Methods of Increasing Miscanthus Biomass Yield for Biofuel Production
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characteristics of Miscanthus and the Need to Increase Its Biomass Yield
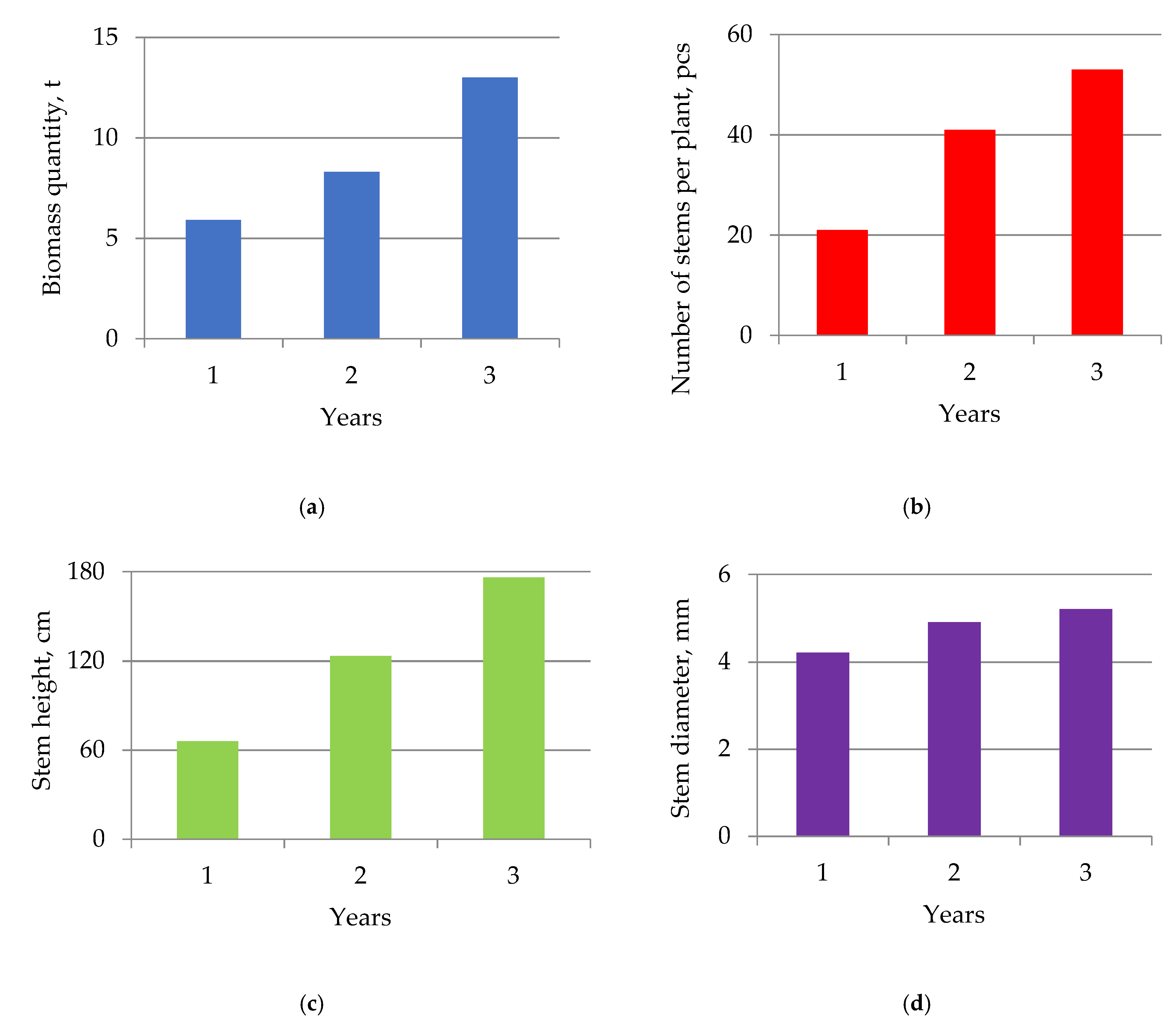
2.2. Influence of the Crop Age on the Accumulation of Miscanthus Biomass
2.3. Influence of Climatic Conditions on Miscanthus Biomass Accumulation
2.4. Influence of the Harvest Date on the Miscanthus Biomass Yield
2.5. Influence of Nitrogen Fertilization and Irrigation on the Miscanthus Biomass Yield
2.6. The Influence of Plant Density on the Miscanthus Biomass Yield
2.7. Influence of Geographic Location on Miscanthus Biomass Yield
2.8. Influence of Drought and Salinity on the Miscanthus Biomass Yield
2.9. Influence of Species and Ploidy Level of Miscanthus on Biomass Yield
2.9.1. Influence of Miscanthus Species on Biomass Yield
2.9.2. Using Miscanthus for Bioethanol Production
2.9.3. Effect of Clones on the Miscanthus Biomass Yield
2.9.4. Effect of Breeding on the Miscanthus Biomass Yield
2.10. Biogenesis and Genes Involved in Cell Wall Assembly
2.11. Genetic Strategies for Improving Lignocellulose Composition
2.12. Genetic Strategies to Increase Miscanthus Biomass Yield
2.13. Molecular Biology Techniques to Increase Miscanthus Biomass Production
2.14. Photosynthesis to Increase the Miscanthus Biomass Yield
2.15. Regulation of Miscanthus Biomass Production
2.16. The Effect of Phytohormones on the Miscanthus Biomass Yield
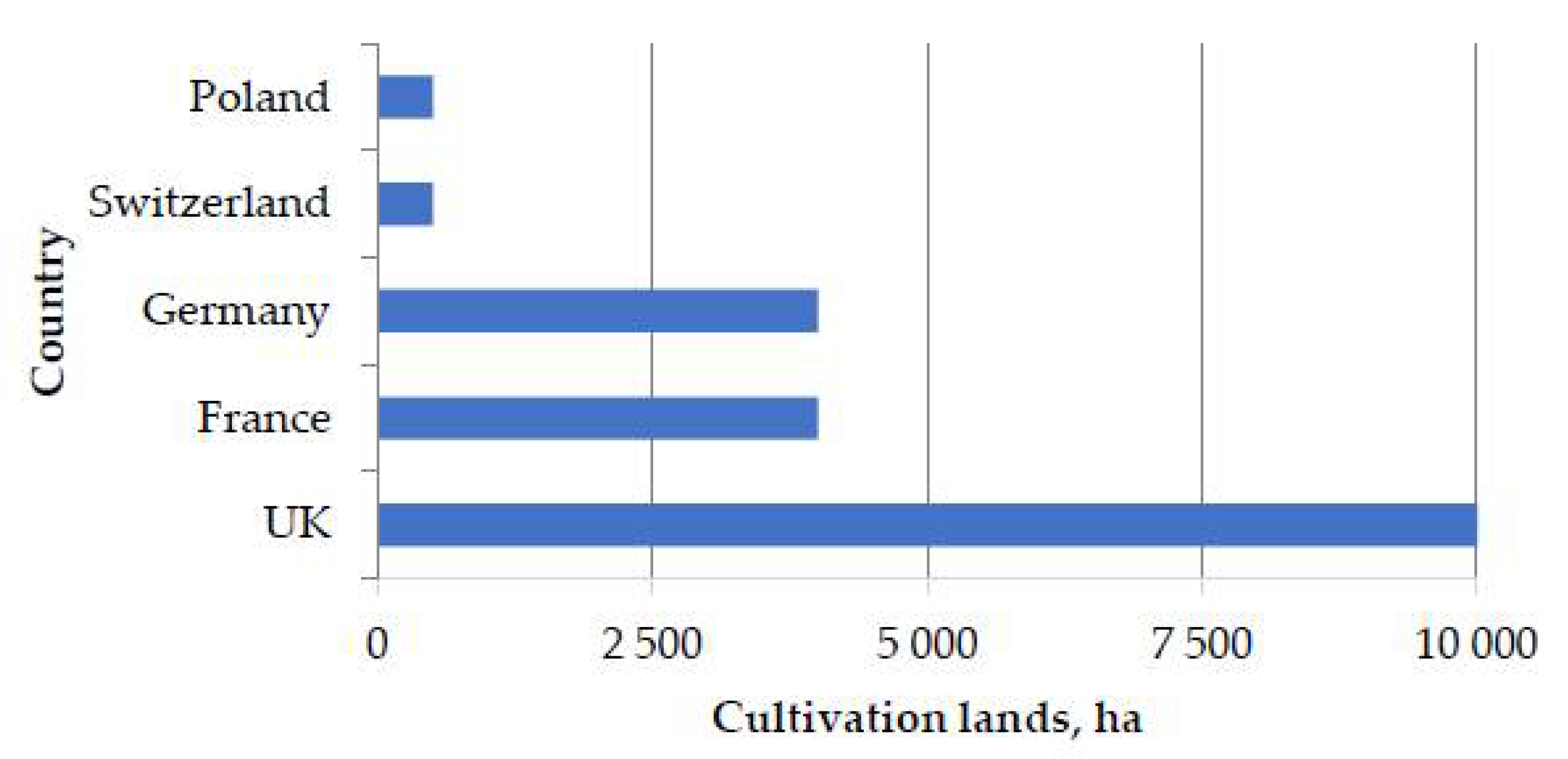
2.17. Peculiarities of Miscanthus Cultivation in Russia Compared with Other Countries
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AraT | arabinosyltransferase |
| ARFCOMT | gene that codes catechol-O-methyltransferase |
| CesA gene | cellulose synthase genes |
| COBITO | genes indirectly involved in cellulose biosynthesis through coding membrane-bound glycoprotein |
| COBRA | genes indirectly involved in cellulose biosynthesis through glycophosphatidylinositol, which encodes the membrane-bound protein GPI |
| CORRIGAN | genes indirectly involved in cellulose biosynthesis through the membrane-bound β-glucanase code |
| CRISPR | special loci of bacteria and archaea, consisting of straight repeating sequences that are separated by unique sequences (spacers) |
| CSLF | cellulose synthase-like gene regulating β-glucan synthesis |
| FT | gene that is expressed in the vascular fibrous bundles of leaves with the formation of FT protein (florigen) |
