environment-steels-contribution-low-carbon-europe-2050 (accessed on 13 January 2022).
- ICF Consulting Services Limited, Fraunhofer Institute for Systems and Innovation Research. Industrial Innovation: Pathways to Deep Decarbonisation of Industry: Part 2: Scenario Analysis and Pathways to Deep Decarbonisation. 2019. Available online: https://www.isi.fraunhofer.de/de/competence-center/energietechnologien-energiesysteme/projekte/pathways.html#3 (accessed on 28 October 2021).
- World Resources Institute, World Business Council for Sustainable Development. The Greenhouse Gas Protocol. Available online: https://ghgprotocol.org/standards (accessed on 19 November 2021).
- Dock, J.; Kienberger, T. Techno-economic case study on Oxyfuel technology implementation in EAF steel mills—Concepts for waste heat recovery and carbon dioxide utilization. Clean. Eng. Technol. 2022, 9, 100525. [Google Scholar] [CrossRef]
- Bussar, C.; Stöcker, P.; Cai, Z.; Moraes Jr., L.; Magnor, D.; Wiernes, P.; van Bracht, N.; Moser, A.; Sauer, D.U. Large-scale integration of renewable energies and impact on storage demand in a European renewable power system of 2050—Sensitivity study. J. Energy Storage 2016, 6, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Greiml, M.; Fritz, F.; Kienberger, T. Increasing installable photovoltaic power by implementing power-to-gas as electricity grid relief—A techno-economic assessment. Energy 2021, 235, 121307. [Google Scholar] [CrossRef]
- Dock, J.; Janz, D.; Weiss, J.; Marschnig, A.; Kienberger, T. Time- and component-resolved energy system model of an electric steel mill. Clean. Eng. Technol. 2021, 4, 100223. [Google Scholar] [CrossRef]
- Madias, J. Electric furnace steelmaking. In Treatise on Process Metallurgy; Elsevier: Kidlington, Oxford, UK; Waltham, MA, USA, 2014; pp. 271–300. ISBN 978-0-08-096988-6. [Google Scholar]
- Holappa, L. Secondary steelmaking. In Treatise on Process Metallurgy; Elsevier: Kidlington, Oxford, UK; Waltham, MA, USA, 2014; pp. 301–345. ISBN 978-0-08-096988-6. [Google Scholar]
- Warren, P. A review of demand-side management policy in the UK. Renew. Sustain. Energy Rev. 2014, 29, 941–951. [Google Scholar] [CrossRef]
- Paulus, M.; Borggrefe, F. The potential of demand-side management in energy-intensive industries for electricity markets in Germany. Appl. Energy 2011, 88, 432–441. [Google Scholar] [CrossRef]
- Strbac, G. Demand side management: Benefits and challenges. Energy Policy 2008, 36, 4419–4426. [Google Scholar] [CrossRef]
- Marchiori, F.; Belloni, A.; Benini, M.; Cateni, S.; Colla, V.; Ebel, A.; Lupinelli, M.; Nastasi, G.; Neuer, M.; Pietrosanti, C.; et al. Integrated dynamic energy management for steel production. Energy Procedia 2017, 105, 2772–2777. [Google Scholar] [CrossRef]
- Zhang, Q.; Grossmann, I.E. Enterprise-wide optimization for industrial demand side management: Fundamentals, advances, and perspectives. Chem. Eng. Res. Des. 2016, 116, 114–131. [Google Scholar] [CrossRef] [Green Version]
- Gorre, J.; Ortloff, F.; van Leeuwen, C. Production costs for synthetic methane in 2030 and 2050 of an optimized Power-to-Gas plant with intermediate hydrogen storage. Appl. Energy 2019, 253, 113594. [Google Scholar] [CrossRef]
- Kato, T.; Kubota, M.; Kobayashi, N.; Suzuoki, Y. Effective utilization of by-product oxygen from electrolysis hydrogen production. Energy 2005, 30, 2580–2595. [Google Scholar] [CrossRef]
- McGuinness, R.M.; Kleinberg, W.T. Oxygen production. In Oxygen-Enhanced Combustion, 2nd ed.; Baukal, C.E., Ed.; CRC Press: Boca Raton, FL, USA, 2013; pp. 44–74. ISBN 978-1-4398-6230-8. [Google Scholar]
- Kerry, F.G. Noncryogenic oxygen production. In Oxygen-Enhanced Combustion, 2nd ed.; Baukal, C.E., Ed.; CRC Press: Boca Raton, FL, USA, 2013; pp. 77–87. ISBN 978-1-4398-6230-8. [Google Scholar]
- Šulc, R.; Ditl, P. A technical and economic evaluation of two different oxygen sources for a small oxy-combustion unit. J. Clean. Prod. 2021, 309, 127427. [Google Scholar] [CrossRef]
- Air Liquide Engineering and Construction. Standard Plants: Fully Packaged Modular Solutions. Available online: https://www.engineering-airliquide.com/standard-plants (accessed on 7 January 2022).