| GAX | gene polymorphism |
| GlcAT | glucosyltransferase |
| GPI | glycophosphatidylinositol (phosphoglyceride, which can bind to the C-terminus of a protein during post-translational modification) |
| GT | gene families |
| GTs | glycosyltransferases |
| IRX | gene that controls the work of several genetic programs in cells at once |
| KNOTTED1 | gene characterized by dominant neomorphic mutations that disrupt certain aspects of leaf development in maize and miscanthus |
| MYB | part of a large family of genes of transcription factors found in plants and animals |
| NAC | N-acetylcysteine |
| QTL | quantitative trait locus |
| SND1 | gene containing onco protein |
| TALEN | effector nucleases like a transcriptional activator are restriction enzymes that can be engineered to cut specific DNA sequences |
| WRKY | family of transcription factors, a class of DNA-binding proteins, mainly specific to plants and algae |
| XylT | xylosyltransferase |
References
- Gontarenko, S.N.; Lashuk, S.A. Method of propagation, stimulation of rhizomes growth in vitro culture and adaptation in the open ground for the genus Miscanthus representatives. Plant Var. Stud. Prot. 2017, 3, 230–238. [Google Scholar]
- Davey, C.L.; Jones, L.E.; Squance, M.; Purdy, S.J.; Maddison, A.L.; Cunniff, J.; Donnison, I.; Clifton-Brown, J. Radiation capture and conversion efficiencies of Miscanthus sacchariflorus, M. sinensis and their naturally occurring hybrid M. × giganteus. Glob. Chang. Biol. 2017, 9, 385–399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gushchina, V.A.; Ostroborodova, N.I. Formation of giant miscanthus biomass in the forest-steppe of the Middle Volga region. Niva Povolzhya 2019, 3, 81–87. [Google Scholar]
- Goryachkovskaya, T.N.; Starostin, K.G.; Meshcheryakova, I.A.; Peltek, S.E.; Slynko, N. Technology of miscanthus biomass saccharification using commercially available enzymes. Vavilov J. Genet. Breed. 2014, 18, 983–988. [Google Scholar]
- Lanzerstorfer, C. Combustion of Miscanthus: Composition of the Ash by Particle Size. Energies 2019, 12, 178. [Google Scholar] [CrossRef] [Green Version]
- IEA/OECD Perspectives des Technologies de l’Energie, Scénarios et Stratégies à l’horizon 2050, Synthèse et Implications Stratégiques. International Energy Agency (IEA). Available online: https://www.iea.org/reports/energy-technology-perspectives-2006 (accessed on 30 April 2021).
- Zegada-Lizarazu, W.; Parrish, D.; Berti, M.; Monti, A. Dedicated crops for advanced biofuels: Consistent and diverging agronomic points of view between the USA and the EU-27. Biofuels Bioprod. Biorefin. 2013, 7, 715–731. [Google Scholar] [CrossRef]
- Dillen, S.Y.; Djomo, S.N.; Al Afas, N.; Vanbeveren, S.; Ceulemans, R. Biomass yield and energy balance of a short-rotation poplar coppice with multiple clones on degraded land during 16 years. Biomass Bioenergy 2013, 56, 157–165. [Google Scholar] [CrossRef] [Green Version]
- Cadoux, S.; Ferchaud, F.; Demay, C.; Boizard, H.; Machet, J.-M.; Fourdinier, E.; Preudhomme, M.; Chabbert, B.; Gosse, G.; Mary, B. Implications of productivity and nutrient requirements on greenhouse gas balance of annual and perennial bioenergy crops. GCB Bioenergy 2014, 6, 425–438. [Google Scholar] [CrossRef]
- Virones, J.; Vo, L.; Arnoult, S.; Brancourt-Hulmel, M.; Navard, P. Miscanthus stem fragment—Reinforced polypropylene composites:development of an optimized preparation procedure at small scaleand its validation for differentiating genotypes. Polym. Test 2016, 55, 166–172. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.; Campbell, L.; Turner, S. Secondary cell walls:biosynthesis and manipulation. J. Exp. Bot. 2016, 67, 515–531. [Google Scholar] [CrossRef]
- Ralph, J.; Lapierre, C.; Boerjan, W. Lignin structure and itsengineering. Curr. Opin. Biotechnol. 2019, 56, 240–249. [Google Scholar] [CrossRef]
- Gallos, A.; Paes, G.; Allais, F.; Beaugrand, J. Lignocellulosicfibers: A critical review of the extrusion process for enhancementof the properties of natural fiber composites. RSC Adv. 2017, 7, 34638–34654. [Google Scholar] [CrossRef]
- Belmokhtar, N.; Arnoult, S.; Chabbert, B.; Charpentier, J.P.; Brancourt-Hulmel, M. Saccharification performances of Miscanthus atthe pilot and miniaturized assay scales: Genotype and year variabil-ities according to the biomass composition. Front. Plant Sci. 2017, 8, 740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vo, L.T.T.; Girones, J.; Jacquemot, M.-P.; Legée, F.; Cézard, L.; Lapierre, C.; Hage, F.E.; Méchin, V.; Reymond, M.; Navard, P. Correlationsbetween genotype biochemical characteristics and mechanicalproperties of maize stem—Polyethylene composites. Ind. Crops Prod. 2020, 143, 111925. [Google Scholar] [CrossRef]
- Yue-Kuang, S.-O.; Alvarado, C.; Antelme, S.; Bouchet, B.; Cézard, L.; le Bris, P.; Legée, F.; Maia-Grondard, A.; Yoshinaga, A.; Saulnier, L.; et al. Mutation in Brachypodium caffeic acid O-methyltransferase 6 al-ters stem and grain lignins and improves straw saccharificationwithout deteriorating grain quality. J. Exp. Bot. 2016, 67, 227–237. [Google Scholar] [CrossRef] [Green Version]
- Hage, F.; Legland, D.; Borrega, N.; Jacquemot, M.-P.; Griveau, Y.; Coursol, S.; Méchin, V.; Reymond, M. Tissue lignification, cell wall p-coumaroylation and degradability of maize stems de-pend on water status. J. Agric. Food Chem. 2018, 66, 4800–4808. [Google Scholar] [CrossRef]
- University of Illinois at Urbana-Champaign Institute for Sustainability, Energy, and Environment (2021). Energy Sorghum May Combine Best of Annual, Perennial Bioenergy Crops. Available online: https://phys.org/news/2021-01-energy-sorghum-combine-annual-perennial.html (accessed on 30 April 2021).
- Weger, J.; Knápek, J.; Bubeník, J.; Vávrová, K.; Strašil, Z. Can Miscanthus Fulfill Its Expectations as an Energy Biomass Source in the Current Conditions of the Czech Republic?—Potentials and Barriers. Agriculture 2021, 11, 40. [Google Scholar] [CrossRef]
- Lewandowski, I.; Clifton-Brown, J.; Trindade, L.M.; van der Linden, G.C.; Schwarz, K.-U.; Müller-Sämann, K.; Anisimov, A.; Chen, C.-L.; Dolstra, O.; Donnison, I.S.; et al. Progress on Optimizing Miscanthus Biomass Production for the European Bioeconomy: Results of the EU FP7. Proj. Optimisc Front. Plant Sci. 2016, 7, 1620. [Google Scholar] [CrossRef] [Green Version]
- Xue, S.; Kalinina, O.; Lewandowski, I. Present and future options for miscanthus propagation and establishment. Renew. Sustain. Energy Rev. 2015, 49, 1233–1246. Available online: http://authors.elsevier.com/a/1R3~O4s9HvhLFW (accessed on 30 April 2021). [CrossRef]
- Moll, L.; Wever, C.; Völkering, G.; Pude, R. Increase of Miscanthus Cultivation with New Roles in Materials Production—A Review. Agronomy 2020, 10, 308. [Google Scholar] [CrossRef] [Green Version]
- Witzel, C.-P.; Finger, R. Economic evaluation of Miscanthus production—A review. Renew. Sustain. Energy Rev. 2016, 53, 681–696. [Google Scholar] [CrossRef]
- Kiesel, A.; Lewandowski, I. Miscanthus as biogas substrate—Cutting tolerance and potential for anaerobic digestion. GCB Bioenergy 2017, 9, 153–167. [Google Scholar] [CrossRef]
- Von Cossel, M.; Möhring, J.; Kiesel, A.; Lewandowski, I. Optimization of specific methane yield prediction models for biogas crops based on lignocellulosic components using non-linear and crop-specific configurations. Ind. Crops Prod. 2018, 120, 330–342. [Google Scholar] [CrossRef]
- Olave, R.; Forbes, E.; Munoz, F.; Laidlaw, A.; Easson, D.; Watson, S. Performance of Miscanthus × giganteus (Greef et Deu) established with plastic mulch and grown from a range of rhizomes sizes and densities in a cool temperate climate. Field Crops Res. 2017, 210, 81–90. [Google Scholar] [CrossRef]
- Grams, J.; Kwapi’nska, M.; Jedrzejczyk, M.; Rze’znicka, I.; Leahy, J.J.; Ruppert, A.M. Surface characterization of Miscanthus giganteus and Willow subjected to torrefaction. J. Anal. Appl. Pyrolysis 2018, 138, 231–241. [Google Scholar] [CrossRef]
- Xue, S.; Lewandowski, I.; Kalinina, O. Miscanthus establishment and management on permanent grassland in southwest Germany. Ind. Crops Prod. 2017, 108, 572–582. [Google Scholar] [CrossRef]
- Gismatulina, Y.A.; Budaeva, V.V. Chemical composition of five Miscanthus sinensis harvests and nitric-acid cellulose therefrom. Ind. Crops Prod. 2017, 109, 227–232. [Google Scholar] [CrossRef]