- PCI Oxygen Solutions. On-Site Oxygen Solutions: Industrial. Available online: https://www.pcigases.com/oxygen-solutions/marketing-literature/ (accessed on 7 January 2022).
- Adsorptech. EcoGen™ Oxygen VPSA Onsite Generator. Available online: https://adsorptech.com/wp-content/uploads/2021Adsorptech-EcoGen-2111-2.pdf (accessed on 7 January 2022).
- Banaszkiewicz, T.; Chorowski, M.; Gizicki, W. Comparative analysis of oxygen production for oxy-combustion application. Energy Procedia 2014, 51, 127–134. [Google Scholar] [CrossRef] [Green Version]
- Bailera, M.; Kezibri, N.; Romeo, L.M.; Espatolero, S.; Lisbona, P.; Bouallou, C. Future applications of hydrogen production and CO2 utilization for energy storage: Hybrid power to gas-oxycombustion power plants. Int. J. Hydrogen Energy 2017, 42, 13625–13632. [Google Scholar] [CrossRef] [Green Version]
- Schiebahn, S.; Grube, T.; Robinius, M.; Tietze, V.; Kumar, B.; Stolten, D. Power to gas: Technological overview, systems analysis and economic assessment for a case study in Germany. Int. J. Hydrogen Energy 2015, 40, 4285–4294. [Google Scholar] [CrossRef]
- Mergel, J.; Carmo, M.; Fritz, D. Status on technologies for hydrogen production by water electrolysis. In Transition to Renewable Energy Systems; Wiley-VCH: Weinheim, Germany, 2013; pp. 425–450. ISBN 978-3-527-33239-7. [Google Scholar]
- Schmidt, O.; Gambhir, A.; Staffell, I.; Hawkes, A.; Nelson, J.; Few, S. Future cost and performance of water electrolysis: An expert elicitation study. Int. J. Hydrogen Energy 2017, 42, 30470–30492. [Google Scholar] [CrossRef]
- Iora, P.; Chiesa, P. High efficiency process for the production of pure oxygen based on solid oxide fuel cell–solid oxide electrolyzer technology. J. Power Sources 2009, 190, 408–416. [Google Scholar] [CrossRef]
- Iora, P.; Taher, M.A.A.; Chiesa, P.; Brandon, N.P. A one dimensional solid oxide electrolyzer-fuel cell stack model and its application to the analysis of a high efficiency system for oxygen production. Chem. Eng. Sci. 2012, 80, 293–305. [Google Scholar] [CrossRef]
- Ghaib, K.; Nitz, K.; Ben-Fares, F.-Z. Chemical methanation of CO2: A review. ChemBioEng Rev. 2016, 3, 266–275. [Google Scholar] [CrossRef]
- Herrmann, F.; Grünewald, M.; Meijer, T.; Gardemann, U.; Feierabend, L.; Riese, J. Operating window and flexibility of a lab-scale methanation plant. Chem. Eng. Sci. 2022, 254, 117632. [Google Scholar] [CrossRef]
- Chwoła, T.; Spietz, T.; Więcław-Solny, L.; Tatarczuk, A.; Krótki, A.; Dobras, S.; Wilk, A.; Tchórz, J.; Stec, M.; Zdeb, J. Pilot plant initial results for the methanation process using CO2 from amine scrubbing at the Łaziska power plant in Poland. Fuel 2020, 263, 116804. [Google Scholar] [CrossRef]
- Gorre, J.; Ruoss, F.; Karjunen, H.; Schaffert, J.; Tynjälä, T. Cost benefits of optimizing hydrogen storage and methanation capacities for Power-to-Gas plants in dynamic operation. Appl. Energy 2020, 257, 113967. [Google Scholar] [CrossRef]
- Van Rossum, G. The Python Library Reference. Release 3.7.8rc1. 2020. Available online: https://docs.python.org/3.7/library/index.html (accessed on 24 June 2020).