- Bergs, M.; Do, X.T.; Rumpf, J.; Kusch, P.; Monakhova, Y.; Konow, C.; Volkering, G.; Pudeef, R.; Schulze, M. Comparing chemical composition and lignin structure of Miscanthus × giganteus and Miscanthus nagara harvested in autumn and spring and separated into stems and leaves. RSC Adv. 2020, 10, 10740. [Google Scholar] [CrossRef] [Green Version]
- El Hage, R.; Perrin, D.; Brosse, N. Effect of the pre-treatment severity on the antioxidant properties of ethanol organosolv Miscanthus × giganteus lignin. Nat. Resour. 2012, 3, 29–34. [Google Scholar] [CrossRef] [Green Version]
- Bauer, S.; Sorek, H.; Mitchell, V.D.; Ibáñez, A.B.; Wemmer, D.E. Characterization of Miscanthus × giganteus lignin isolated by ethanol organosolv process under reflux condition. J. Agric. Food Chem. 2012, 60, 8203–8212. [Google Scholar] [CrossRef] [PubMed]
- Ben Fradj, N.; Rozakis, S.; Borzęcka, M.; Matyka, M. Miscanthus in the European bio-economy: A network analysis. Ind. Crops Prod. 2020, 148, 112281. [Google Scholar] [CrossRef]
- Budenkova, E.; Sukhikh, S.; Ivanova, S.; Babich, O.; Dolganyuk, V.; Michaud, P.; Kriger, O. Improvement of Enzymatic Saccharification of Cellulose-Containing Raw Materials Using Aspergillus niger. Processes 2021, 9, 1360. [Google Scholar] [CrossRef]
- Virlouvet, L.; El Hage, F.; Griveau, Y.; Jacquemot, M.-P.; Gineau, E.; Baldy, A.; Legay, S.; Horlow, C.; Combes, V.; Bauland, C.; et al. Water deficit-responsive QTLs for cell wall degradability and composition inmaize at silage stage. Front. Plant Sci. 2019, 10, 488. [Google Scholar] [CrossRef] [PubMed]
- van der Weijde, T.; Huxley, L.M.; Hawkins, S.; Sembiring, E.H.; Farrar, K.; Dolstra, O.; Visser, R.G.F.; Trindade, L.M. Impact ofdrought stress on growth and quality of miscanthus for biofuelproduction. Glob. Chang. Biol. Bioenergy 2017, 9, 770–782. [Google Scholar] [CrossRef] [Green Version]
- Babich, O.O.; Krieger, O.V.; Chupakhin, E.G.; Kozlova, O.V. Miscanthus plants processing in fuel, energy, chemical and microbiological industries. Foods Raw Mater. 2019, 7, 411. [Google Scholar] [CrossRef]
- Utama, G.L.; Lestari, W.D.; Kayaputri, I.L.; Balia, R.L. Indigenous yeast with cellulose-degrading activity in napa cabbage (Brassica pekinensis L.) waste: Sharacterisation and species identification. Foods Raw Mater. 2019, 7, 321–328. [Google Scholar] [CrossRef]
- Eiland, F.; Leth, M.; Klamer, M.; Lind, A.-M.; Jensen, H.; Iversen, J. C and N Turnover and Lignocellulose Degradation During Composting of Miscanthus Straw and Liquid Pig Manure. Compost. Sci. Util. 2001, 9, 186–196. [Google Scholar] [CrossRef]
- Knapczyk, A.; Francik, S.; Wójcik, A.; Bednarz, G. Influence of Storing Miscanthus × gigantheus on Its Mechanical and Energetic Properties. In Renewable Energy Sources: Engineering, Technology, Innovation; Springer: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Kriger, O.; Budenkova, E.; Babich, O.; Suhih, S.; Patyukov, N.; Masyutin, Y.; Chupakhin, E.; Dolganuk, V. The process of producing bioethanol from delignified cellulose isolated from plants of the miscanthus genus. Bioengineering 2020, 7, 61. [Google Scholar] [CrossRef]
- Kondratenko, V.V.; Kondratenko, T.Y.; Petrov, A.N.; Belozerov, G.A. Assessing protopectin transformation potential of plant tissue using a zoned criterion space. Foods Raw Mater. 2020, 8, 348–361. [Google Scholar] [CrossRef]
- Krieger, O.V.; Budenkova, E.A.; Babich, O.O.; Sukhikh, S.A.; Voblikova, T.V. Features of the processing of cellulose-containing raw materials with Aspergillus niger enzyme systems to obtain bioethanol. IOP Conf. Ser. Earth Environ. 2021, 852, 012056. [Google Scholar] [CrossRef]
- Krieger, O.V.; Dolganyuk, V.F.; Prosekov, A.Y.; Izgaryshev, A.V.; Sukhikh, S.A. Features of processing the biomass of genus miscanthus plants into carbohydrate-containing substrates for biotechnology. IOP Conf. Ser. Earth Environ. Sci. 2021, 699, 012056. [Google Scholar] [CrossRef]
- Dolganuk, V.F.; Kriger, O.V.; Babich, O.O.; Sukhikh, S.A.; Chupakhin, Y.G. Selection of Parameters for Miscanthus Enzymolysis Using Cellulad Ultra in the Collection: Modern Approaches to the Development of Agro-Industrial, Chemical and Forestry Complexes. Problems, Trends, Prospects. Collection of Materials of the All-Russian Scientific-Practical Conference; Yaroslav-the-Wise Novgorod State University Velikiy: Novgorod, Russia, 2021; pp. 182–188. (In Russian) [Google Scholar]
- Lifton-Brown, J.; Lewandowski, I. Overwintering problems of newly established Miscanthus plantations can be overcome by identifying genotypes with improved rhizome cold tolerance. New Phytol. 2000, 148, 287–294. [Google Scholar] [CrossRef]
- Winkler, B.; Mangold, A.; von Cossel, M.; Clifton-Brown, J.; Pogrzeba, M.; Lewandowski, I.; Iqbal, Y.; Kiesel, A. Implementing miscanthus into farming systems: A review of agronomic practices, capital and labour demand. Renew. Sust. Energy Rev. 2020, 132, 110053. [Google Scholar] [CrossRef]
- Moore, K.J.; Kling, C.L.; Raman, D.R. A Midwest USA Perspective on Von Cossel et al.’s Prospects of Bioenergy Cropping Systems for a More Social-Ecologically Sound Bioeconomy. Agronomy 2020, 10, 1658. [Google Scholar] [CrossRef]
- Dorogina, O.V.; Vasilieva, O.Y.; Nuzhdina, N.S.; Buglova, L.V.; Gismatulina, Y.A.; Zhmud, E.V.; Zueva, G.A.; Komina, O.V.; Tsybchenko, E.A. Resource potential of some species of the genus Miscanthus Anderss. under conditions of the continental climate of Western Siberian forest-steppe. Vavilov J. Genet. Breed. 2018, 22, 553–559. [Google Scholar] [CrossRef]
- Ashman, C.; Awty-Carroll, D.; Mos, M.; Robson, P.; Clifton-Brown, J. Assessing seed priming, sowing date, and mulch film to improve the germination and survival of direct-sown Miscanthus sinensis in the United Kingdom. Glob. Chang. Biol. Bioenergy 2018, 10, 612–627. [Google Scholar] [CrossRef]
- Clifton-Brown, J.; Schwarz, K.-U.; Hastings, A. History of the development of Miscanthus as a bioenergy crop: From small be-ginnings to potential realisation. Biol. Environ. 2015, 115, 1–13. [Google Scholar]