- Hilpert, S.; Kaldemeyer, C.; Krien, U.; Günther, S.; Wingenbach, C.; Plessmann, G. The open energy modelling framework (oemof)—A new approach to facilitate open science in energy system modelling. Energy Strategy Rev. 2018, 22, 16–25. [Google Scholar] [CrossRef] [Green Version]
- Krien, U.; Schönfeldt, P.; Launer, J.; Hilpert, S.; Kaldemeyer, C.; Pleßmann, G. Oemof.solph—A model generator for linear and mixed-integer linear optimisation of energy systems. Softw. Impacts 2020, 6, 100028. [Google Scholar] [CrossRef]
- Buttler, A.; Spliethoff, H. Current status of water electrolysis for energy storage, grid balancing and sector coupling via power-to-gas and power-to-liquids: A review. Renew. Sustain. Energy Rev. 2018, 82, 2440–2454. [Google Scholar] [CrossRef]
- Carmo, M.; Fritz, D.L.; Mergel, J.; Stolten, D. A comprehensive review on PEM water electrolysis. Int. J. Hydrogen Energy 2013, 38, 4901–4934. [Google Scholar] [CrossRef]
- International Energy Agency. The Future of Hydrogen: Seizing Today’s Opportunities, Paris. 2019. Available online: https://www.iea.org/reports/the-future-of-hydrogen (accessed on 3 January 2022).
- Mutz, B.; Carvalho, H.W.P.; Mangold, S.; Kleist, W.; Grunwaldt, J.-D. Methanation of CO2: Structural response of a Ni-based catalyst under fluctuating reaction conditions unraveled by operando spectroscopy. J. Catal. 2015, 327, 48–53. [Google Scholar] [CrossRef]
- Energy Brainpool. Trends in the Development of Electricity Prices—EU Energy Outlook 2050. Available online: https://blog.energybrainpool.com/en/trends-in-the-development-of-electricity-prices-eu-energy-outlook-2050/ (accessed on 16 February 2022).
- Energy Brainpool. Update: EU Energy Outlook 2050—How will Europe Evolve over the Next 30 Years? Available online: https://blog.energybrainpool.com/en/update-eu-energy-outlook-2050-how-will-europe-evolve-over-the-next-30-years/ (accessed on 16 February 2022).
- van Leeuwen, C.; Mulder, M. Power-to-gas in electricity markets dominated by renewables. Appl. Energy 2018, 232, 258–272. [Google Scholar] [CrossRef]
- E4tech, E.E. Development of Water Electrolysis in the European Union: Final Report. 2014. Available online: https://www.fch.europa.eu/node/783 (accessed on 20 December 2021).
- Peters, M.S.; Timmerhaus, K.D.; West, R.E. Plant Design and Economics for Chemical Engineers, 5th ed.; McGraw-Hill: Boston, MA, USA, 2004; ISBN 0-07-239266-5. [Google Scholar]
- Austrian Power Grid AG. EXAA Day-Ahead Prices 2020. Available online: https://www.apg.at/de/markt/Markttransparenz/Uebertragung/EXAA-Spotmarkt (accessed on 21 December 2021).
- Traupmann, A.; Greiml, M.; Steinegger, J.; Kühberger, L.; Kienberger, T. Sector coupling technologies as re-purposing options for coal-fired power plants in the austrian grids—A MES approach. Sustain. Energy Technol. Assess. 2022; under review. [Google Scholar]
- International Energy Agency. World Energy Outlook 2021, Paris. 2021. Available online: https://www.iea.org/reports/world-energy-outlook-2021 (accessed on 4 January 2022).
- European Energy Exchange AG. Emission Spot Primary Market Auction Report 2020. Available online: https://www.eex.com/en/market-data/environmental-markets/eua-primary-auction-spot-download (accessed on 21 December 2021).
- Pietzcker, R.C.; Osorio, S.; Rodrigues, R. Tightening EU ETS targets in line with the European Green Deal: Impacts on the decarbonization of the EU power sector. Appl. Energy 2021, 293, 116914. [Google Scholar] [CrossRef]
- Cvetkovska, R.; Nagovnak, P.; Kienberger, T. Pathways for ramping-up hydrogen into the natural gas system. In Proceedings of the 17th Symposium Eneergieinnovation, Future of Energy: Innovationen für Eine Klimaneutrale Zukunft, Graz, Austria, 16–18 February 2022. [Google Scholar]
- Gas-Systemnutzungsentgelte-Verordnung. Available online: https://www.ris.bka.gv.at/GeltendeFassung.wxe?Abfrage=Bundesnormen&Gesetzesnummer=20007992&FassungVom=2022-06-02 (accessed on 28 June 2022).