- Strullu, L.; Ferchaud, F.; Yates, N.; Shield, I.; Beaudoin, N.; Garcia de Cortazar-Atauri, I.; Besnard, A.; Mary, B. Multisite Yield Gap Analysis of Miscanthus × giganteus Using the STICS Model. BioEnergy Res. 2015, 8, 1735–1745. [Google Scholar] [CrossRef]
- Purdy, S.J.; Maddison, A.L.; Nunn, C.P.; Winters, A.; Timms-Taravella, E.; Jones, C.M.; Clifton-Brown, J.C.; Donnison, I.S.; Gallagher, J.A. Could Miscanthus replace maize as the preferred substrate for anaerobic digestion in the United Kingdom? Future breeding strategies. Glob. Chang. Biol. Bioenergy 2017, 9, 1122–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knápek, J.; Vávrová, K.; Valentová, M.; Vašíˇcek, J.; Králík, T. Energy biomass competitiveness-three different views on biomass price. Wiley Interdiscip. Rev. Energy Environ. 2017, 6, e261. [Google Scholar] [CrossRef]
- Stolarski, M.J.; Snieg, M.; Krzyzaniak, M.; Tworkowski, J.; Szczukowski, S.; Graban, Ł.; Lajszner, W. Short rotation coppices, grasses and other herbaceous crops: Biomass properties versus 26 genotypes and harvest time. Ind. Crops Prod. 2018, 119, 22–32. [Google Scholar] [CrossRef]
- Robson, P.R.; Donnison, I.S.; Clifton-Brown, J.C. Stem growth characteristics of high yielding Miscanthus correlate with yield, development and intraspecific competition within plots. GCB Bioenergy 2019, 11, 1075–1085. [Google Scholar] [CrossRef]
- Mantineo, M.; D’agosta, G.M.; Copani, V.; Patanè, C.; Cosentino, S.L. Biomass yield and energy balance of three perennial crops for energy use in the semi-arid Mediterranean environment. Field Crops Res. 2009, 114, 204–213. [Google Scholar] [CrossRef]
- Scebba, F.; Arduini, I.; Ercoli, L.; Sebastiani, L. Cadmium effects on growth and antioxidant enzymes activities in Miscanthus sinensis. Biol. Plant. 2006, 50, 688–692. [Google Scholar] [CrossRef]
- El Achaby, M.; El Miri, N.; Hannache, H.; Gmouh, S.; Trabadelo, V.; Aboulkas, A.; Ben Youcef, H. Cellulose nanocrystals from Miscanthus fibers: Insights into rheological, physico-chemical properties and polymer reinforcing ability. Cellulose 2018, 25, 6603–6619. [Google Scholar] [CrossRef]
- Wever, C.; Van Tassel, D.L.; Pude, R. Third-Generation Biomass Crops in the New Era of De Novo Domestication. Agronomy 2020, 10, 1322. [Google Scholar] [CrossRef]
- Babich, O.; Dyshlyuk, L.; Noskova, S.; Sukhikh, S.; Prosekov, A.; Ivanova, S.; Pavsky, V. In vivo study of the potential of the carbohydrate-mineral complex from pine nut shells as an ingredient of functional food products. Bioact. Carbohydr. Diet. Fibre 2019, 18, 100185. [Google Scholar] [CrossRef]
- Rusinowski, S.; Krzyzak, J.; Clifton-Brown, J.; Jensen, E.; Mos, M.; Webster, R.; Sitko, K.; Pogrzeba, M. New Miscanthus hybrids cultivated at a Polish metal(loid)-contaminated site demonstrate superior stomatal regulation and reduced shoot Pb and Cd concentrations. Environ. Pollut. 2019, 252, 1377–1387. [Google Scholar] [CrossRef]
- Davey, C.L.; Robson, P.; Hawkins, S.; Farrar, K.; Clifton-Brown, J.C.; Donnison, I.S.; Slavov, G.T. Genetic relationships between spring emergence, canopy phenology, and biomass yield increase the accuracy of genomic prediction in Miscanthus. J. Exp. Bot. 2017, 68, 5093–5102. [Google Scholar] [CrossRef] [Green Version]
- Kožnarová, V.; Klabzuba, J. Recommendation of World Meteorological Organization to describing meteorological or climatological conditions—Information. Plant Soil Environ. 2011, 48, 190–192. [Google Scholar] [CrossRef] [Green Version]
- Szulczewski, W.; Zyromski, A.; Jakubowski, W.; Biniak-Pieróg, M. A new method for the estimation of biomass yield of giant Miscanthus (Miscanthus giganteus) in the course of vegetation. Renew. Sustain. Energy Rev. 2018, 82, 1787–1795. [Google Scholar] [CrossRef]
- Zub, H.W.; Brancourt-Hulmel, M. Agronomic and physiological performances of different species of Miscanthus, a major energy crop. A review. Agron. Sustain. Dev. 2010, 30, 201–214. [Google Scholar] [CrossRef] [Green Version]
- Kriger, O.V.; Babich, O.O.; Dolganyuk, V.F.; Kozlova, O.V.; Sukhikh, S.A.; Larichev, T.A. Bioethanol production from Miscanthus sinensis cellulose by bioconversion. Food Process. Tech. Technol. 2021, 51, 387–394. [Google Scholar] [CrossRef]
- Xue, S.; Lewandowski, I.; Wang, X.; Yi, Z. Assessment of the production potentials of Miscanthus on marginal land in China. Renew. Sustain. Energy Rev. 2016, 54, 932–943. [Google Scholar] [CrossRef]
- Vávrová, K.; Knápek, J.; Weger, J.; Králík, T.; Beranovský, J. Model for evaluation of locally available biomass competitiveness for decentralized space heating in villages and small towns. Renew. Energy 2018, 129, 853–865. [Google Scholar] [CrossRef]
- Danielewicz, D.; Dybka-Stępień, K.; Surma-Ślusarska, B. Processing of Miscanthus × giganteus stalks into various soda and kraft pulps. Part I: Chemical composition, types of cells and pulping effects. Cellulose 2018, 25, 6731–6744. [Google Scholar] [CrossRef] [Green Version]
- Zhong, R.; Ye, Z.H. Secondary cell walls: Biosynthesis, patterned deposition and transcriptional regulation. Plant Cell Physiol. 2015, 56, 195–214. [Google Scholar] [CrossRef] [Green Version]
- Hodgson, E.; Lister, S.; Bridgwater, T.; Clifton-Brown, J.; Donnison, I. Genotypic and environmentally derived variation in the cell wall composition of Miscanthus in relation to its use as a biomass feedstock. Biomass Bioenergy 2010, 34, 652–660. [Google Scholar] [CrossRef]
- Hastings, A.; Mos, M.; Yesufu, J.A.; McCalmont, J.; Schwarz, K.; Shafei, R.; Ashman, C.; Nunn, C.; Schuele, H.; Cosentino, S.; et al. Economic and Environmental Assessment of Seed and Rhizome Propagated Miscanthus in the UK. Front. Plant Sci. 2017, 8, 1058. [Google Scholar] [CrossRef] [Green Version]
- Christian, E.J. Seed Development and Germination of Miscanthus sinensis. Ph.D. Thesis, The University of Iowa, Iowa City, IA, USA, 2012; p. 12880. Available online: https://lib.dr.iastate.edu/etd/12880 (accessed on 18 March 2021).