- E-Control. Natural Gas: Taxes and Surcharges. Available online: https://www.e-control.at/en/industrie/gas/gaspreis/steuern-und-abgaben (accessed on 28 June 2022).
- Systemnutzungsentgelte-Verordnung 2018. Available online: https://www.ris.bka.gv.at/GeltendeFassung.wxe?Abfrage=Bundesnormen&Gesetzesnummer=20010107&FassungVom=2022-01-01 (accessed on 28 June 2022).
- E-Control. Electricity: Taxes and Surcharges. Available online: https://www.e-control.at/en/industrie/strom/strompreis/steuern (accessed on 28 June 2022).
- Electricity Industry and Organization Act: ElWOG. 2010. Available online: https://www.ris.bka.gv.at/GeltendeFassung.wxe?Abfrage=Bundesnormen&Gesetzesnummer=20007045 (accessed on 5 April 2022).
- European Commission. A Clean Planet for All—A European Long-Term Strategic Vision for a Prosperous, Modern, Competitive and Climate Neutral Economy: In-Depth Analysis in Support of the Commission Communication COM (2018) 773, 28th 2018. Available online: https://ec.europa.eu/clima/eu-action/climate-strategies-targets/2050-long-term-strategy_en (accessed on 30 June 2022).
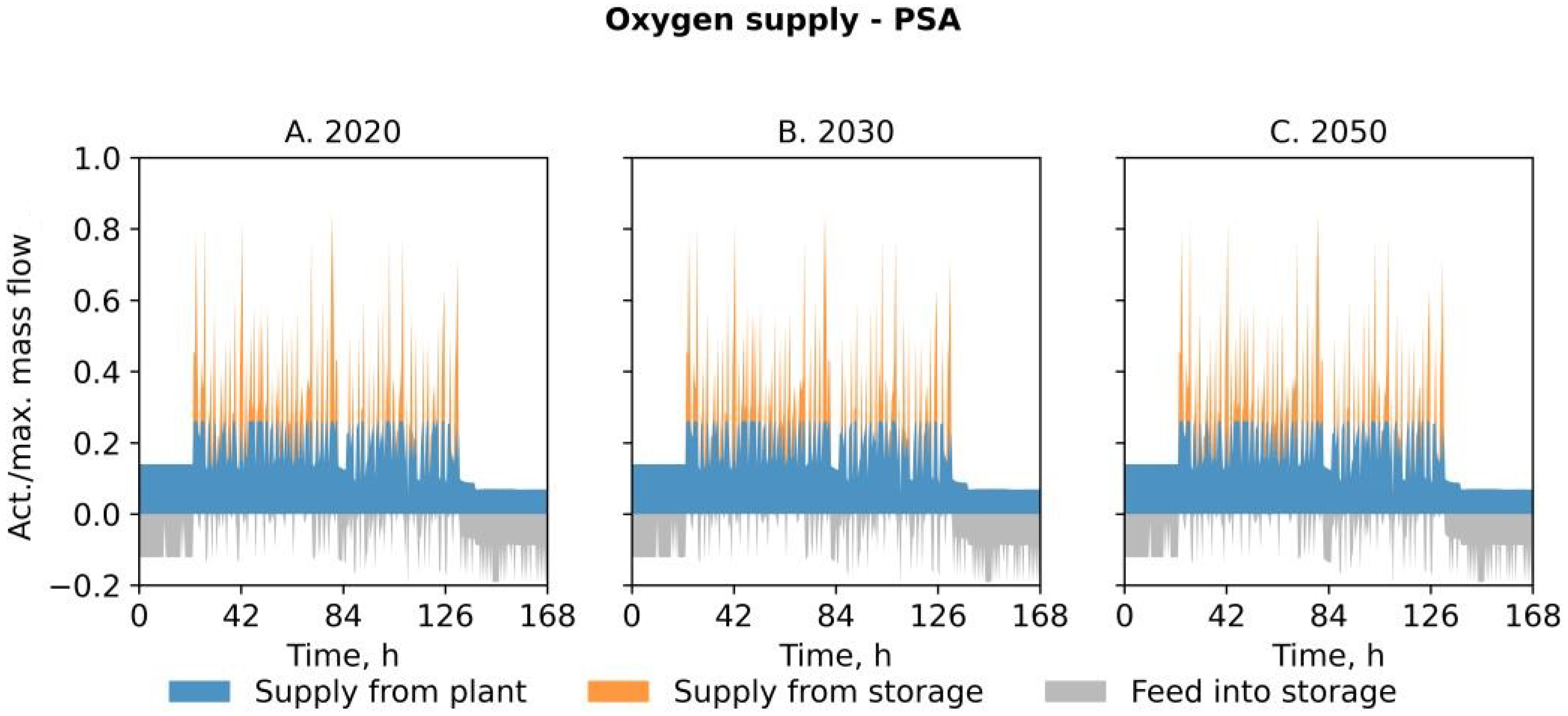
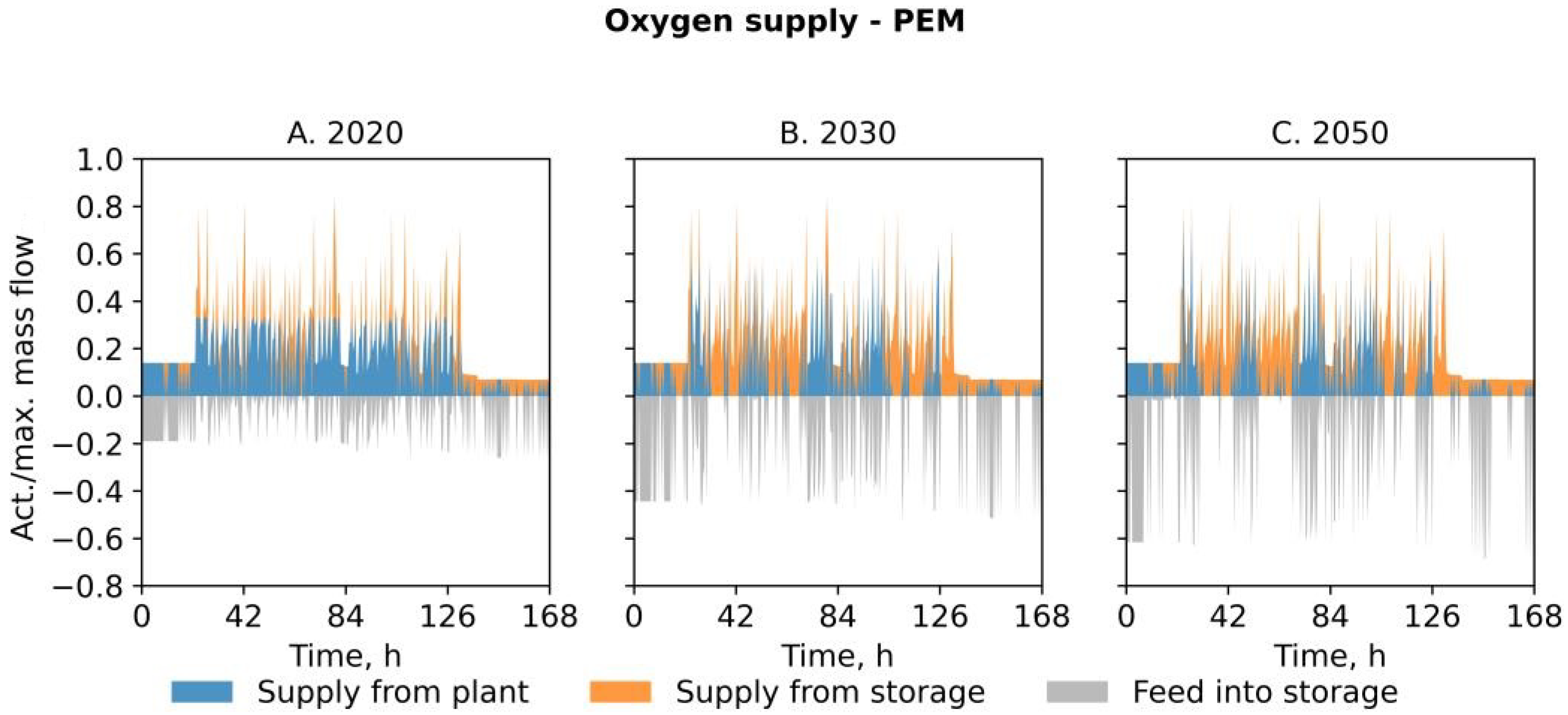
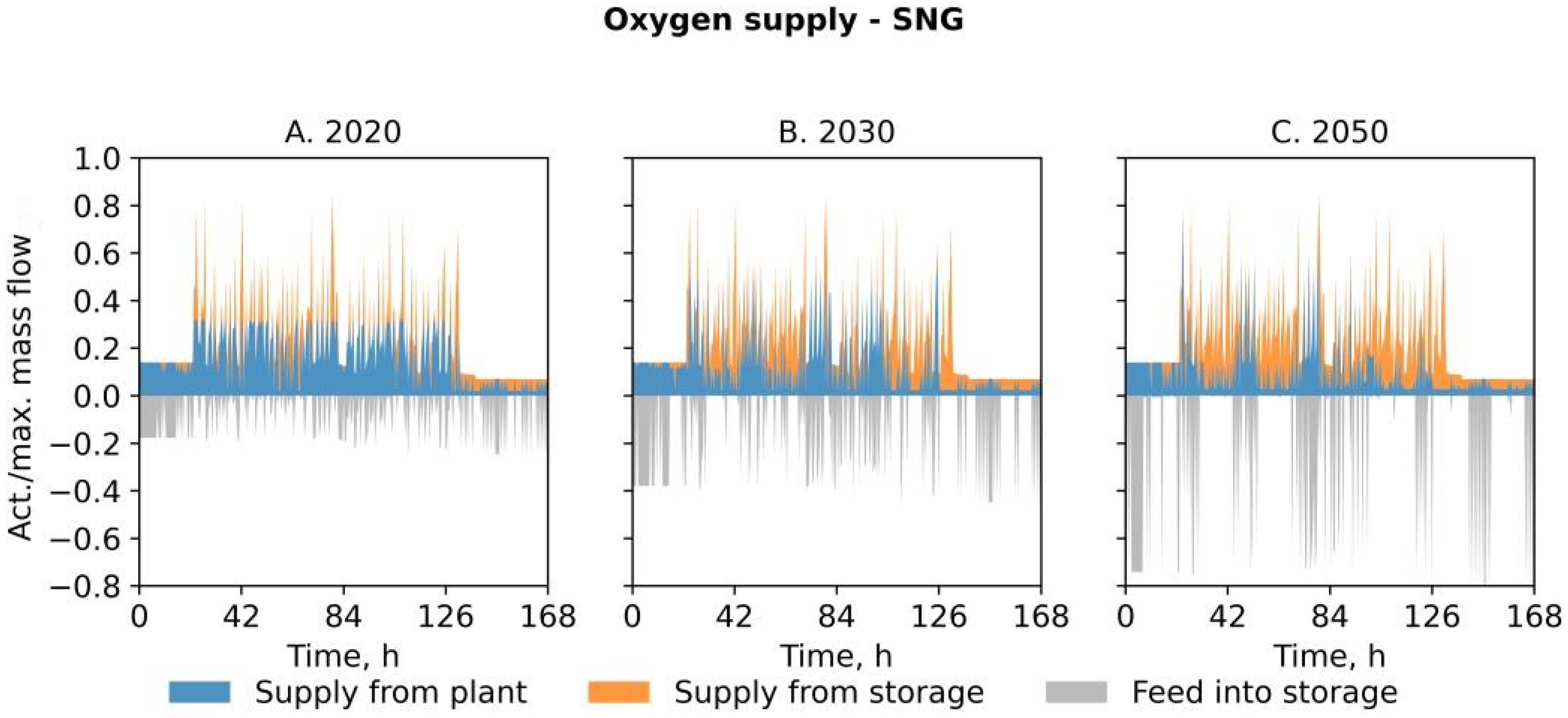
| Feature | PSA | PEM | SNG |
|---|---|---|---|
| Oxygen supply | VPSA | PEM electrolysis | PEM electrolysis |
| Oxygen purchase | no | No | no |
| Hydrogen sales | no | Yes | yes |
| Methanation | no | No | yes |
| Max. Hydrogen admixture 1 | - | 100% | 20% |
| Parameter | Unit | 2020 | 2030 | 2050 | Reference |
|---|---|---|---|---|---|
| VPSA unit | |||||
| Specific energy demand | kWhel/kg | 0.35 | 0.35 | 0.35 | [22,23,24] |
| Load range | % | 60–100 | 60–100 | 60–100 | own assumption |
| Load gradient | %/min | 4 | 4 | 4 | own assumption |
| Pressure | Mpa | 3 | 3 | 3 | [28] |
| PEM electrolysis unit | |||||
| Efficiency | kWhH2/kWhel 1 | 0.68 | 0.75 | 0.83 | [39,40,41] |
| Specific energy demand H2 | kWhel/kg | 58.0 | 52.6 | 47.5 | own calculation |
| Specific energy demand O2 | kWhel/kg | 7.3 | 6.6 | 6.0 | own calculation |
| Load range | % | 0–100 | 0–100 | 0–100 | [35] |
| Methanation unit | |||||
| Efficiency | kWhSNG/kWhH2 1 | 0.62 | 0.62 | 0.62 | own calculation, [42] |
| Load range | % | 20–100 | 20–100 | 20–100 | [33] |