- Bonin, C.L.; Mutegi, E.; Chang, H.; Heaton, E.A. Improved feedstock option or invasive risk? Comparing establishment and productivity of fertile Miscanthus giganteus to Miscanthus sinensis. Bioenergy Res. 2017, 10, 317–328. [Google Scholar] [CrossRef]
- Kalinina, O.; Nunn, C.; Sanderson, R.; Hastings, A.F.S.; van der Weijde, T.; Özgüven, M.; Tarakanov, I.; Schüle, H.; Trindade, L.M.; Dolstra, O.; et al. Extending Miscanthus Cultivation with Novel Germplasm at Six Contrasting Sites. Front. Plant Sci. 2017, 8, 563. [Google Scholar] [CrossRef] [Green Version]
- Tubeilen, F.; Rennie, T.J.; Goss, M.J. A review on biomass production from C4 grasses: Yield and quality for end-use. Curr. Opin. Plant Biol. 2016, 31, 172–180. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Gilley, J.; Eisenhauer, D.; Boldt, A. Soil carbon accumulation under switchgrass barriers. Agron. J. 2014, 106, 2185–2192. [Google Scholar] [CrossRef] [Green Version]
- Robertson, A.D.; Davies, C.A.; Smith, P.; Stott, A.W.; Clark, E.L.; McNamara, N.P. Carbon inputs from Miscanthus displace older soil organic carbon without inducing priming. Bioenergy Res. 2017, 10, 86–102. [Google Scholar] [CrossRef]
- Jones, M.B.; Finnan, J.; Hodkinson, T.R. Morphological and physiological traits for higher biomass production in perennial rhizomatous grasses grown on marginal land. Glob. Chang. Biol. 2015, 7, 375–385. [Google Scholar] [CrossRef]
- Nijsen, M.; Smeets, E.; Stehfest, E. An evaluation of the global potential of bioenergy production on degraded lands. Glob. Chang. Biol. 2012, 4, 130–147. [Google Scholar] [CrossRef]
- Kapustyanchik, S.Y.; Burmakina, N.V.; Yakimenko, V.N. Evaluation of the ecological and agrochemical state of agrocenosis with long-term growing of Miscantus in Western Siberia. Agrohimia 2020, 9, 65–73. (In Russia) [Google Scholar] [CrossRef]
- Lewandowski, I.; Kicherer, A. Combustion quality of biomass: Practical relevance and experiments to modify the biomass quality of Miscanthus × giganteus. Eur. J. Agron. 1997, 6, 163–177. [Google Scholar] [CrossRef]
- Heaton, E.A.; Flavell, R.B.; Mascia, P.N.; Thomas, S.R.; Dohleman, F.G.; Long, S.P. Herbaceous energy crop development: Recent progress and future prospects. Curr. Opin. Plant Biol. 2008, 19, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Cerazy-Waliszewska, J.; Jeżowski, S.; Łysakowski, P.; Waliszewska, B.; Zborowska, M.; Sobańska, K.; Ślusarkiewicz-Jarzina, A.; Białas, W.; Pniewski, T. Potential of bioethanol production from biomass of various Miscanthus genotypes cultivated in three-year plantations in west-central Poland. Ind. Crops Prod. 2019, 141, 111790. [Google Scholar] [CrossRef]
- Głowacka, K.; Ahmed, A.; Sharma, S.; Abbott, T.; Comstock, J.C.; Long, S.P.; Sacks, E.J. Can chilling tolerance of C4 photosynthesis in Miscanthus be transferred to sugarcane? GCB-Bioenergy 2016, 8, 407–418. [Google Scholar] [CrossRef]
- Zhang, Y.; Zahid, I.; Danial, A.; Minaret, J.; Cao, Y.; Dutta, A. Hydrothermal carbonization of Miscanthus: Processing, properties, and synergistic Co-combustion with lignite. Energy 2021, 225, 120200. [Google Scholar] [CrossRef]
- Lewandowski, A.; Lewandowska, W.; Sielski, J.; Dziku’c, M.; Wróbel, M.; Jewiarz, M.; Knapczyk, A. Sustainable Drying and Torrefaction Processes of Miscanthus for Use as a Pelletized Solid Biofuel and Biocarbon-Carrier for Fertilizers. Molecules 2021, 26, 1014. [Google Scholar] [CrossRef]
- Patel, M.K.; Bechu, A.; Villegas, J.D.; Bergez-Lacoste, M.; Yeung, K.; Murphy, R.; Woods, J.; Mwabonje, O.N.; Ni, Y.; Patel, A.D.; et al. Second-generation bio-based plastics are becoming a reality—Non-renewable energy and greenhouse gas (GHG) balance of succinic acid-based plastic end products made from lignocellulosic biomass. Biofuels Bioprod. Biorefin. 2018, 12, 426–441. [Google Scholar] [CrossRef]
- Harris, D.; De Bolt, S. Synthesis, regulation and utilization of lignocellulosic biomass. Plant Biotechnol. J. 2010, 8, 244–262. [Google Scholar] [CrossRef]
- Danielewicz, D.; Surma-Ślusarska, B.; Żurek, G.; Martyniak, D.; Kmiotek, M.; Dybka, K. Selected grass plants as biomass fuels and raw materials for papermaking, Part II: Pulp and paper properties. BioResources 2015, 10, 8552–8564. [Google Scholar] [CrossRef] [Green Version]