| Load gradient | %/min | 3 | 3 | 3 | [35] |
| Buffer storage | |||||
| Minimum pressure | MPa | 1 | 1 | 1 | own assumption, [35] |
| Maximum pressure | Mpa | 3 | 3 | 3 | own assumption, [35] |
| O2 compressor | |||||
| Mean pressure | Mpa | 2 | 2 | 2 | own assumption |
| Specific energy demand | kWhel/kgO2 | 0.14 | 0.14 | 0.14 | own calculation |
| H2 compressor | |||||
| Mean pressure | Mpa | 35 | 35 | 35 | own assumption |
| Specific energy demand | kWhel/kgH2 | 2.14 | 2.14 | 2.14 | own calculation |
| CO2 compressor | |||||
| Mean pressure | Mpa | 2 | 2 | 2 | own assumption |
| Specific energy demand | kWhel/kgCO2 | 0.09 | 0.09 | 0.09 | own calculation |
| KPI | Unit | 2020 | 2030 | 2050 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Scenario | - | PSA | PEM | SNG | PSA | PEM | SNG | PSA | PEM | SNG |
| Nominal power | kWel | 400 | 10,600 | 10,185 | 400 | 16,900 | 15,100 | 400 | 20,000 | 23,300 |
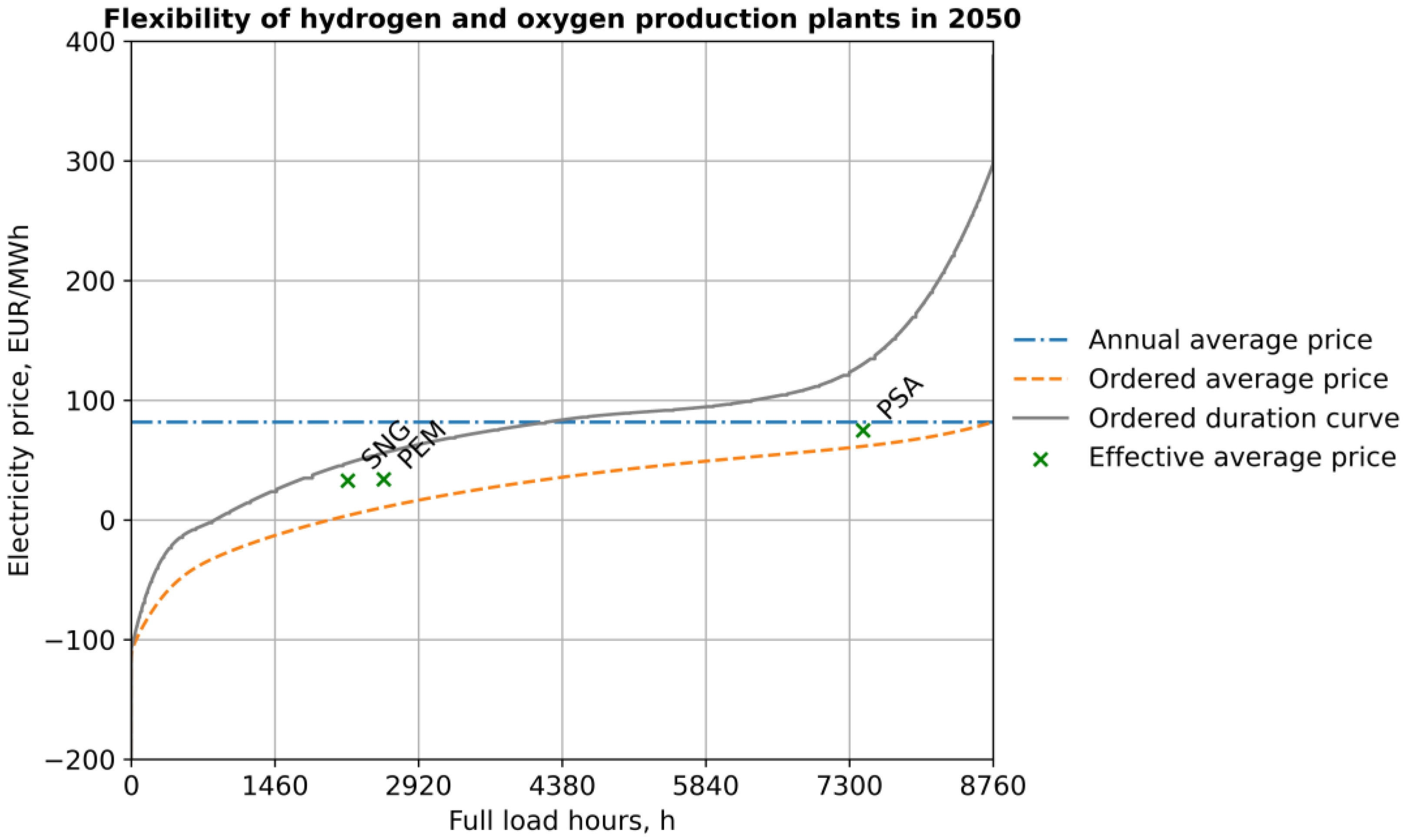
| Full-load hours | hrs | 7439 | 5876 | 6119 | 7439 | 3325 | 3738 | 7439 | 2567 | 2202 |
| H2 production | MWhHHV/a | - | 42,400 | 42,400 | - | 42,400 | 42,400 | - | 42,400 | 42,400 |
| SNG production | MWhHHV/a | - | - | 8600 | - | - | 8600 | - | - | 8600 |
| CAPEX share | % | 45 | 30 | 30 | 37 | 31 | 28 | 34 | 32 | 36 |
| Fuel substitution | %HHV | - | 0 | 15 | - | 0 | 15 | - | 45 | 19 |
| Electricity Prices | Scenario | Unit | 2020 | 2030 | 2050 |
|---|---|---|---|---|---|
| Annual average | - | EUR/MWh | 33 | 62 | 82 |
| Ordered average | PSA | EUR/MWh | 28 | 51 | 62 |
| PEM | EUR/MWh | 24 | 29 | 11 | |
| SNG | EUR/MWh | 25 | 32 | 4 | |
| Effective average | PSA | EUR/MWh | 49 | 76 | 94 |
| PEM | EUR/MWh | 28 | 38 | 34 | |
| SNG | EUR/MWh | 28 | 41 | 33 |
| KPI | Unit | 2020 | 2030 | 2050 | |||
|---|---|---|---|---|---|---|---|
| Scenario | - | PEM | SNG | PEM | SNG | PEM | SNG |
| Internal | tCO2 | 7100 | 4800 | 7100 | 4800 | 2500 | 4500 |
| External | tCO2 | 0 | 1500 | 0 | 1500 | 4600 | 1800 |
| Total | tCO2 | 7100 | 6300 | 7100 | 6300 | 7100 | 6300 |
| KPI | Unit | 2020 | 2030 | 2050 | |||
|---|---|---|---|---|---|---|---|
| Scenario | - | PEM | SNG | PEM | SNG | PEM | SNG |
| LCOO | EUR/MWhHHV | 77 | 77 | 93 | 93 | 103 | 103 |
| LCOH | EUR/MWhHHV | 52 | 52 | 65 | 66 | 47 | 50 |