- Eschenhagen, A.; Raj, M.; Rodrigo, N.; Zamora, A.; Labonne, L.; Evon, P.; Welemane, H. Investigation of Miscanthus and Sunflower Stalk Fiber-Reinforced Composites for Insulation Applications. Adv. Civ. Eng. 2019, 2019, 1–7. [Google Scholar] [CrossRef]
- Wierzbicki, M.P.; Maloney, V.; Mizrachi, E.; Myburg, A.A. Xylan in the Middle: Understanding Xylan Biosynthesis and Its Metabolic Dependencies Toward Improving Wood Fiber for Industrial. Process. Front. Plant Sci. 2019, 10, 176. Available online: https://www.frontiersin.org/article/10.3389/fpls.2019.00176 (accessed on 22 March 2021). [CrossRef] [Green Version]
- Moll, L. Miscanthus für die nachhaltige baustoffliche Nutzung am Beispiel bindemittelfreier Faserplatten. Mitt. Ges. Pflanzenbauwiss. 2018, 30, 189–190. [Google Scholar]
- Klímek, P.; Wimmer, R.; Meinlschmidt, P.; Kúdela, J. Utilizing Miscanthus stalks as raw material for particleboards. Ind. Crops Prod. 2018, 111, 270–276. [Google Scholar] [CrossRef]
- Mangold, A.; Lewandowski, I.; Möhring, J.; Clifton-Brown, J.; Krzyzak, J.; Mos, M.; Pogrzeba, M.; Kiesel, A. Harvest date and leaf: Stem ratio determine methane hectare yield of miscanthus biomass. GCB Bioenergy 2019, 11, 21–33. [Google Scholar] [CrossRef]
- Gaj, T.; Gersbach, C.A.; Barbas, C.F. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 2013, 31, 397–405. [Google Scholar] [CrossRef] [Green Version]
- Bilandzija, N.; Jurisic, V.; Voca, N.; Leto, J.; Matin, A.; Sito, S.; Kricka, T. Combustion properties of Miscanthus × giganteus biomass—Optimization of harvest time. J. Energy Inst. 2017, 90, 528–533. [Google Scholar] [CrossRef]
- Evans, J.R. Improving photosynthesis. Plant Physiol. 2013, 162, 1780–1793. [Google Scholar] [CrossRef] [Green Version]
- Peças, P.; Carvalho, H.; Salman, H.; Leite, M. Natural fiber Composites and Their Applications: A Review. J. Compos. Sci. 2018, 2, 66. [Google Scholar] [CrossRef] [Green Version]
- Kapustyanchik, S.Y.; Danilova, A.A.; Likhenko, I.E. Miscanthus sacchariflorus in Siberia—Biological yield parameters and dynamics of biofilic elements. Agric. Biol. 2021, 56, 121–134. [Google Scholar] [CrossRef]
- Masters, M.D.; Black, C.K.; Kantola, I.B.; Woli, K.P.; Voigt, T.; David, M.V.; DeLucia, E.N. Soil nutrient removal by four potential bioenergy crops: Zea mays, Panicum virgatum, Miscanthus × giganteus, and prairie. Agric. Ecosyst. Environ. 2016, 216, 51–60. [Google Scholar] [CrossRef] [Green Version]
- Maughan, M. Miscanthus × giganteus productivity: The effects of management in different environments. Glob. Chang. Biol. 2012, 4, 253–265. [Google Scholar] [CrossRef]
- Oliveira, J.A.; West, C.P.; Afif, E.; Palencia, P. Comparison of Miscanthus and Switchgrass cultivars for biomass yield, soil nutrients, and nutrient removal in Northwest Spain. Agron. J. 2017, 109, 122–130. [Google Scholar] [CrossRef] [Green Version]
- Dufosse, K.; Drewer, J.; Gabrielle, B.; Drouet, J.-L. Effects of a 20-year old Miscanthus × giganteus stand and its removal on soil characteristics and greenhouse gas emissions. Biomass Bioenergy 2014, 69, 198–210. [Google Scholar] [CrossRef]
- Nunn, C.; Hastings, A.F.S.J.; Kalinina, O.; Özgüven, M.; Schüle, H.; Tarakanov, I.G.; Van Der Weijde, T.; Anisimov, A.A.; Iqbal, Y.; Kiesel, A.; et al. Environmental influences on the growing season duration and ripening of diverse Miscanthus germplasm grown in six countries. Front. Plant Sci. 2017, 8, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Muthuraj, R.; Misra, M.; Mohanty, A.K. Biocomposite consisting of miscanthus fiber and biodegradable binary blend matrix: Compatibilization and performance evaluation. RSC Adv. 2017, 7, 27538–27548. [Google Scholar] [CrossRef] [Green Version]
- Klikocka, H.; Kasztelan, A.; Zakrzewska, A.; Wyłupek, T.; Szostak, B.; Skwaryło-Bednarz, B. The Energy Efficiency of the Production and Conversion of Spring Triticale Grain into Bioethanol. Agronomy 2019, 9, 423. [Google Scholar] [CrossRef] [Green Version]