| LCOM | EUR/MWhHHV | - | 94 | - | 120 | - | 97 |
| LCOHREF | EUR/MWhHHV | 77 | 78 | 87 | 90 | 43 | 35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dock, J.; Wallner, S.; Traupmann, A.; Kienberger, T. Provision of Demand-Side Flexibility through the Integration of Power-to-Gas Technologies in an Electric Steel Mill. Energies 2022, 15, 5815. https://doi.org/10.3390/en15165815
Dock J, Wallner S, Traupmann A, Kienberger T. Provision of Demand-Side Flexibility through the Integration of Power-to-Gas Technologies in an Electric Steel Mill. Energies. 2022; 15(16):5815. https://doi.org/10.3390/en15165815
Chicago/Turabian StyleDock, Johannes, Stefan Wallner, Anna Traupmann, and Thomas Kienberger. 2022. "Provision of Demand-Side Flexibility through the Integration of Power-to-Gas Technologies in an Electric Steel Mill" Energies 15, no. 16: 5815. https://doi.org/10.3390/en15165815
APA StyleDock, J., Wallner, S., Traupmann, A., & Kienberger, T. (2022). Provision of Demand-Side Flexibility through the Integration of Power-to-Gas Technologies in an Electric Steel Mill. Energies, 15(16), 5815. https://doi.org/10.3390/en15165815