- Sokólski, M.; Jankowski, K.J.; Załuski, D.; Szatkowski, A. Productivity, Energy and Economic Balance in the Production of Different Cultivars of Winter Oilseed Rape. A Case Study in North-Eastern Poland. Agronomy 2020, 10, 508. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, Y.; Sugano, S.S.; Shimada, T.; Hara-Nishimura, I. Enhancement of leaf photosynthetic capacity through increased stomatal density in Arabidopsis. New Phytol. 2013, 198, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Robles-Aguilar, A.A.; Temperton, V.M.; Jablonowski, N.D. Maize silage digestate application affecting germination and early growth of maize modulated by soil type. Agronomy 2019, 9, 473. [Google Scholar] [CrossRef] [Green Version]
- Dietrich, C.C.; Rahaman, M.A.; Robles-Aguilar, A.A.; Latif, S.; Intani, K.; Müller, J.; Jablonowski, N.D. Nutrient Loaded Biochar Doubled Biomass Production in Juvenile Maize Plants (Zea mays L.). Agronomy 2020, 10, 567. [Google Scholar] [CrossRef] [Green Version]
- Cumplido-Marin, L.; Graves, A.R.; Burgess, P.J.; Morhart, C.; Paris, P.; Jablonowski, N.D.; Facciotto, G.; Bury, M.; Martens, R.; Nahm, M. Two Novel Energy Crops: Sida hermaphrodita (L.) Rusby and Silphium perfoliatum L.—State of Knowledge. Agronomy 2020, 10, 928. [Google Scholar] [CrossRef]
- Kapustyanchik, S.Y.; Yakimenko, V.N.; Gismatulina, Y.A.; Budaeva, V.V. Miscanthus—A Promising Energy Crop for Industrial Processing. Ecol. Ind. Russ. 2021, 25, 66–71. [Google Scholar] [CrossRef]
- Liu, L.; Li, H.; Lazzaretto, A.; Manente, G.; Tong, C.; Liu, Q.; Li, N. The development history and prospects of biomass-based insulation materials for buildings. Renew. Sustain. Energy Rev. 2017, 69, 912–932. [Google Scholar] [CrossRef]
- Feledyn-Szewczyk, B.; Matyka, M.; Staniak, M. Comparison of the Effect of Perennial Energy Crops and Agricultural Crops on Weed Flora Diversity. Agronomy 2019, 9, 695. [Google Scholar] [CrossRef] [Green Version]
- Eichler-Löbermann, B.; Busch, S.; Jablonowski, N.D.; Kavka, M.; Brandt, C. Mixed cropping as affected by phosphorus and water supply. Agronomy 2020, 10, 1506. [Google Scholar] [CrossRef]
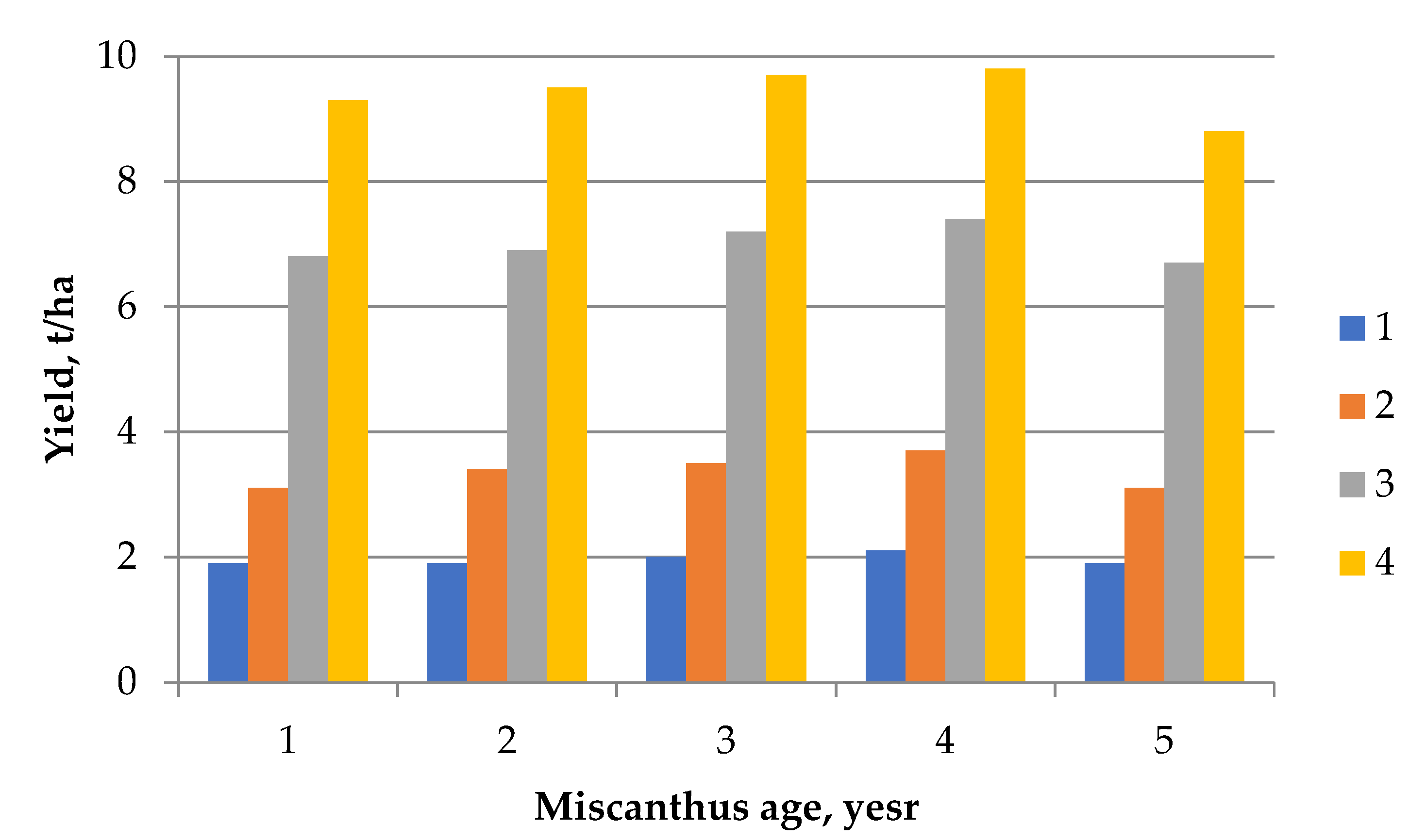
| Harvest Period | Dry Biomass, % | Cellulose, % | Lignin, % | Ash, % |
|---|---|---|---|---|
| autumn | 29.4 | 40.6 | 8.0 | 3.9 |
| winter | 28.8 | 46.4 | 9.4 | 2.5 |
| Miscanthus Species | Number of Stems, pcs. | Stem Diameter, cm | Stem Height, cm | Biomass Yield,% |
|---|---|---|---|---|
| M. × giganteus | 49 | 5.6 | 231 | 324 |
| M. sinensis | 58 | 4.4 | 132 | 276 |
| M. sacchariflorus | 26 | 7.2 | 185 | 308 |
| Pathways of the Bioethanol Synthesis | Enzymes * |
|---|---|
| monoxylphenol pathway in cytosol (via phenylpropane pathway) | PAL, C4H, 4CL, HCT, C3’H, CCoAOM, F5H, COMT, CAD |
| polymerization of monomers in the cell wall | PAL, C4H, 4CL, HCT, CCoAOM, CCR, CAD |
| Region | Climate | Species | Yield, t/ha | Source | |
|---|---|---|---|---|---|
| 1 | 2 | ||||
| Russia (Asian part) | 1.7 | 464 | M. × giganteus | 16.6 | [97] |
| M. sacchariflorus | 12.0 | [97] | |||
| USA (New Jersy) | 11.2 | 1211 | M. × giganteus | 9.5 | [98] |
| USA (Illinois) | 11.1 | 1043 | M. × giganteus | 15.6 | [98] |
| USA (Nebraska) | 9.7 | 704 | M. × giganteus | 27.7 | [98] |
| Spain | 13.9 | 100.3 | M. × giganteus | 17.6 | [99] |
| France | 11.5 | 557 | M. × giganteus | 16.9 | [100] |
| France | 17.8 | 390 | M. × giganteus | 22.0 | [101] |
| Russia (European part) | 4.5 | 620 | M. × giganteus | 7.8 | [102] |
| M. × giganteus | 5.7 | [102] | |||
| M. sacchariflorus | 4.2 | [102] | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chupakhin, E.; Babich, O.; Sukhikh, S.; Ivanova, S.; Budenkova, E.; Kalashnikova, O.; Kriger, O. Methods of Increasing Miscanthus Biomass Yield for Biofuel Production. Energies 2021, 14, 8368. https://doi.org/10.3390/en14248368
Chupakhin E, Babich O, Sukhikh S, Ivanova S, Budenkova E, Kalashnikova O, Kriger O. Methods of Increasing Miscanthus Biomass Yield for Biofuel Production. Energies. 2021; 14(24):8368. https://doi.org/10.3390/en14248368
Chicago/Turabian StyleChupakhin, Evgeny, Olga Babich, Stanislav Sukhikh, Svetlana Ivanova, Ekaterina Budenkova, Olga Kalashnikova, and Olga Kriger. 2021. "Methods of Increasing Miscanthus Biomass Yield for Biofuel Production" Energies 14, no. 24: 8368. https://doi.org/10.3390/en14248368
APA StyleChupakhin, E., Babich, O., Sukhikh, S., Ivanova, S., Budenkova, E., Kalashnikova, O., & Kriger, O. (2021). Methods of Increasing Miscanthus Biomass Yield for Biofuel Production. Energies, 14(24), 8368. https://doi.org/10.3390/en14248368









